A Review of Epithelial Ion Transporters and Their Roles in Equine Infectious Colitis
Abstract
Simple Summary
Abstract
1. Introduction
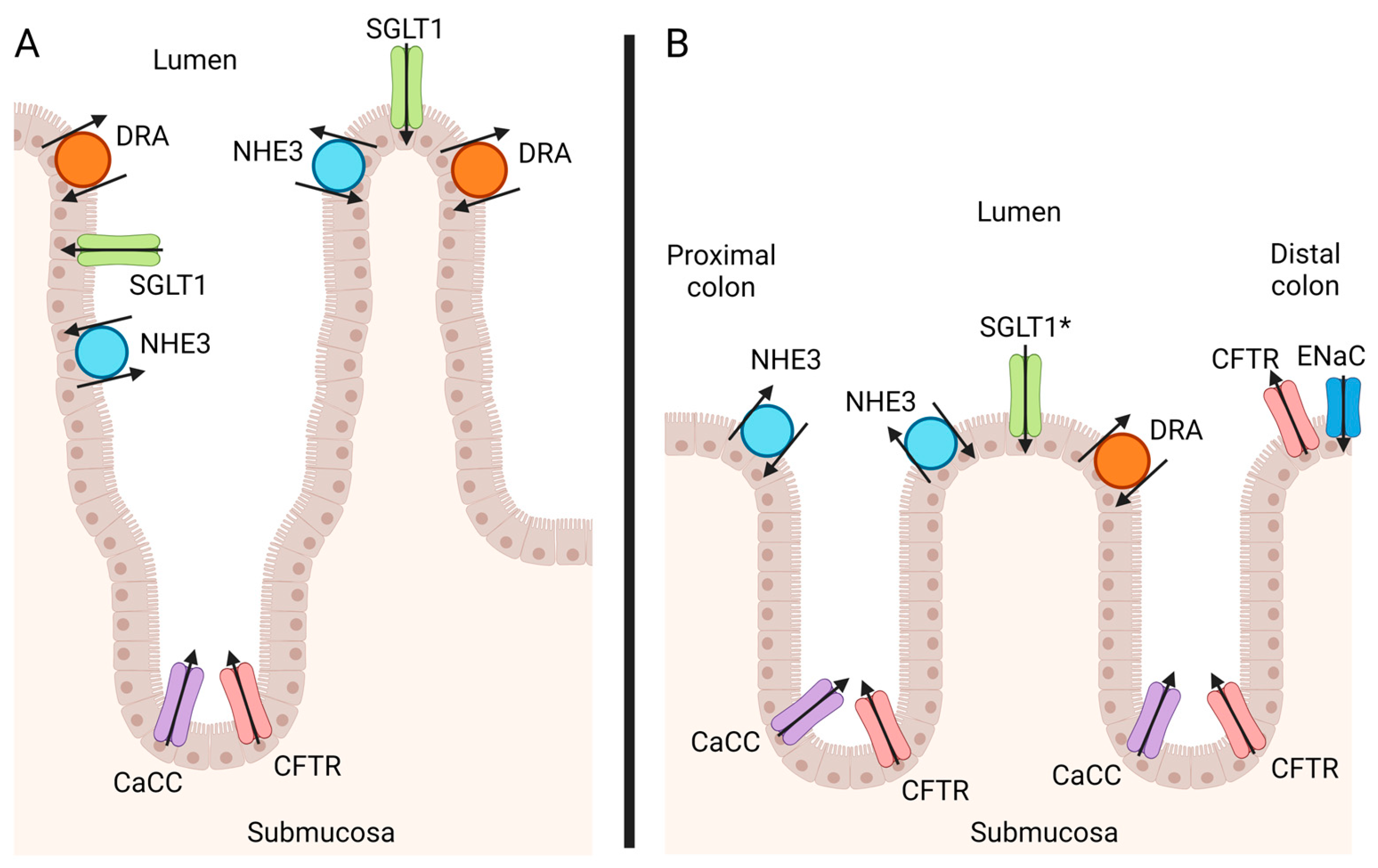
2. Anatomy of the Intestinal Epithelium
3. Epithelial Ion Channels in the Equine Colon
3.1. Chloride
3.2. Sodium
3.3. Potassium
3.4. Bicarbonate
4. The Contributions of Secretory versus Malabsorptive Mechanisms in the Horse
| Pathogen | Serovar | Toxins Produced | Ion Transporter Affected | Absorption or Secretion | Animal/Cell Model | Reference |
|---|---|---|---|---|---|---|
| Clostridioides difficle | TcdA and TcdB | Decrease CFTR function | Decrease secretion–chloride | In vivo and ex vivo murine colon | [57] | |
| Decrease CaCC function | Decrease secretion–chloride | In vivo and ex vivo murine colon | [57] | |||
| Decrease DRA expression | Decrease absorption–chloride | In vivo and ex vivo murine colon | [57] | |||
| Decrease SGLT1 expression | Decrease absorption–sodium, glucose | In vivo and ex vivo murine colon | [57] | |||
| TcdB only | Decrease NHE3 expression | Decrease absorption–sodium | In vivo and ex vivo murine colon | [57] | ||
| Clostridium perfringens | CPE | N/A—Tight junction disruption, pore formation | Passive loss into lumen | Rabbit ileum, Vero kidney cell line (African green monkey) | [58,59] | |
| CPB2 | N/A—Pore formation | Passive loss into lumen | Murine (intravenous administration), I407 cell line (human), rabbit small intestine and colon, ovarian tumor cell line (equine) | [60,61,62] | ||
| CPB | N/A—Endothelial damage | N/A | Neonatal porcine small intestine | [63] | ||
| Salmonella spp. | Typhimurium | Decrease ENaC expression and activity | Decrease absorption–sodium | Ex vivo murine colon | [64] | |
| Typhimurium | Decrease CFTR function | Decrease secretion–chloride | Ex vivo murine colon | [64] | ||
| Typhi | Increase CFTR function | Increase secretion–chloride | Murine small intestine, T-84 cell line (human) | [65] | ||
| Dublin | Increase CFTR function | Increase secretion–chloride | HT-29, Caco-2, T-84 cell lines (human) | [66] | ||
| Typhimurium | Decrease DRA expression | Decrease absorption–chloride | Murine colonic organoids | [67] | ||
| Neorickettsiosis | Decrease NHE3 activity (suspected to also occur in the horse) | Decrease absorption–sodium | Equine colon, T-84 cell line (human) | [54,68] | ||
| Equine Coronavirus | N/A—Villus blunting | Decrease absorption–sodium, glucose | Equine small intestine | [53] | ||
| Increase CaCC activity (demonstrated in SARS-CoV2) | Increase secretion–chloride | Ex vivo human colonic mucosa (SARS-CoV2) | [69] | |||
| Decrease ENaC activity (demonstrated in SARS-CoV) | Decrease absorption–sodium | Expression of human ENaC in Xenopus oocytes (SARS-CoV) | [70] | |||
| Cyathostomins | N/A—Mucosal surface disruption | Passive loss into lumen | Equine colon | [71] | ||
| N/A—Increase mucin production | N/A | Equine enteric monolayers | [72] | |||
| Increase CFTR or CaCC function (demonstrated in other intestinal nematodes) | Increase secretion–chloride | Ex vivo rat colon | [73] |
5. Clinical Overview of Colitis Pathogens in the Horse
5.1. Clostridioides difficile
5.2. Clostridium perfringens
5.3. Salmonella spp.
5.4. Antimicrobial-Associated Diarrhea and the Microbiome
5.5. Neorickettsiosis (Potomac Horse Fever)
5.6. Equine Coronavirus
5.7. Larval Cyathostominosis
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Argenzio, R.A. Functions of the Equine Large Intestine and Their Interrelationship in Disease. Cornell Vet. 1975, 65, 303–330. [Google Scholar] [PubMed]
- Krunkosky, T.M.; Jarrett, C.L.; Moore, J.N. Gross and Microscopic Anatomy of the Equine Gastrointestinal Tract. In The Equine Acute Abdomen; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; pp. 1–18. ISBN 978-1-119-06325-4. [Google Scholar]
- Hines, M. Diarrhea. In Equine Internal Medicine; Reed, S.M., Bayly, W.M., Sellon, D.C., Eds.; Saunders: Philadelphia, PA, USA, 2004; pp. 156–163. [Google Scholar]
- Blikslager, A.; Gonzalez, L. Equine Intestinal Mucosal Pathobiology. Annu. Rev. Anim. Biosci. 2018, 6, 157–175. [Google Scholar] [CrossRef] [PubMed]
- House, J.K.; Smith, B.P. Salmonella in Horses. In Salmonella in Domestic Animals; Wray, C., Wray, A., Eds.; CAB International: Wallingford Oxon, UK, 2000; pp. 219–230. ISBN 9780851992617. [Google Scholar]
- Chidlow, H. Dealing with Acute Diarrhoea in Adult Horses. Practice 2020, 42, 510–519. [Google Scholar] [CrossRef]
- Atherton, R.P.; Furr, M.O.; McKenzie, H.C.; Desrochers, A.M. Efficacy of Hyperimmunized Plasma in the Treatment of Horses with Acute Colitis. J. Equine Vet. Sci. 2011, 31, 19–25. [Google Scholar] [CrossRef]
- Gomez, D.E.; Leclere, M.; Arroyo, L.G.; Li, L.; John, E.; Afonso, T.; Payette, F.; Darby, S. Acute Diarrhea in Horses: A Multicenter Canadian Retrospective Study (2015 to 2019). Can. Vet. J. 2022, 63, 1033–1042. [Google Scholar] [PubMed]
- Ayoub, C.; Arroyo, L.G.; MacNicol, J.L.; Renaud, D.; Weese, J.S.; Gomez, D.E. Fecal Microbiota of Horses with Colitis and Its Association with Laminitis and Survival during Hospitalization. J. Vet. Intern. Med. 2022, 36, 2213–2223. [Google Scholar] [CrossRef]
- Barr, B.S.; Waldridge, B.M.; Morresey, P.R.; Reed, S.M.; Clark, C.; Belgrave, R.; Donecker, J.M.; Weigel, D.J. Antimicrobial-Associated Diarrhoea in Three Equine Referral Practices. Equine Vet. J. 2013, 45, 154–158. [Google Scholar] [CrossRef]
- Liepman, R.S.; Swink, J.M.; Habing, G.G.; Boyaka, P.N.; Caddey, B.; Costa, M.; Gomez, D.E.; Toribio, R.E. Effects of Intravenous Antimicrobial Drugs on the Equine Fecal Microbiome. Animals 2022, 12, 1013. [Google Scholar] [CrossRef]
- Nord, C.E.; Edlund, C. Impact of Antimicrobial Agents on Human Intestinal Microflora. J. Chemother. 1990, 2, 218–237. [Google Scholar] [CrossRef]
- Shaw, S.D.; Stämpfli, H. Diagnosis and Treatment of Undifferentiated and Infectious Acute Diarrhea in the Adult Horse. Vet. Clin. N. Am. Equine Pract. 2018, 34, 39–53. [Google Scholar] [CrossRef]
- Gomez, D.E.; Arroyo, L.G.; Schoster, A.; Renaud, D.L.; Kopper, J.J.; Dunkel, B.; Byrne, D.; Group, T.M.; Toribio, R.E. Diagnostic Approaches, Aetiological Agents and Their Associations with Short-Term Survival and Laminitis in Horses with Acute Diarrhoea Admitted to Referral Institutions. Equine Vet. J. 2023, 56, 959–969. [Google Scholar] [CrossRef] [PubMed]
- International Equine Colitis Research Group. Science-in-Brief: Report on the Havemeyer Foundation Workshop on Acute Colitis of the Adult Horse. Equine Vet. J. 2020, 52, 163–164. [Google Scholar] [CrossRef] [PubMed]
- Barrett, K.E.; Keely, S.J. Chloride Secretion by the Intestinal Epithelium: Molecular Basis and Regulatory Aspects. Annu. Rev. Physiol. 2000, 62, 535–572. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.A. The Emerging Roles of Deep Crypt Secretory Cells in Colonic Physiology. Am. J. Physiol.-Gastrointest. Liver Physiol. 2023, 325, G493–G500. [Google Scholar] [CrossRef] [PubMed]
- Kunzelmann, K.; Mall, M. Electrolyte Transport in the Mammalian Colon: Mechanisms and Implications for Disease. Physiol. Rev. 2002, 82, 245–289. [Google Scholar] [CrossRef]
- Camilleri, M.; Sellin, J.H.; Barrett, K.E. Pathophysiology, Evaluation, and Management of Chronic Watery Diarrhea. Gastroenterology 2017, 152, 515–532.e2. [Google Scholar] [CrossRef]
- Keely, S.J.; Barrett, K.E. Intestinal Secretory Mechanisms and Diarrhea. Am. J. Physiol.-Gastrointest. Liver Physiol. 2022, 322, G405–G420. [Google Scholar] [CrossRef]
- Thiagarajah, J.R.; Verkman, A. CFTR Pharmacology and Its Role in Intestinal Fluid Secretion. Curr. Opin. Pharmacol. 2003, 3, 594–599. [Google Scholar] [CrossRef]
- Thiagarajah, J.R.; Verkman, A.S. New Drug Targets for Cholera Therapy. Trends Pharmacol. Sci. 2005, 26, 172–175. [Google Scholar] [CrossRef]
- Rhodes, J.B.; Schweitzer, D.; Ogg, J.E. Isolation of Non-O1 Vibrio Cholerae Associated with Enteric Disease of Herbivores in Western Colorado. J. Clin. Microbiol. 1985, 22, 572–575. [Google Scholar] [CrossRef]
- Murray, M.J. Enterotoxin Activity of a Salmonella Typhimurium of Equine Origin in Vivo in Rabbits and the Effect of Salmonella Culture Lysates and Cholera Toxin on Equine Colonic Mucosa in Vitro. Am. J. Vet. Res. 1986, 47, 769–773. [Google Scholar] [PubMed]
- Yurko-Mauro, K.A.; Reenstra, W.W. Prostaglandin F2αstimulates CFTR Activity by PKA- and PKC-Dependent Phosphorylation. Am. J. Physiol.-Cell Physiol. 1998, 275, C653–C660. [Google Scholar] [CrossRef] [PubMed]
- Schneider, D.A.; Sayegh, A.I. Gastrointestinal Neuroendocrinology. Vet. Clin. Equine Pract. 2002, 18, 205–217. [Google Scholar] [CrossRef] [PubMed]
- Moore, R.M.; Charalambous, A.C.; Masty, J. Alterations in Colonic Arterial and Venous Plasma Neuropeptide Concentrations in Horses during Low-Flow Ischemia and Reperfusion of the Large Colon. Am. J. Vet. Res. 1996, 57, 1200–1205. [Google Scholar] [CrossRef]
- Daels, P.F.; Stabenfeldt, G.H.; Hughes, J.P.; Odensvik, K.; Kindahl, H. Effects of Flunixin Meglumine on Endotoxin-Induced Prostaglandin F2α Secretion during Early Pregnancy in Mares. Am. J. Vet. Res. 1991, 52, 276–281. [Google Scholar] [CrossRef]
- McConnico, R.S.; Argenzio, R.A.; Roberts, M.C. Prostaglandin E2 and Reactive Oxygen Metabolite Damage in the Cecum in a Pony Model of Acute Colitis. Can. J. Vet. Res. 2002, 66, 50–54. [Google Scholar]
- König, J.; Schreiber, R.; Voelcker, T.; Mall, M.; Kunzelmann, K. The Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) Inhibits ENaC through an Increase in the Intracellular Cl− Concentration. EMBO Rep. 2001, 2, 1047–1051. [Google Scholar] [CrossRef]
- Konstas, A.-A.; Koch, J.-P.; Korbmacher, C. cAMP-Dependent Activation of CFTR Inhibits the Epithelial Sodium Channel (ENaC) without Affecting Its Surface Expression. Pflüg. Arch. 2003, 445, 513–521. [Google Scholar] [CrossRef]
- Wuchu, F.; Ma, X.; Que, Y.; Chen, J.; Ruan, Y.C. Biphasic Regulation of CFTR Expression by ENaC in Epithelial Cells: The Involvement of Ca2+-Modulated cAMP Production. Front. Cell Dev. Biol. 2022, 10, 781762. [Google Scholar] [CrossRef]
- Anand, D.; Hummler, E.; Rickman, O.J. ENaC Activation by Proteases. Acta Physiol. 2022, 235, e13811. [Google Scholar] [CrossRef]
- Rossier, B.C.; Stutts, M.J. Activation of the Epithelial Sodium Channel (ENaC) by Serine Proteases. Annu. Rev. Physiol. 2009, 71, 361–379. [Google Scholar] [CrossRef] [PubMed]
- Clarke, L.L.; Roberts, M.C.; Grubb, B.R.; Argenzio, R.A. Short-Term Effect of Aldosterone on Na-Cl Transport across Equine Colon. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 1992, 262, R939–R946. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, A.; Riber, C.; Trigo, P.; Castejón-Riber, C.; Castejón, F.M. Dehydration, Electrolyte Imbalances and Renin-Angiotensin-Aldosterone-Vasopressin Axis in Successful and Unsuccessful Endurance Horses. Equine Vet. J. 2010, 42, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Geibel, J.P. Secretion and Absorption by Colonic Crypts. Annu. Rev. Physiol. 2005, 67, 471–490. [Google Scholar] [CrossRef]
- Honegger, K.J.; Capuano, P.; Winter, C.; Bacic, D.; Stange, G.; Wagner, C.A.; Biber, J.; Murer, H.; Hernando, N. Regulation of Sodium-Proton Exchanger Isoform 3 (NHE3) by PKA and Exchange Protein Directly Activated by cAMP (EPAC). Proc. Natl. Acad. Sci. USA 2006, 103, 803–808. [Google Scholar] [CrossRef]
- He, P.; Yun, C.C. Mechanisms of the Regulation of the Intestinal Na+/H+Exchanger NHE3. J. Biomed. Biotechnol. 2010, 2010, 238080. [Google Scholar] [CrossRef]
- Musch, M.W.; Bookstein, C.; Xie, Y.; Sellin, J.H.; Chang, E.B. SCFA Increase Intestinal Na Absorption by Induction of NHE3 in Rat Colon and Human Intestinal C2/Bbe Cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 280, G687–G693. [Google Scholar] [CrossRef]
- von Engelhardt, W.; Bartels, J.; Kirschberger, S.; zu Düttingdorf, H.D.M.; Busche, R. Role of Short-chain Fatty Acids in the Hind Gut. Vet. Q. 1998, 20, 52–59. [Google Scholar] [CrossRef]
- Al Jassim, R.A.M.; Andrews, F.M. The Bacterial Community of the Horse Gastrointestinal Tract and Its Relation to Fermentative Acidosis, Laminitis, Colic, and Stomach Ulcers. Vet. Clin. N. Am. Equine Pract. 2009, 25, 199–215. [Google Scholar] [CrossRef]
- Sano, R.; Shinozaki, Y.; Ohta, T. Sodium–Glucose Cotransporters: Functional Properties and Pharmaceutical Potential. J. Diabetes Investig. 2020, 11, 770–782. [Google Scholar] [CrossRef]
- Hirschhorn, N.; Kinzie, J.L.; Sachar, D.B.; Northrup, R.S.; Taylor, J.O.; Ahmad, S.Z.; Phillips, R.A. Decrease in Net Stool Output in Cholera during Intestinal Perfusion with Glucose-Containing Solutions. N. Engl. J. Med. 1968, 279, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Heitzmann, D.; Warth, R. Physiology and Pathophysiology of Potassium Channels in Gastrointestinal Epithelia. Physiol. Rev. 2008, 88, 1119–1182. [Google Scholar] [CrossRef] [PubMed]
- Bauck, A. Mechanisms of Bicarbonate Secretion in the Equine Colon In Vitro. Ph.D. Thesis, University of Florida, Gainesville, FL, USA, 2019. [Google Scholar]
- Barmeyer, C.; Ye, J.H.; Soroka, C.; Geibel, P.; Hingsammer, L.M.; Weitgasser, L.; Atway, D.; Geibel, J.P.; Binder, H.J.; Rajendran, V.M. Identification of Functionally Distinct Na-HCO3 Co-Transporters in Colon. PLoS ONE 2013, 8, e62864. [Google Scholar] [CrossRef] [PubMed]
- Bauck, A.G.; Vidyasagar, S.; Freeman, D.E. Mechanisms of Bicarbonate Secretion in the Equine Colon Ex Vivo. Am. J. Vet. Res. 2022, 83, 8. [Google Scholar] [CrossRef]
- Davis, J.L. Nonsteroidal Anti-Inflammatory Drug Associated Right Dorsal Colitis in the Horse. Equine Vet. Educ. 2017, 29, 104–113. [Google Scholar] [CrossRef]
- Vinijkumthorn, R.; Bauck, A.G.; Vidyasagar, S.; Freeman, D.E. Nonsteroidal Antiinflammatory Drugs Reduce Anion Secretion and Increase Apoptosis in Equine Colonic Mucosa Ex Vivo. Am. J. Vet. Res. 2023, 1. [Google Scholar] [CrossRef]
- Argenzio, R.A. Physiology of Diarrhea—Large Intestine. J. Am. Vet. Med. Assoc. 1978, 173, 667–672. [Google Scholar]
- Ecke, P.; Hodgson, D.R.; Rose, R.J. Induced Diarrhoea in Horses Part 1: Fluid and Electrolytebalance. Vet. J. 1998, 155, 149–159. [Google Scholar] [CrossRef]
- Magdesian, K.G.; Dwyer, R.M.; Arguedas, M.G. Viral Diarrhea. Equine Infect. Dis. 2014, 198–203.e2. [Google Scholar] [CrossRef]
- Rikihisa, Y.; Johnson, G.C.; Wang, Y.Z.; Reed, S.M.; Fertel, R.; Cooke, H.J. Loss of Absorptive Capacity for Sodium and Chloride in the Colon Causes Diarrhoea in Potomac Horse Fever. Res. Vet. Sci. 1992, 52, 353–362. [Google Scholar] [CrossRef]
- Mallicote, M.; House, A.M.; Sanchez, L.C. A Review of Foal Diarrhoea from Birth to Weaning. Equine Vet. Educ. 2012, 24, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Jayaratne, R.; Barrett, K.E. The Role of Ion Transporters in the Pathophysiology of Infectious Diarrhea. Cell. Mol. Gastroenterol. Hepatol. 2018, 6, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Peritore-Galve, F.C.; Kaji, I.; Smith, A.; Walker, L.M.; Shupe, J.A.; Washington, M.K.; Algood, H.M.S.; Dudeja, P.K.; Goldenring, J.R.; Lacy, D.B. Increased Intestinal Permeability and Downregulation of Absorptive Ion Transporters Nhe3, Dra, and Sglt1 Contribute to Diarrhea during Clostridioides Difficile Infection. Gut Microbes 2023, 15, 2225841. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Gurjar, A.; Theoret, J.R.; Garcia, J.P.; Beingesser, J.; Freedman, J.C.; Fisher, D.J.; McClane, B.A.; Uzal, F.A. Synergistic Effects of Clostridium perfringens Enterotoxin and Beta Toxin in Rabbit Small Intestinal Loops. Infect. Immun. 2014, 82, 2958–2970. [Google Scholar] [CrossRef] [PubMed]
- Benz, R.; Popoff, M.R. Clostridium perfringens Enterotoxin: The Toxin Forms Highly Cation-Selective Channels in Lipid Bilayers. Toxins 2018, 10, 341. [Google Scholar] [CrossRef] [PubMed]
- Gibert, M.; Jolivet-Renaud, C.; Popoff, M.R. Beta2 Toxin, a Novel Toxin Produced by Clostridium perfringens. Gene 1997, 203, 65–73. [Google Scholar] [CrossRef]
- Fisher, D.J. Clostridium perfringens Beta2 Toxin: A Potential Accessory Toxin in Gastrointestinal Diseases of Humans and Domestic Animals. Ph.D. Dissertation, University of Pittsburgh, Pittsburgh, PA, USA, 2006. [Google Scholar]
- Gohari, I.M.; Arroyo, L.; MacInnes, J.I.; Timoney, J.F.; Parreira, V.R.; Prescott, J.F. Characterization of Clostridium perfringens in the Feces of Adult Horses and Foals with Acute Enterocolitis. Can. J. Vet. Res. 2014, 78, 1–7. [Google Scholar]
- Posthaus, H.; Kittl, S.; Tarek, B.; Bruggisser, J. Clostridium perfringens Type C Necrotic Enteritis in Pigs: Diagnosis, Pathogenesis, and Prevention. J. Vet. Diagn. Investig. 2020, 32, 203–212. [Google Scholar] [CrossRef]
- Marchelletta, R.R.; Gareau, M.G.; Mccole, D.F.; Okamoto, S.; Roel, E.; Klinkenberg, R.; Guiney, D.G.; Fierer, J.; Barrett, K.E. Altered Expression and Localization of Ion Transporters Contribute to Diarrhea in Mice With Salmonella-Induced Enteritis. Gastroenterology 2013, 145, 1358–1368.e4. [Google Scholar] [CrossRef]
- Lyczak, J.B.; Pier, G.B. Salmonella Enterica Serovar Typhi Modulates Cell Surface Expression of Its Receptor, the Cystic Fibrosis Transmembrane Conductance Regulator, on the Intestinal Epithelium. Infect. Immun. 2002, 70, 6416–6423. [Google Scholar] [CrossRef]
- Resta–Lenert, S.; Barrett, K.E. Enteroinvasive Bacteria Alter Barrier and Transport Properties of Human Intestinal Epithelium: Role of iNOS and COX-2. Gastroenterology 2002, 122, 1070–1087. [Google Scholar] [CrossRef] [PubMed]
- Quach, A.; Jayaratne, R.; Ibeawuchi, S.-R.; Das, S.; Barrett, K. Salmonella Biases Epithelial Differentiation in Murine Enteroids through Wnt and Notch Signaling, Which May Contribute to Diarrheal Pathogenesis. FASEB J. 2020, 34, S1. [Google Scholar] [CrossRef]
- Rikihisa, Y. Growth of Ehrlichia Risticii in Human Colonic Epithelial Cells. Ann. N. Y. Acad. Sci. 1990, 590, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Poeta, M.; Cioffi, V.; Buccigrossi, V.; Corcione, F.; Peltrini, R.; Amoresano, A.; Magurano, F.; Viscardi, M.; Fusco, G.; Tarallo, A.; et al. SARS-CoV-2 Causes Secretory Diarrhea with an Enterotoxin-like Mechanism, Which Is Reduced by Diosmectite. Heliyon 2022, 8, e10246. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.-L.; Song, W.; Gao, Z.; Su, X.-F.; Nie, H.-G.; Jiang, Y.; Peng, J.-B.; He, Y.-X.; Liao, Y.; Zhou, Y.-J.; et al. SARS-CoV Proteins Decrease Levels and Activity of Human ENaC via Activation of Distinct PKC Isoforms. Am. J. Physiol. -Lung Cell. Mol. Physiol. 2009, 296, L372–L383. [Google Scholar] [CrossRef]
- Walshe, N.; Mulcahy, G.; Crispie, F.; Cabrera-Rubio, R.; Cotter, P.; Jahns, H.; Duggan, V. Outbreak of Acute Larval Cyathostominosis—A “Perfect Storm” of Inflammation and Dysbiosis. Equine Vet. J. 2021, 53, 727–739. [Google Scholar] [CrossRef]
- Hellman, S. Generation of Equine Enteroids and Enteroid-Derived 2D Monolayers That Are Responsive to Microbial Mimics. Vet. Res. 2021, 52, 108. [Google Scholar] [CrossRef]
- Baird, A.W.; O’Malley, K.E. Epithelial Ion Transport—Possible Contribution to Parasite Expulsion. Parasitol. Today 1993, 9, 141–143. [Google Scholar] [CrossRef]
- Willette, J.A.; Kopper, J.J.; Kogan, C.J.; Seguin, M.A.; Schott, H.C. Effect of Season and Geographic Location in the United States on Detection of Potential Enteric Pathogens or Toxin Genes in Horses ≥6-Mo-Old. J. Vet. Diagn. Investig. 2022, 34, 407–411. [Google Scholar] [CrossRef]
- Luethy, D.; Feldman, R.; Stefanovski, D.; Aitken, M.R. Risk Factors for Laminitis and Nonsurvival in Acute Colitis: Retrospective Study of 85 Hospitalized Horses (2011–2019). J. Vet. Intern. Med. 2021, 35, 2019–2025. [Google Scholar] [CrossRef]
- Kullmann, A.; Holcombe, S.J.; Hurcombe, S.D.; Roessner, H.A.; Hauptman, J.G.; Geor, R.J.; Belknap, J. Prophylactic Digital Cryotherapy Is Associated with Decreased Incidence of Laminitis in Horses Diagnosed with Colitis. Equine Vet. J. 2014, 46, 554–559. [Google Scholar] [CrossRef] [PubMed]
- Kovač, M.; Huskamp, B.; Scheidemann, W.; Toth, J.; Tambur, Z. Survival and Evaluation of Clinical and Laboratory Variables as Prognostic Indicators in Horses Hospitilized with Acute Diarrhea: 342 Cases (1995–2015). Acta Vet. 2017, 67, 356–365. [Google Scholar] [CrossRef]
- Cohen, N.D.; Woods, A.M. Characteristics and Risk Factors for Failure of Horses with Acute Diarrhea to Survive: 122 Cases (1990–1996). J. Am. Vet. Med. Assoc. 1999, 214, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Sage, S.E.; Bedenice, D.; McKinney, C.A.; Long, A.E.; Pacheco, A.; Wagner, B.; Mazan, M.R.; Paradis, M.R. Assessment of the Impact of Age and of Blood-Derived Inflammatory Markers in Horses with Colitis. J. Vet. Emerg. Crit. Care 2021, 31, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Gomez, D.E.; Arroyo, L.G.; Stämpfli, H.R.; Cruz, L.E.; Oliver, O.J. Physicochemical Interpretation of Acid-Base Abnormalities in 54 Adult Horses with Acute Severe Colitis and Diarrhea. J. Vet. Intern. Med. 2013, 27, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Manship, A.J.; Blikslager, A.T.; Elfenbein, J.R. Disease Features of Equine Coronavirus and Enteric Salmonellosis Are Similar in Horses. J. Vet. Intern. Med. 2019, 33, 912–917. [Google Scholar] [CrossRef]
- Uzal, F.A.; Arroyo, L.G.; Navarro, M.A.; Gomez, D.E.; Asín, J.; Henderson, E. Bacterial and Viral Enterocolitis in Horses: A Review. J. Vet. Diagn. Investig. 2022, 34, 354–375. [Google Scholar] [CrossRef]
- Dolente, B.A.; Wilkins, P.A.; Boston, R.C. Clinicopathologic Evidence of Disseminated Intravascular Coagulation in Horses with Acute Colitis. J. Am. Vet. Med. Assoc. 2002, 220, 1034–1038. [Google Scholar] [CrossRef]
- Winston, J.A.; Theriot, C.M. Impact of Microbial Derived Secondary Bile Acids on Colonization Resistance against Clostridium Difficile in the Gastrointestinal Tract. Anaerobe 2016, 41, 44–50. [Google Scholar] [CrossRef]
- Weese, J.S.; Staempfli, H.R.; Prescott, J.F. A Prospective Study of the Roles of Clostridium Difficile and Enterotoxigenic Clostridium perfringens in Equine Diarrhoea. Equine Vet. J. 2001, 33, 403–409. [Google Scholar] [CrossRef]
- Båverud, V.; Gustafsson, A.; Franklin, A.; Lindholm, A.; Gunnarsson, A. Clostridium Difficile Associated with Acute Colitis in Mature Horses Treated with Antibiotics. Equine Vet. J. 1997, 29, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Båverud, V.; Franklin, A.; Gunnarsson, A.; Gustafsson, A.; Hellander-Edman, A. Clostridium Difficile Associated with Acute Colitis in Mares When Their Foals Are Treated with Erythromycin and Rifampicin for Rhodococcus Equi Pneumonia. Equine Vet. J. 1998, 30, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Voth, D.E.; Ballard, J.D. Clostridium Difficile Toxins: Mechanism of Action and Role in Disease. Clin. Microbiol. Rev. 2005, 18, 247–263. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, L.G.; Gomez, D.E.; Martins, C. Equine Duodenitis-Proximal Jejunitis: A Review. Can. Vet. J. 2018, 59, 510–517. [Google Scholar] [PubMed]
- Diab, S.S.; Rodriguez-Bertos, A.; Uzal, F.A. Pathology and Diagnostic Criteria of Clostridium Difficile Enteric Infection in Horses. Vet. Pathol. 2013, 50, 1028–1036. [Google Scholar] [CrossRef]
- Kuttappan, D.A.; Mooyottu, S.; Sponseller, B.A. An Overview of Equine Enteric Clostridial Diseases. Vet. Clin. N. Am. Equine Pract. 2023, 39, 15–23. [Google Scholar] [CrossRef]
- Weese, J.S.; Toxopeus, L.; Arroyo, L. Clostridium Difficile Associated Diarrhoea in Horses within the Community: Predictors, Clinical Presentation and Outcome. Equine Vet. J. 2006, 38, 185–188. [Google Scholar] [CrossRef]
- Diab, S.S.; Songer, G.; Uzal, F.A. Clostridium Difficile Infection in Horses: A Review. Vet. Microbiol. 2013, 167, 42–49. [Google Scholar] [CrossRef]
- Chapman, A.M. Acute Diarrhea in Hospitalized Horses. Vet. Clin. N. Am. Equine Pract. 2009, 25, 363–380. [Google Scholar] [CrossRef]
- Båverud, V.; Gustafsson, A.; Franklin, A.; Aspán, A.; Gunnarsson, A. Clostridium Difficile: Prevalence in Horses and Environment, and Antimicrobial Susceptibility. Equine Vet. J. 2003, 35, 465–471. [Google Scholar] [CrossRef]
- Rodriguez, C.; Taminiau, B.; Brévers, B.; Avesani, V.; Van Broeck, J.; Leroux, A.; Gallot, M.; Bruwier, A.; Amory, H.; Delmée, M.; et al. Faecal Microbiota Characterisation of Horses Using 16 Rdna Barcoded Pyrosequencing, and Carriage Rate of Clostridium Difficile at Hospital Admission. BMC Microbiol. 2015, 15, 181. [Google Scholar] [CrossRef] [PubMed]
- Madewell, B.R.; Tang, Y.J.; Jang, S.; Madigan, J.E.; Hirsh, D.C.; Gumerlock, P.H.; Silva, J. Apparent Outbreaks of Clostridium Difficile-Associated Diarrhea in Horses in a Veterinary Medical Teaching Hospital. J. Vet. Diagn. Investig. 1995, 7, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Schoster, A.; Arroyo, L.G.; Staempfli, H.R.; Shewen, P.E.; Weese, J.S. Presence and Molecular Characterization of Clostridium Difficile and Clostridium perfringens in Intestinal Compartments of Healthy Horses. BMC Vet. Res. 2012, 8, 94. [Google Scholar] [CrossRef] [PubMed]
- Stark, P.; Lee, A.; Parsonage, B. Colonization of the Large Bowel by Clostridium Difficile in Healthy Infants: Quantitative Study. Infect. Immun. 1982, 35, 895–899. [Google Scholar] [CrossRef] [PubMed]
- Sorg, J.A.; Sonenshein, A.L. Bile Salts and Glycine as Cogerminants for Clostridium Difficile Spores. J. Bacteriol. 2008, 190, 2505–2512. [Google Scholar] [CrossRef] [PubMed]
- Kuehne, S.A.; Cartman, S.T.; Minton, N.P. Both, Toxin A and Toxin B, Are Important in Clostridium Difficile Infection. Gut Microbes 2011, 2, 252–255. [Google Scholar] [CrossRef]
- Arroyo, L.G.; Staempfli, H.; Weese, J.S. Molecular Analysis of Clostridium Difficile Isolates Recovered from Horses with Diarrhea. Vet. Microbiol. 2007, 120, 179–183. [Google Scholar] [CrossRef]
- Davies, A.H.; Roberts, A.K.; Shone, C.C.; Acharya, K.R. Super Toxins from a Super Bug: Structure and Function of Clostridium Difficile Toxins. Biochem. J. 2011, 436, 517–526. [Google Scholar] [CrossRef]
- Orrell, K.E.; Zhang, Z.; Sugiman-Marangos, S.N.; Melnyk, R.A. Clostridium Difficile Toxins A and B: Receptors, Pores, and Translocation into Cells. Crit. Rev. Biochem. Mol. Biol. 2017, 52, 461–473. [Google Scholar] [CrossRef]
- Dierick, E.; Goossens, E.; Prescott, J.F.; Ducatelle, R.; Van Immerseel, F. Enteric Clostridia. In Pathogenesis of Bacterial Infections in Animals; Prescott, J.F., Rycroft, A.N., Boyce, J.D., MacInnes, J.I., Van Immerseel, F., Vázquez-Boland, J.A., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2022; pp. 607–634. ISBN 9781119754862. [Google Scholar]
- Uzal, F.A.; Vidal, J.E.; McClane, B.A.; Gurjar, A.A. Clostridium perfringens Toxins Involved in Mammalian Veterinary Diseases. Open Toxinology J. 2010, 2, 24–42. [Google Scholar] [CrossRef]
- Tillotson, K.; Traub-Dargatz, J.L.; Dickinson, C.E.; Ellis, R.P.; Morley, P.S.; Hyatt, D.R.; Magnuson, R.J.; Riddle, W.T.; Bolte, D.; Salman, M.D. Population-Based Study of Fecal Shedding of Clostridium perfringens in Broodmares and Foals. J. Am. Vet. Med. Assoc. 2002, 220, 342–348. [Google Scholar] [CrossRef] [PubMed]
- East, L.M.; Dargatz, D.A.; Traub-Dargatz, J.L.; Savage, C.J. Foaling-Management Practices Associated with the Occurrence of Enterocolitis Attributed to Clostridium perfringens Infection in the Equine Neonate. Prev. Vet. Med. 2000, 46, 61–74. [Google Scholar] [CrossRef]
- Wierup, M. Equine Intestinal Clostridiosis. An Acute Disease in Horses Associated with High Intestinal Counts of Clostridium perfringens Type A. Acta Vet. Scand. Suppl. 1977, 62, 1–182. [Google Scholar]
- Zakia, L.S.; Arroyo, L.G.; Gomez, D.E.; Boerlin, P.; Surette, M.G.; Lillie, B.N. Postmortem Examination of Equids >1-Year-Old with Enterotyphlocolitis in Ontario: A Retrospective Descriptive Study. J. Vet. Diagn. Investig. 2023, 35, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Macías-Rioseco, M.; Hill, A.E.; Uzal, F.A. Fatal Intestinal Inflammatory Lesions in Equids in California: 710 Cases (1990–2013). J. Am. Vet. Med. Assoc. 2020, 256, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Mendonça, F.S.; Navarro, M.A.; Uzal, F.A. The Comparative Pathology of Enterocolitis Caused by Clostridium perfringens Type C, Clostridioides Difficile, Paeniclostridium Sordellii, Salmonella Enterica Subspecies Enterica Serovar Typhimurium, and Nonsteroidal Anti-Inflammatory Drugs in Horses. J. Vet. Diagn. Investig. 2022, 34, 412–420. [Google Scholar] [CrossRef]
- Diab, S.S.; Kinde, H.; Moore, J.; Shahriar, M.F.; Odani, J.; Anthenill, L.; Songer, G.; Uzal, F.A. Pathology of Clostridium perfringens Type C Enterotoxemia in Horses. Vet. Pathol. 2012, 49, 255–263. [Google Scholar] [CrossRef]
- Griffiths, N.J.; Walton, J.R.; Edwards, G.B. An Investigation of the Prevalence of the Toxigenic Types of Clostridium perfringens in Horses with Anterior Enteritis: Preliminary Results. Anaerobe 1997, 3, 121–125. [Google Scholar] [CrossRef]
- Ochoa, R.; Kern, S.R. The Effects of Clostridium perfringens Type A Enterotoxin in Shetland Ponies—Clinical, Morphologic and Clinicopathologic Changes. Vet. Pathol. 1980, 17, 738–747. [Google Scholar] [CrossRef]
- Mehdizadeh Gohari, I. Clostridium perfringens and Its Potential Role in Equine Colitis. Master’s Thesis, University of Guelph, Guelph, ON, Canada, 2012. [Google Scholar]
- Andersson, G.; Ekman, L.; Månsson, I.; Persson, S.; Rubarth, S.; Tufvesson, G. Lethal Complications Following Administration of Oxytetracycline in the Horse. Nord. Vet. Med. 1971, 23, 9–22. [Google Scholar]
- Sayeed, S.; Uzal, F.A.; Fisher, D.J.; Saputo, J.; Vidal, J.E.; Chen, Y.; Gupta, P.; Rood, J.I.; McClane, B.A. Beta Toxin Is Essential for the Intestinal Virulence of Clostridium perfringens Type C Disease Isolate CN3685 in a Rabbit Ileal Loop Model. Mol. Microbiol. 2008, 67, 15–30. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, M.T.; Palmer, J.E. Prevalence of Clostridium perfringens Enterotoxin and Clostridium Difficile Toxin A in Feces of Horses with Diarrhea and Colic. J. Am. Vet. Med. Assoc. 1999, 215, 358–361. [Google Scholar] [CrossRef] [PubMed]
- Herholz, C.; Miserez, R.; Nicolet, J.; Frey, J.; Popoff, M.; Gibert, M.; Gerber, H.; Straub, R. Prevalence of Β2-Toxigenic Clostridium perfringens in Horses with Intestinal Disorders. J. Clin. Microbiol. 1999, 37, 358–361. [Google Scholar] [CrossRef] [PubMed]
- Bacciarini, L.N.; Boerlin, P.; Straub, R.; Frey, J.; Gröne, A. Immunohistochemical Localization of Clostridium perfringens Β2-Toxin in the Gastrointestinal Tract of Horses. Vet. Pathol. 2003, 40, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Freedman, J.C.; Shrestha, A.; McClane, B.A. Clostridium perfringens Enterotoxin: Action, Genetics, and Translational Applications. Toxins 2016, 8, 73. [Google Scholar] [CrossRef]
- Camargo, A.; Ramírez, J.D.; Kiu, R.; Hall, L.J.; Muñoz, M. Unveiling the Pathogenic Mechanisms of Clostridium perfringens Toxins and Virulence Factors. Emerg. Microbes Infect. 2024, 13, 2341968. [Google Scholar] [CrossRef]
- Mehdizadeh Gohari, I.; Navarro, M.A.; Li, J.; Shrestha, A.; Uzal, F.; McClane, B.A. Pathogenicity and Virulence of Clostridium perfringens. Virulence 2021, 12, 723–753. [Google Scholar] [CrossRef]
- Kiu, R.; Hall, L.J. An Update on the Human and Animal Enteric Pathogen Clostridium perfringens. Emerg. Microbes Infect. 2018, 7, 141. [Google Scholar] [CrossRef]
- Gohari, I.M.; Brefo-Mensah, E.K.; Palmer, M.; Boerlin, P.; Prescott, J.F. Sialic Acid Facilitates Binding and Cytotoxic Activity of the Pore-Forming Clostridium perfringens NetF Toxin to Host Cells. PLoS ONE 2018, 13, e0206815. [Google Scholar] [CrossRef]
- Mehdizadeh Gohari, I.; Parreira, V.R.; Timoney, J.F.; Fallon, L.; Slovis, N.; Prescott, J.F. NetF-Positive Clostridium perfringens in Neonatal Foal Necrotising Enteritis in Kentucky. Vet. Rec. 2016, 178, 216. [Google Scholar] [CrossRef]
- Mehdizadeh Gohari, I.; Unterer, S.; Whitehead, A.E.; Prescott, J.F. NetF-Producing Clostridium perfringens and Its Associated Diseases in Dogs and Foals. J. Vet. Diagn. Investig. 2020, 32, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Finley, A.; Gohari, I.M.; Parreira, V.R.; Abrahams, M.; Staempfli, H.R.; Prescott, J.F. Prevalence of netF-Positive Clostridium perfringens in Foals in Southwestern Ontario. Can. J. Vet. Res. 2016, 80, 242–244. [Google Scholar] [PubMed]
- Traub-Dargatz, J.L.; Jones, R.L. Clostridia-Associated Enterocolitis in Adult Horses and Foals. Vet. Clin. N. Am. Equine Pract. 1993, 9, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Macias Rioseco, M.; Beingesser, J.; Uzal, F.A. Freezing or Adding Trypsin Inhibitor to Equine Intestinal Contents Extends the Lifespan of Clostridium perfringens Beta Toxin for Diagnostic Purposes. Anaerobe 2012, 18, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Traub-Dargatz, J.L.; Salman, M.D.; Jones, R.L. Epidemiologic Study of Salmonellae Shedding in the Feces of Horses and Potential Risk Factors for Development of the Infection in Hospitalized Horses. J. Am. Vet. Med. Assoc. 1990, 196, 1617–1622. [Google Scholar] [CrossRef]
- Ekiri, A.B.; Morton, A.J.; Long, M.T.; MacKay, R.J.; Hernandez, J.A. Review of the Epidemiology and Infection Control Aspects of Nosocomial Salmonella Infections in Hospitalised Horses. Equine Vet. Educ. 2010, 22, 631–641. [Google Scholar] [CrossRef]
- Dallap Schaer, B.L.; Aceto, H.; Caruso, M.A., III; Brace, M.A. Identification of Predictors of Salmonella Shedding in Adult Horses Presented for Acute Colic. J. Vet. Intern. Med. 2012, 26, 1177–1185. [Google Scholar] [CrossRef]
- Timoney, J. Salmonella Infections in Horses. In Salmonella in Domestic Animals; Barrow, P., Methner, U., Eds.; CAB International: Wallingford Oxon, UK, 2013; pp. 305–317. [Google Scholar]
- Rogers, A.W.L.; Tsolis, R.M.; Bäumler, A.J. Salmonella versus the Microbiome. Microbiol. Mol. Biol. Rev. 2020, 85, 10–1128. [Google Scholar] [CrossRef]
- Bavishi, C.; DuPont, H.L. Systematic Review: The Use of Proton Pump Inhibitors and Increased Susceptibility to Enteric Infection. Aliment. Pharmacol. Ther. 2011, 34, 1269–1281. [Google Scholar] [CrossRef]
- Amory, H.; Cesarini, C.; De Maré, L.; Loublier, C.; Moula, N.; Detilleux, J.; Saulmont, M.; Garigliany, M.-M.; Lecoq, L. Relationship between the Cycle Threshold Value (Ct) of a Salmonella Spp. qPCR Performed on Feces and Clinical Signs and Outcome in Horses. Microorganisms 2023, 11, 1950. [Google Scholar] [CrossRef]
- Barrow, P.A.; Jones, M.A.; Mellor, K.C.; Thomson, N.R. Salmonella. In Pathogenesis of Bacterial Infection in Animals; Prescott, J.F., Rycroft, A.N., Boyce, J.D., MacInnes, J.I., Van Immerseel, F., Vázquez-Boland, J.A., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2022; pp. 162–199. ISBN 9781119754862. [Google Scholar]
- Rothers, K.L.; Hackett, E.S.; Mason, G.L.; Nelson, B.B. Atypical Salmonellosis in a Horse: Implications for Hospital Safety. Case Rep. Vet. Med. 2020, 2020, 7062408. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.P.; Reina-Guerra, M.; Hardy, A.J.; Habasha, F. Equine Salmonellosis: Experimental Production of Four Syndromes. Am. J. Vet. Res. 1979, 40, 1072–1077. [Google Scholar] [PubMed]
- Smith, B.P. Salmonella Infections in Horses. Compend. Contin. Educ. Pract. Vet. 1981, 3, S4–S17. [Google Scholar]
- McCain, C.S.; Powell, K.C. Asymptomatic Salmonellosis in Healthy Adult Horses. J. Vet. Diagn. Investig. 1990, 2, 236–237. [Google Scholar] [CrossRef] [PubMed]
- House, J.K.; Smith, B.P.; Wildman, T.R.; Carrigan, M.J.; Kamiya, D.Y. Isolation of Salmonella Organisms from the Mesenteric Lymph Nodes of Horses at Necropsy. J. Am. Vet. Med. Assoc. 1999, 215, 507–510. [Google Scholar] [CrossRef] [PubMed]
- Stevens, M.P.; Kingsley, R.A. Salmonella Pathogenesis and Host-Adaptation in Farmed Animals. Curr. Opin. Microbiol. 2021, 63, 52–58. [Google Scholar] [CrossRef]
- Larsen, J. Acute Colitis in Adult Horses. A Review with Emphasis on Aetiology and Pathogenesis. Vet. Q. 1997, 19, 72–80. [Google Scholar] [CrossRef]
- Lou, L.; Zhang, P.; Piao, R.; Wang, Y. Salmonella Pathogenicity Island 1 (SPI-1) and Its Complex Regulatory Network. Front. Cell. Infect. Microbiol. 2019, 9, 270. [Google Scholar] [CrossRef]
- Kombade, S.; Kaur, N.; Kombade, S.; Kaur, N. Pathogenicity Island in Salmonella. In Salmonella spp.—A Global Challenge; IntechOpen: London, UK, 2021; ISBN 978-1-83969-018-1. [Google Scholar]
- Marijke Keestra, A.; Winter, M.G.; Klein-Douwel, D.; Xavier, M.N.; Winter, S.E.; Kim, A.; Tsolis, R.M.; Bäumler, A.J. A Salmonella Virulence Factor Activates the NOD1/NOD2 Signaling Pathway. mBio 2011, 2, e00266-11. [Google Scholar] [CrossRef]
- Palmer, A.D.; Slauch, J.M. Mechanisms of Salmonella Pathogenesis in Animal Models. Hum. Ecol. Risk Assess. HERA 2017, 23, 1877–1892. [Google Scholar] [CrossRef]
- Quach, A.; Jayaratne, R.R.; Lee, B.J.; Ibeawuchi, S.-R.; Lim, E.; Das, S.; Barrett, K.E. Diarrhoeal Pathogenesis in Salmonella Infection May Result from an Imbalance in Intestinal Epithelial Differentiation through Reduced Notch Signalling. J. Physiol. 2022, 600, 1851–1865. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.J.; Doran, R.E.; Pfeiffer, C.J.; Tyler, D.E.; Moore, J.N.; Sriranganathan, N. Comparative Effects of Cholera Toxin, Salmonella Typhimurium Culture Lysate, and Viable Salmonella Typhimurium in Isolated Colon Segments in Ponies. Am. J. Vet. Res. 1989, 50, 22–28. [Google Scholar] [PubMed]
- Barza, M.; Giuliano, M.; Jacobus, N.V.; Gorbach, S.L. Effect of Broad-Spectrum Parenteral Antibiotics on “Colonization Resistance” of Intestinal Microflora of Humans. Antimicrob. Agents Chemother. 1987, 31, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Stecher, B. The Roles of Inflammation, Nutrient Availability and the Commensal Microbiota in Enteric Pathogen Infection. Microbiol. Spectr. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Keel, M.K.; Songer, J.G. The Comparative Pathology of Clostridium Difficile-Associated Disease. Vet. Pathol. 2006, 43, 225–240. [Google Scholar] [CrossRef]
- Garber, A.; Hastie, P.; Murray, J.-A. Factors Influencing Equine Gut Microbiota: Current Knowledge. J. Equine Vet. Sci. 2020, 88, 102943. [Google Scholar] [CrossRef]
- Collinet, A.; Grimm, P.; Julliand, S.; Julliand, V. Multidimensional Approach for Investigating the Effects of an Antibiotic–Probiotic Combination on the Equine Hindgut Ecosystem and Microbial Fibrolysis. Front. Microbiol. 2021, 12, 646294. [Google Scholar] [CrossRef]
- Vilei, E.M.; Schlatter, Y.; Perreten, V.; Straub, R.; Popoff, M.R.; Gibert, M.; Gröne, A.; Frey, J. Antibiotic-Induced Expression of a Cryptic Cpb2 Gene in Equine Beta2-Toxigenic Clostridium perfringens. Mol. Microbiol. 2005, 57, 1570–1581. [Google Scholar] [CrossRef]
- Staempfli, H.R.; Prescott, J.F.; Brash, M.L. Lincomycin-Induced Severe Colitis in Ponies: Association with Clostridium Cadaveris. Can. J. Vet. Res. 1992, 56, 168–169. [Google Scholar]
- Weese, J.S. Clostridium Difficile Associated Enterocolitis in Adult Horses and Foals. D.V.Sc. Thesis, University of Guelph, Guelph, ON, Canada, 2000. [Google Scholar]
- Wilson, D.A.; MacFadden, K.E.; Green, E.M.; Crabill, M.; Frankeny, R.L.; Thorne, J.G. Case Control and Historical Cohort Study of Diarrhea Associated With Administration of Trimethoprim-Potentiated Sulphonamides to Horses and Ponies. J. Vet. Intern. Med. 1996, 10, 258–264. [Google Scholar] [CrossRef]
- Gustafsson, A.; Båverud, V.; Gunnarsson, A.; Rantzien, M.H.; Lindholm, A.; Franklin, A. The Association of Erythromycin Ethylsuccinate with Acute Colitis in Horses in Sweden. Equine Vet. J. 1997, 29, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Keir, A.A.; Stämpfli, H.R.; Crawford, J. Outbreak of Acute Colitis on a Horse Farm Associated with Tetracycline-Contaminated Sweet Feed. Can. Vet. J. 1999, 40, 718–720. [Google Scholar] [PubMed]
- Gomez, D.E.; Dunkel, B.; Renaud, D.L.; Arroyo, L.G.; Schoster, A.; Kopper, J.J.; Byrne, D.; Group, T.M.; Toribio, R.E. Survival Rates and Factors Associated with Survival and Laminitis of Horses with Acute Diarrhoea Admitted to Referral Institutions. Equine Vet. J. 2024, 56, 970–981. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.C.; Weese, J.S. The Equine Intestinal Microbiome. Anim. Health Res. Rev. 2012, 13, 121–128. [Google Scholar] [CrossRef]
- Dutta, S.K.; Myrup, A.C.; Rice, R.M.; Robl, M.G.; Hammond, R.C. Experimental Reproduction of Potomac Horse Fever in Horses with a Newly Isolated Ehrlichia Organism. J. Clin. Microbiol. 1985, 22, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Budachetri, K.; Lin, M.; Yan, Q.; Chien, R.C.; Hostnik, L.D.; Haanen, G.; Leclère, M.; Waybright, W.; Baird, J.D.; Arroyo, L.G.; et al. Real-Time PCR Differential Detection of Neorickettsia Findlayensis and N. Risticii in Cases of Potomac Horse Fever. J. Clin. Microbiol. 2022, 60, e00250-22. [Google Scholar] [CrossRef]
- Teymournejad, O.; Lin, M.; Bekebrede, H.; Kamr, A.; Toribio, R.E.; Arroyo, L.G.; Baird, J.D.; Rikihisa, Y. Isolation and Molecular Analysis of a Novel Neorickettsia Species That Causes Potomac Horse Fever. mBio 2020, 11, e03429-19. [Google Scholar] [CrossRef]
- Chaichanasiriwithaya, W.; Rikihisa, Y.; Yamamoto, S.; Reed, S.; Crawford, T.B.; Perryman, L.E.; Palmer, G.H. Antigenic, Morphologic, and Molecular Characterization of New Ehrlichia Risticii Isolates. J. Clin. Microbiol. 1994, 32, 3026–3033. [Google Scholar] [CrossRef]
- Bertin, F.R.; Reising, A.; Slovis, N.M.; Constable, P.D.; Taylor, S.D. Clinical and Clinicopathological Factors Associated with Survival in 44 Horses with Equine Neorickettsiosis (Potomac Horse Fever). J. Vet. Intern. Med. 2013, 27, 1528–1534. [Google Scholar] [CrossRef]
- Arroyo, L.G.; Moore, A.; Bedford, S.; Gomez, D.E.; Teymournejad, O.; Xiong, Q.; Budachetri, K.; Bekebrede, H.; Rikihisa, Y.; Baird, J.D. Potomac Horse Fever in Ontario: Clinical, Geographic, and Diagnostic Aspects. Can. Vet. J. 2021, 62, 622–628. [Google Scholar]
- Coffman, E.A.; Abd-Eldaim, M.; Craig, L.E. Abortion in a Horse Following Neorickettsia Risticii Infection. J. Vet. Diagn. Investig. 2008, 20, 827–830. [Google Scholar] [CrossRef]
- McBride, J.W.; Ganta, R.R.; Walker, D.H. Rickettsiales. In Pathogenesis of Bacterial Infections in Animals; Prescott, J.F., Rycroft, A.N., Boyce, J.D., MacInnes, J.I., Van Immerseel, F., Vázquez-Boland, J.A., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2022; pp. 456–485. ISBN 9781119754862. [Google Scholar]
- Palmer, J.E. Potomac Horse Fever. Vet. Clin. N. Am. Equine Pract. 1993, 9, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Madigan, J.E.; Barlough, J.E.; Rikihisa, Y.; Wen, B.; Miller, P.E.; Sampson, T.J. Identification of an Enzootic Diarrhea (“Shasta River Crud”) in Northern California as Potomac Horse Fever. J. Equine Vet. Sci. 1997, 17, 270. [Google Scholar] [CrossRef]
- Dutra, F.; Schuch, L.F.D.; Delucchi, E.; Curcio, B.R.; Coimbra, H.; Raffi, M.B.; Dellagostin, O.; Riet-Correa, F. Equine Monocytic Ehrlichiosis (Potomac Horse Fever) in Horses in Uruguay and Southern Brazil. J. Vet. Diagn. Investig. 2001, 13, 433–437. [Google Scholar] [CrossRef] [PubMed]
- Baird, J.D.; Arroyo, L.G. Historical Aspects of Potomac Horse Fever in Ontario (1924–2010). Can. Vet. J. 2013, 54, 565–572. [Google Scholar]
- Madigan, J.E.; Pusterla, N.; Johnson, E.; Chae, J.-S.; Pusterla, J.B.; Derock, E.; Lawler, S.P. Transmission of Ehrlichia Risticii, the Agent of Potomac Horse Fever, Using Naturally Infected Aquatic Insects and Helminth Vectors: Preliminary Report. Equine Vet. J. 2000, 32, 275–279. [Google Scholar] [CrossRef]
- Tkach, V.V.; Greiman, S.E. Transmission Biology, Host Associations, Distribution and Molecular Diagnostics of Neorickettsia. In Rickettsiales: Biology, Molecular Biology, Epidemiology, and Vaccine Development; Thomas, S., Ed.; Springer International Publishing: Cham, Switzerland, 2016; pp. 295–325. ISBN 978-3-319-46859-4. [Google Scholar]
- Rikihisa, Y.; Perry, B.D.; Cordes, D.O. Ultrastructural Study of Ehrlichial Organisms in the Large Colons of Ponies Infected with Potomac Horse Fever. Infect. Immun. 1985, 49, 505–512. [Google Scholar] [CrossRef]
- Sanchez, L.C. Chapter 12—Disorders of the Gastrointestinal System. In Equine Internal Medicine, 4th ed.; Reed, S.M., Bayly, W.M., Sellon, D.C., Eds.; W.B. Saunders: Philadelphia, PA, USA, 2018; pp. 709–842. ISBN 978-0-323-44329-6. [Google Scholar]
- Sauvanet, C.; Wayt, J.; Pelaseyed, T.; Bretscher, A. Structure, Regulation, and Functional Diversity of Microvilli on the Apical Domain of Epithelial Cells. Annu. Rev. Cell Dev. Biol. 2015, 31, 593–621. [Google Scholar] [CrossRef]
- van der Post, S.; Hansson, G.C. Membrane Protein Profiling of Human Colon Reveals Distinct Regional Differences. Mol. Cell. Proteom. MCP 2014, 13, 2277–2287. [Google Scholar] [CrossRef]
- Pusterla, N. Equine Coronaviruses. Vet. Clin. N. Am. Equine Pract. 2023, 39, 55–71. [Google Scholar] [CrossRef]
- Hepworth-Warren, K.L.; Erwin, S.J.; Moore, C.B.; Talbot, J.R.; Young, K.A.S.; Neault, M.J.; Haugland, J.C.; Robertson, J.B.; Blikslager, A.T. Risk Factors Associated with an Outbreak of Equine Coronavirus at a Large Farm in North Carolina. Front. Vet. Sci. 2023, 10, 1060759. [Google Scholar] [CrossRef] [PubMed]
- Fielding, C.L.; Higgins, J.K.; Higgins, J.C.; McIntosh, S.; Scott, E.; Giannitti, F.; Mete, A.; Pusterla, N. Disease Associated with Equine Coronavirus Infection and High Case Fatality Rate. J. Vet. Intern. Med. 2015, 29, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Berryhill, E.H.; Magdesian, K.G.; Aleman, M.; Pusterla, N. Clinical Presentation, Diagnostic Findings, and Outcome of Adult Horses with Equine Coronavirus Infection at a Veterinary Teaching Hospital: 33 Cases (2012–2018). Vet. J. 2019, 248, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Kooijman, L.J.; James, K.; Mapes, S.M.; Theelen, M.J.P.; Pusterla, N. Seroprevalence and Risk Factors for Infection with Equine Coronavirus in Healthy Horses in the USA. Vet. J. 2017, 220, 91–94. [Google Scholar] [CrossRef] [PubMed]
- Haake, C.; Cook, S.; Pusterla, N.; Murphy, B. Coronavirus Infections in Companion Animals: Virology, Epidemiology, Clinical and Pathologic Features. Viruses 2020, 12, 1023. [Google Scholar] [CrossRef]
- Mattei, D.N.; Kopper, J.J.; Sanz, M.G. Equine Coronavirus-Associated Colitis in Horses: A Retrospective Study. J. Equine Vet. Sci. 2020, 87, 102906. [Google Scholar] [CrossRef]
- Nemoto, M.; Schofield, W.; Cullinane, A. The First Detection of Equine Coronavirus in Adult Horses and Foals in Ireland. Viruses 2019, 11, 946. [Google Scholar] [CrossRef]
- Stummer, M.; Frisch, V.; Glitz, F.; Hinney, B.; Spergser, J.; Krücken, J.; Diekmann, I.; Dimmel, K.; Riedel, C.; Cavalleri, J.-M.V.; et al. Presence of Equine and Bovine Coronaviruses, Endoparasites, and Bacteria in Fecal Samples of Horses with Colic. Pathogens 2023, 12, 1043. [Google Scholar] [CrossRef]
- Giannitti, F.; Diab, S.; Mete, A.; Stanton, J.B.; Fielding, L.; Crossley, B.; Sverlow, K.; Fish, S.; Mapes, S.; Scott, L.; et al. Necrotizing Enteritis and Hyperammonemic Encephalopathy Associated With Equine Coronavirus Infection in Equids. Vet. Pathol. 2015, 52, 1148–1156. [Google Scholar] [CrossRef]
- Ghai, R.R.; Carpenter, A.; Liew, A.Y.; Martin, K.B.; Herring, M.K.; Gerber, S.I.; Hall, A.J.; Sleeman, J.M.; VonDobschuetz, S.; Behravesh, C.B. Animal Reservoirs and Hosts for Emerging Alphacoronaviruses and Betacoronaviruses. Emerg. Infect. Dis. 2021, 27, 1015–1022. [Google Scholar] [CrossRef]
- Pusterla, N.; Lawton, K.; Barnum, S. Investigation of the Seroprevalence to Equine Coronavirus and SARS-CoV-2 in Healthy Adult Horses Recently Imported to the United States. Vet. Q. 2024, 44, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Belouzard, S.; Millet, J.K.; Licitra, B.N.; Whittaker, G.R. Mechanisms of Coronavirus Cell Entry Mediated by the Viral Spike Protein. Viruses 2012, 4, 1011–1033. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, E.; Harms, C.; Viner, M.; Barnum, S.; Pusterla, N. Investigation of an Experimental Infection Model of Equine Coronavirus in Adult Horses. J. Vet. Intern. Med. 2018, 32, 2099–2104. [Google Scholar] [CrossRef] [PubMed]
- Guy, J.S.; Breslin, J.J.; Breuhaus, B.; Vivrette, S.; Smith, L.G. Characterization of a Coronavirus Isolated from a Diarrheic Foal. J. Clin. Microbiol. 2000, 38, 4523–4526. [Google Scholar] [CrossRef] [PubMed]
- Slovis, N.; Elam, J.; Estrada, M.; Leutenegger, C. Infectious Agents Associated with Diarrhoea in Neonatal Foals in Central Kentucky: A Comprehensive Molecular Study. Equine Vet. J. 2014, 46, 311–316. [Google Scholar] [CrossRef]
- Davis, E.; Rush, B.R.; Cox, J.; DeBey, B.; Kapil, S. Neonatal Enterocolitis Associated with Coronavirus Infection in a Foal: A Case Report. J. Vet. Diagn. Investig. 2000, 12, 153–156. [Google Scholar] [CrossRef]
- Perlman, S.; Masters, P.S. Coronavirdae: The Viruses and Their Replication. In Fields Virology: Emerging Viruses; Wolters Kluwer Health: Philadelphia, PA, USA, 2020; ISBN 978-1-975112-55-4. [Google Scholar]
- Pusterla, N.; Vin, R.; Leutenegger, C.M.; Mittel, L.D.; Divers, T.J. Enteric Coronavirus Infection in Adult Horses. Vet. J. 2018, 231, 13–18. [Google Scholar] [CrossRef]
- Pusterla, N.; Mapes, S.; Wademan, C.; White, A.; Ball, R.; Sapp, K.; Burns, P.; Ormond, C.; Butterworth, K.; Bartol, J.; et al. Emerging Outbreaks Associated with Equine Coronavirus in Adult Horses. Vet. Microbiol. 2013, 162, 228–231. [Google Scholar] [CrossRef]
- Suzuki, K.; Matsui, Y.; Miura, Y.; Sentsui, H. Equine Coronavirus Induces Apoptosis in Cultured Cells. Vet. Microbiol. 2008, 129, 390–395. [Google Scholar] [CrossRef]
- D’Amico, F.; Baumgart, D.C.; Danese, S.; Peyrin-Biroulet, L. Diarrhea During COVID-19 Infection: Pathogenesis, Epidemiology, Prevention, and Management. Clin. Gastroenterol. Hepatol. 2020, 18, 1663–1672. [Google Scholar] [CrossRef]
- Tsounis, E.P.; Triantos, C.; Konstantakis, C.; Marangos, M.; Assimakopoulos, S.F. Intestinal Barrier Dysfunction as a Key Driver of Severe COVID-19. World J. Virol. 2023, 12, 68–90. [Google Scholar] [CrossRef] [PubMed]
- Peregrine, A.S.; McEwen, B.; Bienzle, D.; Koch, T.G.; Weese, J.S. Larval Cyathostominosis in Horses in Ontario: An Emerging Disease? Can. Vet. J. 2006, 47, 80–82. [Google Scholar] [PubMed]
- Corning, S. Equine Cyathostomins: A Review of Biology, Clinical Significance and Therapy. Parasit. Vectors 2009, 2, S1. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M. Larval Cyathostominosis in Horses—Digestive System. Available online: https://www.merckvetmanual.com/digestive-system/gastrointestinal-parasites-of-horses/larval-cyathostominosis-in-horses (accessed on 4 April 2024).
- Lawson, A.L.; Malalana, F.; Mair, T.S. Larval Cyathostominosis: Clinicopathological Data and Treatment Outcomes of 38 Hospitalised Horses (2009–2020). Equine Vet. Educ. 2023, 35, 424–435. [Google Scholar] [CrossRef]
- Bodecek, S.; Jahn, P.; Dobesova, O.; Vavrouchova, E. Equine Cyathostomosis: Case Reports. Vet. Med. 2010, 55, 187–193. [Google Scholar] [CrossRef]
- Giles, C.J.; Urquhart, K.A.; Longstaffe, J.A. Larval Cyathostomiasis (Immature Trichonema-Induced Enteropathy): A Report of 15 Clinical Cases. Equine Vet. J. 1985, 17, 196–201. [Google Scholar] [CrossRef]
- Love, S.; Murphy, D.; Mellor, D. Pathogenicity of Cyathostome Infection. Vet. Parasitol. 1999, 85, 113–122. [Google Scholar] [CrossRef]
- Bianchi, M.V.; de Mello, L.S.; Wentz, M.F.; Panziera, W.; Soares, J.F.; Sonne, L.; Driemeier, D.; Pavarini, S.P. Fatal Parasite-Induced Enteritis and Typhlocolitis in Horses in Southern Brazil. Rev. Bras. Parasitol. Vet. 2019, 28, 443–450. [Google Scholar] [CrossRef]
- Lightbody, K.L.; Austin, A.; Lambert, P.A.; von Samson-Himmelstjerna, G.; Jürgenschellert, L.; Krücken, J.; Nielsen, M.K.; Sallé, G.; Reigner, F.; Donnelly, C.G.; et al. Validation of a Serum ELISA Test for Cyathostomin Infection in Equines. Int. J. Parasitol. 2024, 54, 23–32. [Google Scholar] [CrossRef]
- Matthews, J.B.; Hodgkinson, J.E.; Dowdall, S.M.J.; Proudman, C.J. Recent Developments in Research into the Cyathostominae and Anoplocephala Perfoliata. Vet. Res. 2004, 35, 371–381. [Google Scholar] [CrossRef]
- Bellaw, J.L.; Nielsen, M.K. Meta-Analysis of Cyathostomin Species-Specific Prevalence and Relative Abundance in Domestic Horses from 1975-2020: Emphasis on Geographical Region and Specimen Collection Method. Parasit. Vectors 2020, 13, 509. [Google Scholar] [CrossRef] [PubMed]
- McWilliam, H.E.G.; Nisbet, A.J.; Dowdall, S.M.J.; Hodgkinson, J.E.; Matthews, J.B. Identification and Characterisation of an Immunodiagnostic Marker for Cyathostomin Developing Stage Larvae. Int. J. Parasitol. 2010, 40, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Bąska, P.; Norbury, L.J. The Role of the Intestinal Epithelium in the “Weep and Sweep” Response during Gastro—Intestinal Helminth Infections. Animals 2022, 12, 175. [Google Scholar] [CrossRef] [PubMed]
- Walshe, N.; Mulcahy, G.; Hodgkinson, J.; Peachey, L. No Worm Is an Island; The Influence of Commensal Gut Microbiota on Cyathostomin Infections. Animals 2020, 10, 2309. [Google Scholar] [CrossRef] [PubMed]
- Walshe, N.; Duggan, V.; Cabrera-Rubio, R.; Crispie, F.; Cotter, P.; Feehan, O.; Mulcahy, G. Removal of Adult Cyathostomins Alters Faecal Microbiota and Promotes an Inflammatory Phenotype in Horses. Int. J. Parasitol. 2019, 49, 489–500. [Google Scholar] [CrossRef]
- Halliez, M.C.M.; Buret, A.G. Gastrointestinal Parasites and the Neural Control of Gut Functions. Front. Cell. Neurosci. 2015, 9, 452. [Google Scholar] [CrossRef]
- Kosik-Bogacka, D.I.; Baranowska-Bosiacka, I.; Salamatin, R. Hymenolepis Diminuta: Effect of Infection on Ion Transport in Colon and Blood Picture of Rats. Exp. Parasitol. 2010, 124, 285–294. [Google Scholar] [CrossRef]
- Moon, C.; Zhang, W.; Sundaram, N.; Yarlagadda, S.; Reddy, V.S.; Arora, K.; Helmrath, M.A.; Naren, A.P. Drug-Induced Secretory Diarrhea: A Role for CFTR. Pharmacol. Res. 2015, 102, 107. [Google Scholar] [CrossRef]
- Uzal, F.A.; Diab, S.S. Gastritis, Enteritis, and Colitis in Horses. Vet. Clin. N. Am. Equine Pract. 2015, 31, 337–358. [Google Scholar] [CrossRef]
- Tian, F.-J.; Li, J.; Tu, Q.-H.; Xu, S.; Liu, W.-L.; Li, Y.; Bai, Y.; Yu, J.; Liu, W.-H.; Xiao, Y.-Q.; et al. Identification of a Novel Astrovirus from Intestinal Tissue of a Donkey Foal with Severe Diarrhea in China. Zool. Res. 2023, 44, 932–935. [Google Scholar] [CrossRef]
- Tong, P.; Wang, Z.; Dang, Y.; Zhang, L.; Song, G.; Song, X.; Pan, J.; Kuang, L.; Li, J.; Lu, G.; et al. Donkey-like Kirkovirus Is Associated with Diarrhea in Piglets. Virus Genes 2024, 60, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Haywood, L.; Hause, B.M.; Clark, A.; Sheahan, B. A Novel Kirkovirus May Be Associated with Equine Colitis. Equine Vet. Educ. 2024, 36, 47–48. [Google Scholar] [CrossRef]
- Uzal, F.A.; Navarro, M.A.; Asin, J.; Henderson, E.E. Clostridial Diseases of Horses: A Review. Vaccines 2022, 10, 318. [Google Scholar] [CrossRef] [PubMed]
- Kinsella, C.M.; Deijs, M.; Becker, C.; Broekhuizen, P.; van Gool, T.; Bart, A.; Schaefer, A.S.; van der Hoek, L. Host Prediction for Disease-Associated Gastrointestinal Cressdnaviruses. Virus Evol. 2022, 8, veac087. [Google Scholar] [CrossRef]
- Headley, S.A.; Kummala, E.; Sukura, A. Balantidium Coli-Infection in a Finnish Horse. Vet. Parasitol. 2008, 158, 129–132. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haywood, L.M.B.; Sheahan, B.J. A Review of Epithelial Ion Transporters and Their Roles in Equine Infectious Colitis. Vet. Sci. 2024, 11, 480. https://doi.org/10.3390/vetsci11100480
Haywood LMB, Sheahan BJ. A Review of Epithelial Ion Transporters and Their Roles in Equine Infectious Colitis. Veterinary Sciences. 2024; 11(10):480. https://doi.org/10.3390/vetsci11100480
Chicago/Turabian StyleHaywood, Lillian M. B., and Breanna J. Sheahan. 2024. "A Review of Epithelial Ion Transporters and Their Roles in Equine Infectious Colitis" Veterinary Sciences 11, no. 10: 480. https://doi.org/10.3390/vetsci11100480
APA StyleHaywood, L. M. B., & Sheahan, B. J. (2024). A Review of Epithelial Ion Transporters and Their Roles in Equine Infectious Colitis. Veterinary Sciences, 11(10), 480. https://doi.org/10.3390/vetsci11100480






