Comparison of Fish, Krill and Flaxseed as Omega-3 Sources to Increase the Omega-3 Index in Dogs
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Animals and Diet
2.3. Sample Collection
2.4. Food Analysis
2.5. Blood Analysis
2.6. Statistical Analysis
3. Results and Discussion
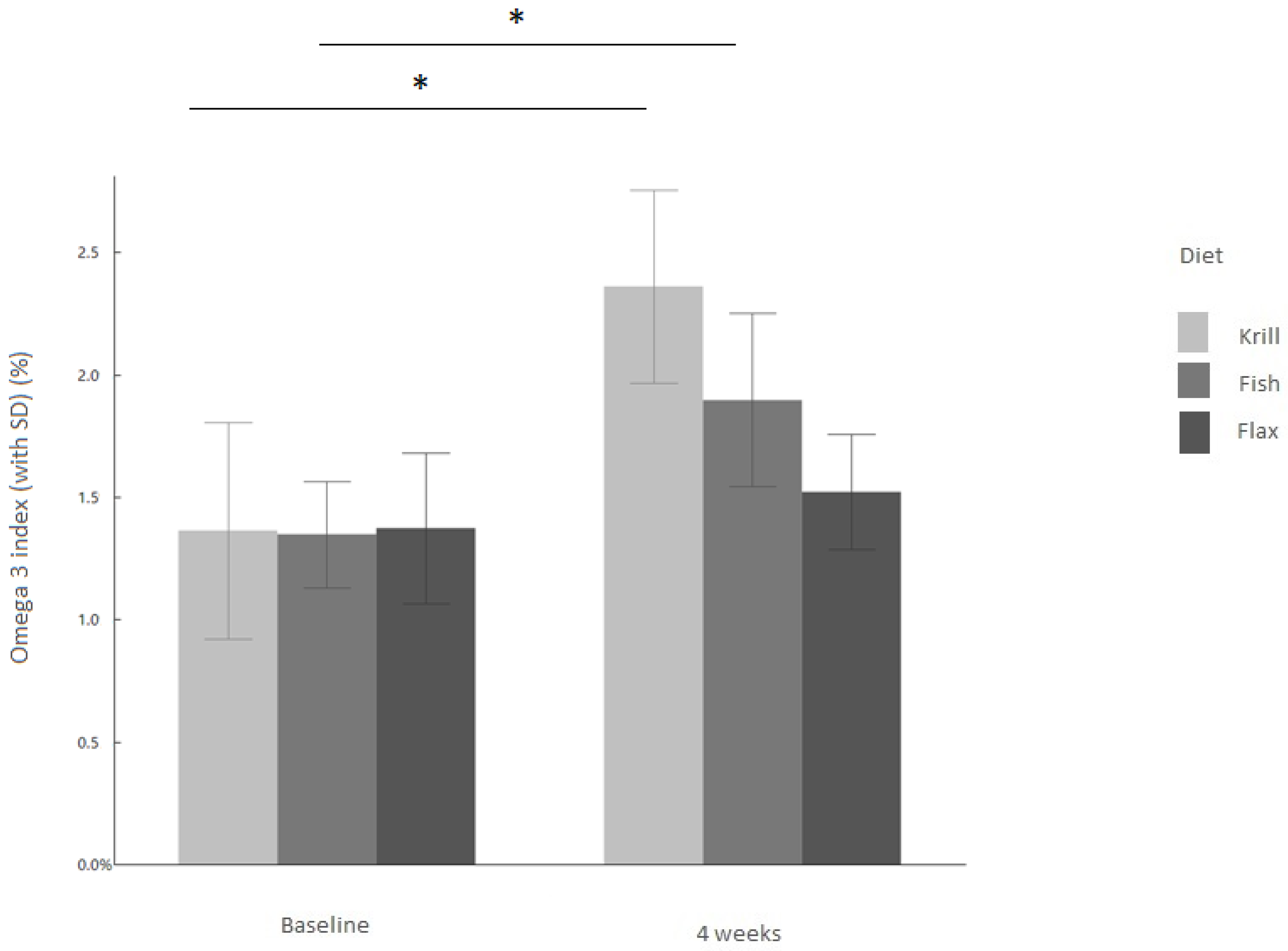
3.1. Omega-3 Index
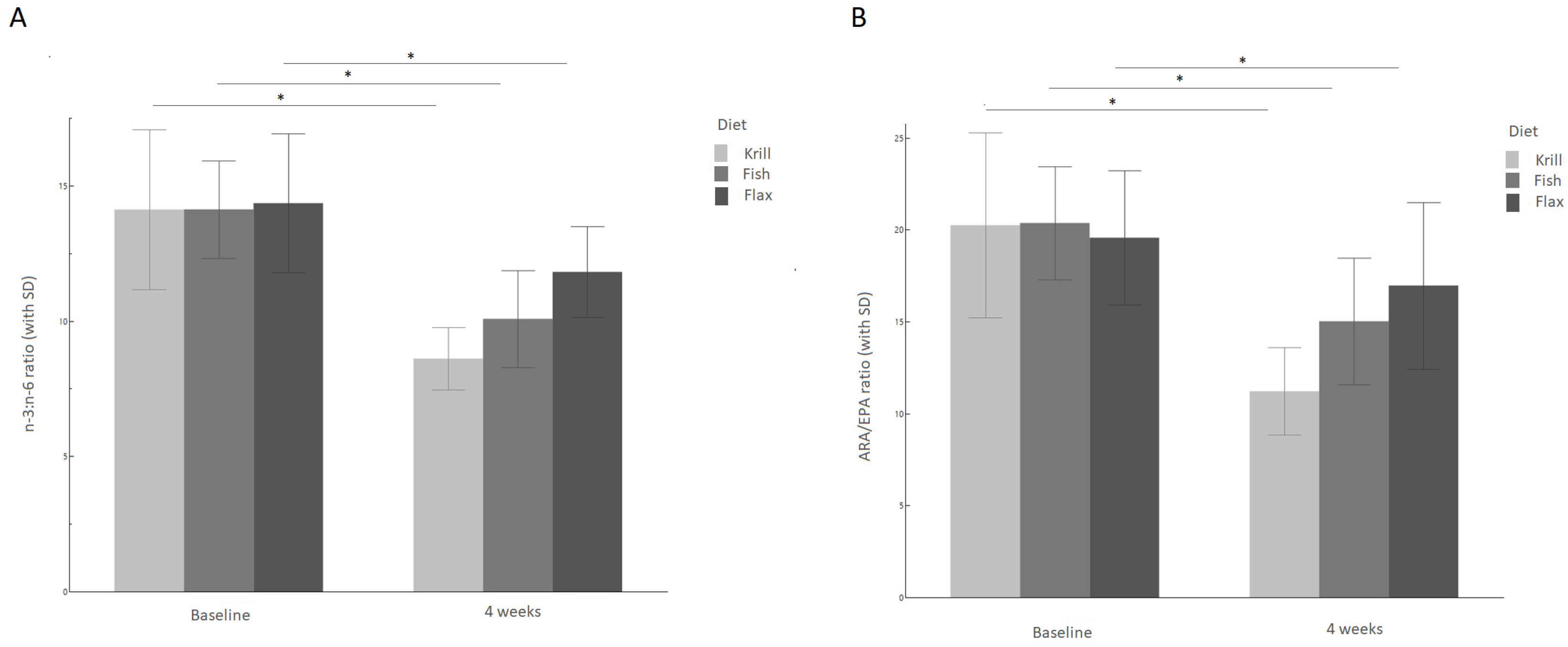
3.2. n-6/n-3 and Arachidonic Acid/EPA Ratios
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abba, C.; Mussa, P.P.; Vercelli, A.; Raviri, G. Essential fatty acids supplementation in different-stage atopic dogs fed on a controlled diet. J. Anim. Physiol. Anim. Nutr. 2005, 89, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.E.; Heinemann, K.M.; Lees, G.E.; Waldron, M.K. Retinal functions of young dogs are improved and maternal plasma phospholipids are altered with diets containing long-chain n-3 polyunsaturated fatty acids during gestation, lactation, and after weaning. J. Nutr. 2006, 136, 1991S–1994S. [Google Scholar] [CrossRef]
- Fritsch, D.; Allen, T.; Dodd, C.; Jewell, D.; Sixby, K.; Leventhal, P.; Hahn, K. Dose-titration effects of fish oil in osteoarthritic dogs. J. Vet. Intern. Med. 2010, 24, 1020–1026. [Google Scholar] [CrossRef]
- Mueller, R.; Fieseler, K.; Fettman, M.; Zabel, S.; Rosychuk, R.; Ogilvie, G.; Greenwalt, T. Effect of omega-3 fatty acids on canine atopic dermatitis. J. Small Anim. Pract. 2004, 45, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.E.; Freeman, L.M.; Rush, J.E.; Cunningham, S.M.; Biourge, V. Omega-3 fatty acids in Boxer dogs with arrhythmogenic right ventricular cardiomyopathy. J. Vet. Intern. Med. 2007, 21, 265–273. [Google Scholar] [CrossRef]
- Trepanier, L. Idiopathic inflammatory bowel disease in cats: Rational treatment selection. J. Feline Med. Surg. 2009, 11, 32–38. [Google Scholar] [CrossRef]
- Ahlstrøm, Ø.; Krogdahl, A.; Vhile, S.G.; Skrede, A. Fatty acid composition in commercial dog foods. J. Nutr. 2004, 134, 2145S–2147S. [Google Scholar] [CrossRef]
- Bauer, J.E. Therapeutic use of fish oils in companion animals. J. Am. Vet. Med. Assoc. 2011, 239, 1441–1451. [Google Scholar] [CrossRef] [PubMed]
- Lenox, C.; Bauer, J. Potential adverse effects of omega-3 fatty acids in dogs and cats. J. Vet. Intern. Med. 2013, 27, 217–226. [Google Scholar] [CrossRef]
- Council, N.R. Nutrient Requirements of Dogs and Cats; National Academies Press: Washington, DC, USA, 2006. [Google Scholar]
- Officials, A.o.A.F.C. Model Bill and Regulations; Association of American Feed Control Officials Official Publication: Atlanta, GA, USA, 2019; pp. 107–232. [Google Scholar]
- Turchini, G.M.; Nichols, P.D.; Barrow, C.; Sinclair, A.J. Jumping on the omega-3 bandwagon: Distinguishing the role of long-chain and short-chain omega-3 fatty acids. Crit. Rev. Food Sci. Nutr. 2012, 52, 795–803. [Google Scholar] [CrossRef]
- Bauer, J.E.; Heinemann, K.M.; Bigley, K.E.; Lees, G.E.; Waldron, M.K. Maternal diet α-linolenic acid during gestation and lactation does not increase docosahexaenoic acid in canine milk. J. Nutr. 2004, 134, 2035S–2038S. [Google Scholar] [CrossRef]
- Chen, H.; Deng, G.; Zhou, Q.; Chu, X.; Su, M.; Wei, Y.; Li, L.; Zhang, Z. Effects of eicosapentaenoic acid and docosahexaenoic acid versus α-linolenic acid supplementation on cardiometabolic risk factors: A meta-analysis of randomized controlled trials. Food & function 2020, 11, 1919–1932. [Google Scholar]
- Calder, P.C. Omega-3 polyunsaturated fatty acids and inflammatory processes: Nutrition or pharmacology? Br. J. Clin. Pharmacol. 2013, 75, 645–662. [Google Scholar] [CrossRef]
- Harris, W.S.; Jackson, K.H.; Carlson, H.; Hoem, N.; Dominguez, T.E.; Burri, L. Derivation of the Omega-3 Index from EPA and DHA Analysis of Dried Blood Spots from Dogs and Cats. Vet. Sci. 2022, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- von Schacky, C. The Omega-3 Index as a risk factor for cardiovascular diseases. Prostaglandins Other Lipid Mediat. 2011, 96, 94–98. [Google Scholar] [CrossRef]
- Harris, W.S.; Von Schacky, C. The Omega-3 Index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Fenton, J.I.; Gurzell, E.A.; Davidson, E.A.; Harris, W.S. Red blood cell PUFAs reflect the phospholipid PUFA composition of major organs. Prostaglandins Leukot. Essent. Fat. Acids 2016, 112, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, R.G.; Cleland, L.G.; Gibson, R.A.; Roberts-Thomson, K.C.; Edwards, J.R.; Sanders, P.; Stuklis, R.; James, M.J.; Young, G.D. Relation between blood and atrial fatty acids in patients undergoing cardiac bypass surgery. Am. J. Clin. Nutr. 2010, 91, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Fediaf. Nutritional Guidelines for Complete and Complementary Pet Food for Cats and Dogs; European Pet Food Industry Federation Brussels: Brussels, Belgium, 2020; pp. 1–96. [Google Scholar]
- Dessen, J.-E.; Weihe, R.; Hatlen, B.; Thomassen, M.S.; Rørvik, K.-A. Different growth performance, lipid deposition, and nutrient utilization in in-season (S1) Atlantic salmon post-smolt fed isoenergetic diets differing in protein-to-lipid ratio. Aquaculture 2017, 473, 345–354. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Firestone, D. AOCS Official Method Ce-1b-89, Fatty Acid Composition by GLC: Marine Oils, Urbana, IL. 1992. Available online: https://myaccount.aocs.org/PersonifyEbusiness/Store/Product-Details?productId=111788 (accessed on 10 November 2022).
- Kjeldahl, J. Neue methode zur bestimmung des stickstoffs in organischen körpern. Z. Für Anal. Chem. 1883, 22, 366–382. [Google Scholar] [CrossRef]
- Harris, W.S.; Polreis, J. Measurement of the omega-3 index in dried blood spots. Ann. Clin. Lab. Res. 2016, 4. [Google Scholar] [CrossRef]
- Team, R.C. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. Available online: http://www.R-project.org (accessed on 10 November 2022).
- Dominguez, T.E.; Kaur, K.; Burri, L. Enhanced omega-3 index after long-versus short-chain omega-3 fatty acid supplementation in dogs. Vet. Med. Sci. 2021, 7, 370–377. [Google Scholar] [CrossRef]
- Burri, L.; Heggen, K.; Storsve, A.B. Higher omega-3 index after dietary inclusion of omega-3 phospholipids versus omega-3 triglycerides in Alaskan Huskies. Vet. World 2020, 13, 1167. [Google Scholar] [CrossRef] [PubMed]
- Burri, L.; Wyse, C.; Gray, S.R.; Harris, W.S.; Lazzerini, K. Effects of dietary supplementation with krill meal on serum pro-inflammatory markers after the Iditarod sled dog race. Res. Vet. Sci. 2018, 121, 18–22. [Google Scholar] [CrossRef]
- Burri, L.; Hoem, N.; Monakhova, Y.B.; Diehl, B.W. Fingerprinting krill oil by 31P, 1H and 13C NMR spectroscopies. J. Am. Oil Chem. Soc. 2016, 93, 1037–1049. [Google Scholar] [CrossRef]
- Liu, L.; Bartke, N.; Van Daele, H.; Lawrence, P.; Qin, X.; Park, H.G.; Kothapalli, K.; Windust, A.; Bindels, J.; Wang, Z. Higher efficacy of dietary DHA provided as a phospholipid than as a triglyceride for brain DHA accretion in neonatal piglets. J. Lipid Res. 2014, 55, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Wijendran, V.; Huang, M.-C.; Diau, G.-Y.; Boehm, G.; Nathanielsz, P.W.; Brenna, J.T. Efficacy of dietary arachidonic acid provided as triglyceride or phospholipid as substrates for brain arachidonic acid accretion in baboon neonates. Pediatr. Res. 2002, 51, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Rossmeisl, M.; Macek Jilkova, Z.; Kuda, O.; Jelenik, T.; Medrikova, D.; Stankova, B.; Kristinsson, B.; Haraldsson, G.G.; Svensen, H.; Stoknes, I. Metabolic effects of n-3 PUFA as phospholipids are superior to triglycerides in mice fed a high-fat diet: Possible role of endocannabinoids. PLoS ONE 2012, 7, e38834. [Google Scholar] [CrossRef]
- Graf, B.; Duchateau, G.; Patterson, A.; Mitchell, E.; Van Bruggen, P.; Koek, J.; Melville, S.; Verkade, H. Age dependent incorporation of 14C-DHA into rat brain and body tissues after dosing various 14C-DHA-esters. Prostaglandins Leukot. Essent. Fat. Acids 2010, 83, 89–96. [Google Scholar] [CrossRef]
- Ramprasath, V.R.; Eyal, I.; Zchut, S.; Jones, P.J. Enhanced increase of omega-3 index in healthy individuals with response to 4-week n-3 fatty acid supplementation from krill oil versus fish oil. Lipids Health Dis. 2013, 12, 178. [Google Scholar] [CrossRef] [PubMed]
- Mueller, R.S.; Fettman, M.J.; Richardson, K.; Hansen, R.A.; Miller, A.; Magowitz, J.; Ogilvie, G.K. Plasma and skin concentrations of polyunsaturated fatty acids before and after supplementation with n-3 fatty acids in dogs with atopic dermatitis. Am. J. Vet. Res. 2005, 66, 868–873. [Google Scholar] [CrossRef]
- Simopoulos, A.P. Omega-6/omega-3 essential fatty acid ratio and chronic diseases. Food Rev. Int. 2004, 20, 77–90. [Google Scholar] [CrossRef]
- Magalhães, R.; Guardiola, F.A.; Guerreiro, I.; Fontinha, F.; Moutinho, S.; Serra, C.R.; Olsen, R.E.; Peres, H.; Oliva-Teles, A. Immunomodulatory effect of different dietary ARA/EPA/DHA ratios in gilthead sea bream (Sparus aurata) juveniles after infection with Photobacterium damselae subsp. piscicida. Aquac. Res. 2022, 53, 6007–6019. [Google Scholar] [CrossRef]
- Bauer, J.E. Responses of dogs to dietary omega-3 fatty acids. J. Am. Vet. Med. Assoc. 2007, 231, 1657–1661. [Google Scholar] [CrossRef]
| Krill | Fish | Flax | |
|---|---|---|---|
| Gender | |||
| Males | 8 | 8 | 8 |
| Females | 7 | 7 | 7 |
| Age (years) | 4 (2–8) | 4 (2–8) | 4 (2–8) |
| Weight (kg) | 28.5 (22–36) | 26.9 (22–33) | 27.5 (22.2–34.4) |
| Ingredient | Krill | Fish | Flax |
|---|---|---|---|
| Beef (meat including lung, tripe and fat) | 40.0 | 42.6 | 40.9 |
| Chicken necks | 12.7 | 8.1 | 15.0 |
| Swine trimmings | 4.0 | 3.0 | 4.0 |
| Maize gluten | 3.4 | 3.6 | 3.5 |
| Oatmeal | 6.7 | 6.8 | 5.6 |
| Wheat and barley mix | 5.5 | 5.8 | 6.0 |
| Rice | 2.0 | 2.0 | 2.0 |
| Wheat bran | 1.0 | 1.0 | 1.0 |
| Water from boiling of rice and porridge | 22.0 | 24.0 | 19.7 |
| Monocalcium phosphate | 0.7 | 0.6 | 0.7 |
| Calcium carbonate | 0.2 | 0.2 | 0.3 |
| Potassium chloride | 0.2 | 0.2 | 0.2 |
| Natrium chloride | 0.2 | 0.2 | 0.2 |
| Vitamin premix | 0.2 | 0.2 | 0.2 |
| Choline Chloride | 0.1 | 0.1 | 0.1 |
| Krill meal | 1.2 | ||
| Fish oil | 0.2 | ||
| Fish meal | 1.5 | ||
| Flaxseed cake | 0.5 |
| Analysed Nutrient Composition | Krill | Fish | Flax |
|---|---|---|---|
| Dry matter (%) | 37.70 | 37.10 | 34.30 |
| Crude protein (%) | 13.70 | 13.90 | 13.20 |
| Crude fat (%) | 13.90 | 13.20 | 12.50 |
| Fatty acids (%) | |||
| C 14:0 | 0.39 | 0.35 | 0.27 |
| C 16:0 | 3.27 | 3.08 | 2.86 |
| C 16:1 | 0.35 | 0.32 | 0.24 |
| C 18:0 | 2.11 | 2.08 | 2.11 |
| C 18:1 | 4.83 | 4.70 | 4.22 |
| C18:2 | 0.71 | 0.76 | 0.78 |
| C 18:3 (ALA) | 0.10 | 0.10 | 0.15 |
| C 18:4 | 0.01 | - | - |
| C 20:5 (EPA) | 0.03 | 0.03 | 0.01 |
| C 22:5 | 0.03 | 0.03 | 0.02 |
| C 22:6 (DHA) | 0.01 | 0.03 | |
| EPA + DHA (g/kg) | 0.42 | 0.51 | 0.12 |
| n-3 (g/kg) | 1.80 | 1.80 | 1.83 |
| n-6 (g/kg) | 7.77 | 8.09 | 8.30 |
| n-6/n-3 | 4.05 | 4.39 | 4.39 |
| Crude fiber (%) | 0.70 | 0.30 | 0.60 |
| Crude ash (%) | 2.30 | 2.30 | 2.30 |
| NFE (%) | 7.10 | 7.40 | 5.70 |
| Kcal ME */kg | 2081 | 2039 | 1881 |
| Analysed Nutrient Composition | Krill | Fish | Flax |
|---|---|---|---|
| Amount (g) | 1000.00 | 1000.00 | 1000.00 |
| Water (g) | 623.00 | 629.00 | 657.00 |
| Crude protein (g) | 137.20 | 139.27 | 132.14 |
| Crude fat (g) | 138.77 | 131.66 | 125.11 |
| Fatty acids (g) | |||
| C 14:0 | 3.88 | 3.46 | 2.68 |
| C 16:0 | 32.70 | 30.80 | 28.60 |
| C 16:1 | 3.46 | 3.20 | 2.44 |
| C 18:0 | 21.09 | 20.80 | 21.13 |
| C 18:1 | 48.29 | 46.98 | 42.25 |
| C18:2 | 7.07 | 7.57 | 7.81 |
| C 18:3 (ALA) | 0.97 | 1.02 | 1.46 |
| C 18:4 | 0.13 | - | - |
| C 20:5 (EPA) | 0.28 | 0.26 | 0.12 |
| C 22:5 | 0.28 | 0.26 | 0.24 |
| C 22:6 (DHA) | 0.14 | 0.26 | - |
| EPA + DHA | 0.42 | 0.51 | 0.12 |
| n-3 | 1.80 | 1.80 | 1.83 |
| n-6 | 7.77 | 8.10 | 8.30 |
| n-6/n-3 | 4.05 | 4.39 | 4.39 |
| Crude fiber (g) | 7.47 | 3.04 | 6.03 |
| Crude ash (g) | 22.80 | 22.83 | 22.84 |
| NFE (g) | 70.76 | 74.20 | 56.58 |
| Calcium (g) | |||
| Phosphorous (g) | |||
| Kcal ME | 2081 | 2039 | 1881 |
| Rec’d Daily Intake | Krill | Fish | Flax | Commercial Premium | |
|---|---|---|---|---|---|
| Daily intake | |||||
| ALA (mg) | 971 | 1027 | 1465 | 1891 | |
| EPA + DHA (mg) | 416 | 513 | 122 | 0 | |
| Daily intake/kg BW0.75 | |||||
| ALA (mg/kg BW0.75) | 14 | 79 | 87 | 122 | 157 |
| EPA + DHA (mg/kg BW0.75) | 30 | 34 | 44 | 10 | 0 |
| Daily intake/1000 kcal | |||||
| ALA/ 1000 kcal ME (mg) | 110 | 467 | 504 | 779 | 1085 |
| DHA + EPA/ 1000 kcal ME (mg) | 110 | 200 | 252 | 65 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lindqvist, H.; Dominguez, T.; Dragøy, R.; Ding, Y.; Burri, L. Comparison of Fish, Krill and Flaxseed as Omega-3 Sources to Increase the Omega-3 Index in Dogs. Vet. Sci. 2023, 10, 162. https://doi.org/10.3390/vetsci10020162
Lindqvist H, Dominguez T, Dragøy R, Ding Y, Burri L. Comparison of Fish, Krill and Flaxseed as Omega-3 Sources to Increase the Omega-3 Index in Dogs. Veterinary Sciences. 2023; 10(2):162. https://doi.org/10.3390/vetsci10020162
Chicago/Turabian StyleLindqvist, Hanna, Tonje Dominguez, Ragnhild Dragøy, Yunpeng Ding, and Lena Burri. 2023. "Comparison of Fish, Krill and Flaxseed as Omega-3 Sources to Increase the Omega-3 Index in Dogs" Veterinary Sciences 10, no. 2: 162. https://doi.org/10.3390/vetsci10020162
APA StyleLindqvist, H., Dominguez, T., Dragøy, R., Ding, Y., & Burri, L. (2023). Comparison of Fish, Krill and Flaxseed as Omega-3 Sources to Increase the Omega-3 Index in Dogs. Veterinary Sciences, 10(2), 162. https://doi.org/10.3390/vetsci10020162





