Non-Human Primate Models of Orthopoxvirus Infections
Abstract
:1. Introduction
2. Monkeypox
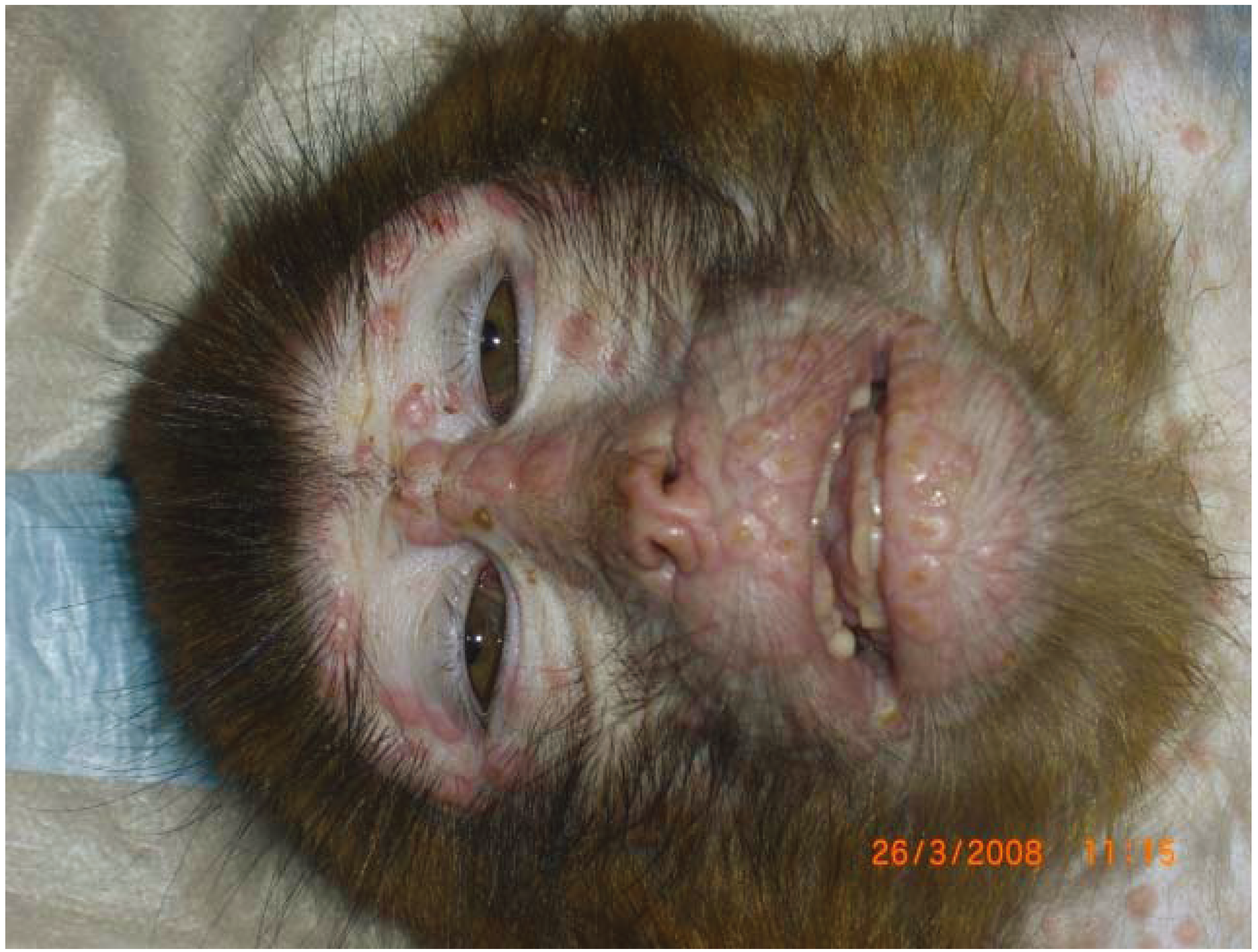
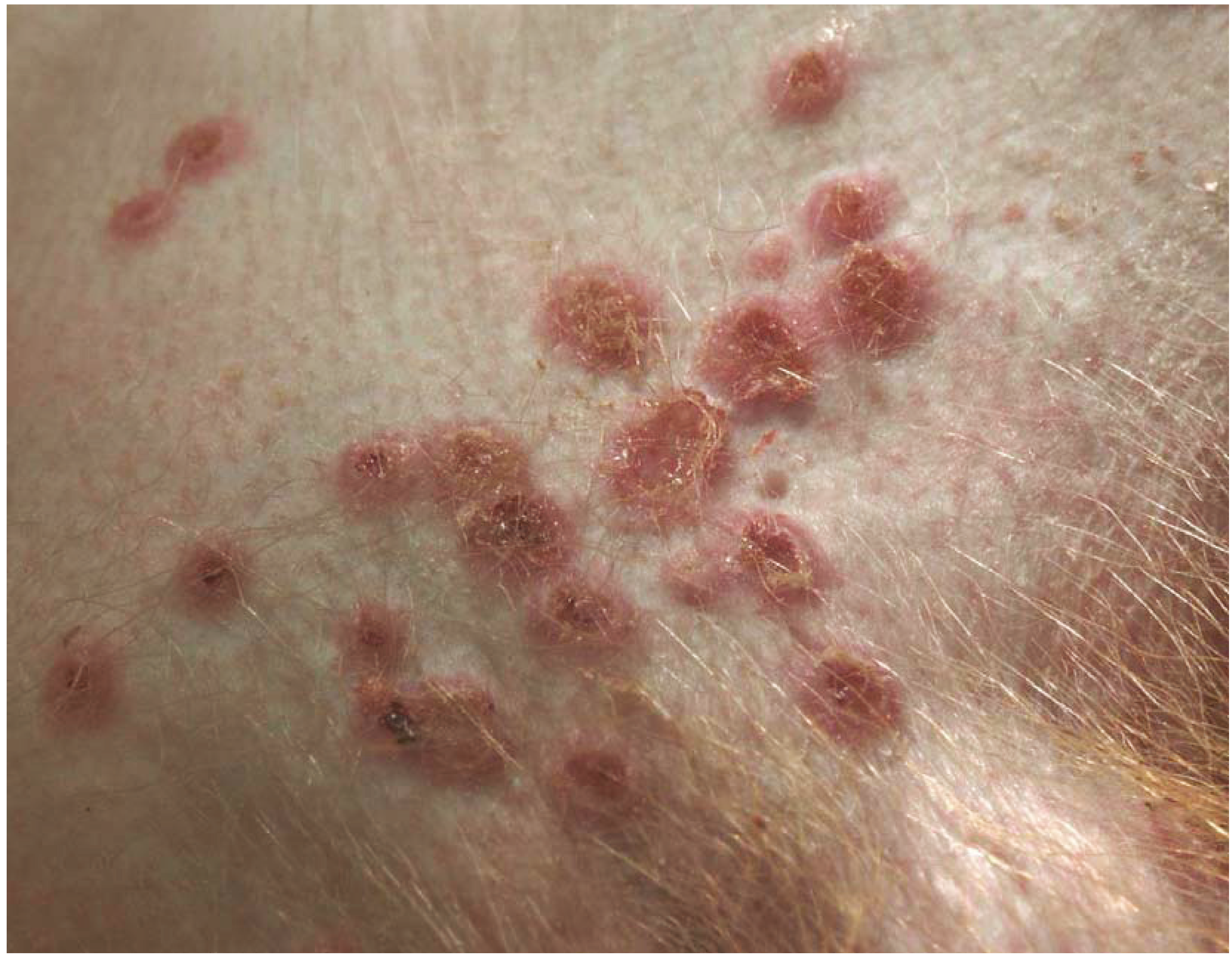
2.1. Intramuscular
2.2. Skin Scarification
2.3. Intravenous
2.4. Intratracheal/Aerosolization
2.5. Microsprayer-Technique and Intrabronchial
2.6. Intranasal
2.7. Subcutaneous
Summary Monkeypox
3. Smallpox
Summary Smallpox
4. Cowpox
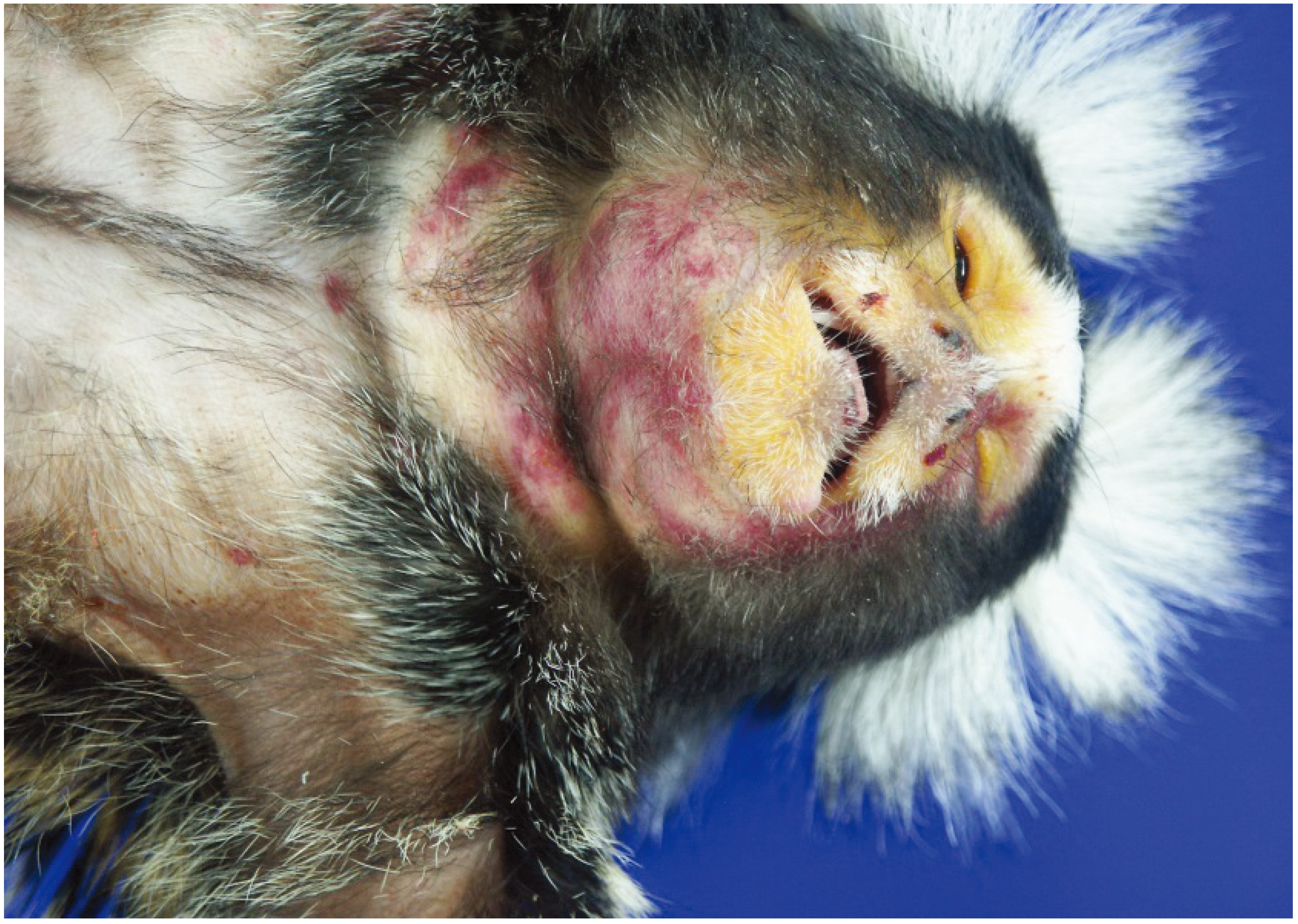
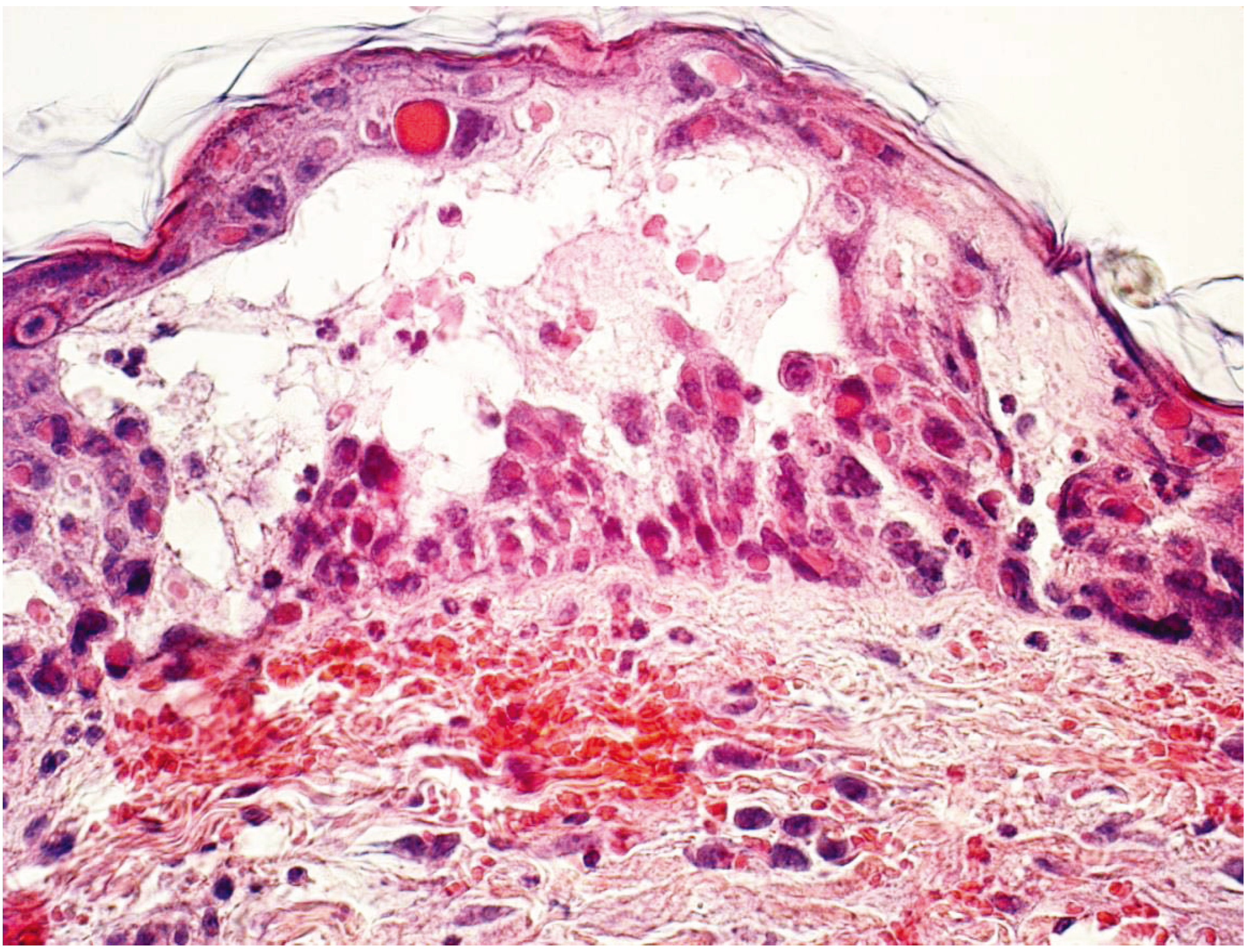
Summary Cowpox
5. Conclusions
| Species | Route of Infection | Virus | Dose | Purpose of Study | Reference |
|---|---|---|---|---|---|
| M.f. and M.m. | i.m. | MPXV | 105 PFU | pathogenesis | [33] |
| P.c. | i.m. | MPXV Copenhagen | 106.5–107.5 TCID50 | pathogenesis | [34] |
| P.c. | via skin scarification | MPXV | 107 TCID50 | pathogenesis | [35] |
| M.f. | i.v. | MPXV Zaire 79 | 5 × 107 PFU | Tecovirimat efficacy study | [20] |
| M.f. | i.v. | MPXV Zaire 79 | 5 × 107 PFU | Tecovirimat efficacy study | [37] |
| M.f. | i.v. | MPXV-GFP Zaire 79 | 5 × 107 PFU | pathogenesis | [38] |
| M.m. | i.v. | MPXV Zaire 79 | 5 × 108 PFU | DNA vaccine study | [39] |
| M.m. | i.v. | MPXV Zaire 79 | 2 × 107 PFU | MVA/gene based vaccine study | [40] |
| M.m. | i.v. | MPXV Zaire 79 | 1.5–2.5 × 106 PFU | pathogenesis | [41] |
| M.f. | i.v. and i.b. | MPXV Zaire 79 | 5 × 106–5 × 107 PFU | pathogenesis | [42] |
| M.f. | i.v. | MPXV Zaire 79 | 5 × 107 PFU | Dryvax/MVA vaccine study | [43] |
| M.m. | i.v. | MPXV Zaire 79 | 5 × 107 PFU | Vaccine study in SIV-infected macaques | [44] |
| M.m. | i.v. | MPXV Zaire 79 | 5 × 107 PFU | MVA/NYVAC/Dry-vax vaccine study | [45] |
| M.m. | i.v. | MPXV Zaire 79 | 5 × 107 PFU | Subunit recombinant vaccine study | [46] |
| M.f. | i.v. | MPXV Zaire 79 | 5 × 107 PFU | Il-15/Wyeth vaccine study | [47] |
| M.f. | i.t. | MPXV MSF #6 | 106–107 PFU in 5 mL | MVA vaccine study | [49] |
| M.f. | i.t. | MPXV MSF #6 | 107 PFU in 5 mL | Comparison post-exposure vaccination with antiviral therapeutics | [50] |
| M.f. | via head-only exposure chamber | MPXV Zaire 79 | 104–105 PFU | pathogenesis | [51] |
| M.f. | via head-only exposure chamber | MPXV Zaire 79 | 4.3 × 104–1.1 × 106 PFU | pathogenesis | [21] |
| M.f. | via head-only exposure system | MPXV Zaire 79 | 2.5 × 104–9.3 × 105 PFU | pathogenesis | [52] |
| M.f. | via Henderson- apparatus and modified anesthesia mask | MPXV Zaire 79 | 2.1 × 105–3.1 × 105 PFU | Imvamune, Acam2000 vaccine study | [53] |
| M.f. | bronchoscope and liquid MicroSprayer aerosolizer | MPXV Zaire | 3.42 × 106–3.53 × 107 PFU | pathogenesis | [36] |
| M.m. | i.b. | MPXV Zaire and D14L KO MPXV | 2 × 105 PFU | pathogenesis | [54] |
| M.f. | i.v. and i.b. | MPXV Zaire 79 | i.v.: 5 × 107–5 × 104 PFU i.b.: 5 × 106–5 × 104 PFU | pathogenesis | [55] |
| M.m. | i.b. | MPXV Zaire 79 | 2 × 105 PFU | pathogenesis | [56] |
| M.f. | i.n. and subcutaneous | MPXV Liberia and Zaire-559 | 1 × 106 PFU | LC16m8 vaccine study | [57] |
| M.f. | i.n. and subcutaneous | MPXV Liberia and Zaire-559 | 1 × 106 PFU | pathogenesis | [58] |
| M.m. | via Henderson apparatus | VARV Higgins | ? | pathogenesis | [64,66] |
| M.f. | aerosol | VARV | >108 PFU | pathogenesis | [67] |
| M.f. | aerosol and/or i.v. | VARV Harper and India 7124 | 106–109 PFU | pathogenesis | [68] |
| M.f. | aerosol and/or i.v. | VARV Harper and India 7124 | Aerosol: 5 × 108 PFU
i.v.: 109 PFU | pathogenesis | [62] |
| M.f. | i.v. | VARV Harper | 108–109 PFU | pathogenesis | [69] |
| M.f. | i.v. | VARV Harper | 1 × 108 PFU | ST-246 efficacy study | [20] |
| M.f. | i.v. | VARV Harper | 1 × 108 PFU | ST-246 efficyacy study | [70] |
| C.j. | i.v. and i.n. | calpox | i.v.: 1.25 × 107–1 × 104 PFU
i.n.: 5 × 102–3.5 × 105 PFU | pathogenesis | [93,94] |
| M.f. | i.b. | CPXV | 5 × 107–5 × 104 PFU | pathogenesis | [98] |
| M.f. | i.v. | CPXV Brighton Red | 5 × 107–5 × 105 PFU | pathogenesis | [99] |
| M.f. | i.v. | CPXV Brighton | 5 × 102–5 × 104 PFU | pathogenesis | [100] |
Acknowledgments
Conflicts of Interest
References
- Fenner, F.; Henderson, D.A.; Arita, I.; Jezek, Z.; Ladnyi, I.D. Variola virus and other orthopoxviruses. In Smallpox and Its Eradication; WHO: Geneva, Switzerland, 1988. [Google Scholar]
- Shchelkunov, S.N. An increasing danger of zoonotic orthopoxvirus infections. PLoS Pathog. 2013, 9. [Google Scholar] [CrossRef]
- Nitsche, A. Untersuchungen zur Diagnostik und Risikobewertung von Emerging und Re-Emerging Orthopockenviren in Deutschland. Habilitation Thesis, Robert Koch-Institute, Berlin, Germany, 2010. [Google Scholar]
- Cardeti, G.; Brozzi, A.; Eleni, C.; Polici, N.; D’Alterio, G.; Carletti, F.; Scicluna, M.T.; Castilletti, C.; Capobianchi, M.R.; di Caro, A.; et al. Cowpox virus in llama, italy. Emerg. Infect. Dis. 2011, 17, 1513–1515. [Google Scholar]
- Bonnekoh, B.; Falk, K.; Reckling, K.F.; Kenklies, S.; Nitsche, A.; Ghebremedhin, B.; Pokrywka, A.; Franke, I.; Thriene, B.; König, W.; et al. Cowpox infection transmitted from a domestic cat. J. Dtsch. Dermatol. Ges. 2008, 6, 210–213. [Google Scholar]
- Baxby, D. Cowpox: Increased incidence or interest? Lancet 1994, 343, 543. [Google Scholar] [CrossRef]
- Dabrowski, P.W.; Radonic, A.; Kurth, A.; Nitsche, A. Genome-wide comparison of cowpox viruses reveals a new clade related to variola virus. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Rimoin, A.W.; Mulembakani, P.M.; Johnston, S.C.; Lloyd Smith, J.O.; Kisalu, N.K.; Kinkela, T.L.; Blumberg, S.; Thomassen, H.A.; Pike, B.L.; Fair, J.N.; et al. Major increase in human monkeypox incidence 30 years after smallpox vaccination campaigns cease in the democratic republic of congo. Proc. Natl. Acad. Sci. USA 2010, 107, 16262–16267. [Google Scholar] [CrossRef]
- Breman, J.G.; Henderson, D.A. Poxvirus dilemmas-monkeypox, smallpox, and biologic terrorism. N. Engl. J. Med. 1998, 339, 556–559. [Google Scholar] [CrossRef]
- Jahrling, P.B.; Fritz, E.A.; Hensley, L.E. Countermeasures to the bioterrorist threat of smallpox. Curr. Mol. Med. 2005, 5, 817–826. [Google Scholar] [CrossRef]
- Anderson, P.D.; Bokor, G. Bioterrorism: Pathogens as weapons. J. Pharm. Pract. 2012, 25, 521–529. [Google Scholar] [CrossRef]
- Hutson, C.L.; Damon, I.K. Monkeypox virus infections in small animal models for evaluation of anti-poxvirus agents. Viruses 2010, 2, 2763–2776. [Google Scholar] [CrossRef]
- Safronetz, D.; Geisbert, T.W.; Feldmann, H. Animal models for highly pathogenic emerging viruses. Curr. Opin. Virol. 2013, 3, 205–209. [Google Scholar] [CrossRef]
- Jordan, R.; Hruby, D. Smallpox antiviral drug development: Satisfying the animal efficacy rule. Expert Rev. Anti-Infect. Ther. 2006, 4, 277–289. [Google Scholar] [CrossRef]
- Parker, S.; Buller, R.M. A review of experimental and natural infections of animals with monkeypox virus between 1958 and 2012. Future Virol. 2013, 8, 129–157. [Google Scholar]
- Chapman, J.L.; Nichols, D.K.; Martinez, M.J.; Raymond, J.W. Animal models of orthopoxvirus infection. Vet. Pathol. 2010, 47, 852–870. [Google Scholar] [CrossRef]
- Smee, D.F.; Sidwell, R.W. A review of compounds exhibiting anti-orthopoxvirus activity in animal models. Antivir. Res. 2003, 57, 41–52. [Google Scholar] [CrossRef]
- Smee, D.F. Progress in the discovery of compounds inhibiting orthopoxviruses in animal models. Antivir. Chem. Chemother. 2008, 19, 115–124. [Google Scholar]
- Smee, D.F. Orthopoxvirus inhibitors that are active in animal models: An update from 2008 to 2012. Future Virol. 2013, 8, 891–901. [Google Scholar] [CrossRef]
- Huggins, J.; Goff, A.; Hensley, L.; Mucker, E.; Shamblin, J.; Wlazlowski, C.; Johnson, W.; Chapman, J.; Larsen, T.; Twenhafel, N.; et al. Nonhuman primates are protected from smallpox virus or monkeypox virus challenges by the antiviral drug st-246. Antimicrob. Agents Chemother. 2009, 53, 2620–2625. [Google Scholar] [CrossRef]
- Nalca, A.; Livingston, V.A.; Garza, N.L.; Zumbrun, E.E.; Frick, O.M.; Chapman, J.L.; Hartings, J.M. Experimental infection of cynomolgus macaques (Macaca fascicularis) with aerosolized monkeypox virus. PLoS ONE 2010, 5. [Google Scholar] [CrossRef]
- Jezek, Z.; Szczeniowski, M.; Paluku, K.M.; Mutombo, M. Human monkeypox: Clinical features of 282 patients. J. Infect. Dis. 1987, 156, 293–298. [Google Scholar] [CrossRef]
- Reynolds, M.G.; Carroll, D.S.; Karem, K.L. Factors affecting the likelihood of monkeypox’s emergence and spread in the post-smallpox era. Curr. Opin. Virol. 2012, 2, 335–343. [Google Scholar] [CrossRef]
- McCollum, A.M.; Damon, I.K. Human monkeypox. Clin. Infect. Dis. 2014, 58, 260–267. [Google Scholar] [CrossRef]
- Damon, I.K. Status of human monkeypox: Clinical disease, epidemiology and research. Vaccine 2011, 29 (Suppl. 4), D54–D59. [Google Scholar] [CrossRef]
- Von Magnus, P.; Anderson, E.; Petersen, K.; Birch-Anderson, A. A pox-like disease in cynomolgus monkeys. Acta Pathol. Microbiol. Scand. 1959, 46, 156–176. [Google Scholar]
- Prier, J.E.; Sauer, R.M. A pox disease of monkeys. Ann. N. Y. Acad. Sci. 1960, 85, 951–959. [Google Scholar] [CrossRef]
- Khodakevich, L.; Jezek, Z.; Kinzanzka, K. Isolation of monkeypox virus from wild squirrel infected in nature. Lancet 1986, 1, 98–99. [Google Scholar] [CrossRef]
- DiGiulio, D.B.; Eckburg, P.B. Monkeypox in the western hemisphere. N. Engl. J. Med. 2004, 350, 1790–1791, author reply 1790–1791. [Google Scholar] [CrossRef]
- Reed, K.D.; Melski, J.W.; Graham, M.B.; Regnery, R.L.; Sotir, M.J.; Wegner, M.V.; Kazmierczak, J.J.; Stratman, E.J.; Li, Y.; Fairley, J.A.; et al. The detection of monkeypox in humans in the western hemisphere. N. Engl. J. Med. 2004, 350, 342–350. [Google Scholar] [CrossRef]
- Centers for Disease, Control and Prevention. Update: Multistate outbreak of monkeypox—Illinois, Indiana, Kansas, Missouri, Ohio, and Wisconsin, 2003. MMWR. Morb. Mortal. Wkly. Rep. 2003, 52, 642–646. [Google Scholar]
- Parker, S.; Nuara, A.; Buller, R.M.; Schultz, D.A. Human monkeypox: An emerging zoonotic disease. Future Microbiol. 2007, 2, 17–34. [Google Scholar] [CrossRef]
- Wenner, H.A.; Bolano, C.R.; Cho, C.T.; Kamitsuka, P.S. Studies on the pathogenesis of monkey pox. 3. Histopathological lesions and sites of immunofluorescence. Arch. Gesamte Virusforsch. 1969, 27, 179–197. [Google Scholar] [CrossRef]
- Heberling, R.L.; Kalter, S.S. Induction, course, and transmissibility of monkeypox in the baboon (Papio cynocephalus). J. Infect. Dis. 1971, 124, 33–38. [Google Scholar] [CrossRef]
- Heberling, R.L.; Kalter, S.S.; Rodriguez, A.R. Poxvirus infection of the baboon (Papio cynocephalus). Bull. World Health Organ. 1976, 54, 285–294. [Google Scholar]
- Goff, A.J.; Chapman, J.; Foster, C.; Wlazlowski, C.; Shamblin, J.; Lin, K.; Kreiselmeier, N.; Mucker, E.; Paragas, J.; Lawler, J.; et al. A novel respiratory model of infection with monkeypox virus in cynomolgus macaques. J. Virol. 2011, 85, 4898–4909. [Google Scholar] [CrossRef]
- Jordan, R.; Goff, A.; Frimm, A.; Corrado, M.L.; Hensley, L.E.; Byrd, C.M.; Mucker, E.; Shamblin, J.; Bolken, T.C.; Wlazlowski, C.; et al. St-246 antiviral efficacy in a nonhuman primate monkeypox model: Determination of the minimal effective dose and human dose justification. Antimicrob. Agents Chemother. 2009, 53, 1817–1822. [Google Scholar] [CrossRef]
- Goff, A.; Mucker, E.; Raymond, J.; Fisher, R.; Bray, M.; Hensley, L.; Paragas, J. Infection of cynomolgus macaques with a recombinant monkeypox virus encoding green fluorescent protein. Arch. Virol. 2011, 156, 1877–1881. [Google Scholar] [CrossRef]
- Hooper, J.W.; Thompson, E.; Wilhelmsen, C.; Zimmerman, M.; Ichou, M.A.; Steffen, S.E.; Schmaljohn, C.S.; Schmaljohn, A.L.; Jahrling, P.B. Smallpox DNA vaccine protects nonhuman primates against lethal monkeypox. J. Virol. 2004, 78, 4433–4443. [Google Scholar] [CrossRef]
- Golden, J.W.; Josleyn, M.; Mucker, E.M.; Hung, C.F.; Loudon, P.T.; Wu, T.C.; Hooper, J.W. Side-by-side comparison of gene-based smallpox vaccine with MVA in nonhuman primates. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Song, H.; Josleyn, N.; Janosko, K.; Skinner, J.; Reeves, R.K.; Cohen, M.; Jett, C.; Johnson, R.; Blaney, J.E.; Bollinger, L.; et al. Monkeypox virus infection of rhesus macaques induces massive expansion of natural killer cells but suppresses natural killer cell functions. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Dyall, J.; Johnson, R.F.; Chen, D.Y.; Huzella, L.; Ragland, D.R.; Mollura, D.J.; Byrum, R.; Reba, R.C.; Jennings, G.; Jahrling, P.B.; et al. Evaluation of monkeypox disease progression by molecular imaging. J. Infect. Dis. 2011, 204, 1902–1911. [Google Scholar] [CrossRef]
- Earl, P.L.; Americo, J.L.; Wyatt, L.S.; Espenshade, O.; Bassler, J.; Gong, K.; Lin, S.; Peters, E.; Rhodes, L., Jr.; Spano, Y.E.; et al. Rapid protection in a monkeypox model by a single injection of a replication-deficient vaccinia virus. Proc. Natl. Acad. Sci. USA 2008, 105, 10889–10894. [Google Scholar] [CrossRef]
- Edghill-Smith, Y.; Bray, M.; Whitehouse, C.A.; Miller, D.; Mucker, E.; Manischewitz, J.; King, L.R.; Robert-Guroff, M.; Hryniewicz, A.; Venzon, D.; et al. Smallpox vaccine does not protect macaques with AIDS from a lethal monkeypox virus challenge. J. Infect. Dis. 2005, 191, 372–381. [Google Scholar] [CrossRef]
- Edghill-Smith, Y.; Golding, H.; Manischewitz, J.; King, L.R.; Scott, D.; Bray, M.; Nalca, A.; Hooper, J.W.; Whitehouse, C.A.; Schmitz, J.E.; et al. Smallpox vaccine-induced antibodies are necessary and sufficient for protection against monkeypox virus. Nat. Med. 2005, 11, 740–747. [Google Scholar] [CrossRef]
- Heraud, J.M.; Edghill-Smith, Y.; Ayala, V.; Kalisz, I.; Parrino, J.; Kalyanaraman, V.S.; Manischewitz, J.; King, L.R.; Hryniewicz, A.; Trindade, C.J.; et al. Subunit recombinant vaccine protects against monkeypox. J. Immunol. 2006, 177, 2552–2564. [Google Scholar] [CrossRef]
- Zielinski, R.J.; Smedley, J.V.; Perera, P.Y.; Silvera, P.M.; Waldmann, T.A.; Capala, J.; Perera, L.P. Smallpox vaccine with integrated il-15 demonstrates enhanced in vivo viral clearance in immunodeficient mice and confers long term protection against a lethal monkeypox challenge in cynomolgus monkeys. Vaccine 2010, 28, 7081–7091. [Google Scholar] [CrossRef]
- Arita, I.; Jezek, Z.; Khodakevich, L.; Ruti, K. Human monkeypox: A newly emerged orthopoxvirus zoonosis in the tropical rain forests of africa. Am. J. Trop. Med. Hyg. 1985, 34, 781–789. [Google Scholar]
- Stittelaar, K.J.; van Amerongen, G.; Kondova, I.; Kuiken, T.; van Lavieren, R.F.; Pistoor, F.H.; Niesters, H.G.; van Doornum, G.; van der Zeijst, B.A.; Mateo, L.; et al. Modified vaccinia virus ankara protects macaques against respiratory challenge with monkeypox virus. J. Virol. 2005, 79, 7845–7851. [Google Scholar] [CrossRef]
- Stittelaar, K.J.; Neyts, J.; Naesens, L.; van Amerongen, G.; van Lavieren, R.F.; Holy, A.; de Clercq, E.; Niesters, H.G.; Fries, E.; Maas, C.; et al. Antiviral treatment is more effective than smallpox vaccination upon lethal monkeypox virus infection. Nature 2006, 439, 745–748. [Google Scholar] [CrossRef]
- Zaucha, G.M.; Jahrling, P.B.; Geisbert, T.W.; Swearengen, J.R.; Hensley, L. The pathology of experimental aerosolized monkeypox virus infection in cynomolgus monkeys (Macaca fascicularis). Lab. Investig. 2001, 81, 1581–1600. [Google Scholar] [CrossRef]
- Barnewall, R.E.; Fisher, D.A.; Robertson, A.B.; Vales, P.A.; Knostman, K.A.; Bigger, J.E. Inhalational monkeypox virus infection in cynomolgus macaques. Front. Cell. Infect. Microbiol. 2012, 2, 117. [Google Scholar]
- Hatch, G.J.; Graham, V.A.; Bewley, K.R.; Tree, J.A.; Dennis, M.; Taylor, I.; Funnell, S.G.; Bate, S.R.; Steeds, K.; Tipton, T.; et al. Assessment of the protective effect of Imvamune and Acam2000 vaccines against aerosolized monkeypox virus in cynomolgus macaques. J. Virol. 2013, 87, 7805–7815. [Google Scholar] [CrossRef]
- Estep, R.D.; Messaoudi, I.; O’Connor, M.A.; Li, H.; Sprague, J.; Barron, A.; Engelmann, F.; Yen, B.; Powers, M.F.; Jones, J.M.; et al. Seletion of the monkeypox virus inhibitor of complement enzymes locus impacts the adaptive immune response to monkeypox virus in a nonhuman primate model of infection. J. Virol. 2011, 85, 9527–9542. [Google Scholar] [CrossRef]
- Johnson, R.F.; Dyall, J.; Ragland, D.R.; Huzella, L.; Byrum, R.; Jett, C.; St Claire, M.; Smith, A.L.; Paragas, J.; Blaney, J.E.; et al. Comparative analysis of monkeypox virus infection of cynomolgus macaques by the intravenous or intrabronchial inoculation route. J. Virol. 2011, 85, 2112–2125. [Google Scholar] [CrossRef]
- Brown, J.N.; Estep, R.D.; Lopez-Ferrer, D.; Brewer, H.M.; Clauss, T.R.; Manes, N.P.; O’Connor, M.; Li, H.; Adkins, J.N.; Wong, S.W.; et al. Characterization of macaque pulmonary fluid proteome during monkeypox infection: Dynamics of host response. Mol. Cell. Proteomics 2010, 9, 2760–2771. [Google Scholar] [CrossRef]
- Saijo, M.; Ami, Y.; Suzaki, Y.; Nagata, N.; Iwata, N.; Hasegawa, H.; Ogata, M.; Fukushi, S.; Mizutani, T.; Sata, T.; et al. Lc16m8, a highly attenuated vaccinia virus vaccine lacking expression of the membrane protein b5r, protects monkeys from monkeypox. J. Virol. 2006, 80, 5179–5188. [Google Scholar] [CrossRef]
- Saijo, M.; Ami, Y.; Suzaki, Y.; Nagata, N.; Iwata, N.; Hasegawa, H.; Iizuka, I.; Shiota, T.; Sakai, K.; Ogata, M.; et al. Virulence and pathophysiology of the congo basin and west african strains of monkeypox virus in non-human primates. J. Gen. Virol. 2009, 90, 2266–2271. [Google Scholar] [CrossRef]
- Olsen, R.G.; Blakeslee, J.R.; Mathes, L.; Nakano, J.H. Preparation and evaluation of a noninfectious monkey pox virus vaccine. J. Clin. Microbiol. 1977, 6, 50–54. [Google Scholar]
- Sauer, R.M.; Prier, J.E.; Buchanan, R.S.; Creamer, A.A.; Fegley, H.C. Studies on a pox disease of monkeys. I. Pathology. Am. J. Vet. Res. 1960, 21, 377–380. [Google Scholar]
- Cann, J.A.; Jahrling, P.B.; Hensley, L.E.; Wahl-Jensen, V. Comparative pathology of smallpox and monkeypox in man and macaques. J. Comp. Pathol. 2013, 148, 6–21. [Google Scholar] [CrossRef]
- Rubins, K.H.; Hensley, L.E.; Jahrling, P.B.; Whitney, A.R.; Geisbert, T.W.; Huggins, J.W.; Owen, A.; Leduc, J.W.; Brown, P.O.; Relman, D.A. The host response to smallpox: Analysis of the gene expression program in peripheral blood cells in a nonhuman primate model. Proc. Natl. Acad. Sci. USA 2004, 101, 15190–15195. [Google Scholar] [CrossRef]
- Brinckerhoff, W.R.; Tyzzer, E.E. Studies upon experimental variola in monkeys (Macacus cynomologus and Macaca nemestrinus) and in the orang utan (Simia satyrus): Part II. J. Med. Res. 1906, 14, 263–320. [Google Scholar]
- Hahon, N.; Wilson, B.J. Pathogenesis of variola in Macaca irus monkeys. Am. J. Hyg. 1960, 71, 69–80. [Google Scholar]
- Westwood, J.C.; Boulter, E.A.; Bowen, E.T.; Maber, H.B. Experimental respiratory infection with poxviruses. I. Clinical virological and epidemiological studies. Br. J. Exp. Pathol. 1966, 47, 453–465. [Google Scholar]
- Lancaster, M.C.; Boulter, E.A.; Westwood, J.C.; Randles, J. Experimental respiratory infection with poxviruses. Pathological studies. Br. J. Exp. Pathol. 1966, 47, 466–471. [Google Scholar]
- LeDuc, J.W.; Jahrling, P.B. Strengthening national preparedness for smallpox: An update. Emerg. Infect. Dis. 2001, 7, 155–157. [Google Scholar] [CrossRef]
- Jahrling, P.B.; Hensley, L.E.; Martinez, M.J.; Leduc, J.W.; Rubins, K.H.; Relman, D.A.; Huggins, J.W. Exploring the potential of variola virus infection of cynomolgus macaques as a model for human smallpox. Proc. Natl. Acad. Sci. USA 2004, 101, 15196–15200. [Google Scholar] [CrossRef]
- Wahl-Jensen, V.; Cann, J.A.; Rubins, K.H.; Huggins, J.W.; Fisher, R.W.; Johnson, A.J.; de Kok-Mercado, F.; Larsen, T.; Raymond, J.L.; Hensley, L.E.; et al. Progression of pathogenic events in cynomolgus macaques infected with variola virus. PLoS ONE 2011, 6. [Google Scholar] [CrossRef]
- Mucker, E.M.; Goff, A.J.; Shamblin, J.D.; Grosenbach, D.W.; Damon, I.K.; Mehal, J.M.; Holman, R.C.; Carroll, D.; Gallardo, N.; Olson, V.A.; et al. Efficacy of Tecovirimat (ST-246) in nonhuman primates infected with variola virus (smallpox). Antimicrob. Agents Chemother. 2013, 57, 6246–6253. [Google Scholar] [CrossRef]
- Alzhanova, D.; Fruh, K. Modulation of the host immune response by cowpox virus. Microbes Infect. 2010, 12, 900–909. [Google Scholar] [CrossRef]
- Wolfs, T.F.; Wagenaar, J.A.; Niesters, H.G.; Osterhaus, A.D. Rat-to-human transmission of cowpox infection. Emerg. Infect. Dis. 2002, 8, 1495–1496. [Google Scholar] [CrossRef]
- Postma, B.H.; Diepersloot, R.J.; Niessen, G.J.; Droog, R.P. Cowpox-virus-like infection associated with rat bite. Lancet 1991, 337, 733–734. [Google Scholar]
- Becker, C.; Kurth, A.; Hessler, F.; Kramp, H.; Gokel, M.; Hoffmann, R.; Kuczka, A.; Nitsche, A. Cowpox virus infection in pet rat owners: Not always immediately recognized. Dtsch. Ärztebl. Int. 2009, 106, 329–334. [Google Scholar]
- Campe, H.; Zimmermann, P.; Glos, K.; Bayer, M.; Bergemann, H.; Dreweck, C.; Graf, P.; Weber, B.K.; Meyer, H.; Büttner, M.; et al. Cowpox virus transmission from pet rats to humans, germany. Emerg. Infect. Dis. 2009, 15, 777–780. [Google Scholar] [CrossRef]
- Baxby, D.; Bennett, M.; Getty, B. Human cowpox 1969–93: A review based on 54 cases. Br. J. Dermatol. 1994, 131, 598–607. [Google Scholar] [CrossRef]
- Schwarzer, H.; Kurth, A.; Hermel, M.; Plange, N. Severe ulcerative keratitis in ocular cowpox infection. Graefe’s Arch. Clin. Exp. Ophthalmol. 2013, 251, 1451–1452. [Google Scholar] [CrossRef]
- Hall, C.J.; Stevens, J.D. Ocular cowpox. Lancet 1987, 1, 111. [Google Scholar] [CrossRef]
- Dugmore, W.N.; Dabir, Z.M. Cowpox virus. Br. J. Ophthalmol. 1992, 76, 510. [Google Scholar] [CrossRef]
- Blackford, S.; Roberts, D.L.; Thomas, P.D. Cowpox infection causing a generalized eruption in a patient with atopic dermatitis. Br. J. Ophthalmol. 1993, 129, 628–629. [Google Scholar]
- Eis-Hubinger, A.M.; Gerritzen, A.; Schneweis, K.E.; Pfeiff, B.; Pullmann, H.; Mayr, A.; Czerny, C.P. Fatal cowpox-like virus infection transmitted by cat. Lancet 1990, 336, 880. [Google Scholar]
- Pelkonen, P.M.; Tarvainen, K.; Hynninen, A.; Kallio, E.R.; Henttonen, K.; Palva, A.; Vaheri, A.; Vapalahti, O. Cowpox with severe generalized eruption, Finland. Emerg. Infect. Dis. 2003, 9, 1458–1461. [Google Scholar] [CrossRef]
- Czerny, C.P.; Eis-Hübinger, A.M.; Mayr, A.; Schneweis, K.E.; Pfeiff, B. Animal poxviruses transmitted from cat to man: Current event with lethal end. Zentralbl. Veterinärmed. B 1991, 38, 421–431. [Google Scholar]
- Klingebiel, T.; Vallbracht, A.; Doller, G.; Stierhof, Y.D.; Gerth, H.J.; Glashauser, E.; Herzau, V. A severe human cowpox infection in south germany. Pediatr. Infect. Dis. J. 1988, 7, 883–885. [Google Scholar] [CrossRef]
- Baxby, D. Is cowpox misnamed? A review of 10 human cases. Br. Med. J. 1977, 1, 1379–1381. [Google Scholar] [CrossRef]
- Essbauer, S.; Pfeffer, M.; Meyer, H. Zoonotic poxviruses. Vet. Microbiol. 2010, 140, 229–236. [Google Scholar] [CrossRef]
- Kinnunen, P.M.; Henttonen, H.; Hoffmann, B.; Kallio, E.R.; Korthase, C.; Laakkonen, J.; Niemimaa, J.; Palva, A.; Schlegel, M.; Ali, H.S.; et al. Orthopox virus infections in eurasian wild rodents. Vector Borne Zoonotic Dis. 2011, 11, 1133–1140. [Google Scholar] [CrossRef]
- Coras, B.; Essbauer, S.; Pfeffer, M.; Meyer, H.; Schroder, J.; Stolz, W.; Landthaler, M.; Vogt, T. Cowpox and a cat. Lancet 2005, 365, 446. [Google Scholar] [CrossRef]
- Kurth, A.; Wibbelt, G.; Gerber, H.P.; Petschaelis, A.; Pauli, G.; Nitsche, A. Rat-to-elephant-to-human transmission of cowpox virus. Emerg. Infect. Dis. 2008, 14, 670–671. [Google Scholar] [CrossRef]
- Wisser, J.; Pilaski, J.; Strauss, G.; Meyer, H.; Burck, G.; Truyen, U.; Rudolph, M.; Frölich, K. Cowpox virus infection causing stillbirth in an asian elephant (Elephas maximus). Vet. Rec. 2001, 149, 244–246. [Google Scholar] [CrossRef]
- Martina, B.E.; van Doornum, G.; Dorrestein, G.M.; Niesters, H.G.; Stittelaar, K.J.; Wolters, M.A.; van Bolhuis, H.G.; Osterhaus, A.D. Cowpox virus transmission from rats to monkeys, The Netherlands. Emerg. Infect. Dis. 2006, 12, 1005–1007. [Google Scholar] [CrossRef]
- Mätz-Rensing, K.; Ellerbrok, H.; Ehlers, B.; Pauli, G.; Floto, A.; Alex, M.; Czerny, C.P.; Kaup, F.J. Fatal poxvirus outbreak in a colony of new world monkeys. Vet. Pathol. 2006, 43, 212–218. [Google Scholar] [CrossRef]
- Kramski, M.; Mätz-Rensing, K.; Stahl-Hennig, C.; Kaup, F.J.; Nitsche, A.; Pauli, G.; Ellerbrok, H. A novel highly reproducible and lethal nonhuman primate model for orthopox virus infection. PLoS ONE 2010, 5. [Google Scholar] [CrossRef]
- Mätz-Rensing, K.; Stahl-Hennig, C.; Kramski, M.; Pauli, G.; Ellerbrok, H.; Kaup, F.J. The pathology of experimental poxvirus infection in common marmosets (Callithrix jacchus): Further characterization of a new primate model for orthopoxvirus infections. J. Comp. Pathol. 2012, 146, 230–242. [Google Scholar] [CrossRef]
- Abbott, D.H.; Barnett, D.K.; Colman, R.J.; Yamamoto, M.E.; Schultz-Darken, N.J. Aspects of common marmorset basic biology and life history important for biomedical research. Comp. Med. 2003, 53, 339–350. [Google Scholar]
- Vorou, R.M.; Papavassiliou, V.G.; Pierroutsakos, I.N. Cowpox virus infection: An emerging health threat. Curr. Opin. Infect. Dis. 2008, 21, 153–156. [Google Scholar] [CrossRef]
- Seet, B.T.; Johnston, J.B.; Brunetti, C.R.; Barrett, J.W.; Everett, H.; Cameron, C.; Sypula, J.; Nazarian, S.H.; Lucas, A.; McFadden, G. Poxviruses and immune evasion. Annu. Rev. Immunol. 2003, 21, 377–423. [Google Scholar] [CrossRef]
- Smith, A.L.; St Claire, M.; Yellayi, S.; Bollinger, L.; Jahrling, P.B.; Paragas, J.; Blaney, J.E.; Johnson, R.F. Intrabronchial inoculation of cynomolgus macaques with cowpox virus. J. Gen. Virol. 2012, 93, 159–164. [Google Scholar] [CrossRef]
- Johnson, R.F.; Yellayi, S.; Cann, J.A.; Johnson, A.; Smith, A.L.; Paragas, J.; Jahrling, P.B.; Blaney, J.E. Cowpox virus infection of cynomolgus macaques as a model of hemorrhagic smallpox. Virology 2011, 418, 102–112. [Google Scholar] [CrossRef]
- Song, H.; Janosko, K.; Johnson, R.F.; Qin, J.; Josleyn, N.; Jett, C.; Byrum, R.; St Claire, M.; Dyall, J.; Blaney, J.E.; et al. Poxvirus antigen staining of immune cells as a biomarker to predict disease outcome in monkeypox and cowpox virus infection in non-human primates. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Schmitt, A.; Mätz-Rensing, K.; Kaup, F.-J. Non-Human Primate Models of Orthopoxvirus Infections. Vet. Sci. 2014, 1, 40-62. https://doi.org/10.3390/vetsci1010040
Schmitt A, Mätz-Rensing K, Kaup F-J. Non-Human Primate Models of Orthopoxvirus Infections. Veterinary Sciences. 2014; 1(1):40-62. https://doi.org/10.3390/vetsci1010040
Chicago/Turabian StyleSchmitt, Anne, Kerstin Mätz-Rensing, and Franz-Josef Kaup. 2014. "Non-Human Primate Models of Orthopoxvirus Infections" Veterinary Sciences 1, no. 1: 40-62. https://doi.org/10.3390/vetsci1010040
APA StyleSchmitt, A., Mätz-Rensing, K., & Kaup, F.-J. (2014). Non-Human Primate Models of Orthopoxvirus Infections. Veterinary Sciences, 1(1), 40-62. https://doi.org/10.3390/vetsci1010040




