Internal Seed Structure of Alpine Plants and Extreme Cold Exposure
Abstract
1. Summary
2. Data Description
2.1. Structure of Dataset
2.2. Descriptive Analyses of Seed Data
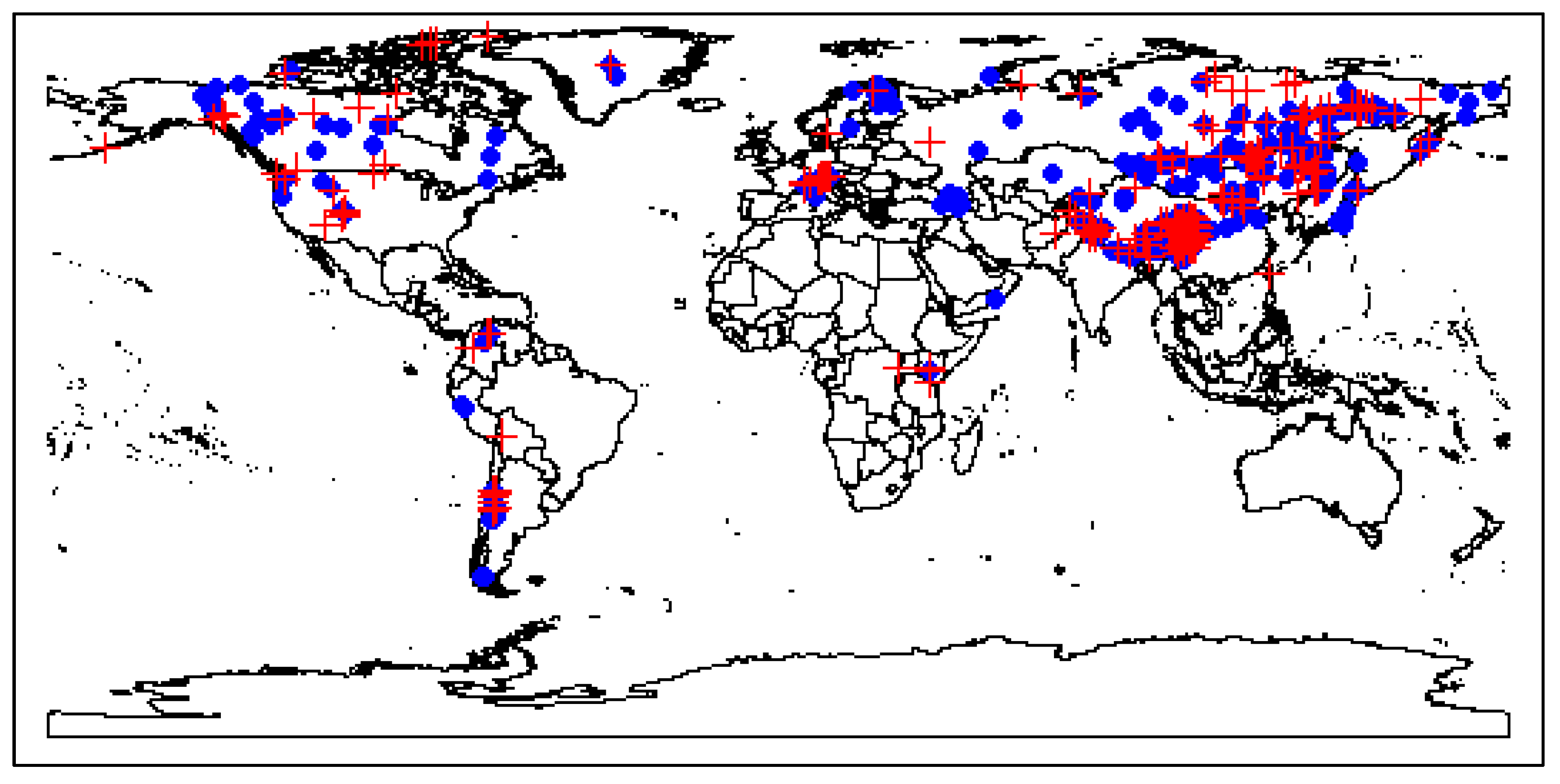
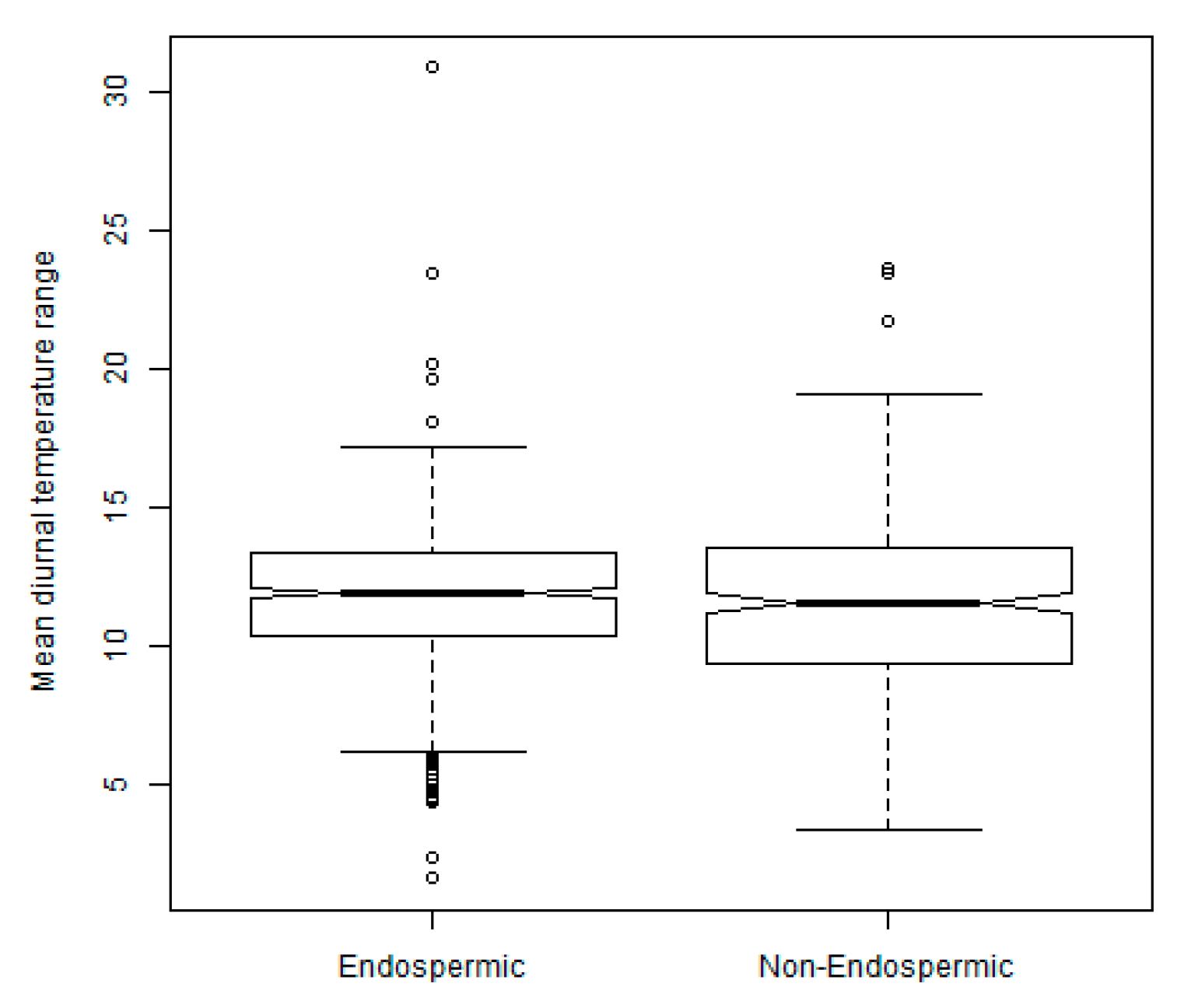
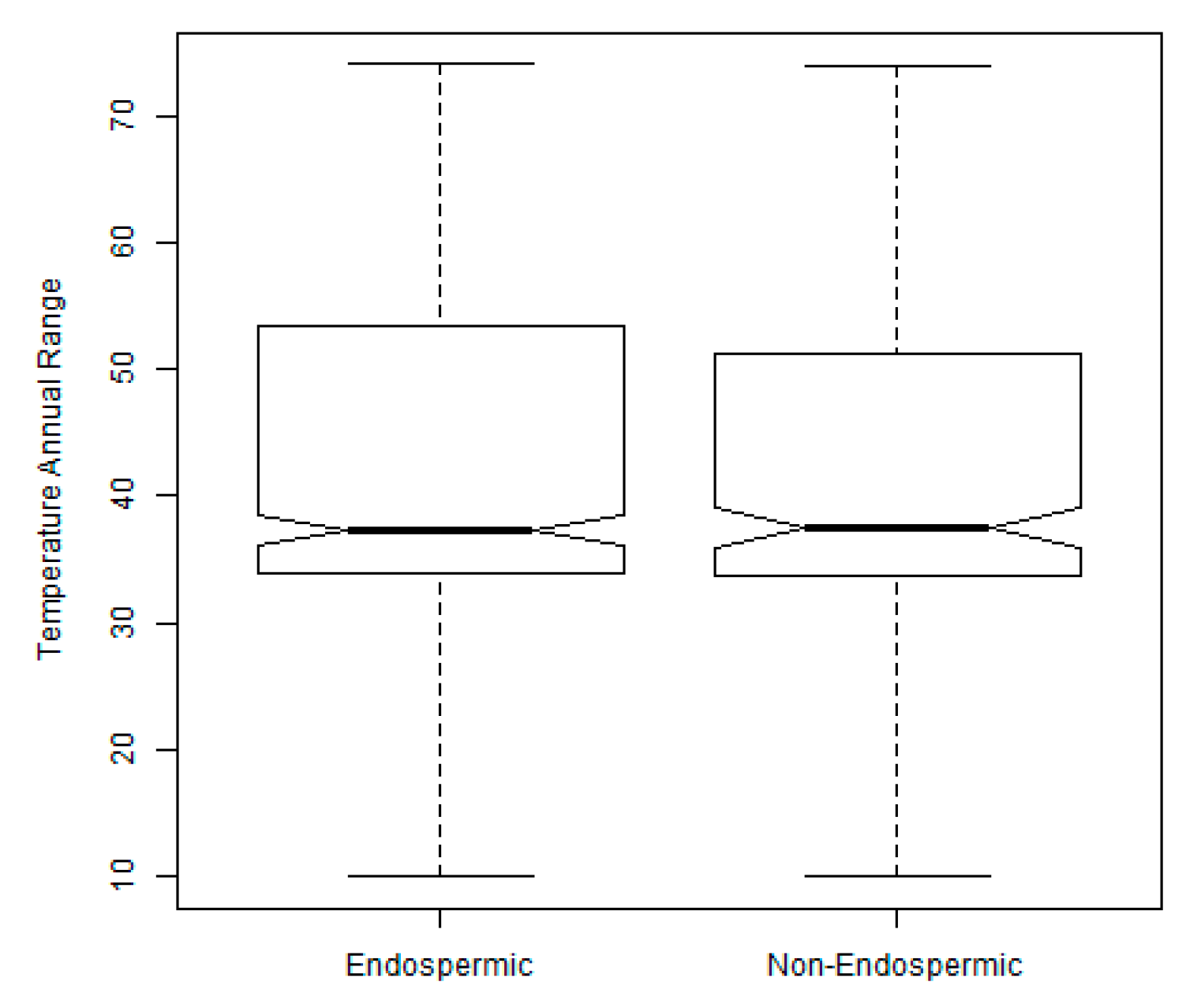
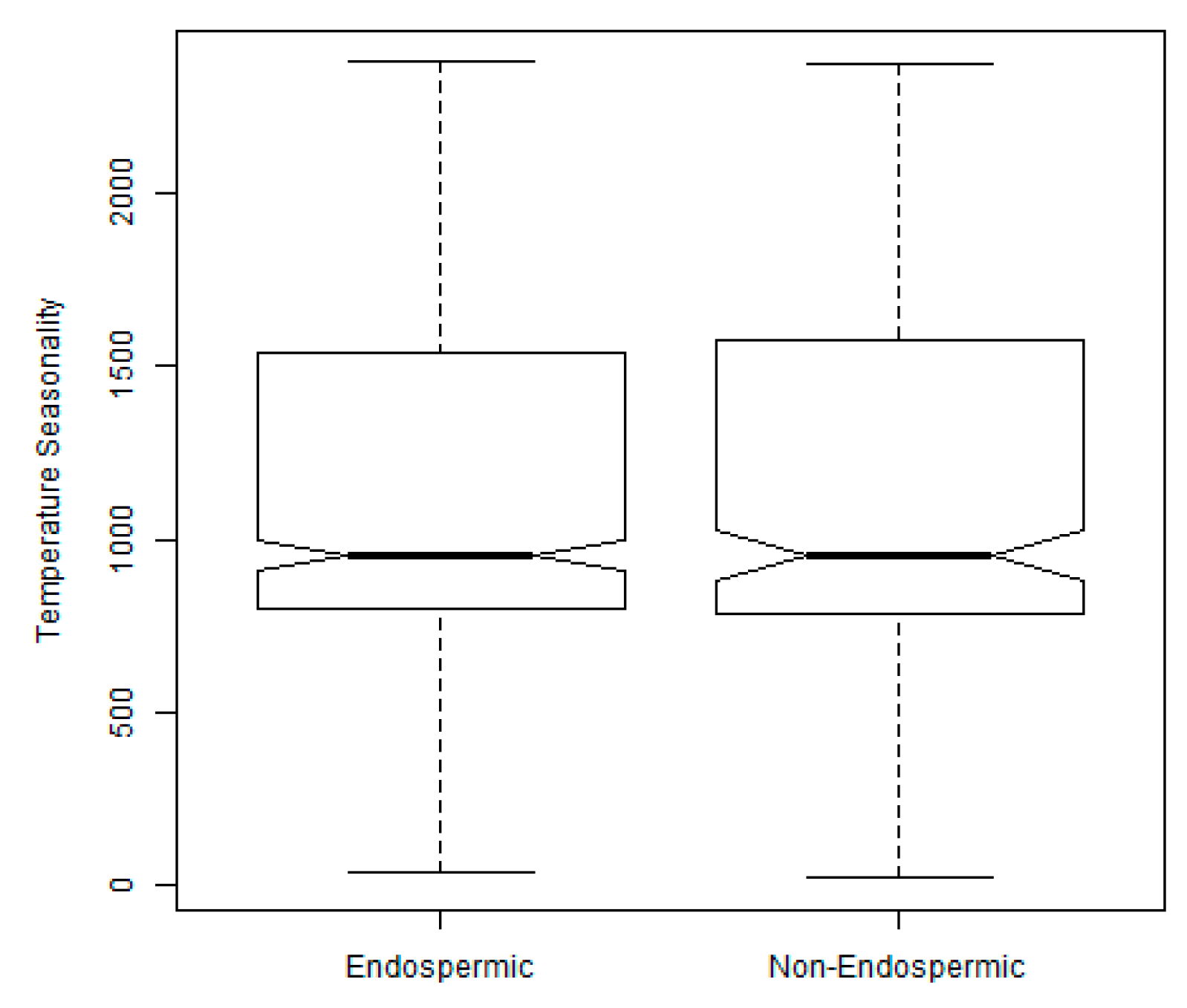
2.3. Exploration of Climatic Data Associated with Temperature Minima Locations
3. Methods
4. User Notes
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Larcher, W. Bioclimatic Temperatures in the High Alps. In Plants in Alpine Regions: Cell Physiology of Adaption and Survival Stratergies; Lutz, C., Ed.; Springer: Berlin, Germany, 2012; pp. 21–27. [Google Scholar]
- Lütz, C. Plants in Alpine Regions: Cell Physiology of Adaption and Survival Strategies; Springer: Berlin, Germany, 2012. [Google Scholar]
- Körner, C. Alpine Plant Life: Functional Plant Ecology of High Mountain Ecosystems; Springer: Berlin, Germany, 2003. [Google Scholar]
- Körner, C. A re-Assessment of High Elevation Treeline Positions and Their Explanation. Oecologia 1998, 115, 445–459. [Google Scholar] [CrossRef] [PubMed]
- Tranquillini, W. Physiological ecology of the alpine timberline. In Tree Existence at High Altitudes with Special Reference to the European Alps; Springer: Berlin, Germany, 1979. [Google Scholar]
- Beck, E. Equilibrium freezing of leaf water and extracellular ice formation in Afroalpine ‘giant rosette’plants. Planta 1984, 162, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Marcante, S. Frost as a limiting factor for recruitment and establishment of early development stages in an alpine glacier foreland? J. Veg. Sci. 2012, 23, 858–868. [Google Scholar] [CrossRef]
- Larcher, W.; Kainmüller, C.; Wagner, J. Survival types of high mountain plants under extreme temperatures. Flora-Morphol. Distrib. Funct. Ecol. Plants 2010, 205, 3–18. [Google Scholar] [CrossRef]
- Sakai, A.; Larcher, W. Frost Survival of Plants. Responses and Adaptation to Freezing Stress; Springer: Berlin, Germany, 1987. [Google Scholar]
- Burke, M. Freezing and injury in plants. Annu. Rev. Plant Physiol. 1976, 27, 507–528. [Google Scholar] [CrossRef]
- Squeo, F. Freezing tolerance and avoidance in high tropical Andean plants: Is it equally represented in species with different plant height? Oecologia 1991, 86, 378–382. [Google Scholar] [CrossRef]
- Levitt, J. Responses of Plants to Environmental Stress. Chilling, Freezing, and High Temperature Stresses; Academic. Press: New York, NY, USA, 1980. [Google Scholar]
- Levitt, J. Responses of Plants to Environmental Stresses. Volume II. Water, Radiation, Salt, and Other Stresses; Academic Press: Cambridge, MA, USA, 1980. [Google Scholar]
- Ristic, Z.; Ashworth, E.N. Response of Xylem Ray Parenchyma Cells of Red Osier Dogwood (Cornus sericea L.) to Freezing Stress (Microscopic Evidence of Protoplasm Contraction). Plant Physiol. 1994, 104, 737–746. [Google Scholar] [CrossRef][Green Version]
- Sakai, A. Survival of the twig of woody plants. Nature 1960, 185, 393–394. [Google Scholar] [CrossRef]
- Hacker, J. Inflorescences of alpine cushion plants freeze autonomously and may survive subzero temperatures by supercooling. Plant Sci. 2011, 180, 149–156. [Google Scholar] [CrossRef]
- Ladinig, U. How endangered is sexual reproduction of high-mountain plants by summer frosts? Frost resistance, frequency of frost events and risk assessment. Oecologia 2013, 171, 1–18. [Google Scholar] [CrossRef]
- Williams, R. Patterns of air temperature and accumulation of snow in subalpine heathlands and grasslands on the Bogong High Plains, Victoria. Aust. J. Ecol. 1987, 12, 153–163. [Google Scholar] [CrossRef]
- Jaganathan, G.K.; Dalrymple, S.E.; Liu, B. Towards an understanding of factors controlling seed bank composition and longevity in the alpine environment. Bot. Rev. 2015, 81, 70–103. [Google Scholar] [CrossRef]
- Lenoir, J. A significant upward shift in plant species optimum elevation during the 20th century. Science 2008, 320, 1768–1771. [Google Scholar] [CrossRef] [PubMed]
- Hughes, L. Biological consequences of global warming: Is the signal already apparent? Trends Ecol. Evol. 2000, 15, 56–61. [Google Scholar] [CrossRef]
- Foden, W.B. Identifying the world’s most climate change vulnerable species: A systematic trait-based assessment of all birds, amphibians and corals. PLoS ONE 2013, 8, 65427. [Google Scholar] [CrossRef] [PubMed]
- Parolo, G.; Rossi, G. Upward migration of vascular plants following a climate warming trend in the Alps. Basic Appl. Ecol. 2008, 9, 100–107. [Google Scholar] [CrossRef]
- Gworek, J.R.; Vander Wall, S.B.; Brussard, P.F. Changes in biotic interactions and climate determine recruitment of Jeffrey pine along an elevation gradient. For. Ecol. Manag. 2007, 239, 57–68. [Google Scholar] [CrossRef]
- Gobiet, A. 21st century climate change in the European Alps—A review. Sci. Total Environ. 2014, 493, 1138–1151. [Google Scholar] [CrossRef] [PubMed]
- Björk, R.G.; Molau, U. Ecology of alpine snowbeds and the impact of global change. Arct. Antarct. Alp. Res. 2007, 39, 34–43. [Google Scholar] [CrossRef]
- Bai, Y.; Booth, D.T.; Romo, G.T. Winterfat (Eurotia lanata (Pursh) Moq.) seedbed ecology: Low temperature exotherms and cold hardiness in hydrated seeds as influenced by imbibition temperature. Ann. Bot. 1998, 81, 595–602. [Google Scholar] [CrossRef][Green Version]
- Gusta, L.V.; Gao, Y.P.; Benning, N.T. Freezing and desiccation tolerance of imbibed canola seed. Physiol. Plant. 2006, 127, 237–246. [Google Scholar] [CrossRef]
- Mondoni, A. Climate warming could shift the timing of seed germination in alpine plants. Ann. Bot. 2012, 110, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Wipf, S.; Rixen, C. A review of snow manipulation experiments in Arctic and alpine tundra ecosystems. Polar Res. 2010, 29, 95–109. [Google Scholar] [CrossRef]
- Junttila, O.; Stushnoff, C. Freezing avoidance by deep supercooling in hydrated lettuce seeds. Nature 1977, 269, 325–327. [Google Scholar] [CrossRef]
- Bourque, J.E.; Wallner, S.J. Endosperm and pericarp involvement in the supercooling of imbibed lettuce seeds. Plant Physiol. 1982, 70, 1571. [Google Scholar] [CrossRef] [PubMed]
- Angell, C.A. Formation of glasses from liquids and biopolymers. Science 1995, 267, 1924–1935. [Google Scholar] [CrossRef] [PubMed]
- Keefe, P.; Moore, K. Freeze desiccation: A second mechanism for the survival of hydrated lettuce (Lactuca sativa L.) seed at sub-zero temperatures. Ann. Bot. 1981, 47, 635–645. [Google Scholar] [CrossRef]
- Keefe, P.; Moore, K. Freezing Tolerance in Hydrated Lactuca sativa (L) Seed: A Model to Explain Observed Variation Between Seed Lots. Ann. Bot. 1983, 51, 373–383. [Google Scholar] [CrossRef]
- GBIF Occurrence Download Accessed from R via rgbif. 2017. Available online: https://github.com/ropensci/rgbif (accessed on 22 June 2019).
- Fick, S.E.; Hijmans, R.J. Worldclim 2: New 1-km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Mabberley, D.J. Mabberley’s Plant-Book: A Portable Dictionary of Plants, their Classification and Uses; Cambridge University Press: Cambridge, UK, 2008. [Google Scholar]
- Martin, A.C. The comparative internal morphology of seeds. Am. Midl. Nat. 1946, 36, 513–660. [Google Scholar] [CrossRef]
- Chamberlain, S.; Barve, V.; Mcglinn, D.; Oldoni, D. rgbif: Interface to the Global Biodiversity Information Facility API. R Package Version 1.1.0. 2019. Available online: https://CRAN.R-project.org/package=rgbif (accessed on 29 June 2019).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- RStudio Team. RStudio: Integrated Development for R Version 1.1.423; RStudio, Inc.: Boston, MA, USA, 2016; Available online: http://www.rstudio.com/ (accessed on 16 February 2018).



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaganathan, G.K.; Dalrymple, S.E. Internal Seed Structure of Alpine Plants and Extreme Cold Exposure. Data 2019, 4, 107. https://doi.org/10.3390/data4030107
Jaganathan GK, Dalrymple SE. Internal Seed Structure of Alpine Plants and Extreme Cold Exposure. Data. 2019; 4(3):107. https://doi.org/10.3390/data4030107
Chicago/Turabian StyleJaganathan, Ganesh K., and Sarah E. Dalrymple. 2019. "Internal Seed Structure of Alpine Plants and Extreme Cold Exposure" Data 4, no. 3: 107. https://doi.org/10.3390/data4030107
APA StyleJaganathan, G. K., & Dalrymple, S. E. (2019). Internal Seed Structure of Alpine Plants and Extreme Cold Exposure. Data, 4(3), 107. https://doi.org/10.3390/data4030107




