Healthy Drinks with Lovely Colors: Phenolic Compounds as Constituents of Functional Beverages
Abstract
1. Introduction
2. The Traditional Mediterranean Diet
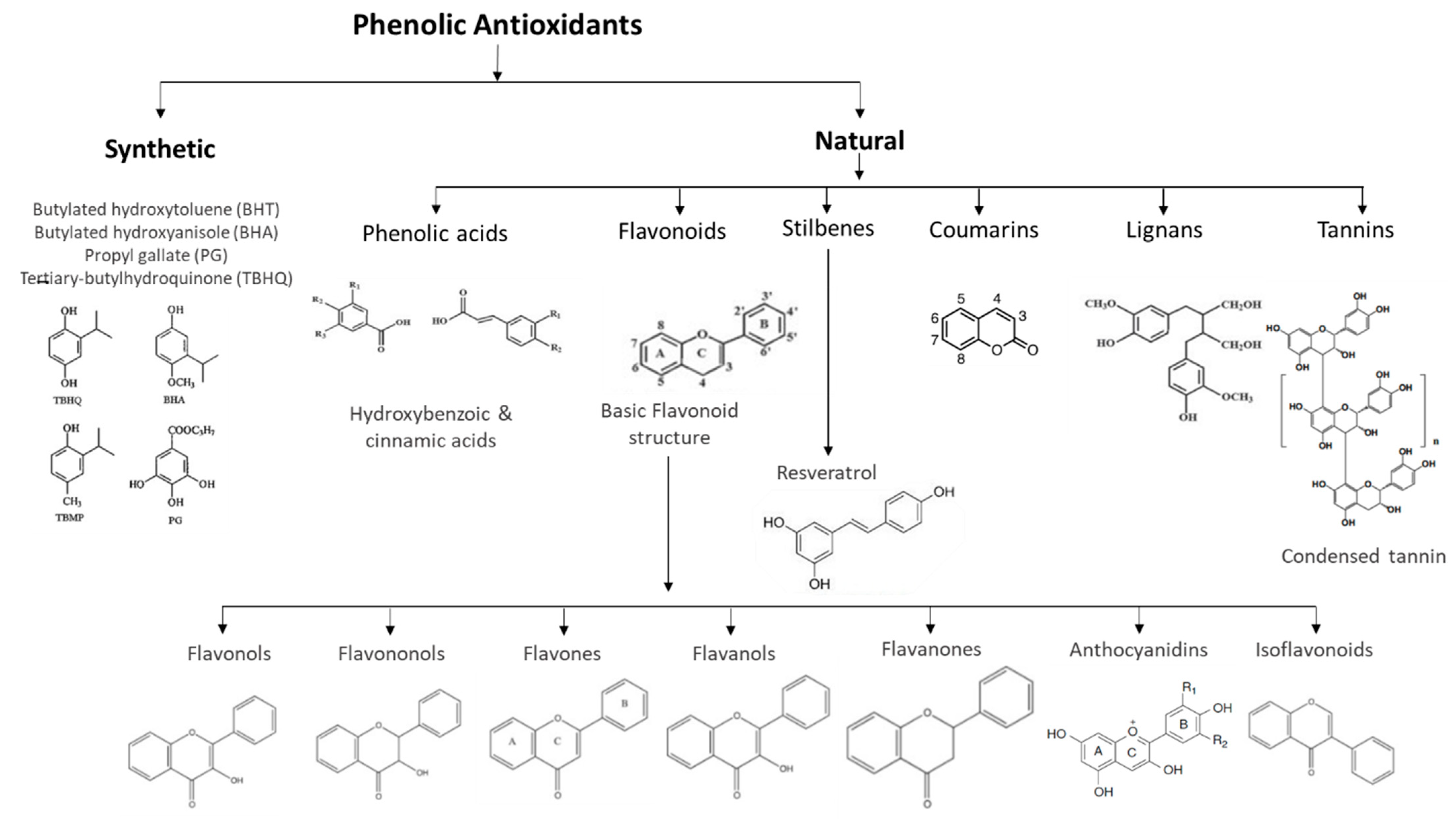
3. Classification of Phenolic Antioxidants
3.1. Natural Phenolic Antioxidants in Beverages
3.2. Synthetic Phenolic Antioxidants in Beverages
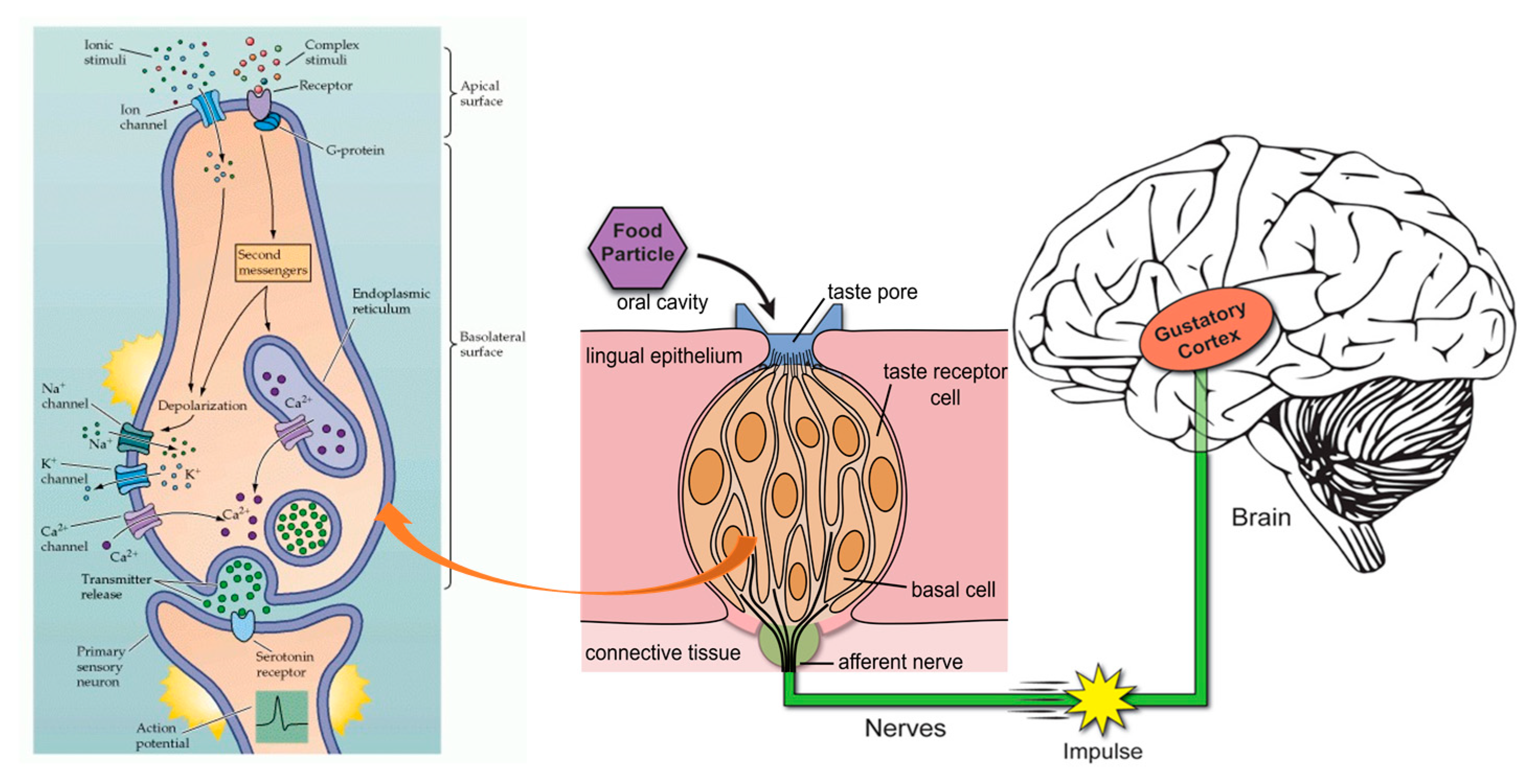
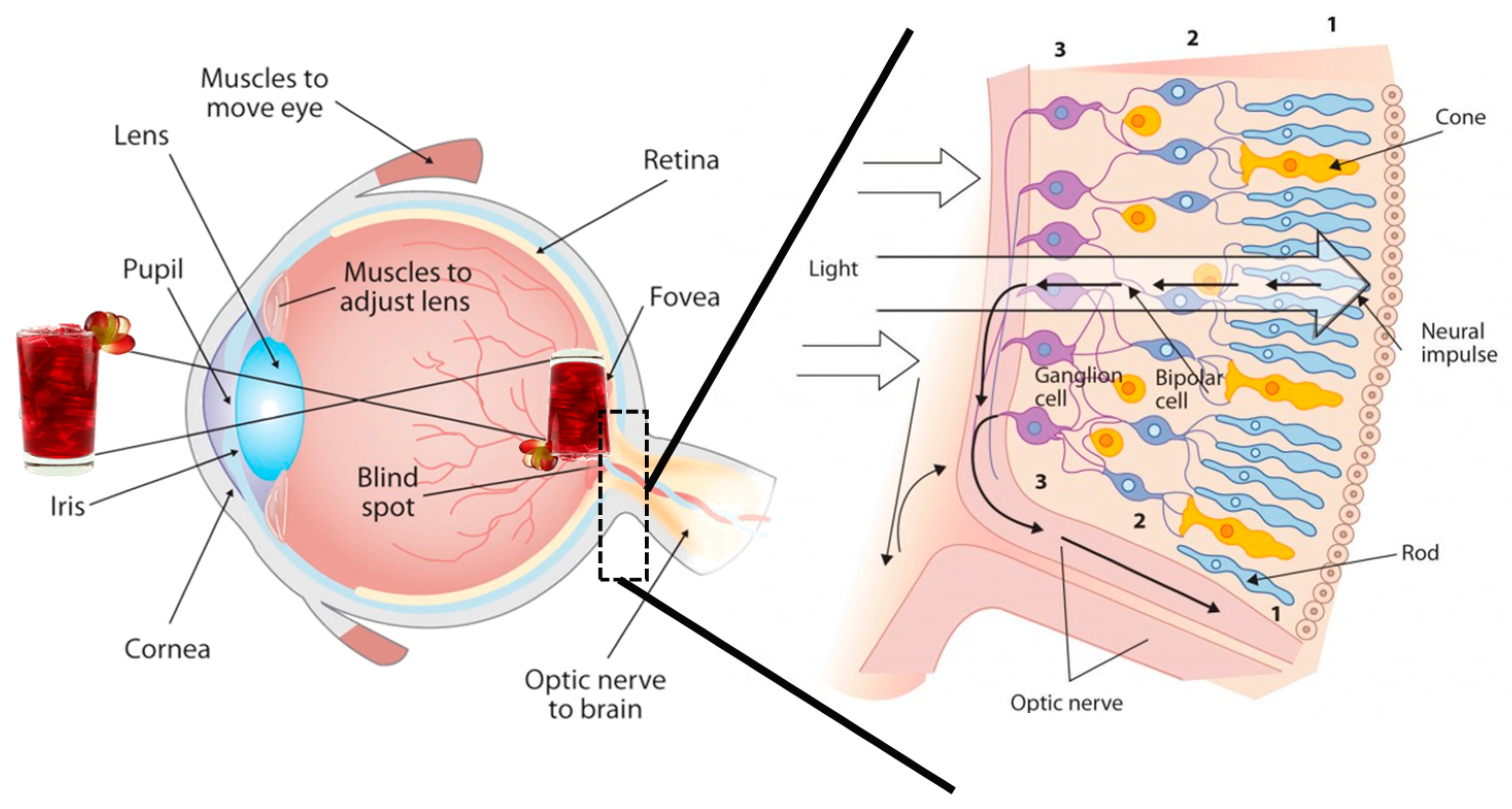
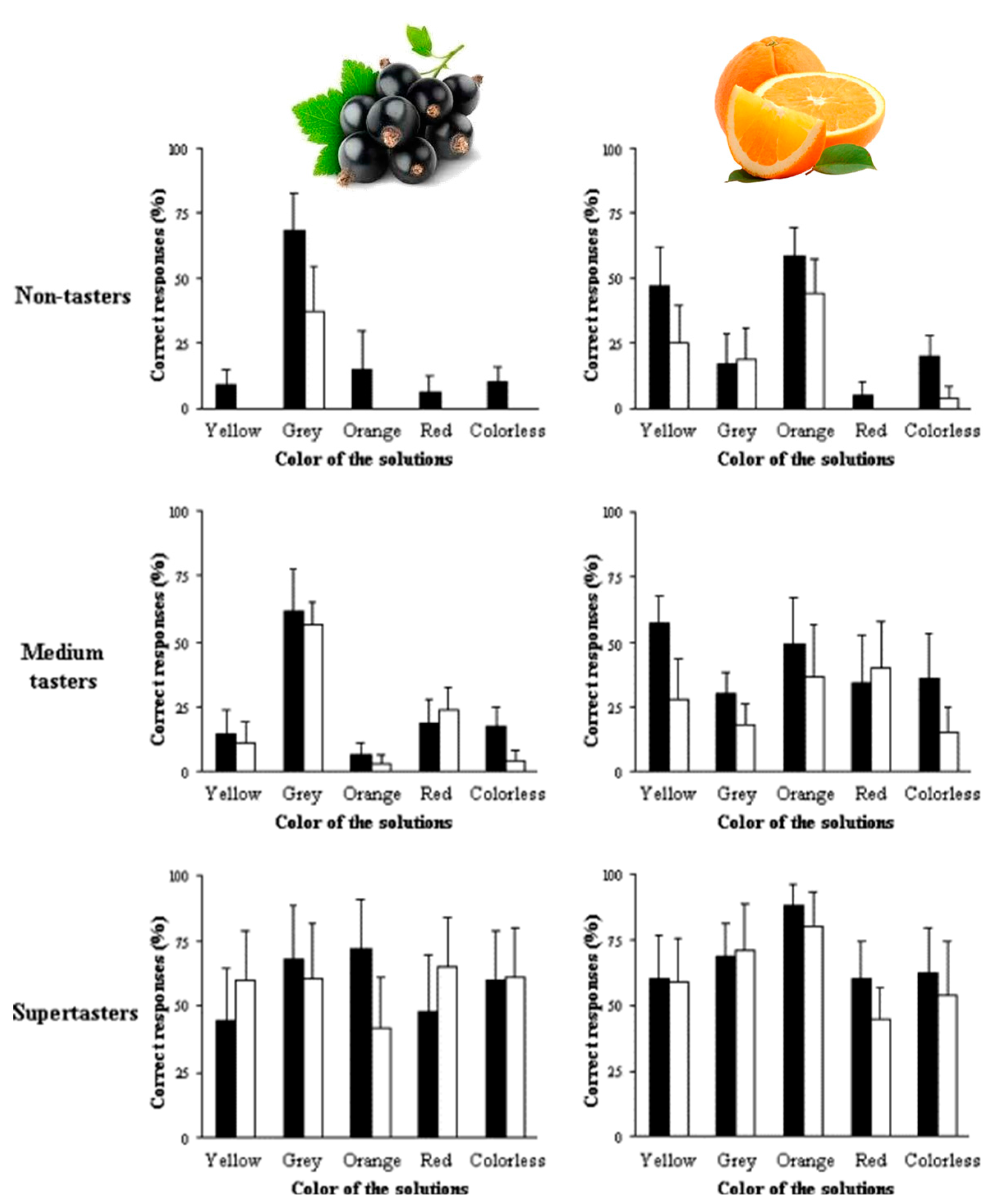
4. Phenolic Antioxidants and Human Health
5. Digestion, Catabolism, and Bioaccessibility of Beverage Phenolic Compounds
6. Final Remarks
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Piqueras-Fizman, B.; Spence, C. Sensory expectations based on product-extrinsic food cues: An interdisciplinary review of the empirical evidence and theoretical accounts. Food Qual. Prefer. 2015, 40, 165–179. [Google Scholar] [CrossRef]
- Vilela, A.; Cosme, F. Drink Red: Phenolic Composition of Red Fruit Juices and Their Sensorial Acceptance. Beverages 2016, 2, 29. [Google Scholar] [CrossRef]
- Spence, C.; Okajima, K.; Cheok, A.D.; Petit, O.; Michel, C. Eating with our eyes: From visual hunger to digital satiation. Brain Cogn. 2016, 110, 53–63. [Google Scholar] [CrossRef]
- Vilela, A.; Inês, A.; Cosme, F. Is wine savory? Umami taste in wine. SDRP J. Food Sci. Technol. 2016, 1. [Google Scholar] [CrossRef][Green Version]
- Purves, D.; Augustine, G.J.; Fitzpatrick, D. (Eds.) Taste Receptors and the Transduction of Taste Signals. In Neuroscience, 2nd ed.; Sinauer Associates: Sunderland, MA, USA, 2001. Available online: https://www.ncbi.nlm.nih.gov/books/NBK11148/ (accessed on 5 December 2020).
- Vera, L.; Wooding, S. Taste: Links in the Chain from Tongue to Brain. Front. Young Minds 2017, 5, 33. [Google Scholar] [CrossRef]
- Mannu, G.S. Retinal phototransduction. Neurosciences 2014, 19, 275–280. [Google Scholar] [PubMed]
- Stangor, C.; Walinga, J. Chapter 5. Sensing and Perceiving. In Introduction to Psychology, 1st ed.; Stangor, C., Walinga, J., Eds.; BCcampus: Victoria, BC, Canada, 2014; ISBN 978-1-77420-005-6. [Google Scholar]
- Moir, H.C. Some observations on the appreciation of flavour in foodstuffs. J. Soc. Chem. Industry 1936, 55, 145–148. [Google Scholar] [CrossRef]
- Zampini, M.; Sanabria, D.; Phillips, N.; Spence, C. The multisensory perception of flavor: Assessing the influence of color cues on flavor discrimination responses. Food Qual. Prefer. 2007, 18, 975–984. [Google Scholar] [CrossRef]
- Zampini, M.; Wantling, E.; Phillips, N.; Spence, C. Multisensory flavor perception: Assessing the influence of fruit acids and color cues on the perception of fruit-flavored beverages. Food Qual. Prefer. 2008, 19, 335–343. [Google Scholar] [CrossRef]
- O’Mahony, M. Gustatory responses to non-gustatory stimuli. Perception 1983, 12, 627–633. [Google Scholar] [CrossRef]
- Koch, C.; Koch, E.C. Preconceptions of taste based on color. J. Psychol. 2003, 137, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Tomasik-Krótki, J.; Strojny, J. Scaling of sensory impressions. J. Sens. Stud. 2008, 23, 251–266. [Google Scholar] [CrossRef]
- Wan, X.; Woods, A.T.; van den Bosch, J.; McKenzie, K.J.; Velasco, C.; Spence, C. Cross-cultural differences in crossmodal correspondences between tastes and visual features. Front. Psychol Cogn. 2014, 5, 1365. [Google Scholar] [CrossRef]
- Vogt, T. Phenylpropanoid Biosynthesis. Mol. Plant. 2010, 3, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, L.L.; Carvalho, M.V.; Melo, L. Health-promoting and sensory properties of phenolic compounds in food. Rev. Ceres 2014, 61, 764–779. [Google Scholar] [CrossRef]
- Emerson Ecologics. Polyphenols—The Star of the Mediterranean Diet. 2018. Available online: https://edu.emersonecologics.com/2018/02/28/polyphenols-star-mediterranean-diet/ (accessed on 19 December 2020).
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef] [PubMed]
- D’Archivio, M.; Filesi, C.; Vari, R.; Scazzocchio, B.; Masella, R. Bioavailability of the polyphenols: Status and controversies. Int. J. Mol. Sci. 2010, 11, 1321–1342. [Google Scholar] [CrossRef]
- Domínguez-Avila, J.A.; Wall-Medrano, A.; Velderrain-Rodríguez, G.R.; Chen, O.; Salazar-López, N.J.; Robles-Sánchez, M.; González-Aguilar, G.A. Gastrointestinal interactions, absorption, splanchnic metabolism, and pharmacokinetics of orally ingested phenolic compounds. Food Funct. 2017, 8, 15–38. [Google Scholar] [CrossRef]
- Day, A.J.; Canada, F.J.; Diaz, J.C.; Kroon, P.A.; Mclauchlan, R.; Faulds, C.B.; Plumb, G.W.; Morgan, M.R.; Williamson, G. Dietary flavonoid and isoflavone glycosides are hydrolysed by the lactase site of lactase phloridzin hydrolase. FEBS Lett. 2000, 468, 166–170. [Google Scholar] [CrossRef]
- Aura, A.M. Microbial metabolism of dietary phenolic compounds in the colon. Phytochem. Rev. 2008, 7, 407–429. [Google Scholar] [CrossRef]
- Espín, J.C.; González-Sarrías, A.; Tomás-Barberán, F.A. The gut microbiota: A key factor in the therapeutic effects of (poly) phenols. Biochem. Pharmacol. 2017, 139, 82–93. [Google Scholar] [CrossRef]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef]
- Kasote, D.M.; Katyare, S.S.; Hegde, M.V.; Bae, H. Significance of antioxidant potential of plants and its relevance to therapeutic applications. Int. J. Biol. Sci. 2015, 11, 982–991. [Google Scholar] [CrossRef]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef]
- Peredo-Lovillo, A.; Romero-Luna, H.E.; Jiménez-Fernández, M. Health promoting microbial metabolites produced by gut microbiota after prebiotics metabolism. Food Res. Int. 2020, 136, 109473. [Google Scholar] [CrossRef]
- Spence, C.; Wan, X.; Woods, A.; Velasco, C.; Deng, J.; Youssef, J.; Deroy, O. On tasty colours and colourful tastes? Assessing, explaining, and utilizing cross-modal correspondences between colours and basic tastes. Flavour 2015, 4, 23. [Google Scholar] [CrossRef]
- Burin, V.M.; Falcão, L.D.; Gonzaga, L.V.; Fett, R.; Rosier, J.P.; Bordignon-Luiz, M.T. Colour, phenolic content and antioxidant activity of grape juice. Ciência e Tecnologia de Alimentos 2010, 30, 1027–1032. [Google Scholar] [CrossRef]
- Spence, C. Multisensory flavour perception. Curr. Biol. 2013, 23, R365–R369. [Google Scholar] [CrossRef] [PubMed]
- Keys, A.; Menotti, A.; Karvonen, M.J.; Aravanis, C.; Blackburn, H.; Buzina, R.; Djordjevic, B.S.; Dontas, A.S.; Fidanza, F.; Keys, M.H.; et al. The diet and 15-year death rate in the seven countries study. Am. J. Epidemiol. 1986, 124, 903–915. [Google Scholar] [CrossRef] [PubMed]
- Radd-Vagenas, S.; Kouris-Blazos, A.; Singh, M.F.; Flood, V.M. Evolution of Mediterranean diets and cuisine: Concepts and definitions. Asia Pac. J. Clin. Nutr. 2017, 26, 749–763. [Google Scholar] [CrossRef]
- Mazzocchi, A.; Leone, L.; Agostoni, C.; Pali-Schöll, I. The Secrets of the Mediterranean Diet. Does [Only] Olive Oil Matter? Nutrients 2019, 11, 2941. [Google Scholar] [CrossRef]
- Sofi, F.; Abbate, R.; Gensini, G.F.; Casini, A. Accruing evidence on benefits of adherence to the Mediterranean diet on health: An updated systematic review and meta-analysis. Am. J. Clin. Nutr. 2010, 92, 1189–1196. [Google Scholar] [CrossRef] [PubMed]
- Altomare, R.; Cacciabaudo, F.; Damiano, G.; Palumbo, V.D.; Gioviale, M.C.; Bellavia, M.; Tomasello, G.; Lo Monte, A.I. The Mediterranean Diet: A History of Health. Iranian J. Publ. Health 2013, 42, 449–457. [Google Scholar]
- Sanchez-Villegas, A.Z.; Zazpe, I. A healthy-eating model called mediterranean diet. In Prevention of Cardiovascular Disease through the Mediterranean Diet; Sánchez-Villegas, A., Sánchez-Tainta, A., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 1–24. [Google Scholar] [CrossRef]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61 (Suppl. 6), 1402S–1406S. [Google Scholar] [CrossRef] [PubMed]
- Leaf, A. Dietary prevention of coronary heart disease: The Lyon Diet Heart Study. Circulation 1999, 99, 733–735. [Google Scholar] [CrossRef] [PubMed][Green Version]
- de Lorgeril, M.; Salen, P. The Mediterranean diet in secondary prevention of coronary heart disease. Clin. Investig. Med. 2006, 29, 154–158. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvadoó, J.; Covas, M.I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of a Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- Vasilopoulou, E.; Georga, K.; Joergensen, M.; Naska, A.; Trichopoulou, A. The antioxidant properties of Greek foods and the flavonoid content of the Mediterranean menu. Curr. Med. Chem. Immunol. Endocr. Metab. Agents 2005, 5, 33–45. [Google Scholar] [CrossRef]
- Salas-Salvadó, J.; Huetos-Solano, M.D.; García-Lorda, P.; Bulló, M. Diet and dietetics in al-Andalus. Br. J. Nutr. 2006, 96, S100–S104. [Google Scholar] [CrossRef]
- Weichselbaum, D.E.; Benelam, B. Synthesis Report No 6: Traditional Foods in Europe; European Food Information Resource (EuroFIR) Consortium: Brussels, Belgium; Institute of Food Research; Norwich Research Park: Norwich, UK, 2009; ISBN 0 907667 67 82009. [Google Scholar]
- Kromhout, D.; Keys, A.; Aravanis, C.; Buzina, R.; Fidanza, F.; Giampaoli, S.; Jansen, A.; Menotti, A.; Nedeljkovic, S.; Pekkarinen, M.; et al. Food consumption patterns in the 1960s in seven countries. Am. J. Clin. Nutr. 1989, 49, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Alberti-Fidanza, A.; Fidanza, F. Mediterranean adequacy index of Italian diets. Public Health Nutr. 2004, 7, 937–941. [Google Scholar] [CrossRef]
- Trichopoulou, A. Mediterranean diet, traditional foods, and health: Evidence from the Greek EPIC cohort. Food Nutr. Bull. 2007, 28, 236–240. [Google Scholar] [CrossRef]
- Kennedy, D.O.; Little, W.; Scholey, A.B. Attenuation of Laboratory-Induced Stress in Humans after Acute Administration of Melissa officinalis (Lemon Balm). Psychosom. Med. 2004, 66, 607–613. [Google Scholar] [CrossRef]
- Herrera, T.; Aguilera, Y.; Rebollo-Hernanza, M.; Bravoa, E.; Beníteza, V.; Martínez-Sáezb, N.; Arribasc, S.M.; Castillo, M.D.; Martín-Cabrejasa, M.A. Teas and herbal infusions as sources of melatonin and other bioactive non-nutrient components. LWT 2018, 89, 65–73. [Google Scholar] [CrossRef]
- Kouris-Blazos, A.; Itsiopoulos, C. Low all-cause mortality despite high cardiovascular risk in elderly Greek-born Australians: Attenuating potential of diet? Asia Pac. J. Clin. Nutr. 2014, 23, 532–544. [Google Scholar] [CrossRef] [PubMed]
- Carluccio, M.A.; Calabriso, N.; Scoditti, E.; Massaro, M.; De Caterina, R. Chapter 27—Mediterranean Diet Polyphenols. In The Mediterranean Diet; Preedy, V.R., Watson, R.R., Eds.; Academic Press: Cambridge, MA, USA, 2015; pp. 291–300. [Google Scholar] [CrossRef]
- Tyrovolas, S.; Polychronopoulos, E.; Bountziouka, V.; Zeimbekis, A.; Tsiligiani, I.; Papoutsou, S.; Gotsis, E.; Metallinos, G.; Lionis, C.; Panagiotakos, D.B. Level of adherence to the Mediterranean diet among elderly individuals living in Mediterranean Islands: Nutritional report from the MEDIS study. Ecol. Food Nutr. 2009, 48, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects—A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Shahidi, F.; Naczk, M. Phenolics in Food and Nutraceuticals; CRC Press: Boca Raton, FL, USA, 2004; pp. 1–558. [Google Scholar]
- Vinson, J.A.; Su, X.; Zubik, L.; Bose, P. Phenol antioxidant quantity and quality in foods: Fruits. J. Agric. Food Chem. 2001, 49, 5315–5321. [Google Scholar] [CrossRef]
- Zhang, Y.; Seeram, N.P.; Lee, R.; Feng, L.; Heber, D. Isolation and identification of strawberry phenolics with antioxidant and human cancer cell antiproliferative properties. J. Agric. Food Chem. 2008, 56, 670–675. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Das, M. Functional foods: An overview. Food Sci. Biotechnol. 2011, 20, 861–875. [Google Scholar] [CrossRef]
- Alarcón-Flores, M.I.; Romero-González, R.; Vidal, J.L.M.; Frenich, A.G. Multiclass determination of phytochemicals in vegetables and fruits by ultra high-performance liquid chromatography coupled to tandem mass spectrometry. Food Chem. 2013, 141, 1120–1129. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Dietary Bioactive Compounds and Their Health Implications. J. Food Sci. 2013, 78, A18–A25. [Google Scholar] [CrossRef]
- McCarty, M.F. Proposal for a dietary “phytochemical index”. Med. Hypotheses 2004, 63, 813–817. [Google Scholar] [CrossRef]
- Pallauf, K.; Bendall, J.K.; Scheiermann, C.; Watschinger, K.; Hoffmann, J.; Roeder, T.; Rimbach, G. Vitamin C and lifespan in model organisms. Food Chem. Toxicol. 2013, 58, 255–263. [Google Scholar] [CrossRef]
- Zingg, J.M. Vitamin E: An overview of major research directions. Mol. Aspects Med. 2007, 28, 400–422. [Google Scholar] [CrossRef]
- Nanditha, B.; Prabhasankar, P. Antioxidants in bakery products: A review. Crit. Rev. Food Sci. Nutr. 2009, 49, 1–27. [Google Scholar] [CrossRef]
- Lourenço, S.C.; Moldão-Martins, M.; Alves, V.D. Antioxidants of Natural Plant Origins: From Sources to Food Industry Applications. Molecules 2019, 24, 4132. [Google Scholar] [CrossRef]
- Sikora, E.; Cieślik, E.; Topolska, K. The sources of natural antioxidants. Acta Sci. Pol. Technol. Aliment. 2008, 7, 5–17. [Google Scholar]
- Jayaprakasha, G.; Singh, R.; Sakariah, K. Antioxidant activity of grape seed (Vitis vinifera) extracts on peroxidation models in vitro. Food Chem. 2001, 73, 285–290. [Google Scholar] [CrossRef]
- Carocho, M.; Barreiro, M.F.; Morales, P.; Ferreira, I.C.F.R. Adding molecules to food, pros and cons: A review on synthetic and natural food additives. Compr. Rev. Food Sci. Food Saf. 2014, 13, 377–399. [Google Scholar] [CrossRef]
- Massini, L.; Rico, D.; Martín-Diana, A.B.; Barry-Ryan, C. Quality Markers of Functional Tomato Juice with Added Apple Phenolic Antioxidants. Beverages 2016, 2, 4. [Google Scholar] [CrossRef]
- Berdahl, D.R.; Nahas, R.I.; Barren, J.P. Synthetic, and natural antioxidant additives in food stabilization: Current applications and future research. In Oxidation in Foods and Beverages and Antioxidant Applications; Decker, E.A., Elias, R.J., McClements, D.J., Eds.; Woodhead Publishing: Cambridge, UK, 2010; Volume 1, pp. 272–313. [Google Scholar]
- Liu, R.; Mabury, S.A. Synthetic Phenolic Antioxidants: A Review of Environmental Occurrence, Fate, Human Exposure, and Toxicity. Environ. Sci. Technol. 2020, 54, 11706–11719. [Google Scholar] [CrossRef]
- Dragovicuzelac, V.; Levaj, B.; Mrkic, V.; Bursac, D.; Boras, M. The Content of Polyphenols and Carotenoids in Three Apricot Cultivars Depending on Stage of Maturity and Geographical Region. Food Chem. 2007, 102, 966–975. [Google Scholar] [CrossRef]
- Abbas, M.; Saeed, F.; Anjum, F.M.; Afzaal, M.; Tufail, T.; Bashir, M.S.; Ishtiaq, A.; Hussain, S.; Suleria, H.A.R. Natural polyphenols: An overview. Int. J. Food Prop. 2017, 20, 1689–1699. [Google Scholar] [CrossRef]
- Fernández-Mar, M.I.; Mateos, R.; Garcıa-Parrilla, M.C.; Puertas, B.; Cantos-Villar, E. Bioactive compounds in wine: Resveratrol, hydroxytyrosol and melatonin: A review. Food Chem. 2012, 130, 797–813. [Google Scholar] [CrossRef]
- Gambini, J.; Inglés, M.; Olaso, G.; Lopez-Grueso, R.; Bonet-Costa, V.; Gimeno-Mallench, L.; Mas-Bargues, C.; Abdelaziz, K.M.; Gomez-Cabrera, M.C.; Vina, J.; et al. Properties of Resveratrol: In Vitro and In Vivo studies about metabolism, bioavailability, and biological effects in animal models and humans. Oxid. Med. Cell Longev. 2015, 837042. [Google Scholar] [CrossRef]
- Gea, A.; Sánchez-Tainta, A. Red Wine Moderate Consumption and at Mealtimes. In Prevention of Cardiovascular Disease through the Mediterranean Diet; Sánchez-Villegas, A., Sánchez-Tainta, A., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 151–157. [Google Scholar] [CrossRef]
- Paixão, N.; Perestrelo, R.; Marques, J.C.; Câmara, J.S. Relationship between antioxidant capacity and total phenolic content of red, rosé and white wines. Food Chem. 2007, 105, 204–214. [Google Scholar] [CrossRef]
- Fiori, L.; de Faveri, D.; Casazza, A.A.; Perego, P. Grape by-products: Extraction of polyphenolic compounds using supercritical CO2 and liquid organic solvent—A preliminary investigation Subproductos de la uva: Extraccion de compuestos polifenolicos usando CO2 supercritico y disolventes organicos liquidos—Una investigacion preliminar. CyTA J. Food 2009, 7, 163–171. [Google Scholar] [CrossRef]
- Obreque-Slier, E.; Peńa-Neira, A.; López-Solís, R.; Zamora-Marín, F.; Ricardo-da Silva, J.M.; Laureano, O. Comparative study of the phenolic composition of seeds and skins from Carménčre and Cabernet Sauvignon grape varieties (Vitis vinifera L.) during ripening. J. Agric Food Chem. 2010, 58, 3591–3599. [Google Scholar] [CrossRef]
- Giuffrč, A.M. HPLC-DAD detection of changes in phenol content of red berry skins during grape ripening. Eur. Food Res. Technol. 2013, 237, 555–564. [Google Scholar] [CrossRef]
- Budić-Leto, I.; Lovrić, T.; Pezo, I.; Gajdoš Kljusurić, J. Study of dynamics of polyphenol extraction during traditional and advanced maceration processes of the Babić grape variety. Food Technol. Biotechnol. 2005, 43, 47–53. [Google Scholar]
- Klenar, I.; Berović, M.; Wondra, M. Phenolic compounds from the fermentation of cultivars Cabernet Sauvignon and Merlot from the Slovenian coastal region. Food Technol. Biotechnol. 2004, 42, 11–17. [Google Scholar]
- Vilela, A. The Importance of Yeasts on Fermentation Quality and Human Health-Promoting Compounds. Fermentation 2019, 5, 46. [Google Scholar] [CrossRef]
- Coletta, A.; Berto, S.; Crupi, P.; Cravero, M.C.; Tamborra, P.; Antonacci, D.; Daniele, P.G.; Prenesti, E. Effect of viticulture practices on concentration of polyphenolic compounds and total antioxidant capacity of Southern Italy red wines. Food Chem. 2014, 152, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Cordova, A.C.; Sumpio, B.E. Polyphenols are medicine: Is it time to prescribe red wine for our patients? Int. J. Angiol. 2009, 18, 111–117. [Google Scholar] [CrossRef]
- Soni, R.P.; Katoch, M.; Kumar, A.; Ladohiya, R.; Verma, P. Tea: Production, Composition, Consumption and it’s Potential as an Antioxidant and Antimicrobial Agent. Int. J. Food. Ferment. Technol. 2015, 5, 95–106. [Google Scholar] [CrossRef]
- Vilela, A.; Pinto, T. Grape Infusions: The Flavor of Grapes and Health-Promoting Compounds in Your Tea Cup. Beverages 2019, 5, 48. [Google Scholar] [CrossRef]
- McCarthy, T.L.; Kerry, J.P.; Kerry, J.F.; Lynch, P.B.; Buckley, D.J. Evaluation of the antioxidant potential of natural food/plant extracts as compared with synthetic antioxidants and vitamin E in raw and cooked pork patties. Meat Sci. 2001, 58, 45–52. [Google Scholar] [CrossRef]
- Bártíková, H.; Skálová, L.; Valentova, K.; Matoušková, P.; Szotáková, B.; Martin, J.; Kvita, V.; Boušová, I. Efect of oral administration of green tea extract in various dosage schemes on oxidative stress status of mice in vivo. Acta Pharm. 2015, 65, 65–73. [Google Scholar] [CrossRef][Green Version]
- Cacho, J.I.; Campillo, N.; Viñas, P.; Hernández-Córdoba, M. Determination of synthetic phenolic antioxidants in soft drinks by stir-bar sorptive extraction coupled to gas chromatography-mass spectrometry. Food Addit. Contam. Part A 2015, 32, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Makahleh, A.; Saad, B.; Bari, M.F. Synthetic phenolics as antioxidants for food preservation. In Handbook of Antioxidants for Food Preservation; Shahidi, F., Ed.; Woodhead Publishing Ltd.: Cambridge, UK, 2015; pp. 51–78. [Google Scholar] [CrossRef]
- Furia, T.E. CRC Handbook of Food Additives, 2nd ed.; CRC Press: Boca Raton, FL, USA, 1980; Volume 2, p. 424. ISBN 0849305438. [Google Scholar]
- Nieva-Echevarría, B.; Manzanos, M.J.; Goicoechea, E.; Guillén, M.D. 2,6-Di-tert-butyl-hydroxytoluene and its metabolites in foods. Compr. Rev. Food Sci. Food Saf. 2015, 14, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Lanigan, R.S.; Yamarik, T.A. Final report on the safety assessment of BHT. Int. J. Toxicol. 2002, 21 (Suppl. 2), 19–94. [Google Scholar] [CrossRef]
- Dolatabadi, J.E.N.; Kashanian, S. A review on DNA interaction with synthetic phenolic food additives. Food Res. Int. 2010, 43, 1223–1230. [Google Scholar] [CrossRef]
- Lavagnini, I.; Urbani, A.; Magno, F. Overall calibration procedure via a statistically based matrix-comprehensive approach in the stir-bar sorptive extraction-thermal desorption-gas chromatography-mass spectrometry analysis of pesticide residues in fruit-based soft drinks. Talanta 2011, 83, 1754–1762. [Google Scholar] [CrossRef] [PubMed]
- Kashanian, S.; Dolatabadi, J.E.N. DNA binding studies of 2-tert-butylhydroquinone (TBHQ) food additive. Food Chem. 2009, 116, 743–747. [Google Scholar] [CrossRef]
- Zurita, J.L.; Jos, A.; Peso, A.D.; Salguero, M.; López-Artíguez, M.; Repetto, G. Ecotoxicological effects of the antioxidant additive propyl gallate in five aquatic systems. Water Res. 2007, 41, 2599–2611. [Google Scholar] [CrossRef]
- Iverson, F. In vivo studies on butylated hydroxyanisole. Food Chem. Toxicol. 1999, 37, 993–997. [Google Scholar] [CrossRef]
- Botterweck, A.A.M.; Verhagen, H.; Goldbohm, R.A.; Kleinjans, J.; van den Brandt, P.A. Intake of butylated hydroxyanisole and butylated hydroxytoluene and stomach cancer risk: Results from analyses in the Netherlands Cohort Study. Food Chem. Toxicol. 2000, 38, 599–605. [Google Scholar] [CrossRef]
- Singh, B.; Mense, S.M.; Remotti, F.; Liu, X.; Bhat, H.K. Antioxidant butylated hydroxyanisole inhibits estrogen-induced breast carcinogenesis in female ACI rats. J. Biochem. Mol. Toxicol. 2009, 23, 202–211. [Google Scholar] [CrossRef]
- Zhang, Y.; Choksi, S.; Liu, Z.-G. Butylated hydroxyanisole blocks the occurrence of tumor-associated macrophages in tobacco smoke carcinogen-induced lung tumorigenesis. Cancers 2013, 5, 1643–1654. [Google Scholar] [CrossRef]
- Liang, X.; Tang, Y.; Duan, L.; Cheng, S.; Luo, L.; Cao, X.; Tu, B. Adverse effect of sub-chronic exposure to benzo(a)pyrene and protective effect of butylated hydroxyanisole on learning and memory ability in male Sprague-Dawley rat. J. Toxicol. Sci. 2014, 39, 739–748. [Google Scholar] [CrossRef]
- Simon, R.A. Adverse reactions to food additives. Curr. Allergy Asthma Rep. 2003, 3, 62–66. [Google Scholar] [CrossRef]
- Horváthová, E.; Slameňová, D.; Bonatti, S.; Abbondandolo, A. Reduction of genotoxic effects of MNNG by butylated hydroxyanisole. Neoplasma 1999, 46, 356–362. [Google Scholar] [PubMed]
- Yang, X.; Song, W.; Liu, N.; Sun, Z.; Liu, R.; Liu, Q.S.; Zhou, Q.; Jiang, G. Synthetic phenolic antioxidants cause perturbation in steroidogenesis in vitro and in vivo. Environ. Sci. Technol. 2018, 52, 850–858. [Google Scholar] [CrossRef]
- Dassarma, B.; Nandi, D.K.; Gangopadhyay, S.; Samanta, S. Hepatoprotective effect of food preservatives (butylated hydroxyanisole, butylated hydroxytoluene) on carbon tetrachloride-induced hepatotoxicity in rat. Toxicol. Rep. 2018, 5, 31–37. [Google Scholar] [CrossRef]
- Pérez-Albaladejo, E.; Lacorte, S.; Porte, C. Differential toxicity of alkylphenols in JEG-3 human placental cells: Alteration of P450 aromatase and cell lipid composition. Toxicol. Sci. 2019, 167, 336–346. [Google Scholar] [CrossRef] [PubMed]
- Valentao, P.; Fernandes, E.; Carvalho, F.; Andrade, P.B.; Seabra, R.M.; Bastos, M.L. Antioxidative properties of cardoon (Cynara cardunculus L.) infusion against superoxide radical, hydroxyl radical, and hypochlorous acid. J. Agric. Food Chem. 2002, 50, 4989–4993. [Google Scholar] [CrossRef]
- Phenolic Antioxidant Market Research Report–Forecast to 2023. Market Research Future. Available online: https://www.marketresearchfuture.com/reports/phenolic-antioxidant-market-3937#answer1 (accessed on 13 December 2020).
- Our World in Data. Death Rate from Cardiovascular Disease and the Death Rate from Cancer Disease. 2017. Available online: https://ourworldindata.org/grapher/cardiovascular-disease-death-rates (accessed on 19 December 2020).
- World Heart Federation. The Costs of CVD. 2020. Available online: http://www.championadvocates.org/en/champion-advocates-programme/the-costs-of-cvd (accessed on 19 December 2020).
- United Nations, Department of Economic and Social Affairs Population Division. World Population Ageing Report 2015, ST/ESA/SER.A/390; United Nations, Department of Economic and Social Affairs Population Division: New York, NY, USA, 2015. [Google Scholar]
- Dauchet, L.; Amouyel, P.; Dallongeville, J. Fruits, vegetables and coronary heart disease. Nat. Rev. Cardiol. 2009, 6, 599–608. [Google Scholar] [CrossRef]
- Goff, D.C., Jr.; Lloyd-Jones, D.M.; Bennett, G.; Coady, S.; D’Agostino, R.B.; Gibbons, R.; Greenland, P.; Lackland, D.T.; Levy, D.; O’Donnell, C.J.; et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 2014, 129 (Suppl. 2), S49–S73. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ouyang, Y.; Liu, J.; Zhu, M.; Zhao, G.; Bao, W.; Hu, F.B. Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2014, 349, g4490. [Google Scholar] [CrossRef] [PubMed]
- Ricordi, C.; Garcia-Contreras, M.; Farnetti, S. Diet and inflammation: Possible effects on immunity, chronic diseases, and life span. J. Am. Coll. Nutr. 2015, 34, 10–13. [Google Scholar] [CrossRef]
- Lutz, M.; Fuentes, E.; Ávila, F.; Alarcón, M.; Palomo, I. Roles of Phenolic Compounds in the Reduction of Risk Factors of Cardiovascular Diseases. Molecules. 2019, 24, 366. [Google Scholar] [CrossRef]
- Palomo, I.G.; Marin, P.; Alarcón, M.; Gubelin, G.; Vinambre, X.; Mora, E.; Icaza, G. Patients with essential hypertension present higher levels of Se-selectin and Svcam-1 than normotensive volunteers. Clin. Exp. Hypertens 2003, 25, 517–523. [Google Scholar] [CrossRef]
- Palomo, I.G.; Gutiérrez, C.L.; Alarcón, M.L.; Jaramillo, J.C.; Segovia, F.M.; Leiva, E.M.; Mujica, V.E.; Icaza, G.N.; Diaz, N.S.; Moore-Carrasco, R. Increased concentration of plasminogen activator inhibitor-1 and fibrinogen in individuals with metabolic syndrome. Mol. Med. Rep. 2009, 2, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Palomo, I.G.; Jaramillo, J.C.; Alarcón, M.L.; Gutiérrez, C.L.; Moore-Carrasco, R.; Segovia, F.M.; Leiva, E.M.; Mujica, V.E.; Icaza, G.; Di, N.S. Increased concentrations of soluble vascular cell adhesion molecule-1 and soluble Cd40l in subjects with metabolic syndrome. Mol. Med. Rep. 2009, 2, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Palomo, I.G.; Moore-Carrasco, R.; Alarcón, M.L.; Rojas, A.; Espana, F.; Andrés, V.; González-Navarro, H. Pathophysiology of the proatherothrombotic state in the metabolic syndrome. Front. Biosci. 2010, 2, 194–208. [Google Scholar] [CrossRef]
- Ruggeri, Z.M. Mechanisms initiating platelet thrombus formation. Thromb. Haemost. 1997, 78, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Mukhtar, H. Green tea catechins: Anticancer effects and molecular targets. In Plant. Phenolics and Human Health; Fraga, C.G., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 1–4. [Google Scholar]
- Kadasi, A.; Maruniakova, N.; Kolesarova, A.; Stochmalova, A.; Alexa, R.; Grossmann, R.; Sirotkin, A.V. The effect of green tea extracts on steroidogenesis, proliferation, and apoptosis in ovarian granulosa cells. Endocr. Abstr. 2014, 34, 348. [Google Scholar] [CrossRef]
- Feskanich, D.; Ziegler, R.G.; Michaud, D.S.; Giovannucci, E.L.; Speizer, F.E.; Willett, W.C.; Coldits, G.A. Prospective study of fruit and vegetable consumption and risk of lung cancer among men and women. J. Nat. Cancer Ins. 2000, 92, 1812–1823. [Google Scholar] [CrossRef]
- Schuler, P. Natural antioxidants exploited commercially. In Food Antioxidants; Hudson, B.J.F., Ed.; Elsevier: London, UK, 1990; pp. 99–170. [Google Scholar]
- Rawal, G.; Yadav, S.; Nagayach, S. Phytosterols and the health. Med. Res. Chron. 2015, 2, 441–444. [Google Scholar]
- Aydar, E.F.; Tutuncu, S.; Ozcelik, B. Plant-based milk substitutes: Bioactive compounds, onventional and novel processes, bioavailability studies, and health effects. J. Funct. Foods 2020, 70, 103975. [Google Scholar] [CrossRef]
- Clifford, M.N.; Ramirez-Martinez, J.R. Phenols and caffeine in wet-processed coffee beans and coffee pulp. Food Chem. 1991, 40, 35–42. [Google Scholar] [CrossRef]
- Tresserra-Rimbau, A.; Medina-Remón, A.; Estruch, R.; Lamuela-Raventós, R.M. Chapter 42—Coffee Polyphenols and High Cardiovascular Risk Parameters. In Coffee in Health and Disease Prevention; Preedy, V.R., Ed.; Academic Press: Cambridge, MA, USA, 2015; pp. 387–394. [Google Scholar] [CrossRef]
- Yamagata, K. Do Coffee Polyphenols Have a Preventive Action on Metabolic Syndrome Associated Endothelial Dysfunctions? An Assessment of the Current Evidence. Antioxidants 2018, 7, 26. [Google Scholar] [CrossRef]
- Keen, C.L. Chocolate: Food as medicine/medicine as food. J. Am. Coll. Nutition 2001, 20, 436S–439S. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Rabaneda, F.; Jauregui, O.; Casals, I.; Andres-Lacueva, C.; Izquierdo-Pulido, M.R.; Lamuela-Raventos, M. Liquid chromatographic/electrospray ionization tandem mass spectrometric study of the phenolic composition of cocoa Theobroma cocao. J. Mass Spectrom. 2003, 38, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Ruzaidi, A.; Amin, I.; Nawalyah, A.G.; Hamid, M.; Faizul, H.A. The effect of Malaysian cocoa extract on glucose levels and lipid profiles in diabetic rats. J. Ethnopharmacol. 2005, 98, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Oracz, J.; Zyzelewicz, D.; Nebesny, E. The Content of Polyphenolic Compounds in Cocoa Beans (Theobroma cacao L.), Depending on Variety, Growing Region, and Processing Operations: A Review. Crit. Rev. Food Sci. Nutition 2015, 55, 1176–1192. [Google Scholar] [CrossRef]
- Arranz, S.; Chiva-Blanch, G.; Valderas-Martínez, P.; Medina-Remón, A.; Lamuela-Raventós, R.M.; Estruch, R. Wine, beer, alcohol and polyphenols on cardiovascular disease and cancer. Nutrients 2012, 4, 759–781. [Google Scholar] [CrossRef]
- Collin, S.; Jerkovic, V.; Bröhan, M.; Callemien, D. Polyphenols and Beer Quality. In Natural Products; Ramawat, K., Mérillon, J.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar] [CrossRef]
- Bianchini, F.; Vainio, H. Wine and resveratrol: Mechanisms of cancer prevention? Eur. J. Can. Prev. 2003, 12, 417–425. [Google Scholar] [CrossRef]
- Merkytė, V.; Longo, E.; Windisch, G.; Boselli, E. Phenolic Compounds as Markers of Wine Quality and Authenticity. Foods 2020, 9, 1785. [Google Scholar] [CrossRef]
- Carocho, M.; Ferreira, I.C. The role of phenolic compounds in the fight against cancer—A review. Anticancer Agents Med. Chem. 2013, 13, 1236–1258. [Google Scholar] [CrossRef] [PubMed]
- Ghasemzadeh, A.; Jaafar, H.Z. Profiling of phenolic compounds and their antioxidant and anticancer activities in pandan (Pandanus amaryllifolius Roxb.) extracts from different locations of Malaysia. BMC Complement. Altern Med. 2013, 13, 341. [Google Scholar] [CrossRef]
- De Oliveira, C.B.; Comunello, L.N.; Maciel, E.S.; Giubel, S.R.; Bruno, A.N.; Chiela, E.C.; Lenz, G.; Gnoatto, S.C.; Buffon, A.; Gosmann, G. The inhibitory effects of phenolic and terpenoid compounds from Baccharis trimera in Siha cells: Differences in their activity and mechanism of action. Molecules 2013, 18, 11022–11032. [Google Scholar] [CrossRef] [PubMed]
- Senawong, T.; Khaopha, S.; Misunaa, S.; Komaikula, J.; Senawonga, G.; Wongphakhama, P.; Yunchalard, S. Phenolic acid composition and anticancer activity against human cancer cell lines of the commercially available fermentation products of Houttuynia cordata. Sci. Asia 2014, 40, 420–427. [Google Scholar] [CrossRef]
- Chen, H.M.; Wu, Y.C.; Chia, Y.C.; Chang, F.R.; Hsu, H.K.; Hsieh, Y.C.; Chen, C.C.; Yuan, S.S. Gallic acid, a major component of Toona sinensis leaf extracts, contains a ROS-mediated anti-cancer activity in human prostate cancer cells. Cancer Lett. 2009, 286, 161–171. [Google Scholar] [CrossRef]
- Anantharaju, P.G.; Gowda, P.C.; Vimalambike, M.G.; Madhunapantula, S.V. An overview on the role of dietary phenolics for the treatment of cancers. Nutr. J. 2016, 15, 99. [Google Scholar] [CrossRef]
- Lee, Y.J.; Liao, P.H.; Chen, W.K.; Yang, C.Y. Preferential cytotoxicity of caffeic acid phenethyl ester analogues on oral cancer cells. Cancer Lett. 2000, 153, 51–56. [Google Scholar] [CrossRef]
- Aguilera, Y.; Martin-Cabrejas, M.A.; González de Mejia, E. Phenolic compounds in fruits and beverages consumed as part of the Mediterranean diet: Their role in prevention of chronic diseases. Phytochem. Rev. 2016, 15, 405–423. [Google Scholar] [CrossRef]
- Chen, M.; Meng, H.; Zhao, Y.; Chen, F.; Yu, S. Antioxidant and in vitro anticancer activities of phenolics isolated from sugar beet molasses. BMC Complement. Altern. Med. 2015, 15, 313. [Google Scholar] [CrossRef]
- Niero, E.L.; Machado-Santelli, G.M. Cinnamic acid induces apoptotic cell death and cytoskeleton disruption in human melanoma cells. J. Exp. Clin. Cancer Res. 2013, 32, 31. [Google Scholar] [CrossRef]
- Rajendra-Prasad, N.; Karthikeyan, A.; Karthikeyan, S.; Reddy, B.V. Inhibitory effect of caffeic acid on cancer cell proliferation by oxidative mechanism in human HT-1080 fibrosarcoma cell line. Mol. Cell Biochem. 2011, 349, 11–19. [Google Scholar] [CrossRef]
- Zhang, X.; Lin, D.; Jiang, R.; Li, H.; Wan, J.; Li, H. Ferulic acid exerts antitumor activity and inhibits metastasis in breast cancer cells by regulating epithelial to mesenchymal transition. Oncol. Rep. 2016, 36, 271–278. [Google Scholar] [CrossRef] [PubMed]
- WHO. World Health Report, 1998. Available online: https://www.who.int/whr/1998/media_centre/press_release/en/ (accessed on 19 December 2020).
- Ramassamy, C. Emerging role of polyphenolic compounds in the treatment of neurodegenerative diseases: A review of their intracellular targets. Eur. J. Pharmacol. 2006, 545, 51–64. [Google Scholar] [CrossRef] [PubMed]
- Checkoway, H.; Powers, K.; Smith-Weller, T.; Franklin, G.M.; Longstreth, W.T., Jr.; Swanson, P.D. Parkinson’s disease risks associated with cigarette smoking, alcohol consumption, and caffeine intake. Am. J. Epidemiol. 2002, 155, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Pan, T.; Fei, J.; Zhou, X.; Jankovic, J.; Le, W. Effects of green tea polyphenols on dopamine uptake and on MPP+-induced dopamine neuron injury. Life Sci. 2003, 72, 1073–1083. [Google Scholar] [CrossRef]
- Sackmann, C.; Hallbeck, M. Oligomeric amyloid-β induces early and widespread changes to the proteome in human iPSC-derived neurons. Sci. Rep. 2020, 10, 6538. [Google Scholar] [CrossRef]
- Bastianetto, S.; Yao, Z.X.; Papadopoulos, V.; Quirion, R. Neuroprotective effects of green and black teas and their catechin gallate esters against beta-amyloid-induced toxicity. Eur. J. Neurosci. 2006, 23, 55–64. [Google Scholar] [CrossRef]
- Choi, Y.T.; Jung, C.H.; Lee, S.R.; Bae, J.H.; Baek, W.K.; Suh, M.H.; Park, J.; Park, C.W.; Suh, S.I. The green tea polyphenol (−)-epigallocatechin gallate attenuates beta-amyloid-induced neurotoxicity in cultured hippocampal neurons. Life Sci. 2001, 70, 603–614. [Google Scholar] [CrossRef]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging. 2018, 13, 757–772. [Google Scholar] [CrossRef]
- Aryaeian, N.; Sedehi, S.K.; Arablou, T. Polyphenols and their effects on diabetes management: A review. Med. J. Islam Repub Iran. 2017, 31, 134. [Google Scholar] [CrossRef]
- Rosillo, M.A.; Alarcón-de-la-Lastra, C.; Sánchez-Hidalgo, M. An update on dietary phenolic compounds in the prevention and management of rheumatoid arthritis. Food Funct. 2016, 7, 2943–2969. [Google Scholar] [CrossRef] [PubMed]
- Llano, D.G.; Liu, H.; Khoo, K.; Moreno-Arribas, M.V.; Bartolomé, B. Some New Findings Regarding the Antiadhesive Activity of Cranberry Phenolic Compounds and Their Microbial-Derived Metabolites against Uropathogenic Bacteria. J. Agric. Food Chem. 2019, 67, 2166–2174. [Google Scholar] [CrossRef] [PubMed]
- Tarko, T.; Duda-Chodak, A.; Zajac, N. Digestion and absorption of phenolic compounds assessed by in vitro simulation methods. A review. Rocz Panstw Zakl Hig. 2013, 64, 79–84. [Google Scholar] [PubMed]
- Quatrin, A.; Rampelotto, C.; Pauletto, R.; Maurer, L.H.; Nichelle, S.N.; Klein, B.; Rodrigues, R.F.; Maróstica Junior, R.M.; Fonseca, B.S.; Menezes, C.R.; et al. Bioaccessibility and catabolism of phenolic compounds from jaboticaba (Myrciaria trunciflora) fruit peel during in vitro gastrointestinal digestion and colonic fermentation. J. Funct. Foods 2020, 65, 103714. [Google Scholar] [CrossRef]
- Danneskiold-Samsøe, N.B.; Barros, H.D.d.F.; Santos, R.; Bicas, J.L.; Cazarin, C.B.B.; Madsen, L.; Maróstica Júnior, M.R. Interplay between food and gut microbiota in health and disease. Food Res. Int. 2019, 115, 23–31. [Google Scholar] [CrossRef]
- Vitaglione, P.; Donnarumma, G.; Napolitano, A.; Galvano, F.; Gallo, A.; Scalfi, L.; Fogliano, V. Protocatechuic acid is the major human metabolite of cyanidin-glucosides. J. Nutition. 2007, 137, 2043–2048. [Google Scholar] [CrossRef] [PubMed]
- Gowd, V.; Karim, N.; Shishir, M.R.I.; Xie, L.; Chen, W. Dietary polyphenols to combat the metabolic diseases via altering gut microbiota. Trends Food Sci. Technol. 2019, 93, 81–93. [Google Scholar] [CrossRef]
- Vilela, A.; Cosme, F.; Inês, A. Wine and Non-Dairy Fermented Beverages: A Novel Source of Pro- and Prebiotics. Fermentation 2020, 6, 113. [Google Scholar] [CrossRef]
- Saarela, M. Probiotics as ingredients in functional beverages. In Functional and Speciality Beverage Technology; Paquin, P., Ed.; Woodhead Publishing Limited and CRC Press LLC: Cambridge, UK, 2009; pp. 55–70. [Google Scholar]
- Soccol, C.R.; De Dea Lindner, J.; Yamaguishi, C.T.; Spier, M.R.; Porto de Souza Vandenberghe, L.; Soccol, V.T. Probiotic nondairy beverages. In Handbook of Plant-Based Fermented Food and Beverage Technology; Hui, Y.H., Ed.; Taylor & Francis Group: Florence, SC, USA, 2012; pp. 707–728. [Google Scholar]
- Silva, S.; Veiga, M.; Costa, E.M.; Oliveira, A.L.S.; Madureira, A.R.; Pintado, M. Nanoencapsulation of Polyphenols towards Dairy Beverage Incorporation. Beverages 2018, 4, 61. [Google Scholar] [CrossRef]
- Teng, H.; Chen, L. Polyphenols and bioavailability: An update. Crit. Rev. Food Sci. Nutr. 2019, 59, 2040–2051. [Google Scholar] [CrossRef]


| Herbal Infusions | Total Phenolic Content (mg GAE/DW) | Total Flavonoid Content (mg CAE/g DW) |
|---|---|---|
| Lemon Balm | 133.3 ± 6.9 | 127.6 ± 2.7 |
| Linden | 33.9 ± 3.2 | 11.8 ± 0.1 |
| Passionflower | 14.9 ± 1.2 | 2.0 ± 0.3 |
| St. John’s Wort | 72.9 ± 2.5 | 45.7 ± 2.4 |
| Valerian | 9.8 ± 0.3 | 2.8 ± 0.5 |
| Black Tea | 21.5 ± 0.4 | 5.1 ± 0.0 |
| Green Tea | 61.0 ± 1.3 | 2.9 ± 0.2 |
| Red Tea | 30.0 ± 1.6 | 14.5 ± 0.6 |
| Boldo | 62.1 ± 0.2 | 51.0 ± 3.2 |
| Chamomile | 6.0 ± 0.4 | 3.7 ± 0.1 |
| Fennel | 19.9 ± 0.8 | 6.4 ± 0.6 |
| Green Anise | 15.9 ± 0.4 | 9.0 ± 0.2 |
| Pennyroyal | 16.2 ± 0.7 | 14.0 ± 0.7 |
| Senna | 6.8 ± 0.1 | 3.5 ± 0.1 |
| Hawthorn | 29.2 ± 1.0 | 17.8 ± 0.5 |
| Horsetail | 29.9 ± 1.1 | 2.0 ± 0.1 |
| Olive Tree | 26.7 ± 0.9 | 18.4 ± 2.2 |
| Rosemary | 47.4 ± 1.3 | 41.5 ± 0.9 |
| Thyme | 40.3 ± 0.7 | 35.9 ± 1.6 |
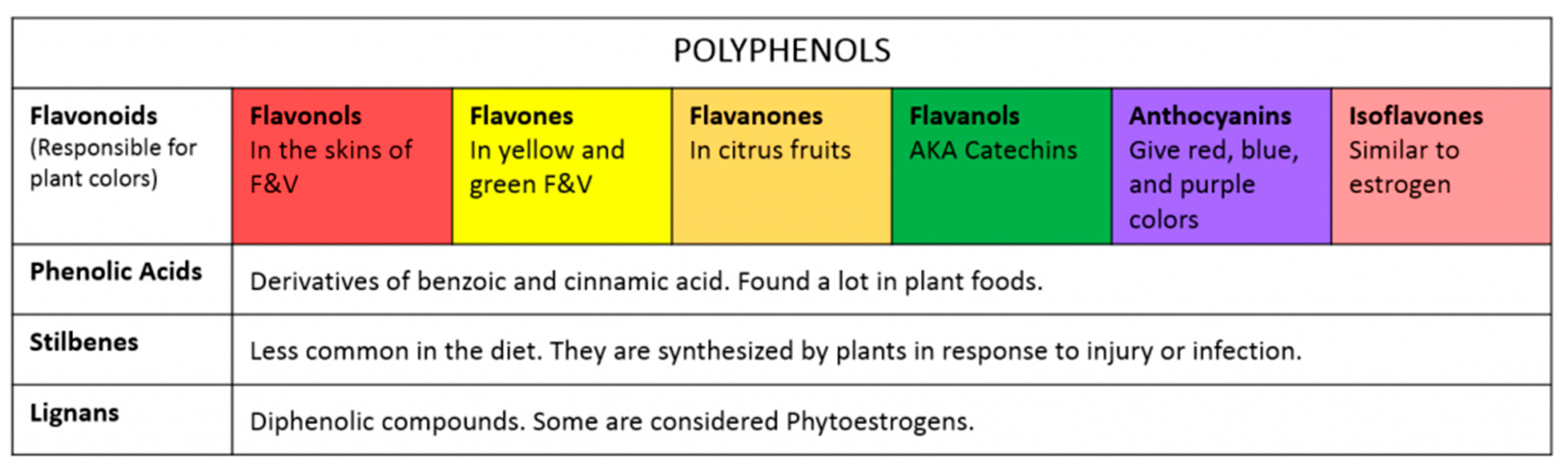
| Flavonoid | Name | Dietary Source |
|---|---|---|
| Flavone | Chrysin | Fruit skins |
| Apigenin | Parsley, celery | |
| Flavanone | Naringin, naringenin, taxifolin, eriodictyol, hesperidin, and isosakuranetin | Citrus, grapefruit, lemons, and oranges |
| Flavonol | Kaempferol | Leek, broccoli, endives, grapefruit, black tea |
| Quercetin | Onion, lettuce, broccoli, tomato, tea, berries, apples, olive oil, cranberry | |
| Rutin | Buckwheat, citrus, red pepper, red wine, tomato skin | |
| Flavononol | Engeletin and astilbin | White grape skin |
| Genistin | Soybean | |
| Taxifolin | Fruits | |
| Isoflavone | Genistein, daidzin, and daidzein | Soybean |
| Flavanol | (+)-Catechin, (+)-Gallocatechin, (−)-Epicatechin, (−)-Epigallocatechin, (−)-Epicatechin gallate, (−)-Epigallocatechin gallate | Tea |
| Anthocyanidin | Epigenidin | Stored fruits |
| Cyanidin | Cherry, raspberry, strawberry, grapes | |
| Delphinium and pelargonidin | Dark fruits |
| Meal | Foods | |
|---|---|---|
| Breakfast | Bread topped with grated fresh tomato, crumbled feta, dried oregano, and drizzled with extra virgin olive oil Herbal tea or boiled Greek coffee |  |
| Lunch (main meal) | Mediterranean penne with courgettes and eggplant, mozzarella cheese, olives, basil leaves. Seasoned with olive oil, and accompanied with red wine |  |
| Dinner | Ratatouille with tomato sauce or kale risotto with pumpkin or fish, rice, olive oil, and natural spices White wine |  |
| Snacks | Water Fresh fruit in season or fruit juices Nuts Dried fruit, e.g., figs, raisins |  |
| Fruit | Total Phenols (µmol/g) (Dry Weight) | Total PAOXI × 10−3 (a) (Dry Weight) |
|---|---|---|
| Apple | 34.1 | 110 |
| Avocado | 12.7 | 60.5 |
| Banana | 42.3 | 108 |
| Blueberry | 62.0 | 273 |
| Cantaloupe | 8.1 | 32.4 |
| Cherry | 52.3 | 523 |
| Cranberry | 158.8 | 212 |
| Grape (white) | 52.3 | 262 |
| Grape (red) | 63.7 | 351 |
| Grapefruit | 7.5 | 39.5 |
| Lemon | 19.6 | 67.6 |
| Melon (honeydew) | 11.4 | 36.8 |
| Nectarine | 12.3 | 64.7 |
| Orange | 18.9 | 55.6 |
| Peach | 27.9 | 60.7 |
| Pear | 41.4 | 81.2 |
| Pineapple | 11.9 | 44.1 |
| Plum | 58.2 | 116 |
| Strawberry | 72.3 | 603 |
| Watermelon | 19.5 | 44.3 |
| Beverages | Main Phenolics | Indicated Bioactivities | Ref. |
|---|---|---|---|
| Tea and infusions (Camelia sinensis and other plants)  | Pure catechins, ((−)-epicatechin (EC), (−)-epicatechin gallate (ECG), (−)-epigallocatechin (EGC), and (−)-epigallocatechin gallate (EGCG)), and phenolic acids, thearubigins, theaflavins, theasinensins, and polyhydroxylated flavan-3-ols. | Potential for use of EGCG in human cancer prevention and treatment. Induction of the suppression of tumor cell proliferation, stimulation of apoptosis, promotion of progestogen, and changes in testosterone release. Reduce the risk of Parkinson’s disease. | [53,86,123,124] |
| Fruit juices (Tree fruits, citrus fruits, and dark fruits)  | Polyphenols, most of which are flavonoids, present mainly in the ester and glycoside forms. | Lower the incidence of degenerative diseases such as cancer, arthritis, arteriosclerosis, heart disease, inflammation, brain dysfunction, and acceleration of the aging process. | [53,125] |
| Plant-based milk substitutes (Oilseed and nut milk substitutes) (1)  | Tocopherol (mainly α-tocopherol) and, to a lesser extent, γ-tocopherol. Isoflavone glycosides and their derivatives. Phenolic acids, namely syringic, vanillic, caffeic, ferulic, p-coumaric, and p-hydroxybenzoic. | Help to support the cardiovascular system. Lowers cholesterol levels and can reduce menopause symptoms due to their elevated content of isoflavones, which are a class of chemical known as “phytoestrogens”. Their high antioxidant content makes them preventative agents against ovarian, breast, stomach, prostate, and lung cancer. | [53,126,127,128] |
| Coffee (Coffea arabica and Coffea canephora)  | Catechol is the predominant volatile phenolic compound found in coffee (after roasting), followed by 4-ethylguaiacol, 4-ethyl catechol, pyrogallol, quinol, and 4-vinyl catechol. Catechin, rutin, ferulic acid, o-dihydroxybenzene, chlorogenic acid, caffeic acid, gallic acid, and protocatechuic acid. | Chlorogenic acid has potential cardiovascular benefits related to the lowering of blood pressure, endothelial function improvement, low-density lipoprotein cholesterol oxidation, and nitric oxide bioavailability prevention of metabolic syndrome, including vascular endothelial function impairment, obesity, and diabetes. | [53,129,130,131] |
| Cocoa drinks (Cocoa liquor, cocoa emulsions)  | Epicatechin, gallocatechin, and epigallocatechin. Phenolics, procyanins, anthocyanins, and flavone and flavonol glycosides such as luteolin-7-O-glucoside and quercetin-3-O-arabinoside. | Consumption of cocoa reduces the risk of cardiovascular disease. Extracts prepared from cocoa powder and cocoa beans were shown to exhibit antihyperglycemic effects. | [132,133,134,135] |
| Beer (Composition of phenolic compounds depends on the brand and country of origin) (2)  | Gallic acid, protocatechuic acid, (+)-catechin, vanillic acid, caffeic acid, syringic acid, (−)-epicatechin, p-coumaric acid, and ferulic acid. The major free phenolic acids in beers are m-, p- and o-coumaric and ferulic. Sinapic, vanillic, chlorogenic, homovanillic, p-hydroxybenzoic, 2,6- and 3,5-dihydroxybenzoic, syringic, gallic, protocatechuic, and caffeic acids. | Positive effect on plasma lipid profile and plasma antioxidant capacity. Increase bile volume and bile acid concentrations. | [136,137] |
| Wine (Depends on the grape variety, the winemaking process, wine-fermenting yeast strain, among others)  | The most common flavonoids in wine are flavonols (quercetin, kaempferol, and myricetin), flavan-3-ols (catechin and epicatechin), tannins, and anthocyanins (cyanin). Nonflavonoids comprise stilbenes, hydroxycinnamic acids, and benzoic acids. | Decrease the levels of lipid peroxidation and a lower the incidence of certain types of cancer. The moderate consumption of red wine has a relatively higher benefit in the prevention of atherosclerosis and coronary heart disease (CHD). Inhibit low-density lipoprotein (LDL) oxidation, increase antioxidative capacity, and modulate vascular function by inducing vasodilation through increased production of nitric oxide (NO). | [82,138,139] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinto, T.; Vilela, A. Healthy Drinks with Lovely Colors: Phenolic Compounds as Constituents of Functional Beverages. Beverages 2021, 7, 12. https://doi.org/10.3390/beverages7010012
Pinto T, Vilela A. Healthy Drinks with Lovely Colors: Phenolic Compounds as Constituents of Functional Beverages. Beverages. 2021; 7(1):12. https://doi.org/10.3390/beverages7010012
Chicago/Turabian StylePinto, Teresa, and Alice Vilela. 2021. "Healthy Drinks with Lovely Colors: Phenolic Compounds as Constituents of Functional Beverages" Beverages 7, no. 1: 12. https://doi.org/10.3390/beverages7010012
APA StylePinto, T., & Vilela, A. (2021). Healthy Drinks with Lovely Colors: Phenolic Compounds as Constituents of Functional Beverages. Beverages, 7(1), 12. https://doi.org/10.3390/beverages7010012