Mycotoxin Contamination of Beverages Obtained from Tropical Crops
Abstract
:1. Introduction
1.1. General Remarks for Mycotoxins
1.2. Consumption Data for Tropical Beverages and Related Products
1.3. Technological Processing and Mycotoxin Reduction
2. Common Drinks Produced in the Tropics Subjected to Mycotoxin Contamination
2.1. Tea (Including Mate)
2.1.1. Mycotoxin Contamination and Fungal Charge Found in Tea
2.1.2. Mycotoxin Transference Rate from Herbs to Infusions
2.1.3. Yerba Mate
2.1.4. The Relationship between Mycotoxins and Bioactive Compounds Found in Tea
2.2. Nutmilk and Similar Beverages
2.3. Fermented Beverages
2.4. Coffee
2.5. Chocolate Beverage
2.6. Fruits and Fruit Drinks
2.6.1. Mycotoxin in Pineapple and Pineapple Juice
2.6.2. Citrus Fruit Juices
2.6.3. Tomato Juice
2.6.4. Mango Juice
2.6.5. Other Cases
3. Methodological Aspects and Approaches for the Determination of Mycotoxins in Selected Matrices
3.1. Approaches in Sample Preparation
3.2. High-Throughput Multi-Analyte LC-Based Techniques
3.2.1. Coupled with MS Detectors
3.2.2. Coupled with UV and FLD Detectors
3.3. Non-Chromatographic Multiple Mycotoxin Analysis
4. Mycotoxin Risk Assessment and Integrated Management Approaches
4.1. Masked and Hidden Mycotoxins
4.2. Mycotoxin Risk and Climate Change
4.3. Multidisciplinary Approaches to Reduce Mycotoxin Contamination
4.4. Mycotoxin Contamination and the Food Chain
4.5. Selected Health Issues Related to Mycotoxin Exposure
5. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- García-Moraleja, A.; Font, G.; Mañes, J.; Ferrer, E. Analysis of mycotoxins in coffee and risk assessment in Spanish adolescents and adults. Food Chem. Toxicol. 2015, 86, 225–233. [Google Scholar]
- García-Moraleja, A.; Font, G.; Mañes, J.; Ferrer, E. Simultaneous determination of mycotoxin in commercial coffee. Food Control 2015, 57, 282–292. [Google Scholar] [CrossRef]
- Ismaiel, A.A.; Papenbrock, J. Mycotoxins: Producing fungi and mechanisms of phytotoxicity. Agriculture 2015, 5, 492–537. [Google Scholar] [CrossRef] [Green Version]
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vecchio, A.; Mineo, V.; Planeta, D. Ochratoxin A in instant coffee in Italy. Food Control 2012, 28, 220–223. [Google Scholar] [CrossRef]
- Fernández-Cruz, M.L.; Mansilla, M.L.; Tadeo, J.L. Mycotoxins in fruits and their processed products: Analysis, occurrence and health implications. J. Adv. Res. 2010, 1, 113–122. [Google Scholar] [CrossRef]
- Mandappa, I.M.; Basavaraj, K.; Manonmani, H.K. Analysis of Mycotoxins in Fruit Juices. In Fruit Juices Extraction, Composition, Quality and Analysis; Rajauria, G., Tiwari, B.K., Eds.; Academic Press: Massachusetts, MA, USA, 2018; pp. 763–777. ISBN 978-0-12-802230-6. [Google Scholar]
- Chen, W.; Li, C.; Zhang, B.; Zhou, Z.; Shen, Y.; Liao, X.; Yanj, J.; Wang, Y.; Li, Y.; Shen, X.L. Advances in biodetoxification of ochratoxin A—A review of the past five decades. Front. Microbiol. 2018, 9, 1386. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.; Fan, Y.; Zhao, L. Review on biological degradation of mycotoxins. Anim. Nutr. 2016, 2, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Zain, M.E. Impact of mycotoxins on humans and animals. J. Saudi Chem. Soc. 2011, 15, 129–144. [Google Scholar] [CrossRef]
- Iheshiulor, O.O.M.; Esonu, B.O.; Chuwuka, O.K.; Omede, A.A.; Okoli, I.C.; Ogbuewu, I.P. Effects of mycotoxins in animal nutrition: A review. Asian J. Anim. Sci. 2011, 5, 19–33. [Google Scholar] [CrossRef]
- Antonissen, G.; Martel, A.; Pasmans, F.; Ducatelle, R.; Verbrugghe, E.; Vandenbroucke, V.; Li, S.; Haesebrouck, F.; Van Immerseel, F.; Croubels, S. The impact of Fusarium mycotoxins on human and animal host susceptibility to infectious diseases. Toxins 2014, 28, 430–452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bezerra da Rocha, M.E.; Freire, F.D.C.O.; Maia, F.E.F.; Guedes, M.I.F.; Rondina, D. Mycotoxins and their effects on human and animal health. Food Control 2014, 36, 159–165. [Google Scholar] [CrossRef]
- Bui-Klimke, T.; Wu, F. Evaluating weight of evidence in the mystery of Balkan endemic nephropathy. Risk Anal. 2014, 3, 1688–1705. [Google Scholar] [CrossRef] [PubMed]
- Galarce-Bustos, O.; Alvarado, M.; Vega, M.; Aranda, M. Occurrence of ochratoxin A in roasted and instant coffees in Chilean market. Food Control 2014, 46, 102–107. [Google Scholar] [CrossRef]
- Alshannaq, A.; Yu, J.-H. Occurrence, toxicity, and analysis of major mycotoxins in food. Int. J. Environ. Res. Public Health 2017, 14, 632. [Google Scholar] [CrossRef] [PubMed]
- Pal, M. Are mycotoxins silent killers of humans and animals? J. Exp. Food Chem. 2017, 3, e110. [Google Scholar] [CrossRef]
- Ezekiel, C.N.; Ayeni, K.I.; Misihariabgwi, J.M.; Somorin, Y.M.; Chibuzor-Onyema, I.E.; Oyedele, O.A.; Abia, W.A.; Sulyok, M.; Shephard, G.S.; Krska, R. Traditionally processed beverages in Africa: A review of the mycotoxin occurrence patterns and exposure assessment. Compr. Rev. Food Sci. Food Saf. 2018, 17, 334–351. [Google Scholar] [CrossRef]
- Aswani, Y.V.V.; Renuka, R.M.; Bodaiah, B.; Mangamu, U.K.; Vijaya, L.M.; Poda, S. Mycotoxin strategies: Impact on global health and wealth. Pharm. Anal. Acta 2016, 7, 498. [Google Scholar]
- Mitchell, N.J.; Bowers, E.; Hurburgh, C.; Wu, F. Potential economic losses to the USA corn industry from aflatoxin contamination. Food Addit. Contam. Part A 2016, 33, 540–550. [Google Scholar] [CrossRef] [PubMed]
- Rico-Sole, R. Economic impact of mycotoxins in nuts and dried fruit chain. Acta Hort. 2012, 963, 155–172. [Google Scholar] [CrossRef]
- Kouadio, J.H.; Lattazio, V.M.T.; Ouattara, D.; Kouakou, B.; Visconti, A. Assessment of mycotoxin exposure in Côte d’ivoire (Ivory Coast) through multi-biomarker analysis and possible correlation with food consumption patterns. Toxins 2014, 21, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Grigg, D. The worlds of tea and coffee: Patterns of consumption. GeoJournal 2002, 57, 283–294. [Google Scholar] [CrossRef]
- Rezaee, E.; Mirlohi, M.; Hassanzadeh, A.; Fallah, A. Factors affecting tea consumption pattern in an urban society in Isfahan, Iran. J. Educ. Health Promot. 2016, 5, 13. [Google Scholar] [PubMed] [Green Version]
- Food and Agriculture Organization of the United Nations (FAO); Intergovernmental Group on Tea. Intersessional Meeting. Report of the Working Group on Global Tea Market Analysis and Promotion. 2017. Available online: http://www.fao.org/fileadmin/templates/est/COMM_MARKETS_MONITORING/Tea/Intersessional_2017/ISM-17-3-GlobalMktAnalysis_Promotion.docx (accessed on 19 September 2018).
- Instituto del Café de Costa Rica (ICAFE). Informe sobre la Actividad Cafetalera de Costa Rica. Preparado en el Instituto del Café de Costa Rica para los Delegados al XLVI Congreso Nacional Cafetalero Ordinario. 2017. Available online: http://www.icafe.cr/wp-content/uploads/informacion_mercado/informes_actividad/actual/InformeActividadCafetalera.pdf.pdf (accessed on 21 September 2018).
- Maurage, P.; Heeren, A.; Pesenti, M. Does chocolate consumption really boost Nobel award chances? The peril of over-interpreting correlations in health studies. J. Nutr. 2013, 143, 931–933. [Google Scholar] [CrossRef] [PubMed]
- Noble, M.D. Chocolate and the consumption of forests: A cross-national examination of ecologically unequal exchange in cocoa exports. J. World Syst. Res. 2017, 23, 236–268. [Google Scholar] [CrossRef]
- Katz, D.L.; Doughty, K.; Ali, A. Cocoa and chocolate in human health and disease. Antioxid. Redox Signal. 2011, 15, 2779–2811. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations (FAO). Medium-Term Prospects for Raw Materials, Horticulture and Tropical Products; FAO: Rome, Italy, 2016; pp. 3–26. ISBN 978-92-5-109202-6. [Google Scholar]
- Duffey, K.J.; Popkin, B.M. Shifts in patterns and consumption of beverages between 1965 and 2002. Obesity 2007, 15, 2739–2747. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.W.; Ostrowski, J.D.; Li, K.-P. Trends in added sugars from packaged beverages available and purchased by US households, 2007–2012. Am. J. Clin. Nutr. 2017, 106, 179–188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neves, M.F.; Trombin, V.G.; Lopes, F.F.; Kalaki, R.; Milan, P. World consumption of fruit juices, nectars, and still drinks. In The Orange Juice Business: A Brazilian Perspective; Neves, M.F., Trombin, V.G., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2011; p. 119. ISBN 978-90-8686-739-4. [Google Scholar]
- Reinhardt, A.; Rodríguez, L.V. Industrial processing of pineapple—Trends and perspectives. Acta Hortic. 2009, 822, 323–328. [Google Scholar] [CrossRef]
- Drusch, S.; Ragab, W. Mycotoxins in fruits, fruit juices, and dried fruits. J. Food Prot. 2003, 66, 1514–1527. [Google Scholar] [CrossRef] [PubMed]
- Hasan, H.A.H. Patulin and aflatoxin in brown rot lesions of apple fruits and their regulation. World J. Microbiol. Biotechnol. 2000, 16, 607–612. [Google Scholar] [CrossRef]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef] [PubMed]
- Manda, P.; Dano, D.S.; Kouadio, J.H.; Diakite, A.; Sangare-Tigori, B.; Ezoulin, M.J.M.; Soumahoro, A.; Dembele, A.; Fourny, G. Impact of industrial treatments on ochratoxin A content in artificially contaminated cocoa beans. Food Addit. Contam. A 2009, 26, 1081–1088. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.; Zhou, T.; Koutchma, T.; Wu, F.; Warriner, K. High hydrostatic pressure assisted degradation of patulin in fruit and vegetable juice blends. Food Control 2016, 62, 237–242. [Google Scholar] [CrossRef]
- Christ, D.; Savi, G.D.; Scussel, V.M. Effectiveness of ozone gas application methods against combined multi-contaminants in food. Food Public Health 2017, 7, 51–58. [Google Scholar]
- Ghanem, I.; Orfi, M.; Shamma, M. Effect of gamma radiation on the inactivation of aflatoxin B1 in food and feed crops. Braz. J. Microbiol. 2008, 39, 787–791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Copetti, M.V.; Iamanaka, B.T.; Pitt, J.I.; Taniwaki, M.H. Fungi and mycotoxins in cocoa: From farm to chocolate. Int. J. Food Microbiol. 2014, 178, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Van de Perre, E.; Jacxsens, L.; Liu, C.; Devlieghere, F.; De Meulenaer, B. Climate impact on Alternaria moulds and their mycotoxins in fresh produce: The case of the tomato chain. Food Res. Int. 2015, 68, 41–46. [Google Scholar] [CrossRef]
- Laitila, A. Toxigenic fungi and mycotoxins in the barley-to-beer chain. In Brewing Microbiology Managing Microbes, Ensuring Quality and Valorising Waste; Hill, A., Ed.; Elsevier Academic Press: Amsterdam, The Netherlands, 2015; pp. 107–139. ISBN 978-1-78242-331-7. [Google Scholar]
- Himery, N.; Vasseur, V.; Coton, M.; Mournier, J.; Jany, J.-L.; Barbier, G.; Coton, E. Filamentous fungi and mycotoxins in cheese: A review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 437–456. [Google Scholar] [CrossRef]
- Garnier, L.; Valence, F.; Mournier, J. Diversity and control of spoilage fungi in dairy products: An update. Microorganism 2017, 5, 42. [Google Scholar] [CrossRef] [PubMed]
- Comisión Económica para América Latina y el Caribe (CEPAL). Perspectivas de la Agricultura y del Desarrollo Rural en las Américas: Una Mirada Hacia América Latina y el Caribe 2015–2016; Organización de las Naciones Unidas para la Agricultura y la Alimentación (FAO): Rome, Italy; Instituto Interamericano de Cooperación para la Agricultura (IICA): San José, Costa Rica, 2016; pp. 63–88. ISBN 978-92-9248-577-1. [Google Scholar]
- Rodríguez, D.I.; Anríquez, G.; Riveros, J.L. Food security and livestock: The case of Latin America and the Caribbean. Cienc. Investig. Agrar. 2016, 43, 5–15. [Google Scholar] [CrossRef]
- Chavan, R.S.; Shraddha, R.C.; Kumar, A.; Nalawade, T. Whey based beverage: Its functionality, formulations, health benefits and applications. J. Food Process. Technol. 2015, 6, 1. [Google Scholar]
- Pereira, C.; Henriques, M.; Gomes, D.; Gomez-Zavaglia, A.; de Antoni, G. Novel functional whey-based drinks with great potential in the dairy industry. Food Technol. Biotechnol. 2015, 53, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Farah, J.S.; Araujo, C.B.; Melo, L. Analysis of yoghurts’, whey-based beverages’ and fermented milks’ labels and differences on their sensory profiles and acceptance. Int. Dairy J. 2017, 68, 17–22. [Google Scholar] [CrossRef]
- Patel, R. Technology for Carbonated Lemon Whey Beverage. Res. Rev. J. Food Dairy Technol. 2017, 5, 30–37. [Google Scholar]
- Janiaski, D.R.; Pimentel, T.C.; Cruz, A.G.; Prudencio, S.H. Strawberry-flavored yogurts and whey beverages: What is the sensory profile of the ideal product? J. Dairy Sci. 2016, 99, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Haas, D.; Pfeifer, B.; Reiterich, C.; Partenheimer, R.; Reck, B.; Buzina, W. Identification and quantification of fungi and mycotoxins from Pu-erh tea. Int. J. Food Microbiol. 2013, 166, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.; Marín, S.; Sanchis, V.; Ramos, A.J. Mycotoxin in medicinal/aromatic herbs—A review. Boletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas 2013, 12, 119–142. [Google Scholar]
- Chang, K. World Tea Production and Trade: Current and Future Development; FAO: Rome, Italy, 2015; pp. 1–13. [Google Scholar]
- Hilal, M.I.M.; Mubarak, K.M. International tea marketing and need for reviving Sri Lankan tea industry. J. Manag. 2013, 9, 25–38. [Google Scholar] [CrossRef]
- Zhao, J.; Ge, L.Y.; Xiong, W.; Leong, F.; Huang, L.Q.; Li, S.P. Advanced development in phytochemicals analysis of medicine and food dual purposes plants used in China (2011–2014). J. Chromatogr. A 2016, 1428, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Trucksess, M.W.; Scott, P.M. Mycotoxins in botanicals and dried fruits: A review. Food Addit. Contam. Part A 2008, 25, 181–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matei, S.; Szakacs, A.; Macri, A. Medicinal plants used for tea, mycological and mycotoxicological potential. Bull. UASVM Vet. Med. 2015, 72, 352–356. [Google Scholar] [CrossRef]
- Wu, J.-Y.; Yang, G.-Y.; Chen, J.-L.; Li, W.-X.; Li, J.-T.; Fu, C.-X.; Jiang, G.-F.; Zhu, W. Investigation for Pu-Erh tea contamination caused by mycotoxins in a tea market in Guangzhou. J. Basic Appl. Sci. 2014, 10, 349–356. [Google Scholar]
- Ning, J.; Sun, J.; Li, S.; Sheng, M.; Zhang, Z. Classification of five Chinese tea categories with different fermentation degrees using visible and near infrared hyperspectral imaging. Int. J. Food Prop. 2017, 20, 1515–1520. [Google Scholar] [CrossRef]
- Li, Q.; Huang, J.; Li, Y.; Zhang, Y.; Luo, Y.; Chen, Y.; Lin, H.; Wang, K.; Liu, Z. Fungal community succession and major components change during manufacturing process of Fu brick tea. Sci. Rep. 2017, 7, 6947. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Chai, S.; Li, Y.; Huang, J.; Luo, Y.; Xiao, L.; Liu, Z. Biochemical components associated with microbial community shift during the pile-fermentation of primary dark tea. Front. Microbiol. 2018, 9, 1509. [Google Scholar] [CrossRef] [PubMed]
- Khanongnuch, C.; Unban, K.; Kanpiengjai, A.; Saenjum, C. Recent research advances and ethno-botanical history of miang, a traditional fermented tea (Camellia sinensis var. assamica) of northern Thailand. J. Ethn. Foods 2017, 4, 135–144. [Google Scholar] [CrossRef]
- Han, T.; Aye, K.N. The legend of laphet: A Myanmar fermented tea leaf. J. Ethn. Foods 2015, 2, 173–178. [Google Scholar] [CrossRef]
- Horie, M.; Nara, K.; Sugino, S.; Umeno, A.; Yoshida, Y. Comparison of antioxidant activities among four kinds of Japanese traditional fermented tea. Feed Sci. Nutr. 2017, 5, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Siddique, N.A.; Mujeeb, M.; Ahmad, S.; Panda, B.P.; Makhmoor, M. Determination of aflatoxins in medicinal plants by high-performance liquid chromatography–tandem mass spectrometry. J. Pharm. Pharm. Sci. 2013, 16, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Blanco, J.; Araya, E.B.; Granados-Chinchilla, F.; Gutiérrez, A.L. Evaluation of the quality of natural products for the preparation of chamomile and mint tea in Costa Rica. World J. Pharmacol. Res. 2017, 6523, 44–55. [Google Scholar]
- Kong, W.-J.; Shen, H.-H.; Zhang, X.-F.; Yang, X.-L.; Qiu, F.; Ou-yang, Z.; Yang, M.-H. Analysis of zearalenone and α-zearalenol in 100 foods and medicinal plants determined by HPLC-FLD and positive confirmation by LC-MS-MS. J. Sci. Food Agric. 2013, 93, 1584–1590. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.; Marín, S.; Sanchis, V.; Ramos, A.J. Screening of mycotoxin multicontamination in medicinal and aromatic herbs sampled in Spain. J. Sci. Food Agric. 2009, 89, 1802–1807. [Google Scholar] [CrossRef]
- Malir, F.; Ostry, V.; Pfohl-Leszkowicz, A.; Toman, J.; Bazin, I.; Roubal, T. Transfer of ochratoxin A into tea and coffee beverages. Toxins 2014, 6, 3438–3453. [Google Scholar] [CrossRef] [PubMed]
- Nian, Y.; Wang, H.; Ying, G.; Yang, M.; Wang, Z.; Kong, W.; Yang, S. Transfer rates of aflatoxins from herbal medicines to decoctions determined by an optimized high-performance liquid chromatography with fluorescence detection method. J. Pharm. Pharmacol. 2018, 70, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Alvarenga, A.A.A.; López, I.P.P.; Abraham, C.M.R.; Caballero, Y.M.R.; Popoff, C.T.; Vázquez, L.; Arrua, J.M.M. Presencia de hongos filamentosos en yerba mate compuesta y eficiencia de medios de cultivo para el aislamiento de Aspergillus. Investig. Agrar. 2016, 18, 49–55. [Google Scholar] [CrossRef]
- Castrillo, M.L.; Jerke, G.; Horianksi, M.A. Detección de la producción de ocratoxina A por cepas de Aspergillus sección Nigri aisladas de yerba mate compuesta. Rev. Mex. Micol. 2014, 40, 1–6. [Google Scholar]
- Yang, Z.; Wang, H.; Ying, G.; Yang, M.; Nian, Y.; Liu, J.; Kong, W. Relationship of mycotoxins accumulation and bioactive components variation in ginger after fungal inoculation. Front. Pharmacol. 2017, 8, 331. [Google Scholar] [CrossRef] [PubMed]
- Jaswal, P.; Kumar, D. Mycobiota and natural incidence of aflatoxins, ochratoxin A, and citrinin in Indian spices confirmed by LC-MS/MS. Int. J. Microbiol. 2015, 2015, 242486. [Google Scholar] [CrossRef] [PubMed]
- Kalaiselvi, P.; Rajashree, K.; Priya, L.B.; Padma, V.V. Cytoprotective effect of epigallocatechin-3-gallate against deoxynivalenol-induced toxicity through anti-oxidative and anti-inflammatory mechanisms in HT-29 cells. Food Chem. Toxicol. 2013, 56, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, K.-I.; Kinoshita, M.; Kamata, Y.; Minai, Y.; Sugita-Konishi, Y. (−)-Epigallocatechin gallate suppresses the cytotoxicity induced by trichothecene mycotoxins in mouse cultural macrophages. Mycotoxin Res. 2011, 27, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Zapata, E.; Fernández-López, J.; Pérez-Álvarez, J.A. Tiger nut (Cyperus esculentus) commercialization: Health aspects, composition, properties, and food applications. Compr. Rev. Food Sci. Food Saf. 2012, 11, 366–377. [Google Scholar] [CrossRef]
- Arranz, I.; Stroka, J.; Neugebauer, M. Determination of aflatoxin B1 in tiger nut-based soft drinks. Food Addit. Contam. Part A 2006, 23, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Sebastià, N.; Soler, C.; Soriano, J.M.; Mañes, J. Occurrence of aflatoxins in tigernuts and their beverages commercialized in Spain. J. Agric. Food Chem. 2010, 58, 2609–2612. [Google Scholar] [CrossRef] [PubMed]
- Rueda, J.; Jimenez, E.; Lobo, M.; Sammán, N. Andean Fermented Beverages. In Fermented Foods of Latin America from Traditional Knowledge to Innovative Applications; Barretto Penna, A.L., Nero, L.A., Todorov, S.D., Eds.; CRC Press/Taylor & Francis Group, LLC: Boca Raton, FL, USA, 2017; ISBN 978-1-4987-3811-8. [Google Scholar]
- Granados-Chinchilla, F.; Molina, A.; Chavarría, G.; Alfaro-Cascante, M.; Bogantes-Ledezma, D.; Murillo-Williams, A. Aflatoxins occurrence through the food chain in Costa Rica: Applying the One Health approach to mycotoxin surveillance. Food Control 2017, 82, 217–226. [Google Scholar] [CrossRef]
- Rodrigues, P.; Venâncio, A.; Lima, N. Mycobiota and mycotoxins of almonds and chestnuts with special reference to aflatoxins. Food Res. Int. 2012, 45, 76–90. [Google Scholar] [CrossRef]
- Rodrigues, P.; Venâncio, A.; Lima, N. Aflatoxigenic fungi and aflatoxins in Portuguese almonds. Sci. World J. 2012, 2012, 471926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lamboni, Y.; Nielsen, K.F.; Linnemann, A.R.; Gezgin, Y.; Hell, K.; Nout, M.J.R.; Smid, E.J.; Tamo, M.; van Boekel, M.A.J.S.; Hoof, J.B.; et al. Diversity in secondary metabolites including mycotoxins from strains of Aspergillus Section Nigri isolated from raw cashew nuts from Benin, West Africa. PLoS ONE 2016, 11, e0164310. [Google Scholar] [CrossRef] [PubMed]
- Valle Garcia, M.; Mallmann, C.A.; Copetti, M.V. Aflatoxigenic, and ochratoxigenic fungi and their mycotoxins in spices marketed in Brazil. Food Res. Int. 2018, 106, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Beherens, A. Soy-Based Beverages in Latin America. In Multinationals in Latin America: Case Studies; Liberman, L., Garcilazo, S., Stal, E., Eds.; Macmillan Distribution Ltd.: Hampshire, UK, 2014; pp. 113–120. ISBN 978-1-349-66879-3. [Google Scholar]
- Iha, M.H.; Trucksess, M.W.; Rader, J.I. The fate of ochratoxin A in soy milk and bean curd (tofu) productions. In Mycotoxin Prevention and Control in Agriculture; Appell, M., Kendra, D.F., Trucksess, M.W., Eds.; American Chemical Society: Washington, DC, USA, 2010; pp. 59–68. ISBN 978-084-12-6990-3. [Google Scholar]
- Marsh, A.J.; Hill, C.; Ross, R.P.; Cotter, P.D. Fermented beverages with health-promoting potential: Past and future perspectives. Trends Food Sci. Technol. 2014, 38, 113–124. [Google Scholar] [CrossRef] [Green Version]
- Misihairabgwi, J.M.; Ishola, A.; Quaye, I.; Sulyok, M.; Krska, R. Diversity and fate of fungal metabolites during the preparation of oshikundu, a Namibian traditional fermented beverage. World Mycotoxin J. 2018, 11, 471–481. [Google Scholar] [CrossRef]
- Faria-Oliveira, F.; Diniz, R.H.S.; Godoy-Santos, F.; Piló, F.B.; Mezadri, H.; Castro, I.M.; Brandão, R.L. The Role of Yeast and Lactic Acid Bacteria in the Production of Fermented Beverages in South America. In Food Production and Industry; Eisa, A.A., Ed.; IntechOpen Limited: London, UK, 2015; pp. 107–135. ISBN 978-953-51-2191-6. [Google Scholar]
- Magnoli, C.E.; Astoreca, A.L.; Chiacchiera, S.M.; Dalcero, A.M. Occurrence of ochratoxin A and ochratoxigenic mycoflora in corn and corn based foods and feeds in some South American countries. Mycopathologia 2007, 163, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Romero, S.M.; Comerio, R.M.; Larumbe, G.; Ritieni, A.; Vaamonde, G.; Fernández Pinto, V. Toxigenic fungi isolated from dried vine fruits in Argentina. Int. J. Food Microbiol. 2005, 104, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Lowe, D.P.; Arendt, E.K. The use and effects of lactic acid bacteria in malting and brewing with their relationships to antifungal activity, mycotoxins and gushing: A review. J. Inst. Brew. 2004, 110, 163–180. [Google Scholar] [CrossRef]
- Inoue, T.; Nagatomi, Y.; Uyama, A.; Mochizuki, N. Degradation of aflatoxin B1 during the fermentation of alcoholic beverages. Toxins 2013, 5, 1219–1229. [Google Scholar] [CrossRef] [PubMed]
- Håkansson, O. Reducing Patulin Levels in Apple Juice by Fermentation with Saccharomyces cerevisiae; G2E; Sveriges Lantbruksuniversitet (SLU): Uppsala, Sweden, 2015. [Google Scholar]
- Casarotti, S.N.; Diamantino, V.R.; Silva, L.F.; Afonso dos Santos, C.L.; Toedoro de Paula, A.; Barretto Penna, A.L. Fermented Dairy Beverages in Latin America. In Fermented Foods of Latin America from Traditional Knowledge to Innovative Applications; Barretto Penna, A.L., Nero, L.A., Todorov, S.D., Eds.; CRC Press/Taylor & Francis Group, LLC: Boca Raton, FL, USA, 2017; ISBN 978-1-4987-3811-8. [Google Scholar]
- Coelho, E.M.; Gomes, R.G.; Souza Machado, B.A.; Santos Oliveira, R.; dos Santos Lima, M.; Cavalcanti de Acevedo, L.; Umsza Guez, M.A. Passion fruit peel flour—Technological properties and application in food products. Food Hydrocoll. 2017, 62, 158–164. [Google Scholar] [CrossRef]
- Célia, J.A.; Pereira da Silva, M.A.; Borges de Oliveira, K.; Freitas e Souza, J.L.; Souza, D.G.; Campos de Moura, L.; Marins da Silva, R.; Cagnin, C.; Morais de Freitas, B.S.; Rocha Plácido, G.; et al. Fermented milk enriched with passion fruit peel flour (Passiflora edulis): Physicochemical and sensory aspects and lactic acid bacteria viability. Afr. J. Microbiol. Res. 2015, 9, 1964–1973. [Google Scholar]
- Vieira, N.F.; Silva, M.A.P.; Martins, Y.A.A.; Souza, D.G.; Lima, M.S.; Plácido, G.R.; Caliari, M. Physicochemical and sensory profile of yogurt added with passion fruit peel flour. Afr. J. Microbiol. Res. 2015, 14, 149–155. [Google Scholar]
- Silva, C.E.F.; Abud, A.K.S. Tropical fruit pulps: Processing, product standardization and main control parameters for quality assurance. Braz. Arch. Biol. Technol. 2017, 60, e160209. [Google Scholar] [CrossRef]
- Viva de Toledo, N.M.; Costa de Camargo, A.; Mendes Ramos, P.B.; Button, D.C.; Granato, D.; Canniatti-Brazaca, S.G. Potentials and pitfalls on the use of passion fruit by-products in drinkable yogurt: Physicochemical, technological, microbiological, and sensory aspects. Beverages 2018, 4, 47. [Google Scholar] [CrossRef]
- Bol-Schoenmakers, M.; Braber, S.; Akbari, P.; de Graaff, P.; van Roest, M.; Kruijssen, L.; Smit, J.J.; van Esch, B.C.; Jeurink, P.V.; Garssen, J.; et al. The mycotoxin deoxynivalenol facilitates allergic sensitization to whey in mice. Mucosal Immunol. 2016, 9, 1477–1486. [Google Scholar] [CrossRef] [PubMed]
- Quintana-Guzmán, E.M.; Antillón-Guerrero, F.; Azofeifa-Chaves, J. Determinación de ocratoxina A en plasma humano y en café de Costa Rica por un método de ELISA. Arch. Latinoam. Nutr. 2007, 57, 168–172. [Google Scholar]
- Santini, A.; Ferracane, R.; Mikušová, P.; Eged, Š.; Šrobárová, A.; Meca, G.; Mañes, J.; Ritieni, A. Influence of different coffee drink preparations on ochratoxin A content and evaluation of the antioxidant activity and caffeine variations. Food Control 2011, 22, 1240–1245. [Google Scholar] [CrossRef]
- Mitchell, N.J.; Chen, C.; Palumbo, J.D.; Bianchini, A.; Cappozzo, J.; Stratton, J.; Ryu, D.; Wu, F. A risk assessment of dietary Ochratoxin a in the United States. Food Chem Toxicol. 2017, 100, 265–273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- International Agency for Research on Cancer Possibly carcinogenic to humans. Monogr. Eval. Carcinog. Risks Hum. 1993, 56, 245–395.
- Nielsen, K.F.; Ngemela, A.F.; Jensen, L.B.; de Medeiros, L.S.; Rasmussen, P.H. UHPLC-MS/MS determination of ochratoxin A and fumonisins in coffee using QuEChERS extraction combined with mixed-Mode SPE purification. J. Agric. Food Chem. 2015, 63, 1029–1034. [Google Scholar] [CrossRef] [PubMed]
- Franco, H.; Vega, A.; Reyes, S.; de León, J.; Bonilla, A. Niveles de Ocratoxina A y Aflatoxinas totales en cafés de exportación de Panamá por un método de ELISA. Arch. Latinoam. Nutr. 2014, 64, 42–49. [Google Scholar]
- Leoni, L.A.; Soares, L.M.; Oliveira, P.L. Ochratoxin A in Brazilian roasted and instant coffees. Food Addit. Contam. 2000, 17, 867–870. [Google Scholar] [CrossRef] [PubMed]
- Casal, S.; Vieira, T.; Cruz, R.; Cunha, S.C. Ochratoxin A in commercial soluble coffee and coffee substitutes. Food Res. Int. 2014, 61, 56–60. [Google Scholar] [CrossRef]
- Joint FAO/WHO Expert Committee on Food Additives (JECFA). Evaluation of Certain Food Additives and Contaminants: 68th Report of the Joint FAO/WHO Expert Committee on Food Additives; WHO: Geneva, Switzerland, 2007. [Google Scholar]
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on Contaminants in the Food Chain on a request from the Commission related to ochratoxin A in food. EFSA J. 2006, 4, 356. [Google Scholar]
- García-Moraleja, A.; Font, G.; Mañes, J.; Ferrer, E. Development of a new method for the simultaneous determination of 21 mycotoxins in coffee beverages by liquid chromatography tandem mass spectrometry. Food Res. Int. 2015, 72, 247–255. [Google Scholar] [CrossRef]
- Sánchez-Hervás, M.; Gil, J.V.; Bisbal, F.; Ramón, D.; Martínez-Culebras, P.V. Mycobiota and mycotoxin producing fungi from cocoa beans. Int. J. Food Microbiol. 2008, 125, 336–340. [Google Scholar] [CrossRef] [PubMed]
- Brera, C.; Debegnach, F.; de Santis, B.; Iafrate, E.; Pannunxi, E.; Berdini, C.; Prantera, E.; Gregori, E.; Miraglia, M. Ochratoxin A in cocoa and chocolate products from the Italian market: Occurrence and exposure assessment. Food Control 2011, 22, 1663–1667. [Google Scholar] [CrossRef]
- Turcotte, A.M.; Scott, P.M.; Tague, B. Analysis of cocoa products for ochratoxin A and aflatoxins. Mycotoxin Res. 2013, 29, 193–201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Copetti, M.V.; Iamanaka, B.T.; Pereira, J.L.; Lemes, D.P.; Nakano, F.; Taniwaki, M.H. Co-occurrence of ochratoxin A and aflatoxins in chocolate marketed in Brazil. Food Control 2012, 26, 36–41. [Google Scholar] [CrossRef]
- Copetti, M.V.; Iamanaka, B.T.; Pereira, J.L.; Fungaro, M.H.; Taniwaki, M.H. Aflatoxigenic fungi and aflatoxin in cocoa. Int. J. Food Microbiol. 2011, 148, 141–144. [Google Scholar] [CrossRef] [PubMed]
- De Magalhães, J.T.; Sodré, G.A.; Viscogliosi, H.; Grenier-Loustalot, M. Occurrence of Ochratoxin A in Brazilian cocoa beans. Food Control 2011, 22, 744–748. [Google Scholar] [CrossRef]
- Barkai-Golan, R. Chapter 7: Penicillium Mycotoxins. In Mycotoxins in Fruits and Vegetables; Barkai-Golan, R., Paster, N., Eds.; Elsevier: Berkeley, CA, USA, 2008; pp. 153–185. [Google Scholar]
- Stepién, L.; Koczyk, G.; Waskiewicz, A. Diversity of Fusarium species and mycotoxins contaminating pineapple. J. Appl. Genet. 2013, 54, 367–380. [Google Scholar]
- Górna, K.; Pawlowicz, I.; Waskiewicz, A.; Stepién, L. Fusarium proliferatum strains change fumonisin biosynthesis and accumulation when exposed to host plant extracts. Fungal Biol. 2016, 120, 884–893. [Google Scholar] [CrossRef] [PubMed]
- Faten; Mansour, S.; Nagy, K.S.; Taqi, A.; Abass, K. Factors affecting the fungal contamination of some fruit juices packaged in Tetra Pack. Afr. J. Biotechnol. 2011, 10, 12957–12962. [Google Scholar]
- Rice, S.L.; Beuchat, R.L.; Worthington, R.E. Patulin production by Byssochlamys spp. in fruit juices. Appl. Environ. Microbiol. 1977, 34, 791–796. [Google Scholar] [PubMed]
- Zimmerman, M.; Miorelli, S.; Massaguer, P.R.; Aragao, G.M.F. Growth of Byssochlamys nivea in pineapple juice under the effect of water activity and ascospore age. Braz. J. Microbiol. 2011, 42, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Bevilacqua, A.; Campaniello, D.; Sinigaglia, M.; Ciccarone, C.; Corbo, M.R. Sodium-benzoate and citrus extract increase the effect of homogenization towards spores of Fusarium oxysporum in pineapple juice. Food Control 2012, 28, 199–2014. [Google Scholar] [CrossRef]
- Mourkas, A.; Panagiotopoulou, V.; Markaki, P. Determination of patulin in fruit juices using HPLC-DAD and GC-MSD techniques. Food Chem. 2008, 109, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.P.; Sakai, R.; Abdul Manaf, N.; Mohd Rodhi, A.; Saad, B. High performance liquid chromatography method for the determination of patulin and 5-hydroxymethylfurfural in fruit juices marketed in Malaysia. Food Control 2014, 38, 142–149. [Google Scholar] [CrossRef]
- De Sylos, C.M.; Rodriguez-Amaya, D.B. Incidence of patulin in fruits and fruit juices marketed in Campinas, Brazil. Food Addit. Contam. 1999, 2, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Filali, A.; Ouammi, L.; Betbeder, A.M.; Baudrimont, I.; Soulaymani, R.; Benayada, A.; Creppy, E.E. Ochratoxin A in beverages from Morocco: A preliminary survey. Food Addit. Contam. 2001, 6, 565–568. [Google Scholar] [CrossRef] [PubMed]
- Nunes, C.; Usall, J.; Manso, T.; Torres, R.; Olmo, M.; García, J.M. Effect of High Temperature Treatments on Growth of Penicillium spp. and their Development on ‘Valencia’ Oranges C. Food Sci. Technol. Int. 2007, 13, 63–68. [Google Scholar] [CrossRef]
- Pallottino, F.; Costa, C.; Antonucci, F.; Strano, M.C.; Calandra, M.; Solainia, S.; Menesattia, P. Electronic nose application for determination of Penicillium digitatum in Valencia oranges. J. Sci. Food Agric. 2012, 92, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Tournas, V.H.; Katsoudas, E. Mould and yeast flora in fresh berries, grapes and citrus fruits. Int. J. Food Microbiol. 2005, 105, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Alderman, G.G.; Marth, E.H. Experimental production of aflatoxin on intact citrus fruit. J. Milk Food. Technol. 1974, 37, 451–456. [Google Scholar] [CrossRef]
- Stinson, E.E.; Osman, S.F.; Heisler, E.G.; Siciliano, J.; Bills, D.D. Mycotoxin production in whole tomatoes, apples, oranges, and lemons. J. Agric. Food Chem. 1981, 29, 790–792. [Google Scholar] [CrossRef] [PubMed]
- Scott, P.; Kanhere, S. Stability of Alternaria toxins in fruit juices and wine. Mycotoxin Res. 2001, 17, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Adegoke, G.O.; Ojo, O.A.; Odelade, K.A. Survey of the post harvest diseases and aflatoxin contamination of marketed orange fruit (Citrus sp.) in major cities in Oyo State, Nigeria. J. Agric. Vet. Sci. 2014, 7, 27–31. [Google Scholar]
- Ragab, W.S.; Rashwan, M.A.; Saleim, M.A. Natural occurrence and experimental proliferation of aflatoxins on orange fruits. J. Agric. Sci. Mansoura Univ. 1999, 9, 4885–4893. [Google Scholar]
- Varma, S.K.; Verma, R.A.B. Aflatoxin B1 production in orange (Citrus reticulata) juice by isolates of Aspergillus flavus Link. Mycopathologia 1987, 97, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Marino, A.; Nostro, A.; Fiorentino, C. Ochratoxin A production by Aspergillus westerdijkiae in orange fruit and juice. Int. J. Food Microbiol. 2009, 132, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.S.; Kim, K.; Seo, E.; Kassim, N.; Mtenga, A.B.; Shim, W.B.; Lee, S.H.; Chung, D.H. Occurrence of patulin in various fruit juices from South Korea: An exposure assessment. Food Sci. Biotechnol. 2010, 19, 1–5. [Google Scholar] [CrossRef]
- Broggi, L.; Reynoso, C.; Resnik, S.; Martinez, F.; Drunday, V.; Romero Bernal, A. Occurrence of alternariol and alternariol monomethyl ether in beverages from the Entre Rios Province market, Argentina. Mycotoxin Res. 2013, 29, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Shao, B.; Yang, D.; Li, F. Natural occurrence of four Alternaria mycotoxins in tomato- and citrus-based foods in China. J. Agric. Food Chem. 2015, 63, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Majerus, P.; Bresch, H.; Ottender, H. Ochratoxin A in wines, fruit juices and seasonings. Arch. Lebensmittelhyg. 2000, 51, 95–97. [Google Scholar]
- Zimmerli, B.; Dick, R. Ochratoxin A in table wine and grape-juice: Occurrence and risk assessment. Food Addit. Contam. 1996, 13, 655–668. [Google Scholar] [CrossRef] [PubMed]
- Mohanlall, V.; Odhav, B. Biocontrol of Aflatoxins B1, B2, G1, G2, and Fumonisin B1 with 6,7-Dimethoxycoumarin, a Phytoalexin from Citrus sinensis. J. Food Prot. 2006, 69, 2224–2229. [Google Scholar] [CrossRef] [PubMed]
- Palou, L.; Smilanickb, J.L.; Crisostoa, C.H.; Mansourb, M.; Plaza, P. Ozone gas penetration and control of the sporulation of Penicillium digitatum and Penicillium italicum within commercial packages of oranges during cold storage. Crop Prot. 2003, 22, 1131–1134. [Google Scholar] [CrossRef]
- Van de Perre, E.; Deschuyffeleer, N.; Jacxsens, L.; Vekeman, F.; Van Der Hauwaert, W.; Asam, S.; Rychlik, M.; Devlieghere, F.; De Meulenaer, B. Screening of moulds and mycotoxins in tomatoes, bell peppers, onions, soft red fruits and derived tomato products. Food Control 2014, 37, 165–170. [Google Scholar] [CrossRef]
- Hegazy, E.M. Mycotoxin and fungal contamination of fresh and dried tomato. Annu. Res. Rev. Biol. 2017, 17, 1–9. [Google Scholar] [CrossRef]
- Meena, M.; Swapnil, P.; Upadhyay, R.S. Isolation, characterization and toxicological potential of Alternaria-mycotoxins (TeA, AOH and AME) in different Alternaria species from various regions of India. Sci. Rep. 2017, 7, 8777. [Google Scholar] [CrossRef] [PubMed]
- Pose, G.; Patriarca, A.; Kyanko, V.; Pardo, A.; Fernández Pinto, V. Water activity and temperature effects on mycotoxin production by Alternaria alternata on a synthetic tomato medium. Int. J. Food Microbiol. 2010, 142, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Ioi, D.J. Occurrence of Alternariol and Alternariol Monomethyl Ether in Apple and Tomato Products and Resistance to Food Processing. Master’s Thesis, University of Guelph, Guelph, ON, Canada, 2017. [Google Scholar]
- Lopez, P.; Venema, D.; Mol, H.; Spanjer, M.; de Stoppelaar, J.; Pfeiffer, E.; de Nijs, M. Alternaria toxins and conjugates in selected foods in the Netherlands. Food Control 2016, 69, 153–159. [Google Scholar] [CrossRef]
- Barros Mariutti, L.R.; Valente Soares, L.M. Survey of aflatoxins in tomato products. Food Sci. Technol. 2009, 29, 431–434. [Google Scholar] [CrossRef] [Green Version]
- Chatha, Z.A.; Anjum, F.M.; Zahoor, T. Comparative effects of postharvest mitigation treatments on mycotoxins production potential of Aspergillus parasiticus in mango (Mangifera indica L.) fruit. Pak. J. Phytopathol. 2014, 26, 97–101. [Google Scholar]
- Marasas, W.F.O.; Ploetz, R.C.; Wingfield, M.J.; Wingfield, B.D.; Steenkamp, E.T. Mango malformation disease and the associated fusarium species. Phypatology 2006, 6, 667–672. [Google Scholar]
- Wafaa Haggag, M.; Hazza, M.; Sehab, A.; Abd El-Wahab, M. Mango Malformation: I. Toxin Production Associated with Fusarium Pathogens. Am. J. Plant Sci. 2011, 2, 276–281. [Google Scholar]
- Ammar, M.I.; El-Naggar, M.A. Screening and characterization of fungi and their associated mycotoxins in some fruit crops. Int. J. Adv. Res. 2014, 2, 1216–1227. [Google Scholar]
- Abdel-Sater, M.A.; Zohri, A.A.; Ismail, M.A. Natural contamination of some Egyptian fruit juices and beverages by mycoflora and mycotoxins. J. Food Sci. Technol. 2001, 398, 407–411. [Google Scholar]
- Kataoka, H.; Itano, M.; Ishizaki, A.; Saito, K. Determination of patulin in fruit juice and dried fruit samples by in-tube solid-phase microextraction coupled with liquid chromatography-mass spectrometry. J. Chromatogr. A 2009, 1216, 3746–3750. [Google Scholar] [CrossRef] [PubMed]
- Anwar, A.; Perween, R.; Nazim, K.; Shaukat, S.S.; Mehmood, T.; Qamar-Ul-Haque. Assessment of physico-chemical properties and fungal contamination in plastic bottles and tetra packed mango juices marketed in Karachi city. Int. J. Biol. Biotechnol. 2013, 10, 411–416. [Google Scholar]
- Chatha, Z.A.; Ahmad, A.; Zahoor, T.; Raza, A.; Kaleem, M. Effect of gamma irradiation, UV irradiation and hot water treatment on fungal growth and aflatoxin in mango fruits (Mangifera indica L.). Pak. J. Nutr. 2013, 12, 1050–1056. [Google Scholar] [CrossRef]
- Shephard, G.S.; Vismer, H.F. Chapter 17: Effect of processing on the mycotoxin content in fruit juice. In Mycotoxins in Fruits and Vegetables; Barkai-Golan, R., Paster, N., Eds.; Elsevier: Berkeley, CA, USA, 2008; pp. 335–350. [Google Scholar]
- Jiménez, M.; Logrieco, A.; Botalico, A. Occurrence and pathogenicity of Fusarium species in banana fruits. J. Phytopathol. 1993, 137, 214–220. [Google Scholar] [CrossRef]
- Li, C.; Zuo, C.; Deng, G.; Kuang, R.; Yang, Q.; Hu, C.; Sheng, O.; Zhang, S.; Ma, L.; Wei, Y.; et al. Contamination of bananas with beauvericin and fusaric acid produced by Fusarium oxysporum f. sp. cubense. PLoS ONE 2013, 8, e70226. [Google Scholar] [CrossRef] [PubMed]
- Baiyewu, R.A.; Amusa, N.A.; Ayoola, O.A.; Babalola, O.O. Survey of the post harvest diseases and aflatoxin contamination of marketed pawpaw fruit (Carica papaya L.) in South Western Nigeria. African J. Agric. Res. 2007, 2, 178–181. [Google Scholar]
- Zakaria, L.; Wan Chik, M.; Wai Heng, K.; Salleh, B. Fusarium species Associated with Fruit Rot of Banana (Musa spp.), Papaya (Carica papaya) and Guava (Psidium guajava). Malays. J. Microbiol. 2012, 8, 127–130. [Google Scholar] [CrossRef]
- Abdallah, M.F.; Krska, R.; Sulyok, M. Mycotoxin contamination in sugarcane grass and juice: First report on detection of multiple mycotoxins and exposure assessment for aflatoxins B1 and G1 in humans. Toxins 2016, 8, 343. [Google Scholar] [CrossRef] [PubMed]
- Yassihuyuk, N.; Kadakal, C.; Otag, M. Ergosterol and patulin contents of conventional and homemade red peppers and hot red peppers pastes. J. Food. Process. Technol. 2014, 5, 379. [Google Scholar] [CrossRef]
- Ioi, J.D.; Zhou, T.; Tsao, R.; Marcone, M.F. Mitigation of patulin in fresh and processed foods and beverages. Toxins 2017, 9, 157. [Google Scholar] [CrossRef] [PubMed]
- Ganzera, M.; Strum, S. Recent advances on HPLC/MS in medicinal plant analysis—An update covering 2011–2016. J. Pharm. Biomed. Anal. 2018, 147, 211–233. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Dou, X.-W.; Zhang, C.; Logrico, A.F.; Yang, M.-H. A review of current methods for analysis of mycotoxins in herbal medicines. Toxins 2018, 10, 65. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Jiang, N.; Xian, H.; Wei, D.; Shi, L.; Feng, X. A single-step solid phase extraction for the simultaneous determination of 8 mycotoxins in fruits by ultra-high performance liquid chromatography tandem mass spectrometry. J. Chromatogr. A 2016, 1429, 22–29. [Google Scholar] [CrossRef] [PubMed]
- La Barbera, G.; Capriotti, A.L.; Cavaliere, C.; Foglia, P.; Montone, C.M.; Chiozzi, R.Z.; Laganà, A. A Rapid Magnetic Solid Phase Extraction Method Followed by Liquid Chromatography-Tandem Mass Spectrometry Analysis for the Determination of Mycotoxins in Cereals. Toxins 2017, 9, 147. [Google Scholar] [CrossRef] [PubMed]
- Granados-Chinchilla, F. Insights into the Interaction of Milk and Dairy Proteins with Aflatoxin M1. In Milk Proteins from Structure to Biological Properties and Health Aspects; Gigli, I., Ed.; IntechOpen Limited: London, UK, 2016; pp. 266–286. ISBN 978-953-51-2537-2. [Google Scholar]
- Capriotti, A.L.; Cavaliere, C.; Foglia, P.; La Barbera, G.; Samperi, R.; Ventura, S.; Laganà, A. Mycoestrogen determination in cow milk: Magnetic solid-phase extraction followed by liquid chromatography and tandem mass spectrometry analysis. J. Sep. Sci. 2016, 39, 4794–4808. [Google Scholar] [CrossRef] [PubMed]
- Socas-Rodríguez, B.; Herrera-Herrera, A.V.; Asencio-Ramos, M.; Harnández-Borges, J. Recent applications of carbon nanotube sorbents in analytical chemistry. J. Chromatogr. A 2014, 1357, 110–146. [Google Scholar] [CrossRef] [PubMed]
- Herrero-LaTorre, C.; Barcela-García, J.; García-Martín, S.; Peña-Crecente, R.M.; Otárola-Jiménez, J. Magnetic solid-phase extraction using carbon nanotubes as sorbents: A review. Anal. Chim. Acta 2015, 892, 10–26. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.; Si, W.; Wang, W.; Bai, B.; Nie, D.; Song, W.; Zhao, Z.; Guo, Y.; Han, Z. Determination of type A trichothecenes in coix seed by magnetic solid-phase extraction based on magnetic multi-walled carbon nanotubes coupled with ultra-high performance liquid chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2016, 408, 6823–6831. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Liu, S.; Wang, S.; Guo, Y.; Jiang, S. Carbon-based sorbents: Carbon nanotubes. J. Chromatogr. A 2014, 1357, 53–67. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Deng, Y.; Liu, Y.; Ding, Z.; Li, Y.; Jin, Y. High Throughput Detections. Toxicol. Open Access 2017, 3, 131. [Google Scholar] [CrossRef]
- Arroyo-Manzanares, N.; Huertas-Pérez, J.F.; García-Campaña, A.M.; Gámiz-Gracia, L. Mycotoxin Analysis: New Proposals for Sample Treatment. Adv. Chem. 2014, 2014, 547506. [Google Scholar] [CrossRef]
- Berthiller, F.; Cramer, B.; Iha, M.H.; Krska, R.; Lattanzio, V.M.T.; MacDonald, S.; Malone, R.J.; Maragos, C.; Solfizzo, M.; Stranska-Zachariasova, M.; et al. Developments in mycotoxin analysis: An update for 2016–2017. World Mycotoxin J. 2018, 11, 5–31. [Google Scholar] [CrossRef]
- Escrivá, L.; Oueslati, S.; Font, G.; Manyes, L. Alternaria Mycotoxins in Food and Feed: An Overview. J. Food Qual. 2017, 2017, 1569748. [Google Scholar] [CrossRef]
- Tölgyesi, Á.; Stroka, J.; Tamosiunas, V.; Zwickel, T. Simultaneous analysis of Alternaria toxins and citrinin in tomato: An optimised method using liquid chromatography-tandem mass spectrometry. Food Addit. Contam. Part A 2015, 32, 1512–1522. [Google Scholar] [CrossRef] [PubMed]
- Da Cruz Cabral, L.; Terminiello, L.; Fernández Pinto, V.; Fog Nielsen, K.; Patriarca, A. Natural occurrence of mycotoxins and toxigenic capacity of Alternaria strains from mouldy peppers. Int. J. Food Microbiol. 2016, 236, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Santos, G.G.; Mattos, L.M.; Moretti, C.L. Quality and Occurrence of Mycotoxins in Tomato Products in the Brazilian Market. Enzym. Eng. 2016, 5, 3. [Google Scholar] [CrossRef]
- Chen, A.J.; Tang, D.; Zhou, Y.Q.; Sun, D.S.; Li, X.J.; Wang, L.Z.; Gao, W.W. Identification of Ochratoxin A Producing Fungi Associated with Fresh and Dry Liquorice. PLoS ONE 2013, 8, e78285. [Google Scholar] [CrossRef] [PubMed]
- Romero-González, R.; Frenich, A.G.; Vidal, J.L.; Aguilera-Luiz, M.M. Determination of ochratoxin A and T-2 toxin in alcoholic beverages by hollow fiber liquid phase microextraction and ultra high-pressure liquid chromatography coupled to tandem mass spectrometry. Talanta 2010, 82, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Maham, M.; Kiarostami, V.; Waqif-Husain, S.; Karami-Osboo, R.; Mirabolfathy, M. Analysis of Ochratoxin A in Malt Beverage Samples using Dispersive Liquid–Liquid Microextraction Coupled with Liquid Chromatography-Fluorescence Detection. Czech J. Food Sci. 2013, 31, 520–525. [Google Scholar] [CrossRef]
- Meena, M.; Zehra, A.; Dubey, M.K.; Aamir, M.; Gupta, V.K.; Upadhyay, R.S. A Comparative Evaluation of Biochemical Changes in Tomato (Lycopersicon esculentum Mill.) Infected by Alternaria alternata and Its Toxic Metabolites (TeA, AOH, and AME). Front. Plant Sci. 2016, 7, 1408. [Google Scholar] [CrossRef] [PubMed]
- Walravens, J.; Mikula, H.; Rychlik, M.; Asam, S.; Ediage, E.N.; Di Mavungu, J.D.; Van Landschoot, A.; Vanhaecke, L.; De Saeger, S. Development and validation of an ultra-high-performance liquid chromatography tandem mass spectrometric method for the simultaneous determination of free and conjugated Alternaria toxins in cereal-based foodstuffs. J. Chromatogr. A 2014, 1372, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Jin, H.; Sun, L.; Ma, S.; Lin, R. Determination of Aflatoxins in Medicinal Herbs by High-performance Liquid Chromatography–Tandem Mass Spectrometry. Phytochem. Anal. 2012, 23, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Yao, K.; Zhao, S.; Zheng, P.; Wang, S.; Zeng, Y.; Liang, D.; Ke, Y.; Jiang, H. Determination of aflatoxin and zearalenone analogs in edible and medicinal herbs using a group-specific immunoaffinity column coupled to ultra-high performance liquid chromatography with tandem mass spectrometry. J. Chromatogr. A 2018, 1092, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Monbaliu, S.; Wu, A.; Zhang, D.; Van Peteghem, C.; De Saeger, S. Multimycotoxin UPLC-MS/MS for Tea, Herbal Infusions and the Derived Drinkable Products. J. Agric. Food Chem. 2010, 58, 12664–12671. [Google Scholar] [CrossRef] [PubMed]
- Pallarés, N.; Font, G.; Mañes, J.; Ferrer, E. Multimycotoxin LC-MS/MS analysis in tea beverages after dispersive liquid-liquid microextraction (DLLME). J. Agric. Food Chem. 2017, 65, 10282–10289. [Google Scholar] [CrossRef] [PubMed]
- Miró-Abella, E.; Herrero, P.; Canela, N.; Arola, L.; Borrull, F.; Ras, R.; Fontanals, N. Determination of mycotoxins in plant-based beverages using QuEChERS and liquid chromatography–tandem mass spectrometry. Food Chem. 2017, 229, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Vaclavik, L.; Vaclavikova, M.; Begley, T.H.; Krynitsky, A.J.; Rader, J.I. Determination of Multiple Mycotoxins in Dietary Supplements Containing Green Coffee Bean Extracts Using Ultrahigh-Performance Liquid Chromatography-Tandem Mass Spectrometry (UHPLC-MS/MS). J. Agric. Food Chem. 2013, 61, 4822–4830. [Google Scholar] [CrossRef] [PubMed]
- Juan, C.; Mañes, J.; Font, G.; Juan-García, A. Determination of mycotoxins in fruit berry by-products using QuEChERS extraction method. LWT Food Sci. Technol. 2017, 86, 344–351. [Google Scholar] [CrossRef]
- Flores-Flores, M.E.; González-Peñas, E. Analysis of Mycotoxins in Peruvian Evaporated Cow Milk. Beverages 2018, 4, 34. [Google Scholar] [CrossRef]
- Lippolis, V.; Maragos, C. Fluorescence polarisation immunoassays for rapid, accurate and sensitive determination of mycotoxins. World Mycotoxin J. 2014, 7, 479–489. [Google Scholar] [CrossRef]
- Anfossi, L.; Baggiani, C.; Giovannoli, C.; D’Arco, G.; Giraudi, G. Lateral-flow immunoassays for mycotoxins and phycotoxins: A review. Anal. Bioanal. Chem. 2013, 405, 467–480. [Google Scholar] [CrossRef] [PubMed]
- Sajid, M.; Kawde, A.-N.; Daud, M. Designs, formats and applications of lateral flow assay: A literature review. J. Saudi Chem. Soc. 2015, 19, 689–705. [Google Scholar] [CrossRef]
- Tripathi, P.; Upadhyay, N.; Nara, S. Recent advancements in lateral flow immunoassays: A journey for toxin detection in food. Crit. Rev. Food Sci. Nutr. 2017, 58, 1715–1734. [Google Scholar] [CrossRef] [PubMed]
- Suman, M.; Poms, R. Foreword: Rapid methods for mycotoxin detection. World Mycotoxin J. 2014, 7, 401–405. [Google Scholar]
- Joshi, S.; Annida, R.M.; Zuilhof, H.; van Beek, T.A.; Nielen, M.W.F. Analysis of Mycotoxins in Beer Using a Portable Nanostructured Imaging Surface Plasmon Resonance Biosensor. J. Agric. Food Chem. 2016, 64, 8263–8271. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.; van Dam, R.; van Doom, R.; Katerere, D.; Berthiller, F.; Haasnoot, W.; Nielen, M.W.F. Mycotoxin profiling of 1000 beer samples with a special focus on craft beer. PLoS ONE 2017, 12, e0185887. [Google Scholar] [CrossRef] [PubMed]
- Anfossi, L.; Giovannoli, C.; Giraudi, G.; Biagioli, F.; Passini, C.; Baggiani, C. A Lateral Flow Immunoassay for the Rapid Detection of Ochratoxin A in Wine and Grape Must. J. Agric. Food Chem. 2012, 60, 11491–11497. [Google Scholar] [CrossRef] [PubMed]
- Anfossi, L.; Baggiani, C.; Giovannoli, C.; Giraudi, G. Lateral Flow Immunoassays for Aflatoxins B and G and for Aflatoxin M1. In Aflatoxins—Recent Advances and Future Prospects; Razzaghi-Abyaneh, M., Ed.; IntechOpen Limited: London, UK, 2013; pp. 315–339. ISBN 978-953-51-0904-4. [Google Scholar]
- Mataboro, E.; Ishimwe, N.; Uwimbabazi, E.; Lee, B.H. Current Immunoassay Methods for the Rapid Detection of Aflatoxin in Milk and Dairy Products. Compr. Rev. Food Sci. Food Saf. 2017, 16, 808–820. [Google Scholar] [CrossRef] [Green Version]
- Song, S.; Liu, N.; Zhao, Z.; Ediage, E.N.; Wu, S.; Sun, C.; De Saeger, S.; Wu, A. Multiplex Lateral Flow Immunoassay for Mycotoxin Determination. Anal. Chem. 2014, 86, 4995–5001. [Google Scholar] [CrossRef] [PubMed]
- Man, Y.; Liang, G.; Jia, F.; Li, A.; Fu, H.; Wang, M.; Pan, L. Development of an Immunochromatographic Strip Test for the Rapid Detection of Alternariol Monomethyl Ether in Fruit. Toxins 2017, 9, 152. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.; Liu, L.; Song, S.; Zhng, Q.; Wu, X.; Kuang, H. Rapid detection of tenuazonic acid in cereal and fruit juice using a lateral-flow immunochromatographic assay strip. Food Agric. Immunol. 2017, 28, 1293–1303. [Google Scholar] [CrossRef]
- Urusov, A.E.; Zherdev, A.V.; Petrakova, A.V.; Sadykhov, E.G.; Koroleva, O.V.; Dzantiev, B.N. Rapid Multiple Immunoenzyme Assay of Mycotoxins. Toxins 2015, 7, 238–254. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, W.; Powers, S.; Dai, S.Y. Using commercial immunoassay kits for mycotoxins: ‘joys and sorrows’? World Mycotoxin J. 2014, 7, 417–430. [Google Scholar] [CrossRef]
- O’Farrell, B. Lateral Flow Immunoassay Systems: Evolution from the Current State of the Art to the Next Generation of Highly Sensitive, Quantitative Rapid Assays. In The Immunoassay Handbook—Theory and Applications of Ligand Binding, ELISA and Related Techniques, 4th ed.; Wild, D., Ed.; Elsevier Science: Amsterdam, The Netherlands, 2013; pp. 90–107. ISBN 978-008-09-7037-0. [Google Scholar]
- Man, Y.; Liang, G.; Li, A.; Pan, L. Recent Advances in Mycotoxin Determination for Food Monitoring via Microchip. Toxins 2017, 9, 324. [Google Scholar] [CrossRef] [PubMed]
- Ngundi, M.M.; Shriver-Lake, L.C.; Moore, M.H.; Lassman, M.E.; Ligler, F.S.; Taitt, C.R. Array Biosensor for Detection of Ochratoxin A in Cereals and Beverages. Anal. Chem. 2005, 77, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Karczmarczyk, A. Development of Biosensors for Mycotoxins Detection in Food and Beverages. Ph.D. Thesis, Universität Regensburg, Regensburg, Germany, 2017. [Google Scholar]
- Xu, L.; Zhang, Z.; Zhang, Q.; Li, P. Mycotoxin Determination in Foods Using Advanced Sensors Based on Antibodies or Aptamers. Toxins 2016, 8, 239. [Google Scholar] [CrossRef] [PubMed]
- Tomita, Y.; Morita, Y.; Suga, H.; Fujiwara, D. DNA module platform for developing colorimetric aptamer sensors. BioTechniques 2016, 60, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Mahdavi, R.; Khorrami, S.A.H.; Jabbari, M.V. Evaluation of Ochratoxin A Contamination in Non Alcoholic Beers in Iran. Res. J. Biol. Sci. 2007, 2, 546–550. [Google Scholar]
- Heussner, A.H.; Ausländer, S.; Dietrich, D.R. Development and Characterization of a Monoclonal Antibody against Ochratoxin B and Its Application in ELISA. Toxins 2010, 2, 1582–1594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Mukhtar, H.; Ma, L.; Pang, Q.; Wang, X. VHH Antibodies: Reagents for Mycotoxin Detection in Food Products. Sensors 2018, 18, 485. [Google Scholar] [CrossRef] [PubMed]
- de Nijs, M.; Mengelers, M.J.B.; Boon, P.E.; Heyndrickx, E.; Hoogenboom, L.A.P.; Lopez, P.; Mol, H.G.J. Strategies for estimating human exposure to mycotoxins via food. World Mycotoxin J. 2016, 9, 831–845. [Google Scholar] [CrossRef] [Green Version]
- Baker, R.C.; Ford, R.M.; Helander, M.E.; Marecki, J.; Natarajan, R.; Ray, B. Framework for Managing Mycotoxin Risks in the Food Industry. J. Food Prot. 2014, 77, 2181–2188. [Google Scholar] [CrossRef] [PubMed]
- Van de Brug, F.J.; Lucas Luijckx, N.B.; Cnossen, H.J.; Houben, G.F. Early signals for emerging food safety risks: From past cases to future identification. Food Control 2014, 39, 75–86. [Google Scholar] [CrossRef]
- Costa, M.C.; Goumperis, T.; Andresson, W.; Badiola, J.; Ooms, W.; Pongolini, S.; Saegerman, C.; Jurkovic, M.; Tuominen, P.; Tsigarida, E.; et al. Risk identification in food safety: Strategy and outcomes of the EFSA emerging risks exchange network (EREN), 2010–2014. Food Control 2017, 73, 255–264. [Google Scholar] [CrossRef]
- Freire, L.; Sant’Ana, A.S. Modified mycotoxins: An updated review on their formation, detection, occurrence, and toxic effects. Food Chem. Toxicol. 2018, 111, 189–205. [Google Scholar] [CrossRef] [PubMed]
- Kovač, M.; Šubarić, D.; Bulaić, M.; Kovač, T.; Šarkanj, B. Yesterday masked, today modified; what do mycotoxins bring next? Arh Hig Rada Toksikol. 2018, 69, 169–214. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.; Bignmi, M.; Brüschweiler, B.J.; Rose, M.D. Risks for animal health related to the presence of fumonisins, their modified forms and hidden forms in feed. EFSA J. 2018, 16, 5242. [Google Scholar]
- Medina, Á.; Rodríguez, A.; Magan, N. Climate change and mycotoxigenic fungi: Impacts on mycotoxin production. Curr. Opin. Food Sci. 2015, 5, 99–104. [Google Scholar] [CrossRef]
- Baranyi, N.; Kocsubé, S.; Varga, J. Aflatoxins: Climate change and biodegradation. Curr. Opin. Food Sci. 2015, 5, 60–66. [Google Scholar] [CrossRef]
- Medina, Á.; Akbar, A.; Baazeem, A.; Rodriguez, A.; Magan, N. Climate change, food security and mycotoxins: Do we know enough? Fungal Biol. Rev. 2017, 31, 143–145. [Google Scholar] [CrossRef]
- Moretti, A.; Pascale, M.; Logrieco, A.F. Mycotoxin risks under a climate change scenario in Europe. Trends Food Sci. Technol. 2018, in press. [Google Scholar] [CrossRef]
- Van de Perre, E. Farm to Fork Risk Assessment of Emerging Mycotoxins in Fresh Produce: The Case of Tomato Considering Climate Change. Ph.D. Thesis, Ghent University, Gent, Belgium, 2014. [Google Scholar]
- Vaquera, S.; Patriarca, A.; Fernández Pinto, V. Influence of environmental parameters on mycotoxin production by Alternaria arborescens. Int. J. Food Microbiol. 2016, 219, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Marvin, H.J.P.; Kleter, G.A.; Van der Fels-Klerx, H.J.; Noordam, M.Y.; Franz, E.; Willems, D.J.M.; Boxall, A. Proactive systems for early warning of potential impacts of natural disasters on food safety: Climate-change-induced extreme events as case in point. Food Control 2013, 34, 444–456. [Google Scholar] [CrossRef]
- Rahmianna, A.A.; Purnomo, J.; Yusnawan, E. Assessment of Groundnut Varietal Tolerant to Aflatoxin Contamination in Indonesia. Procedia Food Sci. 2015, 3, 330–339. [Google Scholar] [CrossRef]
- Torre, A.M.; Barros, G.G.; Palacios, S.A.; Chulze, S.N.; Battiani, P. Review on pre- and post-harvest management of peanuts to minimize aflatoxin contamination. Food Res. Int. 2014, 62, 11–19. [Google Scholar] [CrossRef]
- Panjak, S.K.; Shi, H.; Keener, K.M. A review of novel physical and chemical decontamination technologies for aflatoxin in food. Trends Food Sci. Technol. 2018, 71, 73–83. [Google Scholar]
- Chulze, S.N.; Palazzini, J.M.; Torres, A.M.; Barros, G.; Ponsone, M.L.; Geisen, R.; Schmidt-Heydt, M.; Köhl, J. Biological control as a strategy to reduce the impact of mycotoxins in peanuts, grapes and cereals in Argentina. Food Addit. Contam. Part A 2015, 32, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Udomkan, P.; Wiredu, A.N.; Nagle, M.; Müller, J.; Vanlauwe, B.; Bandyopadhyay, R. Innovative technologies to manage aflatoxins in foods and feeds and the profitability of application—A review. Food Control 2017, 76, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar-Mathur, P.; Sunkara, S.; Bhatnagar-Panwar, M.; Waliyar, F.; Sharma, K.K. Biotechnological advances for combating Aspergillus flavus and aflatoxin contamination in crops. Plant Sci. 2015, 234, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Fountain, J.C.; Khera, P.; Yang, L.; Nayak, S.N.; Scully, B.T.; Lee, R.D.; Chen, Z.-Y.; Kemerait, R.C.; Varshney, R.K.; Guo, B. Resistance to Aspergillus flavus in maize and peanut: Molecular biology, breeding, environmental stress, and future perspectives. Crop J. 2015, 3, 229–237. [Google Scholar] [CrossRef]
- Scussel, V. Mycotoxins and fungi food decontamination by GRAS methods. Food Process. Technol. 2016, 7. [Google Scholar] [CrossRef]
- Shah, L.; Ali, A.; Yahya, M.; Zhu, Y.; Wang, S.; Si, H.; Rahman, H.; Ma, C. Integrated control of Fusarium head blight and deoxynivalenol mycotoxin in wheat. Plant Pathol. 2017, 67, 532–548. [Google Scholar] [CrossRef]
- Granados-Chinchilla, F. A Focus on Aflatoxin in Feedstuffs: New Developments in Analysis and Detection, Feed Composition Affecting Toxin Contamination, and Interdisciplinary Approaches to Mitigate It. In Aflatoxin—Control, Analysis, Detection and Health Risks; Abdulra’uf, L.B., Ed.; InTechOpen: London, UK, 2017; pp. 251–280. ISBN 978-953-51-3458-9. [Google Scholar]
- Juvonen, R.; Virkajärvi, V.; Priha, O.; Laitila, A. Microbiological Spoilage and Safety Risks in Non-Beer Beverages; VTT Technical Research Centre of Finland: Vuorimiehentie, Finland, 2011; pp. 37–44. ISBN 978-951-38-7786-6. [Google Scholar]
- Rawat, S. Food Spoilage: Microorganisms and their prevention. Asian J. Plant Sci. Res. 2015, 5, 47–56. [Google Scholar]
- Kregiel, D. Health Safety of Soft Drinks: Contents, Containers, and Microorganisms. BioMed Res. Int. 2015, 2015, 128697. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.L.; Hocking, A.D. Fungi and Food Spoilage, 3rd ed.; Springer: New York, NY, USA, 2009; pp. 401–416. ISBN 978-0-387-92207-2. [Google Scholar]
- Wu, F.; Stacy, S.L.; Kensler, T.W. Global Risk Assessment of Aflatoxins in Maize and Peanuts: Are Regulatory Standards Adequately Protective? Toxicol. Sci. 2013, 135, 251–259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang’ethe, E.K.; Korhonen, H.; Marimba, K.A.; Nduhiu, G.; Mungatu, J.K.; Okoth, S.A.; Joutsjoki, V.; Wamae, L.W.; Shalo, P. Management and mitigation of health risks associated with the occurrence of mycotoxins along the maize value chain in two counties in Kenya. Food Qual. Saf. 2017, 1, 268–274. [Google Scholar] [CrossRef] [Green Version]
- Adetunji, M.C.; Atanda, O.O.; Ezekiel, C.N. Risk Assessment of Mycotoxins in Stored Maize Grains Consumed by Infants and Young Children in Nigeria. Children 2017, 4, 58. [Google Scholar] [CrossRef] [PubMed]
- Assunção, R.; Silva, M.J.; Alvito, P. Challenges in risk assessment of multiple mycotoxins in food. World Mycotoxin J. 2016, 9, 791–811. [Google Scholar] [CrossRef] [Green Version]
- International Agency for Research on Cancer (IARC). Practical approaches to control mycotoxins. In Improving Public Health Thorugh Mycotoxin Control; Pitt, J.I., Wild, C.P., Baan, R.A., Gelderblom, W.C.A., Miller, J.D., Riley, R.T., Wu, F., Eds.; IARC: Lyon, France, 2012; pp. 131–146. ISBN 978-92-832-2158-6. [Google Scholar]
- United States Food and Drug Administration (US FDA). Juice HACCP and the FDA Food Safety Modernization Act: Guidance for Industry. Available online: https://www.fda.gov/downloads/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/UCM569777.pdf (accessed on 25 September 2018).
- Ozturkoglu-Budak, S. A model for implementation of HACCP system for prevention and control of mycotoxins during the production of red dried chili pepper. Food Sci. Technol. 2017, 37, 24–29. [Google Scholar] [CrossRef] [Green Version]
- Gil, L.; Ruiz, P.; Font, G.; Manyes, L. An overview of the applications of hazards analysis and critical control point (HACCP) system to mycotoxins. Rev. Toxicol. 2016, 33, 50–55. [Google Scholar]
- Kristina, S.; Wijaya, B.M. Risk management for food and beverage industry using Australia/New Zealand 4360 Standard. IOP Conf. Ser. Mater. Sci. Eng. 2017, 277, 012025. [Google Scholar] [CrossRef] [Green Version]
- Akbari, P.; Braber, S.; Varasteh, S.; Alizadeh, A.; Garssen, J.; Fink-Gremmels, J. The intestinal barrier as an emerging target in the toxicological assessment of mycotoxins. Arch. Toxicol. 2017, 91, 1007–1029. [Google Scholar] [CrossRef] [PubMed]
- Abreu de Assunção, R.M. Children Exposure to Multiple Mycotoxins through Food Consumption: A Holistic Approach for Risk Assessment. Ph.D. Thesis, Universidade de Évora, Évora, Portugal, 2017. [Google Scholar]
- Benedict, K.; Chiller, T.M.; Mody, R.K. Invasive Fungal Infections Acquired from Contaminated Food or Nutritional Supplements: A Review of the Literature. Foodborne Pathog. Dis. 2016, 13, 343–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vieira, N.O.; Peres, A.; Aquino, V.R.; Pasqualatto, A.C. Drinking yerba mate infusion: A potential risk factor for invasive fungal diseases? Transpl. Infect. Dis. 2010, 12, 565–569. [Google Scholar] [CrossRef] [PubMed]
- Chhonker, S.K.; Rawat, D.; Naik, R.A.; Koiri, R.K. An Overview of Mycotoxins in Human Health with Emphasis on Development and Progression of Liver Cancer. Clin. Oncol. 2018, 3, 1408. [Google Scholar]
- Torres, O.; Matute, J.; Eglineau Waes, J.; Maddox, J.R.; Gregory, S.G.; Ashley-Kock, A.E.; Showker, J.L.; Voss, K.A.; Riley, R.T. Human health implications from co-exposure to aflatoxins and fumonisins in maize based foods in Latin America: Guatemala as a case study. World Mycotoxin J. 2015, 8, 143–159. [Google Scholar] [CrossRef]
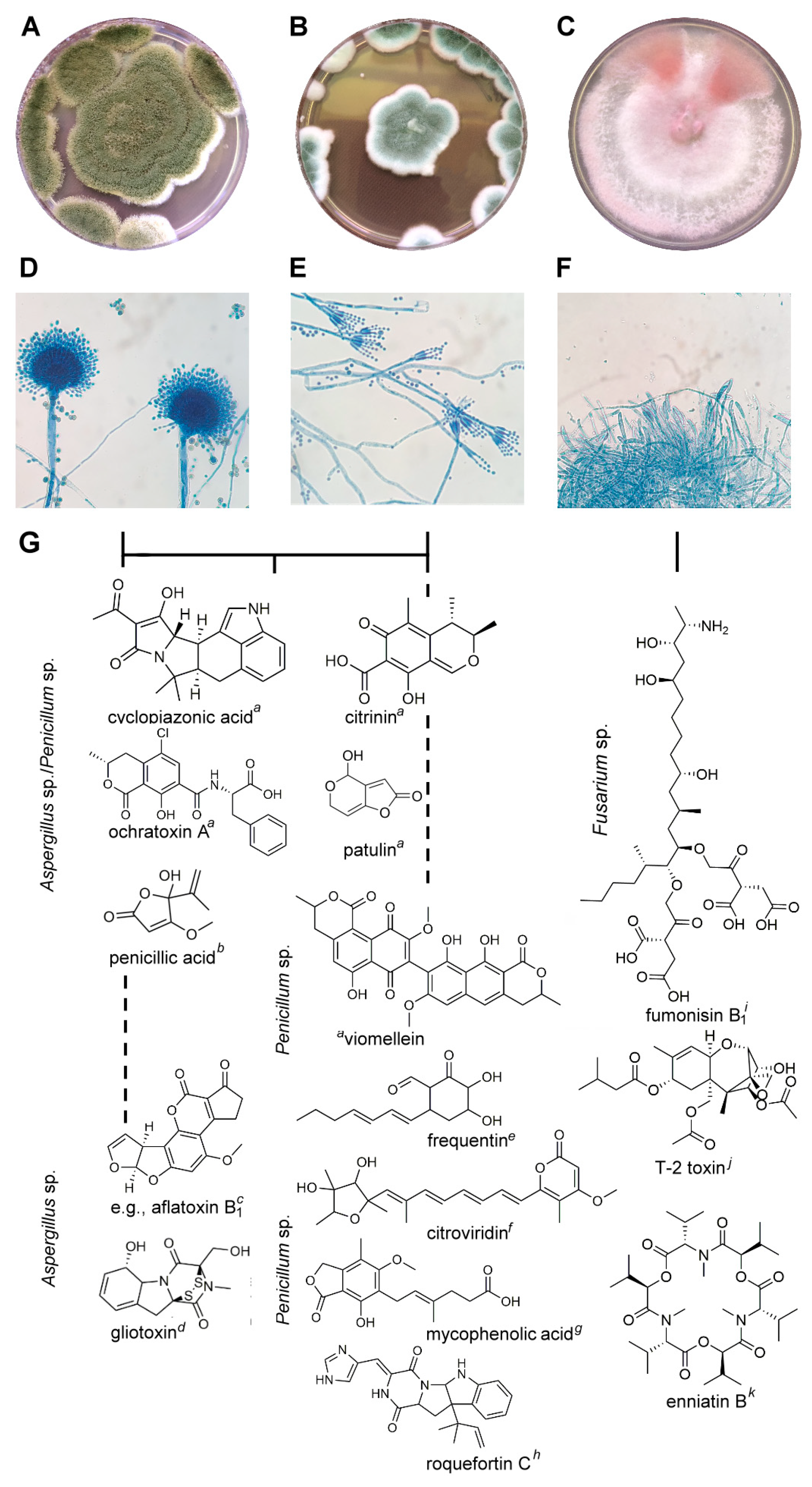
 Pineapple juice production
Pineapple juice production  Canned pineapple chunks production
Canned pineapple chunks production  Waste disposal and treatment; and (B) Chocolate: 1. Receipt and depulping 2. Fermentation 3. Drying (greenhouse) 4. Raw material storage house 5. Packaging material storage 6. Packaging 7. Baler 8. Storage tank 9. Rectification 10. Elevator 11. Mill 12. Toaster 13. Dehuller 14. Fuel and maintenance material storage 15. Cocoa dry bean storage 16. Waste.
Waste disposal and treatment; and (B) Chocolate: 1. Receipt and depulping 2. Fermentation 3. Drying (greenhouse) 4. Raw material storage house 5. Packaging material storage 6. Packaging 7. Baler 8. Storage tank 9. Rectification 10. Elevator 11. Mill 12. Toaster 13. Dehuller 14. Fuel and maintenance material storage 15. Cocoa dry bean storage 16. Waste.
 Pineapple juice production
Pineapple juice production  Canned pineapple chunks production
Canned pineapple chunks production  Waste disposal and treatment; and (B) Chocolate: 1. Receipt and depulping 2. Fermentation 3. Drying (greenhouse) 4. Raw material storage house 5. Packaging material storage 6. Packaging 7. Baler 8. Storage tank 9. Rectification 10. Elevator 11. Mill 12. Toaster 13. Dehuller 14. Fuel and maintenance material storage 15. Cocoa dry bean storage 16. Waste.
Waste disposal and treatment; and (B) Chocolate: 1. Receipt and depulping 2. Fermentation 3. Drying (greenhouse) 4. Raw material storage house 5. Packaging material storage 6. Packaging 7. Baler 8. Storage tank 9. Rectification 10. Elevator 11. Mill 12. Toaster 13. Dehuller 14. Fuel and maintenance material storage 15. Cocoa dry bean storage 16. Waste.
| Consumption | Production Export Value | |||
|---|---|---|---|---|
| Tea (including mate) [23,25] | ||||
| Rank | Country | kg per capita per year | Country | Metric tons |
| 1 | Paraguay | 12.22 | China | 2,414,802 |
| 2 | Uruguay | 9.66 | India | 1,252,174 |
| 3 | Argentina | 6.05 | Kenya | 473,000 |
| 4 | Kenya | 3.24 | Sri Lanka | 349,308 |
| 5 | Gambia | 3.22 | Turkey | 243,000 |
| Coffee [26] | ||||
| Rank | Country | kg per capita per year | Country | Thousand 60 kg bags |
| 1 | Finland | 12.2 | Brazil | 52,735 |
| 2 | Sweden | 10.1 | Vietnam | 29,500 |
| 3 | Norway | 8.9 | Colombia | 14,000 |
| 4 | Austria | 7.8 | Indonesia | 10,902 |
| 5 | Switzerland | 7.6 | Ethiopia | 7650 |
| Chocolate [27,28] | ||||
| Rank | Country | kg per capita per year | Country | Thousand USD |
| 1 | Switzerland | 8.98 | Cote d’Ivoire | 2,595,897 |
| 2 | Germany | 7.89 | Ghana | 1,090,910 |
| 3 | Ireland | 7.39 | Indonesia | 1,087,485 |
| 4 | United Kingdom | 7.39 | Nigeria | 599,000 |
| 5 | Norway | 6.62 | Cameroon | 540,281 |
| Tropical beverage crops, fruits, and sugar [30] | ||||
| Country | Million USD | Country | Million USD (percentage of the total agricultural products traded) | |
| 1 | United States | 2402 | Brazil | 16,466 (19.6) |
| 2 | Mexico | 3071 | Colombia | 2951 (45.8) |
| 3 | Spain | 3373 | Ecuador | 2782 (56.5) |
| 4 | China | 3386 | Vietnam | 2678 (26.5) |
| 5 | Germany | 4360 | Guatemala | 2403 (44.2) |
| 6 | Netherlands | 4708 | Ivory Coast | 2276 (48.5) |
| 7 | United Kingdom | 8602 | Costa Rica | 2015 (50.7) |
| Mycotoxin Analysis in Tea | |||||||
| Matrix | Country | Analyzed Toxin | Positive Samples | Minimum, µg kg−1 | Maximum, µg kg−1 | Analysis Method | Reference |
| Pu-Erh tea | China | OTA | n = 4/36 | 6.50 × 10−1 | 9.47 × 101 | HPLC-FLD | [54] |
| Medicinal plants | Rumania | AFs | n = 7/10 | 1.30 × 101 | 3.80 × 102 | ELISA | [60] |
| FBs | n = 1/10 | 4.60 × 101 | 2.19 × 104 | ||||
| Pu-Erh tea | China | AFB1 | n = 8/70 | 2.10 × 10−2 | 8.52 × 100 | ELISA and HPLC | [61] |
| DON | n = 63/70 | 3.57 × 102 | 3.11 × 103 | ||||
| Medicinal plants | India | AFB1 | n = 1/3 | 4.00 × 10−2 | 1.68 × 100 | HPLC-MS/MS | [68] |
| AFB2 | n = 1/3 | 5.00 × 10−2 | 1.34 × 100 | ||||
| Chamomile | Costa Rica | OTA | n = 13/17 | 1.40 × 10−1 | 4.10 × 10−1 | HPLC-FLD | [69] |
| Mint | n =5/5 | 3.20 × 10−1 | 5.30 × 10−1 | ||||
| Job’s tears | China | ZEA | n = 5/8 | 6.89 × 101 | 2.96 × 102 | HPLC-FLD-MS/MS | [70] |
| Herbs | Spain | OTA | n = 19/88 | 8.00 × 10−1 | 1.06 × 101 | ELISA | [71] |
| FBs | n = 3/88 | 1.40 × 102 | 2.37 × 102 | ||||
| AFs | n = 30/88 | 2.60 × 100 | 8.53 × 102 | ||||
| ZEA | n = 29/88 | 1.50 × 100 | 4.41 × 101 | ||||
| T-2 | n = 29/88 | 6.00 × 10−1 | 2.57 × 102 | ||||
| DON | n = 22/88 | 3.60 × 101 | 3.43 × 102 | ||||
| CIT | n = 19/88 | 1.49 × 101 | 3.55 × 102 | ||||
| Mycotoxin Analysis Nut Milk and Related Beverages | |||||||
| Soft drinks | Spain and Belgium | AFB1 | n = 1/22 | 2.00 × 10−2 | 6.00 × 10−2 | HPLC-FLD | [81] |
| Tiger nuts | Spain | AFB1 | n = 3/37 | 7.00 × 10−1 | 4.50 × 100 | HPLC-FLD | [82] |
| Tiger nut beverages | AFB1 | n = 3/25 | 1.20 × 100 | 3.10 × 100 | |||
| Peanuts | Costa Rica | AFs | n = 125/572 | 4.80 × 10−1 | 4.00 × 102 | Fluorimetry | [84] |
| Almonds | n = 3/65 | 4.80 × 10−1 | 8.90 × 100 | ||||
| Almonds | Portugal | AFB1 | n = 1/21 | 4.60 × 10−1 | 4.97 × 100 | HPLC-FLD | [85] |
| Country | Positive Samples | OTA Concentration (μg kg−1) | Analysis Method | Reference | |
|---|---|---|---|---|---|
| Mean | Max | ||||
| Decaffeinated coffee | |||||
| Spain | 36/40 e | 4.4 × 100 | 32.40 | LC-MS/MS | [2] |
| Green coffee beans | |||||
| Denmark | 7/18 | 1.7 × 100 | 2.8 × 100 | UHPLC-MS/MS | [110] |
| Panamá | 4/21 a | 2.6 × 101 | 3.8 × 101 | ELISA | [111] |
| Roasted coffee | |||||
| Spain | 61/169 | 1.9 × 100 | 4.7 × 100 | LC-MS/MS-QqQ-IT | [1] |
| Spain | 7/52 b | 4.1 × 100 | 1.1 × 101 | LC-MS/MS | [2] |
| Chile | 18/24 | 4.7 × 10−1 | 8.5 × 10−1 | HPLC/fluorescence detector (FLD) | [15] |
| Costa Rica | 54/57 | 3.5 × 10−1 | 9.6 × 10−1 | ELISA (RIDASCREEN® Ochratoxin A) | [106] |
| Denmark | 26/57 c | 2.3 × 100 | 2.1 × 101 | UHPLC-MS/MS | [110] |
| Brazil | 23/34 d | 9.0 ×10−1 | 6.5 × 100 | HPLC/FLD | [111] |
| Soluble coffee | |||||
| Italy | 42/44 | 1.3 × 100 | 6.4 × 100 | HPLC/FLD | [5] |
| Chile | 37/39 | 1.8 × 100 | 7.3 × 100 | HPLC/FLD | [15] |
| Denmark | 14/25 | 4.5 × 100 | 8.3 × 100 | UHPLC-MS/MS | [110] |
| Brazil | 14/14 | 2.2 × 100 | 5.1 × 100 | HPLC/FLD | [112] |
| Portugal | 9/10 f | 2.8 × 100 | 1.2 × 101 | HPLC/FLD | [113] |
| Country | Analyzed Toxin | Positive Samples | Mycotoxin Concentration (µg/kg) | Analysis Methods | References | |
|---|---|---|---|---|---|---|
| Mean | Max | |||||
| Cocoa beans | ||||||
| Brazil | OTA | 38/54 | 1,0 × 100 | >2 | HPLC/FLD | [122] |
| Brazil | AFB1 | 11/85 a | 1.1 × 10−1 | 6.7 × 100 | HPLC/FLD | [121] |
| Cocoa powder | ||||||
| Italy | OTA | 40/40 | 5.1 × 10−1 | 1.82 × 100 | HPLC | [118] |
| Brazil | OTA | 25/25 | 3.9 × 10−1 | 9.2 × 10−1 | HPLC/FLD | [120] |
| Canada | OTA | n = 15 | 1.2 × 100 | 4.7 × 100 | HPLC/FLD | [119] |
| Brazil | AF | 24/25 | 5.3 × 10−1 | 1.7 × 100 | HPLC/FLD | [120] |
| Canada | AF | n = 15 | 1.2 × 100 | 3.52 × 100 | HPLC/FLD | [119] |
| Chocolate | ||||||
| Italy | OTA | 139/260 | 1.4 × 10−1 | 7.4 × 10−1 | HPLC | [118] |
| Brazil | OTA | 98/100 | 2.0 × 10−1 | 8.7 × 10−1 | HPLC/FLD | [120] |
| Canada | OTA | n = 30 | 2.9 × 10−1 | 6.5 × 10−1 | HPLC/FLD | [119] |
| Brazil | AF | 73/100 | 3.0 × 10−1 | 9.1 × 10−1 | HPLC/FLD | [120] |
| Canada | AF | n = 30 | 1.9 × 10−1 | 9.1 × 10−1 | HPLC/FLD | [119] |
| Baking chocolate | ||||||
| Canada | OTA | n = 9 | 4.9 × 10−1 | 9.1 × 10−1 | HPLC/FLD | [119] |
| Canada | AF | n = 9 | 2.7 × 10−1 | 6.7 × 10−1 | HPLC/FLD | [119] |
| Cocoa liquor | ||||||
| Canada | OTA | n = 5 | 4.3 × 10−1 | 5.6 × 10−1 | HPLC/FLD | [119] |
| Canada | AF | n = 5 | 5.1 × 10−1 | 7.6 × 10−1 | HPLC/FLD | [119] |
| Matrix | Target Toxins | Extraction Method | Column, Detection Method | Concentrations Found, µg kg−1 or µg L−1 (Total of Samples Assayed, n) | Reference |
|---|---|---|---|---|---|
| Ochratoxin Determination | |||||
| Liquorice | OTA | 80:20 MeOH:H2O, NERCB-Solid phase extraction (SPE) | Xbridge™ C18 150 × 2.1 mm, 3.5 µm, LC-MS/MS QTRAP®, ESI+ | 12.99–39.03 (26) | [191] |
| Fermented beverages | OTA, T-2 toxin | 1-octanol, 80:20 methanol (MeOH):H2O, hollow fiber liquid phase microextraction | UPLC-MS/MS, ESI+ | <0.02–1.1 (9) | [192] |
| Malt beverages | OTA | Dispersive liquid-liquid acetone/CHCl3 (73:27) | LC-FLD λex 330 nm, λem 460 nm Chromolith, RP18 HPLC column (100 × 4.6 mm) | <0.5–4 (Validation data) | [193] |
| Alternaria Toxins | |||||
| Tomato and tomato juice | ALT, AOH, TEN, TEA, AME, and CIT | MeOH, cleanup Strata-XL SPE cartridges (200 mg, 6 mL, 100 μm) | Ascentis Express C-18 (100 × 2.1 mm, 2.7 μm) TEA derivatized using 2,4-dinitrophenylhydrazine TSQ Quantum Ultra triple quadrupole MS detector, ESI- | 2–50 (Validation data) | [188] |
| Tomato products (pulp, paste, ketchup) | AOH and AME | MeOH, 10 g/100 mL NH4SO4, liquid-liquid extraction CHCl3 | C18 300 × 2.9 mm, 10 μm. 300 mg ZnSO4/L in mobile phase. UV at 250 nm | Ketchup: AOH 0.42–1.16 | [189] |
| Peppers | AOH, AME, TEA | 1. EtOAc, 1 mL/100 mL HCOOH and 2. Liquid-liquid 20 g/100 mL NH4SO4, CHCl3, HCl | 1. Kinetex 100 × 2.1 mm, 2.6 μm. UPLC-DAD/QTOF mass spectrometer ESI 2. Phenomenex Jupiter 250 × 4.6 mm, 5 μm C18. 300 mg ZnSO4/L in the mobile phase. UV 258 nm for AOH and AME, and 280 nm for TEA | TEA: 8–11 422 AOH: 3–98 AME: 7–262 | [190] |
| Tomato | AOH, AME, TEA | Liquid-liquid extraction MeOH/EtOAc | Spherisorb, ODS2 250 × 4.6 mm, 5.0 µm. UV 254 nm | UV absorption match (Qualitative analysis) | [194] |
| Cereals and cereal products | AOH, AME, ALT, TEA, TEN, altertoxin-I, and conjugated (sulfates and glucosides) of AOH and AME | Acetonitrile (ACN)/H2O/CH3COOH (79/19.5/1.5), combined with a hexane defatting step | UPLC-ESI±-MS/MS AcquityUPLC HSS T3 (1.8 µm, 2.1 mm × 100 mm) | Rice: 71% (n = 22/31), 35% (n = 11/31), 19% (6/31) contaminated with TEA, ranges (1.90 ± 0.12–113 ± 12), TEN (3.6 ± 0.7–15.6 ± 2.9), and AOH (1.83 ± 0.14–2.97 ± 0.23). Oats flakes: 31% (n = 5/16) contaminated with TEA (2.13 ± 0.18–39 ± 5) | [195] |
| Aflatoxin Determination | |||||
| Ginger | AFB1, AFB2, AFG1, AFG2, OTA | Immunoaffinity column, MeOH | Agilent Poroshell 120 ECC18 column (50 × 4.6 mm, 2.7 µm) UFLC 5500 QTRAP® hybrid triple quadrupole/near ion trap mass spectrometer equipped, ESI+ | <0.25–13.98 AFB1 <0.10–3 045.37 OTA (3 inoculated ginger powders with A. flavus and A. carbonarius) | [71] |
| Medicinal herbs | AFB1, AFB2, AFG1, AFG2 | 70:30 MeOH:H2O, Immunoafinity column | Agilent XDB C18-column 4.6 × 50 mm, 1.8 μm, MS/MS ESI+ | <0.14–290.80 (174) | [196] |
| Edible and medicinal herbs | AFB1, AFB2, AFG1, AFG2, AFM1, AFM2, ZEA, zearalanone, α/β-zeralanol, and α/β-zearalenol | Immunoaffinity column, N-hydroxysuccinimide-activated Sepharose 4B Fast Flow gel with two group-specific monoclonal antibodies | Acquity HSS T3 2.1 mm × 100 mm; 1.8 μm and Acquity BEH C18 2.1 mm × 100 mm; 1.7 μm, MS/MS ESI± | AFB1 < 0.03–0.15 AFB2 < 0.03–0.54 ZEA < 0.05–2.78 ZAN < 0.06–10.5 (15) | [197] |
| Multi-Toxin Analysis | |||||
| Coffee beverages | OTA, AFB1, AFB2, AFG1, AFG2, STG, NIV, DON, 3-aDON, 15-aDON, DAS, NEO, HT-2, T-2, FB1, FB 2, ENA, ENA1, ENB, ENB1, and BEA | Ethyl acetate/formic acid (95:5). Reconstitution H2O/MeOH (50:50) | LC-MS/MS-TI, C18 column (150 mm × 2 mm, 3 µm, 110 Å), MRM; ESI+ | OTA < 0.24–4.93 AFB1 < 0.05–3.66 AFB2 < 0.04–5.64 AFG1 < 0.04–6.65 STG < 1.00–36.54 NIV < 0.02–24.46 DON <8.23–18.34 3-aDON < 1.00–5.17 NEO < 1.22–30.24 T2 < 0.21–3.57 HT2 < 5.41–14.39 FB1 < 2.78–5.18 ENA1 < 0.02–4.82 ENB < 0.14–36.14 ENB1 < 0.15– 5.33 BEA < 0.03–0.37 (6) | [116] |
| Fresh tomatoes, bell peppers, onions, and soft red fruits | AOH, AME, OTA, FB1, FB2, and FB3 | ACN/EtAOc/HCOOH (60:39:1) | Agilent Zorbax SB-C8, LC-TOF-MS, ESI± | Tomato: TEA 0.7–4.8; Overall < 1.3–90.0 (319) | [151] |
| Fruits (i.e., apple, orange, sweet cherry, and tomato fruits) | AOH, AME, ALT, TEN, TEA, OTA, PAT, CIT | ACN, NaCl. SPE ACN/H2O (3:7) containing 5 mmol L−1 ammonium acetate | Acquity Cortecs UPLC C18 column (100 × 2.1 mm, 1.6 µm), UPLC–MS/MS, ESI± | <1–50 (Validation data) | [176] |
| Dry ginger | AFG1, AFG2, AFB1, AFB2, OTA, CIT | ACN/H2O (80:20) | Agilent Poroshell 120 EC C18, 100 × 2.1 mm, LC-MS/MS | Ginger AFB1 n = 16/28, OTA n = 20/28, CIT n = 16/28 | [76] |
| Cereals (corn and wheat meal) | AFB1, AFB2, AFG1, AFG2, OTA, and ZEA | mSPE, ACN/H2O/HCOOH (80:19.8:0.2). Elution form mGCB CH2Cl2/MeOH 80:20 containing 0.2 mL/100 mL | Thermo Fisher Hypersil Gold C18 column 50 × 2.1 mm, 1.9 µm, UHPLC/ESI± MS/MS (TSQ, triple stage quadrupole) | OTA < 0.10–1.3 (10) ZEA 1.0 <–72.9 (10) | [177] |
| Medicinal plants | ZEA, α-ZON | MeOH/H2O (80:20), NaCl. VICAM® ZearalaTest column, elution MeOH. | LC-FLD λex 274 nm, λem 440 nm, Ultimate XB-C18 column 250 × 4.6, 5 μm LC-MS/MS, ESI-, Gemini C18 20 mm × 2 mm, 3 μm | ZEA < 4–295.8 α-ZON < 2.5 (100) | [70] |
| Tea, Herbal Infusions and the Derived Drinkable Products (from Camellia sinensis) | Nivalenol (NIV), DON, fusarenon-X (FSX), neosolaniol (NEO), 3a-DON, 15a-DON, AFG1, AFG2, AFB1, AFB2, sterigmatocystin (STE), OTA, FB1, FB2, FB3, AOH, AME, ALT, HT-2, T-2 toxin, diacetoxyscirpenol (DAS), ZEA | Raw material: Ethyl acetate (EtOAc)/HCOOH (99:1) NH2 SPE Drinkable products: C18 SPE, elution methanol | Acquity UPLC BEH C18, 100 × 2.1 mm, 1.7 μm, UPLC-MS/MS, ESI+ | FB1 Ceylon mélange < 37–76 (91) | [198] |
| Tea Beverages | AFB1, AFB2, AFG1, AFG2, 3aDON, 15aDON, NIV, HT-2, T-2, ZEA, OTA, ENN, BEA | Dispersion liquid-liquid: NaCl, ACN/EtOAc (60:40), MeOH/CHCl3 (60:40) | LC-MS/MS Turbo ion spray (TI), ESI+, Gemini NX C18 150 × 4.6 mm, 5 µm | Black, red, green and green/teas (44). Green/mint tea: n = 6/8 AFB2 14.4–32.2, n = 2/8 15aDON 60.5–61, n = 4/8 AFG2 1.9–2.6 | [199] |
| Soy, oat and rice plant-based Beverages | AFB1, AFB2, AFG1, AFG2, OTA, DON, ZEA, T-2, HT-2, FB1, and FB2 | ACN with 1 mL/100 mL HCOOH, MgSO4, and NaCl | Cortecs UHPLC C18 column (100 × 2.1 mm, 1.6 µm), Triple Quadrupole MS/MS, ESI+ | Oat: 0.1 AFG1, 0.4 AFB2, AFB2 0.2–0.3, T-2 0.4–1.3, OTA 0.2 Rice: DON 15–19 (9) | [200] |
| Green Coffee Bean Extracts | OTA, OTB, AFB1, AFB2, AFG1, AFG2, TRC A, and B, Alternaria toxins, FB1, FB2, FB3, enniatins (enniatin A, A1, B, B1), BEA, CIT, cyclopiazonic, mycophenolic, and, PA, penitrem A, roquefortine C, gliotoxin, STE. | H2O with 2 mL/100 mL HCOOH and ACN (50:50). MgSO4, NaCl (QuEChERS) | UFLC-QTRAP® TurboIon electrospray, ESI and UPLC quadrupole-orbitrap HESI-II ESI | OTA, OTB, FB1 and mycophenolic acid prevalence: 36%, 32%, 10%, and 16%, respectively. OTA < 1.0−136.9, OTB < 1.0−20.2, FB1 < 50.0−415.0, mycophenolic acid, < 5.0−395.0 | [201] |
| Berry by-products Jam/Juice (i.e., strawberries, blackberries, blueberries, cranberries, and raspberries) and mixed in minor percentage with grape, pomegranate, cherry, apple and plum juice. | AFB1, AFB2, AFG1, AFG2, OTA, AOH, AME, TEN | ACN sodium citrate, MgSO4, and NaCl (QuEChERS) | Phenomenex Gemini-NX C18 (150 × 2.0 mm, 3 µm) LC-MS/MS QTRAP®, ESI+ (Turbo-VTM) | Jams: 1.6 AFG1 Juices: 0.9 AFB2 0.4 AFG1, AFG2 0.7–79, Alternaria toxins (AOH 2.5–85 and AME 267–308) 47% prevalence (52) | [202] |
| Evaporated cow milk | AFM1, AFB1, AFB2, AFG1, AFG2; OTA, OTB, FB1, FB2, FB3, HT-2, T-2, NIV, DON, deepoxy-DON, 3 and 15a-DON, DAS, FSX, NEO, STE, and ZEA | ACN acidified with HCOOH | Ascentis Express C18 column (150 × 2.1 mm, 2.7 µm), Triple Quad (QqQ) LC-MS/MS | OTA: 4/30 < 0.2 (30) | [203] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Granados-Chinchilla, F.; Redondo-Solano, M.; Jaikel-Víquez, D. Mycotoxin Contamination of Beverages Obtained from Tropical Crops. Beverages 2018, 4, 83. https://doi.org/10.3390/beverages4040083
Granados-Chinchilla F, Redondo-Solano M, Jaikel-Víquez D. Mycotoxin Contamination of Beverages Obtained from Tropical Crops. Beverages. 2018; 4(4):83. https://doi.org/10.3390/beverages4040083
Chicago/Turabian StyleGranados-Chinchilla, Fabio, Mauricio Redondo-Solano, and Daniela Jaikel-Víquez. 2018. "Mycotoxin Contamination of Beverages Obtained from Tropical Crops" Beverages 4, no. 4: 83. https://doi.org/10.3390/beverages4040083





