Comparative Physicochemical Properties and Antioxidant Activity of Dietary Soursop Milkshake
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Soursop Drink
2.2. Determination of Physicochemical Properties
2.3. Determination of Proximate Composition
2.4. Evaluation of Antioxidant Properties
2.5. Microbiological Analysis
2.6. Sensory Evaluation
2.7. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Properties of Formulated Soursop Drink
3.2. Proximate Composition of Formulated Drink
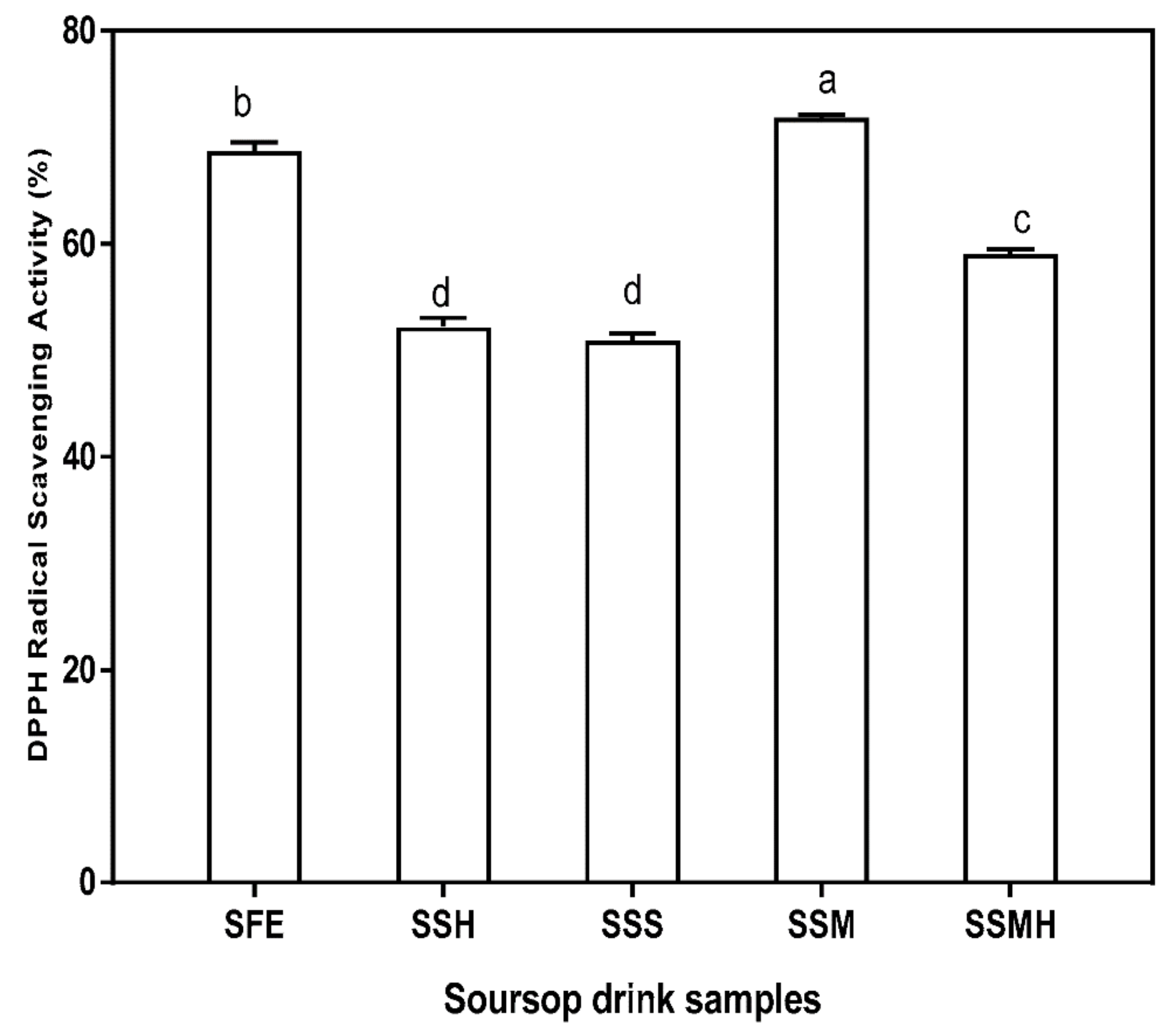
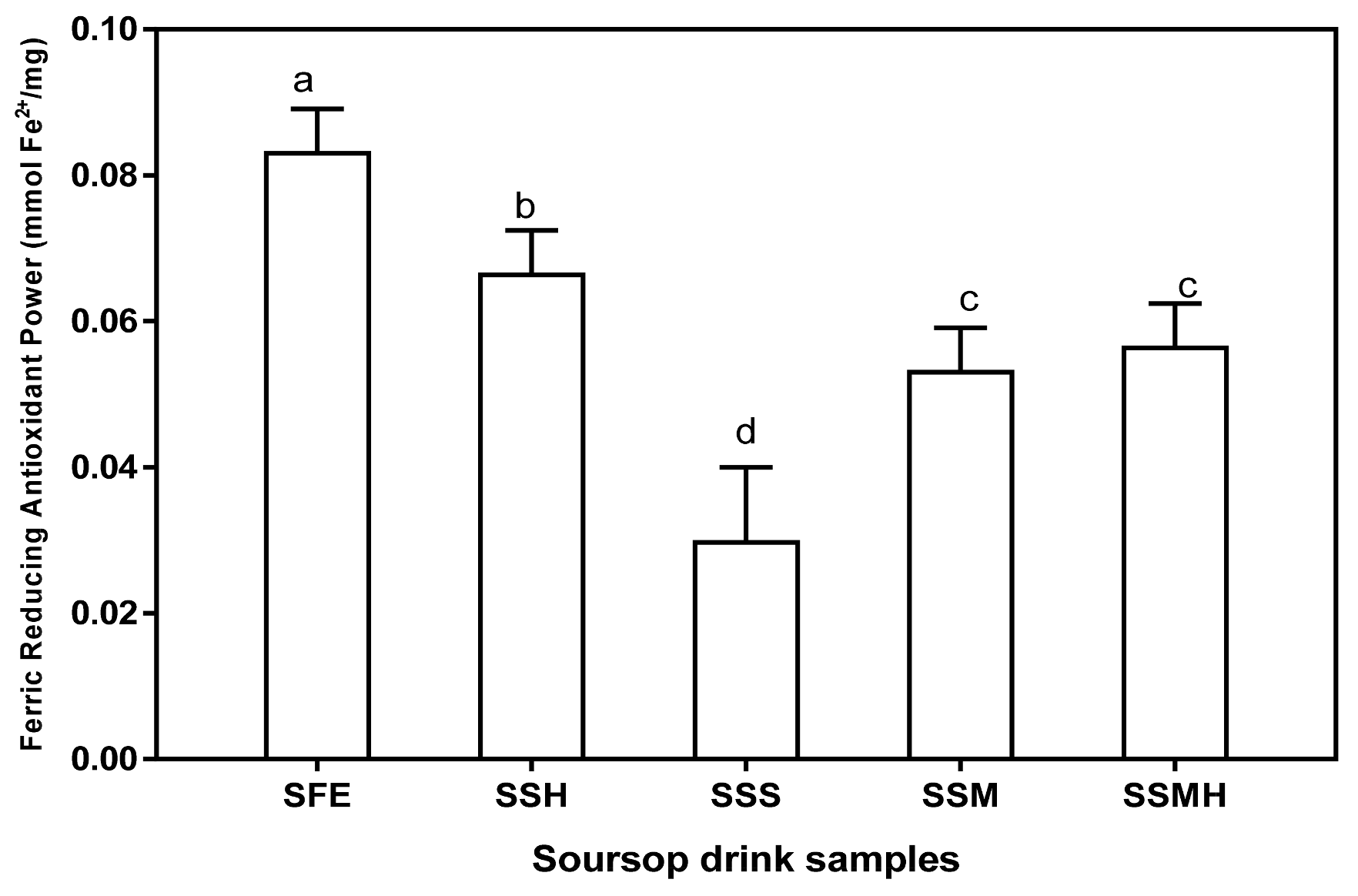
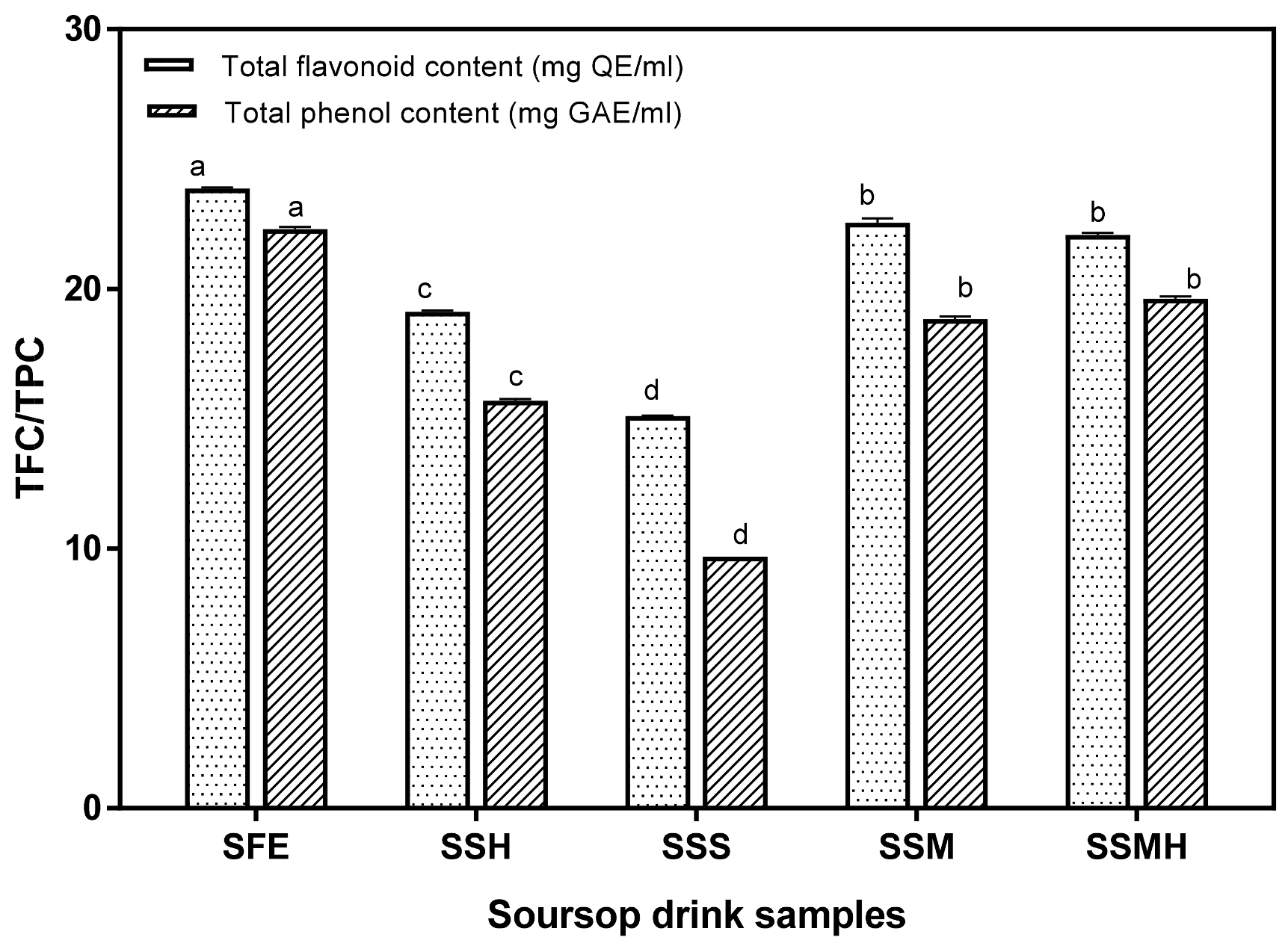
3.3. Antioxidant Properties of Soursop Drink
3.4. Microbiological Assessment of the Soursop Drink Formulations
3.5. Sensory Attributes of the Soursop Drink Formulations
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Abbo, E.S.; Olurin, T.O.; Odeyemi, G. Studies on the storage stability of soursop (Annona muricate L.) juice. Afr. J. Biotechnol. 2006, 5, 1808–1812. [Google Scholar]
- Luzia, D.M.M.; Jorge, N. Soursop (Annona muricata L.) and sugar apple (Annona squamosa L.): Antioxidant activity, fatty acids profile and determination of tocopherols. Nutr. Food Sci. 2012, 42, 434–441. [Google Scholar] [CrossRef]
- Singh, D.R.; Medhi, R.P. Soursop Cultivation in Andaman; Director CIARI: Port Blair, India, 2003. [Google Scholar]
- Minh, N.P. Application of alginate film for soursop fruit preservation. Int. J. Appl. Eng. Res. 2017, 12, 15287–15291. [Google Scholar]
- Othman, O.C.; Fabian, C.; Lugwisha, E. Postharvest physicochemical properties of soursop (Annona muricate L.) fruits of Coast region, Tanzania. J. Food Nutr. Sci. 2014, 2, 220–226. [Google Scholar]
- Almeida-Muradian, L.B.; Stramm, K.M.; Horita, A.; Barth, O.M.; Freitas, A.S.; Estevinho, L.M. Comparative study of the physicochemical and palynological characteristics of honey from Melipona subnitida and Apis mellifera. Int. J. Food Sci. Technol. 2013, 48, 1698–1706. [Google Scholar] [CrossRef]
- Hussein, A.M.S.; Hegazy, N.A.; Kamil, M.M.; Ola, S.S.M. Formulation and evaluation of some healthy natural juice blends. Asian J. Sci. Res. 2016, 10, 160–168. [Google Scholar] [CrossRef]
- Awolu, O.O.; Aderinola, T.A.; Adebayo, I.A. Physicochemical and rheological behaviour of African star apple (Chrysophyllum albidium) juice as affected by concentration and temperature variation. J. Food Process. Technol. 2013, 4, 229–235. [Google Scholar]
- Akusu, O.M.; Kiin-Kabari, D.B.; Ebere, C.O. Quality characteristics of orange/pineapple fruit juice blends. Am. J. Food Sci. Technol. 2016, 4, 43–47. [Google Scholar]
- Karthikeyan, K.; Abitha, S.; Kumar, V.G. Identification of bioactive constituents in peel, pulp of prickly custard apple (Annona muricate) and its antimicrobial activity. Int. J. Pharm. Phytochem. Res. 2016, 8, 1833–1838. [Google Scholar]
- Ndife, J.; Kwaya, P.J.; Bello, S. Production and evaluation of storage changes in soursop-juice. Asian J. Agric. Food Sci. 2014, 2, 425–433. [Google Scholar]
- Mau, J.L.; Tsai, S.Y.; Tseng, Y.H.; Huang, S.J. Antioxidant properties of methanolic extracts from Ganoderma tsugae. Food Chem. 2005, 93, 641–649. [Google Scholar] [CrossRef]
- Association of official Analytical Chemists (AOAC). Official Methods of Analysis, Association of Official Analytical Chemists, 19th ed.; AOAC: Washington, DC, USA, 2012. [Google Scholar]
- Gyamfi, M.A.; Yonamine, M.; Aniya, Y. Free radical scavenging activity of medicinal herb of Ghana: Thonningia sanguinea on experimentally induced liver injuries. Gen. Pharmacol. 1999, 32, 661–667. [Google Scholar] [CrossRef]
- Bao, J.Y.; Cai, M.; Sun, G.; Wang, G.; Corke, H. Anthocyanins, flavonoid and free radical scavenging activity of myrialrubia extracts and their colour properties and stability. J. Agric. Food Chem. 2005, 53, 2327–2332. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Method Enzymol. 1999, 299, 152–178. [Google Scholar]
- Pulido, R.; Bravo, L.; Sauro-Calixo, F. Antioxidant activity of dietary polyphenols as determined by a modified ferric reducing/antioxidant power assay. J. Agric. Food Chem. 2000, 48, 3396–3402. [Google Scholar] [CrossRef] [PubMed]
- Morton, D. Aerobic plate count. In Compendium of Methods for the Microbiological Examination of Foods, 4th ed.; Downes, F.P., Ito, K., Eds.; American Public Health Association: Washington, DC, USA, 2001; pp. 63–67. [Google Scholar]
- Harrigan, W.F.; MacCance, M.E. Laboratory Methods in Food and Dairy Microbiology, 1st ed.; Academic Press: London, UK, 1976; pp. 25–29. [Google Scholar]
- Yapo, E.S.; Kouakou, K.L.; Bognonkpe, T.P.; Kouame, P.; Kouakou, T.H. Comparison of pineapple fruit characteristics of plants propagated in three different ways: by suckers, micropropagation and somatic embryogenesis. J. Nutr. Food Sci. 2011, 1, 110–118. [Google Scholar]
- Islam, M.K.; Khan, M.Z.; Sarkar, M.A.; Absar, N.; Sarkar, S.K. Changes in acidity, TSS, and sugar content at different storage periods of the postharvest mango (Mangifera indica L.) influenced by Bavistin DF. Int. J. Food Sci. 2013. [Google Scholar] [CrossRef] [PubMed]
- Duarte, M.E.M.; Gouveia, D.S.; Mata, M.E.; Queiroz, A.J. Rheological behaviour of mixed drink of Annona and milk. Eng. Agric. Jaboticabal 2012, 32, 333–341. [Google Scholar]
- De Moura, L.C.; da Silva, M.A.; Plácido, G.R.; Caliari, M.; Souza, D.G.; e Souza, J.L.F.; Célia, J.A.; de Oliveira, K.B.; Leão, K.M.; do Nascimento, L.E.C. Functional properties of milk drinks flavoured with mangaba pulp and enriched with passion fruit bark flour. Afr. J. Biotechnol. 2016, 15, 1846–1854. [Google Scholar]
- Nwachukwu, E.; Ezeigbo, C.G. Changes in microbial population of pasteurized soursop juice treated with benzoate and lime during storage. Afr. J. Microbiol. Res. 2013, 7, 3992–3995. [Google Scholar]
- Obasi, B.C.; Whong, C.M.; Ameh, J.B. Nutritional and sensory qualities of commercially and laboratory prepared orange juice. Afr. J. Food Sci. 2017, 11, 189–199. [Google Scholar] [CrossRef]
- Chavan, R.S.; Shraddha, R.C.; Kumar, A.; Nalawade, T. Whey based beverage: Its functionality, formulations, health benefits and applications. J. Food Process. Technol. 2015, 6, 495–503. [Google Scholar]
- Orsi, D.C.; Carvalho, V.S.; Nishr, A.C.; Damiani, C.; Asquieri, E.R. Use of sugar apple, atemoya, and soursop for technological development of jams-chemical and sensorial composition. Ciênc. Agrotecnol. Lavras 2012, 36, 560–566. [Google Scholar] [CrossRef]
- Bajwa, U.; Mittal, S. Quality characteristics of no added sugar ready to drink milk supplemented with mango pulp. J. Food Sci. Technol. 2015, 52, 2112–2120. [Google Scholar] [CrossRef] [PubMed]
- Waston, R.R.; Preedy, V. Bioactive Foods in Promoting Health: Fruits and Vegetables; Academic Press: London, UK, 2009; pp. 628–629. [Google Scholar]
- Badrie, N.; Schauss, A.G. Composition, nutritional value, medicinal uses, and toxicology. In Bioactive Foods in Promoting Health; Waston, R.R., Preedy, V.R., Eds.; Academic Press: London, UK, 2009; pp. 621–641. [Google Scholar]
- Amoo, I.A.; Emenike, A.E.; Akpambang, V.O.E. Compositional evaluation of Annona cherimoya (custard apple) fruit. Trends Appl. Sci. Res. 2008, 3, 216–220. [Google Scholar]
- Chua, L.S.; Rahaman, N.L.; Adnan, N.A.; Tan, T.T. Antioxidant activity of three honey samples in relation with their biochemical components. J. Anal. Methods Chem. 2013. [Google Scholar] [CrossRef] [PubMed]
- Awsi, J.; Er-Dorcus, M. Development and quality evaluation of pineapple juice blend with carrot and orange juice. Int. J. Sci. Res. Publ. 2012, 2, 1–7. [Google Scholar]
- Padayatty, S.J.; Katz, A.; Wang, Y.; Eck, P.; Kwon, O.; Lee, J.H.; Chen, S.; Corpe, C.; Dutta, A.; Dutta, S.K.; et al. Vitamin C as an antioxidant: Evaluation of its role in disease prevention. J. Am. Coll. Nutr. 2003, 22, 18–25. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health, Office of Dietary Supplements. Vitamin C Fact Sheet for Consumers. pp. 1–3. Available online: https://ods.od.nih.gov/pdf/factsheets/VitaminC-Consumer.pdf (accessed on 3 February 2018).
- Onimawo, I.A. Proximate composition and selected physicochemical properties of the seed, pulp and oil of soursop (Annona muricata). Plant Foods Hum. Nutr. 2002, 57, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Cancer.Vg. Cancer Information & Resources. Available online: http://cancer.vg/en/annona-muricata-soursop (accessed on 26 February 2018).
- Onyechi, A.C.; Ibeanu, V.N.; Eme, P.E.; Kelechi, M. Nutrient, phytochemical composition and sensory evaluation of soursop (Annona muricate) pulp and drink in south eastern Nigeria. Int. J. Basic Appl. Sci. 2012, 12, 53–57. [Google Scholar]
- Emelike, N.J.T.; Hart, A.D.; Ebere, C.O. Influence of drying techniques on the properties, physicochemical and mineral composition of beetroot juice. IOSR J. Environ. Sci. Toxicol. Food Technol. 2015, 9, 20–26. [Google Scholar]
- Asgary, S.; Sahebkar, A.; Afshani, M.R.; Keshvari, M.; Haghjooyjavanmard, S.; Rafieian-Kopaei, M. Clinical Evaluation of Blood Pressure Lowering, Endothelial Function Improving, Hypolipidemic and Anti-Inflammatory Effects of Pomegranate Juice in Hypertensive Subjects. Phytopathol. Res. 2014, 28, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Atoui, A.K.; Mansouri, A.; Boskou, G.; Kefalas, P. Tea and herbal infusions; their antioxidant activity and phenolic profile. Food Chem. 2005, 89, 27–36. [Google Scholar] [CrossRef]
- Syahida, M.; Maskat, M.Y.; Suri, R.; Mamot, S.; Hadijah, H. Soursop (Anona muricate L.): Blood hematology and serum biochemistry of Sprague-dawley rats. Int. Food Res. J. 2012, 19, 955–959. [Google Scholar]
- Madurangi, G.W.; Gunathilake, D. Development of an antioxidant rich beverage using soursop fruit and ginger extract. Ann. Food Sci. Technol. 2016, 17, 179–185. [Google Scholar]
- Akomolafe, S.F.; Ajayi, O.B. A comparative study on antioxidant properties, proximate and mineral composition of peel and pulp of ripe Annona muricata (L.) fruit. Int. Food Res. J. 2015, 22, 2381–2388. [Google Scholar]
- Bonoli-Carbognin, M.; Cerretani, L.; Bendini, A.; Almajano, M.P.; Gordon, M.H. Bovine serum albumin produces a synergistic increase in the antioxidant activity of virgin olive oil phenolic compounds in oil-in-water emulsions. J. Agric. Food Chem. 2008, 56, 7076–7081. [Google Scholar] [CrossRef] [PubMed]
- Benavente, G.O.; Castillo, J.; Marin, F.R. Uses and properties of Citrus flavonoids. J. Agric. Food Chem. 1997, 45, 4505–4515. [Google Scholar] [CrossRef]
- Vinson, J.A.; Zubik, L.; Bose, P.; Samman, N.; Proch, J. Dried fruits: Excellent in vitro and in vivo antioxidants. J. Am. Coll. Nutr. 2005, 24, 44–50. [Google Scholar] [CrossRef] [PubMed]
| Sample Code | Pulp (g) | Sugar Syrup (mL) | Honey (mL) | Milk (g) | Water (mL) |
|---|---|---|---|---|---|
| SFE | 200.86 | - | - | - | - |
| SSH | 201.30 | - | 15 | - | 400 |
| SSS | 201.51 | 150 | - | - | 400 |
| SSM | 200.95 | - | - | 75 (30%) | 400 |
| SSMH | 200.00 | - | 15 | 25 (10%) | 400 |
| Soursop Formulations | pH | Brix (°) | Viscosity (dPa) | TTA (%) | Ascorbic Acid (mg/100 mL) |
|---|---|---|---|---|---|
| SFE | 3.70 ± 0.01 c | 18.80 ± 0.10 b | 0.89 ± 0.01 b | 0.52 ± 0.10 a | 44.00 ± 0.75 a |
| SSH | 4.50 ± 0.00 b | 16.00 ± 0.00 c | 0.40 ± 0.01 d | 0.42 ± 0.07 b | 30.86 ± 0.11 c |
| SSS | 4.50 ± 0.00 b | 15.00 ± 0.50 c | 0.40 ± 0.00 d | 0.42 ± 0.03 b | 28.67 ± 0.90 d |
| SSM | 5.60 ± 0.01 a | 22.00 ± 1.00 a | 0.98 ± 0.02 a | 0.25 ± 0.11 c | 32.20 ± 0.18 b |
| SSMH | 4.80 ± 0.00 b | 18.50 ± 0.50 b | 0.78 ± 0.00 c | 0.46 ± 0.10 b | 32.92 ± 0.40 b |
| Sample Code | Composition (%) | Energy (kJ) | |||||
|---|---|---|---|---|---|---|---|
| Moisture | Fat | Protein | Ash | Crude Fibre | Carbohydrate | ||
| SFE | 70.26 ± 0.11 c | 2.40 ± 0.15 c | 2.77 ± 0.12 d | 0.73 ± 0.14 a | 6.30 ± 0.09 a | 17.54 ± 0.15 a | 104.63 ± 1.55 c |
| SSH | 81.30 ± 0.24 a | 1.06 ± 0.0 d | 2.61 ± 0.11 c | 0.46 ± 0.03 b | 3.21 ± 0.16 b | 11.36 ± 0.22 d | 65.33 ± 0.32 e |
| SSS | 79.27 ± 1.02 a | 1.42 ± 0.0 d | 2.03 ± 0.08 d | 0.32 ± 0.06 c | 1.63 ± 0.18 c | 15.33 ± 0.30 b | 83.62 ± 0.38 d |
| SSM | 71.73 ± 0.20 b | 7.44 ± 0.0 a | 5.35 ± 0.16 a | 0.51 ± 0.10 b | 1.55 ± 0.11 c | 13.42 ± 0.12 c | 146.60 ± 1.23 a |
| SSMH | 74.35 ± 1.16 b | 5.41 ± 0.4 b | 4.24 ± 0.07 b | 0.49 ± 0.08 b | 2.98 ± 0.32 b | 12.53 ± 0.51 cd | 117.75 ± 1.56 b |
| Sample Code | Total Viable Count (cfu/mL) | Mould Count (sfu/mL) |
|---|---|---|
| SFE | NVG | NVG |
| SSH | NVG | 1.00 × 103 |
| SSS | 2.30 × 103 | 1.00 × 103 |
| SSM | 4.50 × 103 | 3.20 × 104 |
| SSMH | 2.50 × 103 | 1.00 × 103 |
| Sample | Appearance | Mouthfeel | Aroma | Consistency | Taste | Overall Acceptability |
|---|---|---|---|---|---|---|
| VMD | 7.10 ± 0.13 c | 8.10 ± 0.24 a | 8.00 ± 0.70 a | 8.30 ± 0.70 a | 8.10 ± 0.18 a | 8.00 ± 0.10 a |
| SFE | 7.80 ± 0.20 b | 7.40 ± 0.22 b | 7.60 ± 0.34 b | 7.10 ± 0.89 b | 7.70 ± 0.37 ab | 8.10 ± 0.10 a |
| SSH | 4.60 ± 0.16 e | 5.70 ± 0.08 d | 6.20 ± 0.37 c | 5.20 ± 0.26 d | 5.50 ± 0.12 d | 6.00 ± 0.40 b |
| SSS | 4.60 ± 1.30 e | 6.50 ± 0.26 c | 5.60 ± 0.20 d | 6.00 ± 0.12 c | 6.30 ± 0.25 c | 6.10 ± 0.22 b |
| SSM | 8.30 ± 0.45 a | 7.60 ± 0.12 b | 7.60 ± 0.09 b | 8.00 ± 0.10 a | 7.40 ± 0.10 b | 8.00 ± 0.04 a |
| SSMH | 6.10 ± 0.11 d | 5.70 ± 0.42 d | 6.00 ± 0.8 c | 6.40 ± 0.13 c | 5.90 ± 0.22 d | 6.00 ± 0.50 b |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olagunju, A.I.; Sandewa, O.E. Comparative Physicochemical Properties and Antioxidant Activity of Dietary Soursop Milkshake. Beverages 2018, 4, 38. https://doi.org/10.3390/beverages4020038
Olagunju AI, Sandewa OE. Comparative Physicochemical Properties and Antioxidant Activity of Dietary Soursop Milkshake. Beverages. 2018; 4(2):38. https://doi.org/10.3390/beverages4020038
Chicago/Turabian StyleOlagunju, Aderonke I., and Oreoluwa E. Sandewa. 2018. "Comparative Physicochemical Properties and Antioxidant Activity of Dietary Soursop Milkshake" Beverages 4, no. 2: 38. https://doi.org/10.3390/beverages4020038
APA StyleOlagunju, A. I., & Sandewa, O. E. (2018). Comparative Physicochemical Properties and Antioxidant Activity of Dietary Soursop Milkshake. Beverages, 4(2), 38. https://doi.org/10.3390/beverages4020038