Physicochemical Stability, Antioxidant Activity, and Acceptance of Beet and Orange Mixed Juice During Refrigerated Storage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of the Juices
2.2. Physicochemical Analysis
2.3. Ascorbic Acid Determination by High Performance Liquid Chromatography (HPLC)
2.4. Betalains Determination
2.5. Total Phenolic Compounds Content Determination
2.6. Antioxidant Activity Evaluated by the DPPH Free Radical Scavenging Method
2.7. ABTS+ Cation Radical Scavenging Activity
2.8. Sensory Analysis
2.9. Experimental Design and Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Characteristics
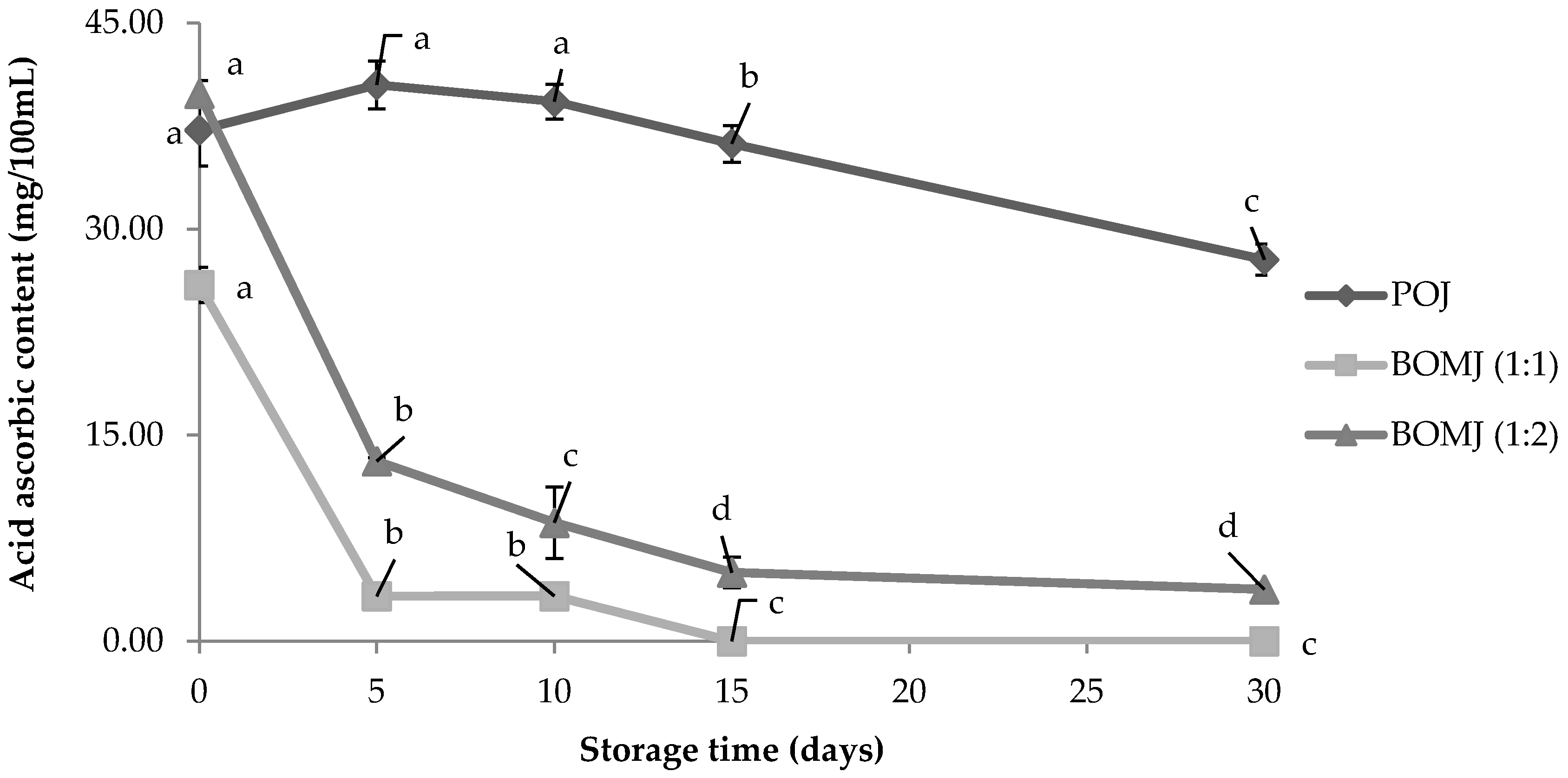
3.2. Ascorbic Acid Content
3.3. Betalain Content
3.4. Total Phenolic Compounds Content and Antioxidant Activity
3.5. Sensory Acceptance
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- FAO. Crop Water Information: Sugarbeet. 2015. Available online: http://www.fao.org/nr/water/cropinfo_sugarbeet.html (accessed on 22 February 2017).
- Wootton-Beard, P.C.; Moran, A.; Ryan, L. Stability of the antioxidant capacity and total polyphenol content of 23 commercially available vegetable jucies before and after in vitro digestion as measured by FRAP, DPPH, ABTS and Folin Ciocalteau methods. Food Res. Int. 2011, 44, 217–224. [Google Scholar] [CrossRef]
- Wruss, J.; Waldenberger, G.; Huemer, S.; Uygun, P.; Lazerstorfer, P.; Müller, U.; Höglinger, O.; Weghuber, J. Compositional characteristics of commercial beetroot products and beetroot juice prepared from seven beetroot varieties grown in Upper Austria. J. Food Comps. Anal. 2015, 42, 46–55. [Google Scholar] [CrossRef]
- Gengatharan, A.; Dykes, G.A.; Choo, W.S. Betalains: Natural plant pigments with potential application in functional foods. LWT Food Sci. Technol. 2015, 64, 645–649. [Google Scholar] [CrossRef]
- Farabegoli, F.; Scarpa, E.S.; Frati, A.; Serafini, G.; Papi, A.; Spisni, E.; Antonini, E.; Benedetti, S.; Ninfali, P. Betalains increase vitexin-2-O-xyloside cytotoxicity in CaCo-2 cancer cells. Food Chem. 2017, 218, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Tarrago-Trani, M.T.; Phillips, K.M.; Cotty, M. Matrix-specific method validation for quantitative analysis of vitamin c in diverse foods. J. Food Comps. Anal. 2012, 26, 12–25. [Google Scholar] [CrossRef]
- Oikeh, E.I.; Omoregie, E.S.; Oviasogie, F.E.; Oriakhi, K. Phytochemical, antimicrobial, and antioxidant activities of different citrus juice concentrates. Food Sci. Nutr. 2016, 4, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Fusco, R.; Cirmi, S.; Gugliandolo, E.; di Paola, R.; Cuzzocrea, S.; Navarra, M. A flavonoid-rich extract of orange juice reduced oxidative stress in an experimental model of inflammatory bowel disease. J. Funct. Foods 2017, 30, 168–178. [Google Scholar] [CrossRef]
- FAO. Food Based Dietary Guidelines for Sri Lankans. 2011. Available online: http://www.fao.org/nutrition/education/food-based-dietary-guidelines/regions/countries/sri-lanka/en/ (accessed on 22 February 2017).
- Vigitel Brasil 2011: Vigilância de Fatores de Risco e Proteção para Doenças Crônicas por Inquérito Telefônico. Available online: http://bvsms.saude.gov.br/bvs/publicacoes/vigitel_brasil_2011_fatores_risco_doencas_cronicas.pdf (accessed on 18 July 2017).
- Wootton-Beard, P.C.; Ryan, L. Improving public health?: The role of antioxidant-rich fruit and vegetable beverages. Food Res. Int. 2011, 44, 3135–3148. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemistry (AOAC). Official Methods of Analysis, 15th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 2004. [Google Scholar]
- Souza, M.C.C.; Benassi, M.T.; Meneghel, R.F.A.; Silva, R.S.S.F. Stability of unpasteurized and refrigerated orange juice. Braz. Arch. Biol. Technol. 2004, 47, 391–397. [Google Scholar] [CrossRef]
- Stintzing, F.C.; Schieber, A.; Carle, R. Evaluation of colour properties and chemical quality parameters of cactus juices. Eur. Food Res. Technol. 2003, 216, 303–311. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin–Ciocalteau reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free-radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Ozgen, M.; Reese, R.N.; Tulio, A.Z.; Scheerens, J.C.; Miller, A.R. Modified 2,2- azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) method to measure antioxidant capacity of selected small fruits and comparison to ferric reducing antioxidant power (FRAP) and 2,2-diphenyl-1-picrylhydrazyl (DPPH) methods. J. Agric. Food Chem. 2006, 54, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.Y.; Woodams, E.E.; Hang, Y.D. Fermentation of beet juice by beneficial lactic acid bacteria. LWT Food Sci. Technol. 2005, 38, 73–75. [Google Scholar] [CrossRef]
- Pimentel, T.C.; Madrona, G.S.; Garcia, S.; Prudencio, S.H. Probiotic viability, physicochemical characteristics and acceptability during refrigerated storage of clarified apple juice supplemented with Lactobacillus paracasei ssp. paracasei and oligofructose in different package type. LWT Food Sci. Technol. 2015, 63, 415–422. [Google Scholar] [CrossRef]
- Costa, G.M.; Silva, J.V.C.; Mingotti, J.D.; Barão, C.E.; Klososki, S.J.; Pimentel, T.C. Effect of ascorbic acid or oligofructose supplementation on L. paracasei viability, physicochemical characteristics and acceptance of probiotic orange juice. LWT Food Sci. Technol. 2017, 75, 195–201. [Google Scholar] [CrossRef]
- Delgado-Vargas, F.; Jiménez, A.R.; Paredes-López, O. Natural pigments: Carotenoids, anthocyanins, and betalains—Characteristics, biosynthesis, processing, and stability. Crit. Rev. Food Sci. Nutr. 2000, 40, 173–289. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, B.K.; Donnell, C.P.O.; Muthukumarappan, K.; Cullen, P.J. Ascorbic acid degradation kinetics of sonicated orange juice during storage and comparison with thermally pasteurised juice. LWT Food Sci. Technol. 2009, 42, 700–704. [Google Scholar] [CrossRef]
- Ministério da Agricultura e do Abastecimento Instrução Normativa n° 1, de 7 de Janeiro de 2000. Complementa Padrões de Identidade e Qualidade Para Suco de Laranja; Diário Oficial da União da República Federativa do Brasil: Brasília (DF), Brasil, 2000; Seção 1; p. 54.
- Cardoso-Ugarte, G.A.; Sosa-Morales, M.E.; Ballard, T.; Liceaga, A.; San Martín-González, M.F. Microwave-assisted extraction of betalains from red beet (Beta vulgaris). LWT Food Sci. Technol. 2014, 59, 276–282. [Google Scholar] [CrossRef]
- Gliszczynska-Swiglo, A.; Wroblewska, J.; Lemanska, K.; Klimczak, I.; Tyrakowska, B. The contribution of polyphenols and vitamin C to the antioxidant activity of commercial orange juices and drinks. In Proceedings of the 14th IGWT Symposium Focussing New Century, Commodity-trade Environment, Beijng, China, August 2004; pp. 121–126. [Google Scholar]
- Zitnanova, I.; Ranostajova, S.; Sobotova, H.; Demelova, D.; Pechan, I.; Durackova, Z. Antioxidative activity of selected fruits and vegetables. Biologia 2006, 61, 279–284. [Google Scholar] [CrossRef]
- Ryan, L.; Prescott, S.L. Stability of the antioxidant capacity of twenty-five commercially available fruit juices subjected to an in vitro digestion. Int. J. Food Sci. Technol. 2010, 45, 1191–1197. [Google Scholar] [CrossRef]
- Ravichandran, K.; Ahmed, A.R.; Knorr, D.; Smetanska, I. The effect of different processing methods on phenolic acid content and antioxidant activity of red beet. Food Res. Int. 2012, 48, 16–20. [Google Scholar] [CrossRef]
- Ravichandran, K.; Saw, N.M.M.T.; Mohdaly, A.A.A.; Gabr, A.M.M.; Kastell, A.; Riedel, H.; Cai, Z.; Knorr, D.; Smetanska, I. Impact of processing of red beet on betalain content and antioxidant activity. Food Res. Int. 2013, 50, 670–675. [Google Scholar] [CrossRef]
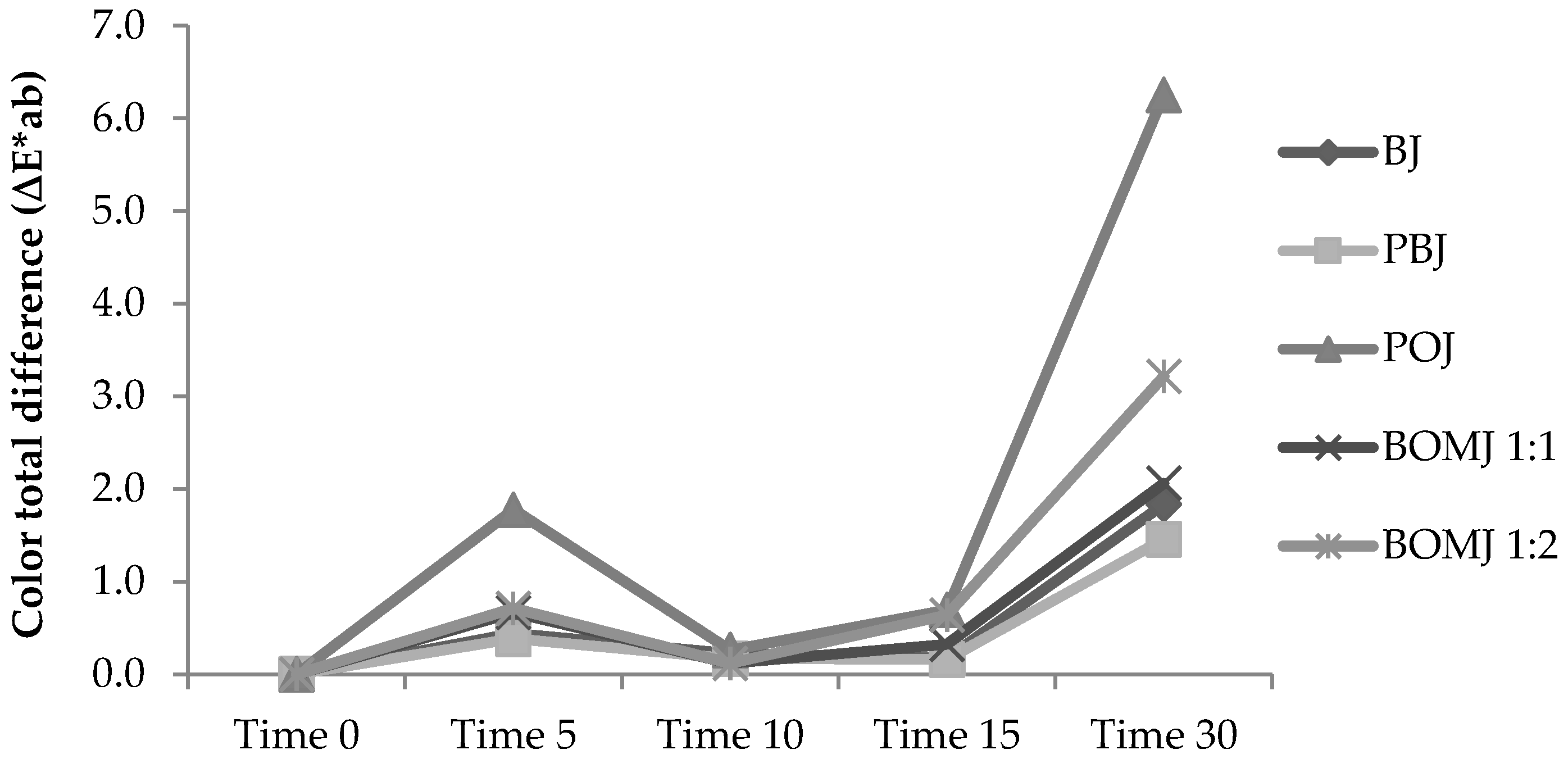
| Titratable Acidity (% Citric Acid) | |||||
| Juices | Storage Time (Days) | ||||
| 0 | 5 | 10 | 15 | 30 | |
| BJ | 0.08 ± 0.00 cD | 0.08 ± 0.00 cD | 0.09 ± 0.00 cbD | 0.09 ± 0.01 bD | 0.14 ± 0.06 aA |
| PBJ | 0.08 ± 0.00 aD | 0.08 ± 0.00 aD | 0.09 ± 0.00 aD | 0.08 ± 0.00 aD | 0.09 ± 0.01 aB |
| POJ | 0.78 ± 0.00 abA | 0.78 ± 0.01 abA | 0.78 ± 0.00 abA | 0.79 ± 0.00 aA | 0.77 ± 0.01 bC |
| BOMJ 1:1 | 0.43 ± 0.01 aC | 0.42 ± 0.00 bC | 0.43 ± 0.01 aC | 0.42 ± 0.00 bC | 0.41 ± 0.00 bD |
| BOMJ 1:2 | 0.59 ± 0.01 aB | 0.57 ± 0.01 bB | 0.57 ± 0.00 bcB | 0.56 ± 0.01 cB | 0.54 ± 0.00 dE |
| pH | |||||
| Juices | Storage Time (Days) | ||||
| 0 | 5 | 10 | 15 | 30 | |
| BJ | 5.71 ± 0.02 aA | 5.62 ± 0.01 abA | 5.56 ± 0.02 bB | 5.36 ± 0.21 cB | 4.84 ± 0.26 dB |
| PBJ | 5.70 ± 0.01 aA | 5.69 ± 0.01 aA | 5.68 ± 0.01 aA | 5.68 ± 0.01 aA | 5.49 ± 0.40 bA |
| POJ | 3.88 ± 0.00 aD | 3.83 ± 0.01 aD | 3.87 ± 0.00 aE | 3.83 ± 0.00 aE | 3.88 ± 0.00 aE |
| BOMJ 1:1 | 4.17 ± 0.01 aB | 4.18 ± 0.01 aB | 4.16 ± 0.00 aC | 4.21 ± 0.03 aC | 4.16 ± 0.01 aC |
| BOMJ 1:2 | 4.03 ± 0.00 aC | 4.00 ± 0.01 aC | 4.02 ± 0.00 aD | 4.03 ± 0.02 aD | 4.03 ± 0.01 aD |
| L* | |||||
| Juices | Storage Time (Days) | ||||
| 0 | 5 | 10 | 15 | 30 | |
| BJ | 22.45 ± 0.16 cD | 22.87 ± 0.04 bD | 22.31 ± 0.07 Cd | 22.37 ± 0.02 cD | 24.16 ± 0.47 aD |
| PBJ | 22.37 ± 0.02 cD | 22.71 ± 0.10 bD | 22.34 ± 0.14 cD | 22.23 ± 0.06 cD | 23.72 ± 0.20 aE |
| POJ | 36.61 ± 0.14 dA | 38.18 ± 0.04 bA | 36.73 ± 0.03 dA | 37.04 ± 0.17 cA | 42.42 ± 0.73 aA |
| BOMJ 1:1 | 23.21 ± 0.04 cC | 23.76 ± 0.06 bC | 23.15 ± 0.02 cC | 23.14 ± 0.05 cC | 24.78 ± 0.17 aC |
| BOMJ 1:2 | 23.77 ± 0.04 cB | 24.33 ± 0.07 bB | 23.81 ± 0.06 cB | 24.15 ± 0.04 bB | 26.18 ± 0.27 aB |
| a* | |||||
| Juices | Storage Time (Days) | ||||
| 0 | 5 | 10 | 15 | 30 | |
| BJ | 0.78 ± 0.06 bD | 0.68 ± 0.03 cD | 0.74 ± 0.03 bcD | 0.81 ± 0.03 bC | 1.07 ± 0.16 aD |
| PBJ | 0.95 ± 0.03 bC | 0.82 ± 0.04 cC | 0.87 ± 0.06 bcC | 0.91 ± 0.03 bcC | 1.19 ± 0.13 aC |
| POJ | −2.49 ± 0.02 dE | −2.87 ± 0.02 bE | −2.51 ± 0.04 dE | −2.63 ± 0.05 cD | −3.64 ± 0.14 aE |
| BOMJ 1:1 | 4.80 ± 0.05 cB | 5.18 ± 0.03 bB | 4.78 ± 0.07 cB | 4.59 ± 0.13 dB | 6.13 ± 0.18 aB |
| BOMJ 1:2 | 6.65 ± 0.04 cA | 7.07 ± 0.05 cA | 6.65 ± 0.03 cA | 6.68 ± 0.10 cA | 8.76 ± 0.32 aA |
| b* | |||||
| Juices | Storage Time (Days) | ||||
| 0 | 5 | 10 | 15 | 30 | |
| BJ | 1.56 ± 0.04 aE | 1.52 ± 0.03 aD | 1.56 ± 0.03 aD | 1.57 ± 0.03 aC | 0.97 ± 0.16 bE |
| PBJ | 1.67 ± 0.02 aD | 1.61 ± 0.03 aD | 1.63 ± 0.05 aD | 1.64 ± 0.03 aC | 1.21 ± 0.11 bD |
| POJ | 16.34 ± 0.31 dA | 17.03 ± 0.02 bA | 16.46 ± 0.08 cA | 15.95 ± 0.16 eA | 18.34 ± 0.17 aA |
| BOMJ 1:1 | 2.39 ± 0.03 aC | 2.38 ± 0.03 aC | 2.35 ± 0.04 aC | 2.19 ± 0.14 bB | 2.23 ± 0.07 bC |
| BOMJ 1:2 | 2.75 ± 0.03 abB | 2.82 ± 0.03 aB | 2.68 ± 0.02 bB | 2.26 ± 0.06 dB | 2.47 ± 0.02 cB |
| Chroma | |||||
| Juices | Storage Time (Days) | ||||
| 0 | 5 | 10 | 15 | 30 | |
| BJ | 1.74 ± 0.03 aE | 1.66 ± 0.02 aE | 1.73 ± 0.03 aA | 1.77 ± 0.02 aA | 1.45 ± 0.12 bA |
| PBJ | 1.92 ± 0.02 aD | 1.80 ± 0.03 abD | 1.85 ± 0.03 aB | 1.88 ± 0.03 aA | 1.69 ± 0.16 bB |
| POJ | 16.53 ± 0.31 dA | 17.27 ± 0.02 bA | 16.65 ± 0.08 cC | 16.16 ± 0.16 eB | 18.70 ± 0.19 aC |
| BOMJ 1:1 | 5.36 ± 0.05 cC | 5.70 ± 0.03 bC | 5.33 ± 0.05 cD | 5.08 ± 0.17 dC | 6.52 ± 0.17 aD |
| BOMJ 1:2 | 7.19 ± 0.04 cB | 7.61 ± 0.04 bB | 7.17 ± 0.02 cdE | 7.05 ± 0.11 dD | 9.10 ± 0.31 aE |
| Juices | Storage Time (Days) | ||||
|---|---|---|---|---|---|
| 0 | 5 | 10 | 15 | 30 | |
| Betacyanin | |||||
| BJ | 65.32 ± 1.58 aA | 61.18 ± 1.72 bA | 55.34 ± 2.09 cA | 51.76 ± 3.51 cA | 40.64 ± 7.79 dA |
| PBJ | 53.39 ± 3.31 aB | 50.52 ± 5.89 bcB | 49.48 ± 2.61 cB | 42.31 ± 3.68 dB | 31.55 ± 5.64 eB |
| BOMJ 1:1 | 31.12 ± 0.89 aC | 31.99 ± 0.59 aC | 31.63 ± 0.76 aC | 28.27 ± 5.19 bC | 30.29 ± 1.94 abC |
| BOMJ 1:2 | 22.31 ± 0.98 abD | 23.17 ± 0.77 aD | 22.85 ± 0.78 abD | 20.37 ± 3.77 bD | 22.32 ± 1.40 abD |
| Betaxanthins | |||||
| BJ | 29.99 ± 1.86 aA | 28.84 ± 0.79 abB | 28.42 ± 2.34 bB | 27.82 ± 2.77 bA | 22.79 ± 5.61 cB |
| PBJ | 31.76 ± 0.51 aA | 30.43 ± 0.08 bA | 30.40 ± 2.29 bA | 27.80 ± 1.44 cA | 25.69 ± 0.22 dA |
| BOMJ 1:1 | 11.96 ± 0.30 abB | 12.08 ± 0.16 aC | 11.76 ± 0.25 abC | 11.44 ± 0.29 abB | 10.74 ± 0.93 bC |
| BOMJ 1:2 | 8.80 ± 0.29 aC | 8.84 ± 0.16 aD | 8.52 ± 0.16 aD | 8.21 ± 0.18 aC | 7.82 ± 0.60 aD |
| Juices | Total Phenolic Compounds | ||||
|---|---|---|---|---|---|
| Storage Time (Days) | |||||
| 0 | 5 | 10 | 15 | 30 | |
| BJ | 521 ± 19 bA | 518 ± 9 bA | 571 ± 86 aA | 515 ± 62 bA | 448 ± 48 cB |
| PBJ | 497 ± 26 bABD | 494 ± 20 bAB | 555 ± 76 aA | 517 ± 54 bA | 431 ± 19 cB |
| POJ | 448 ± 19 abC | 473 ± 10 aB | 477 ± 18 aBD | 458 ± 15 abB | 433 ± 41 bB |
| BOMJ 1:1 | 484 ± 33 cDB | 499 ± 18 abcAB | 519 ± 50 aC | 514 ± 48 abA | 485 ± 65 bcA |
| BOMJ 1:2 | 485 ± 50 abB | 506 ± 29 aA | 505 ± 42 aCD | 490 ± 42 aA | 459 ± 59 bAB |
| DPPH | |||||
| BJ | 2733 ± 188 aA | 2527 ± 255 abA | 2307 ± 325 bcA | 2323 ± 419 bA | 2157 ± 243 cA |
| PBJ | 2813 ± 264 aA | 2210 ± 285 bB | 2040 ± 296 bcB | 1870 ± 236 cdB | 1760 ± 261 dB |
| POJ | 1408 ± 245 aC | 773 ± 96 bcD | 1037 ± 153 bD | 797 ± 167 bcD | 957 ± 135 bcC |
| BOMJ 1:1 | 2083 ± 89 aB | 2027 ± 187 abBC | 1317 ± 318 cC | 1670 ± 369 bBC | 1853 ± 231 abB |
| BOMJ 1:2 | 1930 ± 246 aB | 1803 ± 146 abC | 1452 ± 120 cC | 1596 ± 200 bcC | 1554 ± 119 cB |
| ABTS | |||||
| BJ | 2179 ± 126 aA | 1972 ± 209 bA | 1950 ± 173 bA | 2059 ± 145 bA | 1720 ± 352 cAD |
| PBJ | 2081 ± 142 aA | 1741 ± 155 cB | 1957 ± 206 bA | 1999 ± 63 abA | 1798 ± 62 cA |
| POJ | 1872 ± 241 aB | 1515 ± 93 bC | 1507 ± 103 bcB | 1575 ± 148 bB | 1400 ± 113 cB |
| BOMJ 1:1 | 1854 ± 75 aB | 1746 ± 136 abB | 1642 ± 61 bcC | 1711 ± 92 bcC | 1604 ± 54 cC |
| BOMJ 1:2 | 1840 ± 131 aB | 1667 ± 68 bB | 1552 ± 51 cBC | 1605 ± 84 bcBC | 1614 ± 146 bcCD |
| Juices | Color | Aroma | Flavor | Overall Acceptance |
|---|---|---|---|---|
| PBJ | 7.5 ± 1.8 b | 5.9 ± 1.7 b | 4.9 ± 2.2 b | 5.6 ± 1.8 b |
| BOMJ (1:1) | 8.0 ± 0.9 a | 6.6 ± 1.7 a | 6.3 ± 1.7 a | 6.8 ± 1.5 a |
| BOMJ (1:2) | 7.8 ± 1.1 ab | 6.9 ± 1.9 a | 6.6 ± 2.0 a | 6.9 ± 1.8 a |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Porto, M.R.A.; Okina, V.S.; Pimentel, T.C.; Prudencio, S.H. Physicochemical Stability, Antioxidant Activity, and Acceptance of Beet and Orange Mixed Juice During Refrigerated Storage. Beverages 2017, 3, 36. https://doi.org/10.3390/beverages3030036
Porto MRA, Okina VS, Pimentel TC, Prudencio SH. Physicochemical Stability, Antioxidant Activity, and Acceptance of Beet and Orange Mixed Juice During Refrigerated Storage. Beverages. 2017; 3(3):36. https://doi.org/10.3390/beverages3030036
Chicago/Turabian StylePorto, Maria Rita A., Vivian S. Okina, Tatiana C. Pimentel, and Sandra Helena Prudencio. 2017. "Physicochemical Stability, Antioxidant Activity, and Acceptance of Beet and Orange Mixed Juice During Refrigerated Storage" Beverages 3, no. 3: 36. https://doi.org/10.3390/beverages3030036
APA StylePorto, M. R. A., Okina, V. S., Pimentel, T. C., & Prudencio, S. H. (2017). Physicochemical Stability, Antioxidant Activity, and Acceptance of Beet and Orange Mixed Juice During Refrigerated Storage. Beverages, 3(3), 36. https://doi.org/10.3390/beverages3030036





