Ultra High Pressure Homogenization of Soy Milk: Effect on Quality Attributes during Storage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Soy Milk
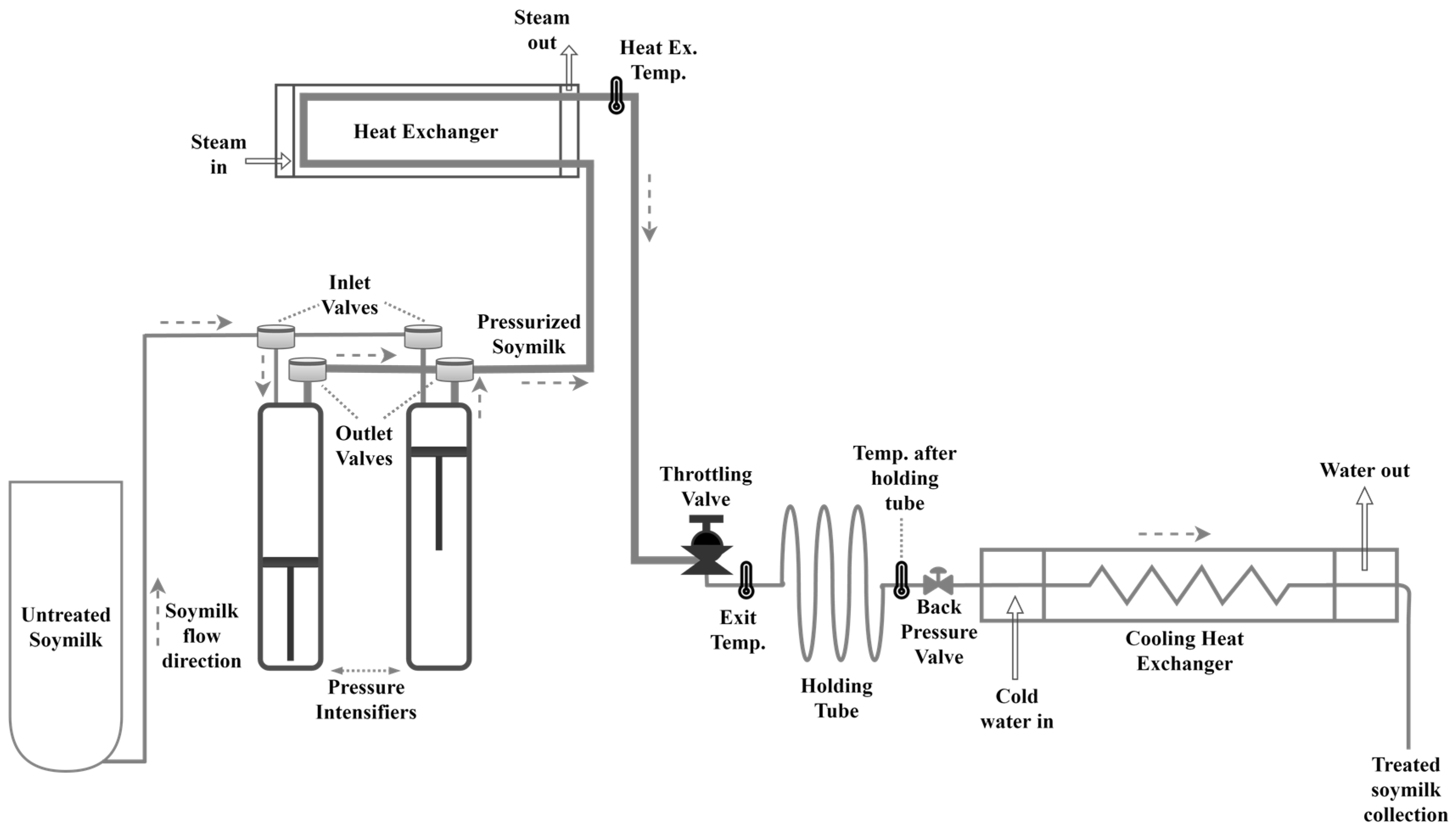
2.2. Ultra High Pressure Homogenization (UHPH) of Soy Milk (Figure 1)
2.3. Microbiology
2.4. pH
2.5. Dry Solids Content
2.6. Particle Size Distribution (PSD)
2.7. Visible Layer Separation/Sedimentation
2.8. Lipoxygenase Activity
2.9. Sensory Analysis
2.10. Data Analysis
3. Results and Discussion
3.1. Effect of UHPH on Temperature Rise
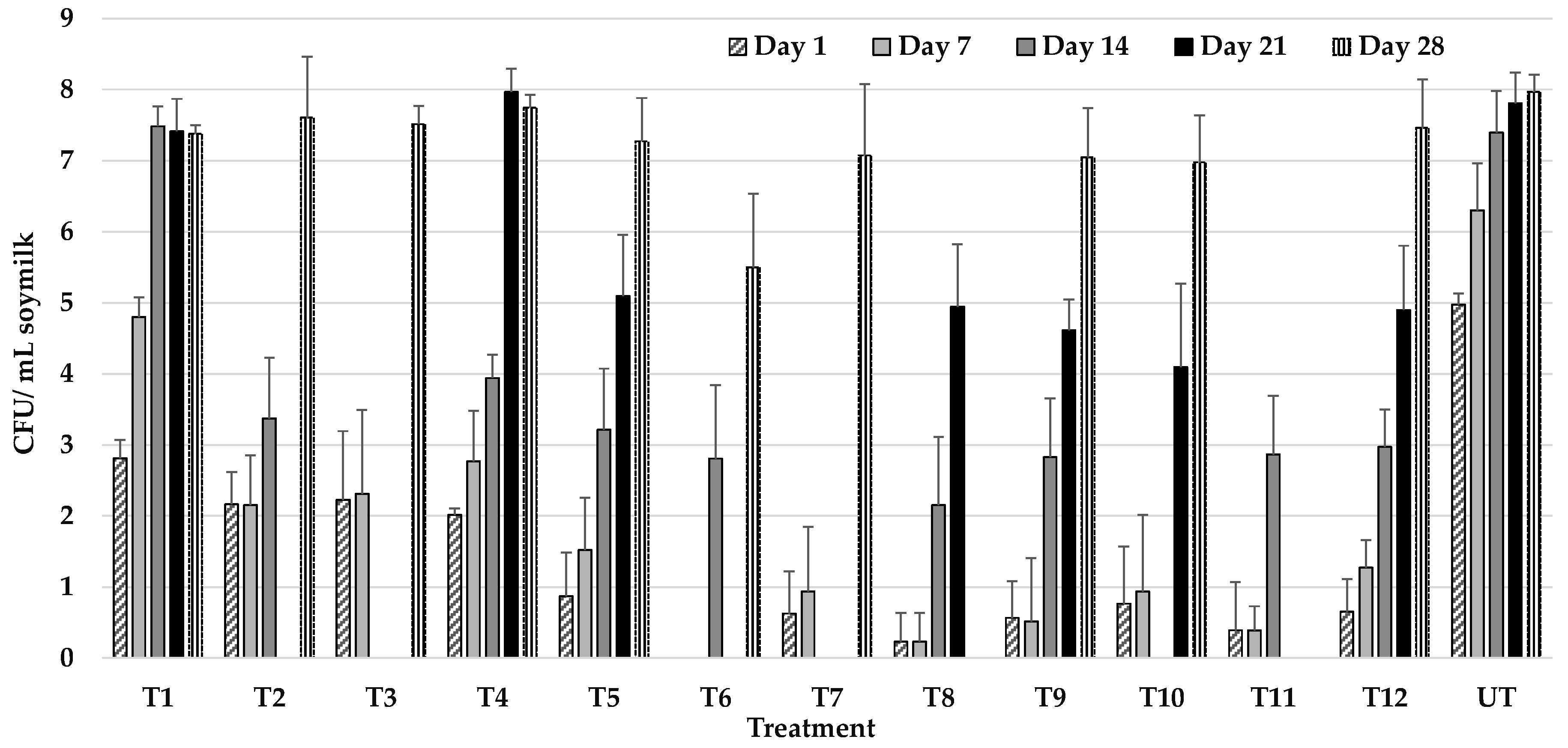
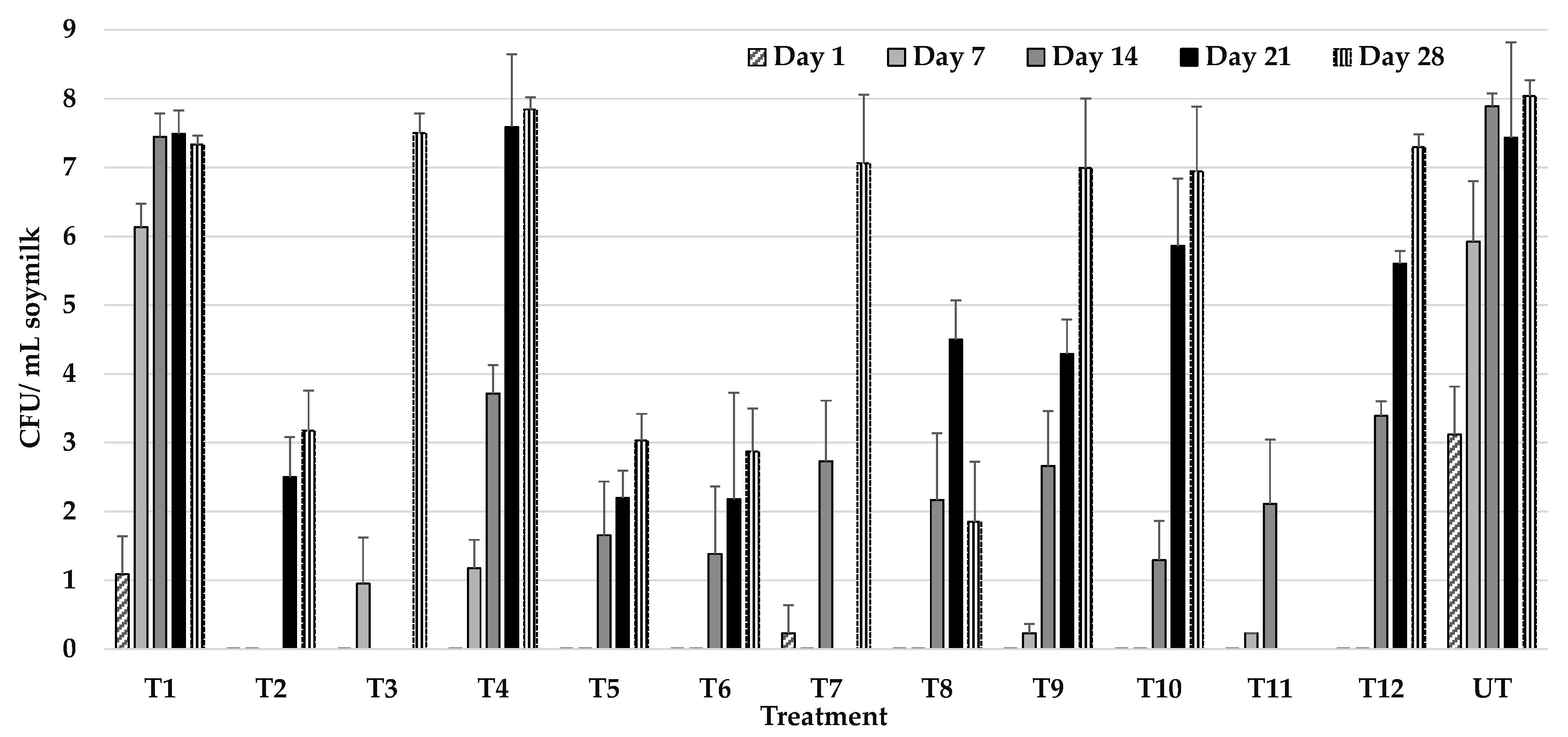
3.2. Effect of UHPH on the Microbiological Quality of Soy Milk
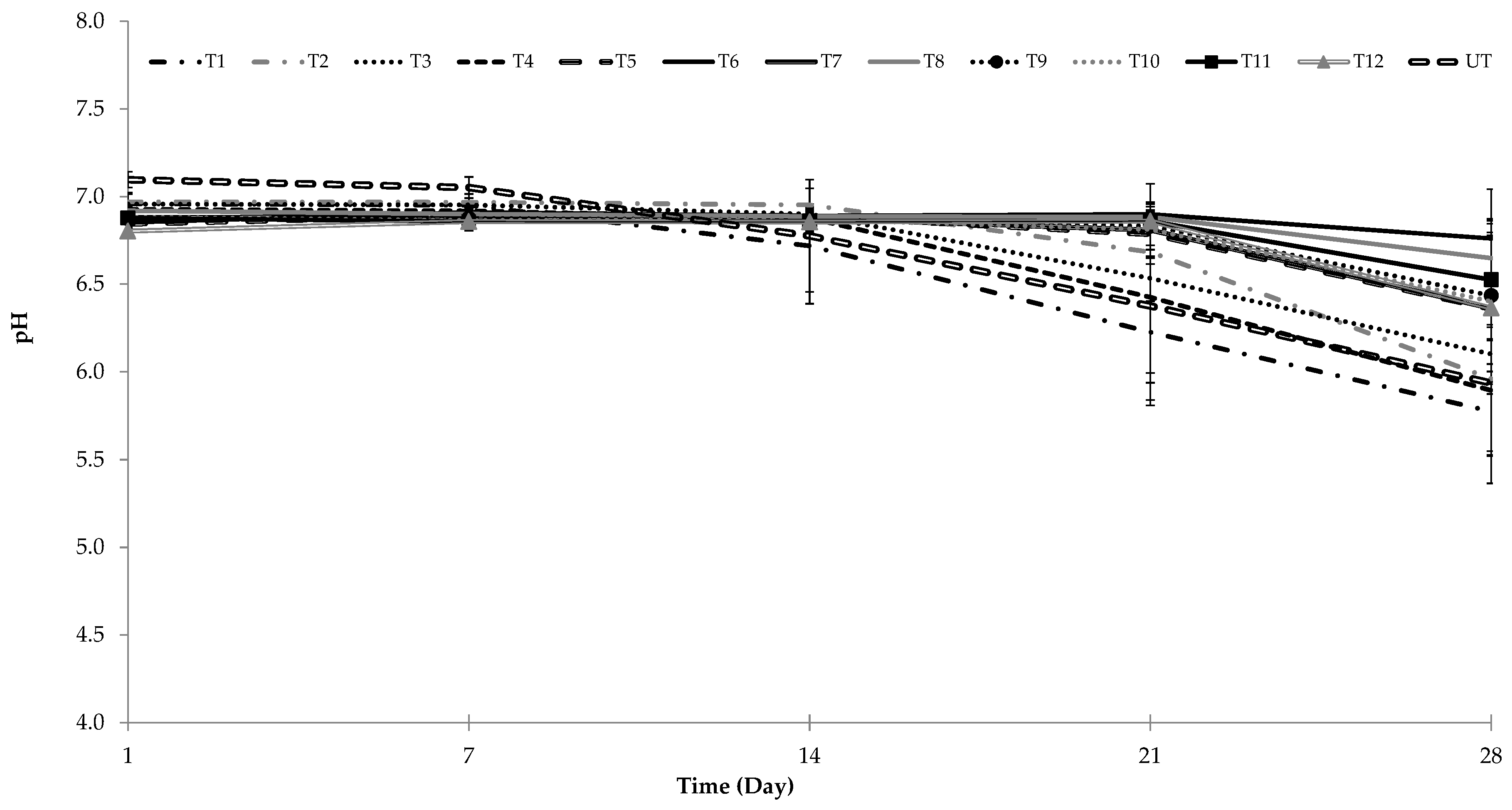
3.3. Effect of UHPH on pH
3.4. Total Solids Content (%) and Comparisons with Commercial Samples
3.5. Effect of UHPH on the Particle Size Distribution (PSD) of Soy Milk
3.6. Visible Layer Separation
3.7. Effect of UHPH on the Lipoxygenase (LOX) Activity in Soy Milk
3.8. Effect of UHPH on the Sensory Attributes of Soy Milk
- Silk: Calcium Carbonate, Sea Salt, Flavors, Gum, Vitamins
- SoyCow: Emulsifier
- Vita Soy: Tricalcium Phosphate, Salt, Zinc Oxide, Vitamins
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| APC | Aerobic Plate Counts |
| CFU | Colony Forming Units |
| CHP | Continuous High Pressure |
| CFHPT | Continuous Flow High Pressure Throttling |
| DW | Deionized Water |
| HDPE | High Density Polyethylene |
| HHP | High Hydrostatic Pressure |
| HPP | High Pressure Processing |
| LOX | Lipoxygenase |
| PSD | Particle Size Distribution |
| RH | Relative Humidity |
| RI | Refractive Index |
| UHPH | Ultra High Pressure Homogenization |
| UT | Untreated |
References
- Diels, A.M.J.; Michiels, C.W. High-pressure homogenization as a non-thermal technique for the inactivation of microorganisms. Crit. Rev. Microbiol. 2006, 33, 201–216. [Google Scholar] [CrossRef] [PubMed]
- Yaldagard, M.; Mortazavi, S.A.; Tabatabaie, F. The principles of ultra high pressure technology and its application in food processing/preservation: A review of microbiological and quality aspects. Afr. J. Biotechnol. 2008, 7, 2739–2767. [Google Scholar]
- Martin, S.; Barbosa-Cánovas, G.V.; Swanson, B.G. Food processing by high hydrostatic pressure. Crit. Rev. Food Sci. Nutr. 2002, 42, 627–645. [Google Scholar] [CrossRef] [PubMed]
- Dumay, E.; Chevalier-Lucia, D.; Picart-Palmade, L.; Benzaria, A.; Gracia-Julia, A.; Blayo, C. Technological aspects and potential applications of (ultra) high-pressure homogenisation. Trends Food Sci. Technol. 2013, 31, 13–26. [Google Scholar] [CrossRef]
- Adapa, S.; Schmidt, K.A.; Toledo, R. Functional properties of skim milk processed with continuous high pressure throttling. J. Dairy Sci. 1997, 80, 1941–1948. [Google Scholar] [CrossRef]
- Sivanandan, L.; Toledo, R.T.; Singh, R.K. Effect of continuous flow high-pressure throttling on rheological and ultrastructural properties of soymilk. J. Food Sci. 2008, 73, E288–E296. [Google Scholar] [CrossRef] [PubMed]
- Toledo, R.T.; Moorman, J.E. Microbial Inactivation by High-Pressure Throttling. U.S. Patent 6120732, 19 September 2000. [Google Scholar]
- Popper, L.; Knorr, D. Applications of high-pressure homogenization for food preservation. Food Technol. 1990, 44, 84–89. [Google Scholar]
- Areekul, V. High Pressure Sterilization of Honey: Physicochemical Changes, Sensory Attributes and Shelf-Life. Ph.D. Thesis, Department of Food Science and Technology, University of Georgia, Athens, GA, USA, 2003. [Google Scholar]
- Cavender, G.A.; Kerr, W.L. Inactivation of vegetative cells by continuous high-pressure processing: New insights on the contribution of thermal effects and release device. J. Food Sci. 2011, 76, E525–E529. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Singh, R.K.; Toledo, R.T. Microbial inactivation kinetics in soymilk during continuous flow high-pressure throttling. J. Food Sci. 2009, 74, M268–M275. [Google Scholar] [CrossRef] [PubMed]
- Lo, W.Y.-L.; Steinkraus, K.H.; Hand, D.B.; Hackler, L.R.; Wilkens, W.F. Soaking soybeans before extraction as it affects composition & yield of soymilk. Food Technol. 1968, 33, 1188–1190. [Google Scholar]
- Nelson, A.I.; Steinberg, M.P.; Wei, L.S. Illinois process for preparation of soymilk. J. Food Sci. 1976, 41, 57–61. [Google Scholar] [CrossRef]
- Malaki Nik, A.M.; Tosh, S.M.; Poysa, V.; Woodrow, L.; Corredig, M. Physicochemical characterization of soymilk after step-wise centrifugation. Food Res. Int. 2008, 41, 286–294. [Google Scholar] [CrossRef]
- Poliseli-Scopel, F.H.; Hernandez-Herrero, M.; Guamis, B.; Ferragut, V. Comparison of ultra high pressure homogenization and conventional thermal treatments on the microbiological, physical and chemical quality of soymilk. LWT Food Sci. Technol. 2012, 46, 42–48. [Google Scholar] [CrossRef]
- Van der Ven, C.; Master, A.M.; van den Berg, R.W. Inactivation of soybean trypsin inhibitors and lipoxygenase by high-pressure processing. J. Agric. Food Chem. 2005, 53, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhou, X.; Chen, Z. High pressure inactivation of lipoxygenase in soy milk and crude soybean extract. Food Chem. 2008, 106, 603–611. [Google Scholar] [CrossRef]
- Poliseli-Scopel, F.H.; Hernández-Herrero, M.; Guamis, B.; Ferragut, V. Characteristics of soymilk pasteurized by ultra high pressure homogenization (UHPH). Innov. Food Sci. Emerg. Technol. 2013, 20, 73–80. [Google Scholar] [CrossRef]
- Smith, K.; Mendonca, A.; Jung, S. Impact of high-pressure processing on microbial shelf-life and protein stability of refrigerated soymilk. Food Microbiol. 2009, 26, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Black, E.P.; Setlow, P.; Hocking, A.D.; Stewart, C.M.; Kelly, A.L.; Hoover, D.G. Response of spores to high-pressure processing. Compr. Rev. Food Sci. Food Saf. 2007, 6, 103–119. [Google Scholar] [CrossRef]
- Wang, B.; Xiong, Y.L.; Wang, C. Physicochemical and sensory characteristics of flavored soymilk during refrigeration storage. J. Food Qual. 2001, 24, 513–526. [Google Scholar] [CrossRef]
- Poliseli-Scopel, F.H.; Hernández-Herrero, M.; Guamis, B.; Ferragut, V. Sterilization and aseptic packaging of soymilk treated by ultra high pressure homogenization. Innov. Food Sci. Emerg. Technol. 2014, 22, 81–88. [Google Scholar] [CrossRef]
- Horiba Scientific. A Guidebook to Particle Size Analysis; Horiba Instruments, Inc.: Irvine, CA, USA, 2012. [Google Scholar]
- Malvern Instruments. A Basic Guide to Particle Characterization; MRK1806-01; Malvern Instruments Limited: Worcestershire, UK, 2012. [Google Scholar]
- Floury, J.; Bellettre, J.; Legrand, J.; Desrumaux, A. Analysis of a new type of high pressure homogeniser. A study of the flow pattern. Chem. Eng. Sci. 2004, 59, 843–853. [Google Scholar] [CrossRef]
- Floury, J.; Desrumaux, A.; Legrand, J. Effect of ultra-high-pressure homogenization on structure and on rheological properties of soy protein-stabilized emulsions. J. Food Sci. 2002, 67, 3388–3395. [Google Scholar] [CrossRef]
- Hayes, M.G.; Fox, P.F.; Kelly, A.L. Potential applications of high pressure homogenisation in processing of liquid milk. J. Dairy Res. 2005, 72, 25–33. [Google Scholar] [CrossRef] [PubMed]
- De Heij, W.B.C.; van Schepdael, L.J.M.M.; Moezelaar, R.; Hoogland, H.; Master, A.M.; van den Berg, R.W. High-pressure sterilization: Maximizing the benefits of adiabatic heating. Food Technol. 2003, 57, 37–41. [Google Scholar]
- Cruz, N.; Capellas, M.; Hernandez, M.; Trujillo, A.J.; Guamis, B.; Ferragut, V. Ultra high pressure homogenization of soymilk: Microbiological, physicochemical & microstructural characteristics. Food Res. Int. 2007, 40, 725–732. [Google Scholar]
- Smiddy, M.A.; Martin, J.-E.; Huppertz, T.; Kelly, A.L. Microbial shelf-life of high-pressure-homogenised milk. Int. Dairy J. 2007, 17, 29–32. [Google Scholar] [CrossRef]
- Toda, K.; Chiba, K.; Ono, T. Effect of components extracted from okara on the physicochemical properties of soymilk and tofu texture. J. Food Sci. 2007, 72, C108–C113. [Google Scholar] [CrossRef] [PubMed]
- Hayes, M.G.; Kelly, A.L. High pressure homogenisation of raw whole bovine milk (a) effects on fat globule size and other properties. J. Dairy Res. 2003, 70, 279–305. [Google Scholar] [CrossRef]
- Pereda, J.; Ferragut, V.; Quevedo, J.M.; Guamis, B.; Trujillo, A.J. Effects of ultra-high pressure homogenization on microbial and physicochemical shelf life of milk. J. Dairy Sci. 2007, 90, 1081–1093. [Google Scholar] [CrossRef]
- Farkas, D.F.; Hoover, D.G. High pressure processing. J. Food Sci. 2000, 65, 47–64. [Google Scholar] [CrossRef]
- Lakshmanan, R.; De Lamballerie, M.; Jung, S. Effect of soybean-to-water ratio and pH on pressurized soymilk properties. J. Food Sci. 2006, 71, E384–E391. [Google Scholar] [CrossRef]
- Zhang, H.; Li, L.; Tatsumi, E.; Isobe, S. High-pressure treatment effects on proteins in soy milk. LWT Food Sci. Technol. 2005, 38, 7–14. [Google Scholar] [CrossRef]
- Achouri, A.; Boye, J.I.; Zamani, Y. Changes in soymilk quality as a function of composition and storage. J. Food Qual. 2007, 30, 731–744. [Google Scholar] [CrossRef]
- Thiebaud, M.; Dumay, E.; Picart, L.; Guiraud, J.P.; Cheftel, J.C. High-pressure homogenisation of raw bovine milk. Effects on fat globule size distribution and microbial inactivation. Int. Dairy J. 2003, 13, 427–439. [Google Scholar] [CrossRef]
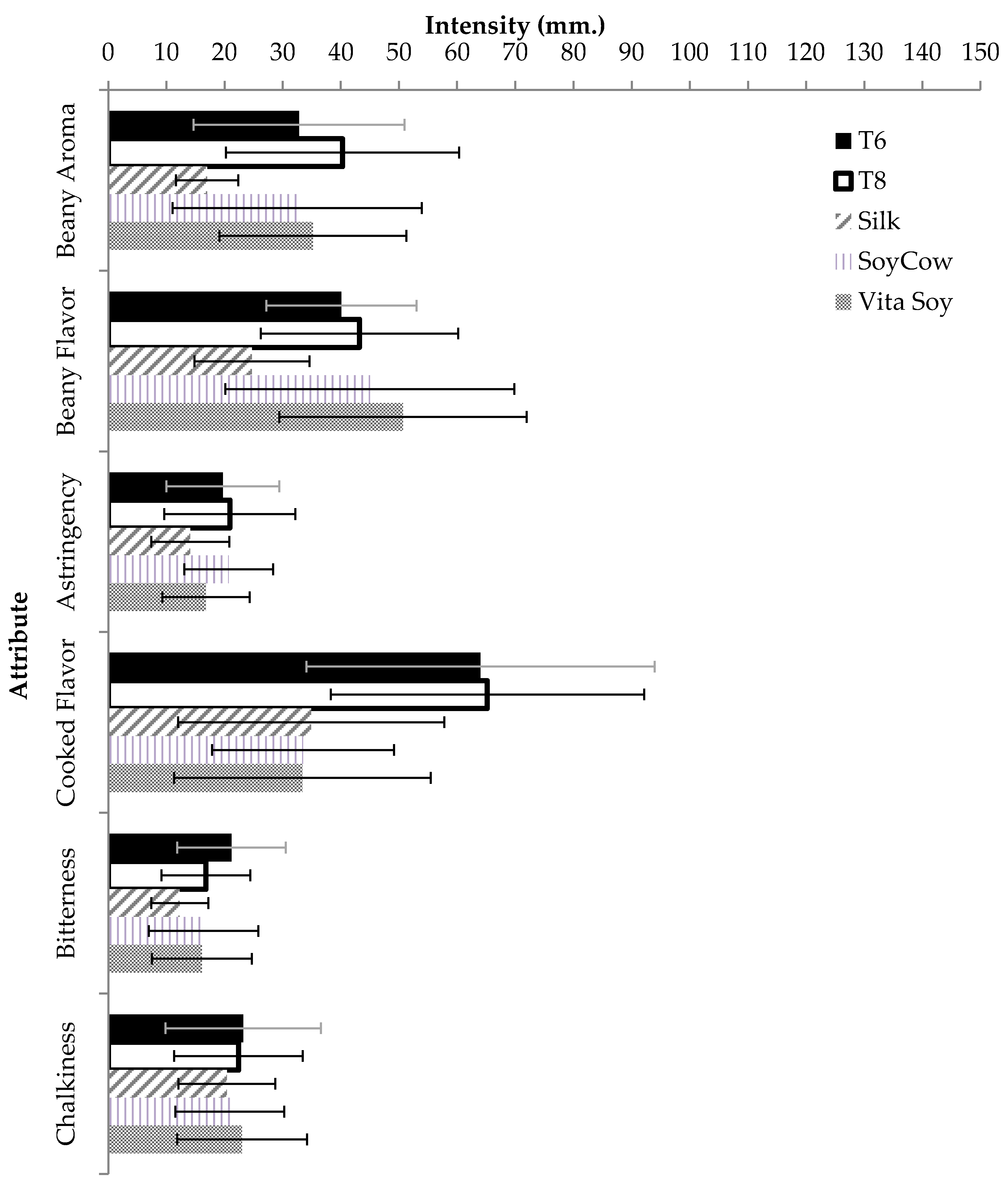
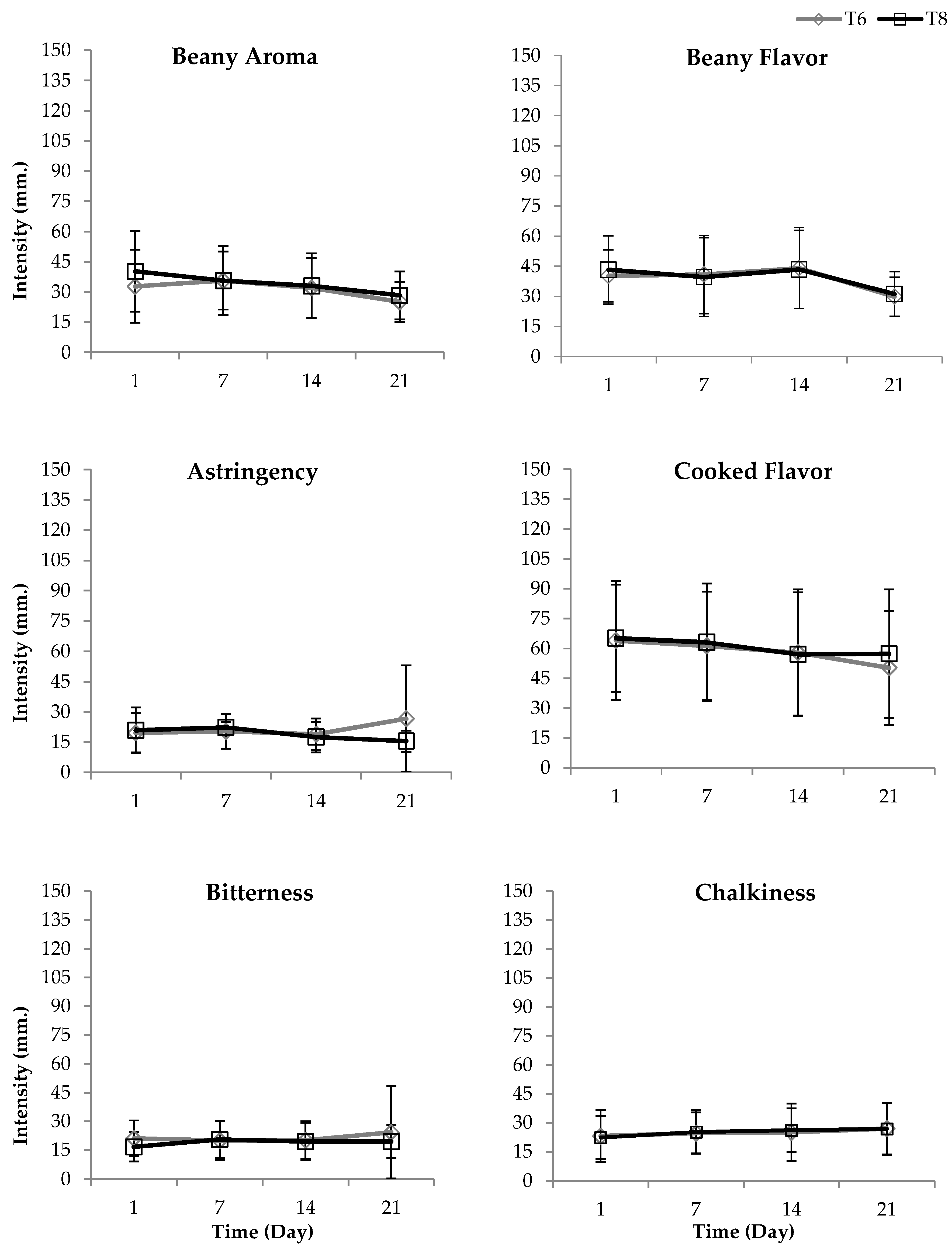
| Attribute | Reference Sample | Preparation Method | Intensity (mm) |
|---|---|---|---|
| Beany Aroma | Raw soybeans soaked in deionized water for 16 h (1:12 w/w) | Drained and ground with deionized water (1:4 w/w) | 60 |
| Beany Flavor | Same as Beany Aroma | Same as Beany Aroma | 60 |
| Astringency | Alum Powder a | 0.01% solution in water b | 20 |
| Cooked Flavor | Evaporated Milk c | Diluted with water (1:6 w/w) | 45 |
| Bitterness | Caffeine d | 0.03% solution in water | 20 |
| Chalkiness | Protein Juice e | Diluted with water (1:4 w/w) | 55 |
| T. No. b | Pressure (MPa) | Heat Ex. c Temp. (°C) | Temp. Rise (°C) | Exit d Temp. (°C) | Rise Per Unit Applied Pressure (°C/MPa) | Residence Time (s) | Temp. After Holding Tube (°C) |
|---|---|---|---|---|---|---|---|
| T1 e | 207 | 26.28 (0.025) | 46.23 (0.325) | 72.51 (0.300) | 0.22 (0.002) | 20.80 | 68.68 (0.525) |
| T2 e | 207 | 27.25 (0.750) | 44.90 (2.300) | 72.15 (1.550) | 0.22 (0.011) | 12.48 | 70.08 (0.725) |
| T3 e | 276 | 26.98 (0.375) | 59.35 (0.550) | 86.33 (0.925) | 0.22 (0.002) | 20.80 | 82.28 (0.225) |
| T4 e | 276 | 29.10 (1.500) | 58.05 (0.950) | 87.15 (0.550) | 0.21 (0.003) | 12.48 | 85.30 (0.300) |
| T5 f | 276 | 72.20 (4.700) | 53.50 (7.800) | 125.70 (3.100) | 0.19 (0.028) | 20.80 | 121.15 (0.300) |
| T6 f | 276 | 59.85 (8.350) | 64.85 (13.050) | 124.70 (4.700) | 0.23 (0.047) | 12.48 | 121.38 (1.375) |
| T7 f | 207 | 75.50 (2.200) | 53.35 (3.350) | 128.85 (5.550) | 0.26 (0.016) | 20.80 | 120.98 (0.875) |
| T8 f | 207 | 72.25 (3.750) | 61.25 (6.250) | 133.50 (2.500) | 0.30 (0.030) | 12.48 | 122.30 (0.400) |
| T9 g | 276 | 84.40 (1.400) | 63.95 (2.750) | 148.35 (1.350) | 0.23 0.010) | 20.80 | 144.15 (1.550) |
| T10 g | 276 | 84.60 (5.600) | 59.80 (7.000) | 144.40 (1.400) | 0.22 (0.025) | 12.48 | 146.65 (1.250) |
| T11 g | 207 | 95.45 (1.250) | 50.85 (1.550) | 146.30 (0.300) | 0.25 (0.008) | 20.80 | 141.88 (0.075) |
| T12 g,h | 207 | 93.40 (0.400) | 52.30 (1.900) | 145.70 (2.300) | 0.25 (0.009) | 12.48 | 143.80 (1.300) |
| UT h | - | - | - | - | - | - | - |
| Sample | Mean (%) | SD (%) |
|---|---|---|
| T6 b | 8.71 | 0.045 |
| T8 c | 8.78 | 0.170 |
| Silk | 7.03 | 0.085 |
| SoyCow | 3.77 | 0.030 |
| Vita Soy | 6.89 | 0.045 |
| T. No. b | Temp. c (°C) | Residence Time (s) | Pressure (kPa) | D[4,3] d (µm) | D[3,2] e (µm) | D(v,0.9) f (µm) |
|---|---|---|---|---|---|---|
| UT g | - | - | - | 129.86 (10.659) | 17.04 (0.690) | 335.90 (30.196) |
| T1 | No heating | 20.80 | 207 | 23.34 (3.903) | 10.60 (0.940) | 46.90 (8.560) |
| T2 | No heating | 12.48 | 207 | 25.21 (3.076) | 10.63 (0.820) | 51.13 (6.247) |
| T3 | No heating | 20.80 | 276 | 20.91 (1.131) | 10.75 (0.757) | 40.90 (2.885) |
| T4 | No heating | 12.48 | 276 | 19.74 (1.294) | 9.54 (0.350) | 39.21 (3.002) |
| T5 | 121 | 20.80 | 276 | 22.44 (2.517) | 12.63 (0.841) | 43.70 (5.469) |
| T6 | 121 | 12.48 | 276 | 23.47 (0.240) | 12.31 (1.039) | 46.70 (0.106) |
| T7 | 121 | 20.80 | 207 | 22.60 (1.584) | 12.38 (0.686) | 43.51 (3.140) |
| T8 | 121 | 12.48 | 207 | 26.24 (2.058) | 13.91 (1.336) | 51.98 (4.179) |
| T9 | 145 | 20.80 | 276 | 23.63 (6.039) | 13.59 (1.937) | 46.53 (12.459) |
| T10 | 145 | 12.48 | 276 | 28.25 (8.775) | 14.34 (1.648) | 56.39 (19.958) |
| T11 | 145 | 20.80 | 207 | 30.19 (3.543) | 17.08 (1.478) | 58.35 (7.266) |
| T12 | 145 | 12.48 | 207 | 30.10 (7.6374) | 16.14 (0.778) | 58.65 (14.711) |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sidhu, J.S.; Singh, R.K. Ultra High Pressure Homogenization of Soy Milk: Effect on Quality Attributes during Storage. Beverages 2016, 2, 15. https://doi.org/10.3390/beverages2020015
Sidhu JS, Singh RK. Ultra High Pressure Homogenization of Soy Milk: Effect on Quality Attributes during Storage. Beverages. 2016; 2(2):15. https://doi.org/10.3390/beverages2020015
Chicago/Turabian StyleSidhu, Jaideep S., and Rakesh K. Singh. 2016. "Ultra High Pressure Homogenization of Soy Milk: Effect on Quality Attributes during Storage" Beverages 2, no. 2: 15. https://doi.org/10.3390/beverages2020015






