Performance of Hollow Fiber Ultrafiltration Membranes in the Clarification of Blood Orange Juice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Blood Orange Juice
2.2. Ultrafiltration Experimental Setup
| Membrane Type | DCQ II-006C-PS50 | DCQ II-006C-PS100 | DCQ II-006C-PAN50 |
|---|---|---|---|
| Membrane material | Polysulfone | Polysulfone | Polyacrylonitrile |
| Internal diameter of fibers (mm) | 2.1 | 2.1 | 2.1 |
| Operating pressure (bar) | 0–1.5 | 0–1.5 | 0–1.2 |
| Operating temperature (°C) | 5–45 | 5–45 | 5–45 |
| Operating pH | 2–13 | 2–13 | 2–10 |
| Molecular weight cut-off (kDa) | 50 | 100 | 50 |
2.3. Analytical Methods
2.3.1. Physicochemical Analysis
2.3.2. Total Phenolic Content (TPC)
2.3.3. Total Antioxidant Activity (TAA)
2.3.4. Anthocyanins
2.4. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Properties of Blood Orange Juice
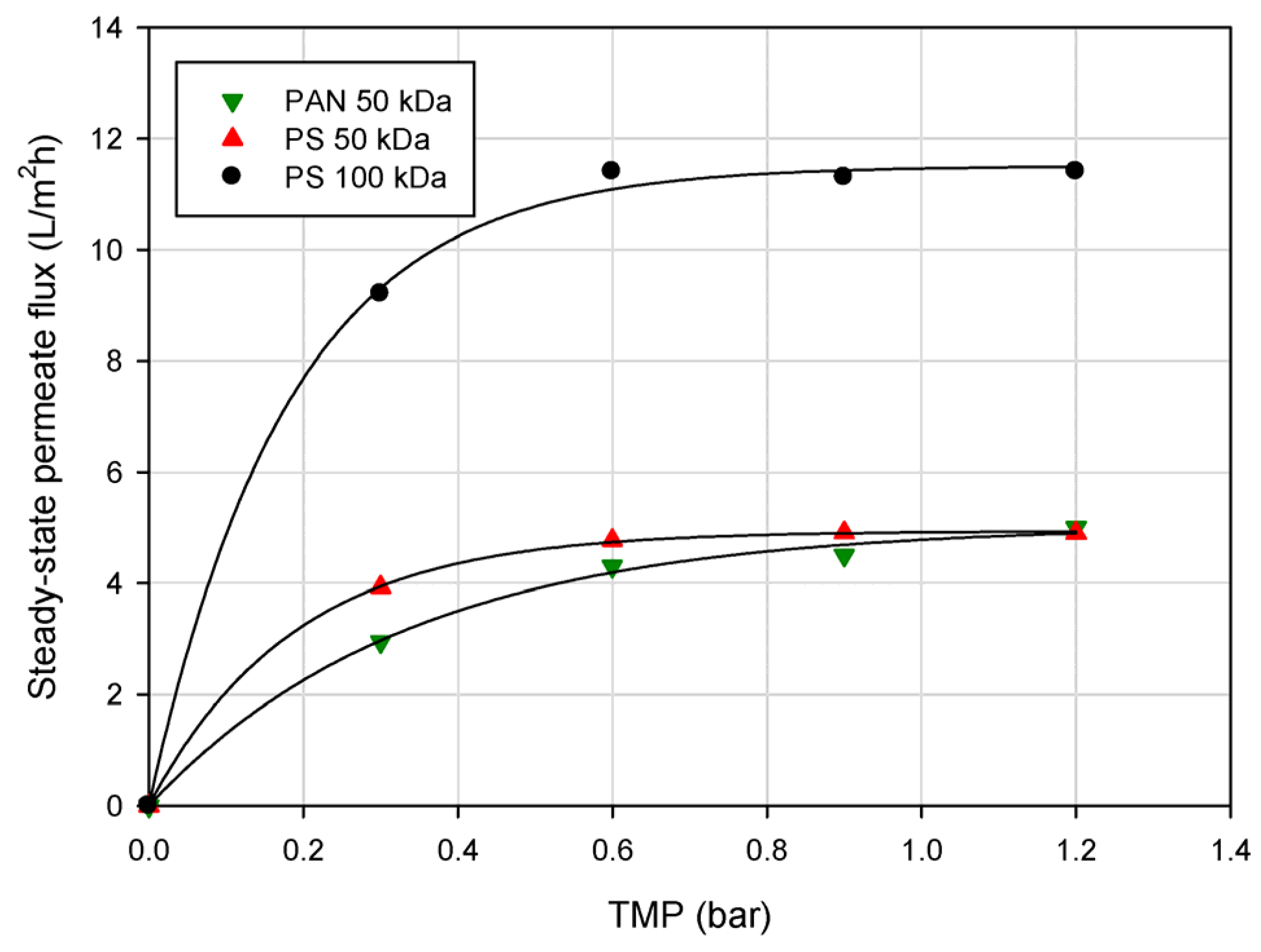
3.2. Effect of Operating Conditions on Permeate Flux
| pH | 3.5 ± 0.1 |
|---|---|
| TSS (°Brix) | 10.2 ± 0.2 |
| Suspended solids (%, w/w) | 12.0 ± 0.2 |
| Viscosity (mPa s) | 1.90 ± 0.04 |
| TPC (mg GAE/L) | 907.22 ± 66.34 |
| TAA (mM Trolox) | 6.50 ± 0.46 |
| TAC (mg/L) | 7.56 ± 0.87 |
| Cyanidin-3-glucoside (mg/L) | 5.98 ± 0.75 |
| Cyanidin-3,5-diglucoside (mg/L) | 0.20 ± 0.04 |
| Delfinidin-3-glucoside (mg/L) | 0.84 ± 0.15 |
| Peonidin-3-glucoside (mg/L) | 0.54 ± 0.01 |
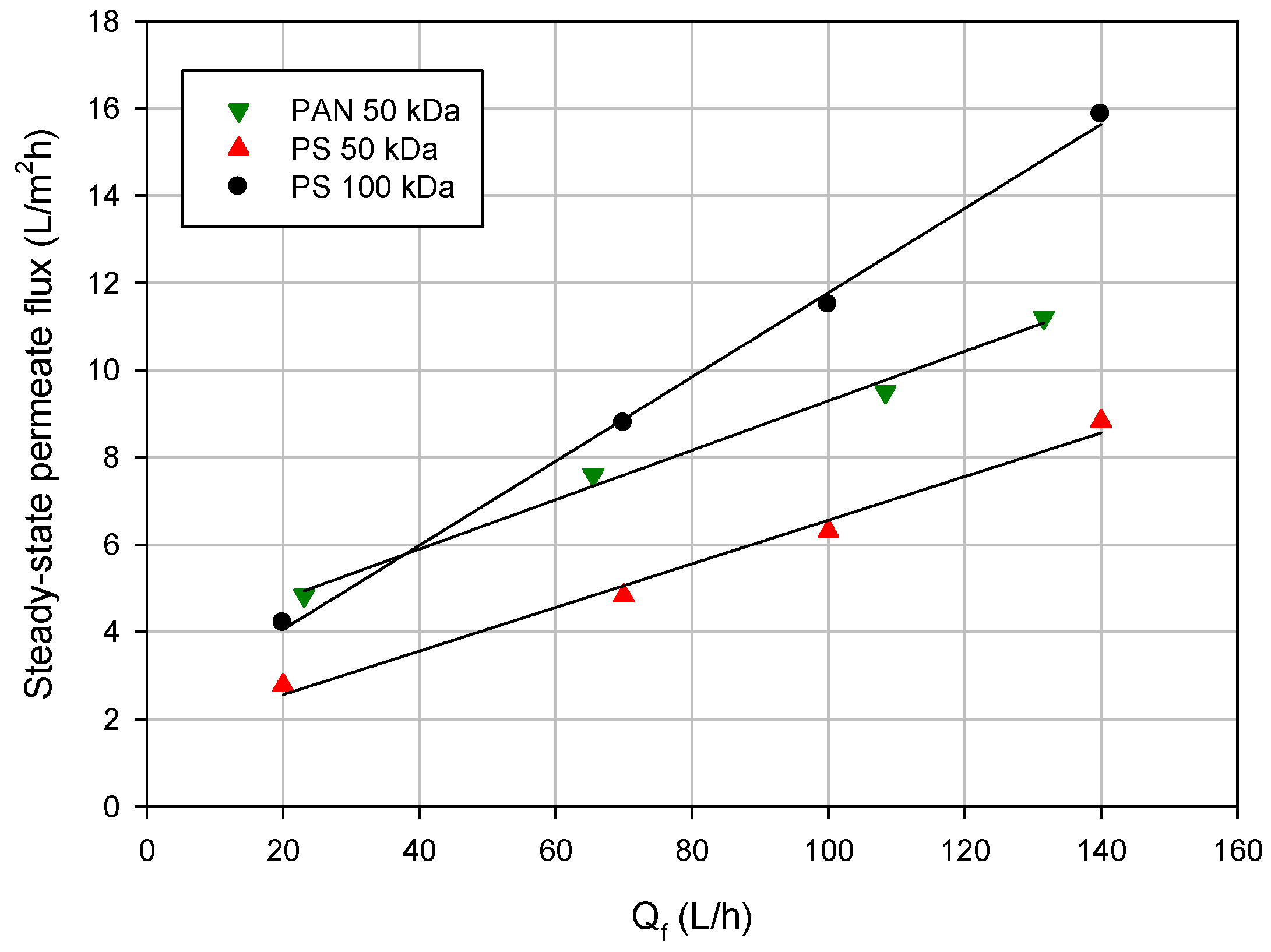
3.3. Effect of Volume Reduction Factor on Permeate Flux
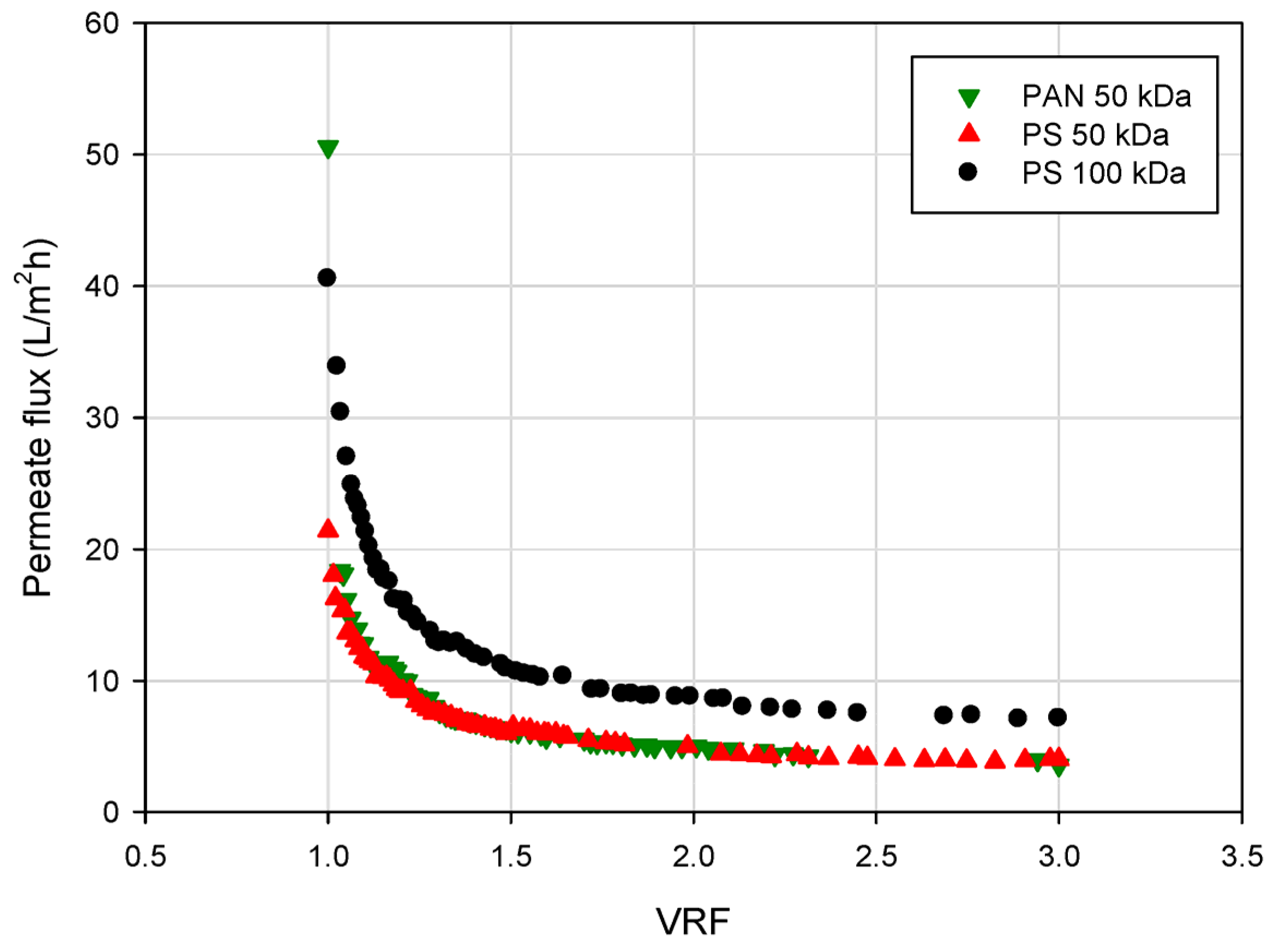
3.4. Effect of UF on Chemical Parameters of Blood Orange Juice
| UF Membrane | Sample | TSS (° Brix) | TPC (mg GAE/L) | TAC (mg/L) | TAA (mM Trolox) |
|---|---|---|---|---|---|
| PS 100 kDa | Feed | 10.0 ± 0.2 a | 835.42 ± 0.43 a | 7.33 ± 0.14 a | 6.0 ± 0.8 a,b |
| Permeate | 9.8 ± 0.2 a | 687.08 ± 0.22 b | 6.79 ± 0.13 b | 5.1 ± 0.2 a | |
| Retentate | - | 950.83 ± 0.21 c | 7.71 ± 0.15 a | 7.1 ± 1.2 b | |
| PS 50 kDa | Feed | 10.2 ± 0.2 a | 920.00 ± 0.47 a | 6.83 ± 0.13 a | 6.6 ± 0.5 a |
| Permeate | 10.0 ± 0.2 a | 705.00 ± 0.34 b | 6.20 ± 0.12 b | 5.1 ± 0.3 b | |
| Retentate | - | 1217.08 ± 0.82 c | 7.02 ± 0.14 a | 8.9 ± 1.0 c | |
| PAN 50 kDa | Feed | 10.4 ± 0.2 a | 966.25 ± 0.27 a | 8.52 ± 0.17 a | 6.9 ± 0.9 a |
| Permeate | 10.2 ± 0.2 a | 738.33 ± 0.19 b | 8.03 ± 0.16 b | 5.3 ± 0.4 b | |
| Retentate | - | 1095.80 ± 0.70 c | 8.80 ± 0.17 a | 7.8 ± 0.8 a |
| UF Membrane | Rejection (%) | |||
|---|---|---|---|---|
| TSS | TPC | TAC | TAA | |
| PS 100 kDa | 2.0 ± 0.2 a | 17.75 ± 0.43 a | 7.4 ± 0.14 a | 15.0 ± 0.8 a |
| PS 50 kDa | 1.9 ± 0.2 a | 23.37 ± 0.47 b | 9.2 ± 0.13 b | 22.7 ± 0.5 b |
| PAN 50 kDa | 1.9 ± 0.2 a | 23.60 ± 0.30 b | 5.7 ± 0.17 c | 23.2 ± 0.9 b |
| Membrane type | Anthocyanins | Feed (mg/L) | Permeate (mg/L) | Retentate (mg/L) |
|---|---|---|---|---|
| PS 100kDa | Cyanidin-3,5-diglucoside | 0.24 ± 0.01 a | 0.20 ± 0.01 b | 0.29 ± 0.01 c |
| Cyanidin-3-glucoside | 5.64 ± 0.11 a | 5.23 ± 0.10 b | 6.00 ± 0.12 c | |
| Delfinidin-3-glucoside | 0.91 ± 0.01 a | 0.90 ± 0.01 ab | 0.88 ± 0.01 b | |
| Peonidin-3-glucoside | 0.54 ± 0.01 a | 0.46 ± 0.01 b | 0.54 ± 0.01 a | |
| PS 50kDa | Cyanidin-3,5-diglucoside | 0.17 ± 0.01 a | 0.16 ± 0.01 a | 0.18 ± 0.01 a |
| Cyanidin-3-glucoside | 5.46 ± 0.10 a | 4.98 ± 0.10 b | 5.56 ± 0.11 a | |
| Delfinidin-3-glucoside | 0.67 ± 0.01 a | 0.60 ± 0.01 b | 0.70 ± 0.01 a | |
| Peonidin-3-glucoside | 0.53 ± 0.01 a | 0.46 ± 0.01 b | 0.58 ± 0.01 c | |
| PAN 50kDa | Cyanidin-3,5-diglucoside | 0.19 ± 0.01 a | 0.18 ± 0.01 a | 0.24 ± 0.01 b |
| Cyanidin-3-glucoside | 6.84 ± 0.13 a | 6.45 ± 0.13 b | 6.94 ± 0.13 a | |
| Delfinidin-3-glucoside | 0.95 ± 0.02 a | 0.90 ± 0.02 b | 0.98 ± 0.02 a | |
| Peonidin-3-glucoside | 0.54 ± 0.01 a | 0.50 ± 0.01 b | 0.64 ± 0.01 c |
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Mondello, L.; Cotroneo, A.; Errante, G.; Dugo, G.; Dugo, P. Determination of anthocyanins in blood orange juices by HPLC analysis. J. Pharm. Biomed. Anal. 2000, 23, 191–195. [Google Scholar] [CrossRef]
- Dugo, P.; Mondello, L.; Morabito, D.; Dugo, G. Characterization of the anthocyanins fraction of Sicilian blood orange juice by micro-HPLC ESI/MS. J. Agric. Food Chem. 2003, 51, 1173–1176. [Google Scholar] [CrossRef] [PubMed]
- Maccarone, E.; Rapisarda, P.; Fanella, F.; Arena, E.; Mondello, L. Cyanidin-3-(6′′-malonyl)-glucoside. An important anthocyanin of blood orange juice. Ital. J. Food Sci. 1998, 10, 367–372. [Google Scholar]
- Arena, E.; Fallico, B.; Maccarone, E. Evaluation of antioxidant capacity of blood orange juices as influenced by constituents, concentration process and storage. Food Chem. 2001, 74, 423–427. [Google Scholar] [CrossRef]
- Wang, H.; Cao, G.; Prior, R.L. Oxygen radical absorbing capacity of anthocyanins. J. Agric. Food Chem. 1997, 45, 304–309. [Google Scholar] [CrossRef]
- Lo Scalzo, R.; Iannoccari, T.; Summa, C.; Morelli, R.; Rapisarda, P. Effect of thermal treatments on antioxidant and antiradical activity of blood orange juice. Food Chem. 2004, 85, 41–47. [Google Scholar] [CrossRef]
- Rapisarda, P.; Tomaino, A.; Lo Cascio, R.; Bonina, F.; de Pasquale, A.; Saija, A. Antioxidant effectiveness as influenced by phenolic content of fresh orange juice. J. Agric. Food Chem. 1999, 47, 4718–4723. [Google Scholar] [CrossRef] [PubMed]
- Scordino, M.; Sabatino, L.; Lazzaro, F.; Borzi, M.A.; Gargano, M.; Traulo, P.; Gagliano, G. Blood orange anthocyanins in fruit beverages: How the commercial shelf life reflects the quality parameter. Beverages 2015, 1, 82–94. [Google Scholar] [CrossRef]
- Rai, P.; De, S. Clarification of pectin-containing juice using ultrafiltration. Curr. Sci. 2009, 96, 1361–1371. [Google Scholar]
- Girard, B.; Fukumoto, L.R. Membrane processing of fruit juices and beverages: A review. Crit. Rev. Food Sci. Nutr. 2000, 40, 91–157. [Google Scholar] [CrossRef] [PubMed]
- Cassano, A.; Donato, L.; Drioli, E. Ultrafiltration of kiwifruit juice: Operating parameters. Juice quality and membrane fouling. J. Food Eng. 2007, 79, 613–621. [Google Scholar] [CrossRef]
- Rai, P.; Majumdar, G.C.; Sharma, G.; DasGupta, S.; De, S. Effect of various cutoff membranes on permeate flux and quality during filtration of mosambi (Citrus sinensis L.) Osbeck juice. Food Bioprod. Process. 2006, 84, 213–219. [Google Scholar] [CrossRef]
- Vladisavljevíc, G.T.; Vukosavljevíc, P.; Bukvíc, B. Permeate flux and fouling resistance in ultrafiltration of depectinized apple juice using ceramic membranes. J. Food Eng. 2003, 60, 241–247. [Google Scholar] [CrossRef]
- Hernandez, E.; Chen, C.S.; Shaw, P.E.; Carter, R.D.; Barros, S. Ultrafiltration of orange juice: Effect on soluble solids, suspended solids, and aroma. J. Agric. Food Chem. 1992, 40, 986–988. [Google Scholar] [CrossRef]
- Cassano, A.; Marchio, M.; Drioli, E. Clarification of blood orange juice by ultrafiltration: Analyses of operating parameters, membrane fouling and juice quality. Desalination 2007, 212, 15–27. [Google Scholar] [CrossRef]
- Jiraratananon, R.; Chanachai, A. A study of fouling in the ultrafiltration of passion fruit juice. J. Membr. Sci. 1996, 111, 39–48. [Google Scholar] [CrossRef]
- De Barros, S.T.D.; Andrade, C.M.G.; Mendes, E.S.; Peres, L. Study of fouling mechanism in pineapple juice clarification by ultrafiltration. J. Membr. Sci. 2003, 215, 213–224. [Google Scholar] [CrossRef]
- Cassano, A.; Conidi, C.; Drioli, E. Physico-chemical parameters of Cactus Pear (Opuntia Ficus-Indica) juice clarified by microfiltration and ultrafiltration processes. Desalination 2010, 250, 1101–1104. [Google Scholar] [CrossRef]
- Naidu, N.G.S.; Panda, T. Performance of pectolytic enzymes during hydrolysis of pectic substances under assay conditions: A statistical approach. Enzyme Microb. Technol. 1999, 25, 116–124. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. [Google Scholar]
- Rice-Evans, C.A.; Miller, N.J. Total antioxidant status in plasma and body fluids. Methods Enzymol. 1994, 243, 279–298. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C.A. Antioxidant activity applying and improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Destani, F.; Cassano, A.; Fazio, A.; Vincken, J.P.; Gabriele, B. Recovery and concentration of phenolic compounds in blood orange juice by membrane operations. J. Food Eng. 2013, 117, 263–271. [Google Scholar] [CrossRef]
- Ingallinera, B.; Barbagallo, R.N.; Spagna, G.; Palmeri, R.; Todaro, A. Effects of thermal treatments on pectinesterase activity determined in blood oranges juices. Enzyme Microb. Technol. 2005, 36, 258–263. [Google Scholar] [CrossRef]
- Lutz, H. Ultrafiltration: Fundamentals and Engineering. In Comprehensive Membrane Science and Engineering; Drioli, E., Giorno, L., Eds.; Elsevier B.V.: Kidlington, UK, 2010; Volume 2, pp. 115–139. [Google Scholar]
- Nilsson, S.L. Protein fouling of UF membrane: Causes and consequences. J. Membr. Sci. 1990, 52, 121–142. [Google Scholar] [CrossRef]
- Toker, R.; Karhan, M.; Tetik, N.; Turhan, I.; Oziyci, H.R. Effect of ultrafiltration and concentration processes on the physical and chemical composition of blood orange juice. J. Food Process. Preserv. 2014, 38, 1321–1329. [Google Scholar] [CrossRef]
- Laorko, A.; Li, Z.Y.; Tongchitpakdee, S.; Chantachum, S.; Youravong, W. Effect of membrane property and operating conditions on phytochemical properties and permeate flux during clarification of pineapple juice. J. Food Eng. 2010, 100, 514–521. [Google Scholar] [CrossRef]
- Onsekizoglu, P.; Bahceci, K.S.; Acar, M.J. Clarification and the concentration of apple juice using membrane processes: A comparative quality assessment. J. Membr. Sci. 2010, 352, 160–165. [Google Scholar] [CrossRef]
- Roginsky, V.; Lissi, A.E. Review of methods to determine chain breaking antioxidant activity in food. Food Chem. 2005, 92, 235–254. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Conidi, C.; Destani, F.; Cassano, A. Performance of Hollow Fiber Ultrafiltration Membranes in the Clarification of Blood Orange Juice. Beverages 2015, 1, 341-353. https://doi.org/10.3390/beverages1040341
Conidi C, Destani F, Cassano A. Performance of Hollow Fiber Ultrafiltration Membranes in the Clarification of Blood Orange Juice. Beverages. 2015; 1(4):341-353. https://doi.org/10.3390/beverages1040341
Chicago/Turabian StyleConidi, Carmela, Fitim Destani, and Alfredo Cassano. 2015. "Performance of Hollow Fiber Ultrafiltration Membranes in the Clarification of Blood Orange Juice" Beverages 1, no. 4: 341-353. https://doi.org/10.3390/beverages1040341
APA StyleConidi, C., Destani, F., & Cassano, A. (2015). Performance of Hollow Fiber Ultrafiltration Membranes in the Clarification of Blood Orange Juice. Beverages, 1(4), 341-353. https://doi.org/10.3390/beverages1040341







