Body Acoustics for the Non-Invasive Diagnosis of Medical Conditions
Abstract
:1. Introduction
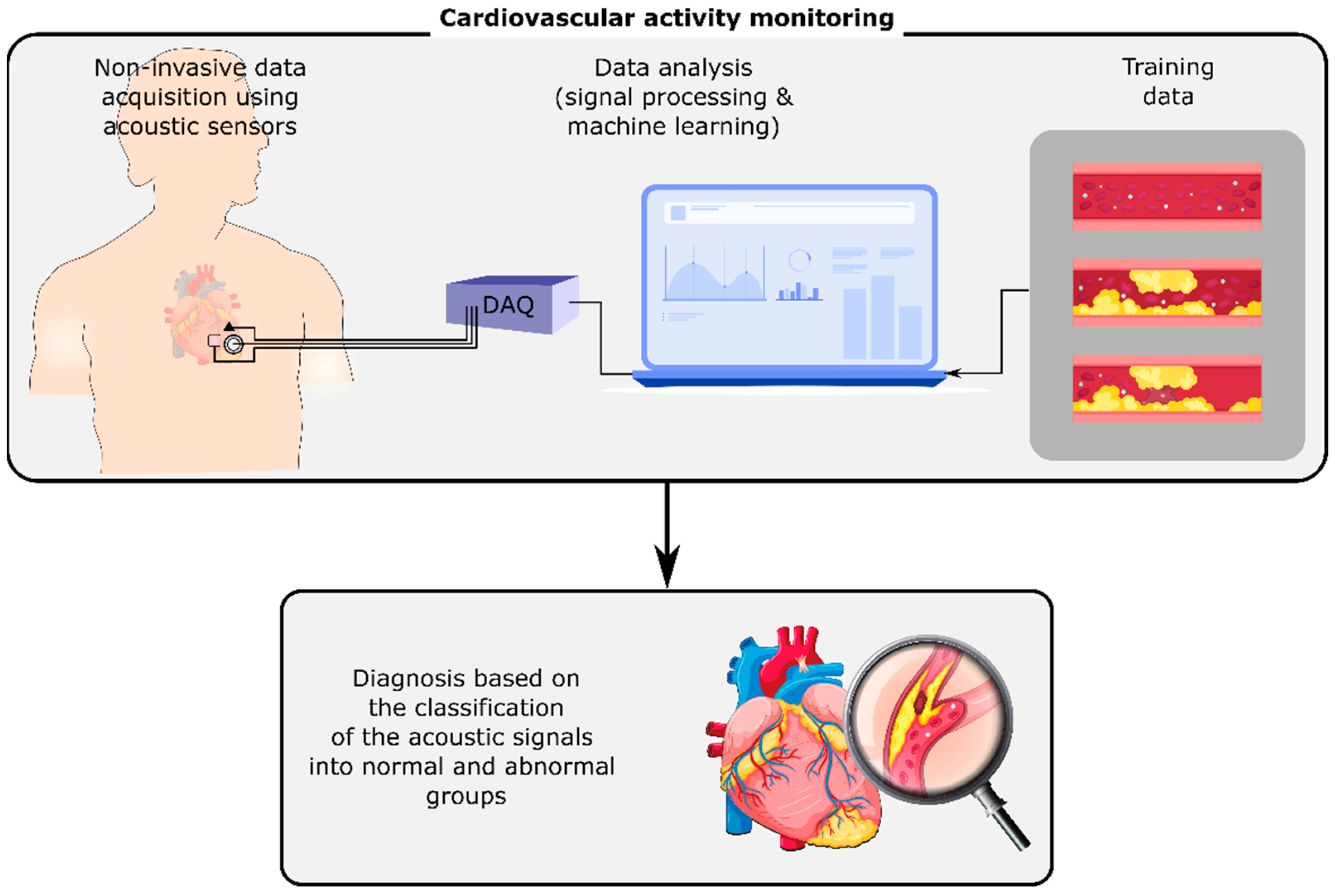
2. Cardiovascular Diseases
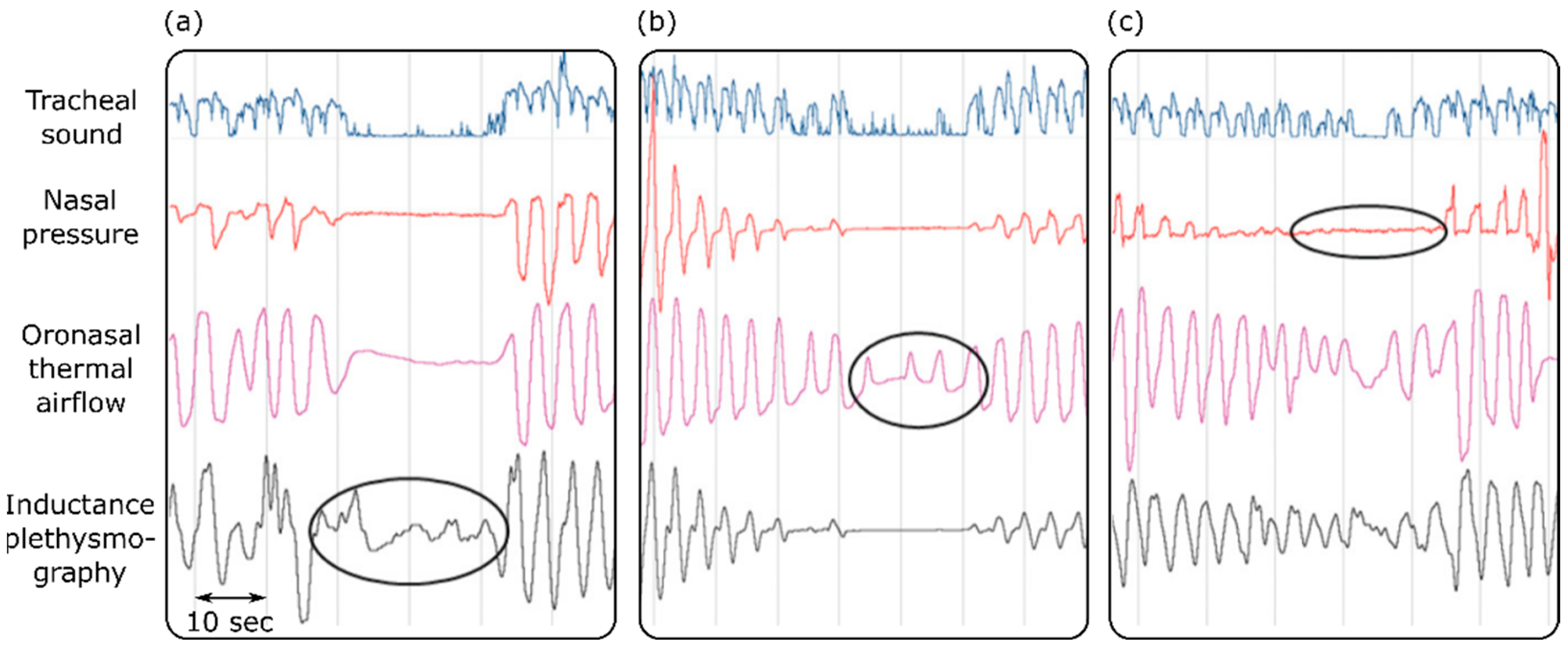
3. Respiratory Illnesses
4. Other Diseases
5. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Murphy, R.L.H. In Defense of the Stethoscope. Respir. Care 2008, 53, 355–369. [Google Scholar] [PubMed]
- Virani, S.S.; Alonso, A.; Aparicio, H.J.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Cheng, S.; Delling, F.N.; et al. Heart Disease and Stroke Statistics—2021 Update. Circulation 2021, 143, e254–e743. [Google Scholar] [CrossRef] [PubMed]
- Celermajer, D.S.; Chow, C.K.; Marijon, E.; Anstey, N.M.; Woo, K.S. Cardiovascular Disease in the Developing World: Prevalences, Patterns, and the Potential of Early Disease Detection. J. Am. Coll. Cardiol. 2012, 60, 1207–1216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, Y.; Kim, E.G.; Cao, G.; Liu, S.; Xu, Y. Physiological Acoustic Sensing Based on Accelerometers: A Survey for Mobile Healthcare. Ann. Biomed. Eng. 2014, 42, 2264–2277. [Google Scholar] [CrossRef]
- Taebi, A.; Solar, B.; Bomar, A.; Sandler, R.; Mansy, H. Recent Advances in Seismocardiography. Vibration 2019, 2, 64–86. [Google Scholar] [CrossRef] [Green Version]
- Inan, O.T.; Migeotte, P.-F.; Park, K.-S.; Etemadi, M.; Tavakolian, K.; Casanella, R.; Zanetti, J.; Tank, J.; Funtova, I.; Prisk, G.K.; et al. Ballistocardiography and Seismocardiography: A Review of Recent Advances. IEEE J. Biomed. Health Inform. 2015, 19, 1414–1427. [Google Scholar] [CrossRef] [Green Version]
- Gupta, P.; Moghimi, M.J.; Jeong, Y.; Gupta, D.; Inan, O.T.; Ayazi, F. Precision wearable accelerometer contact microphones for longitudinal monitoring of mechano-acoustic cardiopulmonary signals. NPJ Digit. Med. 2020, 3, 19. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, S.K.; Tripathy, R.K.; Ponnalagu, R.N.; Pachori, R.B. Automated Detection of Heart Valve Disorders from the PCG Signal Using Time-Frequency Magnitude and Phase Features. IEEE Sens. Lett. 2019, 3, 1–4. [Google Scholar] [CrossRef]
- Chowdhury, T.H.; Poudel, K.N.; Hu, Y. Time-Frequency Analysis, Denoising, Compression, Segmentation, and Classification of PCG Signals. IEEE Access 2020, 8, 160882–160890. [Google Scholar] [CrossRef]
- Yaseen; Son, G.-Y.; Kwon, S. Classification of Heart Sound Signal Using Multiple Features. Appl. Sci. 2018, 8, 2344. [Google Scholar] [CrossRef] [Green Version]
- Saeidi, A.; Almasganj, F. Cardiac valves disorder classification based on active valves appearance periodic sequences tree of murmurs. Biomed. Signal Process. Control 2020, 57, 101775. [Google Scholar] [CrossRef]
- Taebi, A.; Mansy, H.A. Time-Frequency Distribution of Seismocardiographic Signals: A Comparative Study. Bioengineering 2017, 4, 32. [Google Scholar] [CrossRef] [PubMed]
- Taebi, A.; Mansy, H.A. Analysis of Seismocardiographic Signals Using Polynomial Chirplet Transform and Smoothed Pseudo Wigner-Ville Distribution. In Proceedings of the IEEE Signal Processing in Medicine and Biology Symposium (SPMB), Philadelphia, PA, USA, 2 December 2017; pp. 1–6. [Google Scholar]
- Taebi, A.; Mansy, H.A. Effect of Noise on Time-frequency Analysis of Vibrocardiographic Signals. J. Bioeng. Biomed. Sci. 2016, 6, 202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taebi, A.; Solar, B.E.; Mansy, H.A. An Adaptive Feature Extraction Algorithm for Classification of Seismocardiographic Signals. In Proceedings of the IEEE SoutheastCon 2018, St. Petersburg, Russia, 19–22 April 2018; pp. 1–5. [Google Scholar]
- Taebi, A.; Mansy, H.A. Noise Cancellation from Vibrocardiographic Signals Based on the Ensemble Empirical Mode Decomposition. J. Appl. Biotechnol. Bioeng. 2017, 2, 49–54. [Google Scholar] [CrossRef] [Green Version]
- Choudhary, T.; Bhuyan, M.K.; Sharma, L.N. A Novel Method for Aortic Valve Opening Phase Detection Using SCG Signal. IEEE Sens. J. 2020, 20, 899–908. [Google Scholar] [CrossRef]
- Zakeri, V.; Tavakolian, K. Identification of respiratory phases using seismocardiogram: A machine learning approach. Comput. Cardiol. 2015, 42, 305–308. [Google Scholar]
- Taebi, A.; Mansy, H.A. Grouping Similar Seismocardiographic Signals Using Respiratory Information. In Proceedings of the IEEE Signal Processing in Medicine and Biology Symposium (SPMB17), Philadelphia, PA, USA, 2 December 2017; pp. 1–6. [Google Scholar]
- Gamage, P.T.; Azad, M.K.; Taebi, A.; Sandler, R.H.; Mansy, H.A. Clustering of SCG Events Using Unsupervised Machine Learning. In Signal Processing in Medicine and Biology; Springer International Publishing: Cham, Switzerland, 2020; pp. 205–233. [Google Scholar]
- Solar, B.E.; Taebi, A.; Mansy, H.A. Classification of Seismocardiographic Cycles into Lung Volume Phases. In Proceedings of the IEEE Signal Processing in Medicine and Biology Symposium (SPMB), Philadelphia, PA, USA, 2 December 2017; pp. 1–2. [Google Scholar]
- Taebi, A.; Bomar, A.J.; Sandler, R.H.; Mansy, H.A. Heart Rate Monitoring During Different Lung Volume Phases Using Seismocardiography. In Proceedings of the IEEE SoutheastCon 2018, St. Petersburg, Russia, 19–22 April 2018; pp. 1–5. [Google Scholar]
- Sandler, R.H.; Azad, K.; Rahman, B.; Taebi, A.; Gamage, P.; Raval, N.; Mentz, R.J.; Mansy, H.A. Minimizing Seismocardiography Variability by Accounting for Respiratory Effects. J. Card. Fail. 2019, 25, S185. [Google Scholar] [CrossRef] [Green Version]
- Rai, D.; Thakkar, H.K.; Rajput, S.S.; Santamaria, J.; Bhatt, C.; Roca, F. A Comprehensive Review on Seismocardiogram: Current Advancements on Acquisition, Annotation, and Applications. Mathematics 2021, 9, 2243. [Google Scholar] [CrossRef]
- Abbas, A.K.; Bassam, R. Phonocardiography Signal Processing. Synth. Lect. Biomed. Eng. 2009, 4, 1–194. [Google Scholar] [CrossRef]
- Ismail, S.; Siddiqi, I.; Akram, U. Localization and classification of heart beats in phonocardiography signals—A comprehensive review. EURASIP J. Adv. Signal Process. 2018, 2018, 26. [Google Scholar] [CrossRef] [Green Version]
- Zanetti, J.; Salerno, D. Seismocardiography: A new technique for recording cardiac vibrations. Concept, method, and initial observations. J. Cardiovasc. Technol. 1990, 9, 111–120. [Google Scholar]
- Zanetti, J.M.; Tavakolian, K. Seismocardiography: Past, present and future. In Proceedings of the 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; pp. 7004–7007. [Google Scholar]
- Tadi, M.J.; Lehtonen, E.; Saraste, A.; Tuominen, J.; Koskinen, J.; Teräs, M.; Airaksinen, J.; Pänkäälä, M.; Koivisto, T. Gyrocardiography: A new non-invasive monitoring method for the assessment of cardiac mechanics and the estimation of hemodynamic variables. Sci. Rep. 2017, 7, 6823. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Tavassolian, N. Combined Seismo- and Gyro-Cardiography: A More Comprehensive Evaluation of Heart-Induced Chest Vibrations. IEEE J. Biomed. Health Inform. 2018, 22, 1466–1475. [Google Scholar] [CrossRef] [PubMed]
- Sadek, I.; Biswas, J.; Abdulrazak, B. Ballistocardiogram signal processing: A review. Health Inf. Sci. Syst. 2019, 7, 10. [Google Scholar] [CrossRef]
- Giovangrandi, L.; Inan, O.T.; Wiard, R.M.; Etemadi, M.; Kovacs, G.T.A. Ballistocardiography–A method worth revisiting. In Proceedings of the 33rd Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011; pp. 4279–4282. [Google Scholar]
- Morbiducci, U.; Scalise, L.; De Melis, M.; Grigioni, M. Optical Vibrocardiography: A Novel Tool for the Optical Monitoring of Cardiac Activity. Ann. Biomed. Eng. 2007, 35, 45–58. [Google Scholar] [CrossRef]
- Hossein, A.; Rabineau, J.; Gorlier, D.; Del Rio, J.I.J.; van de Borne, P.; Migeotte, P.-F.; Nonclercq, A. Kinocardiography Derived from Ballistocardiography and Seismocardiography Shows High Repeatability in Healthy Subjects. Sensors 2021, 21, 815. [Google Scholar] [CrossRef]
- Jaakkola, J.; Jaakkola, S.; Lahdenoja, O.; Hurnanen, T.; Koivisto, T.; Pänkäälä, M.; Knuutila, T.; Kiviniemi, T.O.; Vasankari, T.; Airaksinen, K.E.J. Mobile Phone Detection of Atrial Fibrillation with Mechanocardiography. Circulation 2018, 137, 1524–1527. [Google Scholar] [CrossRef]
- Bowen, W.P. Changes in Heart-Rate, Blood-Pressure, and Duration of Systole Resulting from Bicycling. Am. J. Physiol. Content 1904, 11, 59–77. [Google Scholar] [CrossRef]
- Sieciński, S.; Kostka, P.S.; Tkacz, E.J. Heart Rate Variability Analysis on Electrocardiograms, Seismocardiograms and Gyrocardiograms on Healthy Volunteers. Sensors 2020, 20, 4522. [Google Scholar] [CrossRef]
- D’Mello, Y.; Skoric, J.; Xu, S.; Roche, P.J.R.; Lortie, M.; Gagnon, S.; Plant, D.V. Real-Time Cardiac Beat Detection and Heart Rate Monitoring from Combined Seismocardiography and Gyrocardiography. Sensors 2019, 19, 3472. [Google Scholar] [CrossRef] [Green Version]
- Sessa, F.; Anna, V.; Messina, G.; Cibelli, G.; Monda, V.; Marsala, G.; Ruberto, M.; Biondi, A.; Cascio, O.; Bertozzi, G.; et al. Heart Rate Variability as Predictive Factor for Sudden Cardiac Death. Aging 2018, 10, 166–177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Etemadi, M.; Inan, O.T.; Giovangrandi, L.; Kovacs, G.T.A. Rapid Assessment of Cardiac Contractility on a Home Bathroom Scale. IEEE Trans. Inf. Technol. Biomed. 2011, 15, 864–869. [Google Scholar] [CrossRef] [PubMed]
- Ha, T.; Tran, J.; Liu, S.; Jang, H.; Jeong, H.; Mitbander, R.; Huh, H.; Qiu, Y.; Duong, J.; Wang, R.L.; et al. A Chest-Laminated Ultrathin and Stretchable E-Tattoo for the Measurement of Electrocardiogram, Seismocardiogram, and Cardiac Time Intervals. Adv. Sci. 2019, 6, 1900290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mora, N.; Cocconcelli, F.; Matrella, G.; Ciampolini, P. Fully automated annotation of seismocardiogram for noninvasive vital sign measurements. IEEE Trans. Instrum. Meas. 2020, 69, 1241–1250. [Google Scholar] [CrossRef]
- Dehkordi, P.; Khosrow-Khavar, F.; Di Rienzo, M.; Inan, O.T.; Schmidt, S.E.; Blaber, A.P.; Sørensen, K.; Struijk, J.J.; Zakeri, V.; Lombardi, P.; et al. Comparison of Different Methods for Estimating Cardiac Timings: A Comprehensive Multimodal Echocardiography Investigation. Front. Physiol. 2019, 10, 1057. [Google Scholar] [CrossRef]
- Hoff, L.; Elle, O.J.; Grimnes, M.J.; Halvorsen, S.; Alker, H.J.; Fosse, E. Measurements of Heart Motion Using Accelerometers. In Proceedings of the the 26th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Francisco, CA, USA, 1–5 September 2004; Volume 3, pp. 2049–2051. [Google Scholar]
- Salerno, D.M.; Zanetti, J. Seismocardiography for monitoring changes in left ventricular function during ischemia. Chest 1991, 100, 991–993. [Google Scholar] [CrossRef] [Green Version]
- Inan, O.T.; Baran Pouyan, M.; Javaid, A.Q.; Dowling, S.; Etemadi, M.; Dorier, A.; Heller, J.A.; Bicen, A.O.; Roy, S.; De Marco, T.; et al. Novel Wearable Seismocardiography and Machine Learning Algorithms Can Assess Clinical Status of Heart Failure Patients. Circ. Heart Fail. 2018, 11, e004313. [Google Scholar] [CrossRef]
- Sandler, R.; Gamage, P.; Azad, M.K.; Dhar, R.; Raval, N.; Mentz, R.; Mansy, H. Potential SCG Predictors of Heart Failure Readmission. J. Card. Fail. 2020, 26, S87. [Google Scholar] [CrossRef]
- Dehkordi, P.; Bauer, E.P.; Tavakolian, K.; Zakeri, V.; Blaber, A.P.; Khosrow-Khavar, F. Identifying Patients with Coronary Artery Disease Using Rest and Exercise Seismocardiography. Front. Physiol. 2019, 10, 1211. [Google Scholar] [CrossRef]
- Yao, J.; Tridandapani, S.; Wick, C.A.; Bhatti, P.T. Seismocardiography-Based Cardiac Computed Tomography Gating Using Patient-Specific Template Identification and Detection. IEEE J. Transl. Eng. Health Med. 2017, 5, 1–14. [Google Scholar] [CrossRef]
- Iftikhar, Z.; Lahdenoja, O.; Jafari Tadi, M.; Hurnanen, T.; Vasankari, T.; Kiviniemi, T.; Airaksinen, J.; Koivisto, T.; Pänkäälä, M. Multiclass Classifier based Cardiovascular Condition Detection Using Smartphone Mechanocardiography. Sci. Rep. 2018, 8, 9344. [Google Scholar] [CrossRef] [PubMed]
- Hurnanen, T.; Lehtonen, E.; Tadi, M.J.; Kuusela, T.; Kiviniemi, T.; Saraste, A.; Vasankari, T.; Airaksinen, J.; Koivisto, T.; Pankaala, M. Automated Detection of Atrial Fibrillation Based on Time–Frequency Analysis of Seismocardiograms. IEEE J. Biomed. Health Inform. 2017, 21, 1233–1241. [Google Scholar] [CrossRef] [PubMed]
- Jafari Tadi, M.; Mehrang, S.; Kaisti, M.; Lahdenoja, O.; Hurnanen, T.; Jaakkola, J.; Jaakkola, S.; Vasankari, T.; Kiviniemi, T.; Airaksinen, J.; et al. Comprehensive Analysis of Cardiogenic Vibrations for Automated Detection of Atrial Fibrillation Using Smartphone Mechanocardiograms. IEEE Sens. J. 2019, 19, 2230–2242. [Google Scholar] [CrossRef]
- Paukkunen, M.; Parkkila, P.; Hurnanen, T.; Pankaala, M.; Koivisto, T.; Nieminen, T.; Kettunen, R.; Sepponen, R. Beat-by-Beat Quantification of Cardiac Cycle Events Detected from Three-Dimensional Precordial Acceleration Signals. IEEE J. Biomed. Health Inform. 2016, 20, 435–439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elnaggar, I.; Hurnanen, T.; Lahdenoja, O.; Airola, A.; Kaisti, M.; Vasankari, T.; Pykari, J.; Savontaus, M.; Koivisto, T. Detecting Aortic Stenosis Using Seismocardiography and Gryocardiography Combined with Convolutional Neural Networks. In Proceedings of the IEEE 28th Computing in Cardiology (CinC), Brno, Czech Republic, 12–15 September 2021; pp. 1–4. [Google Scholar]
- Yang, C.; Ojha, B.D.; Aranoff, N.D.; Green, P.; Tavassolian, N. Classification of aortic Stenosis Using Conventional Machine Learning and Deep Learning Methods Based on Multi-Dimensional Cardio-Mechanical Signals. Sci. Rep. 2020, 10, 17521. [Google Scholar] [CrossRef]
- Tavakolian, K.; Khosrow-Khavar, F.; Kajbafzadeh, B.; Marzencki, M.; Blaber, A.P.; Kaminska, B.; Menon, C. Precordial Acceleration Signals Improve the Performance of Diastolic Timed Vibrations. Med. Eng. Phys. 2013, 35, 1133–1140. [Google Scholar] [CrossRef]
- Hasan Shandhi, M.M.; Aras, M.; Wynn, S.; Fan, J.; Heller, J.A.; Etemadi, M.; Klein, L.; Inan, O.T. Cardiac Function Monitoring for Patients Undergoing Cancer Treatments Using Wearable Seismocardiography: A Proof-of-Concept Study. In Proceedings of the 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Montreal, QC, Canada, 20–24 July 2020; pp. 4075–4078. [Google Scholar]
- Mehta, N.J.; Khan, I.A. Third heart sound: Genesis and clinical importance. Int. J. Cardiol. 2004, 97, 183–186. [Google Scholar] [CrossRef]
- Khalili, F.; Gamage, P.T.; Taebi, A.; Johnson, M.E.; Roberts, R.B.; Mitchel, J. Spectral Decomposition and Sound Source Localization of Highly Disturbed Flow through a Severe Arterial Stenosis. Bioengineering 2021, 8, 34. [Google Scholar] [CrossRef]
- Khalili, F.; Gamage, P.T.; Taebi, A.; Johnson, M.E.; Roberts, R.B.; Mitchell, J. Spectral Decomposition of the Flow and Characterization of the Sound Signals through Stenoses with Different Levels of Severity. Bioengineering 2021, 8, 41. [Google Scholar] [CrossRef]
- Schmidt, S.E.; Holst-Hansen, C.; Graff, C.; Toft, E.; Struijk, J.J. Detection of Coronary Artery Disease with an Electronic Stethoscope. In Proceedings of the IEEE 34th Computers in Cardiology, Durham, NC, USA, 30 September–3 October 2007; pp. 757–760. [Google Scholar]
- Schmidt, S.; Holst-Hansen, C.; Toft, E.; Struijk, J. Detection of Coronary Artery Disease with an Electronic Stethoscope: Is it possible? In Proceedings of the Summer Meeting, Danish Cardiovascular Research Academy, Sønderborg, Denmark, 7–9 June 2007; p. 26. [Google Scholar]
- Khalili, F.; Taebi, A. Advances in Computational Fluid Dynamics Modeling of Cardiac Sounds as a Non-Invasive Diagnosis Method. In Biomedical and Biotechnology; American Society of Mechanical Engineers: Brussels, Belgium, 2021; Volume 5. [Google Scholar]
- Taebi, A.; Khalili, F. Advances in Noninvasive Diagnosis Based on Body Sounds and Vibrations–A Review. In Biomedical and Biotechnology; American Society of Mechanical Engineers: Brussels, Belgium, 2021; Volume 5. [Google Scholar]
- Khalili, F.; Gamage, P.P.T.; Mansy, H.A. Hemodynamics of a Bileaflet Mechanical Heart Valve with Different Levels of Dysfunction. J. Appl. Biotechnol. Bioeng. 2017, 2, 187–191. [Google Scholar] [CrossRef] [Green Version]
- Khalili, F.; Gamage, P.; Sandler, R.; Mansy, H. Adverse Hemodynamic Conditions Associated with Mechanical Heart Valve Leaflet Immobility. Bioengineering 2018, 5, 74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khalili, F.; Gamage, P.P.T.; Mansy, H.A. Prediction of Turbulent Shear Stresses through Dysfunctional Bileaflet Mechanical Heart Valves using Computational Fluid Dynamics. In Proceedings of the 3rd Thermal and Fluids Engineering Conference (TFEC), Fort Lauderdale, FL, USA, 4–7 March 2018; pp. 1–9. [Google Scholar]
- Khalili, F.; Gamage, P.P.T.; Mansy, H.A. Verification of Turbulence Models for Flow in a Constricted Pipe at Low Reynolds Number. In Proceedings of the 3rd Thermal and Fluids Engineering Conference (TFEC), Fort Lauderdale, FL, USA, 4–7 March 2018; pp. 1–10. [Google Scholar]
- Khalili, F. Fluid Dynamics Modeling and Sound Analysis of a Bileaflet Mechanical Heart Valve. Ph.D. Thesis, University of Central Florida, Orlando, FL, USA, May 2018. [Google Scholar]
- Khalili, F.; Gamage, P.P.T.; Meguid, I.A.; Mansy, H.A. A Coupled CFD-FEA study of the Sound Generated in a Stenosed Artery and Transmitted Through Tissue Layers. In Proceedings of the IEEE SoutheastCon, Orlando, FL, USA, 19–22 April 2018; pp. 1–6. [Google Scholar]
- Le, T.B.; Akerkouch, L. On the Modal Analysis of Blood Flow Dynamics in Brain Aneurysms. In Proceedings of the 2020 Design of Medical Devices Conference, Minneapolis, MN, USA, 6–9 April 2020; American Society of Mechanical Engineers: Brussels, Belgium, 2020. [Google Scholar]
- Fathi, M.F.; Bakhshinejad, A.; Baghaie, A.; Saloner, D.; Sacho, R.H.; Rayz, V.L.; D’Souza, R.M. Denoising and spatial Resolution Enhancement of 4D Flow MRI Using Proper Orthogonal Decomposition and Lasso Regularization. Comput. Med. Imaging Graph. 2018, 70, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Darwish, A.; Di Labbio, G.; Saleh, W.; Kadem, L. Proper Orthogonal Decomposition Analysis of the Flow Downstream of a Dysfunctional Bileaflet Mechanical Aortic Valve. Cardiovasc. Eng. Technol. 2021, 12, 286–299. [Google Scholar] [CrossRef] [PubMed]
- Habibi, M.; Dawson, S.T.M.; Arzani, A. Data-Driven Pulsatile Blood Flow Physics with Dynamic Mode Decomposition. Fluids 2020, 5, 111. [Google Scholar] [CrossRef]
- Janiga, G. Novel Feature-Based Visualization of the Unsteady Blood Flow in Intracranial Aneurysms with the Help of Proper orthogonal Decomposition (POD). Comput. Med. Imaging Graph. 2019, 73, 30–38. [Google Scholar] [CrossRef]
- Ballarin, F.; Faggiano, E.; Ippolito, S.; Manzoni, A.; Quarteroni, A.; Rozza, G.; Scrofani, R. Fast Simulations of Patient-Specific Haemodynamics of Coronary Artery Bypass Grafts Based on a POD–Galerkin Method and a Vascular Shape Parametrization. J. Comput. Phys. 2016, 315, 609–628. [Google Scholar] [CrossRef]
- Rao, A.; Huynh, E.; Royston, T.J.; Kornblith, A.; Roy, S. Acoustic Methods for Pulmonary Diagnosis. IEEE Rev. Biomed. Eng. 2019, 12, 221–239. [Google Scholar] [CrossRef] [Green Version]
- Bohadana, A.; Izbicki, G.; Kraman, S.S. Fundamentals of Lung Auscultation. N. Engl. J. Med. 2014, 370, 744–751. [Google Scholar] [CrossRef] [Green Version]
- Hanna, N.; Smith, J.; Wolfe, J. How the Acoustic Resonances of The Subglottal Tract Affect the Impedance Spectrum Measured Through the Lips. J. Acoust. Soc. Am. 2018, 143, 2639–2650. [Google Scholar] [CrossRef]
- Harper, P.; Kraman, S.S.; Pasterkamp, H.; Wodicka, G.R. An acoustic Model of the Respiratory Tract. IEEE Trans. Biomed. Eng. 2001, 48, 543–550. [Google Scholar] [CrossRef]
- Gavriely, N.; Cugell, D.W. Airflow Effects on Amplitude and Spectral Content of Normal Breath Sounds. J. Appl. Physiol. 1996, 80, 5–13. [Google Scholar] [CrossRef]
- Gavriely, N.; Palti, Y.; Alroy, G. Spectral Characteristics of Normal Breath Sounds. J. Appl. Physiol. 1981, 50, 307–314. [Google Scholar] [CrossRef]
- Gross, V.; Dittmar, A.; Penzel, T.; Schüttler, F.; Von Wichert, P. The Relationship between Normal Lung Sounds, Age, and Gender. Am. J. Respir. Crit. Care Med. 2000, 162, 905–909. [Google Scholar] [CrossRef]
- Schreur, H.J.; Diamant, Z.; Vanderschoot, J.; Zwinderman, A.H.; Dijkman, J.H.; Sterk, P.J. Lung Sounds During Allergen-Induced Asthmatic Responses in Patients with Asthma. Am. J. Respir. Crit. Care Med. 1996, 153, 1474–1480. [Google Scholar] [CrossRef]
- Goettel, N.; Herrmann, M.J. Breath Sounds. Anesth. Analg. 2019, 128, e42. [Google Scholar] [CrossRef]
- Mansy, H.A.; Balk, R.A.; Warren, W.H.; Royston, T.J.; Dai, Z.; Peng, Y.; Sandler, R.H. Pneumothorax Effects on Pulmonary Acoustic Transmission. J. Appl. Physiol. 2015, 119, 250–257. [Google Scholar] [CrossRef] [Green Version]
- Yigla, M.; Gat, M.; Meyer, J.-J.; Friedman, P.J.; Maher, T.M.; Madison, J.M. Vibration Response Imaging Technology in Healthy Subjects. Am. J. Roentgenol. 2008, 191, 845–852. [Google Scholar] [CrossRef]
- Becker, H.D. Vibration Response Imaging–Finally a Real Stethoscope. Respiration 2009, 77, 236–239. [Google Scholar] [CrossRef]
- Aliboni, L.; Pennati, F.; Royston, T.J.; Woods, J.C.; Aliverti, A. Simulation of Bronchial Airway Acoustics in Healthy and Asthmatic Subjects. PLoS ONE 2020, 15, e0228603. [Google Scholar] [CrossRef]
- Fiz, J.A.; Jané, R.; Salvatella, D.; Izquierdo, J.; Lores, L.; Caminal, P.; Morera, J. Analysis of Tracheal Sounds During Forced Exhalation in Asthma Patients and Normal Subjects. Chest 1999, 116, 633–638. [Google Scholar] [CrossRef]
- Song, T.W.; Kim, K.W.; Kim, E.S.; Park, J.-W.; Sohn, M.H.; Kim, K.-E. Utility of Impulse Oscillometry in Young Children with Asthma. Pediatr. Allergy Immunol. 2008, 19, 763–768. [Google Scholar] [CrossRef]
- Barua, M.; Nazeran, H.; Nava, P.; Diong, B.; Goldman, M. Classification of Impulse Oscillometric Patterns of Lung Function in Asthmatic Children using Artificial Neural Networks. In Proceedings of the IEEE 27th Annual International Conference on IEEE Engineering in Medicine and Biology, Shanghai, China, 31 August–3 September 2005; pp. 327–331. [Google Scholar]
- Guntupalli, K.K.; Reddy, R.M.; Loutfi, R.H.; Alapat, P.M.; Bandi, V.D.; Hanania, N.A. Evaluation of Obstructive Lung Disease with Vibration Response Imaging. J. Asthma 2008, 45, 923–930. [Google Scholar] [CrossRef]
- Homs-Corbera, A.; Fiz, J.A.; Morera, J.; Jane, R. Time-Frequency Detection and Analysis of Wheezes During Forced Exhalation. IEEE Trans. Biomed. Eng. 2004, 51, 182–186. [Google Scholar] [CrossRef]
- Pasterkamp, H.; Schäfer, J.; Wodicka, G.R. Posture-Dependent Change of Tracheal Sounds at Standardized Flows in Patients with Obstructive Sleep Apnea. Chest 1996, 110, 1493–1498. [Google Scholar] [CrossRef]
- Elwali, A.; Moussavi, Z. Obstructive Sleep Apnea Screening and Airway Structure Characterization During Wakefulness Using Tracheal Breathing Sounds. Ann. Biomed. Eng. 2017, 45, 839–850. [Google Scholar] [CrossRef]
- Alshaer, H.; Levchenko, A.; Bradley, T.D.; Pong, S.; Tseng, W.-H.; Fernie, G.R. A System for Portable Sleep Apnea Diagnosis Using an Embedded Data Capturing Module. J. Clin. Monit. Comput. 2013, 27, 303–311. [Google Scholar] [CrossRef]
- Goncharoff, V.; Jacobs, J.E.; Cugell, D.W. Wideband Acoustic Transmission of Human Lungs. Med. Biol. Eng. Comput. 1989, 27, 513–519. [Google Scholar] [CrossRef]
- Crim, C.; Celli, B.; Edwards, L.D.; Wouters, E.; Coxson, H.O.; Tal-Singer, R.; Calverley, P.M.A. Respiratory System Impedance with Impulse Oscillometry in Healthy and COPD Subjects: ECLIPSE Baseline Results. Respir. Med. 2011, 105, 1069–1078. [Google Scholar] [CrossRef] [Green Version]
- Gong, S.-G.; Yang, W.-L.; Zheng, W.; Liu, J.-M. Evaluation of Respiratory Impedance in Patients with Chronic Obstructive Pulmonary Disease by an Impulse Oscillation System. Mol. Med. Rep. 2014, 10, 2694–2700. [Google Scholar] [CrossRef]
- Fernandez-Granero, M.; Sanchez-Morillo, D.; Leon-Jimenez, A. Computerised Analysis of Telemonitored Respiratory Sounds for Predicting Acute Exacerbations of COPD. Sensors 2015, 15, 26978–26996. [Google Scholar] [CrossRef] [Green Version]
- Yonemaru, M.; Kikuchi, K.; Mori, M.; Kawai, A.; Abe, T.; Kawashiro, T.; Ishihara, T.; Yokoyama, T. Detection of Tracheal Stenosis by Frequency Analysis of Tracheal Sounds. J. Appl. Physiol. 1993, 75, 605–612. [Google Scholar] [CrossRef]
- Royston, T.J.; Zhang, X.; Mansy, H.A.; Sandler, R.H. Modeling Sound Transmission Through the Pulmonary System and Chest with Application to Diagnosis of a Collapsed Lung. J. Acoust. Soc. Am. 2002, 111, 1931–1946. [Google Scholar] [CrossRef]
- Lichtenstein, D.A.; Menu, Y. A Bedside Ultrasound Sign Ruling Out Pneumothorax in the Critically III. Chest 1995, 108, 1345–1348. [Google Scholar] [CrossRef] [Green Version]
- Blanco, M.; Mor, R.; Fraticelli, A.; Breen, D.P.; Dutau, H. Distribution of Breath Sound Images in Patients with Pneumothoraces Compared to Healthy Subjects. Respiration 2009, 77, 173–178. [Google Scholar] [CrossRef]
- Kompis, M.; Pasterkamp, H.; Wodicka, G.R. Acoustic Imaging of the Human Chest. Chest 2001, 120, 1309–1321. [Google Scholar] [CrossRef] [Green Version]
- Rao, A.; Ruiz, J.; Bao, C.; Roy, S. Tabla: A Proof-of-Concept Auscultatory Percussion Device for Low-Cost Pneumonia Detection. Sensors 2018, 18, 2689. [Google Scholar] [CrossRef] [Green Version]
- Lichtenstein, D.; Mezière, G.; Seitz, J. The Dynamic Air Bronchogram. Chest 2009, 135, 1421–1425. [Google Scholar] [CrossRef]
- Anantham, D.; Herth, F.J.F.; Majid, A.; Michaud, G.; Ernst, A. Vibration Response Imaging in the Detection of Pleural Effusions: A Feasibility Study. Respiration 2009, 77, 166–172. [Google Scholar] [CrossRef]
- Osman, L.P.; Roughton, M.; Hodson, M.E.; Pryor, J.A. Short-Term Comparative Study of High Frequency Chest Wall Oscillation and European Airway Clearance Techniques in Patients with cystic fibrosis. Thorax 2010, 65, 196–200. [Google Scholar] [CrossRef] [Green Version]
- Hristara-Papadopoulou, A.; Tsanakas, J.; Diomou, G.; Papadopoulou, O. Current Devices of Respiratory Physiotherapy. Hippokratia 2008, 12, 211–220. [Google Scholar]
- Muguli, A.; Pinto, L.; Sharma, N.; Krishnan, P.; Ghosh, P.K.; Kumar, R.; Ramoji, S.; Bhat, S.; Chetupalli, S.R.; Ganapathy, S.; et al. DiCOVA Challenge: Dataset, task, and baseline system for COVID-19 diagnosis using acoustics. arXiv 2021, arXiv:2103.09148. [Google Scholar]
- Rogers, J.; Ni, X.; Ouyang, W.; Jeong, H.; Kim, J.-T.; Tzavelis, A.; Mirzazadeh, A.; Wu, C.; Lee, J.Y.; Keller, M.; et al. Long-term, Continuous, and Multimodal Monitoring of Respiratory Digital Biomarkers Via Wireless Epidermal Mechano-Acoustic Sensing in Clinical and Home Settings for COVID-19 patients. Proc. Natl. Acad. Sci. USA 2020, 118, e2026610118. [Google Scholar]
- Brashier, B.; Salvi, S. Measuring Lung Function Using Sound Waves: Role of the Forced Oscillation Technique and Impulse Oscillometry System. Breathe 2015, 11, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Beydon, N. Pulmonary Function Testing in Young Children. Paediatr. Respir. Rev. 2009, 10, 208–213. [Google Scholar] [CrossRef]
- Penzel, T.; Sabil, A. The Use of Tracheal Sounds for the Diagnosis of Sleep Apnoea. Breathe 2017, 13, e37–e45. [Google Scholar] [CrossRef]
- Berry, R.B.; Budhiraja, R.; Gottlieb, D.J.; Gozal, D.; Iber, C.; Kapur, V.K.; Marcus, C.L.; Mehra, R.; Parthasarathy, S.; Quan, S.F.; et al. Rules for Scoring Respiratory Events in Sleep: Update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. J. Clin. Sleep Med. 2012, 08, 597–619. [Google Scholar] [CrossRef] [Green Version]
- Sabil, A.; Glos, M.; Günther, A.; Schöbel, C.; Veauthier, C.; Fietze, I.; Penzel, T. Comparison of Apnea Detection Using Oronasal Thermal Airflow Sensor, Nasal Pressure Transducer, Respiratory Inductance Plethysmography and Tracheal Sound Sensor. J. Clin. Sleep Med. 2019, 15, 285–292. [Google Scholar] [CrossRef] [Green Version]
- Maughan, R.J.; Shirreffs, S.M. Muscle Cramping During Exercise: Causes, Solutions, and Questions Remaining. Sport Med. 2019, 49, 115–124. [Google Scholar] [CrossRef] [Green Version]
- Kuo, Y.-C.; Song, T.-T.; Bernard, J.R.; Liao, Y.-H. Short-Term Expiratory Muscle Strength Training Attenuates Sleep Apnea and Improves Sleep Quality in Patients with Obstructive Sleep Apnea. Respir. Physiol. Neurobiol. 2017, 243, 86–91. [Google Scholar] [CrossRef]
- Ruuskanen, O.; Lahti, E.; Jennings, L.C.; Murdoch, D.R. Viral Pneumonia. Lancet 2011, 377, 1264–1275. [Google Scholar] [CrossRef]
- Ayan, E.; Unver, H.M. Diagnosis of Pneumonia from Chest X-Ray Images Using Deep Learning. In Proceedings of the IEEE 2019 Scientific Meeting on Electrical-Electronics & Biomedical Engineering and Computer Science (EBBT), Istanbul, Turkey, 24–26 April 2019; pp. 1–5. [Google Scholar]
- Bartziokas, K.; Daenas, C.; Preau, S.; Zygoulis, P.; Triantaris, A.; Kerenidi, T.; Makris, D.; Gourgoulianis, K.I.; Daniil, Z. Vibration Response Imaging: Evaluation of Rater Agreement in Healthy Subjects and Subjects with Pneumonia. BMC Med. Imaging 2010, 10, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, H.; Chen, J.; Cao, J.; Mu, L.; Hu, Z.; He, J. Application of Vibration Response Imaging Technology in Patients with Community-Acquired Pneumonia Before and After the Treatment. Exp. Ther. Med. 2017, 13, 3433–3437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rennard, S.I.; Drummond, M.B. Early Chronic Obstructive Pulmonary Disease: Definition, Asessment, and Prevention. Lancet 2015, 385, 1778–1788. [Google Scholar] [CrossRef] [Green Version]
- WHO The top 10 Causes of Death. Available online: https://www.who.int/en/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 15 February 2022).
- Altan, G.; Kutlu, Y.; Allahverdi, N. Deep Learning on Computerized Analysis of Chronic Obstructive Pulmonary Disease. IEEE J. Biomed. Health Inform. 2020, 24, 1344–1350. [Google Scholar] [CrossRef]
- Kim, V.; Criner, G.J. Chronic Bronchitis and Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2013, 187, 228–237. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Lin, Y.; Zhang, X.; Wang, Z.; Gao, Y.; Chen, G.; Xiong, H. Classifying Respiratory Sounds Using Electronic Stethoscope. In Proceedings of the 2017 IEEE SmartWorld, Ubiquitous Intelligence & Computing, Advanced & Trusted Computed, Scalable Computing & Communications, Cloud & Big Data Computing, Internet of People and Smart City Innovation (SmartWorld/SCALCOM/UIC/ATC/CBDCom/IOP/SCI), San Francisco, CA, USA, 4–8 August 2017; IEEE: Picataway, NJ, USA, 2017; pp. 1–8. [Google Scholar]
- Emmanouilidou, D.; McCollum, E.D.; Park, D.E.; Elhilali, M. Computerized Lung Sound Screening for Pediatric Auscultation in Noisy Field Environments. IEEE Trans. Biomed. Eng. 2018, 65, 1564–1574. [Google Scholar] [CrossRef]
- Henry, B.; Royston, T.J. A Multiscale Analytical Model of Bronchial Airway Acoustics. J. Acoust. Soc. Am. 2017, 142, 1774–1783. [Google Scholar] [CrossRef]
- Hu, P.; Cai, C.; Yi, H.; Zhao, J.; Feng, Y.; Wang, Q. Aiding Airway Obstruction Diagnosis with Computational Fluid Dynamics and Convolutional Neural Network: A New Perspective and Numerical Case Study. J. Fluids Eng. 2022, 144, 081206. [Google Scholar] [CrossRef]
- Gamage, P.P.T.; Khalili, F.; Khurshidul Azad, M.D.; Mansy, H.A. Modeling Inspiratory Flow in a Porcine Lung Airway. J. Biomech. Eng. 2018, 140, 061003. [Google Scholar] [CrossRef]
- Wang, L.; Ge, H.; Chen, L.; Hajipour, A.; Feng, Y.; Cui, X. LES Study on the Impact of Airway Deformation on the Airflow Structures in the Idealized Mouth–Throat Model. J. Braz. Soc. Mech. Sci. Eng. 2022, 44, 23. [Google Scholar] [CrossRef]
- Du, X.; Allwood, G.; Webberley, K.M.; Osseiran, A.; Wan, W.; Volikova, A.; Marshall, B.J. A Mathematical Model of Bowel Sound Generation. J. Acoust. Soc. Am. 2018, 144, EL485–EL491. [Google Scholar] [CrossRef] [PubMed]
- Dimoulas, C.; Kalliris, G.; Papanikolaou, G.; Kalampakas, A. Long-Term Signal Detection, Segmentation and Summarization Using Wavelets and Fractal Dimension: A Bioacoustics Application in Gastrointestinal-Motility Monitoring. Comput. Biol. Med. 2007, 37, 438–462. [Google Scholar] [CrossRef] [PubMed]
- Dimoulas, C.; Kalliris, G.; Papanikolaou, G.; Petridis, V.; Kalampakas, A. Bowel-Sound Pattern Analysis Using Wavelets and Neural Networks with Application to Long-Term, Unsupervised, Gastrointestinal Motility Monitoring. Expert Syst. Appl. 2008, 34, 26–41. [Google Scholar] [CrossRef]
- Massey, R.L. Return of Bowel Sounds Indicating an End of Postoperative Ileus: Is it Time to Cease This Long-Standing Nursing Tradition? Medsurg Nurs. 2012, 21, 146–150. [Google Scholar] [PubMed]
- Sugrue, M.; Redfern, M.; Kurata, J.H. Computerized Phonoenterography. J. Clin. Gastroenterol. 1994, 18, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Tomomasa, T.; Morikawa, A.; Sandler, R.H.; Mansy, H.A.; Koneko, H.; Masahiko, T.; Hyman, P.E.; Itoh, Z. Gastrointestinal Sounds and Migrating Motor Complex in Fasted Humans. Am. J. Gastroenterol. 1999, 94, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Yuki, M.; Adachi, K.; Fujishiro, H.; Uchida, Y.; Miyaoka, Y.; Yoshino, N.; Yuki, T.; Ono, M.; Kinoshita, Y. Is A Computerized Bowel Sound Auscultation System Useful for The Detection of Increased Bowel Motility? Am. J. Gastroenterol. 2002, 97, 1846–1848. [Google Scholar] [CrossRef]
- Tomomasa, T.; Takahashi, A.; Nako, Y.; Kaneko, H.; Tabata, M.; Tsuchida, Y.; Morikawa, A. Analysis of Gastrointestinal Sounds in Infants with Pyloric Stenosis Before and After Pyloromyotomy. Pediatrics 1999, 104, e60. [Google Scholar] [CrossRef] [Green Version]
- Lu, Q.; Yadid-Pecht, O.; Sadowski, D.; Mintchev, M.P. Acoustic and Intraluminal Ultrasonic Technologies in the Diagnosis of Diseases in Gastrointestinal Tract: A Review. Engineering 2013, 05, 73–77. [Google Scholar] [CrossRef] [Green Version]
- Du, X.; Allwood, G.; Webberley, K.M.; Inderjeeth, A.-J.; Osseiran, A.; Marshall, B.J. Noninvasive Diagnosis of Irritable Bowel Syndrome via Bowel Sound Features: Proof of Concept. Clin. Transl. Gastroenterol. 2019, 10, e00017. [Google Scholar] [CrossRef]
- Spiegel, B.M.R.; Kaneshiro, M.; Russell, M.M.; Lin, A.; Patel, A.; Tashjian, V.C.; Zegarski, V.; Singh, D.; Cohen, S.E.; Reid, M.W.; et al. Validation of an Acoustic Gastrointestinal Surveillance Biosensor for Postoperative Ileus. J. Gastrointest. Surg. 2014, 18, 1795–1803. [Google Scholar] [CrossRef] [PubMed]
- Kaneshiro, M.; Kaiser, W.; Pourmorady, J.; Fleshner, P.; Russell, M.; Zaghiyan, K.; Lin, A.; Martinez, B.; Patel, A.; Nguyen, A.; et al. Postoperative Gastrointestinal Telemetry with an Acoustic Biosensor Predicts Ileus vs. Uneventful GI Recovery. J. Gastrointest. Surg. 2016, 20, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Wang, J.-R.; Ma, Y.-L. Bowel Sounds and Monitoring Gastrointestinal Motility in Critically Ill Patients. Clin. Nurse Spec. 2012, 26, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Lord, S.; Rochester, L.; Baker, K.; Nieuwboer, A. Concurrent Validity of Accelerometry to Measure Gait in Parkinsons Disease. Gait Posture 2008, 27, 357–359. [Google Scholar] [CrossRef]
- Ornelas-Vences, C.; Sánchez-Fernández, L.P.; Sánchez-Pérez, L.A.; Martínez-Hernández, J.M. Computer Model for Leg Agility Quantification and Assessment for Parkinson’s Disease Patients. Med. Biol. Eng. Comput. 2019, 57, 463–476. [Google Scholar] [CrossRef]
- Aminian, S.; Ezeugwu, V.E.; Motl, R.W.; Manns, P.J. Sit Less and Move More: Perspectives of Adults with Multiple Sclerosis. Disabil. Rehabil. 2019, 41, 904–911. [Google Scholar] [CrossRef]
- Charles, P.D.; Esper, G.J.; Davis, T.J.; Maciunas, R.J.; Robertson, D. Classification of Tremor and Update on Treatment. Am. Fam. Phys. 1999, 59, 1565. [Google Scholar]
- Preeti, M.; Guha, K.; Baishnab, K.L.; Dusarlapudi, K.; Narasimha Raju, K. Low Frequency MEMS Accelerometers in Health Monitoring–A Review Based on Material and Design Aspects. Mater. Today Proc. 2019, 18, 2152–2157. [Google Scholar] [CrossRef]
- Cortés, J.P.; Espinoza, V.M.; Ghassemi, M.; Mehta, D.D.; Van Stan, J.H.; Hillman, R.E.; Guttag, J.V.; Zañartu, M. Ambulatory Assessment of Phonotraumatic Vocal Hyperfunction Using Glottal Airflow Measures Estimated from Neck-Surface Acceleration. PLoS ONE 2018, 13, e0209017. [Google Scholar] [CrossRef] [Green Version]
- Mehdizadehfar, V.; Almasganj, F.; Torabinezhad, F. Investigation of the Effects of Speech Signal Length on Vocal Disorder Sorting Done Via Dynamic Pattern Modeling. J. Voice 2017, 31, 515.e1–515.e8. [Google Scholar] [CrossRef]
- Santamato, A.; Panza, F.; Solfrizzi, V.; Russo, A.; Frisardi, V.; Megna, M.; Ranieri, M.; Fiore, P. Acoustic Analysis of Swallowing Sounds: A New Technique for Assessing Dysphagia. J. Rehabil. Med. 2009, 41, 639–645. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dudik, J.M.; Kurosu, A.; Coyle, J.L.; Sejdić, E. Dysphagia and Its Effects on Swallowing Sounds and Vibrations in Adults. Biomed. Eng. Online 2018, 17, 69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lazareck, L.J.; Moussavi, Z.M.K. Classification of Normal and Dysphagic Swallows by Acoustical Means. IEEE Trans. Biomed. Eng. 2004, 51, 2103–2112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chetlur Adithya, P.; Sankar, R.; Moreno, W.A.; Hart, S. Trends in Fetal Monitoring Through Phonocardiography: Challenges and Future Directions. Biomed. Signal. Process. Control. 2017, 33, 289–305. [Google Scholar] [CrossRef]
- Mollan, R.A.; McCullagh, G.C.; Wilson, R.I. A Critical Appraisal of Auscultation of Human Joints. Clin. Orthop. Relat. Res. 1982, 170, 231–237. [Google Scholar] [CrossRef]
- Shark, L.-K. Discovering Differences in Acoustic Emission Between Healthy and Osteoarthritic Knees Using a Four-Phase Model of Sit-Stand-Sit Movements. Open Med. Inform. J. 2010, 4, 116–125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Warnecke, J.M.; Haghi, M.; Deserno, T.M. Unobtrusive Health Monitoring in Private Spaces: The Smart Vehicle. Sensors 2020, 20, 2442. [Google Scholar] [CrossRef]
- Di Rienzo, M.; Piccirillo, S. Wearables for Life in Space. In Wearable Sensors; Elsevier: Amsterdam, The Netherlands, 2021; pp. 463–486. [Google Scholar]
- McKee, A.M.; Goubran, R.A. Sound Localization in the Human Thorax. In Proceedings of the IEEE Instrumentationand Measurement Technology Conference Proceedings, Ottawa, ON, Canada, 16–19 May 2005; Volume 1, pp. 117–122. [Google Scholar]
- Saeidi, A.; Almasganj, F. 3D Heart Sound Source Localization Via Combinational Subspace Methods for Long-Term Heart Monitoring. Biomed. Signal. Process. Control. 2017, 31, 434–443. [Google Scholar] [CrossRef]

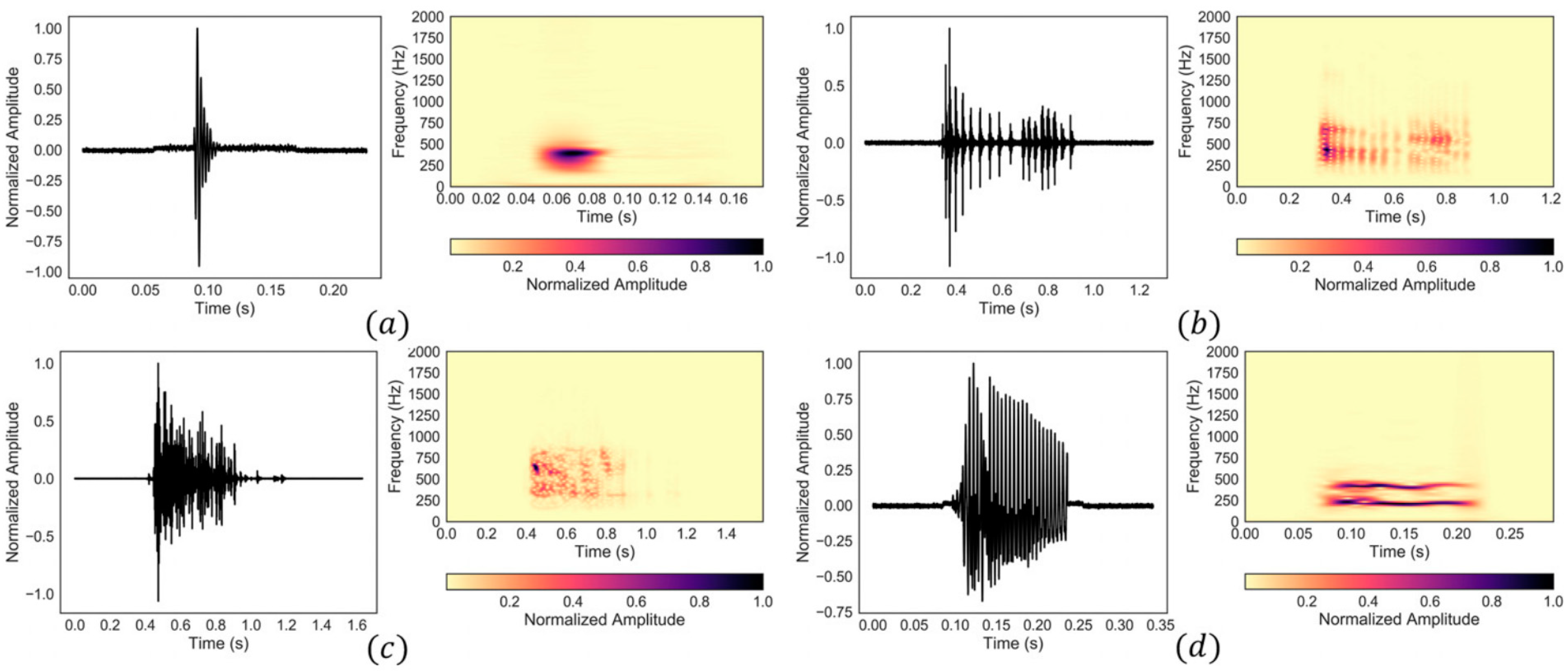
| Disease | Frequency Range [Hz] |
|---|---|
| Pneumonia | 300–600 [1,83] |
| Asthma | 165 [84] 239 [4] 329 [85] |
| Chronic obstructive pulmonary disorder | 233–311 [85] |
| Pneumothorax | 400–600 [86] |
| Type | Duration [ms] | Spectral Centroid [Hz] |
|---|---|---|
| Single burst | 18–58 | 347–681 |
| Multiple bursts | 100–1030 | 345–753 |
| Continuous random sound | 119–1637 | 316–609 |
| Harmonic sound | 73–763 | 269–630 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cook, J.; Umar, M.; Khalili, F.; Taebi, A. Body Acoustics for the Non-Invasive Diagnosis of Medical Conditions. Bioengineering 2022, 9, 149. https://doi.org/10.3390/bioengineering9040149
Cook J, Umar M, Khalili F, Taebi A. Body Acoustics for the Non-Invasive Diagnosis of Medical Conditions. Bioengineering. 2022; 9(4):149. https://doi.org/10.3390/bioengineering9040149
Chicago/Turabian StyleCook, Jadyn, Muneebah Umar, Fardin Khalili, and Amirtahà Taebi. 2022. "Body Acoustics for the Non-Invasive Diagnosis of Medical Conditions" Bioengineering 9, no. 4: 149. https://doi.org/10.3390/bioengineering9040149
APA StyleCook, J., Umar, M., Khalili, F., & Taebi, A. (2022). Body Acoustics for the Non-Invasive Diagnosis of Medical Conditions. Bioengineering, 9(4), 149. https://doi.org/10.3390/bioengineering9040149








