Recent Developments and Current Applications of Hydrogels in Osteoarthritis
Abstract
:1. Introduction
2. OA
3. Hydrogels as a Biomaterial
4. Therapeutic Effects of Hydrogels
5. Hydrogel Implantation in Joints
6. Hydrogel-Based Intra-Articular Drug Delivery
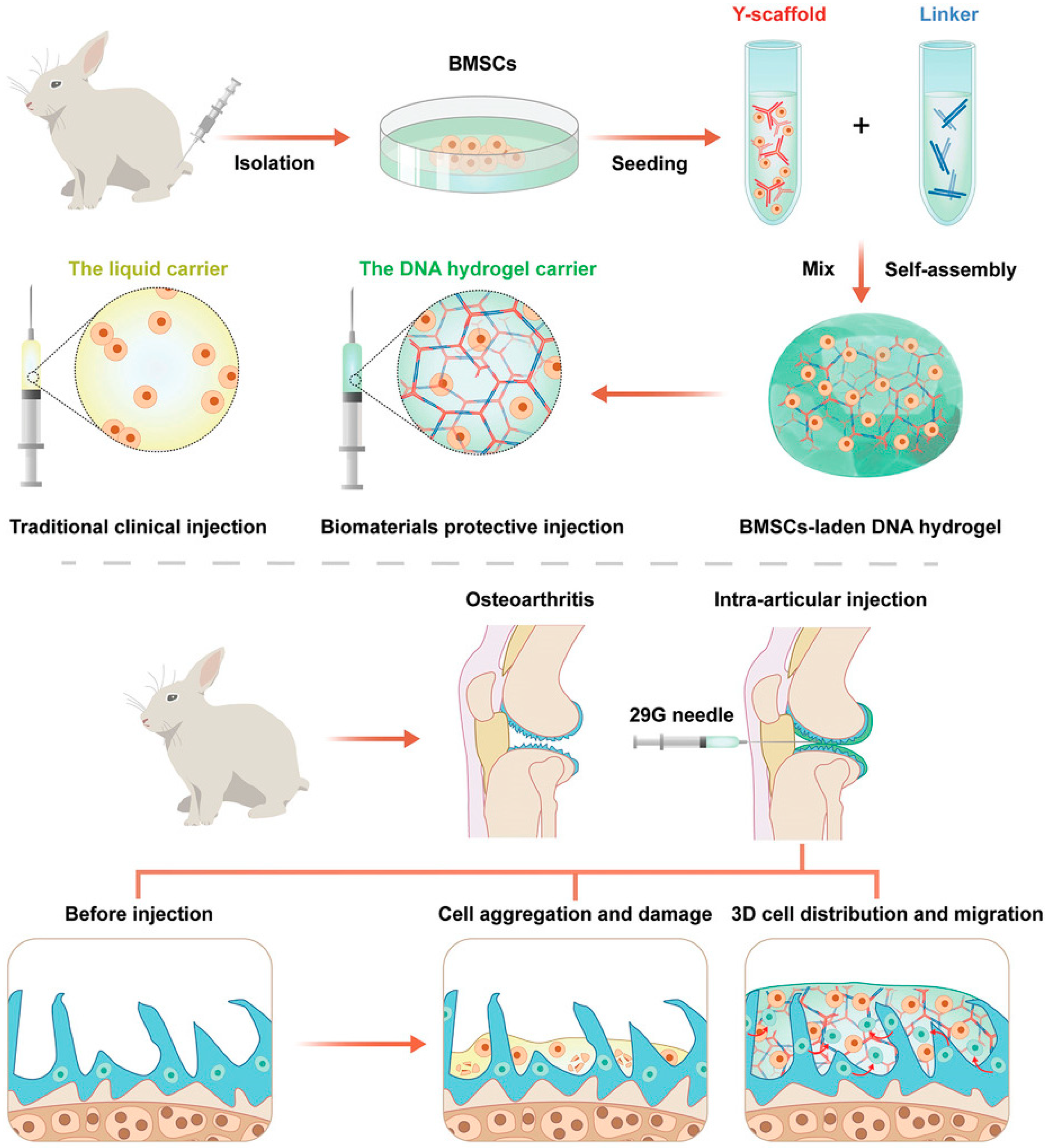
7. MSC Entrapment
8. Articular Cartilage Regeneration
9. Conclusions and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, W.; Ouyang, H.W.; Dass, C.R.; Xu, J.K. Current research on pharmacologic and regenerative therapies for osteoarthritis. Bone Res. 2016, 4, 15040. [Google Scholar] [PubMed]
- Quicke, J.G.; Conaghan, P.G.; Corp, N.; Peat, G. Osteoarthritis year in review 2021: Epidemiology & therapy. Osteoarthr. Cartil. 2022, 30, 196–206. [Google Scholar]
- Nelson, A.E. Osteoarthritis year in review 2017: Clinical. Osteoarthr. Cartil. 2018, 26, 319–325. [Google Scholar]
- Zhang, Y.S.; Khademhosseini, A. Advances in engineering hydrogels. Science 2017, 356, eaaf3627. [Google Scholar]
- Hennink, W.E.; van Nostrum, C.F. Novel crosslinking methods to design hydrogels. Adv. Drug Deliv. Rev. 2012, 64, 223–236. [Google Scholar]
- Kim, I.L.; Mauck, R.L.; Burdick, J.A. Hydrogel design for cartilage tissue engineering: A case study with hyaluronic acid. Biomaterials 2011, 32, 8771–8782. [Google Scholar] [PubMed] [Green Version]
- Van der Kraan, P.M.; van den Berg, W.B. Osteophytes: Relevance and biology. Osteoarthr. Cartil. 2007, 15, 237–244. [Google Scholar]
- Benito, M.J.; Veale, D.J.; Fitzgerald, O.; van den Berg, W.B.; Bresnihan, B. Synovial tissue inflammation in early and late osteoarthritis. Ann. Rheum. Dis. 2005, 64, 1263–1267. [Google Scholar]
- Jin, Z.Y.; Wang, D.D.; Zhang, H.Y.; Liang, J.; Feng, X.B.; Zhao, J.K.; Sun, L.Y. Incidence trend of five common musculoskeletal disorders from 1990 to 2017 at the global, regional and national level: Results from the global burden of disease study 2017. Ann. Rheum. Dis. 2020, 79, 1014–1022. [Google Scholar]
- Hunter, D.J.; Bierma-Zeinstra, S. Osteoarthritis. Lancet 2019, 393, 1745–1759. [Google Scholar]
- Zhang, Y.Q.; Jordan, J.M. Epidemiology of Osteoarthritis. Clin. Geriatr. Med. 2010, 26, 355–369. [Google Scholar] [PubMed] [Green Version]
- Lawrence, R.C.; Felson, D.T.; Helmick, C.G.; Arnold, L.M.; Choi, H.; Deyo, R.A.; Gabriel, S.; Hirsch, R.; Hochberg, M.C.; Hunder, G.G.; et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Arthritis Rheum. 2008, 58, 26–35. [Google Scholar] [PubMed] [Green Version]
- Felson, D.T.; Lawrence, R.C.; Dieppe, P.A.; Hirsch, R.; Helmick, C.G.; Jordan, J.M.; Kington, R.S.; Lane, N.E.; Nevitt, M.C.; Zhang, Y.Q.; et al. Osteoarthritis: New Insights. Part 1: The Disease and Its Risk Factors. Ann. Intern. Med. 2000, 133, 635–646. [Google Scholar] [PubMed]
- Silverwood, V.; Blagojevic-Bucknall, M.; Jinks, C.; Jordan, J.L.; Protheroe, J.; Jordan, K.P. Current evidence on risk factors for knee osteoarthritis in older adults: A systematic review and meta-analysis. Osteoarthr. Cartil. 2015, 23, 507–515. [Google Scholar]
- Hochberg, M.C. Osteoarthritis year 2012 in review: Clinical. Osteoarthr. Cartil. 2012, 20, 1465–1469. [Google Scholar]
- Hochberg, M.C.; Altman, R.D.; April, K.T.; Benkhalti, M.; Guyatt, G.; McGowan, J.; Towheed, T.; Welch, V.; Wells, G.; Tugwell, P. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res. 2012, 64, 465–474. [Google Scholar]
- Nasr, M. In Vitro and In Vivo Evaluation of Proniosomes Containing Celecoxib for Oral Administration. Aaps Pharmscitech 2010, 11, 85–89. [Google Scholar]
- Makela, A.L.; Lempiainen, M.; Ylijoki, H. Ibuprofen Levels in Serum and Synovial-Fluid. Scand. J. Rheumatol. 1981, 39, 15–17. [Google Scholar]
- Zhang, W.; Moskowitz, R.W.; Nuki, G.; Abramson, S.; Altman, R.D.; Arden, N.; Bierma-Zeinstra, S.; Brandt, K.D.; Croft, P.; Doherty, M.; et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. Osteoarthr. Cartil. 2008, 16, 137–162. [Google Scholar]
- Xue, X.; Hu, Y.; Wang, S.; Chen, X.; Jiang, Y.; Su, J. Fabrication of physical and chemical crosslinked hydrogels for bone tissue engineering. Bioact. Mater. 2022, 12, 327–339. [Google Scholar]
- Seliktar, D. Designing Cell-Compatible Hydrogels for Biomedical Applications. Science 2012, 336, 1124–1128. [Google Scholar] [PubMed]
- Zhu, J.Y.; Tang, X.D.; Jia, Y.; Ho, C.T.; Huang, Q.R. Applications and delivery mechanisms of hyaluronic acid used for topical/transdermal delivery—A review. Int. J. Pharm. 2020, 578, 119127. [Google Scholar]
- Lohrasbi, S.; Mirzaei, E.; Karimizade, A.; Takallu, S.; Rezaei, A. Collagen/cellulose nanofiber hydrogel scaffold: Physical, mechanical and cell biocompatibility properties. Cellulose 2020, 27, 927–940. [Google Scholar]
- Hernandez-Gonzalez, A.C.; Tellez-Jurado, L.; Rodriguez-Lorenzo, L.M. Alginate hydrogels for bone tissue engineering, from injectables to bioprinting: A review. Carbohydr. Polym. 2020, 229, 115514. [Google Scholar] [PubMed]
- Reakasame, S.; Boccaccini, A.R. Oxidized Alginate-Based Hydrogels for Tissue Engineering Applications: A Review. Biomacromolecules 2018, 19, 3–21. [Google Scholar]
- Phelps, E.A.; Enemchukwu, N.O.; Fiore, V.F.; Sy, J.C.; Murthy, N.; Sulchek, T.A.; Barker, T.H.; Garcia, A.J. Maleimide Cross-Linked Bioactive PEG Hydrogel Exhibits Improved Reaction Kinetics and Cross-Linking for Cell Encapsulation and In Situ Delivery. Adv. Mater. 2012, 24, 64–70. [Google Scholar]
- Mao, X.Y.; Cheng, R.Y.; Zhang, H.B.; Bae, J.H.; Cheng, L.Y.; Zhang, L.; Deng, L.F.; Cui, W.G.; Zhang, Y.G.; Santos, H.A.; et al. Self-Healing and Injectable Hydrogel for Matching Skin Flap Regeneration. Adv. Sci. 2019, 6, 1801555. [Google Scholar]
- Rizwan, M.; Yahya, R.; Hassan, A.; Yar, M.; Azzahari, A.D.; Selvanathan, V.; Sonsudin, F.; Abouloula, C.N. pH Sensitive Hydrogels in Drug Delivery: Brief History, Properties, Swelling, and Release Mechanism, Material Selection and Applications. Polymers 2017, 9, 137. [Google Scholar]
- Faivre, J.; Pigweh, A.I.; Iehl, J.; Maffert, P.; Goekjian, P.; Bourdon, F. Crosslinking hyaluronic acid soft-tissue fillers: Current status and perspectives from an industrial point of view. Expert Rev. Med. Devices 2021, 18, 1175–1187. [Google Scholar]
- Reeff, J.; Gaignaux, A.; Goole, J.; Siepmann, J.; Siepmann, F.; Jerome, C.; Thomassin, J.M.; De Vriese, C.; Amighi, K. Characterization and optimization of GMO-based gels with long term release for intraarticular administration. Int. J. Pharm. 2013, 451, 95–103. [Google Scholar]
- Wang, Y.; Kankala, R.K.; Ou, C.W.; Chen, A.Z.; Yang, Z.L. Advances in hydrogel-based vascularized tissues for tissue repair and drug screening. Bioact. Mater. 2022, 9, 198–220. [Google Scholar] [PubMed]
- Grimaudo, M.A.; Krishnakumar, G.S.; Giusto, E.; Furlani, F.; Bassi, G.; Rossi, A.; Molinari, F.; Lista, F.; Montesi, M.; Panseri, S. Bioactive injectable hydrogels for on demand molecule/cell delivery and for tissue regeneration in the central nervous system. Acta Biomater. 2022, 140, 88–101. [Google Scholar] [PubMed]
- Wang, G.H.E.; Zhang, X.L.; Bu, X.; An, Y.; Bi, H.S.; Zhao, Z.M. The Application of Cartilage Tissue Engineering with Cell-Laden Hydrogel in Plastic Surgery: A Systematic Review. Tissue Eng. Regen. Med. 2022, 19, 1–9. [Google Scholar]
- Mauro, G.L.; Scaturro, D.; Sanfilippo, A.; Benedetti, M.G. Intra-Articular Hyaluronic Acid Injections for Hip Osteoarthritis. J. Biol. Regul. Homeost. Agents 2018, 32, 1303–1309. [Google Scholar]
- Maudens, P.; Meyer, S.; Seemayer, C.A.; Jordan, O.; Allemann, E. Self-assembled thermoresponsive nanostructures of hyaluronic acid conjugates for osteoarthritis therapy. Nanoscale 2018, 10, 1845–1854. [Google Scholar]
- Pirkle, S.; Seidel, H.; Bhattacharjee, S.; Shi, L.L.; Lee, M.J.; Strelzow, J.A. Analysis of the Cost and Efficacy of Intra-Articular Knee Injections. J. Am. Acad. Orthop. Surg. Glob. Res. Rev. 2022, 6, e21.00203. [Google Scholar]
- Bucci, J.; Chen, X.Y.; LaValley, M.; Nevitt, M.; Torner, J.; Lewis, C.E.; Felson, D.T. Progression of Knee Osteoarthritis with Use of Intraarticular Glucocorticoids Versus Hyaluronic Acid. Arthritis Rheumatol. 2022, 74, 223–226. [Google Scholar] [PubMed]
- Wang, C.P.; Lee, W.C.; Hsieh, R.L. Effects of Repeated Coinjections of Corticosteroids and Hyaluronic Acid on Knee Osteoarthritis: A Prospective, Double-Blind Randomized Controlled Trial: Repeated Coinjections for Knee Osteoarthritis. Am. J. Med. 2021; in press. [Google Scholar]
- Alexander, L.A.M.; Denisov, L.N.; Zotkin, E.G.; Dydykina, I.S.; Kochish, A.Y.; Rodionova, S.S.; Trofimov, E.A.; Yakupova, S.P.; Yakupov, E.Z.; Gallelli, L. Pharmacological Management of Osteoarthritis with a Focus on Symptomatic Slow-Acting Drugs Recommendations from Leading Russian Experts. J. Clin. Rheumatol. 2021, 27, E533–E539. [Google Scholar]
- Mou, D.G.; Yu, Q.Y.; Zhang, J.M.; Zhou, J.P.; Li, X.M.; Zhuang, W.Y.; Yang, X.M. Intra-articular Injection of Chitosan-Based Supramolecular Hydrogel for Osteoarthritis Treatment. Tissue Eng. Regen. Med. 2021, 18, 113–125. [Google Scholar]
- Tsukuda, Y.; Onodera, T.; Ito, M.; Izumisawa, Y.; Kasahara, Y.; Igarashi, T.; Ohzawa, N.; Todoh, M.; Tadano, S.; Iwasaki, N. Therapeutic effects of intra-articular ultra-purified low endotoxin alginate administration on an experimental canine osteoarthritis model. J. Biomed. Mater. Res. Part A 2015, 103, 3441–3448. [Google Scholar]
- Chejara, D.R.; Mabrouk, M.; Kumar, P.; Choonara, Y.E.; Kondiah, P.P.D.; Badhe, R.V.; du Toit, L.C.; Bijukumar, D.; Pillay, V. Synthesis and Evaluation of a Sodium Alginate-4-Aminosalicylic Acid Based Microporous Hydrogel for Potential Viscosupplementation for Joint Injuries and Arthritis-Induced Conditions. Mar. Drugs 2017, 15, 257. [Google Scholar]
- Fakhari, A.; Berkland, C. Applications and emerging trends of hyaluronic acid in tissue engineering, as a dermal filler and in osteoarthritis treatment. Acta Biomater. 2013, 9, 7081–7092. [Google Scholar]
- Jeuken, R.M.; Roth, A.K.; Peters, R.; van Donkelaar, C.C.; Thies, J.C.; van Rhijn, L.W.; Emans, P.J. Polymers in Cartilage Defect Repair of the Knee: Current Status and Future Prospects. Polymers 2016, 8, 219. [Google Scholar]
- Ha, W.; Wang, Z.H.; Zhao, X.B.; Shi, Y.P. Reinforced Supramolecular Hydrogels from Attapulgite and Cyclodextrin Pseudopolyrotaxane for Sustained Intra-Articular Drug Delivery. Macromol. Biosci. 2021, 21, e2000299. [Google Scholar]
- Bao, W.R.; Li, M.L.; Yang, Y.Y.; Wan, Y.; Wang, X.; Bi, N.; Li, C.L. Advancements and Frontiers in the High Performance of Natural Hydrogels for Cartilage Tissue Engineering. Front. Chem. 2020, 8, 53. [Google Scholar]
- Cho, H.; Kim, J.; Kim, S.; Jung, Y.C.; Wang, Y.; Kang, B.J.; Kim, K. Dual delivery of stem cells and insulin-like growth factor-1 in coacervate-embedded composite hydrogels for enhanced cartilage regeneration in osteochondral defects. J. Control. Release 2020, 327, 284–295. [Google Scholar]
- Zhao, W.; Jin, X.; Cong, Y.; Liu, Y.; Fu, J. Degradable natural polymer hydrogels for articular cartilage tissue engineering. J. Chem. Technol. Biotechnol. 2013, 88, 327–339. [Google Scholar]
- Wei, W.; Ma, Y.Z.; Yao, X.D.; Zhou, W.Y.; Wang, X.Z.; Li, C.L.; Lin, J.X.; He, Q.L.; Leptihn, S.; Ouyang, H.W. Advanced hydrogels for the repair of cartilage defects and regeneration. Bioact. Mater. 2021, 6, 998–1011. [Google Scholar]
- Fussenegger, M.; Meinhart, J.; Hobling, W.; Kullich, W.; Funk, S.; Bernatzky, G. Stabilized autologous fibrin-chondrocyte constructs for cartilage repair in vivo. Ann. Plast. Surg. 2003, 51, 493–498. [Google Scholar]
- Frisman, I.; Orbach, R.; Seliktar, D.; Bianco-Peled, H. Structural investigation of PEG-fibrinogen conjugates. J. Mater. Sci.-Mater. Med. 2010, 21, 73–80. [Google Scholar] [PubMed]
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [PubMed] [Green Version]
- Choi, J.H.; Choi, O.K.; Lee, J.; Noh, J.; Lee, S.; Park, A.; Rim, M.A.; Reis, R.L.; Khang, G. Evaluation of double network hydrogel of poloxamer-heparin/gellan gum for bone marrow stem cells delivery carrier. Colloids Surf. B Biointerfaces 2019, 181, 879–889. [Google Scholar] [PubMed]
- Klotz, B.J.; Oosterhoff, L.A.; Utomo, L.; Lim, K.S.; Vallmajo-Martin, Q.; Clevers, H.; Woodfield, T.B.F.; Rosenberg, A.; Malda, J.; Ehrbar, M.; et al. A Versatile Biosynthetic Hydrogel Platform for Engineering of Tissue Analogues. Adv. Healthc. Mater. 2019, 8, 1900979. [Google Scholar]
- Owen, S.G.; Francis, H.W.; Roberts, M.S. Disappearance kinetics of solutes from synovial-fluid after intraarticular injection. Br. J. Clin. Pharmacol. 1994, 38, 349–355. [Google Scholar]
- Hoare, T.R.; Kohane, D.S. Hydrogels in drug delivery: Progress and challenges. Polymer 2008, 49, 1993–2007. [Google Scholar]
- Garcia-Fernandez, L.; Olmeda-Lozano, M.; Benito-Garzon, L.; Perez-Caballer, A.; San Roman, J.; Vazquez-Lasa, B. Injectable hydrogel-based drug delivery system for cartilage regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 110, 110702. [Google Scholar]
- Kim, K.S.; Park, S.J.; Yang, J.A.; Jeon, J.H.; Bhang, S.H.; Kim, B.S.; Hahn, S.K. Injectable hyaluronic acid-tyramine hydrogels for the treatment of rheumatoid arthritis. Acta Biomater. 2011, 7, 666–674. [Google Scholar]
- Petit, A.; Sandker, M.; Muller, B.; Meyboom, R.; van Midwoud, P.; Bruin, P.; Redout, E.M.; Versluijs-Helder, M.; van der Lest, C.H.A.; Buwalda, S.J.; et al. Release behavior and intra-articular biocompatibility of celecoxib-loaded acetyl-capped PCLA-PEG-PCLA thermogels. Biomaterials 2014, 35, 7919–7928. [Google Scholar]
- Diaz-Rodriguez, P.; Landin, M. Controlled release of indomethacin from alginate-poloxamer-silicon carbide composites decrease in-vitro inflammation. Int. J. Pharm. 2015, 480, 92–100. [Google Scholar]
- Yin, N.; Guo, X.T.; Sun, R.; Liu, H.B.; Tang, L.H.; Gou, J.X.; Yin, T.; He, H.B.; Zhang, Y.; Tang, X. Intra-articular injection of indomethacin-methotrexate in situ hydrogel for the synergistic treatment of rheumatoid arthritis. J. Mater. Chem. B 2020, 8, 993–1007. [Google Scholar] [PubMed]
- Fattahpour, S.; Shamanian, M.; Tavakoli, N.; Fathi, M.; Sadeghi-Aliabadi, H.; Sheykhi, S.R.; Fesharaki, M.; Fattahpour, S. An injectable carboxymethyl chitosan-methylcellulose-pluronic hydrogel for the encapsulation of meloxicam loaded nanoparticles. Int. J. Biol. Macromol. 2020, 151, 220–229. [Google Scholar] [PubMed]
- Johnson, K.; Zhu, S.T.; Tremblay, M.S.; Payette, J.N.; Wang, J.N.; Bouchez, L.C.; Meeusen, S.; Althage, A.; Cho, C.Y.; Wu, X.; et al. A Stem Cell-Based Approach to Cartilage Repair. Science 2012, 336, 717–721. [Google Scholar]
- Massaro, M.; Buscemi, G.; Arista, L.; Biddeci, G.; Cavallaro, G.; D’Anna, F.; Di Blasi, F.; Ferrante, A.; Lazzara, G.; Rizzo, C.; et al. Multifunctional Carrier Based on Halloysite/Laponite Hybrid Hydrogel for Kartogenin Delivery. ACS Med. Chem. Lett. 2019, 10, 419–424. [Google Scholar]
- Kang, M.L.; Jeong, S.Y.; Im, G.I. Hyaluronic Acid Hydrogel Functionalized with Self-Assembled Micelles of Amphiphilic PEGylated Kartogenin for the Treatment of Osteoarthritis. Tissue Eng. Part A 2017, 23, 630–639. [Google Scholar] [PubMed]
- Xia, C.; Chen, P.F.; Mei, S.; Ning, L.; Lei, C.Y.; Wang, J.Y.; Zhang, J.F.; Ma, J.J.; Fan, S.W. Photo-crosslinked HAMA hydrogel with cordycepin encapsulated chitosan microspheres for osteoarthritis treatment. Oncotarget 2017, 8, 2835–2849. [Google Scholar]
- Chen, P.F.; Zhu, S.A.; Wang, Y.Y.; Mu, Q.; Wu, Y.; Xia, Q.Q.; Zhang, X.L.; Sun, H.; Tao, J.D.; Hu, H.; et al. The amelioration of cartilage degeneration by ADAMTS-5 inhibitor delivered in a hyaluronic acid hydrogel. Biomaterials 2014, 35, 2827–2836. [Google Scholar]
- Chen, P.F.; Xia, C.; Mei, S.; Wang, J.Y.; Shan, Z.; Lin, X.F.; Fan, S.W. Intra-articular delivery of sinomenium encapsulated by chitosan microspheres and photo-crosslinked GelMA hydrogel ameliorates osteoarthritis by effectively regulating autophagy. Biomaterials 2016, 81, 1–13. [Google Scholar]
- Matsuzaki, T.; Matsushita, T.; Tabata, Y.; Saito, T.; Matsumoto, T.; Nagai, K.; Kuroda, R.; Kurosaka, M. Intra-articular administration of gelatin hydrogels incorporating rapamycin-micelles reduces the development of experimental osteoarthritis in a murine model. Biomaterials 2014, 35, 9904–9911. [Google Scholar]
- Feng, Q.; Lin, S.; Zhang, K.Y.; Dong, C.Q.; Wu, T.Y.; Huang, H.Q.; Yan, X.H.; Zhang, L.; Li, G.; Bian, L.M. Sulfated hyaluronic acid hydrogels with retarded degradation and enhanced growth factor retention promote hMSC chondrogenesis and articular cartilage integrity with reduced hypertrophy. Acta Biomater. 2017, 53, 329–342. [Google Scholar]
- Berney, M.; McCarroll, P.; Glynn, L.; Lenehan, B. Platelet-rich plasma injections for hip osteoarthritis: A review of the evidence. Ir. J. Med. Sci. 2021, 190, 1021–1025. [Google Scholar] [PubMed]
- Saito, M.; Takahashi, K.A.; Arai, Y.; Inoue, A.; Sakao, K.; Tonomura, H.; Honjo, K.; Nakagawa, S.; Inoue, H.; Tabata, Y.; et al. Intraarticular administration of platelet-rich plasma with biodegradable gelatin hydrogel microspheres prevents osteoarthritis progression in the rabbit knee. Clin. Exp. Rheumatol. 2009, 27, 201–207. [Google Scholar] [PubMed]
- Lu, H.T.; Chang, W.T.; Tsai, M.L.; Chen, C.H.; Chen, W.Y.; Mi, F.L. Development of Injectable Fucoidan and Biological Macromolecules Hybrid Hydrogels for Intra-Articular Delivery of Platelet-Rich Plasma. Mar. Drugs 2019, 17, 236. [Google Scholar]
- Thakur, A.; Parra, D.C.; Motallebnejad, P.; Brocchi, M.; Chen, H.J. Exosomes: Small vesicles with big roles in cancer, vaccine development, and therapeutics. Bioact. Mater. 2022, 10, 281–294. [Google Scholar] [PubMed]
- Zhang, Y.; Wang, X.W.; Chen, J.; Qian, D.F.; Gao, P.; Qin, T.; Jiang, T.; Yi, J.; Xu, T.; Huang, Y.F.; et al. Exosomes derived from platelet-rich plasma administration in site mediate cartilage protection in subtalar osteoarthritis. J. Nanobiotechnol. 2022, 20, 56. [Google Scholar]
- Wang, Q.S.; Xu, B.X.; Fan, K.J.; Fan, Y.S.; Teng, H.; Wang, T.Y. Dexamethasone-loaded thermo-sensitive hydrogel attenuates osteoarthritis by protecting cartilage and providing effective pain relief. Ann. Transl. Med. 2021, 9, 1120. [Google Scholar] [PubMed]
- Zhao, G.; Ren, R.G.; Wei, X.; Jia, Z.S.; Chen, N.R.; Sun, Y.Y.; Zhao, Z.F.; Lele, S.M.; Zhong, H.A.; Goldring, M.B.; et al. Thermoresponsive polymeric dexamethasone prodrug for arthritis pain. J. Control. Release 2021, 339, 484–497. [Google Scholar]
- Zhang, T.T.; Chen, S.Q.; Dou, H.B.; Liu, Q.J.; Shu, G.; Lin, J.C.; Zhang, W.; Peng, G.N.; Zhong, Z.J.; Fu, H.L. Novel glucosamine-loaded thermosensitive hydrogels based on poloxamers for osteoarthritis therapy by intra-articular injection. Mater. Sci. Eng. CMater. Biol. Appl. 2021, 118, 111352. [Google Scholar]
- Hanafy, A.S.; El-Ganainy, S.O. Thermoresponsive Hyalomer intra-articular hydrogels improve monoiodoacetate-induced osteoarthritis in rats. Int. J. Pharm. 2020, 573, 118859. [Google Scholar]
- Kristjansson, B.; Honsawek, S. Current Perspectives in Mesenchymal Stem Cell Therapies for Osteoarthritis. Stem Cells Int. 2014, 2014, 194318. [Google Scholar]
- Hached, F.; Vinatier, C.; Le Visage, C.; Gonde, H.; Guicheux, J.; Grimandi, G.; Billon-Chabaud, A. Biomaterial-assisted cell therapy in osteoarthritis: From mesenchymal stem cells to cell encapsulation. Best Pract. Res. Clin. Rheumatol. 2017, 31, 730–745. [Google Scholar] [PubMed]
- Park, Y.B.; Ha, C.W.; Lee, C.H.; Yoon, Y.C.; Park, Y.G. Cartilage Regeneration in Osteoarthritic Patients by a Composite of Allogeneic Umbilical Cord Blood-Derived Mesenchymal Stem Cells and Hyaluronate hydrogel: Results from a Clinical Trial for Safety and Proof-of-Concept with 7 Years of Extended Follow-Up. Stem Cells Transl. Med. 2017, 6, 613–621. [Google Scholar] [PubMed]
- Yan, X.; Yang, B.; Chen, Y.R.; Song, Y.F.; Ye, J.; Pan, Y.F.; Zhou, B.N.; Wang, Y.Q.; Mao, F.B.A.; Dong, Y.C.; et al. Anti-Friction MSCs Delivery System Improves the Therapy for Severe Osteoarthritis. Adv. Mater. 2021, 33, 2104758. [Google Scholar]
- Snyder, T.N.; Madhavan, K.; Intrator, M.; Dregalla, R.C.; Park, D. A fibrin/hyaluronic acid hydrogel for the delivery of mesenchymal stem cells and potential for articular cartilage repair. J. Biol. Eng. 2014, 8, 10. [Google Scholar] [PubMed] [Green Version]
- Xing, D.; Liu, W.; Wang, B.; Li, J.J.; Zhao, Y.; Li, H.; Liu, A.F.; Du, Y.N.; Lin, J.H. Intra-articular Injection of Cell-laden 3D Microcryogels Empower Low-dose Cell Therapy for Osteoarthritis in a Rat Model. Cell Transplant. 2020, 29, 0963689720932142. [Google Scholar]
- Choi, B.; Kim, S.; Lin, B.; Wu, B.M.; Lee, M. Cartilaginous Extracellular Matrix-Modified Chitosan Hydrogels for Cartilage Tissue Engineering. Acs Appl. Mater. Interfaces 2014, 6, 20110–20121. [Google Scholar]
- Liu, X.L.; Yang, Y.L.; Niu, X.; Lin, Q.N.; Zhao, B.Z.; Wang, Y.; Zhu, L.Y. An in situ photocrosslinkable platelet rich plasma—Complexed hydrogel glue with growth factor controlled release ability to promote cartilage defect repair. Acta Biomater. 2017, 62, 179–187. [Google Scholar]
- Ahmadian, E.; Eftekhari, A.; Dizaj, S.M.; Sharifi, S.; Mokhtarpour, M.; Nasibova, A.N.; Khalilov, R.; Samiei, M. The effect of hyaluronic acid hydrogels on dental pulp stem cells behavior. Int. J. Biol. Macromol. 2019, 140, 245–254. [Google Scholar]
- Gonzalez-Fernandez, T.; Tierney, E.G.; Cunniffe, G.M.; O’Brien, F.J.; Kelly, D.J. Gene Delivery of TGF-beta 3 and BMP2 in an MSC-Laden Alginate Hydrogel for Articular Cartilage and Endochondral Bone Tissue Engineering. Tissue Eng. Part A 2016, 22, 776–787. [Google Scholar]
- Krebs, M.D.; Salter, E.; Chen, E.; Sutter, K.A.; Alsberg, E. Calcium alginate phosphate-DNA nanoparticle gene delivery from hydrogels induces in vivo osteogenesis. J. Biomed. Mater. Res. Part A 2010, 92A, 1131–1138. [Google Scholar]
- Liu, H.; Xiang, X.; Huang, J.B.; Zhu, B.H.; Wang, L.Y.; Tang, Y.J.; Du, F.X.; Li, L.; Yan, F.; Ma, L.; et al. Ultrasound augmenting injectable chemotaxis hydrogel for articular cartilage repair in osteoarthritis. Chin. Chem. Lett. 2021, 32, 1759–1764. [Google Scholar]
- Yang, Y.L.; Zhu, Z.C.; Gao, R.Z.; Yuan, J.; Zhang, J.T.; Li, H.Y.; Xie, Z.P.; Wang, Y. Controlled release of MSC-derived small extracellular vesicles by an injectable Diels-Alder crosslinked hyaluronic acid/PEG hydrogel for osteoarthritis improvement. Acta Biomater. 2021, 128, 163–174. [Google Scholar] [PubMed]
- Schneider, M.C.; Barnes, C.A.; Bryant, S.J. Characterization of the Chondrocyte Secretome in Photoclickable Poly(Ethylene Glycol) Hydrogels. Biotechnol. Bioeng. 2017, 114, 2096–2108. [Google Scholar]
- Cai, Y.P.; Lopez-Ruiz, E.; Wengel, J.; Creemers, L.B.; Howard, K.A. A hyaluronic acid-based hydrogel enabling CD44-mediated chondrocyte binding and gapmer oligonucleotide release for modulation of gene expression in osteoarthritis. J. Control. Release 2017, 253, 153–159. [Google Scholar] [PubMed] [Green Version]
- Sharma, B.; Fermanian, S.; Gibson, M.; Unterman, S.; Herzka, D.A.; Cascio, B.; Coburn, J.; Hui, A.Y.; Marcus, N.; Gold, G.E.; et al. Human Cartilage Repair with a Photoreactive Adhesive-Hydrogel Composite. Sci. Transl. Med. 2013, 5, 167ra6. [Google Scholar] [PubMed] [Green Version]
- Little, C.J.; Kulyk, W.M.; Chen, X. The Effect of Chondroitin Sulphate and Hyaluronic Acid on Chondrocytes Cultured. J. Funct. Biomater. 2014, 5, 197–210. [Google Scholar]
- Agas, D.; Laus, F.; Lacava, G.; Marchegiani, A.; Deng, S.Y.; Magnoni, F.; Silva, G.G.; Di Martino, P.; Sabbieti, M.G.; Censi, R. Thermosensitive hybrid hyaluronan/p(HPMAm-lac)-PEG hydrogels enhance cartilage regeneration in a mouse model of osteoarthritis. J. Cell. Physiol. 2019, 234, 20013–20027. [Google Scholar]
- Guo, T.; Noshin, M.; Baker, H.B.; Taskoy, E.; Meredith, S.J.; Tang, Q.Q.; Ringel, J.P.; Lerman, M.J.; Chen, Y.; Packer, J.D.; et al. 3D printed biofunctionalized scaffolds for microfracture repair of cartilage defects. Biomaterials 2018, 185, 219–231. [Google Scholar]
- Tamai, N.; Myoui, A.; Hirao, M.; Kaito, T.; Ochi, T.; Tanaka, J.; Takaoka, K.; Yoshikawa, H. A new biotechnology for articular cartilage repair: Subchondral implantation of a composite of interconnected porous hydroxyapatite, synthetic polymer (PLA-PEG), and bone morphogenetic protein-2 (rhBMP-2). Osteoarthr. Cartil. 2005, 13, 405–417. [Google Scholar]
- Chen, P.F.; Xia, C.; Mo, J.; Mei, S.; Lin, X.F.; Fan, S.W. Interpenetrating polymer network scaffold of sodium hyaluronate and sodium alginate combined with berberine for osteochondral defect regeneration. Mater. Sci. Eng. C-Mater. Biol. Appl. 2018, 91, 190–200. [Google Scholar]
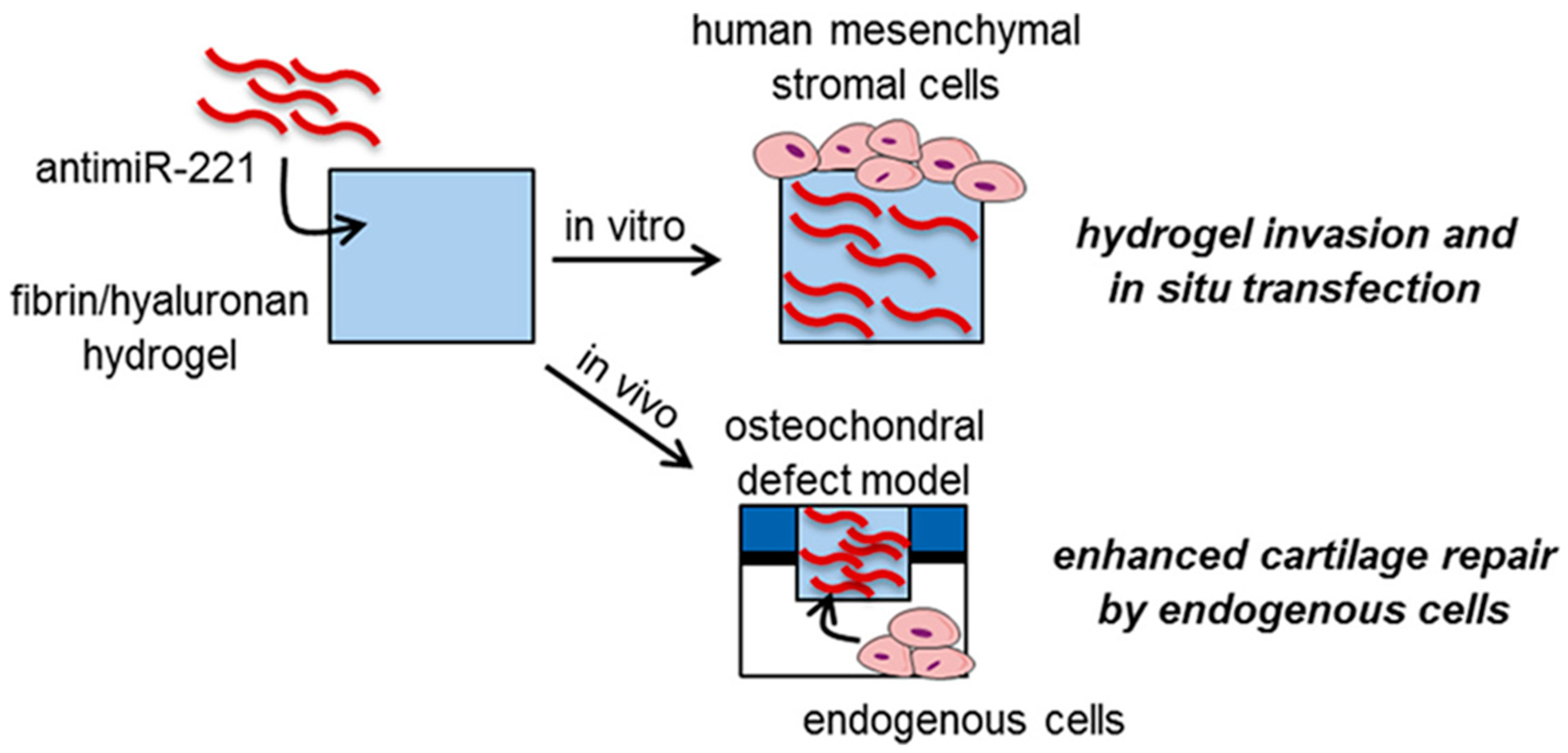
- Lolli, A.; Sivasubramaniyan, K.; Vainieri, M.L.; Oieni, J.; Kops, N.; Yayon, A.; van Osch, G. Hydrogel-based delivery of antimiR-221 enhances cartilage regeneration by endogenous cells. J. Control. Release 2019, 309, 220–230. [Google Scholar] [PubMed]
- Zhang, F.X.; Liu, P.; Ding, W.; Meng, Q.B.; Su, D.H.; Zhang, Q.C.; Lian, R.X.; Yu, B.Q.; Zhao, M.D.; Dong, J.; et al. Injectable Mussel-Inspired highly adhesive hydrogel with exosomes for endogenous cell recruitment and cartilage defect regeneration. Biomaterials 2021, 278, 121169. [Google Scholar] [PubMed]
- Hu, X.B.; Kang, R.R.; Tang, T.T.; Li, Y.J.; Wu, J.Y.; Wang, J.M.; Liu, X.Y.; Xiang, D.X. Topical delivery of 3,5,4-trimethoxy-trans-stilbene-loaded microemulsion-based hydrogel for the treatment of osteoarthritis in a rabbit model. Drug Deliv. Transl. Res. 2019, 9, 357–365. [Google Scholar] [PubMed]
- Ghosh, S.; Mukherjee, B.; Chaudhuri, S.; Roy, T.; Mukherjee, A.; Sengupta, S. Methotrexate Aspasomes Against Rheumatoid Arthritis: Optimized Hydrogel Loaded Liposomal Formulation with In Vivo Evaluation in Wistar Rats. Aaps Pharmscitech 2018, 19, 1320–1336. [Google Scholar] [PubMed]
- Tryfonidou, M.A.; de Vries, G.; Hennink, W.E.; Creemers, L.B. “Old Drugs, New Tricks”—Local controlled drug release systems for treatment of degenerative joint disease. Adv. Drug Deliv. Rev. 2020, 160, 170–185. [Google Scholar]
- Colella, F.; Garcia, J.P.; Sorbona, M.; Lolli, A.; Antunes, B.; D’Atri, D.; Barre, F.P.Y.; Oieni, J.; Vainieri, M.L.; Zerrillo, L.; et al. Drug delivery in intervertebral disc degeneration and osteoarthritis: Selecting the optimal platform for the delivery of disease-modifying agents. J. Control. Release 2020, 328, 985–999. [Google Scholar]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, T.; Wei, Z.; Zhu, W.; Weng, X. Recent Developments and Current Applications of Hydrogels in Osteoarthritis. Bioengineering 2022, 9, 132. https://doi.org/10.3390/bioengineering9040132
Zhao T, Wei Z, Zhu W, Weng X. Recent Developments and Current Applications of Hydrogels in Osteoarthritis. Bioengineering. 2022; 9(4):132. https://doi.org/10.3390/bioengineering9040132
Chicago/Turabian StyleZhao, Tianhao, Zhanqi Wei, Wei Zhu, and Xisheng Weng. 2022. "Recent Developments and Current Applications of Hydrogels in Osteoarthritis" Bioengineering 9, no. 4: 132. https://doi.org/10.3390/bioengineering9040132
APA StyleZhao, T., Wei, Z., Zhu, W., & Weng, X. (2022). Recent Developments and Current Applications of Hydrogels in Osteoarthritis. Bioengineering, 9(4), 132. https://doi.org/10.3390/bioengineering9040132






