Characterization of Macrophage and Cytokine Interactions with Biomaterials Used in Negative-Pressure Wound Therapy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture and Media
2.2. Transwell Culture
2.3. Cell Viability Assay
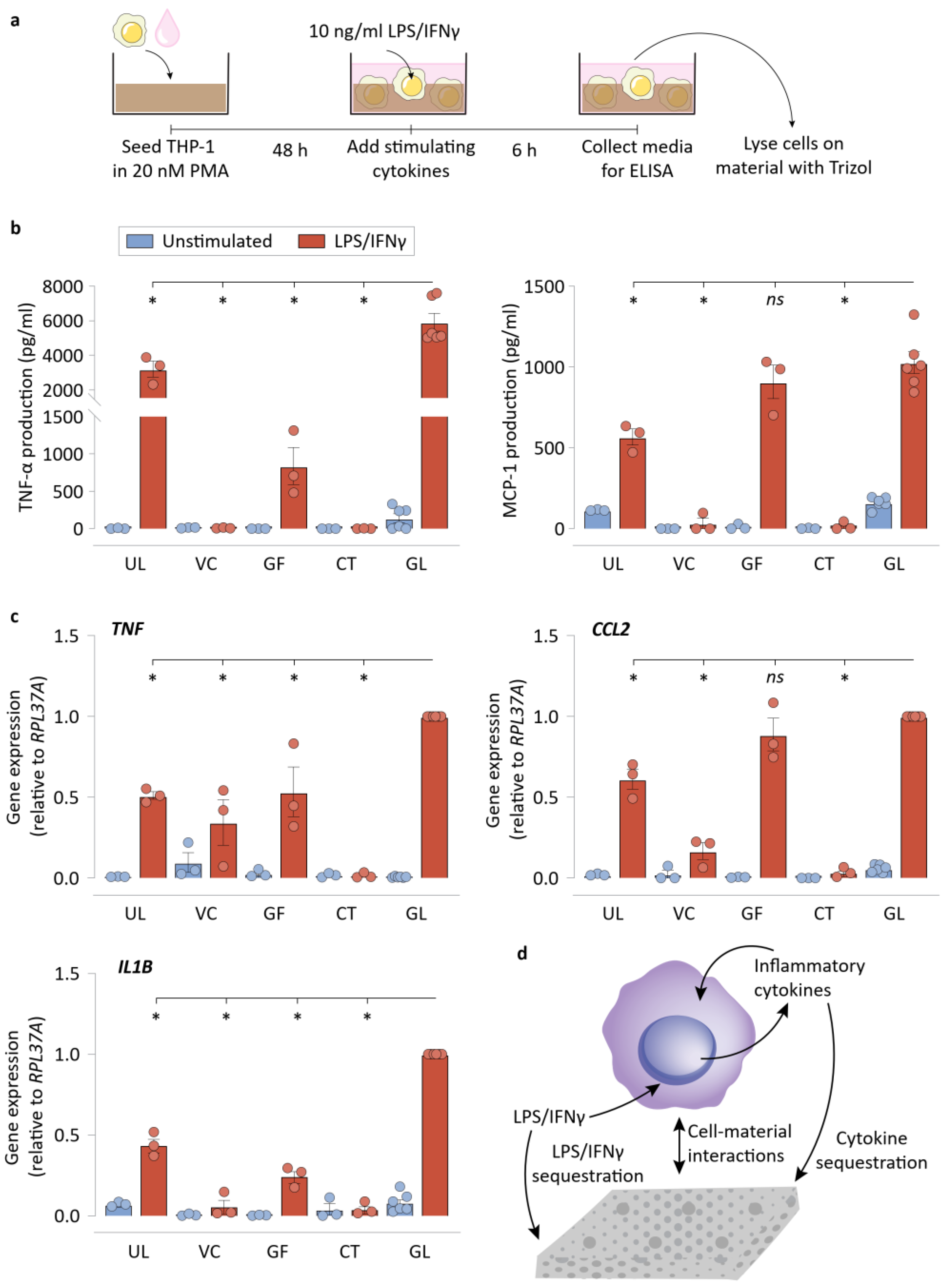
2.4. LPS/IFNγ and TNF-α Cytokine Sequestration Studies
2.5. ELISA on Media Supernatant and on NPWT Materials
2.6. RNA Extraction and qRT-PCR
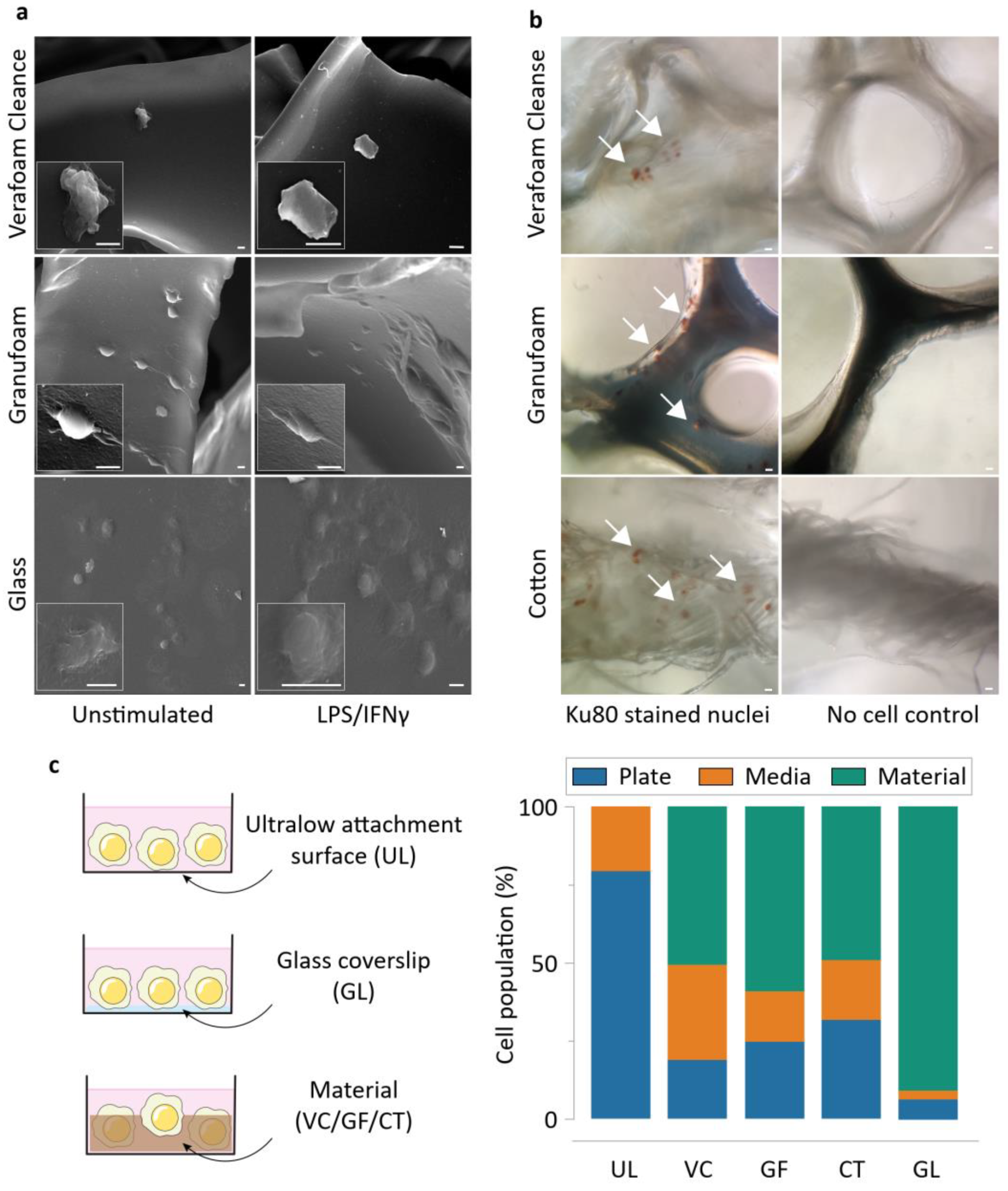
2.7. Determination of Cell Distribution in Culture on the Materials
2.8. Calorimetric Staining
2.9. Scanning Electron Microscopy
2.10. Statistical Analyses
3. Results and Discussion
3.1. NPWT Materials Elicit Different Macrophage Adhesion and Attenuate Inflammatory Gene Activation
3.2. NPWT Materials Do Not Require Direct Contact with Cells to Suppress Inflammatory Activation
3.3. Sequestration of Biochemical Signals by NPWT Materials Results in Reduced Macrophage Inflammatory Activation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Orgill, D.P.; Bayer, L.R. Negative pressure wound therapy: Past, present and future. Int. Wound. J. 2013, 10 (Suppl. S1), 15–19. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Leavitt, T.; Bayer, L.R.; Orgill, D.P. Effect of negative pressure wound therapy on wound healing. Curr. Probl. Surg. 2014, 51, 301–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fagerdahl, A.-M.; Boström, L.; Ulfvarson, J.; Ottosson, C. Risk factors for unsuccessful treatment and complications with negative pressure wound therapy. Wounds 2012, 24, 168. [Google Scholar] [PubMed]
- Koh, T.J.; DiPietro, L.A. Inflammation and wound healing: The role of the macrophage. Expert Rev. Mol. Med. 2011, 13, e23. [Google Scholar] [CrossRef] [Green Version]
- Sheikh, Z.; Brooks, P.J.; Barzilay, O.; Fine, N.; Glogauer, M. Macrophages, foreign body giant cells and their response to implantable biomaterials. Materials 2015, 8, 5671–5701. [Google Scholar] [CrossRef] [Green Version]
- Hind, L.E.; Dembo, M.; Hammer, D.A. Macrophage motility is driven by frontal-towing with a force magnitude dependent on substrate stiffness. Integr. Biol. 2015, 7, 447–453. [Google Scholar] [CrossRef] [Green Version]
- Labernadie, A.; Bouissou, A.; Delobelle, P.; Balor, S.; Voituriez, R.; Proag, A.; Fourquaux, I.; Thibault, C.; Vieu, C.; Poincloux, R.; et al. Protrusion force microscopy reveals oscillatory force generation and mechanosensing activity of human macrophage podosomes. Nat. Commun. 2014, 5, 5343. [Google Scholar] [CrossRef] [Green Version]
- Meli, V.S.; Veerasubramanian, P.K.; Atcha, H.; Reitz, Z.; Downing, T.L.; Liu, W.F. Biophysical regulation of macrophages in health and disease. J. Leukoc. Biol. 2019, 106, 283–299. [Google Scholar] [CrossRef]
- Owen, K.A.; Pixley, F.J.; Thomas, K.S.; Vicente-Manzanares, M.; Ray, B.J.; Horwitz, A.F.; Parsons, J.T.; Beggs, H.E.; Stanley, E.R.; Bouton, A.H. Regulation of lamellipodial persistence, adhesion turnover, and motility in macrophages by focal adhesion kinase. J. Cell Biol. 2007, 179, 1275–1287. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, D.T.; Orgill, D.P.; Murphy, G.F. 4—The pathophysiologic basis for wound healing and cutaneous regeneration. In Biomaterials for Treating Skin Loss; Orgill, D., Blanco, C., Eds.; Woodhead Publishing: Cambridge, UK, 2009; pp. 25–57. [Google Scholar]
- Hsieh, J.Y.; Smith, T.D.; Meli, V.S.; Tran, T.N.; Botvinick, E.L.; Liu, W.F. Differential regulation of macrophage inflammatory activation by fibrin and fibrinogen. Acta Biomater. 2017, 47, 14–24. [Google Scholar] [CrossRef] [Green Version]
- Meli, V.S.; Atcha, H.; Veerasubramanian, P.K.; Nagalla, R.R.; Luu, T.U.; Chen, E.Y.; Guerrero-Juarez, C.F.; Yamaga, K.; Pandori, W.; Hsieh, J.Y.; et al. YAP-mediated mechanotransduction tunes the macrophage inflammatory response. Sci. Adv. 2020, 6, eabb8471. [Google Scholar] [CrossRef] [PubMed]
- Blakney, A.K.; Swartzlander, M.D.; Bryant, S.J. The effects of substrate stiffness on the in vitro activation of macrophages and in vivo host response to poly(ethylene glycol)-based hydrogels. J. Biomed. Mater. Res. A 2012, 100, 1375–1386. [Google Scholar] [CrossRef] [Green Version]
- Varela, P.; Sartori, S.; Viebahn, R.; Salber, J.; Ciardelli, G. Macrophage immunomodulation: An indispensable tool to evaluate the performance of wound dressing biomaterials. J. Appl. Biomater Funct. Mater. 2019, 17, 2280800019830355. [Google Scholar] [CrossRef]
- Eisenhardt, S.U.; Schmidt, Y.; Thiele, J.R.; Iblher, N.; Penna, V.; Torio-Padron, N.; Stark, G.B.; Bannasch, H. Negative pressure wound therapy reduces the ischaemia/reperfusion-associated inflammatory response in free muscle flaps. J. Plast. Reconstr. Aesthet. Surg. 2012, 65, 640–649. [Google Scholar] [CrossRef]
- Glass, G.E.; Murphy, G.F.; Esmaeili, A.; Lai, L.M.; Nanchahal, J. Systematic review of molecular mechanism of action of negative-pressure wound therapy. Br. J. Surg. 2014, 101, 1627–1636. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Xu, Y.; Chang, W.; Zhuang, J.; Wu, X. Negative pressure wound therapy promotes wound healing by suppressing macrophage inflammation in diabetic ulcers. Regen. Med. 2020, 15, 2341–2349. [Google Scholar] [CrossRef]
- Lewis, C.; Murdoch, C. Macrophage responses to hypoxia: Implications for tumor progression and anti-cancer therapies. Am. J. Pathol. 2005, 167, 627–635. [Google Scholar] [CrossRef]
- Scherer, S.S.; Pietramaggiori, G.; Mathews, J.C.; Prsa, M.J.; Huang, S.; Orgill, D.P. The mechanism of action of the vacuum-assisted closure device. Plast. Reconstr. Surg. 2008, 122, 786–797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, P.J.; Attinger, C.E.; Constantine, T.; Crist, B.D.; Faust, E.; Hirche, C.R.; Lavery, L.A.; Messina, V.J.; Ohura, N.; Punch, L.J.; et al. Negative pressure wound therapy with instillation: International consensus guidelines update. Int. Wound. J. 2020, 17, 174–186. [Google Scholar] [CrossRef] [Green Version]
- Davis, K.; Bills, J.; Barker, J.; Kim, P.; Lavery, L. Simultaneous irrigation and negative pressure wound therapy enhances wound healing and reduces wound bioburden in a porcine model. Wound. Repair Regen. 2013, 21, 869–875. [Google Scholar] [CrossRef]
- Gustafsson, R.I.; Sjogren, J.; Ingemansson, R. Deep sternal wound infection: A sternal-sparing technique with vacuum-assisted closure therapy. Ann. Thorac. Surg. 2003, 76, 2048–2053, discussion 2053. [Google Scholar] [CrossRef]
- Zannis, J.; Angobaldo, J.; Marks, M.; DeFranzo, A.; David, L.; Molnar, J.; Argenta, L. Comparison of fasciotomy wound closures using traditional dressing changes and the vacuum-assisted closure device. Ann. Plast. Surg. 2009, 62, 407–409. [Google Scholar] [CrossRef]
- Luu, T.U.; Gott, S.C.; Woo, B.W.; Rao, M.P.; Liu, W.F. Micro- and Nanopatterned Topographical Cues for Regulating Macrophage Cell Shape and Phenotype. ACS Appl. Mater. Interfaces 2015, 7, 28665–28672. [Google Scholar] [CrossRef] [Green Version]
- Sussman, E.M.; Halpin, M.C.; Muster, J.; Moon, R.T.; Ratner, B.D. Porous implants modulate healing and induce shifts in local macrophage polarization in the foreign body reaction. Ann. Biomed. Eng. 2014, 42, 1508–1516. [Google Scholar] [CrossRef]
- Veerasubramanian, P.K.; Shao, H.; Meli, V.S.; Phan, T.A.Q.; Luu, T.U.; Liu, W.F.; Downing, T.L. A Src-H3 acetylation signaling axis integrates macrophage mechanosensation with inflammatory response. Biomaterials 2021, 279, 121236. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, S.L. Advanced wound therapies in the management of severe military lower limb trauma: A new perspective. Eplasty 2009, 9, e28. [Google Scholar]
- Riss, T.L.; Moravec, R.A.; Niles, A.L.; Duellman, S.; Benink, H.A.; Worzella, T.J.; Minor, L. Cell Viability Assays. In Assay Guidance Manual; Markossian, S., Grossman, A., Brimacombe, K., Arkin, M., Auld, D., Austin, C.P., Baell, J., Chung, T.D.Y., Coussens, N.P., Dahlin, J.L., et al., Eds.; Eli Lilly & Company and the National Center for Advancing Translational Sciences: Bethesda, MD, USA, 2004. [Google Scholar]
- Chongrak, K.; Eric, H.; Noureddine, A.; Jean, P.G. Application of methylene blue adsorption to cotton fiber specific surface area measurement: Part, I. Methodology. J. Cotton. Sci. 1998, 2, 164–173. [Google Scholar]
- Lin, W.; Zhang, J.; Wang, Z.; Chen, S. Development of robust biocompatible silicone with high resistance to protein adsorption and bacterial adhesion. Acta Biomater. 2011, 7, 2053–2059. [Google Scholar] [CrossRef]
- Shi, X.; Wang, Y.; Li, D.; Yuan, L.; Zhou, F.; Wang, Y.; Song, B.; Wu, Z.; Chen, H.; Brash, J.L. Cell Adhesion on a POEGMA-Modified Topographical Surface. Langmuir 2012, 28, 17011–17018. [Google Scholar] [CrossRef]
- Hasan, A.; Saxena, V.; Pandey, L.M. Surface Functionalization of Ti6Al4V via Self-assembled Monolayers for Improved Protein Adsorption and Fibroblast Adhesion. Langmuir 2018, 34, 3494–3506. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Gabriel, A.; Lantis, J.; Téot, L. Clinical recommendations and practical guide for negative pressure wound therapy with instillation. Int. Wound. J. 2016, 13, 159–174. [Google Scholar] [CrossRef] [PubMed]
- Elliott, J.R. The development of cotton wool as a wound dressing. Med. Hist. 1957, 1, 362–366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xue, Q.; Lu, Y.; Eisele, M.R.; Sulistijo, E.S.; Khan, N.; Fan, R.; Miller-Jensen, K. Analysis of single-cell cytokine secretion reveals a role for paracrine signaling in coordinating macrophage responses to TLR4 stimulation. Sci. Signal. 2015, 8, ra59. [Google Scholar] [CrossRef] [PubMed] [Green Version]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Veerasubramanian, P.K.; Joe, V.C.; Liu, W.F.; Downing, T.L. Characterization of Macrophage and Cytokine Interactions with Biomaterials Used in Negative-Pressure Wound Therapy. Bioengineering 2022, 9, 2. https://doi.org/10.3390/bioengineering9010002
Veerasubramanian PK, Joe VC, Liu WF, Downing TL. Characterization of Macrophage and Cytokine Interactions with Biomaterials Used in Negative-Pressure Wound Therapy. Bioengineering. 2022; 9(1):2. https://doi.org/10.3390/bioengineering9010002
Chicago/Turabian StyleVeerasubramanian, Praveen Krishna, Victor C. Joe, Wendy F. Liu, and Timothy L. Downing. 2022. "Characterization of Macrophage and Cytokine Interactions with Biomaterials Used in Negative-Pressure Wound Therapy" Bioengineering 9, no. 1: 2. https://doi.org/10.3390/bioengineering9010002
APA StyleVeerasubramanian, P. K., Joe, V. C., Liu, W. F., & Downing, T. L. (2022). Characterization of Macrophage and Cytokine Interactions with Biomaterials Used in Negative-Pressure Wound Therapy. Bioengineering, 9(1), 2. https://doi.org/10.3390/bioengineering9010002





