Innovative High-Pressure Fabrication Processes for Porous Biomaterials—A Review
Abstract
1. Introduction
2. Discussion on Different Biomaterials
2.1. Biodegradable Polymers for Tissue Engineering
2.1.1. Synthetic Polymers for Biomedical Applications
2.1.2. Natural Polymers for Biomedical Applications
2.2. Porous Bioceramics
2.3. Bioactive Glasses
2.4. Metallic Biomaterials
2.5. Porous Scaffold Fabrication Methods
2.5.1. Solvent Casting
2.5.2. Freeze Drying
2.5.3. Gas Foaming
2.5.4. Electrospinning
2.5.5. Three-Dimensional Printing
2.5.6. Other Processing Techniques
2.6. Pressure-Assisted Porous Scaffolds Fabrication
2.6.1. Isostatic Pressure at Negative Temperature: Freeze Isostatic Pressure
2.6.2. Isostatic Pressure at Positive Temperature
2.6.3. Gas-Reinforced (GASAR) Technique
2.6.4. Hydrothermal Sintering (HyS)
2.6.5. Thermosensitive Materials Processing with High Hydrostatic Pressure
2.7. Examples of Porous Silica and Porous Copper by Innovative High-Pressure Processing
2.7.1. Freeze Isostatic Pressure (FIP) Processing of Amorphous SiO2
2.7.2. Hydrothermal Sintering of Porous Amorphous SiO2
2.7.3. Hydrothermal Sintering of Porous Spherical Copper
3. Summary and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jodati, H.; Yılmaz, B.; Evis, Z. A review of bioceramic porous scaffolds for hard tissue applications: Effects of structural features. Ceram. Int. 2020, 46, 15725–15739. [Google Scholar] [CrossRef]
- Baino, F.; Ferraris, M. Learning from Nature: Using bioinspired approaches and natural materials to make porous bioceramics. Int. J. Appl. Ceram. Technol. 2017, 14, 507–520. [Google Scholar] [CrossRef]
- Baino, F.; Fiume, E.; Barberi, J.; Kargozar, S.; Marchi, J.; Massera, J.; Verné, E. Processing methods for making porous bioactive glass-based scaffolds—A state-of-the-art review. Int. J. Appl. Ceram. Technol. 2019, 16, 1762–1796. [Google Scholar] [CrossRef]
- Prasad, K.; Bazaka, O.; Chua, M.; Rochford, M.; Fedrick, L.; Spoor, J.; Symes, R.; Tieppo, M.; Collins, C.; Cao, A.; et al. Metallic Biomaterials: Current Challenges and Opportunities. Materials 2017, 10, 884. [Google Scholar] [CrossRef]
- Hedayat, N.; Du, Y.; Ilkhani, H. Review on fabrication techniques for porous electrodes of solid oxide fuel cells by sacrificial template methods. Renew. Sustain. Energy Rev. 2017, 77, 1221–1239. [Google Scholar] [CrossRef]
- Largeteau, A.; Prakasam, M. Trends in high pressure developments for new perspectives. Solid State Sci. 2018, 80, 141–146. [Google Scholar] [CrossRef]
- Asghari, F.; Samiei, M.; Adibkia, K.; Akbarzadeh, A.; Davaran, S. Biodegradable and biocompatible polymers for tissue engineering application: A review. Artif. Cells, Nanomed. Biotechnol. 2016, 45, 185–192. [Google Scholar] [CrossRef]
- Kobayashi, H.; Terada, D.; Yokoyama, Y.; Moon, D.W.; Yasuda, Y.; Koyama, H.; Takato, T. Vascular-Inducing Poly(glycolic acid)-Collagen Nanocomposite-Fiber Scaffold. J. Biomed. Nanotechnol. 2013, 9, 1318–1326. [Google Scholar] [CrossRef]
- Patrascu, J.M.; Krüger, J.P.; Böss, H.G.; Ketzmar, A.-K.; Freymann, U.; Sittinger, M.; Notter, M.; Endres, M.; Kaps, C. Polyglycolic acid-hyaluronan scaffolds loaded with bone marrow-derived mesenchymal stem cells show chondrogenic differentiationin vitroand cartilage repair in the rabbit model. J. Biomed. Mater. Res. Part B Appl. Biomater. 2013, 101, 1310–1320. [Google Scholar] [CrossRef]
- Lin, C.-C.; Fu, S.-J.; Lin, Y.-C.; Yang, I.-K.; Gu, Y. Chitosan-coated electrospun PLA fibers for rapid mineralization of calcium phosphate. Int. J. Biol. Macromol. 2014, 68, 39–47. [Google Scholar] [CrossRef]
- Mi, H.-Y.; Salick, M.R.; Jing, X.; Jacques, B.R.; Crone, W.C.; Peng, X.-F.; Turng, L.-S. Characterization of thermoplastic polyurethane/polylactic acid (TPU/PLA) tissue engineering scaffolds fabricated by microcellular injection molding. Mater. Sci. Eng. C 2013, 33, 4767–4776. [Google Scholar] [CrossRef]
- Zheng, R.; Duan, H.; Xue, J.; Liu, Y.; Feng, B.; Zhao, S.; Zhu, Y.; Liu, Y.; He, A.; Zhang, W.; et al. The influence of Gelatin/PCL ratio and 3-D construct shape of electrospun membranes on cartilage regeneration. Biomaterials 2014, 35, 152–164. [Google Scholar] [CrossRef]
- Maheshwari, S.U.; Samuel, V.K.; Nagiah, N. Fabrication and evaluation of (PVA/HAp/PCL) bilayer composites as potential scaffolds for bone tissue regeneration application. Ceram. Int. 2014, 40, 8469–8477. [Google Scholar] [CrossRef]
- Qian, J.; Xu, W.; Yong, X.; Jin, X.; Zhang, W. Fabrication and in vitro biocompatibility of biomorphic PLGA/nHA composite scaffolds for bone tissue engineering. Mater. Sci. Eng. C 2014, 36, 95–101. [Google Scholar] [CrossRef]
- Sá-Lima, H.; Tuzlakoglu, K.; Mano, J.F.; Reis, R.L. Thermoresponsive poly(N-isopropylacrylamide)-gmethylcellulose hydrogel as a three-dimensional extracellular matrix for cartilage-engineered applications. J. Biomed. Mater. Res. Part A 2011, 98, 596–603. [Google Scholar] [CrossRef]
- Sidney, L.; Heathman, T.R.; Britchford, E.R.; Abed, A.A.; Rahman, C.V.; Buttery, L.D. Investigation of Localized Delivery of Diclofenac Sodium from Poly(D,L-Lactic Acid-co-Glycolic Acid)/Poly(Ethylene Glycol) Scaffolds Using an In Vitro Osteoblast Inflammation Model. Tissue Eng. Part A 2015, 21, 362–373. [Google Scholar] [CrossRef]
- Niu, Y.; Li, L.; Chen, K.C.; Chen, F.; Liu, X.; Ye, J.; Li, W.; Xu, K. Scaffolds from alternating block polyurethanes of poly(ε-caprolactone) and poly(ethylene glycol) with stimulation and guidance of nerve growth and better nerve repair than autograft. J. Biomed. Mater. Res. Part A 2015, 103, 2355–2364. [Google Scholar] [CrossRef]
- Karami, Z.; Rezaeian, I.; Zahedi, P.; Abdollahi, M. Preparation and performance evaluations of electrospun poly(ε-caprolactone), poly(lactic acid), and their hybrid (50/50) nanofibrous mats containing thymol as an herbal drug for effective wound healing. J. Appl. Polym. Sci. 2013, 129, 756–766. [Google Scholar] [CrossRef]
- Min, B.-M.; Lee, S.W.; Lim, J.N.; You, Y.; Lee, T.S.; Kang, P.H.; Park, W.H. Chitin and chitosan nanofibers: Electrospinning of chitin and deacetylation of chitin nanofibers. Polymer 2004, 45, 7137–7142. [Google Scholar] [CrossRef]
- Liakos, I.; Rizzello, L.; Bayer, I.S.; Pompa, P.P.; Cingolani, R.; Athanassiou, A. Controlled antiseptic release by alginate polymer films and beads. Carbohydr. Polym. 2013, 92, 176–183. [Google Scholar] [CrossRef]
- He, W.; Ma, Z.; Yong, T.; Teo, W.E.; Ramakrishna, S. Fabrication of collagen-coated biodegradable polymer nanofiber mesh and its potential for endothelial cells growth. Biomaterials 2005, 26, 7606–7615. [Google Scholar] [CrossRef]
- Prakasam, M.; Locs, J.; Salma-Ancane, K.; Loca, D.; Largeteau, A.; Berzina-Cimdina, L. Biodegradable Materials and Metallic Implants—A Review. J. Funct. Biomater. 2017, 8, 44. [Google Scholar] [CrossRef]
- Dorozhkin, S.; Epple, M. Biological and Medical Significance of Calcium Phosphates. Angew. Chem. Int. Ed. 2002, 41, 3130–3146. [Google Scholar] [CrossRef]
- Koutsopoulos, S. Synthesis and characterization of hydroxyapatite crystals: A review study on the analytical methods. J. Biomed. Mater. Res. 2002, 62, 600–612. [Google Scholar] [CrossRef]
- Carrodeguas, R.G.; De Aza, S. α-Tricalcium phosphate: Synthesis, properties and biomedical applications. Acta Biomater. 2011, 7, 3536–3546. [Google Scholar] [CrossRef]
- Miyazaki, T.; Ohtsuki, C.; Iwasaki, H.; Ogata, S.; Tanihara, M. Organic modification of porousalpha-tricalcium phosphate to improve chemical durability. Mater. Sci. Forum 2003, 426, 3201–3206. Available online: https://www.cheric.org/research/tech/periodicals/view.php?seq=12548742 (accessed on 5 November 2020). [CrossRef]
- Miao, X.; Lim, W.-K.; Huang, X.; Chen, Y. Preparation and characterization of interpenetrating phased TCP/HA/PLGA composites. Mater. Lett. 2005, 59, 4000–4005. [Google Scholar] [CrossRef]
- Zaichick, S.; Zaichick, V. The Content of Silver, Cobalt, Chromium, Iron, Mercury, Rubidium, Antimony, Selenium, and Zinc in Osteogenic Sarcoma. J. Cancer Ther. 2015, 06, 493–503. [Google Scholar] [CrossRef][Green Version]
- Jeong, C.G.; Hollister, S.J. Mechanical and Biochemical Assessments of Three-Dimensional Poly(1,8-Octanediol-co-Citrate) Scaffold Pore Shape and Permeability Effects on In Vitro Chondrogenesis Using Primary Chondrocytes. Tissue Eng. Part A 2010, 16, 3759–3768. [Google Scholar] [CrossRef]
- Lefebvre, L.; Gremillard, L.; Chevalier, J.; Zenati, R.; Bernache-Assolant, D. Sintering behaviour of 45S5 bioactive glass. Acta Biomater. 2008, 4, 1894–1903. [Google Scholar] [CrossRef]
- Palmero, P. 15-Ceramic–polymer nanocomposites for bone-tissue regeneration. In Nanocomposites for Musculoskeletal Tissue Regeneration; Liu, H., Ed.; Woodhead Publishing: Sawston, UK, 2016; pp. 331–367. [Google Scholar] [CrossRef]
- Andersson, J.; Stenhamre, H.; Bäckdahl, H.; Gatenholm, P. Behavior of human chondrocytes in engineered porous bacterial cellulose scaffolds. J. Biomed. Mater. Res. Part A 2010, 9999A, 1124–1132. [Google Scholar] [CrossRef] [PubMed]
- Popescu, I.N.; Vidu, R.; Bratu, V. Porous Metallic Biomaterials Processing (Review) Part 1: Compaction, Sintering Behavior, Properties and Medical Applications. Sci. Bull. Valahia Univ.-Mater. Mech. 2017, 15, 28–40. [Google Scholar] [CrossRef]
- Grądzka-Dahlke, M.; Dąbrowski, J.; Dąbrowski, B. Modification of mechanical properties of sintered implant materials on the base of Co–Cr–Mo alloy. J. Mater. Process. Technol. 2008, 204, 199–205. [Google Scholar] [CrossRef]
- Gepreel, M.A.-H.; Niinomi, M. Biocompatibility of Ti-alloys for long-term implantation. J. Mech. Behav. Biomed. Mater. 2013, 20, 407–415. [Google Scholar] [CrossRef]
- Levine, B.R.; Sporer, S.; Poggie, R.A.; Della Valle, C.J.; Jacobs, J.J. Experimental and clinical performance of porous tantalum in orthopedic surgery. Biomaterials 2006, 27, 4671–4681. [Google Scholar] [CrossRef]
- Vahidgolpayegani, A.; Wen, C.; Hodgson, P.; Li, Y. Production Methods and Characterization of Porous Mg and Mg Alloys for Biomedical Applications; Bone, M.F., Wen, C., Eds.; Woodhead Publishing: Sawston, UK, 2017; pp. 25–82. [Google Scholar] [CrossRef]
- Prakasam, M. Method for Producing a Porous Monolithic Material. U.S. Patent 10,500,313, 10 December 2019. [Google Scholar]
- Rhim, J.-W.; Mohanty, A.K.; Singh, S.P.; Ng, P.K.W. Effect of the processing methods on the performance of polylactide films: Thermocompression versus solvent casting. J. Appl. Polym. Sci. 2006, 101, 3736–3742. [Google Scholar] [CrossRef]
- Franks, F. Freeze-drying of bioproducts: Putting principles into practice. Eur. J. Pharm. Biopharm. 1998, 45, 221–229. [Google Scholar] [CrossRef]
- Salerno, A.; Oliviero, M.; Di Maio, E.; Iannace, S.; Netti, P.A. Design of porous polymeric scaffolds by gas foaming of heterogeneous blends. J. Mater. Sci. Mater. Med. 2009, 20, 2043–2051. [Google Scholar] [CrossRef]
- Lannutti, J.; Reneker, D.; Ma, T.; Tomasko, D.; Farson, D. Electrospinning for tissue engineering scaffolds. Mater. Sci. Eng. C 2007, 27, 504–509. [Google Scholar] [CrossRef]
- Bose, S.; Vahabzadeh, S.; Bandyopadhyay, A. Bone tissue engineering using 3D printing. Mater. Today 2013, 16, 496–504. [Google Scholar] [CrossRef]
- Andersson, L.; Bergström, L. Gas-filled microspheres as an expandable sacrificial template for direct casting of complex-shaped macroporous ceramics. J. Eur. Ceram. Soc. 2008, 28, 2815–2821. [Google Scholar] [CrossRef]
- Barg, S.; Soltmann, C.; Andrade, M.; Koch, D.; Grathwohl, G. Cellular Ceramics by Direct Foaming of Emulsified Ceramic Powder Suspensions. J. Am. Ceram. Soc. 2008, 91, 2823–2829. [Google Scholar] [CrossRef]
- Ros-Tárraga, P.; Murciano, A.; Mazón, P.; Gehrke, S.A.; De Aza, P. New 3D stratified Si-Ca-P porous scaffolds obtained by sol-gel and polymer replica method: Microstructural, mineralogical and chemical characterization. Ceram. Int. 2017, 43, 6548–6553. [Google Scholar] [CrossRef]
- Eliaz, N.; Metoki, N. Calcium Phosphate Bioceramics: A Review of Their History, Structure, Properties, Coating Technologies and Biomedical Applications. Materials 2017, 10, 334. [Google Scholar] [CrossRef]
- Huang, Y.; Bazarnik, P.; Wan, D.; Luo, D.; Pereira, P.H.; Lewandowska, M.; Yao, J.; Hayden, B.E.; Langdon, T.G. The fabrication of graphene-reinforced Al-based nanocomposites using high-pressure torsion. Acta Mater. 2019, 164, 499–511. [Google Scholar] [CrossRef]
- Edalati, K.; Horita, Z. Application of high-pressure torsion for consolidation of ceramic powders. Scr. Mater. 2010, 63, 174–177. [Google Scholar] [CrossRef]
- Grasso, S.; Biesuz, M.; Zoli, L.; Taveri, G.; Duff, A.I.; Ke, D.; Jiang, A.; Reece, M.J. A review of cold sintering processes. Adv. Appl. Ceram. 2020, 119, 115–143. [Google Scholar] [CrossRef]
- Maria, J.P.; Kang, X.; Floyd, R.D.; Dickey, E.C.; Guo, H.; Guo, J.; Baker, A.; Funihashi, S.; Randall, C.A. Cold sintering: Current status and prospects. Mater. Res. 2017, 32, 3205. [Google Scholar] [CrossRef]
- Kähäri, H.; Teirikangas, M.; Juuti, J.; Jantunen, H. Dielectric properties of lithium molybdate ceramic fabricated at room temperature. J. Am. Ceram. Soc. 2014, 97, 3378. [Google Scholar] [CrossRef]
- Guo, J.; Guo, H.; Baker, A.L.; Lanagan, M.T.; Kupp, E.R.; Messing, G.L.; Randall, C.A. Cold sintering: A paradigm shift for processing and integration of ceramics. Angew. Chem. Int. Ed. 2016, 55, 11457. [Google Scholar] [CrossRef]
- Guo, H.; Guo, J.; Baker, A.; Randall, C.A. A New Guidance for Low-Temperature Ceramic Sintering. ACS Appl. Mater. Interfaces 2016, 8, 20909. [Google Scholar] [CrossRef]
- Floyd, R.; Lowum, S.; Maria, J.-P. Instrumentation for automated and quantitative low temperature compaction and sintering. Rev. Sci. Instrum. 2019, 90, 055104. [Google Scholar] [CrossRef]
- Vakifahmetoglu, C.; Karacasulu, L. Cold sintering of ceramics and glasses: A review. Curr. Opin. Solid State Mater. Sci. 2020, 24, 100807. [Google Scholar] [CrossRef]
- Eksi, A.; Yuzbasioglu, A. Effect of sintering and pressing parameters on the densification of cold isostatically pressed Al and Fe powders. Mater. Des. 2007, 28, 1364–1368. [Google Scholar] [CrossRef]
- Sur un Nouveau Procédé de Frittage de Céramiques à Basse Température: Le Frittage Hydrothermal. Développement et Approche Mécanistique—Thèse Doctorat Bordeaux. Available online: https://tel.archives-ouvertes.fr/tel-01910467 (accessed on 5 November 2020).
- Bocanegra-Bernal, M.H. Hot Isostatic Pressing (HIP) technology and its applications to metals and ceramics. J. Mater. Sci. 2004, 39, 6399–6420. [Google Scholar] [CrossRef]
- Balasubramaniam, V.M.; Barbosa-Cánovas, G.V.; Lelieveld, H. (Eds.) High Pressure Processing of Food Principles, Technology and Application; Springer: New York, NY, USA, 2016. [Google Scholar]
- Dion, M.; Parker, W. Steam sterilization principles. Pharm. Eng. 2013, 33, 60–69. [Google Scholar]
- Gutmanas, E.; Rabinkin, A.; Roitberg, M. Cold sintering under high pressure. Scr. Met. 1979, 13, 11–15. [Google Scholar] [CrossRef]
- Prakasam, M.; Chirazi, A.; Pyka, G.; Prokhodtseva, A.; Lichau, D.; Largeteau, A. Fabrication and Multiscale Structural Properties of Interconnected Porous Biomaterial for Tissue Engineering by Freeze Isostatic Pressure (FIP). J. Funct. Biomater. 2018, 9, 51. [Google Scholar] [CrossRef]
- Katsnel’Son, L.M.; Kerbel’, B.M. Determination of the Optimal Uniaxial Pressing Pressure for Ceramic Powders. Glas. Ceram. 2014, 70, 319–323. [Google Scholar] [CrossRef]
- Taveri, G.; Grasso, S.; Gucci, F.; Toušek, J.; Dlouhy, I. Bio-Inspired Hydro-Pressure Consolidation of Silica. Adv. Funct. Mater. 2018, 28, 1805794. [Google Scholar] [CrossRef]
- Bouville, F.; Studart, A.R. Geologically-inspired strong bulk ceramics made with water at room temperature. Nat. Commun. 2017, 8, 14655. [Google Scholar] [CrossRef] [PubMed]
- Prakasam, M.; Largeteau, A.; (ICMCB-CNRS, Pessac, France). Personal communication, 2019.
- Prakasam, M.; Morvan, A.; Azina, C.; Constantin, L.; Goglio, G.; Largeteau, A.; Bordère, S.; Heintz, J.-M.; Lu, Y.; Silvain, J.-F. Ultra-low temperature fabrication of copper carbon fibre composites by hydrothermal sintering for heat sinks with enhanced thermal efficiency. Compos. Part A Appl. Sci. Manuf. 2020, 133, 105858. [Google Scholar] [CrossRef]
- Mungekar, D.S.; Bhatt, K.B.; Singh, A.J. A laboratory design for uniaxial hot pressing. Bull. Mater. Sci. 1990, 13, 365–369. [Google Scholar] [CrossRef]
- Matamoros-Veloza, Z.; Rendón-Angeles, J.; Yanagisawa, K.; Mejia-Martínez, E.; Parga, J. Low temperature preparation of porous materials from TV panel glass compacted via hydrothermal hot pressing. Ceram. Int. 2015, 41, 12700–12709. [Google Scholar] [CrossRef]
- Vakifahmetoglu, C.; Anger, J.F.; Atakan, V.; Quinn, S.; Gupta, S.; Li, Q.; Tang, L.; Riman, R.E. Reactive Hydrothermal Liquid-Phase Densification (rHLPD) of Ceramics—A Study of the BaTiO3 [TiO2] Composite System. J. Am. Ceram. Soc. 2016, 99, 3893–3901. [Google Scholar] [CrossRef]
- Toraya, H.; Yoshimura, M.; Somiya, S. Hydrothermal Reaction-Sintering of Monoclinic HfO2. J. Am. Ceram. Soc. 1982, 65, c159–c160. [Google Scholar] [CrossRef]
- Han, Y.; Li, S.; Zhu, T.; Xie, Z. An oscillatory pressure sintering of zirconia powder: Rapid densification with limited grain growth. J. Am. Ceram. Soc. 2017, 100, 2774–2780. [Google Scholar] [CrossRef]
- Khasanov, O.; Reichel, U.; Khasanov, D. Lower sintering temperature of nanostructured dense ceramics compacted from dry nanopowders using powerful ultrasonic action. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2011; Volume 18, p. 082004. [Google Scholar]
- Gan, H.; Wang, C.; Li, L.; Shen, Q.; Zhang, L. Structural and magnetic properties of La2NiMnO6 ceramic prepared by ultra-high pressure sintering. J. Alloys Compd. 2018, 735, 2486–2490. [Google Scholar] [CrossRef]
- Sumiya, H. Novel Development of High-Pressure Synthetic Diamonds. SEI Tech. Rev. 2012, 74, 15–23. [Google Scholar] [CrossRef]
- Wilkinson, D. A Pressure-sintering Model for the Densification of Polar Firn and Glacier Ice. J. Glaciol. 1988, 34, 40–45. [Google Scholar] [CrossRef]
- Bridgman, P.W. The Phase Diagram of Water to 45,000 kg/cm2. J. Chem. Phys. 1937, 5, 964. [Google Scholar] [CrossRef]
- Blackford, J.R. Sintering and microstructure of ice: A review. J. Phys. D Appl. Phys. 2007, 40, R355–R385. [Google Scholar] [CrossRef]
- Ishizaki, K.; Nanko, M. A hot isostatic process for fabricating porous materials. J. Porous Mater. 1995, 1, 19–27. [Google Scholar] [CrossRef]
- Shapovalov, V.; Boyko, L. Gasar—A new Class of Porous Materials. Adv. Eng. Mater. 2004, 6, 407–410. [Google Scholar] [CrossRef]
- Jiang, A.; Ke, D.; Xu, L.; Xu, Q.; Li, J.; Wei, J.; Hu, C.; Grasso, S. Cold Hydrostatic Sintering: From shaping to 3D printing. J. Mater. 2019, 5, 496–501. [Google Scholar] [CrossRef]
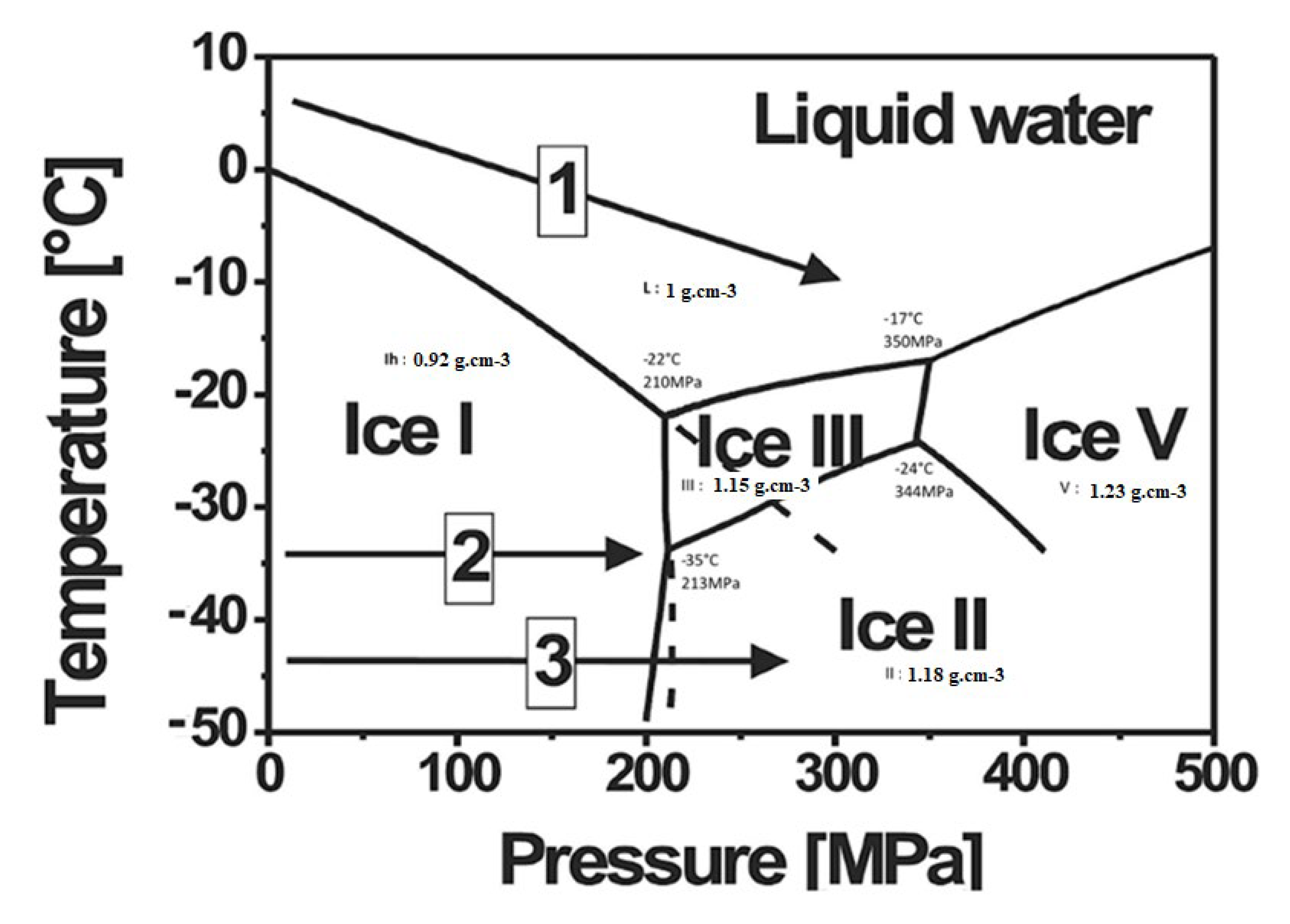



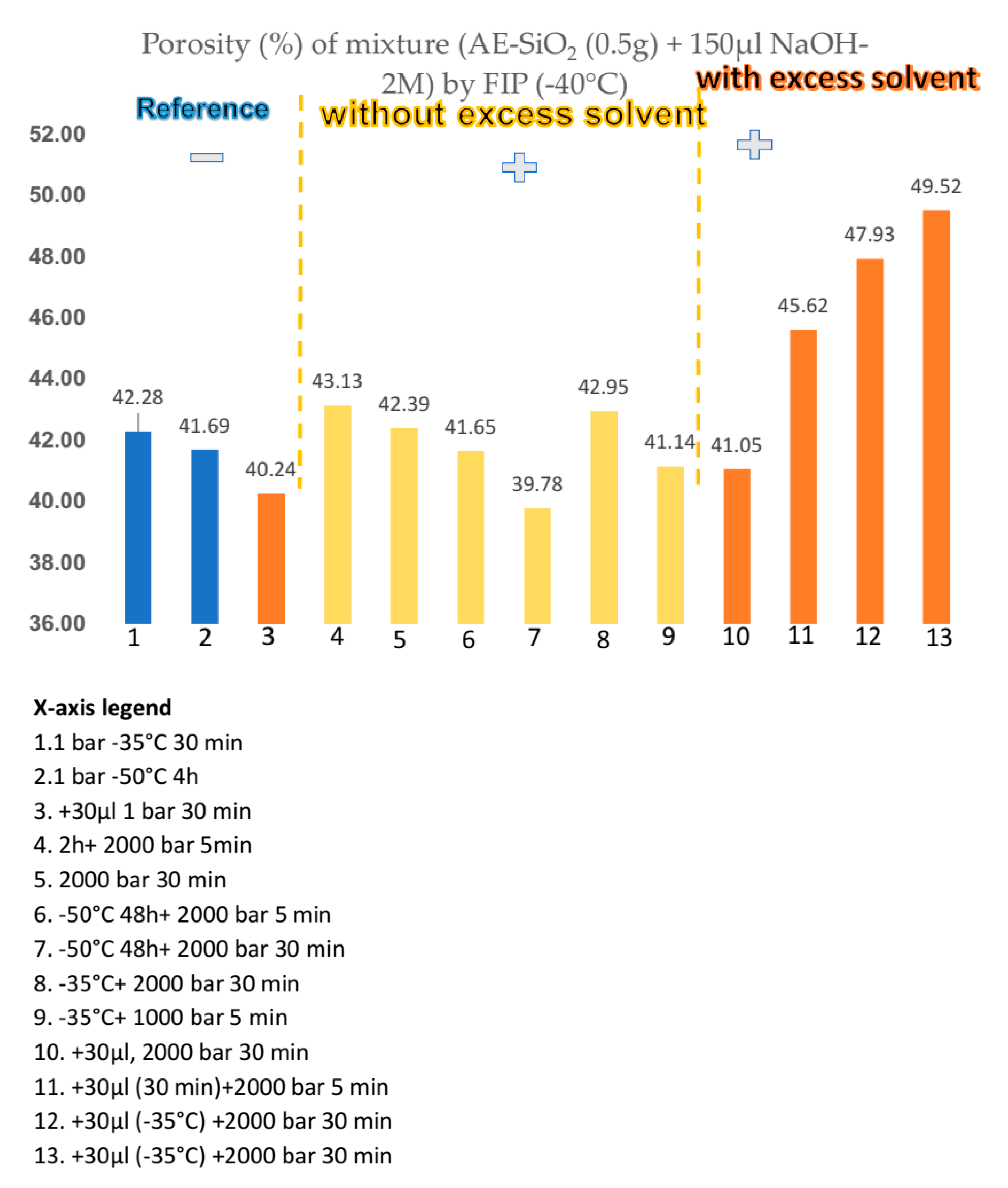

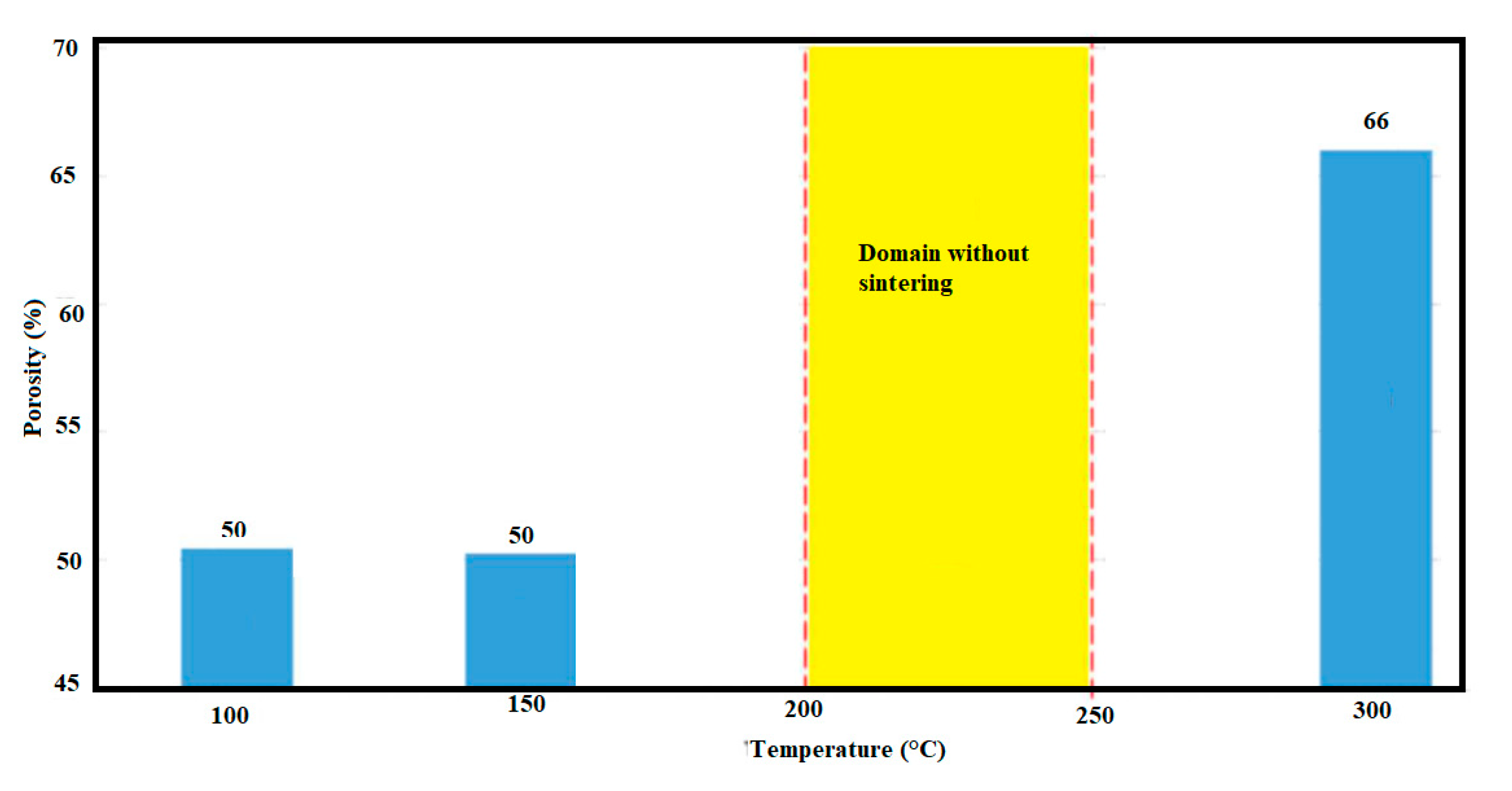
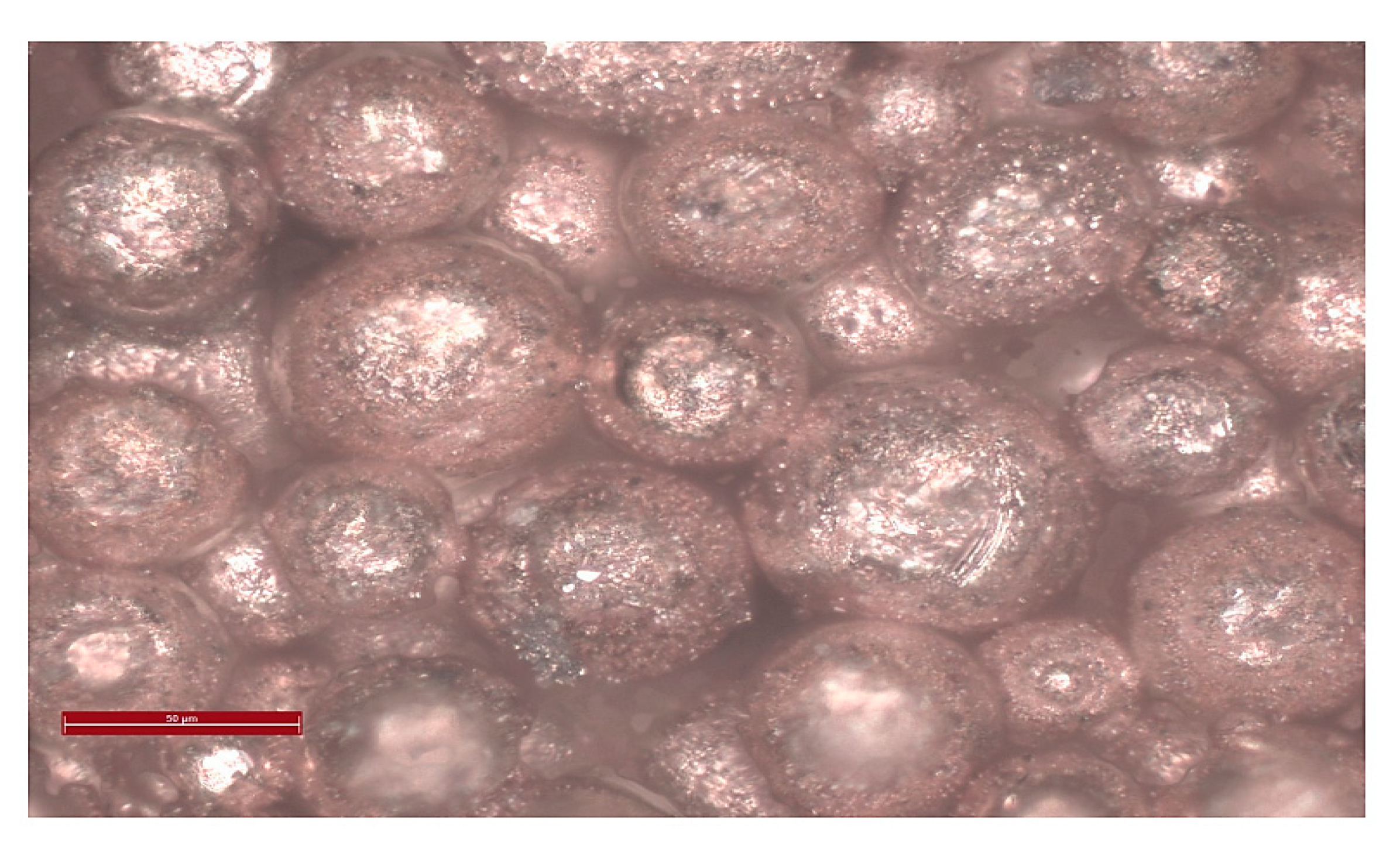

| High Pressure Processes | Acronyms | Equipment (Tool) | Applications Material Processing | T (°C) | P (MPa) | |
|---|---|---|---|---|---|---|
| Vessel (Force isostatic) | High Hydrostatic Pressing High Pressure Processing | HHP HPP | Tank, autoclave | Pascalization, decontamination, sterilization, disinfection of biological materials (Foods, Pharmacology, Medical) | 20 | xxx |
| Cold isostatic Pressing | CIP | Tank, vessel | Compaction of powder | 20 | xxx | |
| Freeze Isostatic Pressing | FIP | Vessel | Consolidation of powder | T < 0 °C | xxx | |
| Autoclaving (Steam sterilization) | Autoclave, tank | Decontamination, sterilization, disinfection in medical | 132 | P < 1 | ||
| High Pressure (isostatic) | HyCG HyCr HyPu | Reactor, autoclave, bomb, vessel | Hydrothermal Crystal Growth Hydrothermal Crystallization Hydrothermal Purification | 1000 | xxx | |
| Hot Isostatic Pressing | HIP | Tank, autoclave, bomb | Compaction of powder, sintering | 1000 | xxx | |
| Reactive Hydrothermal Liquid-Phase Densification | rHLPD | Autoclave | Infiltration of permeable green compacts by aqueous solutions + reaction under hydrothermal conditions | 240 | unknown | |
| Hydrothermal Reaction-Sintering | HRS | Sealed capsule | Sintering of powder by hydrothermal oxidation of a metal + diffusion of H2 from the capsule + sintering of the oxide powder formed | 900 | xxx | |
| Piston –cylinder (Force on 1 axe) | Uniaxial Pressing (ambient T) | UP | Non Leak-proof set-up: die, chamber, mold, cylinder, pelletizer | Compaction of powder | 20 | xxx |
| Hydro Pressure Sintering (ambient T) | HyPS(≈HyS) | Leak-proof set-up | Compaction of powder, consolidation | 20 | xxx | |
| Cold Sintering Process(ambient T) | CSP (20 °C) | Non leak-proof set-up | Compaction of powder, consolidation | 20 | xxx | |
| High-pressure torsion | HPT | Anvils in rotation while pressing | Pre-compaction & subsequent consolidation | 20 | GPa | |
| Uniaxial pressing ultrasonic | PUA | Non Leak-proof set-up: mold | Compaction of powder by Uniaxial pressing + simultaneous powerful ultrasonic action | 20 | xxx | |
| Uniaxial Hot Pressing (dry materials) | UHP | Chamber = Non leak-proof set-up (Heating by Induction RF exists) | Sintering of powder | 1000 | xx | |
| Uniaxial Hot Pressing (humid materials) | HyS CSP (T > 20 °C) | Autoclave (2 openings) = Leak-proof set-up (Heating by Induction RF exists) Non leak-proof set-up | Sintering of powder Sintering of powder | 500 200 | xxx | |
| Hydrothermal Hot Pressing | HHP (=HyS) | Autoclave (2 openings) = Leak-proof set-up | Sintering of powder | 250 | xxx | |
| Oscillatory pressure sintering | OPS | Non leak-proof set-up: Graphite die | Sintering of powder | 1300 | xx + x | |
| High Pressure | HP-HT, HP-SPS | Belt, Bridgman | Sintering of powder | 1800 | GPa | |
| Multi-anvils (Force on multi axes) | Ultra-high pressure sintering | UHPS | Multianvils (1 stage: 3 axes, 2 stages: Kawai, Walker) | Sintering of powder | 2200 | GPa |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prakasam, M.; Silvain, J.-F.; Largeteau, A. Innovative High-Pressure Fabrication Processes for Porous Biomaterials—A Review. Bioengineering 2021, 8, 170. https://doi.org/10.3390/bioengineering8110170
Prakasam M, Silvain J-F, Largeteau A. Innovative High-Pressure Fabrication Processes for Porous Biomaterials—A Review. Bioengineering. 2021; 8(11):170. https://doi.org/10.3390/bioengineering8110170
Chicago/Turabian StylePrakasam, Mythili, Jean-François Silvain, and Alain Largeteau. 2021. "Innovative High-Pressure Fabrication Processes for Porous Biomaterials—A Review" Bioengineering 8, no. 11: 170. https://doi.org/10.3390/bioengineering8110170
APA StylePrakasam, M., Silvain, J.-F., & Largeteau, A. (2021). Innovative High-Pressure Fabrication Processes for Porous Biomaterials—A Review. Bioengineering, 8(11), 170. https://doi.org/10.3390/bioengineering8110170








