Biotechnological Conversion of Grape Pomace to Poly(3-hydroxybutyrate) by Moderately Thermophilic Bacterium Tepidimonas taiwanensis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microorganisms and Cultivation
2.2. Verification of PHA Production at Genotype and Phenotype Level
2.3. Grape Pomace Extract
3. Results and Discussion
3.1. PHA-Related Genes of Strain T. taiwanensis LMG 22826
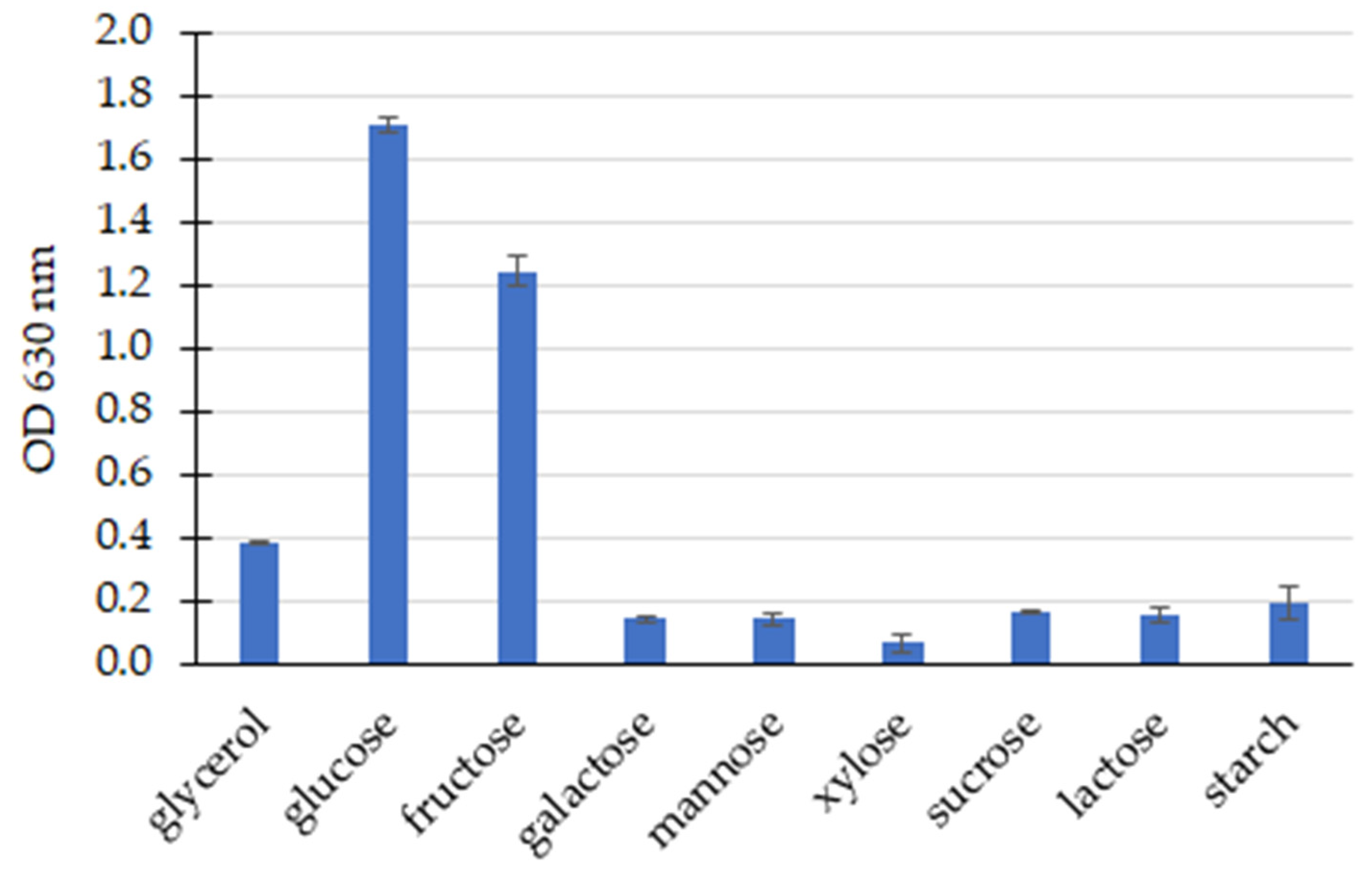
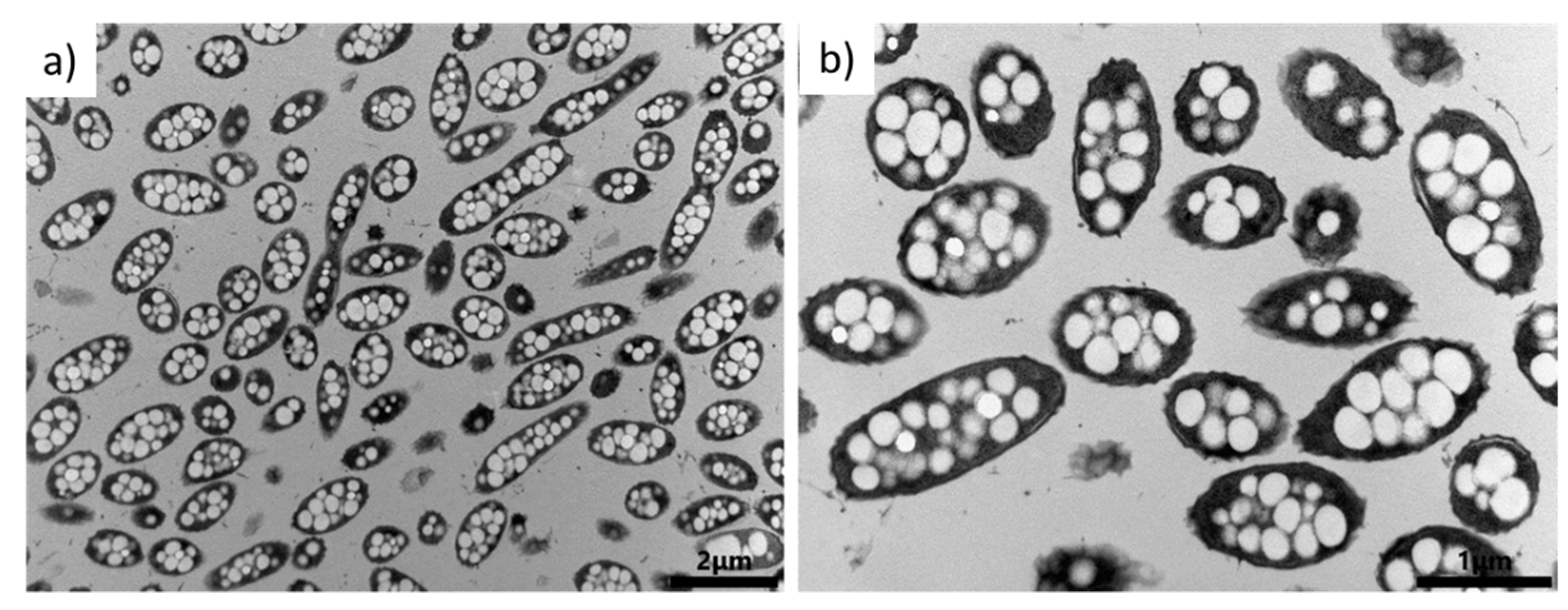
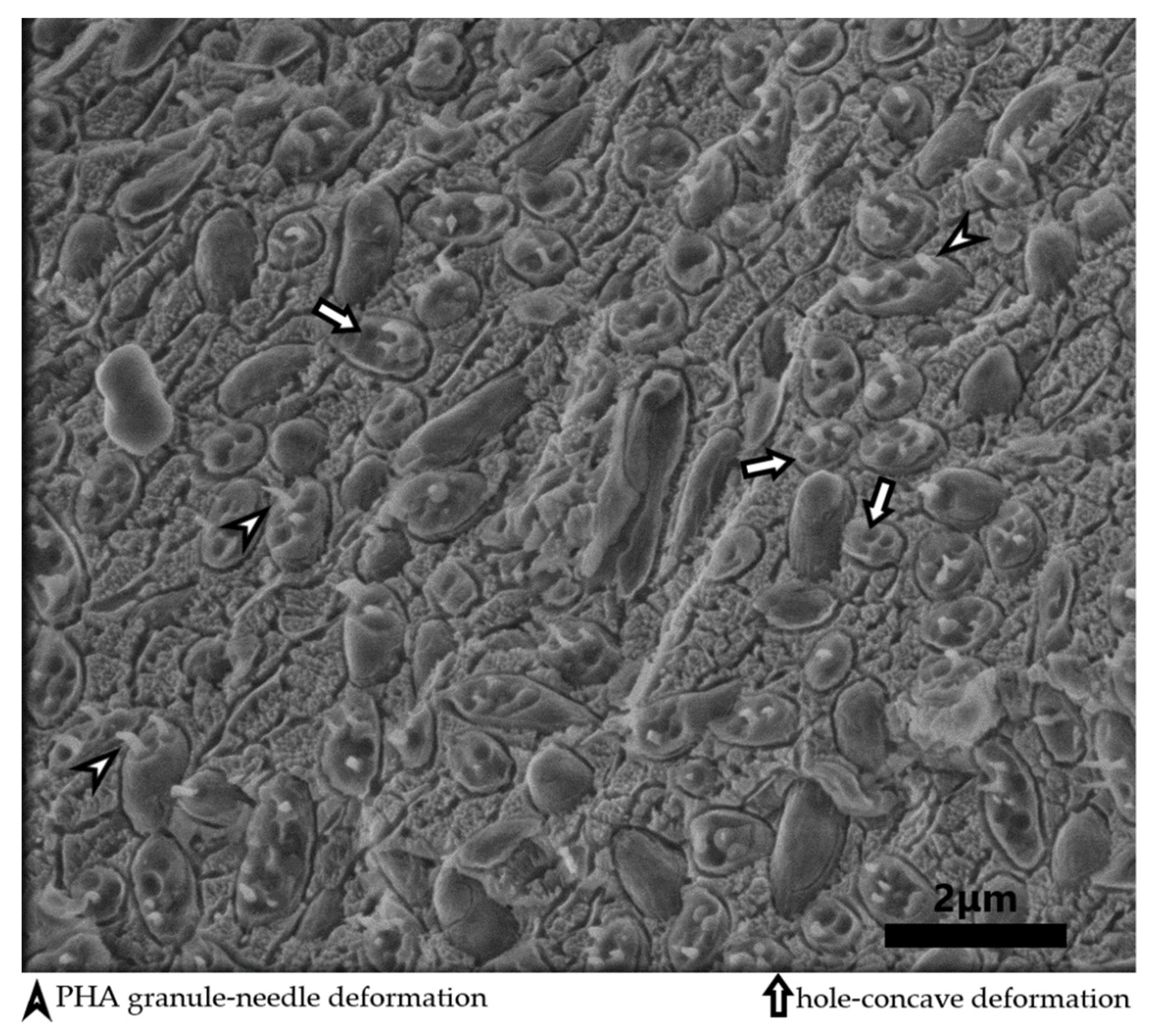
3.2. Screening of PHA Production and the Influence of Culture Parameters on PHA Production
3.3. Use of Grape Pomace to Produce PHA
3.3.1. Characterization of Extract of Grape Pomace
3.3.2. Grape Pomace Extracts as a Carbon Source for PHA Production
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Lim, H.; Chuah, J.; Chek, M.; Tan, H.; Hakoshima, T.; Sudesh, K. Identification of regions affecting enzyme activity, substrate binding, dimer stabilization and polyhydroxyalkanoate (PHA) granule morphology in the PHA synthase of Aquitalea sp. USM4. Int. J. Biol. Macromol. 2021, 186, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Raza, Z.; Abid, S.; Banat, I. Polyhydroxyalkanoates: Characteristics, production, recent developments and applications. Int. Biodeterior. Biodegrad. 2018, 126, 45–56. [Google Scholar] [CrossRef]
- Peña, C.; Castillo, T.; García, A.; Millán, M.; Segura, D. Biotechnological strategies to improve production of microbial poly-(3-hydroxybutyrate): A review of recent research work. Microb. Biotechnol. 2014, 7, 278–293. [Google Scholar] [CrossRef] [PubMed]
- Yadav, B.; Talan, A.; Tyagi, R.; Drogui, P. Concomitant production of value-added products with polyhydroxyalkanoate (PHA) synthesis: A review. Bioresour. Technol. 2021, 337, 125419. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.; Kalia, V. Biomedical Applications of Polyhydroxyalkanoates. Indian J. Microbiol. 2017, 57, 261–269. [Google Scholar] [CrossRef]
- Chen, G.; Patel, M. Plastics Derived from Biological Sources: Present and Future. Chem. Rev. 2012, 112, 2082–2099. [Google Scholar] [CrossRef]
- Poltronieri, P.; Kumar, P. Polyhydroxyalcanoates (PHAs) in Industrial Applications. In Handbook of Ecomaterials; Martínez, L., Kharissova, O., Kharisov, B., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 1–30. ISBN 978-3-319-48281-1. [Google Scholar]
- Crutchik, D.; Franchi, O.; Caminos, L.; Jeison, D.; Belmonte, M.; Pedrouso, A.; Val del Rio, A.; Mosquera-Corral, A.; Campos, J. Polyhydroxyalkanoates (PHAs) Production: A Feasible Economic Option for the Treatment of Sewage Sludge in Municipal Wastewater Treatment Plants? Water 2020, 12, 1118. [Google Scholar] [CrossRef]
- Rampelotto, P. Extremophiles and Extreme Environments. Life 2013, 3, 482–485. [Google Scholar] [CrossRef]
- Berlemont, R.; Gerday, C. Extremophiles. In Comprehensive Biotechnology; Elsevier: Amsterdam, The Netherlands, 2011; pp. 229–242. [Google Scholar]
- Irwin, J. Overview of extremophiles and their food and medical applications. Physiol. Biotechnol. Asp. Extrem. 2020, 65–87. [Google Scholar] [CrossRef]
- Dumorne, K.; Cordova, D.; Astorga-Elo, M.; Renganathan, P. Extremozymes: A Potential Source for Industrial Applications. J. Microbiol. Biotechnol. 2017, 27, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Jiang, X. Next generation industrial biotechnology based on extremophilic bacteria. Curr. Opin. Biotechnol. 2018, 50, 94–100. [Google Scholar] [CrossRef]
- Quillaguamán, J.; Guzmán, H.; Van-Thuoc, D.; Hatti-Kaul, R. Synthesis and production of polyhydroxyalkanoates by halophiles: Current potential and future prospects. Appl. Microbiol. Biotechnol. 2010, 85, 1687–1696. [Google Scholar] [CrossRef]
- Sedlacek, P.; Slaninova, E.; Koller, M.; Nebesarova, J.; Marova, I.; Krzyzanek, V.; Obruca, S. PHA granules help bacterial cells to preserve cell integrity when exposed to sudden osmotic imbalances. New Biotechnol. 2019, 49, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Alsafadi, D.; Al-Mashaqbeh, O. A one-stage cultivation process for the production of poly-3-(hydroxybutyrate-co-hydroxyvalerate) from olive mill wastewater by Haloferax mediterranei. New Biotechnol. 2017, 34, 47–53. [Google Scholar] [CrossRef]
- Tan, D.; Wu, Q.; Chen, J.; Chen, G. Engineering Halomonas TD01 for the low-cost production of polyhydroxyalkanoates. Metab. Eng. 2014, 26, 34–47. [Google Scholar] [CrossRef]
- Kucera, D.; Pernicová, I.; Kovalcik, A.; Koller, M.; Mullerova, L.; Sedlacek, P.; Mravec, F.; Nebesarova, J.; Kalina, M.; Marova, I.; et al. Characterization of the promising poly(3-hydroxybutyrate) producing halophilic bacterium Halomonas halophila. Bioresour. Technol. 2018, 256, 552–556. [Google Scholar] [CrossRef]
- Rivera-Terceros, P.; Tito-Claros, E.; Torrico, S.; Carballo, S.; Van-Thuoc, D.; Quillaguamán, J. Production of poly(3-hydroxybutyrate) by Halomonas boliviensis in an air-lift reactor. J. Biol. Res.-Thessalon. 2015, 22, 8. [Google Scholar] [CrossRef] [Green Version]
- Kulkarni, S.; Kanekar, P.; Nilegaonkar, S.; Sarnaik, S.; Jog, J. Production and characterization of a biodegradable poly (hydroxybutyrate-co-hydroxyvalerate) (PHB-co-PHV) copolymer by moderately haloalkalitolerant Halomonas campisalis MCM B-1027 isolated from Lonar Lake, India. Bioresour. Technol. 2010, 101, 9765–9771. [Google Scholar] [CrossRef] [PubMed]
- Pernicova, I.; Kucera, D.; Nebesarova, J.; Kalina, M.; Novackova, I.; Koller, M.; Obruca, S. Production of polyhydroxyalkanoates on waste frying oil employing selected Halomonas strains. Bioresour. Technol. 2019, 292, 122028. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.; Willems, A.; Steinbüchel, A. Isolation and characterization of new poly(3HB)-accumulating star-shaped cell-aggregates-forming thermophilic bacteria. J. Appl. Microbiol. 2010, 109, 1579–1590. [Google Scholar] [CrossRef]
- Sheu, D.; Chen, W.; Yang, J.; Chang, R. Thermophilic bacterium Caldimonas taiwanensis produces poly(3-hydroxybutyrate-co-3-hydroxyvalerate) from starch and valerate as carbon sources. Enzym. Microb. Technol. 2009, 44, 289–294. [Google Scholar] [CrossRef]
- Pernicova, I.; Novackova, I.; Sedlacek, P.; Kourilova, X.; Kalina, M.; Kovalcik, A.; Koller, M.; Nebesarova, J.; Krzyzanek, V.; Hrubanova, K.; et al. Introducing the Newly Isolated Bacterium Aneurinibacillus sp. H1 as an Auspicious Thermophilic Producer of Various Polyhydroxyalkanoates (PHA) Copolymers–1. Isolation and Characterization of the Bacterium. Polymers 2020, 12, 1235. [Google Scholar] [CrossRef]
- Kourilova, X.; Pernicova, I.; Sedlar, K.; Musilova, J.; Sedlacek, P.; Kalina, M.; Koller, M.; Obruca, S. Production of polyhydroxyalkanoates (PHA) by a thermophilic strain of Schlegelella thermodepolymerans from xylose rich substrates. Bioresour. Technol. 2020, 315, 123885. [Google Scholar] [CrossRef] [PubMed]
- 2019 Statistical Report on World Vitiviniculture, 1st ed.; International Organisation of Vine and Wine Intergovernmental Organisation: International Organisation of Vine and Wine Intergovernmental Organisation. 2020. Available online: http://oiv.int/public/medias/6782/oiv-2019-statistical-report-on-world-vitiviniculture.pdf (accessed on 9 September 2021).
- Moreno, A.; Ballesteros, M.; Negro, M. Biorefineries for the valorization of food processing waste. In The Interaction of Food Industry and Environment; Academic Press: Cambridge, MA, USA, 2020; pp. 155–190. [Google Scholar]
- Dávila, I.; Robles, E.; Egüés, I.; Labidi, J.; Gullón, P. The Biorefinery Concept for the Industrial Valorization of Grape Processing By-Products. In Handbook of Grape Processing By-Products; Academic Press: Cambridge, MA, USA, 2017; pp. 29–53. [Google Scholar]
- Antonić, B.; Jančíková, S.; Dordević, D.; Tremlová, B. Grape Pomace Valorization: A Systematic Review and Meta-Analysis. Foods 2020, 9, 1627. [Google Scholar] [CrossRef]
- González-Paramás, A.; Esteban-Ruano, S.; Santos-Buelga, C.; de Pascual-Teresa, S.; Rivas-Gonzalo, J. Flavanol Content and Antioxidant Activity in Winery Byproducts. J. Agric. Food Chem. 2004, 52, 234–238. [Google Scholar] [CrossRef]
- Manios, T. The composting potential of different organic solid wastes: Experience from the island of Crete. Environ. Int. 2004, 29, 1079–1089. [Google Scholar] [CrossRef]
- Sánchez, A.; Ysunza, F.; Beltrán-García, M.; Esqueda, M. Biodegradation of Viticulture Wastes by Pleurotus: A Source of Microbial and Human Food and Its Potential Use in Animal Feeding. J. Agric. Food Chem. 2002, 50, 2537–2542. [Google Scholar] [CrossRef]
- Zacharof, M. Grape Winery Waste as Feedstock for Bioconversions: Applying the Biorefinery Concept. Waste Biomass Valorization 2017, 8, 1011–1025. [Google Scholar] [CrossRef] [Green Version]
- Taurino, R.; Ferretti, D.; Cattani, L.; Bozzoli, F.; Bondioli, F. Lightweight clay bricks manufactured by using locally available wine industry waste. J. Build. Eng. 2019, 26, 100892. [Google Scholar] [CrossRef]
- Kovalcik, A.; Pernicova, I.; Obruca, S.; Szotkowski, M.; Enev, V.; Kalina, M.; Marova, I. Grape winery waste as a promising feedstock for the production of polyhydroxyalkanoates and other value-added products. Food Bioprod. Process. 2020, 124, 1–10. [Google Scholar] [CrossRef]
- Follonier, S. Pilot-scale Production of Functionalized mcl-PHA from Grape Pomace Supplemented with Fatty Acids. Chem. Biochem. Eng. Q. 2015, 29, 113–121. [Google Scholar] [CrossRef]
- Chen, T.; Chou, Y.; Chen, W.; Arun, B.; Young, C. Tepidimonas taiwanensis sp. nov., a novel alkaline-protease-producing bacterium isolated from a hot spring. Extremophiles 2006, 10, 35–40. [Google Scholar] [CrossRef]
- Obruca, S.; Petrik, S.; Benesova, P.; Svoboda, Z.; Eremka, L.; Marova, I. Utilization of oil extracted from spent coffee grounds for sustainable production of polyhydroxyalkanoates. Appl. Microbiol. Biotechnol. 2014, 98, 5883–5890. [Google Scholar] [CrossRef]
- Mravec, F.; Obruca, S.; Krzyzanek, V.; Sedlacek, P.; Hrubanova, K.; Samek, O.; Kucera, D.; Benesova, P.; Nebesarova, J.; Steinbüchel, A. Accumulation of PHA granules in Cupriavidus necator as seen by confocal fluorescence microscopy. FEMS Microbiol. Lett. 2016, 363, fnw094. [Google Scholar] [CrossRef] [Green Version]
- Kouřilová, X.; Schwarzerová, J.; Pernicová, I.; Sedlář, K.; Mrázová, K.; Krzyžánek, V.; Nebesářová, J.; Obruča, S. The First Insight into Polyhydroxyalkanoates Accumulation in Multi-Extremophilic Rubrobacter xylanophilus and Rubrobacter spartanus. Microorganisms 2021, 9, 909. [Google Scholar] [CrossRef]
- Obruca, S.; Sedlacek, P.; Krzyzanek, V.; Mravec, F.; Hrubanova, K.; Samek, O.; Kucera, D.; Benesova, P.; Marova, I.; Chen, G.-Q. Accumulation of Poly(3-hydroxybutyrate) Helps Bacterial Cells to Survive Freezing. PLoS ONE 2016, 11, e0157778. [Google Scholar] [CrossRef] [Green Version]
- Singleton, V.; Rossi, J. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Eiticulture 1965, 16, 144–158. [Google Scholar]
- Tan, I.; Foong, C.; Tan, H.; Lim, H.; Zain, N.; Tan, Y.; Hoh, C.; Sudesh, K. Polyhydroxyalkanoate (PHA) synthase genes and PHA-associated gene clusters in Pseudomonas spp. and Janthinobacterium spp. isolated from Antarctica. J. Biotechnol. 2020, 313, 18–28. [Google Scholar] [CrossRef]
- Pan, W.; Perrotta, J.; Stipanovic, A.; Nomura, C.; Nakas, J. Production of polyhydroxyalkanoates by Burkholderia cepacia ATCC 17759 using a detoxified sugar maple hemicellulosic hydrolysate. J. Ind. Microbiol. Biotechnol. 2012, 39, 459–469. [Google Scholar] [CrossRef]
- Kourmentza, C.; Costa, J.; Azevedo, Z.; Servin, C.; Grandfils, C.; De Freitas, V.; Reis, M. Burkholderia thailandensis as a microbial cell factory for the bioconversion of used cooking oil to polyhydroxyalkanoates and rhamnolipids. Bioresour. Technol. 2018, 247, 829–837. [Google Scholar] [CrossRef]
- Ibrahim, M.; Steinbüchel, A. High-Cell-Density Cyclic Fed-Batch Fermentation of a Poly(3-Hydroxybutyrate)-Accumulating Thermophile, Chelatococcus sp. Strain MW10. Appl. Environ. Microbiol. 2010, 76, 7890–7895. [Google Scholar] [CrossRef] [Green Version]
- Keenan, T.; Nakas, J.; Tanenbaum, S. Polyhydroxyalkanoate copolymers from forest biomass. J. Ind. Microbiol. Biotechnol. 2006, 33, 616–626. [Google Scholar] [CrossRef]
- Norhafini, H.; Huong, K.; Amirul, A. High PHA density fed-batch cultivation strategies for 4HB-rich P(3HB-co-4HB) copolymer production by transformant Cupriavidus malaysiensis USMAA1020. Int. J. Biol. Macromol. 2019, 125, 1024–1032. [Google Scholar] [CrossRef]
- Venkitasamy, C.; Zhao, L.; Zhang, R.; Pan, Z. Grapes. In Integrated Processing Technologies for Food and Agricultural By-Products; Elsevier: Amsterdam, The Netherlands, 2019; pp. 133–163. ISBN 9780128141380. [Google Scholar]
- Chowdhary, P.; Gupta, A.; Gnansounou, E.; Pandey, A.; Chaturvedi, P. Current trends and possibilities for exploitation of Grape pomace as a potential source for value addition. Environ. Pollut. 2021, 278, 116796. [Google Scholar] [CrossRef]
- Hogan, S.; Zhang, L.; Li, J.; Sun, S.; Canning, C.; Zhou, K. Antioxidant rich grape pomace extract suppresses postprandial hyperglycemia in diabetic mice by specifically inhibiting alpha-glucosidase. Nutr. Metab. 2010, 7, 71. [Google Scholar] [CrossRef] [Green Version]
- Pinto, D.; Cádiz-Gurrea, M.; Silva, A.; Delerue-Matos, C.; Rodrigues, F. Cosmetics—food waste recovery. In Food Waste Recovery; Elsevier: Amsterdam, The Netherlands, 2021; pp. 503–528. ISBN 9780128205631. [Google Scholar]
- Luchian, C.; Cotea, V.; Vlase, L.; Toiu, A.; Colibaba, L.; Răschip, I.; Nadăş, G.; Gheldiu, A.; Tuchiluş, C.; Rotaru, L.; et al. Antioxidant and antimicrobial effects of grape pomace extracts. BIO Web Conf. 2019, 15, 04006. [Google Scholar] [CrossRef]
- Shang, L.; Jiang, M.; Chang, H.N. P oly(3-hydroxybutyrate) synthesis in fed-batch culture of Ralstonia eutropha with phosphate limitation under different glucose concentrations. Biotechnol. Lett. 2003, 25, 1415–1419. [Google Scholar] [CrossRef]

| Temperature (°C) | CDM (g/L) | P(3HB) (%) | PHB (g/L) |
|---|---|---|---|
| 45 | 5.08 ± 0.11 | 67.3 ± 2.1 | 3.417 ± 0.076 |
| 50 | 5.45 ± 0.22 | 65.5 ± 5.7 | 3.559 ± 0.169 |
| 55 | 5.36 ± 0.16 | 63.9 ± 2.9 | 3.421 ± 0.056 |
| 60 | 1.60 ± 0.08 | 49.8 ± 8.5 | 0.801 ± 0.178 |
| Precursors | CDM (g/L) | P(3HB) (%) | PHA (g/L) | 3HV (mol. %) | 4HB (mol. %) |
|---|---|---|---|---|---|
| control | 4.21 ± 0.19 | 49.2 ± 4.1 | 2.069 ± 0.041 | n.d. | n.d. |
| 1,4-butanediol | 0.32 ± 0.12 | 14.2 ± 2.5 | 0.091 ± 0.022 | n.d. | n.d. |
| γ-butyrolactone | 0.08 ± 0.01 | n.d | n.d. | n.d. | n.d. |
| valeric acid | 0.03 ± 0.01 | n.d | n.d. | n.d. | n.d. |
| sodium propionate | 4.16 ± 0.17 | 44.8 ± 3.8 | 1.860 ± 0.130 | 6.6 | n.d. |
| Composition of Grape Pomace Extracts | Muller Thurgau weiss | Veltliner Fruehrot | Palava | Sauvignon | Pinot Blanc | Blaufraenkisch (Red) | Blaufraenkisch (Rose) | |
|---|---|---|---|---|---|---|---|---|
| Organic acid (g/L) | Citric acid | 0.009 | 0.096 | 0.027 | 0.147 | 0.137 | 0.017 | 0.104 |
| Malic acid | 0.620 | 0.939 | 0.956 | 0.979 | 0.0849 | 0.319 | 0.877 | |
| Succinic acid | 0.093 | 0.054 | 0.045 | 0.101 | 0.115 | 0.107 | 0.012 | |
| Lactic acid | n.d. | 0.006 | 0.006 | 0.009 | 0.014 | 0.022 | 0.001 | |
| Acetic acid | 0.131 | 0.156 | 0.059 | 0.100 | 0.119 | 0.071 | 0.054 | |
| Formic acid | n.d. | 0.004 | 0005 | 0.004 | 0.006 | 0.004 | n.d. | |
| Propionic acid | 0.062 | 0.067 | 0.064 | 0.064 | 0.066 | 0.061 | 0.052 | |
| Total polyphenols (g/L) | 0.453 | 0.301 | 0.520 | 0.160 | 0.231 | 0.821 | 0.548 | |
| Sugars (g/L) | Fructose | 5.49 | 4.73 | 6.68 | 2.84 | 2.97 | 11.65 | 9.04 |
| Glucose | 5.71 | 6.61 | 6.66 | 4.13 | 4.31 | 11.09 | 8.22 | |
| Total sugars (g/L) | 11.20 | 11.35 | 13.34 | 6.97 | 7.28 | 22.73 | 17.26 | |
| Grape Pomace From | CDM (g/L) | P(3HB) (%) | P(3HB) (g/L) | Residual Fructose (g/L) | Residual Glucose (g/L) | YP/S |
|---|---|---|---|---|---|---|
| Muller Thurgau weiss | 0.35 ± 0.02 | 1.4 ± 0.8 | 0.005 ± 0.001 | 4.65 | 4.66 | 0.00 |
| Veltliner fruehrot | 4.36 ± 0.04 | 47.9 ± 2.3 | 2.090 ± 0.081 | 0.74 | 0.38 | 0.21 |
| Palava | 0.80 ± 0.04 | 3.7 ± 1.2 | 0.030 ± 0.001 | 6.06 | 6.11 | 0.03 |
| Sauvignon | 3.37 ± 0.13 | 44.5 ± 1.7 | 1.498 ± 0.041 | 0.05 | n.d. | 0.22 |
| Pinot blanc | 3.62 ± 0.01 | 43.8 ± 2.5 | 1.585 ± 0.046 | 0.12 | n.d. | 0.22 |
| Blaufraenkisch (red) | 0.27 ± 0.03 | 8.4 ± 2.6 | 0.022 ± 0.001 | 9.06 | 8.69 | 0.00 |
| Blaufraenkisch (rose) | 1.92 ± 0.35 | 12.3 ± 1.9 | 0.236 ± 0.049 | 8.62 | 8.04 | 0.39 |
| Grape Pomace From | CDM [g/L] | P(3HB) [%] | P(3HB) [g/L] | Residual Fructose [g/L] | Residual Glucose [g/L] | YP/S |
|---|---|---|---|---|---|---|
| Muller Thurgau weiss | 1.09 ± 0.24 | 11.4 ± 1.2 | 0.115 ± 0.013 | n.d. | n.d. | 0.02 |
| Veltliner fruehrot | 2.39 ± 0.12 | 36.9 ± 2.4 | 0.883 ± 0.044 | n.d. | n.d. | 0.16 |
| Palava | 2.59 ± 0.05 | 31.2 ± 3.4 | 0.619 ± 0.070 | n.d. | n.d. | 0.09 |
| Sauvignon | 1.32 ± 0.08 | 27.2 ± 1.7 | 0.359 ± 0.054 | n.d. | n.d. | 0.10 |
| Pinot blanc | 1.97 ± 0.06 | 28.6 ± 5.2 | 0.564 ± 0.013 | n.d. | n.d. | 0.15 |
| Blaufraenkisch (red) | 2.87 ± 0.81 | 41.9 ± 3.7 | 1.201 ± 0.076 | n.d. | n.d. | 0.11 |
| Blaufraenkisch (rose) | 2.94 ± 0.09 | 47.7 ± 2.5 | 1.399 ± 0.041 | n.d. | n.d. | 0.16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kourilova, X.; Pernicova, I.; Vidlakova, M.; Krejcirik, R.; Mrazova, K.; Hrubanova, K.; Krzyzanek, V.; Nebesarova, J.; Obruca, S. Biotechnological Conversion of Grape Pomace to Poly(3-hydroxybutyrate) by Moderately Thermophilic Bacterium Tepidimonas taiwanensis. Bioengineering 2021, 8, 141. https://doi.org/10.3390/bioengineering8100141
Kourilova X, Pernicova I, Vidlakova M, Krejcirik R, Mrazova K, Hrubanova K, Krzyzanek V, Nebesarova J, Obruca S. Biotechnological Conversion of Grape Pomace to Poly(3-hydroxybutyrate) by Moderately Thermophilic Bacterium Tepidimonas taiwanensis. Bioengineering. 2021; 8(10):141. https://doi.org/10.3390/bioengineering8100141
Chicago/Turabian StyleKourilova, Xenie, Iva Pernicova, Michaela Vidlakova, Roman Krejcirik, Katerina Mrazova, Kamila Hrubanova, Vladislav Krzyzanek, Jana Nebesarova, and Stanislav Obruca. 2021. "Biotechnological Conversion of Grape Pomace to Poly(3-hydroxybutyrate) by Moderately Thermophilic Bacterium Tepidimonas taiwanensis" Bioengineering 8, no. 10: 141. https://doi.org/10.3390/bioengineering8100141
APA StyleKourilova, X., Pernicova, I., Vidlakova, M., Krejcirik, R., Mrazova, K., Hrubanova, K., Krzyzanek, V., Nebesarova, J., & Obruca, S. (2021). Biotechnological Conversion of Grape Pomace to Poly(3-hydroxybutyrate) by Moderately Thermophilic Bacterium Tepidimonas taiwanensis. Bioengineering, 8(10), 141. https://doi.org/10.3390/bioengineering8100141







