Operative Workflow from CT to 3D Printing of the Heart: Opportunities and Challenges
Abstract
:1. Introduction
2. State of the Art
2.1. Reconstruction of an Anatomical District: Methods and Tools
2.2. Additive Manufacturing in the Cardiovascular Field: A Framework
2.3. Additive Manufacturing in the Cardiovascular Field: Applications
- (1)
- Through 3D printing, accurate educational tools able to illustrate complex cardiovascular anatomy and pathology can be created [15]. Compared to 2D images, 3D renderings guarantee a better understanding of the human body and of fine anatomical details that may influence the management of the underlying disease [18]. This is especially true if we think about the complexity of the heart, above all in the presence of congenital heart diseases [19]. For this kind of application, the model is usually intended for visual inspection only, so the focus is on creating a high-resolution replica of the anatomy, while mechanical aspects are of secondary importance [18].
- (2)
- Communication between cardiologists, cardiac surgeons and patients is very challenging, given also the complexity of medical terminology [20]. Therefore, the introduction of 3D-printed heart models during routine clinical consultations could be an appreciated improvement, as a preliminary study in the domain of congenital heart defects confirms [21].
- (3)
- Moreover, 3D printing can be used to create and analyze models before starting actual surgery on the patient. The creation of high-fidelity training simulators for specific surgical procedures is also a possibility. Every patient’s anatomy is different, so surgeons’ practice on human cadavers, animal models and generic mannequins has often little relevance to the actual patient on the table. Decision-making in those cases considered complex and non-routine can surely benefit from the availability of physical 3D models, allowing an effective replication of surgical procedures, such as dissections, suturing or device sizing and placement (e.g., heart valves [22]), thus reducing operative risks [23] and operative room time. The employment of distensible resins in these cases surely helps to increase the realism and the reliability of the simulation. Indeed, here, differently from point (1), there is the need to carefully mimic the biomechanical properties of the involved tissue or organ, thus providing more realistic haptic feedback. The careful choice and characterization of materials become compulsory. As some studies suggest [24,25,26], when experimented, the adaption of 3DP has shown a reduction in procedure time and optimization of device deployment by improving the anticipation of potential obstacles in surgical procedures [27].
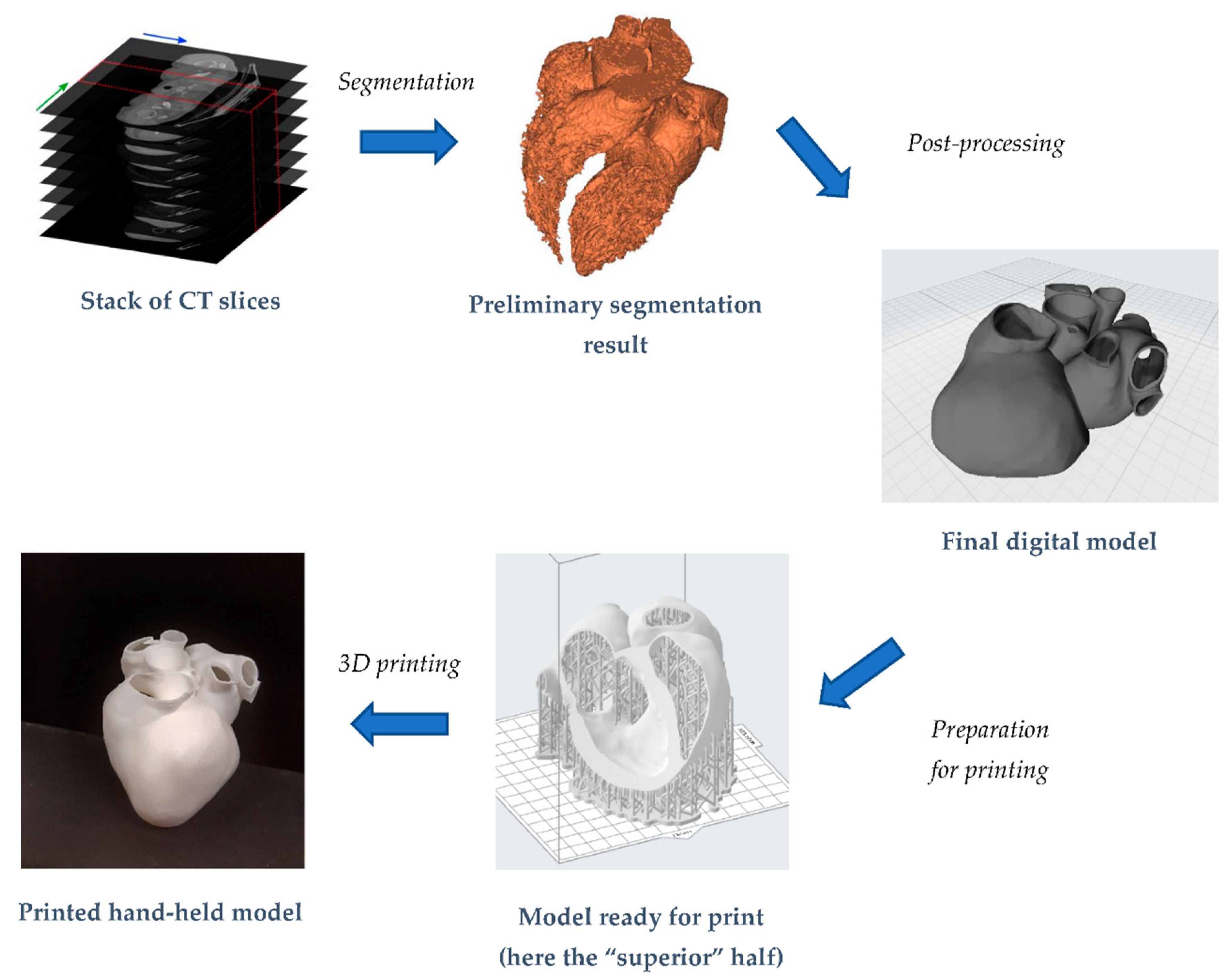
3. Digital Manufacturing of a Whole Heart Model
3.1. Reconstruction of the 3D Digital Model
3.2. Heart Model 3D Print
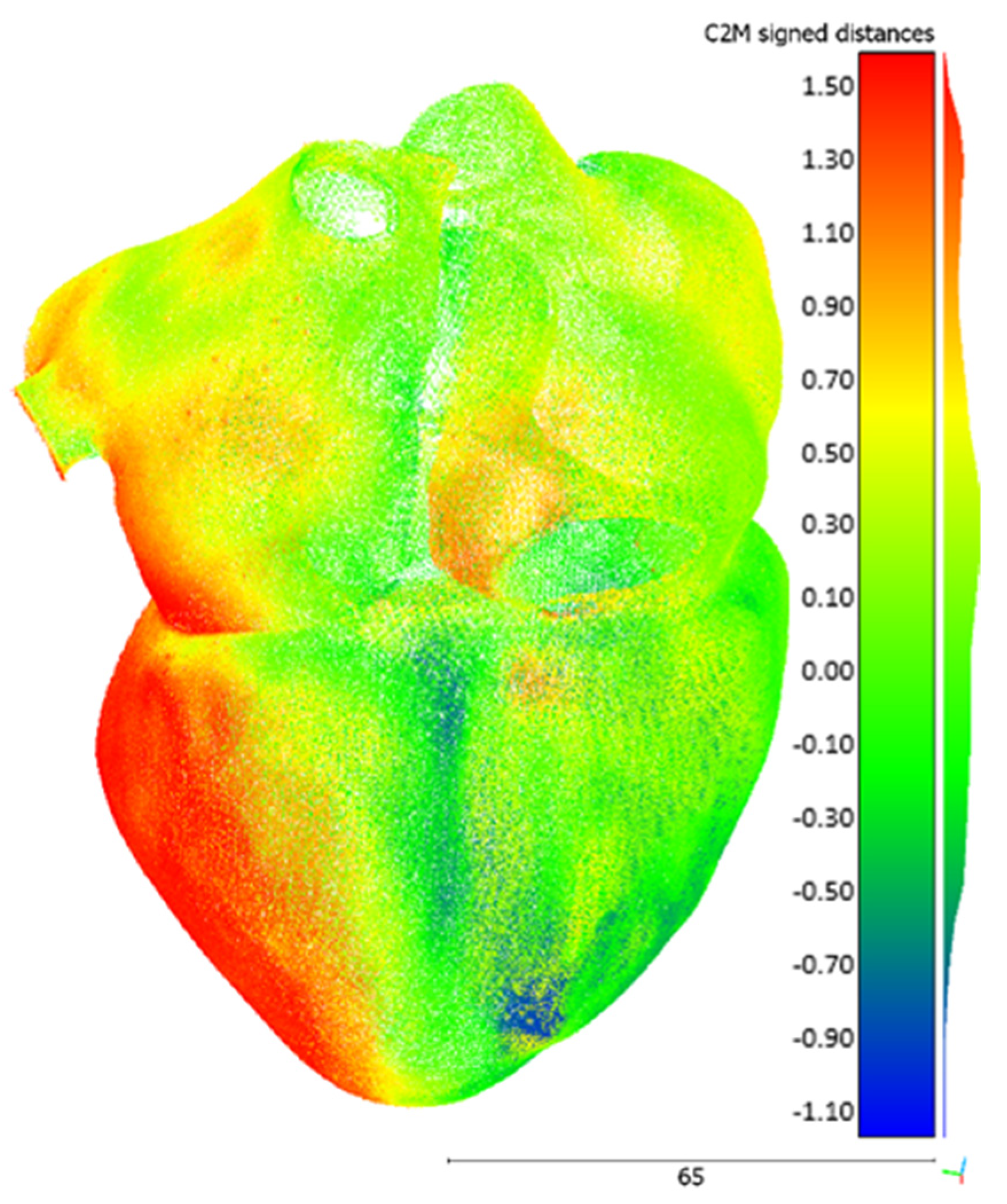
3.3. Dimensional Accuracy Evaluation
4. Conclusions and Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Coppini, G.; Diciotti, S.; Valli, G. Bioimmagini; Pàtron Editore: Bologna, Italy, 2012. [Google Scholar]
- Schmauss, D.; Haeberle, S.; Hagl, C.; Sodian, R. Three-dimensional printing in cardiac surgery and interventional cardiology: A single-centre experience. Eur. J. Cardio-Thorac. Surg. 2014, 47, 1044–1052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robinson, R.; Valindria, V.V.; Bai, W.; Oktay, O.; Kainz, B.; Suzuki, H.; Sanghvi, M.M.; Aung, N.; Paiva, J.M.; Zemrak, F.; et al. Automated quality control in image segmentation: Application to the UK Biobank cardiovascular magnetic resonance imaging study. J. Cardiovasc. Magn. Reson. 2019, 21, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Bade, R.; Haase, J.; Preim, B. Comparison of Fundamental Mesh Smoothing Algorithms for Medical Surface Models. In SimVis; Citeseer: Princeton, NJ, USA, 2006; pp. 289–304. [Google Scholar]
- Heimann, T.; Meinzer, H. Statistical shape models for 3D medical image segmentation: A review. Med. Image Anal. 2009, 13, 543–563. [Google Scholar] [CrossRef] [PubMed]
- Rathnayaka, K.; Sahama, T.; Schuetz, M.A.; Schmutz, B. Effects of CT image segmentation methods on the accuracy of long bone 3D reconstructions. Med. Eng. Phys. 2011, 33, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Lundervold, A.S.; Lundervold, A. An overview of deep learning in medical imaging focusing on MRI. Zeitschrift Medizinische Physik 2019, 29, 102–127. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Qin, C.; Qiu, H.; Tarroni, G.; Duan, J.; Bai, W.; Rueckert, D. Deep Learning for Cardiac Image Segmentation: A Review. Front. Cardiovasc. Med. 2020, 7, 25. [Google Scholar] [CrossRef]
- Wang, L.; Chitiboi, T.; Meine, H.; Günther, M.; Hahn, H.K. Principles and methods for automatic and semi-automatic tissue segmentation in MRI data. MAGMA 2016, 29, 95–110. [Google Scholar] [CrossRef]
- Ghelich Oghli, M.; Mohammadzadeh, M.; Mohammadzadeh, V.; Kadivar, S.; Mohammad Zadeh, A. Left Ventricle Segmentation Using a Combination of Region Growing and Graph Based. Iran. J. Radiol. 2017, 14, 2. [Google Scholar] [CrossRef] [Green Version]
- Abdullah, J.Y.; Omar, M.; Pritam, H.M.H.; Husein, A.; Rajion, Z.A. Comparison of 3D Reconstruction of Mandible for Pre-operative Planning Using Commercial and Open-Source Software; AIP: Melville, NY, USA, 2016; Volume 1791. [Google Scholar]
- Giannopoulos, A.A.; Mitsouras, D.; Yoo, S.; Liu, P.P.; Chatzizisis, Y.S.; Rybicki, F.J. Applications of 3D printing in cardiovascular diseases. Nat. Rev. Cardiol. 2016, 13, 701–718. [Google Scholar] [CrossRef]
- Lau, I.; Sun, Z. Three-dimensional printing in congenital heart disease: A systematic review. J. Med. Radial. Sci. 2018, 65, 226–236. [Google Scholar] [CrossRef]
- Uccheddu, F.; Carfagni, M.; Governi, L.; Furferi, R.; Volpe, Y.; Nocerino, E. 3D printing of cardiac structures from medical images: An overview of methods and interactive tools. Int. J. Interact. Des. Manuf. 2018, 12, 597–609. [Google Scholar] [CrossRef]
- Meier, L.M.; Meineri, M.; Qua Hiansen, J.; Horlick, E.M. Structural and congenital heart disease interventions: The role of three-dimensional printing. Neth. Heart J. 2017, 25, 65–75. [Google Scholar] [CrossRef] [Green Version]
- Ahangar, P.; Cooke, M.E.; Weber, M.H.; Rosenzweig, D.H. Current biomedical applications of 3D printing and additive manufacturing. Appl. Sci. 2019, 9, 1713. [Google Scholar] [CrossRef] [Green Version]
- Shearn, A.I.U.; Yeong, M.; Richard, M.; Ordoñez, M.V.; Pinchbeck, H.; Milano, E.G.; Hayes, A.; Caputo, M.; Biglino, G. Use of 3D Models in the Surgical Decision-Making Process in a Case of Double-Outlet Right Ventricle With Multiple Ventricular Septal Defects. Front. Pediatr. 2019, 7, 330. [Google Scholar] [CrossRef] [PubMed]
- Pietrabissa, A.; Marconi, S.; Negrello, E.; Mauri, V.; Peri, A.; Pugliese, L.; Marone, E.M.; Auricchio, F. An overview on 3D printing for abdominal surgery. Surg. Endosc. 2020, 34, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Vijayavenkataraman, S.; Fuh, J.Y.H.; Lu, W.F. 3D Printing and 3D Bioprinting in Pediatrics. Bioengineering 2017, 4, 63. [Google Scholar] [CrossRef] [Green Version]
- Milano, E.G.; Capelli, C.; Wray, J.; Biffi, B.; Layton, S.; Lee, M.; Caputo, M.; Taylor, A.M.; Schievano, S.; Biglino, G. Current and future applications of 3D printing in congenital cardiology and cardiac surgery. Br. J. Radiol. 2019, 92, 20180389. [Google Scholar] [CrossRef] [Green Version]
- Biglino, G.; Capelli, C.; Wray, J.; Schievano, S.; Leaver, L.; Khambadkone, S.; Giardini, A.; Derrick, G.; Jones, A.; Taylor, A.M. 3D-manufactured patient-specific models of congenital heart defects for communication in clinical practice: Feasibility and acceptability. BMJ Open 2015, 5, e007165. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, R.; Otto, C.; Bonow, R.; Carabello, B.; Erwin, J.; Guyton, R.; O’Gara, P.; Ruiz, C.; Skubas, N.; Sorajja, P.; et al. 2014 AHA/ACC Guideline for the Management of Patients with Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 2014, 129, e521–e643. [Google Scholar] [CrossRef] [Green Version]
- Haleem, A.; Javaid, M.; Saxena, A. Additive manufacturing applications in cardiology: A review. Egypt. Heart J. 2018, 70, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, S.; Grunert, R.; Mohr, F.W.; Falk, V. 3D-Imaging of cardiac structures using 3D heart models for planning in heart surgery: A preliminary study. Interact. Cardio Vasc. Thorac. Surg. 2008, 7, 6–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knox, K.; Kerber, C.W.; Singel, S.A.; Bailey, M.J.; Imbesi, S.G. Rapid prototyping to create vascular replicas from CT scan data: Making tools to teach, rehearse, and choose treatment strategies. Catheter. Cardiovasc. Interv. 2005, 65, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Valverde, I.; Gomez, G.; Coserria, J.F.; Suarez-Mejias, C.; Uribe, S.; Sotelo, J.; Velasco, M.N.; Santos De Soto, J.; Hosseinpour, A.; Gomez-Cia, T. 3D printed models for planning endovascular stenting in transverse aortic arch hypoplasia. Catheter. Cardiovasc. Interv. 2015, 85, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Abdelkarim, A.; Hageman, A.; Levi, D.S.; Aboulhosn, J. Operationalizing low-cost three-dimensional printing in planning for complex congenital cardiac interventions. Mater. Today Commun. 2018, 15, 171–174. [Google Scholar] [CrossRef]
- Gallo, M.; D’Onofrio, A.; Tarantini, G.; Nocerino, E.; Remondino, F.; Gerosa, G. 3D-printing model for complex aortic transcatheter valve treatment. Int. J. Cardiol. 2015, 210, 139–140. [Google Scholar] [CrossRef]
- Witschey, W.R.T.; Pouch, A.M.; McGarvey, J.R.; Ikeuchi, K.; Contijoch, F.; Levack, M.M.; Yushkevick, P.A.; Sehgal, C.M.; Jackson, B.M.; Gorman, R.C.; et al. Three-Dimensional Ultrasound-Derived Physical Mitral Valve Modeling. Annal. Thorac. Surg. 2014, 98, 691–694. [Google Scholar] [CrossRef] [Green Version]
- Costello, J.P.; Olivieri, L.J.; Krieger, A.; Thabit, O.; Marshall, M.B.; Yoo, S.; Kim, P.C.; Jonas, R.A.; Nath, D.S. Utilizing Three-Dimensional Printing Technology to Assess the Feasibility of High-Fidelity Synthetic Ventricular Septal Defect Models for Simulation in Medical Education. World J. Pediatr. Congenit. Heart Surg. 2014, 5, 421–426. [Google Scholar] [CrossRef]
- Yoo, S.; Spray, T.; Austin, E.H.; Yun, T.; van Arsdell, G.S. Hands-on surgical training of congenital heart surgery using 3-dimensional print models. J. Thorac. Cardiovasc. Surg. 2017, 153, 1530–1540. [Google Scholar] [CrossRef] [Green Version]
- Gómez-Ciriza, G.; Hussain, T.; Gómez-Cía, T.; Valverde, I. Potential of 3D-printed models in planning structural interventional procedures. Intervent. Cardiol. 2015, 7, 343–350. [Google Scholar] [CrossRef]
- Pellegrino, P.L.; Fassini, G.; Biase, M.; Tondo, C. Left Atrial Appendage Closure Guided by 3D Printed Cardiac Reconstruction: Emerging Directions and Future Trends. J. Cardiovasc. Electrophysiol. 2016, 27, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Wunderlich, N.C.; Beigel, R.; Swaans, M.J.; Ho, S.Y.; Siegel, R.J. Percutaneous interventions for left atrial appendage exclusion: Options, assessment, and imaging using 2D and 3D echocardiography. JACC Cardiovasc. Imag. 2015, 8, 472–488. [Google Scholar] [CrossRef] [Green Version]
- Chaowu, Y.; Hua, L.; Xin, S. Three-Dimensional Printing as an Aid in Transcatheter Closure of Secundum Atrial Septal Defect with Rim Deficiency: In Vitro Trial Occlusion Based on a Personalized Heart Model. Circulation 2016, 133, e608–e610. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmauss, D.; Schmitz, C.; Bigdeli, A.K.; Weber, S.; Gerber, N.; Beiras-Fernandez, A.; Schwarz, F.; Becker, C.; Kupatt, C.; Sodian, R. Three-Dimensional Printing of Models for Preoperative Planning and Simulation of Transcatheter Valve Replacement. Annal. Thorac. Surg. 2012, 93, e31–e33. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, K.M.; Lengua, C.G.; Weinberg, A.D.; Nielsen, J.C.; Sanz, J. Blood Pool Segmentation Results in Superior Virtual Cardiac Models than Myocardial Segmentation for 3D Printing. Pediatr. Cardiol. 2016, 37, 1028–1036. [Google Scholar] [CrossRef] [PubMed]
- Shiraishi, I.; Yamagishi, M.; Hamaoka, K.; Fukuzawa, M.; Yagihara, T. Simulative operation on congenital heart disease using rubber-like urethane stereolithographic biomodels based on 3D datasets of multislice computed tomography. Eur. J. Cardio-Thorac. Surg. 2010, 37, 302–306. [Google Scholar] [CrossRef] [Green Version]
- Bertolini, M.; Rossoni, M.; Colombo, G. Additive Manufacturing of a Compliant Multimaterial Heart Model. Comput. Aided Des. Appl. 2022, 19. in press. [Google Scholar]
- Zhuang, X. Challenges and Methodologies of Fully Automatic Whole Heart Segmentation: A Review. J. Healthc. Eng. 2013, 4, 371–408. [Google Scholar] [CrossRef] [Green Version]
- Lo Giudice, A.; Ronsivalle, V.; Grippaudo, C.; Lucchese, A.; Muraglie, S.; Lagravère, M.O.; Isola, G. One Step before 3D Printing—Evaluation of Imaging Software Accuracy for 3-Dimensional Analysis of the Mandible: A Comparative Study Using a Surface-to-Surface Matching Technique. Materials 2020, 13, 2798. [Google Scholar] [CrossRef]






| 3DP Technology | Employed Materials | Producer (Example) | Spatial Resolution | Costs (Printer + Material) | Printing Time | Print Volume | Mono-/Multi- Material | Further Notes |
|---|---|---|---|---|---|---|---|---|
| Fused Deposition Modeling (FDM) | Thermoplastic filaments | Ultimaker | Generally quite low | Relatively cheap | Long | Limited | Multi- | Anisotropy Wide variety of materials |
| Stereolithography (SLA) | Photo-sensitive resin | Formlabs | Very good (up to 0.025 mm) | Relatively cheap | Long | Limited | Mono- | Extensive post-processing |
| Selective Laser Sintering (SLS) | Powdered polymers | EOS | Good (up to 0.060 mm) | Expensive | Very long (heating and cooling phases) | Large | Mono- | Complex machine preparation Safety issues |
| Material jetting | Photo-polymers | Stratasys | Excellent (up to 0.014 mm) | Very expensive | Shorter | Large | Multi- | High printer encumbrance |
| Green | Post-Cured | Method | |
|---|---|---|---|
| Ultimate tensile strength | 38 MPa | 65 MPa | ASTM D 638-10 |
| Tensile modulus | 1.6 GPa | 2.8 GPa | ASTM D 638-10 |
| Elongation at break | 12% | 6% | ASTM D 638-10 |
| Notched IZOD | 16 J/m | 25 J/m | ASTM D 638-10 |
| Green | Post-Cured | Method | |
|---|---|---|---|
| Ultimate tensile strength | 1.61 MPa | 3.23 MPa | ASTM D 412-06 |
| Elongation at break | 100% | 160% | ASTM D 412-06 |
| Tear strength | 8.9 kN/m | 19.1 kN/m | ASTM D 624-00 |
| Shore hardness | 40 A | 50 A | ASTM 2240 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertolini, M.; Rossoni, M.; Colombo, G. Operative Workflow from CT to 3D Printing of the Heart: Opportunities and Challenges. Bioengineering 2021, 8, 130. https://doi.org/10.3390/bioengineering8100130
Bertolini M, Rossoni M, Colombo G. Operative Workflow from CT to 3D Printing of the Heart: Opportunities and Challenges. Bioengineering. 2021; 8(10):130. https://doi.org/10.3390/bioengineering8100130
Chicago/Turabian StyleBertolini, Michele, Marco Rossoni, and Giorgio Colombo. 2021. "Operative Workflow from CT to 3D Printing of the Heart: Opportunities and Challenges" Bioengineering 8, no. 10: 130. https://doi.org/10.3390/bioengineering8100130
APA StyleBertolini, M., Rossoni, M., & Colombo, G. (2021). Operative Workflow from CT to 3D Printing of the Heart: Opportunities and Challenges. Bioengineering, 8(10), 130. https://doi.org/10.3390/bioengineering8100130






