Neurocognitive and Psychosocial Outcomes in Pediatric Brain Tumor Survivors
Abstract
1. Introduction
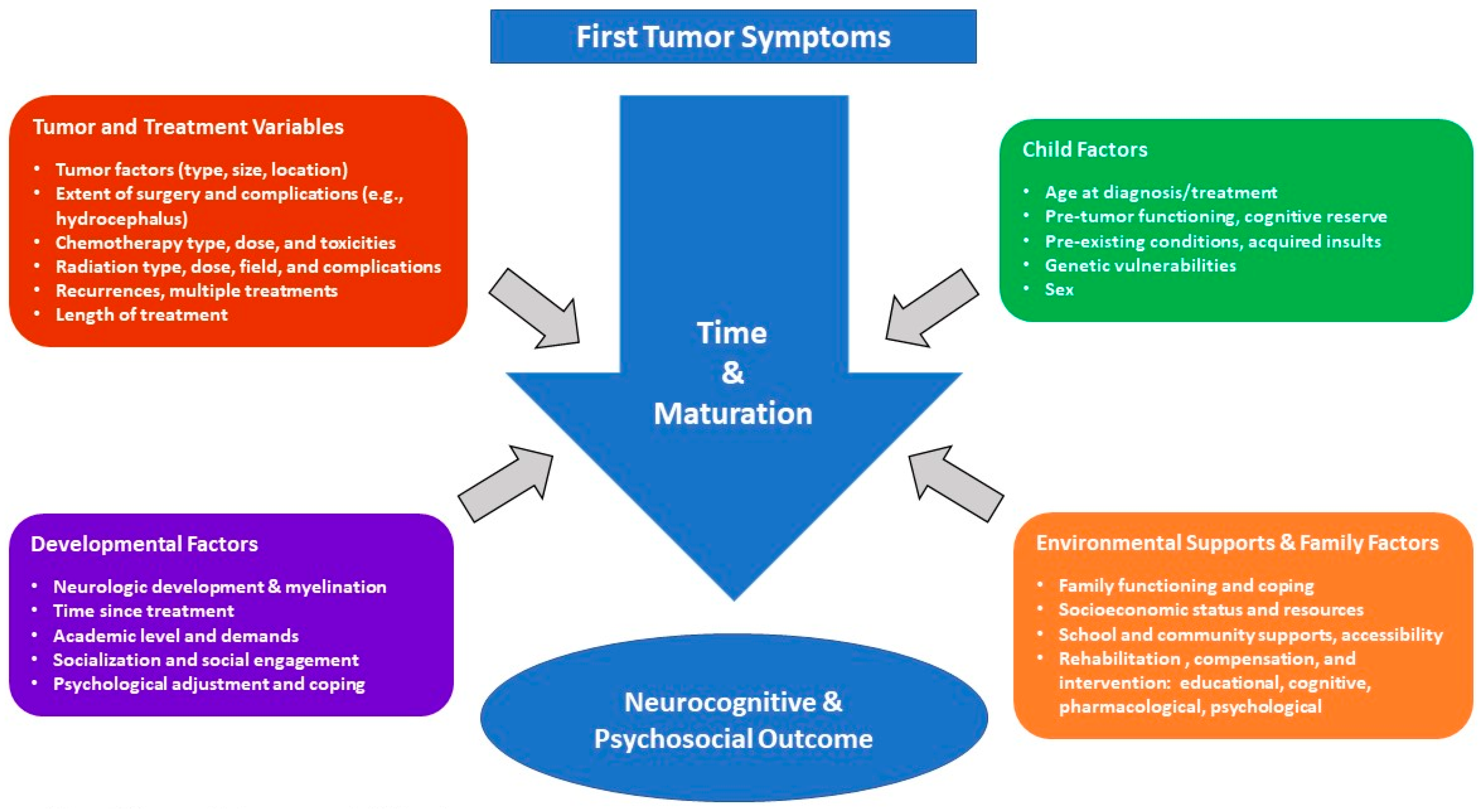
2. Factors Related to Expression of Late Effects
2.1. Tumor Variables
2.2. Treatment Variables
2.3. Individual Patient and Environmental Characteristics
3. Late Effects of PBT
3.1. Neurocognitive Outcomes
3.1.1. Intellectual Functioning
3.1.2. Core Deficits—Attention, Processing Speed, & Working Memory
3.1.3. Other Cognitive Functions
3.2. Psychosocial Outcomes
4. Interventions to Support PBT Survivors
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Olsson, I.T.; Perrin, S.; Lundgren, J.; Hjorth, L.; Johanson, A. Long-term cognitive sequelae after pediatric brain tumor related to medical risk factors, age, and sex. Pediatr. Neurol. 2014, 51, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.L.; Armstrong, C.; Onar-Thomas, A.; Wu, S.; Wallace, D.; Bonner, M.J.; Schreiber, J.; Swain, M.; Chapieski, L.; Mabbott, D.; et al. Processing speed, attention, and working memory after treatment for medulloblastoma: An international, prospective, and longitudinal study. J. Clin. Oncol. 2013, 31, 3494–3500. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, J.E.; Gurney, J.G.; Palmer, S.L.; Bass, J.K.; Wang, M.; Chen, S.; Zhang, H.; Swain, M.; Chapieski, M.L.; Bonner, M.J. Examination of risk factors for intellectual and academic outcomes following treatment for pediatric medulloblastoma. Neuro-Oncology 2014, 16, 1129–1136. [Google Scholar] [CrossRef] [PubMed]
- Di Rocco, F.; Jucá, C.E.; Zerah, M.; Sainte-Rose, C. Endoscopic third ventriculostomy and posterior fossa tumors. World Neurosurg. 2013, 79, S18.e15–S18.e19. [Google Scholar] [CrossRef] [PubMed]
- Hardy, K.K.; Bonner, M.J.; Willard, V.W.; Watral, M.A.; Gururangan, S. Hydrocephalus as a possible additional contributor to cognitive outcome in survivors of pediatric medulloblastoma. Psycho-Oncol. J. Psychol. Soc. Behav. Dimens. Cancer 2008, 17, 1157–1161. [Google Scholar] [CrossRef] [PubMed]
- Duffner, P.K. Risk factors for cognitive decline in children treated for brain tumors. Eur. J. Paediatr. Neurol. 2010, 14, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Mullins, W.; O’neil, S.; Wilson, K. Neuropsychological differences between survivors of supratentorial and infratentorial brain tumours. J. Intell. Disabil. Res. 2011, 55, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Beebe, D.W.; Ris, M.D.; Armstrong, F.D.; Fontanesi, J.; Mulhern, R.; Holmes, E.; Wisoff, J.H. Cognitive and adaptive outcome in low-grade pediatric cerebellar astrocytomas: Evidence of diminished cognitive and adaptive functioning in National Collaborative Research Studies (CCG 9891/POG 9130). J. Clin. Oncol. 2005, 23, 5198–5204. [Google Scholar] [CrossRef] [PubMed]
- Jalali, R.; Mallick, I.; Dutta, D.; Goswami, S.; Gupta, T.; Munshi, A.; Deshpande, D.; Sarin, R. Factors influencing neurocognitive outcomes in young patients with benign and low-grade brain tumors treated with stereotactic conformal radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2010, 77, 974–979. [Google Scholar] [CrossRef] [PubMed]
- Ris, M.D.; Beebe, D.W.; Armstrong, F.D.; Fontanesi, J.; Holmes, E.; Sanford, R.A.; Wisoff, J.H. Cognitive and adaptive outcome in extracerebellar low-grade brain tumors in children: A report from the Children’s Oncology Group. J. Clin. Oncol. 2008, 26, 4765. [Google Scholar] [CrossRef] [PubMed]
- Avula, S.; Spiteri, M.; Kumar, R.; Lewis, E.; Harave, S.; Windridge, D.; Ong, C.; Pizer, B. Post-operative pediatric cerebellar mutism syndrome and its association with hypertrophic olivary degeneration. Quant. Imaging Med. Surg. 2016, 6, 535. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, J.E.; Palmer, S.L.; Conklin, H.M.; Mabbott, D.J.; Swain, M.A.; Bonner, M.J.; Chapieski, M.L.; Huang, L.; Zhang, H.; Gajjar, A. Posterior fossa syndrome and long-term neuropsychological outcomes among children treated for medulloblastoma on a multi-institutional, prospective study. Neuro-Oncology 2017, 19, 1673–1682. [Google Scholar] [CrossRef] [PubMed]
- De Ruiter, M.A.; van Mourik, R.; Schouten-van Meeteren, A.Y.; Grootenhuis, M.A.; Oosterlaan, J. Neurocognitive consequences of a paediatric brain tumour and its treatment: A meta-analysis. Dev. Med. Child. Neurol. 2013, 55, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Spiegler, B.J.; Bouffet, E.; Greenberg, M.L.; Rutka, J.T.; Mabbott, D.J. Change in neurocognitive functioning after treatment with cranial radiation in childhood. J. Clin. Oncol. 2004, 22, 706–713. [Google Scholar] [CrossRef] [PubMed]
- Fouladi, M.; Chintagumpala, M.; Laningham, F.H.; Ashley, D.; Kellie, S.J.; Langston, J.W.; McCluggage, C.W.; Woo, S.; Kocak, M.; Krull, K. White matter lesions detected by magnetic resonance imaging after radiotherapy and high-dose chemotherapy in children with medulloblastoma or primitive neuroectodermal tumor. J. Clin. Oncol. 2004, 22, 4551–4560. [Google Scholar] [CrossRef] [PubMed]
- Jacola, L.M.; Ashford, J.M.; Reddick, W.E.; Glass, J.O.; Ogg, R.J.; Merchant, T.E.; Conklin, H.M. The relationship between working memory and cerebral white matter volume in survivors of childhood brain tumors treated with conformal radiation therapy. J. Neuro-Oncol. 2014, 119, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Reddick, W.E.; Glass, J.O.; Palmer, S.L.; Wu, S.; Gajjar, A.; Langston, J.W.; Kun, L.E.; Xiong, X.; Mulhern, R.K. Atypical white matter volume development in children following craniospinal irradiation. Neuro-Oncology 2005, 7, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Monje, M.L.; Vogel, H.; Masek, M.; Ligon, K.L.; Fisher, P.G.; Palmer, T.D. Impaired human hippocampal neurogenesis after treatment for central nervous system malignancies. Ann. Neurol. Off. J. Am. Neurol. Assoc. Child Neurol. Soc. 2007, 62, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Nagel, B.J.; Delis, D.C.; Palmer, S.L.; Reeves, C.; Gajjar, A.; Mulhern, R.K. Early patterns of verbal memory impairment in children treated for medulloblastoma. Neuropsychology 2006, 20, 105. [Google Scholar] [CrossRef] [PubMed]
- Law, N.; Bouffet, E.; Laughlin, S.; Laperriere, N.; Brière, M.-E.; Strother, D.; McConnell, D.; Hukin, J.; Fryer, C.; Rockel, C. Cerebello–thalamo–cerebral connections in pediatric brain tumor patients: Impact on working memory. Neuroimage 2011, 56, 2238–2248. [Google Scholar] [CrossRef] [PubMed]
- Taylor, O.A.; Hockenberry, M.J.; McCarthy, K.; Gundy, P.; Montgomery, D.; Ross, A.; Scheurer, M.E.; Moore, I.M. Evaluation of biomarkers of oxidative stress and apoptosis in patients with severe methotrexate neurotoxicity: A case series. J. Pediatr. Oncol. Nurs. 2015, 32, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Rutkowski, S.; Bode, U.; Deinlein, F.; Ottensmeier, H.; Warmuth-Metz, M.; Soerensen, N.; Graf, N.; Emser, A.; Pietsch, T.; Wolff, J.E. Treatment of early childhood medulloblastoma by postoperative chemotherapy alone. N. Engl. J. Med. 2005, 352, 978–986. [Google Scholar] [CrossRef] [PubMed]
- Verstappen, C.C.; Heimans, J.J.; Hoekman, K.; Postma, T.J. Neurotoxic complications of chemotherapy in patients with cancer. Drugs 2003, 63, 1549–1563. [Google Scholar] [CrossRef] [PubMed]
- McHaney, V.A.; Thibadoux, G.; Hayes, F.A.; Green, A.A. Hearing loss in children receiving cisplatin chemotherapy. J. Pediatr. 1983, 102, 314–317. [Google Scholar] [CrossRef]
- Warrier, R.; Chauhan, A.; Davluri, M.; Tedesco, S.L.; Nadell, J.; Craver, R. Cisplatin and cranial irradiation-related hearing loss in children. Ochsner J. 2012, 12, 191–196. [Google Scholar] [PubMed]
- Bull, K.S.; Spoudeas, H.A.; Yadegarfar, G.; Kennedy, C.R. Reduction of health status 7 years after addition of chemotherapy to craniospinal irradiation for medulloblastoma: A follow-up study in PNET 3 trial survivors—On behalf of the CCLG (formerly UKCCSG). J. Clin. Oncol. 2007, 25, 4239–4245. [Google Scholar] [CrossRef] [PubMed]
- Di Pinto, M.; Conklin, H.M.; Li, C.; Merchant, T.E. Learning and memory following conformal radiation therapy for pediatric craniopharyngioma and low-grade glioma. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, e363–e369. [Google Scholar] [CrossRef] [PubMed]
- Ris, M.D.; Packer, R.; Goldwein, J.; Jones-Wallace, D.; Boyett, J.M. Intellectual outcome after reduced-dose radiation therapy plus adjuvant chemotherapy for medulloblastoma: A Children’s Cancer Group study. J. Clin. Oncol. 2001, 19, 3470–3476. [Google Scholar] [CrossRef] [PubMed]
- Andreotti, C.; Root, J.C.; Ahles, T.A.; McEwen, B.S.; Compas, B.E. Cancer, coping, and cognition: A model for the role of stress reactivity in cancer-related cognitive decline. Psycho-Oncology 2015, 24, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Brouwers, P.; Okcu, M.F.; Cirino, P.T.; Krull, K.R. Sex-specific attention problems in long-term survivors of pediatric acute lymphoblastic leukemia. Cancer Interdiscip. Int. J. Am. Cancer Soc. 2009, 115, 4238–4245. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.L. Neurodevelopmental impact on children treated for medulloblastoma: A review and proposed conceptual model. Dev. Disabil. Res. Rev. 2008, 14, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.K.; Fernandez, N.; Dekel, N.; Turk, A.; Meier, A.; Ross, P.; Rosenthal, J. Socioeconomic status as a possible moderator of neurocognitive outcomes in children with cancer. Psycho-Oncology 2016, 25, 115–118. [Google Scholar] [CrossRef] [PubMed]
- Robinson, K.E.; Wolfe, K.R.; Yeates, K.O.; Mahone, E.M.; Cecil, K.M.; Ris, M.D. Predictors of adaptive functioning and psychosocial adjustment in children with pediatric brain tumor: A report from the brain radiation investigative study consortium. Pediatr. Blood Cancer 2015, 62, 509–516. [Google Scholar] [CrossRef] [PubMed]
- French, A.E.; Tsangaris, E.; Barrera, M.; Guger, S.; Brown, R.; Urbach, S.; Stephens, D.; Nathan, P.C. School attendance in childhood cancer survivors and their siblings. J. Pediatr. 2013, 162, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Butler, R.W.; Haser, J.K. Neurocognitive effects of treatment for childhood cancer. Ment. Retard. Dev. Disabil. Res. Rev. 2006, 12, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.L.; Reddick, W.E.; Gajjar, A. Understanding the cognitive impact on children who are treated for medulloblastoma. J. Pediatr. Psychol. 2007, 32, 1040–1049. [Google Scholar] [CrossRef] [PubMed]
- Mabbott, D.J.; Spiegler, B.J.; Greenberg, M.L.; Rutka, J.T.; Hyder, D.J.; Bouffet, E. Serial evaluation of academic and behavioral outcome after treatment with cranial radiation in childhood. J. Clin. Oncol. 2005, 23, 2256–2263. [Google Scholar] [CrossRef] [PubMed]
- Mabbott, D.J.; Penkman, L.; Witol, A.; Strother, D.; Bouffet, E. Core neurocognitive functions in children treated for posterior fossa tumors. Neuropsychology 2008, 22, 159. [Google Scholar] [CrossRef] [PubMed]
- Dennis, M. Childhood medical disorders and cognitive impairment: Biological risk, time, development, and reserve. In Pediatric Neuropsychology: Research, Theory, and Practice; Yeates, K.O., Ris, M.D., Taylor, H.G., Eds.; Guilford Press: New York, NY, USA, 2000; pp. 3–22. [Google Scholar]
- Baum, K.T.; Powell, S.K.; Jacobson, L.A.; Gragert, M.N.; Janzen, L.A.; Paltin, I.; Rey-Casserly, C.M.; Wilkening, G.N. Implementing guidelines: Proposed definitions of neuropsychology services in pediatric oncology. Pediatr. Blood Cancer 2017, 64, e26446. [Google Scholar] [CrossRef] [PubMed]
- Nathan, P.C.; Patel, S.K.; Dilley, K.; Goldsby, R.; Harvey, J.; Jacobsen, C.; Kadan-Lottick, N.; McKinley, K.; Millham, A.K.; Moore, I.; et al. Guidelines for identification of, advocacy for, and intervention in neurocognitive problems in survivors of childhood cancer: A report from the Children’s Oncology Group. Arch. Pediatr. Adolesc. Med. 2007, 161, 798–806. [Google Scholar] [CrossRef] [PubMed]
- Annett, R.D.; Patel, S.K.; Phipps, S. Monitoring and Assessment of Neuropsychological Outcomes as a Standard of Care in Pediatric Oncology. Pediatr. Blood Cancer 2015, 62 (Suppl. 5), S460–S513. [Google Scholar] [CrossRef] [PubMed]
- Bledsoe, J.C. Effects of Cranial Radiation on Structural and Functional Brain Development in Pediatric Brain Tumors. J. Pediatr. Neuropsychol. 2016, 2, 3–13. [Google Scholar] [CrossRef]
- Grill, J.; Renaux, V.K.; Bulteau, C.; Viguier, D.; Levy-Piebois, C.; Sainte-Rose, C.; Dellatolas, G.; Raquin, M.A.; Jambaque, I.; Kalifa, C. Long-term intellectual outcome in children with posterior fossa tumors according to radiation doses and volumes. Int. J. Radiat. Oncol. Biol. Phys. 1999, 45, 137–145. [Google Scholar] [CrossRef]
- Moxon-Emre, I.; Bouffet, E.; Taylor, M.D.; Laperriere, N.; Scantlebury, N.; Law, N.; Spiegler, B.J.; Malkin, D.; Janzen, L.; Mabbott, D. Impact of craniospinal dose, boost volume, and neurologic complications on intellectual outcome in patients with medulloblastoma. J. Clin. Oncol. 2014, 32, 1760–1768. [Google Scholar] [CrossRef] [PubMed]
- Chevignard, M.; Câmara-Costa, H.; Doz, F.; Dellatolas, G. Core deficits and quality of survival after childhood medulloblastoma: A review. Neuro-Oncol. Pract. 2017, 4, 82–97. [Google Scholar] [CrossRef]
- Brinkman, T.M.; Reddick, W.E.; Luxton, J.; Glass, J.O.; Sabin, N.D.; Srivastava, D.K.; Robison, L.L.; Hudson, M.M.; Krull, K.R. Cerebral white matter integrity and executive function in adult survivors of childhood medulloblastoma. Neuro-Oncology 2012, 14 (Suppl. 4), iv25–iv36. [Google Scholar] [CrossRef] [PubMed]
- Robinson, K.E.; Kuttesch, J.F.; Champion, J.E.; Andreotti, C.F.; Hipp, D.W.; Bettis, A.; Barnwell, A.; Compas, B.E. A quantitative meta-analysis of neurocognitive sequelae in survivors of pediatric brain tumors. Pediatr. Blood Cancer 2010, 55, 525–531. [Google Scholar] [CrossRef] [PubMed]
- King, T.Z.; Ailion, A.S.; Fox, M.E.; Hufstetler, S.M. Neurodevelopmental model of long-term outcomes of adult survivors of childhood brain tumors. Child Neuropsychol. 2017, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Robinson, K.E.; Fraley, C.E.; Pearson, M.M.; Kuttesch, J.F., Jr.; Compas, B.E. Neurocognitive late effects of pediatric brain tumors of the posterior fossa: A quantitative review. J. Int. Neuropsychol. Soc. 2013, 19, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.L.; Goloubeva, O.; Reddick, W.E.; Glass, J.O.; Gajjar, A.; Kun, L.; Merchant, T.E.; Mulhern, R.K. Patterns of intellectual development among survivors of pediatric medulloblastoma: A longitudinal analysis. J. Clin. Oncol. 2001, 19, 2302–2308. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.L.; Gajjar, A.; Reddick, W.E.; Glass, J.O.; Kun, L.E.; Wu, S.; Xiong, X.; Mulhern, R.K. Predicting intellectual outcome among children treated with 35–40 Gy craniospinal irradiation for medulloblastoma. Neuropsychology 2003, 17, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Radcliffe, J.; Bunin, G.R.; Sutton, L.N.; Goldwein, J.W.; Phillips, P.C. Cognitive deficits in long-term survivors of childhood medulloblastoma and other noncortical tumors: Age-dependent effects of whole brain radiation. Int. J. Dev. Neurosci. 1994, 12, 327–334. [Google Scholar] [CrossRef]
- Kieffer-Renaux, V.; Bulteau, C.; Grill, J.; Kalifa, C.; Viguier, D.; Jambaque, I. Patterns of neuropsychological deficits in children with medulloblastoma according to craniospatial irradiation doses. Dev. Med. Child Neurol. 2000, 42, 741–745. [Google Scholar] [CrossRef] [PubMed]
- Mulhern, R.K.; Kepner, J.L.; Thomas, P.R.; Armstrong, F.D.; Friedman, H.S.; Kun, L.E. Neuropsychologic functioning of survivors of childhood medulloblastoma randomized to receive conventional or reduced-dose craniospinal irradiation: A Pediatric Oncology Group study. J. Clin. Oncol. 1998, 16, 1723–1728. [Google Scholar] [CrossRef] [PubMed]
- Wilson, V.C.; McDonough, J.; Tochner, Z. Proton beam irradiation in pediatric oncology: An overview. J. Pediatr. Hematol. Oncol. 2005, 27, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Warren, E.; Child, A.; Cirino, P.; Grosshans, D.; Mahajan, A.; Paulino, A.; Chintagumpala, M.; Okcu, F.; Douglas Ris, M.; Minard, C. QOL-42. Better social, cognitive, and academic outcomes among pediatric brain tumor survivors treated with proton versus photon radiation therapy. Neuro-Oncology 2018, 20, i166. [Google Scholar] [CrossRef]
- Kahalley, L.S.; Ris, M.D.; Grosshans, D.R.; Okcu, M.F.; Paulino, A.C.; Chintagumpala, M.; Moore, B.D.; Guffey, D.; Minard, C.G.; Stancel, H.H.; et al. Comparing Intelligence Quotient Change After Treatment With Proton Versus Photon Radiation Therapy for Pediatric Brain Tumors. J. Clin. Oncol. 2016, 34, 1043–1049. [Google Scholar] [CrossRef] [PubMed]
- Antonini, T.N.; Ris, M.D.; Grosshans, D.R.; Mahajan, A.; Okcu, M.F.; Chintagumpala, M.; Paulino, A.; Child, A.E.; Orobio, J.; Stancel, H.H.; et al. Attention, processing speed, and executive functioning in pediatric brain tumor survivors treated with proton beam radiation therapy. Radiother. Oncol. 2017, 124, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Conklin, H.M.; Ashford, J.M.; Howarth, R.A.; Merchant, T.E.; Ogg, R.J.; Santana, V.M.; Reddick, W.E.; Wu, S.; Xiong, X. Working memory performance among childhood brain tumor survivors. J. Int. Neuropsychol. Soc. 2012, 18, 996–1005. [Google Scholar] [CrossRef] [PubMed]
- Reddick, W.E.; White, H.A.; Glass, J.O.; Wheeler, G.C.; Thompson, S.J.; Gajjar, A.; Leigh, L.; Mulhern, R.K. Developmental model relating white matter volume to neurocognitive deficits in pediatric brain tumor survivors. Cancer 2003, 97, 2512–2519. [Google Scholar] [CrossRef] [PubMed]
- Kahalley, L.S.; Conklin, H.M.; Tyc, V.L.; Hudson, M.M.; Wilson, S.J.; Wu, S.; Xiong, X.; Hinds, P.S. Slower processing speed after treatment for pediatric brain tumor and acute lymphoblastic leukemia. Psychooncology 2013, 22, 1979–1986. [Google Scholar] [CrossRef] [PubMed]
- Schatz, J.; Kramer, J.H.; Ablin, A.; Matthay, K.K. Processing speed, working memory, and IQ: A developmental model of cognitive deficits following cranial radiation therapy. Neuropsychology 2000, 14, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Fry, A.F.; Hale, S. Relationships among processing speed, working memory, and fluid intelligence in children. Biol. Psychol. 2000, 54, 1–34. [Google Scholar] [CrossRef]
- Holland, A.A.; Hughes, C.W.; Stavinoha, P.L. School competence and fluent academic performance: Informing assessment of educational outcomes in survivors of pediatric medulloblastoma. Appl. Neuropsychol. Child 2015, 4, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Roddy, E.; Mueller, S. Late effects of treatment of pediatric central nervous system tumors. J. Child Neurol. 2016, 31, 237–254. [Google Scholar] [CrossRef] [PubMed]
- Starowicz-Filip, A.; Chrobak, A.A.; Milczarek, O.; Kwiatkowski, S. The visuospatial functions in children after cerebellar low-grade astrocytoma surgery: A contribution to the pediatric neuropsychology of the cerebellum. J. Neuropsychol. 2017, 11, 201–221. [Google Scholar] [CrossRef] [PubMed]
- Koustenis, E.; Driever, P.H.; de Sonneville, L.; Rueckriegel, S.M. Executive function deficits in pediatric cerebellar tumor survivors. Eur. J. Paediatr. Neurol. 2016, 20, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Robison, L.L.; Hudson, M.M. Survivors of childhood and adolescent cancer: Life-long risks and responsibilities. Nat. Rev. Cancer 2014, 14, 61. [Google Scholar] [CrossRef] [PubMed]
- Wochos, G.; Semerjian, C.; Walsh, K.S. Differences in parent and teacher rating of everyday executive function in pediatric brain tumor survivors. Clin. Neuropsychol. 2014, 28, 1243–1257. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, K.R.; Walsh, K.S.; Reynolds, N.C.; Mitchell, F.; Reddy, A.T.; Paltin, I.; Madan-Swain, A. Executive functions and social skills in survivors of pediatric brain tumor. Child Neuropsychol. 2013, 19, 370–384. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, G.T.; Liu, Q.; Yasui, Y.; Huang, S.; Ness, K.K.; Leisenring, W.; Hudson, M.M.; Donaldson, S.S.; King, A.A.; Stovall, M.; et al. Long-term outcomes among adult survivors of childhood central nervous system malignancies in the Childhood Cancer Survivor Study. J. Natl. Cancer Inst. 2009, 101, 946–958. [Google Scholar] [CrossRef] [PubMed]
- Ribi, K.; Relly, C.; Landolt, M.A.; Alber, F.D.; Boltshauser, E.; Grotzer, M.A. Outcome of medulloblastoma in children: Long-term complications and quality of life. Neuropediatrics 2005, 36, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Ronning, C.; Sundet, K.; Due-Tonnessen, B.; Lundar, T.; Helseth, E. Persistent cognitive dysfunction secondary to cerebellar injury in patients treated for posterior fossa tumors in childhood. Pediatr. Neurosurg. 2005, 41, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Roncadin, C.; Dennis, M.; Greenberg, M.L.; Spiegler, B.J. Adverse medical events associated with childhood cerebellar astrocytomas and medulloblastomas: Natural history and relation to very long-term neurobehavioral outcome. Childs Nerv. Syst. 2008, 24, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- Maddrey, A.M.; Bergeron, J.A.; Lombardo, E.R.; McDonald, N.K.; Mulne, A.F.; Barenberg, P.D.; Bowers, D.C. Neuropsychological performance and quality of life of 10 year survivors of childhood medulloblastoma. J. Neurooncol. 2005, 72, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Edelstein, K.; Spiegler, B.J.; Fung, S.; Panzarella, T.; Mabbott, D.J.; Jewitt, N.; D’Agostino, N.M.; Mason, W.P.; Bouffet, E.; Tabori, U.; et al. Early aging in adult survivors of childhood medulloblastoma: Long-term neurocognitive, functional, and physical outcomes. Neuro-Oncology 2011, 13, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Mulhern, R.K.; Palmer, S.L.; Reddick, W.E.; Glass, J.O.; Kun, L.E.; Taylor, J.; Langston, J.; Gajjar, A. Risks of young age for selected neurocognitive deficits in medulloblastoma are associated with white matter loss. J. Clin. Oncol. 2001, 19, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Hocking, M.C.; Hobbie, W.L.; Deatrick, J.A.; Hardie, T.L.; Barakat, L.P. Family functioning mediates the association between neurocognitive functioning and health-related quality of life in young adult survivors of childhood brain tumors. J. Adolesc. Young Adult Oncol. 2015, 4, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Redmond, K.J.; Mahone, E.M.; Terezakis, S.; Ishaq, O.; Ford, E.; McNutt, T.; Kleinberg, L.; Cohen, K.J.; Wharam, M.; Horska, A. Association between radiation dose to neuronal progenitor cell niches and temporal lobes and performance on neuropsychological testing in children: A prospective study. Neuro-Oncology 2013, 15, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Huber, J.F.; Bradley, K.; Spiegler, B.; Dennis, M. Long-term neuromotor speech deficits in survivors of childhood posterior fossa tumors: Effects of tumor type, radiation, age at diagnosis, and survival years. J. Child Neurol. 2007, 22, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Catsman-Berrevoets, C.E.; Aarsen, F.K. The spectrum of neurobehavioural deficits in the Posterior Fossa Syndrome in children after cerebellar tumour surgery. Cortex 2010, 46, 933–946. [Google Scholar] [CrossRef] [PubMed]
- Hua, C.; Bass, J.K.; Khan, R.; Kun, L.E.; Merchant, T.E. Hearing loss after radiotherapy for pediatric brain tumors: Effect of cochlear dose. Int. J. Radiat. Oncol. Biol. Phys. 2008, 72, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.S.; Dellarole, A.; Peterson, E.C.; Bregy, A.; Komotar, R.; Harvey, P.D.; Elhammady, M.S. Long-term psychiatric outcomes in pediatric brain tumor survivors. Child’s Nerv. Syst. 2015, 31, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Memmesheimer, R.M.; Lange, K.; Dölle, M.; Heger, S.; Mueller, I. Psychological well-being and independent living of young adults with childhood-onset craniopharyngioma. Dev. Med. Child Neurol. 2017, 59, 829–836. [Google Scholar] [CrossRef] [PubMed]
- Quast, L.F.; Phillips, P.C.; Li, Y.; Kazak, A.E.; Barakat, L.P.; Hocking, M.C. A prospective study of family predictors of health-related quality of life in pediatric brain tumor survivors. Pediatr. Blood Cancer 2018, 65, e26976. [Google Scholar] [CrossRef] [PubMed]
- Adduci, A.; Jankovic, M.; Strazzer, S.; Massimino, M.; Clerici, C.; Poggi, G. Parent–child communication and psychological adjustment in children with a brain tumor. Pediatr. Blood Cancer 2012, 59, 290–294. [Google Scholar] [CrossRef] [PubMed]
- De Ruiter, M.A.; Schouten-van Meeteren, A.Y.N.; van Vuurden, D.G.; Maurice-Stam, H.; Gidding, C.; Beek, L.R.; Granzen, B.; Oosterlaan, J.; Grootenhuis, M.A. Psychosocial profile of pediatric brain tumor survivors with neurocognitive complaints. Qual. Life Res. 2016, 25, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Fuemmeler, B.F.; Elkin, T.D.; Mullins, L.L. Survivors of childhood brain tumors: Behavioral, emotional, and social adjustment. Clin. Psychol. Rev. 2002, 22, 547–585. [Google Scholar] [CrossRef]
- Emond, A.; Edwards, L.; Peacock, S.; Norman, C.; Evangeli, M. Social competence in children and young people treated for a brain tumour. Support. Care Cancer 2016, 24, 4587–4595. [Google Scholar] [CrossRef] [PubMed]
- Schulte, F. Social competence in pediatric brain tumor survivors: Breadth versus depth. Curr. Opin. Oncol. 2015, 27, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Holland, A.A.; Colaluca, B.; Bailey, L.; Stavinoha, P.L. Impact of attention on social functioning in pediatric medulloblastoma survivors. Pediatr. Hematol. Oncol. 2018, 35, 76–89. [Google Scholar] [CrossRef] [PubMed]
- Sands, S.A.; Pasichow, K.P. Psychological and social impact of being a pediatric brain tumor survivor. In Late Effects of Treatment for Brain Tumors; Springer: New York, NY, USA, 2009; pp. 297–307. [Google Scholar]
- Nassau, J.H.; Drotar, D. Social competence among children with central nervous system-related chronic health conditions: A review. J. Pediatr. Psychol. 1997, 22, 771–793. [Google Scholar] [CrossRef] [PubMed]
- Poggi, G.; Liscio, M.; Galbiati, S.; Adduci, A.; Massimino, M.; Gandola, L.; Spreafico, F.; Clerici, C.A.; Fossati-Bellani, F.; Sommovigo, M. Brain tumors in children and adolescents: Cognitive and psychological disorders at different ages. Psycho-Oncol. J. Psychol. Soc. Behav. Dimens. Cancer 2005, 14, 386–395. [Google Scholar] [CrossRef] [PubMed]
- Kahalley, L.S.; Wilson, S.J.; Tyc, V.L.; Conklin, H.M.; Hudson, M.M.; Wu, S.; Xiong, X.; Stancel, H.H.; Hinds, P.S. Are the psychological needs of adolescent survivors of pediatric cancer adequately identified and treated? Psycho-Oncology 2013, 22, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Ventura, L.M.; Grieco, J.A.; Evans, C.L.; Kuhlthau, K.A.; MacDonald, S.M.; Tarbell, N.J.; Yock, T.I.; Pulsifer, M.B. Executive functioning, academic skills, and quality of life in pediatric patients with brain tumors post-proton radiation therapy. J. Neuro-Oncol. 2018, 137, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Marusak, H.A.; Iadipaolo, A.S.; Harper, F.W.; Elrahal, F.; Taub, J.W.; Goldberg, E.; Rabinak, C.A. Neurodevelopmental consequences of pediatric cancer and its treatment: Applying an early adversity framework to understanding cognitive, behavioral, and emotional outcomes. Neuropsychol. Rev. 2017, 28, 123–175. [Google Scholar] [CrossRef] [PubMed]
- Hay, G.H.; Nabors, M.; Sullivan, A.; Zygmund, A. Students with Pediatric Cancer: A Prescription for School Success. Phys. Disabil. Educ. Relat. Serv. 2015, 34, 1–13. [Google Scholar] [CrossRef]
- Mitby, P.A.; Robison, L.L.; Whitton, J.A.; Zevon, M.A.; Gibbs, I.C.; Tersak, J.M.; Meadows, A.T.; Stovall, M.; Zeltzer, L.K.; Mertens, A.C.; et al. Utilization of special education services and educational attainment among long-term survivors of childhood cancer: A report from the Childhood Cancer Survivor Study. Cancer 2003, 97, 1115–1126. [Google Scholar] [CrossRef] [PubMed]
- Kirchhoff, A.C.; Leisenring, W.; Krull, K.R.; Ness, K.K.; Friedman, D.L.; Armstrong, G.T.; Stovall, M.; Park, E.R.; Oeffinger, K.C.; Hudson, M.M.; et al. Unemployment among adult survivors of childhood cancer: A report from the childhood cancer survivor study. Med. Care 2010, 48, 1015–1025. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.L.; Christiansen, H.L.; Elam, M.; Hoag, J.; Irwin, M.K.; Pao, M.; Voll, M.; Noll, R.B.; Kelly, K.P. Academic Continuity and School Reentry Support as a Standard of Care in Pediatric Oncology. Pediatr. Blood Cancer 2015, 62 (Suppl. 5), S805–S817. [Google Scholar] [CrossRef] [PubMed]
- Olson, K.; Sands, S.A. Cognitive training programs for childhood cancer patients and survivors: A critical review and future directions. Child Neuropsychol. 2016, 22, 509–536. [Google Scholar] [CrossRef] [PubMed]
- Butler, R.W.; Copeland, D.R. Attentional processes and their remediation in children treated for cancer: A literature review and the development of a therapeutic approach. J. Int. Neuropsychol. Soc. 2002, 8, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Butler, R.W.; Copeland, D.R.; Fairclough, D.L.; Mulhern, R.K.; Katz, E.R.; Kazak, A.E.; Noll, R.B.; Patel, S.K.; Sahler, O.J.Z. A multicenter, randomized clinical trial of a cognitive remediation program for childhood survivors of a pediatric malignancy. J. Consult. Clin. Psychol. 2008, 76, 367. [Google Scholar] [CrossRef] [PubMed]
- Pearson Education, Incorporated Cogmed. Available online: https://www.cogmed.com/ (accessed on 20 July 2018).
- Hardy, K.K.; Willard, V.W.; Allen, T.M.; Bonner, M.J. Working memory training in survivors of pediatric cancer: A randomized pilot study. Psycho-Oncology 2013, 22, 1856–1865. [Google Scholar] [CrossRef] [PubMed]
- Conklin, H.M.; Ogg, R.J.; Ashford, J.M.; Scoggins, M.A.; Zou, P.; Clark, K.N.; Martin-Elbahesh, K.; Hardy, K.K.; Merchant, T.E.; Jeha, S. Computerized cognitive training for amelioration of cognitive late effects among childhood cancer survivors: A randomized controlled trial. J. Clin. Oncol. 2015, 33, 3894. [Google Scholar] [CrossRef] [PubMed]
- Conklin, H.M.; Ashford, J.M.; Clark, K.N.; Martin-Elbahesh, K.; Hardy, K.K.; Merchant, T.E.; Ogg, R.J.; Jeha, S.; Huang, L.; Zhang, H. Long-term efficacy of computerized cognitive training among survivors of childhood cancer: A single-blind randomized controlled trial. J. Pediatr. Psychol. 2016, 42, 220–231. [Google Scholar] [CrossRef] [PubMed]
- Chacko, A.; Bedard, A.; Marks, D.; Feirsen, N.; Uderman, J.; Chimiklis, A.; Rajwan, E.; Cornwell, M.; Anderson, L.; Zwilling, A. A randomized clinical trial of Cogmed working memory training in school-age children with ADHD: A replication in a diverse sample using a control condition. J. Child Psychol. Psychiatry 2014, 55, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.K.; Ross, P.; Cuevas, M.; Turk, A.; Kim, H.; Lo, T.T.; Wong, L.F.; Bhatia, S. Parent-directed intervention for children with cancer-related neurobehavioral late effects: A randomized pilot study. J. Pediatr. Psychol. 2014, 39, 1013–1027. [Google Scholar] [CrossRef] [PubMed]
- Holland, A.A.; Hughes, C.W.; Harder, L.; Silver, C.; Bowers, D.C.; Stavinoha, P.L. Effect of motivation on academic fluency performance in survivors of pediatric medulloblastoma. Child Neuropsychol. 2016, 22, 570–586. [Google Scholar] [CrossRef] [PubMed]
- Riggs, L.; Piscione, J.; Laughlin, S.; Cunningham, T.; Timmons, B.W.; Courneya, K.S.; Bartels, U.; Skocic, J.; de Medeiros, C.; Liu, F. Exercise training for neural recovery in a restricted sample of pediatric brain tumor survivors: A controlled clinical trial with crossover of training versus no training. Neuro-Oncology 2016, 19, 440–450. [Google Scholar] [CrossRef] [PubMed]
- Conklin, H.M.; Reddick, W.E.; Ashford, J.; Ogg, S.; Howard, S.C.; Morris, E.B.; Brown, R.; Bonner, M.; Christensen, R.; Wu, S. Long-term efficacy of methylphenidate in enhancing attention regulation, social skills, and academic abilities of childhood cancer survivors. J. Clin. Oncol. 2010, 28, 4465. [Google Scholar] [CrossRef] [PubMed]
- Castellino, S.M.; Tooze, J.A.; Flowers, L.; Hill, D.F.; McMullen, K.P.; Shaw, E.G.; Parsons, S.K. Toxicity and efficacy of the acetylcholinesterase (AChe) inhibitor donepezil in childhood brain tumor survivors: A pilot study. Pediatr. Blood Cancer 2012, 59, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Boele, F.W.; Douw, L.; de Groot, M.; van Thuijl, H.F.; Cleijne, W.; Heimans, J.J.; Taphoorn, M.J.; Reijneveld, J.C.; Klein, M. The effect of modafinil on fatigue, cognitive functioning, and mood in primary brain tumor patients: A multicenter randomized controlled trial. Neuro-Oncology 2013, 15, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Brown, P.D.; Pugh, S.; Laack, N.N.; Wefel, J.S.; Khuntia, D.; Meyers, C.; Choucair, A.; Fox, S.; Suh, J.H.; Roberge, D. Memantine for the prevention of cognitive dysfunction in patients receiving whole-brain radiotherapy: A randomized, double-blind, placebo-controlled trial. Neuro-Oncology 2013, 15, 1429–1437. [Google Scholar] [CrossRef] [PubMed]
- Poggi, G.; Liscio, M.; Pastore, V.; Adduci, A.; Galbiati, S.; Spreafico, F.; Gandola, L.; Massimino, M. Psychological intervention in young brain tumor survivors: The efficacy of the cognitive behavioural approach. Disabil. Rehabil. 2009, 31, 1066–1073. [Google Scholar] [CrossRef] [PubMed]
- Barrera, M.; Atenafu, E.G.; Sung, L.; Bartels, U.; Schulte, F.; Chung, J.; Cataudella, D.; Hancock, K.; Janzen, L.; Saleh, A. A randomized control intervention trial to improve social skills and quality of life in pediatric brain tumor survivors. Psycho-Oncology 2018, 27, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.C.; Enderby, K.; O’Toole, M.; Thomas, S.A.; Ashley, D.; Rosenfeld, J.V.; Simos, E.; Tokatlian, N.; Gedye, R. The role of social support in families coping with childhood brain tumor. J. Psychosoc. Oncol. 2009, 27, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Macartney, G.; Stacey, D.; Harrison, M.B.; VanDenKerkhof, E. Symptoms, coping, and quality of life in pediatric brain tumor survivors: A qualitative study. Oncol. Nurs. Forum 2014, 41, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Ris, M.D.; Grosch, M.; Fletcher, J.M.; Metah, P.; Kahalley, L.S. Measurement of neurodevelopmental changes in children treated with radiation for brain tumors: What is a true ‘baseline?’. Clin. Neuropsychol. 2017, 31, 307–328. [Google Scholar] [CrossRef] [PubMed]
- Kesler, S.R.; Gugel, M.; Huston-Warren, E.; Watson, C. Atypical structural connectome organization and cognitive impairment in young survivors of acute lymphoblastic leukemia. Brain Connect. 2016, 6, 273–282. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stavinoha, P.L.; Askins, M.A.; Powell, S.K.; Pillay Smiley, N.; Robert, R.S. Neurocognitive and Psychosocial Outcomes in Pediatric Brain Tumor Survivors. Bioengineering 2018, 5, 73. https://doi.org/10.3390/bioengineering5030073
Stavinoha PL, Askins MA, Powell SK, Pillay Smiley N, Robert RS. Neurocognitive and Psychosocial Outcomes in Pediatric Brain Tumor Survivors. Bioengineering. 2018; 5(3):73. https://doi.org/10.3390/bioengineering5030073
Chicago/Turabian StyleStavinoha, Peter L., Martha A. Askins, Stephanie K. Powell, Natasha Pillay Smiley, and Rhonda S. Robert. 2018. "Neurocognitive and Psychosocial Outcomes in Pediatric Brain Tumor Survivors" Bioengineering 5, no. 3: 73. https://doi.org/10.3390/bioengineering5030073
APA StyleStavinoha, P. L., Askins, M. A., Powell, S. K., Pillay Smiley, N., & Robert, R. S. (2018). Neurocognitive and Psychosocial Outcomes in Pediatric Brain Tumor Survivors. Bioengineering, 5(3), 73. https://doi.org/10.3390/bioengineering5030073



