Stem Cells and Engineered Scaffolds for Regenerative Wound Healing
Abstract
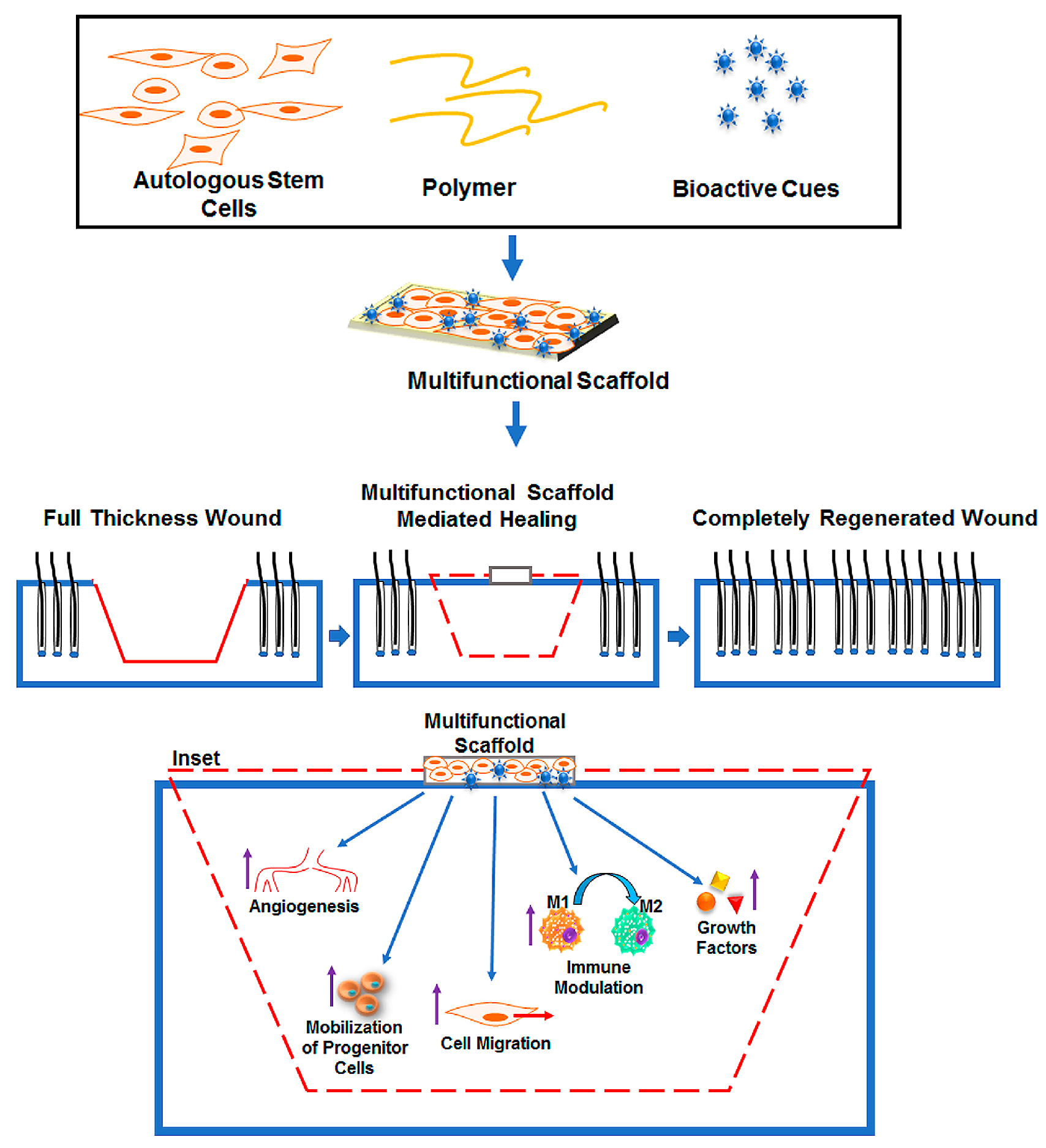
1. Introduction
2. Stem Cells for Regenerative Wound Healing
3. Polymer-Based Biomaterials for Stem Cell Delivery for Regenerative Wound Healing
3.1. Natural Polymer-Based Biomaterials
3.2. Synthetic Polymer-Based Biomaterials
4. Modifications to Biomaterials
5. Conclusions and Future Directions
Acknowledgments
Conflicts of Interest
References
- Atiyeh, B.S.; Ioannovich, J.; Al-Amm, C.A.; El-Musa, K.A. Management of acute and chronic open wounds: The importance of moist environment in optimal wound healing. Curr. Pharm. Biotechnol. 2002, 3, 179–195. [Google Scholar] [CrossRef] [PubMed]
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci. Transl. Med. 2014, 6, 265sr266. [Google Scholar] [CrossRef] [PubMed]
- Sorg, H.; Tilkorn, D.J.; Hager, S.; Hauser, J.; Mirastschijski, U. Skin wound healing: An update on the current knowledge and concepts. Eur. Surg. Res. 2017, 58, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Werner, S.; Grose, R. Regulation of wound healing by growth factors and cytokines. Physiol. Rev. 2003, 83, 835–870. [Google Scholar] [CrossRef] [PubMed]
- Qing, C. The molecular biology in wound healing & non-healing wound. Chin. J. Traumatol. Zhonghua Chuang Shang Za Zhi 2017, 20, 189–193. [Google Scholar] [PubMed]
- Demidova-Rice, T.N.; Hamblin, M.R.; Herman, I.M. Acute and impaired wound healing: Pathophysiology and current methods for drug delivery, part 2: Role of growth factors in normal and pathological wound healing: Therapeutic potential and methods of delivery. Adv. Skin Wound Care 2012, 25, 349–370. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.J.; Badylak, S.F. The use of biologic scaffolds in the treatment of chronic nonhealing wounds. Adv. Wound Care 2015, 4, 490–500. [Google Scholar] [CrossRef] [PubMed]
- Ojeh, N.; Pastar, I.; Tomic-Canic, M.; Stojadinovic, O. Stem cells in skin regeneration, wound healing, and their clinical applications. Int. J. Mol. Sci. 2015, 16, 25476–25501. [Google Scholar] [CrossRef] [PubMed]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.K.; Gordillo, G.M.; Roy, S.; Kirsner, R.; Lambert, L.; Hunt, T.K.; Gottrup, F.; Gurtner, G.C.; Longaker, M.T. Human skin wounds: A major and snowballing threat to public health and the economy. Wound Repair Regen. 2009, 17, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, L.E.; Gerecht, S. Engineered biopolymeric scaffolds for chronic wound healing. Front. Physiol. 2016, 7, 341. [Google Scholar] [CrossRef] [PubMed]
- Duscher, D.; Barrera, J.; Wong, V.W.; Maan, Z.N.; Whittam, A.J.; Januszyk, M.; Gurtner, G.C. Stem cells in wound healing: The future of regenerative medicine? A mini-review. Gerontology 2016, 62, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Rosemann, A. Why regenerative stem cell medicine progresses slower than expected. J. Cell. Biochem. 2014, 115, 2073–2076. [Google Scholar] [CrossRef] [PubMed]
- Kirby, G.T.; Mills, S.J.; Cowin, A.J.; Smith, L.E. Stem cells for cutaneous wound healing. BioMed Res. Int. 2015, 2015, 285869. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, M.T.; Pirraco, R.P.; Marques, A.P. Stem cells in skin wound healing: Are we there yet? Adv. Wound Care 2016, 5, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Major, M.R.; Wong, V.W.; Nelson, E.R.; Longaker, M.T.; Gurtner, G.C. The foreign body response: At the interface of surgery and bioengineering. Plast. Reconstr. Surg. 2015, 135, 1489–1498. [Google Scholar] [CrossRef] [PubMed]
- Morais, J.M.; Papadimitrakopoulos, F.; Burgess, D.J. Biomaterials/tissue interactions: Possible solutions to overcome foreign body response. AAPS J. 2010, 12, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Baraniak, P.R.; McDevitt, T.C. Stem cell paracrine actions and tissue regeneration. Regen. Med. 2010, 5, 121–143. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Ding, Y.; Zhang, Y.; Tse, H.F.; Lian, Q. Paracrine mechanisms of mesenchymal stem cell-based therapy: Current status and perspectives. Cell Transplant. 2014, 23, 1045–1059. [Google Scholar] [CrossRef] [PubMed]
- Quan, R.; Zheng, X.; Xu, S.; Zhang, L.; Yang, D. Gelatin-chondroitin-6-sulfate-hyaluronic acid scaffold seeded with vascular endothelial growth factor 165 modified hair follicle stem cells as a three-dimensional skin substitute. Stem Cell Res. Ther. 2014, 5, 118. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Dai, L.; Li, X.; Liang, R.; Guan, G.; Zhang, Z.; Cao, W.; Liu, Z.; Mei, S.; Liang, W.; et al. Epidermal stem cells cultured on collagen-modified chitin membrane induce in situ tissue regeneration of full-thickness skin defects in mice. PLoS ONE 2014, 9, e87557. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, H.; Keshel, S.H.; Chari, A.J.; Biazar, E. Human unrestricted somatic stem cells loaded in nanofibrous pcl scaffold and their healing effect on skin defects. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1556–1560. [Google Scholar] [CrossRef] [PubMed]
- Metallo, C.M.; Azarin, S.M.; Moses, L.E.; Ji, L.; de Pablo, J.J.; Palecek, S.P. Human embryonic stem cell-derived keratinocytes exhibit an epidermal transcription program and undergo epithelial morphogenesis in engineered tissue constructs. Tissue Eng. Part A 2010, 16, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Cherbuin, T.; Movahednia, M.M.; Toh, W.S.; Cao, T. Investigation of human embryonic stem cell-derived keratinocytes as an in vitro research model for mechanical stress dynamic response. Stem Cell Rev. 2015, 11, 460–473. [Google Scholar] [CrossRef] [PubMed]
- Guenou, H.; Nissan, X.; Larcher, F.; Feteira, J.; Lemaitre, G.; Saidani, M.; Del Rio, M.; Barrault, C.C.; Bernard, F.X.; Peschanski, M.; et al. Human embryonic stem-cell derivatives for full reconstruction of the pluristratified epidermis: A preclinical study. Lancet 2009, 374, 1745–1753. [Google Scholar] [CrossRef]
- Wu, D.C.; Boyd, A.S.; Wood, K.J. Embryonic stem cell transplantation: Potential applicability in cell replacement therapy and regenerative medicine. Front. Biosci. 2007, 12, 4525–4535. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Avior, Y.; Sagi, I.; Benvenisty, N. Pluripotent stem cells in disease modelling and drug discovery. Nat. Rev. Mol. Cell Biol. 2016, 17, 170–182. [Google Scholar] [CrossRef] [PubMed]
- Sebastiano, V.; Zhen, H.H.; Haddad, B.; Bashkirova, E.; Melo, S.P.; Wang, P.; Leung, T.L.; Siprashvili, Z.; Tichy, A.; Li, J.; et al. Human col7a1-corrected induced pluripotent stem cells for the treatment of recessive dystrophic epidermolysis bullosa. Sci. Transl. Med. 2014, 6, 264ra163. [Google Scholar] [CrossRef] [PubMed]
- Umegaki-Arao, N.; Pasmooij, A.M.; Itoh, M.; Cerise, J.E.; Guo, Z.; Levy, B.; Gostynski, A.; Rothman, L.R.; Jonkman, M.F.; Christiano, A.M. Induced pluripotent stem cells from human revertant keratinocytes for the treatment of epidermolysis bullosa. Sci. Transl. Med. 2014, 6, 264ra164. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Zheng, Y.; Burrows, M.; Liu, S.; Wei, Z.; Nace, A.; Guo, W.; Kumar, S.; Cotsarelis, G.; Xu, X. Generation of folliculogenic human epithelial stem cells from induced pluripotent stem cells. Nat. Commun. 2014, 5, 3071. [Google Scholar] [CrossRef] [PubMed]
- Itoh, M.; Umegaki-Arao, N.; Guo, Z.; Liu, L.; Higgins, C.A.; Christiano, A.M. Generation of 3d skin equivalents fully reconstituted from human induced pluripotent stem cells (ipscs). PLoS ONE 2013, 8, e77673. [Google Scholar] [CrossRef] [PubMed]
- Asti, A.; Gioglio, L. Natural and synthetic biodegradable polymers: Different scaffolds for cell expansion and tissue formation. Int. J. Artif. Organs 2014, 37, 187–205. [Google Scholar] [PubMed]
- Hinderer, S.; Layland, S.L.; Schenke-Layland, K. Ecm and ecm-like materials—Biomaterials for applications in regenerative medicine and cancer therapy. Adv. Drug Deliv. Rev. 2016, 97, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Kohane, D.S.; Langer, R. Polymeric biomaterials in tissue engineering. Pediatr. Res. 2008, 63, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Stoppel, W.L.; Ghezzi, C.E.; McNamara, S.L.; Black, L.D., 3rd; Kaplan, D.L. Clinical applications of naturally derived biopolymer-based scaffolds for regenerative medicine. Ann. Biomed. Eng. 2015, 43, 657–680. [Google Scholar] [CrossRef] [PubMed]
- Rabotyagova, O.S.; Cebe, P.; Kaplan, D.L. Protein-based block copolymers. Biomacromolecules 2011, 12, 269–289. [Google Scholar] [CrossRef] [PubMed]
- Chow, D.; Nunalee, M.L.; Lim, D.W.; Simnick, A.J.; Chilkoti, A. Peptide-based biopolymers in biomedicine and biotechnology. Mater. Sci. Eng. R Rep. 2008, 62, 125–155. [Google Scholar] [CrossRef] [PubMed]
- Dawson, E.; Mapili, G.; Erickson, K.; Taqvi, S.; Roy, K. Biomaterials for stem cell differentiation. Adv. Drug Deliv. Rev. 2008, 60, 215–228. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Tian, L.; Shamirzaei-Jeshvaghani, E.; Dehghani, L.; Ramakrishna, S. Structural properties of scaffolds: Crucial parameters towards stem cells differentiation. World J. Stem Cells 2015, 7, 728–744. [Google Scholar] [CrossRef] [PubMed]
- Willerth, S.M.; Sakiyama-Elbert, S.E. Combining stem cells and biomaterial scaffolds for constructing tissues and cell delivery. In StemBook; Harvard Stem Cell Institute: Cambridge, MA, USA, 2008. [Google Scholar]
- Formigli, L.; Paternostro, F.; Tani, A.; Mirabella, C.; Quattrini Li, A.; Nosi, D.; D’Asta, F.; Saccardi, R.; Mazzanti, B.; Lo Russo, G.; et al. Mscs seeded on bioengineered scaffolds improve skin wound healing in rats. Wound Repair Regen. 2015, 23, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.H.; Hsieh, P.S. Self-assembled adult adipose-derived stem cell spheroids combined with biomaterials promote wound healing in a rat skin repair model. Wound Repair Regen. 2015, 23, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Kosaraju, R.; Rennert, R.C.; Maan, Z.N.; Duscher, D.; Barrera, J.; Whittam, A.J.; Januszyk, M.; Rajadas, J.; Rodrigues, M.; Gurtner, G.C. Adipose-derived stem cell-seeded hydrogels increase endogenous progenitor cell recruitment and neovascularization in wounds. Tissue Eng. Part A 2016, 22, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Zhu, L.; Han, Q.; Liu, W.; Mao, X.; Li, Y.; Yu, N.; Feng, S.; Fu, Q.; Wang, X.; et al. Preformed gelatin microcryogels as injectable cell carriers for enhanced skin wound healing. Acta Biomater. 2015, 25, 291–303. [Google Scholar] [CrossRef] [PubMed]
- Bhowmick, S.; Scharnweber, D.; Koul, V. Co-cultivation of keratinocyte-human mesenchymal stem cell (hmsc) on sericin loaded electrospun nanofibrous composite scaffold (cationic gelatin/hyaluronan/chondroitin sulfate) stimulates epithelial differentiation in hmscs: In vitro study. Biomaterials 2016, 88, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Guan, J.; Guo, S.; Guo, F.; Niu, X.; Liu, Q.; Zhang, C.; Nie, H.; Wang, Y. Human urine-derived stem cells in combination with polycaprolactone/gelatin nanofibrous membranes enhance wound healing by promoting angiogenesis. J. Transl. Med. 2014, 12, 274. [Google Scholar] [CrossRef] [PubMed]
- Machula, H.; Ensley, B.; Kellar, R. Electrospun tropoelastin for delivery of therapeutic adipose-derived stem cells to full-thickness dermal wounds. Adv. Wound Care 2014, 3, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Ozpur, M.A.; Guneren, E.; Canter, H.I.; Karaaltin, M.V.; Ovali, E.; Yogun, F.N.; Baygol, E.G.; Kaplan, S. Generation of skin tissue using adipose tissue-derived stem cells. Plast. Reconstr. Surg. 2016, 137, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Caiado, F.; Carvalho, T.; Silva, F.; Castro, C.; Clode, N.; Dye, J.F.; Dias, S. The role of fibrin e on the modulation of endothelial progenitors adhesion, differentiation and angiogenic growth factor production and the promotion of wound healing. Biomaterials 2011, 32, 7096–7105. [Google Scholar] [CrossRef] [PubMed]
- Assi, R.; Foster, T.R.; He, H.; Stamati, K.; Bai, H.; Huang, Y.; Hyder, F.; Rothman, D.; Shu, C.; Homer-Vanniasinkam, S.; et al. Delivery of mesenchymal stem cells in biomimetic engineered scaffolds promotes healing of diabetic ulcers. Regen. Med. 2016, 11, 245–260. [Google Scholar] [CrossRef] [PubMed]
- Navone, S.E.; Pascucci, L.; Dossena, M.; Ferri, A.; Invernici, G.; Acerbi, F.; Cristini, S.; Bedini, G.; Tosetti, V.; Ceserani, V.; et al. Decellularized silk fibroin scaffold primed with adipose mesenchymal stromal cells improves wound healing in diabetic mice. Stem Cell Res. Ther. 2014, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Chlapanidas, T.; Tosca, M.C.; Farago, S.; Perteghella, S.; Galuzzi, M.; Lucconi, G.; Antonioli, B.; Ciancio, F.; Rapisarda, V.; Vigo, D.; et al. Formulation and characterization of silk fibroin films as a scaffold for adipose-derived stem cells in skin tissue engineering. Int. J. Immunopathol. Pharmacol. 2013, 26, 43–49. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Shen, L.; Wu, Y.; Li, L.; Chen, W.; Hou, C.; Yang, M.; Zeng, W.; Zhu, C. Effect of brain-derived neurotrophic factor on mesenchymal stem cell-seeded electrospinning biomaterial for treating ischemic diabetic ulcers via milieu-dependent differentiation mechanism. Tissue Eng. Part A 2015, 21, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhang, H.; Zhang, X.; Lu, W.; Huang, X.; Xie, H.; Zhou, J.; Wang, W.; Zhang, Y.; Liu, Y.; et al. Synergistic angiogenesis promoting effects of extracellular matrix scaffolds and adipose-derived stem cells during wound repair. Tissue Eng. Part A 2011, 17, 725–739. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, A.; Rodel, P.; Anamur, C.; Seeliger, C.; Imhoff, A.B.; Herbst, E.; Vogt, S.; van Griensven, M.; Winter, G.; Engert, J. Calcium alginate gels as stem cell matrix-making paracrine stem cell activity available for enhanced healing after surgery. PLoS ONE 2015, 10, e0118937. [Google Scholar] [CrossRef] [PubMed]
- Bussche, L.; Harman, R.M.; Syracuse, B.A.; Plante, E.L.; Lu, Y.C.; Curtis, T.M.; Ma, M.; Van de Walle, G.R. Microencapsulated equine mesenchymal stromal cells promote cutaneous wound healing in vitro. Stem Cell Res. Ther. 2015, 6, 66. [Google Scholar] [CrossRef] [PubMed]
- Bellini, M.Z.; Caliari-Oliveira, C.; Mizukami, A.; Swiech, K.; Covas, D.T.; Donadi, E.A.; Oliva-Neto, P.; Moraes, A.M. Combining xanthan and chitosan membranes to multipotent mesenchymal stromal cells as bioactive dressings for dermo-epidermal wounds. J. Biomater. Appl. 2015, 29, 1155–1166. [Google Scholar] [CrossRef] [PubMed]
- Zeinali, R.; Biazar, E.; Keshel, S.H.; Tavirani, M.R.; Asadipour, K. Regeneration of full-thickness skin defects using umbilical cord blood stem cells loaded into modified porous scaffolds. ASAIO J. 2014, 60, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Hao, H.; Tong, C.; Liu, J.; Dong, L.; Ti, D.; Hou, Q.; Liu, H.; Han, W.; Fu, X. Transdifferentiation of umbilical cord-derived mesenchymal stem cells into epidermal-like cells by the mimicking skin microenvironment. Int. J. Low Extrem. Wounds 2015, 14, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Garg, R.K.; Rennert, R.C.; Duscher, D.; Sorkin, M.; Kosaraju, R.; Auerbach, L.J.; Lennon, J.; Chung, M.T.; Paik, K.; Nimpf, J.; et al. Capillary force seeding of hydrogels for adipose-derived stem cell delivery in wounds. Stem Cells Transl. Med. 2014, 3, 1079–1089. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, M.T.; da Silva, L.P.; Santos, T.C.; Pirraco, R.P.; Correlo, V.M.; Reis, R.L.; Marques, A.P. Gellan gum-hyaluronic acid spongy-like hydrogels and cells from adipose tissue synergize promoting neoskin vascularization. ACS Appl. Mater. Interfaces 2014, 6, 19668–19679. [Google Scholar] [CrossRef] [PubMed]
- O’Loughlin, A.; Kulkarni, M.; Creane, M.; Vaughan, E.E.; Mooney, E.; Shaw, G.; Murphy, M.; Dockery, P.; Pandit, A.; O’Brien, T. Topical administration of allogeneic mesenchymal stromal cells seeded in a collagen scaffold augments wound healing and increases angiogenesis in the diabetic rabbit ulcer. Diabetes 2013, 62, 2588–2594. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.H.; Lee, J.H.; Won, J.H.; Cho, M.K. Mesenchymal stem cells improve wound healing in vivo via early activation of matrix metalloproteinase-9 and vascular endothelial growth factor. J. Korean Med. Sci. 2011, 26, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Lequeux, C.; Oni, G.; Wong, C.; Damour, O.; Rohrich, R.; Mojallal, A.; Brown, S.A. Subcutaneous fat tissue engineering using autologous adipose-derived stem cells seeded onto a collagen scaffold. Plast. Reconstr. Surg. 2012, 130, 1208–1217. [Google Scholar] [CrossRef] [PubMed]
- Lough, D.M.; Wetter, N.; Madsen, C.; Reichensperger, J.; Cosenza, N.; Cox, L.; Harrison, C.; Neumeister, M.W. Transplantation of an lgr6+ epithelial stem cell-enriched scaffold for repair of full-thickness soft-tissue defects: The in vitro development of polarized hair-bearing skin. Plast. Reconstr. Surg. 2016, 137, 495–507. [Google Scholar] [CrossRef] [PubMed]
- Falanga, V.; Iwamoto, S.; Chartier, M.; Yufit, T.; Butmarc, J.; Kouttab, N.; Shrayer, D.; Carson, P. Autologous bone marrow-derived cultured mesenchymal stem cells delivered in a fibrin spray accelerate healing in murine and human cutaneous wounds. Tissue Eng. 2007, 13, 1299–1312. [Google Scholar] [CrossRef] [PubMed]
- Almine, J.F.; Bax, D.V.; Mithieux, S.M.; Nivison-Smith, L.; Rnjak, J.; Waterhouse, A.; Wise, S.G.; Weiss, A.S. Elastin-based materials. Chem. Soc. Rev. 2010, 39, 3371–3379. [Google Scholar] [CrossRef] [PubMed]
- Wise, S.G.; Mithieux, S.M.; Weiss, A.S. Engineered tropoelastin and elastin-based biomaterials. Adv. Protein Chem. Struct. Biol. 2009, 78, 1–24. [Google Scholar] [PubMed]
- Shen, Y.I.; Cho, H.; Papa, A.E.; Burke, J.A.; Chan, X.Y.; Duh, E.J.; Gerecht, S. Engineered human vascularized constructs accelerate diabetic wound healing. Biomaterials 2016, 102, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, C.; de Assis, A.M.; Moura, D.J.; Halmenschlager, G.; Saffi, J.; Xavier, L.L.; Fernandes Mda, C.; Wink, M.R. New therapy of skin repair combining adipose-derived mesenchymal stem cells with sodium carboxymethylcellulose scaffold in a pre-clinical rat model. PLoS ONE 2014, 9, e96241. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.L.; Han, D.K.; Park, K.; Song, S.H.; Kim, J.Y.; Kim, J.M.; Ki, H.Y.; Yie, S.W.; Roh, C.R.; Jeon, E.S.; et al. Enhanced dermal wound neovascularization by targeted delivery of endothelial progenitor cells using an RGD-g-PLLA scaffold. Biomaterials 2009, 30, 3742–3748. [Google Scholar] [CrossRef] [PubMed]
- Geesala, R.; Bar, N.; Dhoke, N.R.; Basak, P.; Das, A. Porous polymer scaffold for on-site delivery of stem cells--protects from oxidative stress and potentiates wound tissue repair. Biomaterials 2016, 77, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.Y.; Cobain, E.; Huard, J.; Huang, L. Thermosensitive hydrogel peg-plga-peg enhances engraftment of muscle-derived stem cells and promotes healing in diabetic wound. Mol. Ther. 2007, 15, 1189–1194. [Google Scholar] [CrossRef] [PubMed]
- Natesan, S.; Zamora, D.O.; Wrice, N.L.; Baer, D.G.; Christy, R.J. Bilayer hydrogel with autologous stem cells derived from debrided human burn skin for improved skin regeneration. J. Burn Care Res. 2013, 34, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Cho, S.W.; Son, S.M.; Bogatyrev, S.R.; Singh, D.; Green, J.J.; Mei, Y.; Park, S.; Bhang, S.H.; Kim, B.S.; et al. Genetic engineering of human stem cells for enhanced angiogenesis using biodegradable polymeric nanoparticles. Proc. Natl. Acad. Sci. USA 2010, 107, 3317–3322. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Shi, J.; Zhang, M.; Chen, Y.; Wang, X.; Zhang, L.; Tian, Z.; Yan, Y.; Li, Q.; Zhong, W.; et al. Mesenchymal stem cell-laden anti-inflammatory hydrogel enhances diabetic wound healing. Sci. Rep. 2015, 5, 18104. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Liu, N.; Yang, X.; Feng, Z.; Qi, F. Adiposed-derived stem cells seeded on plcl/p123 eletrospun nanofibrous scaffold enhance wound healing. Biomed. Mater. 2014, 9, 035012. [Google Scholar] [CrossRef] [PubMed]
- Baker, M.I.; Walsh, S.P.; Schwartz, Z.; Boyan, B.D. A review of polyvinyl alcohol and its uses in cartilage and orthopedic applications. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100, 1451–1457. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, J.; Pereira, T.; Amorim, I.; Caseiro, A.R.; Lopes, M.A.; Lima, J.; Gartner, A.; Santos, J.D.; Bartolo, P.J.; Rodrigues, J.M.; et al. Cell therapy with human mscs isolated from the umbilical cord wharton jelly associated to a pva membrane in the treatment of chronic skin wounds. Int. J. Med. Sci. 2014, 11, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Hassan, W.U.; Kennedy, R.; Greiser, U.; Pandit, A.; Garcia, Y.; Wang, W. Performance of an in situ formed bioactive hydrogel dressing from a peg-based hyperbranched multifunctional copolymer. Acta Biomater. 2014, 10, 2076–2085. [Google Scholar] [CrossRef] [PubMed]
- Hassan, W.; Dong, Y.; Wang, W. Encapsulation and 3d culture of human adipose-derived stem cells in an in-situ crosslinked hybrid hydrogel composed of peg-based hyperbranched copolymer and hyaluronic acid. Stem Cell Res. Ther. 2013, 4, 32. [Google Scholar] [CrossRef] [PubMed]
- Martino, S.; D’Angelo, F.; Armentano, I.; Kenny, J.M.; Orlacchio, A. Stem cell-biomaterial interactions for regenerative medicine. Biotechnol. Adv. 2012, 30, 338–351. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Deng, Z.; Han, S.; Liu, T.; Wen, N.; Lu, W.; Geng, X.; Huang, S.; Jin, Y. Tissue-engineered skin containing mesenchymal stem cells improves burn wounds. Artif. Organs 2008, 32, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Catanzano, O.; D’Esposito, V.; Acierno, S.; Ambrosio, M.R.; De Caro, C.; Avagliano, C.; Russo, P.; Russo, R.; Miro, A.; Ungaro, F.; et al. Alginate-hyaluronan composite hydrogels accelerate wound healing process. Carbohydr. Polym. 2015, 131, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Dash, B.C.; Mandal, B.B.; Kundu, S.C. Silk gland sericin protein membranes: Fabrication and characterization for potential biotechnological applications. J. Biotechnol. 2009, 144, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Dinescu, S.; Galateanu, B.; Albu, M.; Cimpean, A.; Dinischiotu, A.; Costache, M. Sericin enhances the bioperformance of collagen-based matrices preseeded with human-adipose derived stem cells (hadscs). Int. J. Mol. Sci. 2013, 14, 1870–1889. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.O.; Lee, Y.; Hwang, J.W.; Kim, H.; Kim, S.M.; Chang, S.W.; Lee, H.S.; Choi, Y.S. Wound healing properties of a 3-D scaffold comprising soluble silkworm gland hydrolysate and human collagen. Colloids Surf. B 2014, 116, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Cantu, D.A.; Fu, Y.; Kim, J.; Zheng, X.; Hematti, P.; Kao, W.J. Thiol-ene michael-type formation of gelatin/poly(ethylene glycol) biomatrices for three-dimensional mesenchymal stromal/stem cell administration to cutaneous wounds. Acta Biomater. 2013, 9, 8802–8814. [Google Scholar] [CrossRef] [PubMed]
| Biological Polymer | Structure | Selective Wound Healing Application | Important Findings | Reference |
|---|---|---|---|---|
| Collagen | A fibrous triple helical protein. Collagen type I, a major subtype consists of two alpha 1 units and one alpha 2 | 3D scaffold Hydrogel Composite material | MSCs within the scaffolds greatly ameliorated the quality of regenerated skin, reduced collagen deposition. Enhanced reepithelization, increased neo-angiogenesis, and promoted a greater return of hair follicles and sebaceous glands. The mechanisms involved in these beneficial effects were likely related to the ability of MSCs to release paracrine factors modulating the wound healing response. Self-assembled ASC spheroids on chitosan-hyaluronan membranes expressed more cytokine genes (fibroblast growth factor 1, vascular endothelial growth factor, and chemokine [C-C motif] ligand 2) as well as migration-associated genes (chemokine) [C-X-C motif] receptor type 4 and matrix metalloprotease 1. Spheroids combined with the use of biomaterials can enhance skin wound healing and more capillary formation. This study shows the potential use of biomaterial-derived 3D MSC spheroids in wound treatment. Increased the recruitment of provascular circulating bone marrow-derived mesenchymal progenitor cells in vivo. Significant increase in BM-MPC migration, proliferation, and tubulization when exposed to hydrogel-seeded ASC conditioned medium. BM-MPC expression of genes related to cell stemness and angiogenesis was also significantly increased following exposure to hydrogel-seeded ASC conditioned medium. ASC-seeded hydrogels improve both progenitor cell recruitment and functionality to effect greater neovascularization. | [42,43,44] |
| Gelatin | A hydrolytic byproduct of collagen | Microgel Composite material; nanofibers; electrospun scaffold | Biodegradable gelatin microgels (GMs), as 3D micro-scaffolds, could provide suitable microenvironment for stem cell proliferation. GMs could greatly improve growth factor secretion from hASCs and might be an enhanced strategy to promote hASCs-assisted wound healing. GMs could protect hASCs in the micro-niches and exhibit excellent injectability through syringe without obvious cell damages. hASCs delivered via GMs assisted injection could retain hASCs in situ and improve cell survival in wounds. Besides, hASCs retention improved by GMs could enhance secretion of positive tissue growth factors for wound healing, indicating an advanced injection strategy superior to free cell suspension injection GMs assisted cell delivery could accelerate wound healing by constant modulation of local growth factor expression to enhance regeneration and vascularization Treated wounds closed much faster, with increased re-epithelialization, collagen formation, and angiogenesis in vivo. USCs could secrete VEGF and TGF-β1. USC-conditioned medium enhanced the migration, proliferation, and tube formation of endothelial cells in vitro The composite nanofibrous scaffold was found to be biocompatible. Electrospun scaffold containing sericin promoted epithelial differentiation of hMSC. | [45,46,47] |
| Elastin | An elastic protein made up of water soluble tropoelastin | Electrospun scaffold | Electrospun tropoelastin membranes form stable structures that retain their integrity and strength in tissue culture medium. ADSCs rapidly proliferate on the scaffold and secrete an ECM that eventually covers the entire scaffold in vitro. The populated scaffold is well tolerated in a murine excisional wound model. Wounds treated with ADSC-populated tropoelastin scaffolds showed greater rate of closure and restoration of normal epithelium. | [48] |
| Fibrin | A fibrous non-globular protein produced by the cleavage of fibrinogen. | Gel Composite material | Enhanced wound healing for the scaffold containing ADSC and keratinocyte. Total epithelialization and higher collagen deposition and higher vascularization. Co-administration of FbnE enriched scaffold (SM) with CB-EPC accelerated wound closure and vascularization compared to FbnE scaffold alone. No differences in number of pericytes and myofibroblasts seen. No comparison to Integra treated mice as all died before study was complete. | [49,50] |
| Laminin | A heterotrimeric glycoprotein and binds to ECM proteins and cell membranes | Surface modification | The biomimetic collagen scaffold increases VEGF secretion from MSC in vitro. Activated MSC in collagen scaffolds increase wound healing in vivo. Activated MSCs increase wound healing in a splinted back wound model. Laminin improves wound healing efficiency. MSC delivered topically increase wound healing. | [51] |
| Silk protein | Extracted from cocoon of silk worms. It contains fibrous protein fibroin and water soluble sericin protein. | Scaffold Composite material Film | Scaffolds significantly improved tissue regeneration, reducing the wound area. Decellularized patches are almost as effective as cellularized patches in the treatment of diabetic wounds Scaffolds improve healing through the release of angiogenic and collagen deposition stimulating molecules. Silk Fibroin can be used a scaffold and is biocompatible, biodegradable and has excellent tensile strength The scaffold can support adipose derived stem cells for skin tissue engineering. Added Pectin and Glycerol can benefit scaffold by promoting protein conformation transition and biomaterial flexibility, respectively. BDNF-induced proliferation and migration of MSCs. BDNF stimulation affects the ability of MSCs to secrete IL-8, NGF, and MMP-9 and that this process depends largely on the Akt signaling pathway. The upregulation of NGF, IL-8, and MMP-9 in the BDNF-CM group contributed to angiogenesis. The BDNF-CM-modified materials also significantly accelerated wound healing. BDNF promotes angiogenesis and enhances the milieu-dependent endothelial differentiation of MSCs in ischemic ulcers. | [46,52,53,54] |
| Hyaluronic acid/Hyaluronan | An anionic nonsulfated glycosaminoglyacan that helps in cell migration and proliferation | Surface modification Composite material | Scaffolds seeded with VEGF165-modified rHFSCs, resulted in promotion of angiogenesis during wound healing and facilitation of vascularization in skin substitutes Increased VEGF165 level in the repair microenvironment improved vascularization ability. It is believed that HFSCs secrete a variety of cytokines to promote wound healing. Subcutaneous implantation showed that vascularization capacity of non-seeded SJS and ADM were greater than that of Co-CS-HA in subcutaneous wounds. ADSCs cultured on SIS secreted more VEGF compared to those seeded on ADM and Co-CS-HA ADSC-seeded scaffolds enhanced angiogenesis and wound healing rate compared to non-seeded scaffold in in vivo mouse models. ADSC-SIS and ADSC-ADM had greater microvessel densities than ADSC-co-CS-HA in vivo. | [20,55] |
| Alginate | An anionic polysachharide consisting of homopolymeric blocks of (1-4)-linked β-d-mannuronate (M) and C-5 epimer α-l-guluronate (G) residues. | Hydrogel Composite material | Humans MSCs remained viable for the duration of 6 weeks within the gels. Human VEGF and bFGF was found in quantifiable concentrations in cell culture supernatants of gels loaded with MSCs and incubated for a period of 6 weeks. Conditioned medium from mesenchymal stromal cells stimulates migration of dermal fibroblasts in scratch assays. Conditioned medium of mesenchymal stromal cells induces alterations in the expression of genes involved in wound healing. Encapsulated mesenchymal stromal cells retain stem cell characteristics and remain viable during long-term encapsulation. Encapsulated mesenchymal stromal cell-derived conditioned medium stimulates migration of dermal fibroblasts and induces alterations in the expression of genes involved in wound healing. Cells attached proliferated on the porous membrane. Accelerated wound healing. | [56,57] |
| Chitosan | A linear polysaccharide consisting of β-(1→4)-linked d-glucosamine and N-acetyl-d-glucosamine. | Membrane Scaffold Composite material | Conditioned medium from mesenchymal stromal cells stimulates migration of dermal fibroblasts in scratch assays. Conditioned medium of mesenchymal stromal cells induces alterations in the expression of genes involved in wound healing. Encapsulated mesenchymal stromal cells retain stem cell characteristics and remain viable during long-term encapsulation. Encapsulated mesenchymal stromal cell-derived conditioned medium stimulates migration of dermal fibroblasts and induces alterations in the expression of genes involved in wound healing. The scaffold was biocompatible UC-MSC differentiated to epidermis and positive for the epidermal markers cytokeratin 19 and involucrin at 14 days. The constructed epidermis substitutes helped rapid wound healing. Better cell adhesion, growth, and proliferation inside the modified scaffolds. Showed a good resilience and compliance with movement as a skin graft. All scaffolds, especially those with stem cells, exhibited pronounced effects on wound closure. The reconstructed skin in grafted groups demonstrated an intact epithelium with the formation of new hair follicles and sebaceous glands, which were reminiscent of the structures of normal skin. | [58,59,60] |
| Pullulan | A polysaccharide consisting of maltotriose units, connected by an α-1,6 glycosidic bond. | Hydrogel Composite material | Described above. Hydrogel seeding of ASCs resulted in the enhanced expression of multiple stemness and angiogenesis-related genes (Oct4, Vegf, Mcp-1, and Sdf-1) in vitro. ASCs seeded within hydrogel scaffolds showed minimal proliferation and maintained baseline levels of metabolic activity. Hydrogel delivery improved ASC survival in vivo. Resulted in accelerated wound closure and increased vascularity in splinted murine wounds. | [44,61] |
| Xanthan | A hetero-polysaccharide with main containing glucose units and side chain of trisaccharides | Membrane | Described above. | [58] |
| Gellan gum | A water soluble anionic polysaccharide with repeated tetrasaccharide units containing two d-glucose and one of each l-rhamnose and d-glucuronic acid | Hydrogel | The hydrogels absorbed early inflammatory cell infiltrate and led to formulation of granulation tissue in vivo. Improved wound closure, re-epithelialization, and matrix remodeling. Promoted superior neo-vascularization. | [62] |
| Synthetic Polymer | Structure | Selective Wound Healing Application | Important Findings | Reference |
|---|---|---|---|---|
| Poly(l-lactic acid) (PLA or PLLA) | A biodegradable and thermoplastic polymer synthesized using monomers of lactic acid or lactide. | Scaffold | Scaffolds had a high porosity and a 50–75% increase in swelling, along with complete protein release in the presence of phosphate-buffered saline. Accelerated wound re-epithelialization in mouse model Maintained optimal hydration of the exposed tissues and decreased wound healing time in vivo. | [72] |
| Poly(ethylene glycol) (PEG) | A hydrophilic polymer synthesized by anionic ring-opening polymerization of ethylene oxide. | Scaffold Hydrogel Composite material | Polymer network/porous scaffold helps cells from oxidative stress. The implant showed fibroblast proliferation, collagen deposition, and anti-oxidant enzyme activity. Enhanced wound healing by enhanced engraftment and increased vascularization. Decreased proinflammatory cytokines and increased anti-inflammatory cytokines. Wounds treated with MDSC and PEG-PLGA-PEG showed enhanced wound closure rate, epithelium migration, and collagen deposition. There was increased engraftment of MDSCs into the wound bed compared to controls (MDSC treatment without hydrogel and MDSC with control dressing). In wounds, 25% MDSCs differentiated into fibroblasts, 10% into myofibroblasts, 10% into endothelial cells, and none into macrophages. Rat excision wounds treated with PEGylated fibrin-collagen bilayer hydrogels show decreased wound contraction over time. They show faster wound closure in comparison with control Within bilayer hydrogels, dsASCs proliferate, differentiate, maintain a spindle-shaped morphology in collagen, and develop tubular microvascular networks in PEGylated fibrin. | [73,74,75] |
| Poly(lactic-co-glycolic acid) (PLGA) | A biodegradable copolymer of glycolic and lactic acid. | Scaffold | Scaffolds seeded with VEGF-transfected stem cells led to increased blood vessel migration into the constructs compared to control cells or cells transfected with VEGF using a commercial reagent. There was increased endothelial cell density compared to the controls. | [74,76] |
| Polyurethane | A polymer synthesized by reacting poly-isocyanate and polyol. Contains urethane to join organic units. | Composite material | Described above. | [73] |
| Poly(N-isopropylacrylamide) (PNIPAM) | A thermoresponsive polymer synthesized using free radical polymerization of N-isopropylacrylamide. | Thermosensitive hydrogel | Hydrogel and BMSC combination therapy promoted wound contraction. The hydrogel inhibited chronic inflammation. Hydrogel and BMSCs combination therapy promoted the formation of granulation tissue. Hydrogel and BMSCs combination therapy promoted keratinocyte proliferation and differentiation. Hydrogel and BMSCs combination therapy improved the quality of wound healing. | [77] |
| Polycaprolactone (PCL) | A biodegradable polyester prepared by ring opening polymerization of ε-caprolactone. | Composite material | ADSCs differentiated into epidermal-like structures Observed higher microvessel density in rat skin tissue injury models. Improved healing was observed in vivo. ADSCs-PLCL/P123 scaffolds with the thickness of 150–250 μm match well with the epidermis layer (200–250 μm). | [47,78] |
| PAA-poly(amidoamine) | A dendrimer with repetitively branched subunits of amide and amine | Hydrogel | Described above. | [77] |
| Biopolymers for Surface Modification | Effect on Stem Cells and Wound Healing |
|---|---|
| Glycosaminoglycan | Promoted MSC survival. Improved healing, keratinization and vascularization [84]. |
| Laminin | Enhanced MSC survival and VEGF secretion. Promoted healing [51]. |
| HA | Maintained cell survival of ADSC and keratinocytes and improved wound closure [85]. Enhanced cell adhesion and survival and secretion of paracrine factors such as VEGF and bFGF [56]. Enhanced neovascularization, wound closure, re-epithelialization, matrix remodeling and reduced inflammation [62]. |
| Sericin/Silk derivative | Increased cell proliferation of ADSC and maintained adipogenicity of the cells by stimulating the expression of PPARγ2 [87]. Reduced oxidative stress in the cells, enhanced re-epithelialization and wound closure [88]. Promoted cell viability and proliferation of MSCs and keratinocytes and fibroblasts and helped differentiation of MSCs to epithelial lineage [46]. |
| Fibrin | Fibrin fragment E promoted cell adhesion and differentiation of cord blood epidermal progenitor cells to endothelial cells and enhanced vascularization and wound closure [50]. PEGylated fibrin promoted cell proliferation of ADSCs and tubular microvascular formation in the scaffold and enhance wound closure, re-epithelialization [75]. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dash, B.C.; Xu, Z.; Lin, L.; Koo, A.; Ndon, S.; Berthiaume, F.; Dardik, A.; Hsia, H. Stem Cells and Engineered Scaffolds for Regenerative Wound Healing. Bioengineering 2018, 5, 23. https://doi.org/10.3390/bioengineering5010023
Dash BC, Xu Z, Lin L, Koo A, Ndon S, Berthiaume F, Dardik A, Hsia H. Stem Cells and Engineered Scaffolds for Regenerative Wound Healing. Bioengineering. 2018; 5(1):23. https://doi.org/10.3390/bioengineering5010023
Chicago/Turabian StyleDash, Biraja C., Zhenzhen Xu, Lawrence Lin, Andrew Koo, Sifon Ndon, Francois Berthiaume, Alan Dardik, and Henry Hsia. 2018. "Stem Cells and Engineered Scaffolds for Regenerative Wound Healing" Bioengineering 5, no. 1: 23. https://doi.org/10.3390/bioengineering5010023
APA StyleDash, B. C., Xu, Z., Lin, L., Koo, A., Ndon, S., Berthiaume, F., Dardik, A., & Hsia, H. (2018). Stem Cells and Engineered Scaffolds for Regenerative Wound Healing. Bioengineering, 5(1), 23. https://doi.org/10.3390/bioengineering5010023






