Spatiotemporal Mapping of Biomechanical Stress Predicts Region-Specific Retinal Injury in a Murine Model of Blunt Ocular Trauma
Abstract
1. Introduction
2. Materials and Methods
2.1. Computational Simulation of Weight Drop
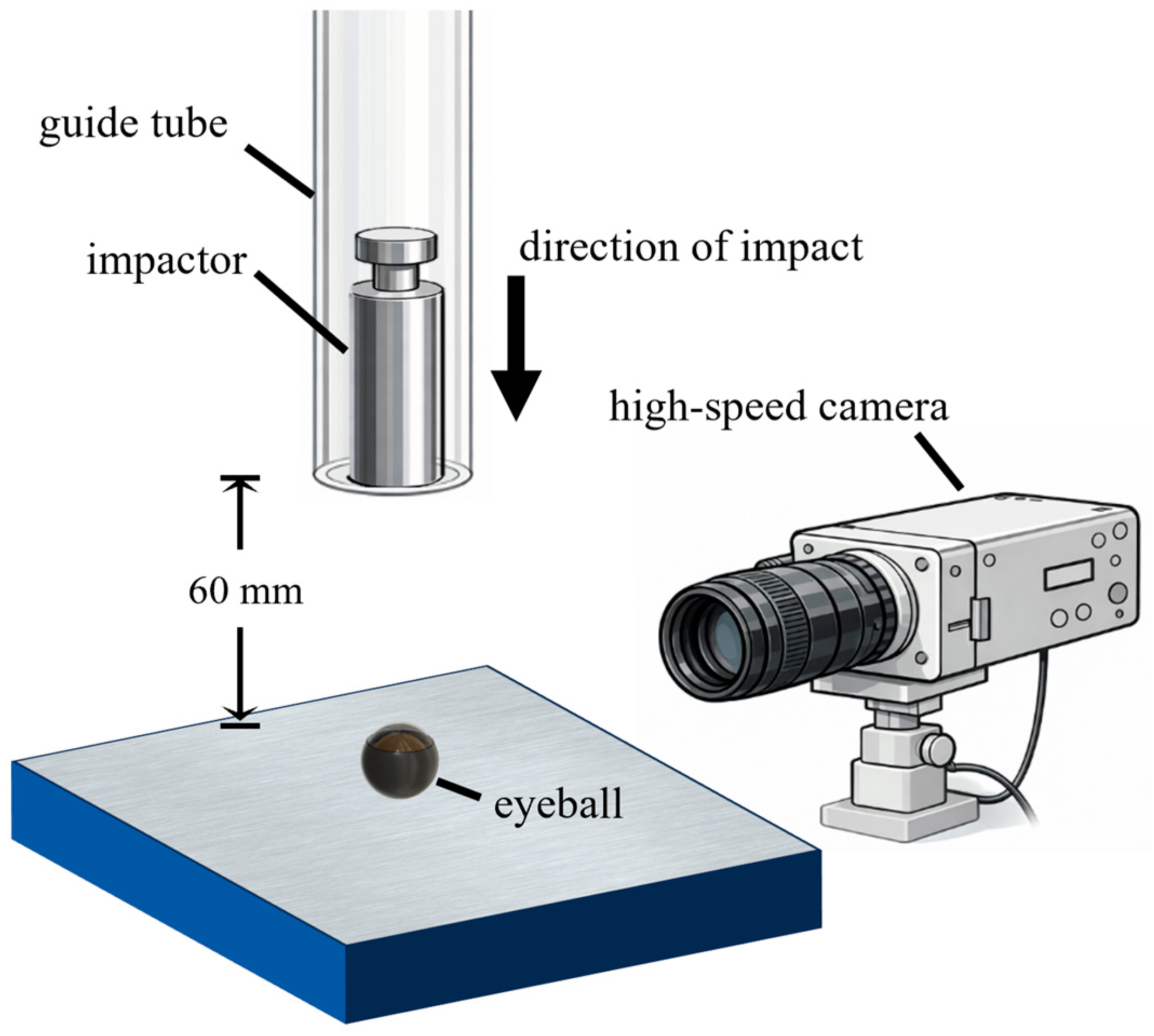
2.2. In Vitro Weight-Drop Test on Mouse Eye and Histological Assessment
3. Results
3.1. Stress Distribution in Retina
3.2. Histological Analysis of Retinal Damage
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| FEA | Finite Element Analysis |
| H&E | Hematoxylin and Eosin |
| OCT | Optical Coherence Tomography |
| RPE | Retinal Pigment Epithelium |
| σr,σθ,σφ | Radial, Circumferential and Azimuthal Stress |
| TRD | Traumatic Retinal Detachment |
References
- Thylefors, B. Epidemiological Patterns of Ocular Trauma. Aust. N. Z. J. Ophthalmol. 1992, 20, 95–98. [Google Scholar] [CrossRef]
- Li, C.; Fu, Y.; Liu, S.; Yu, H.; Yang, X.; Zhang, M.; Liu, L. The Global Incidence and Disability of Eye Injury: An Analysis from the Global Burden of Disease Study 2019. eClinicalMedicine 2023, 62, 102134. [Google Scholar] [CrossRef]
- Iftikhar, M.; Latif, A.; Farid, U.Z.; Usmani, B.; Canner, J.K.; Shah, S.M.A. Changes in the Incidence of Eye Trauma Hospitalizations in the United States from 2001 Through 2014. JAMA Ophthalmol. 2019, 137, 48. [Google Scholar] [CrossRef]
- Kuhn, F.; Morris, R.; Witherspoon, C.D.; Mann, L. Epidemiology of Blinding Trauma in the United States Eye Injury Registry. Ophthalmic Epidemiol. 2006, 13, 209–216. [Google Scholar] [CrossRef]
- Rohowetz, L.; Fan, J.; Flynn, H., Jr. Vitreoretinal Injury Associated with Sports Ball Ocular Trauma. Clin. Ophthalmol. 2025, 19, 1931–1943. [Google Scholar] [CrossRef]
- Sebag, J. Anatomy and Pathology of the Vitreo-Retinal Interface. Eye 1992, 6, 541–552. [Google Scholar] [CrossRef]
- Ghazi, N.G.; Green, W.R. Pathology and Pathogenesis of Retinal Detachment. Eye 2002, 16, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Blanch, R.J.; Ahmed, Z.; Sik, A.; Snead, D.R.J.; Good, P.A.; O’Neill, J.; Berry, M.; Scott, R.A.H.; Logan, A. Neuroretinal Cell Death in a Murine Model of Closed Globe Injury: Pathological and Functional Characterization. Investig. Ophthalmol. Vis. Sci. 2012, 53, 7220. [Google Scholar] [CrossRef] [PubMed]
- Sponsel, W.E.; Gray, W.; Scribbick, F.W.; Stern, A.R.; Weiss, C.E.; Groth, S.L.; Walker, J.D. Blunt Eye Trauma: Empirical Histopathologic Paintball Impact Thresholds in Fresh Mounted Porcine Eyes. Investig. Ophthalmol. Vis. Sci. 2011, 52, 5157. [Google Scholar] [CrossRef]
- Chen, D.; Liu, X.; Sun, X.; Liu, Y.; Geng, X.; Huo, H.; Tang, M.; Tang, Z.; Dong, Y.; Wang, J.; et al. Experimental Evidence to Understand Mechanical Causes of Retinal Detachment Following Blunt Trauma. Exp. Eye Res. 2023, 233, 109552. [Google Scholar] [CrossRef] [PubMed]
- Beckmann, L.; Cai, Z.; Cole, J.; Miller, D.A.; Liu, M.; Grannonico, M.; Zhang, X.; Ryu, H.J.; Netland, P.A.; Liu, X.; et al. In Vivo Imaging of the Inner Retinal Layer Structure in Mice after Eye-Opening Using Visible-Light Optical Coherence Tomography. Exp. Eye Res. 2021, 211, 108756. [Google Scholar] [CrossRef]
- Liu, X.; Wang, L.; Wang, C.; Sun, G.; Liu, S.; Fan, Y. Mechanism of Traumatic Retinal Detachment in Blunt Impact: A Finite Element Study. J. Biomech. 2013, 46, 1321–1327. [Google Scholar] [CrossRef]
- Rossi, T.; Boccassini, B.; Esposito, L.; Iossa, M.; Ruggiero, A.; Tamburrelli, C.; Bonora, N. The Pathogenesis of Retinal Damage in Blunt Eye Trauma: Finite Element Modeling. Investig. Ophthalmol. Vis. Sci. 2011, 52, 3994. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.D.; Colmenarez, J.A.; Choi, E.H.; Suh, A.; Suh, A.; Lam, M.; Hoskin, A.; Minckler, D.S.; Lin, K.Y.; Shahraki, K.; et al. Finite Element Analysis of Mechanical Ocular Sequelae from Badminton Shuttlecock Projectile Impact. Ophthalmol. Sci. 2025, 5, 100625. [Google Scholar] [CrossRef]
- Rydz, C.; Colmenarez, J.A.; Shahraki, K.; Dong, P.; Gu, L.; Suh, D.W. Finite Element Analysis of Ocular Impact Forces and Potential Complications in Pickleball-Related Eye Injuries. Bioengineering 2025, 12, 570. [Google Scholar] [CrossRef] [PubMed]
- Mohd Rasidin, A.H.; Muhammad-Ikmal, M.K.; Raja Omar, R.N.; Yaakub, A.; Ahmad Tajudin, L.S. Clinical Audit on Badminton-Related Ocular Injuries in a Tertiary Hospital in Malaysia. Cureus 2022, 14, e30769. [Google Scholar] [CrossRef] [PubMed]
- Rangarajan, N.; Kamalakkannan, S.B.; Hasija, V.; Shams, T.; Jenny, C.; Serbanescu, I.; Ho, J.; Rusinek, M.; Levin, A.V. Finite Element Model of Ocular Injury in Abusive Head Trauma. J. Am. Assoc. Pediatr. Ophthalmol. Strabismus 2009, 13, 364–369. [Google Scholar] [CrossRef]
- Gray, W.; Sponsel, W.E.; Scribbick, F.W.; Stern, A.R.; Weiss, C.E.; Groth, S.L.; Walker, J.D. Numerical Modeling of Paintball Impact Ocular Trauma: Identification of Progressive Injury Mechanisms. Investig. Ophthalmol. Vis. Sci. 2011, 52, 7506. [Google Scholar] [CrossRef]
- Chen, D.; Sun, X.; Wu, Y.; Tang, M.; Wang, J.; Qiao, X.; Zhu, Y.; Zhang, Z.; Du, X.; Guo, J.; et al. A Finite Element Model of the Eye Matched with in Vitro Experiments for the Prediction of Traumatic Retinal Detachment. Theor. Appl. Mech. Lett. 2024, 14, 100539. [Google Scholar] [CrossRef]
- Pang, J.; Le, L.; Zhou, Y.; Tu, R.; Hou, Q.; Tsuchiya, D.; Thomas, N.; Wang, Y.; Yu, Z.; Alexander, R.; et al. NOTCH Signaling Controls Ciliary Body Morphogenesis and Secretion by Directly Regulating Nectin Protein Expression. Cell Rep. 2021, 34, 108603. [Google Scholar] [CrossRef]
- Zhai, Y.; Wang, J.; Mendoza, V.O.; Ye, M.; Shahraki, K.; Suh, D.W.; Minckler, D.S.; Karpova, T.; Nunes, K.; Dong, P.; et al. Spatial Relationship between Histological Staining Intensity and Corneal Stiffness Variations: Insights from AFM Indentation in Infant African Green Monkeys. J. Mech. Behav. Biomed. Mater. 2025, 169, 107047. [Google Scholar] [CrossRef]
- Colmenarez, J.A.; Zhai, Y.; Mendoza, V.O.; Dong, P.; Nunes, K.; Suh, D.; Gu, L. Damage-Induced Softening of the Sclera: A Pseudo-Elastic Modeling Approach. J. Eng. Sci. Med. Diagn. Ther. 2024, 7, 031001. [Google Scholar] [CrossRef]
- Franze, K.; Francke, M.; Günter, K.; Christ, A.F.; Körber, N.; Reichenbach, A.; Guck, J. Spatial Mapping of the Mechanical Properties of the Living Retina Using Scanning Force Microscopy. Soft Matter 2011, 7, 3147. [Google Scholar] [CrossRef]
- Ariza-Gracia, M.Á.; Wu, W.; Calvo, B.; Malvè, M.; Büchler, P.; Rodriguez Matas, J.F. Fluid–Structure Simulation of a General Non-Contact Tonometry. A Required Complexity? Comput. Methods Appl. Mech. Eng. 2018, 340, 202–215. [Google Scholar] [CrossRef]
- Aung, O.; Rossi, P.J.; Dyer, M.R.; Stellpflug, A.; Zhai, Y.; Kenneth, A.; Wang, X.; Chang, J.; Chen, Y.; Tefft, B.; et al. Biofabrication of Small Vascular Graft with Acellular Human Amniotic Membrane: A Proof-of-Concept Study in Pig. Biofabrication 2024, 17. [Google Scholar] [CrossRef]
- Delori, F.; Pomerantzeff, O.; Cox, M.S. Deformation of the Globe under High-Speed Impact: Its Relation to Contusion Injuries. Investig. Ophthalmol. Vis. Sci. 1969, 8, 290–301. [Google Scholar]
- Chauhan, K.; Dave, V.P.; De Ribot, F.M.; Agrawal, R.; Sallam, A.B.; Andayani, G.; Chang, C.-J.; Hsiao, C.-H.; Bastion, M.-L.C.; Hattenbach, L.-O.; et al. Traumatic Retinal Detachment: A Contemporary Update. Surv. Ophthalmol. 2025, 70, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Yu, J.; Liao, S.-H.; Duan, X.-C. Biomechanics of the Sclera and Effects on Intraocular Pressure. Int. J. Ophthalmol. 2016, 9, 1824–1831. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.J.; Woo, S.J.; Park, K.H.; Lee, B.R. Retinal Pigment Epithelium Sequelae Caused by Blunt Ocular Trauma: Incidence, Visual Outcome, and Associated Factors. Sci. Rep. 2017, 7, 14184. [Google Scholar] [CrossRef]
- Morad, Y.; Wygnansky-Jaffe, T.; Levin, A.V. Retinal Haemorrhage in Abusive Head Trauma. Clin. Exper Ophthalmol. 2010, 38, 514–520. [Google Scholar] [CrossRef]
- Faghihi, H.; Ghassemi, F.; Falavarjani, K.G.; Saeedi Anari, G.; Safizadeh, M.; Shahraki, K. Spontaneous Closure of Traumatic Macular Holes. Can. J. Ophthalmol. 2014, 49, 395–398. [Google Scholar] [CrossRef]
- Zigiotti, G.L.; Cavarretta, S.; Morara, M.; Nam, S.M.; Ranno, S.; Pichi, F.; Lembo, A.; Lupo, S.; Nucci, P.; Meduri, A. Standard Enucleation with Aluminium Oxide Implant (Bioceramic) Covered with Patient’s Sclera. Sci. World J. 2012, 2012, 481584. [Google Scholar] [CrossRef]
- Frisina, R.; Besozzi, G.; Gius, I.; Greggio, A.; De Salvo, G.; Meduri, A. Pole to Pole Surgery in Ocular Trauma: Standardizing Surgical Steps. Ophthalmol. Ther. 2022, 11, 1951–1959. [Google Scholar] [CrossRef]
- Bhatia, K.; Sharma, R. Eye Emergencies. In Emergency Medicine; Elsevier: Amsterdam, The Netherlands, 2013; pp. 209–225.e1. ISBN 978-1-4377-3548-2. [Google Scholar]
- Wang, S.; Li, F.; Jin, S.; Zhang, Y.; Yang, N.; Zhao, J. Biomechanics of Open-Globe Injury: A Review. BioMed. Eng. OnLine 2023, 22, 53. [Google Scholar] [CrossRef] [PubMed]
- Stitzel, J.D.; Duma, S.M.; Cormier, J.M.; Herring, I.P. A Nonlinear Finite Element Model of the Eye with Experimental Validation for the Prediction of Globe Rupture. Stapp. Car. Crash J. 2002, 46, 81–102. [Google Scholar] [CrossRef] [PubMed]
- Lam, M.R.; Colmenarez, J.A.; Dong, P.; Gu, L.; Suh, D.W. Vascular Insult in Neonatal Retinal Hemorrhage: Computational Analysis of a Fundus-Segmented Blood Vessel Network. Sci. Rep. 2024, 14, 29331. [Google Scholar] [CrossRef]
- Hoogewoud, F.; Chronopoulos, A.; Varga, Z.; Souteyrand, G.; Thumann, G.; Schutz, J.S. Traumatic Retinal Detachment—The Difficulty and Importance of Correct Diagnosis. Surv. Ophthalmol. 2016, 61, 156–163. [Google Scholar] [CrossRef]
- Mishra, A.; Bhirud, A.; Agrawal, M.; Tripathi, A.; Baranwal, V.K.; Kapoor, G. A Prospective Study to Evaluate the Effectiveness of Preventive Aspects in Relation to Sports Related Ocular Injuries. Int. Ophthalmol. 2024, 44, 436. [Google Scholar] [CrossRef]
- Mazarelo, J.F.D.; Winter, S.L.; Fong, D.T.P. A Systematic Review on the Effectiveness of Eyewear in Reducing the Incidence and Severity of Eye Injuries in Racket Sports. Physician Sportsmed. 2024, 52, 115–124. [Google Scholar] [CrossRef]
- Cohen, L.M.; Habib, L.A.; Yoon, M.K. Post-Traumatic Enophthalmos Secondary to Orbital Fat Atrophy: A Volumetric Analysis. Orbit 2020, 39, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Jafari, S.; Hollister, J.; Kavehpour, P.; Demer, J.L. Shear Viscoelastic Properties of Human Orbital Fat. J. Biomech. 2024, 177, 112416. [Google Scholar] [CrossRef] [PubMed]
- Evans, L.P.; Newell, E.A.; Mahajan, M.; Tsang, S.H.; Ferguson, P.J.; Mahoney, J.; Hue, C.D.; Vogel, E.W.; Morrison, B.; Arancio, O.; et al. Acute Vitreoretinal Trauma and Inflammation after Traumatic Brain Injury in Mice. Ann. Clin. Transl. Neurol. 2018, 5, 240–251. [Google Scholar] [CrossRef] [PubMed]
- Karimi, A.; Razaghi, R.; Sera, T.; Kudo, S. A Combination of the Finite Element Analysis and Experimental Indentation via the Cornea. J. Mech. Behav. Biomed. Mater. 2019, 90, 146–154. [Google Scholar] [CrossRef] [PubMed]




| Structure | Mesh Number | Material Model | Material Parameters |
|---|---|---|---|
| Cornea [21] | 15,265 | Neo-Hookean hyperelastic | µ = 0.079 MPa, D1 = 1 × 10−7 MPa |
| Sclera [22] | 144,570 | Yeoh’s 3rd hyperelastic | C10 = 0.91 MPa, C20 = 19.02 MPa; C30 = −64.73 MPa |
| Lens [22] | 29,843 | Neo-Hookean hyperelastic | µ = 2.29 MPa, D1 = 1 × 10−7 MPa |
| Retina [23] | 244,564 | Neo-Hookean hyperelastic | µ = 0.005 MPa, D1 = 1 × 10−7 MPa |
| Zonules [24] | 17,559 | Linear elastic | E = 0.35 MPa, ν = 0.47 |
| Aqueous [12] | Eulerian | Shock EOS linear | C1 = 1.64 × 106 mm∙s−1, S1 = 1.92 |
| Vitreous [12] | Eulerian | Shock EOS linear | C1 = 1.64 × 106 mm∙s−1, S1 = 1.92 |
| Region | FEA | In Vitro Experiment |
|---|---|---|
| Posterior pole | Compressive stress during impact; tensile stress during retraction. | Evidence of retinoschisis or detachment observed in photoreceptor layers |
| Peripheral | Compressive stress during impact and retraction | Minimal structural disruption; intact retinal layers |
| Equatorial | Radial stress variation during impact and retraction | Detachment observed in pigment epithelium and photoreceptor layers. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, J.; Lee, J.A.; Zhai, Y.; Shahraki, K.; Dong, P.; Suh, D.W.; Gu, L. Spatiotemporal Mapping of Biomechanical Stress Predicts Region-Specific Retinal Injury in a Murine Model of Blunt Ocular Trauma. Bioengineering 2026, 13, 431. https://doi.org/10.3390/bioengineering13040431
Wang J, Lee JA, Zhai Y, Shahraki K, Dong P, Suh DW, Gu L. Spatiotemporal Mapping of Biomechanical Stress Predicts Region-Specific Retinal Injury in a Murine Model of Blunt Ocular Trauma. Bioengineering. 2026; 13(4):431. https://doi.org/10.3390/bioengineering13040431
Chicago/Turabian StyleWang, Jianing, Ji An Lee, Yingnan Zhai, Kourosh Shahraki, Pengfei Dong, Donny W. Suh, and Linxia Gu. 2026. "Spatiotemporal Mapping of Biomechanical Stress Predicts Region-Specific Retinal Injury in a Murine Model of Blunt Ocular Trauma" Bioengineering 13, no. 4: 431. https://doi.org/10.3390/bioengineering13040431
APA StyleWang, J., Lee, J. A., Zhai, Y., Shahraki, K., Dong, P., Suh, D. W., & Gu, L. (2026). Spatiotemporal Mapping of Biomechanical Stress Predicts Region-Specific Retinal Injury in a Murine Model of Blunt Ocular Trauma. Bioengineering, 13(4), 431. https://doi.org/10.3390/bioengineering13040431








