1. Introduction
Ventricular septal defect (VSD) is among the most common congenital heart diseases and remains a major cause of morbidity in infants and children [
1,
2]. Large defects that do not close spontaneously can increase left-to-right shunting, elevate pulmonary blood flow and pressure, and, in severe cases, progress to irreversible pulmonary hypertension and heart failure [
2]. For defects that require intervention, device closure has become a key option because it can avoid open surgery and shorten recovery time.
Despite broad clinical adoption, VSD occluders must simultaneously achieve secure anchoring, reliable sealing, and long-term mechanical compatibility with the beating myocardium. Computational and experimental studies have shown that occluder performance is highly sensitive to membrane stiffness, thickness, and support architecture, and that excessive deformation or localized strain can compromise sealing stability and safety [
3]. Large clinical series have demonstrated the effectiveness of transcatheter closure for muscular and perimembranous VSDs, but device–tissue interaction remains a critical design constraint [
4,
5]. In particular, late complications such as conduction disturbance and complete atrioventricular block have been reported after transcatheter closure, motivating efforts to reduce chronic irritation and improve compliance [
6,
7,
8].
Most commercial VSD occluders still rely on permanent nitinol wire meshes to provide radial support and shape stability. That design works well clinically, but the metal frame remains in the heart long after endothelialization and may contribute to chronic mechanical mismatch and late complications. To address this issue, partially or fully degradable occluders have been explored using polymeric frameworks and membranes, with encouraging preclinical and early clinical results [
9,
10,
11,
12]. Even so, many polymer-based designs still face a practical mechanical problem: they are either too compliant to resist pressure-driven bulging or require a separate stiff support that partly defeats the goal of reducing mismatch.
Biodegradable shape-memory polymers offer an attractive route to reconcile compliance with deployability. Poly(glycerol dodecanedioate) (PGD) is a soft, biodegradable polyester elastomer whose mechanical behavior is closer to that of soft tissue than metal-based devices, and its properties can be tuned through synthesis and curing conditions [
13,
14,
15,
16,
17]. As a thermally activated shape-memory material, PGD can be programmed into a compact temporary shape at lower temperature and recover near physiological temperature, enabling catheter-based delivery and in situ deployment [
18,
19,
20]. However, PGD alone can be overly compliant and prone to pressure-driven bulging, indicating that internal reinforcement is needed to improve structural stability without sacrificing tissue compliance.
Poly(vinyl alcohol) (PVA) is widely used in biomedical applications because of its favorable biocompatibility, processability, and established use in hydrogel and scaffold systems [
21,
22,
23]. Here we combine PGD with a 3D-printed PVA double-layer grid as an internal support, aiming to create an integrated composite membrane that preserves near-body-temperature shape recovery while improving bulge resistance and reducing strain localization.
In this study, we fabricated co-cured PGD–PVA composite membranes with tunable grid spacing (0.1–0.5 mm). We characterized their chemical structure, thermal transition, and temperature-triggered shape-memory behavior, and measured tensile properties and swelling in water at 37 ± 1 °C. We then used finite-element analysis and an in vitro occlusion test to compare deformation and bulge resistance, followed by cytocompatibility and hemocompatibility assays. The aim was not to present a finished occluder, but to test whether this material combination could strengthen a compliant polymer membrane while retaining near-body-temperature recovery.
2. Materials and Methods
2.1. Preparation of PGD–PVA Composite Membranes
PGD prepolymer synthesis: dodecanedioic acid and glycerol were mixed at a 1:1 molar ratio in a three-neck flask. The reaction was carried out at 120 °C under nitrogen and then continued under vacuum (−0.08 MPa). Total reaction time was 24 h to obtain a PGD prepolymer.
3D printing of PVA grids: PVA filament (ePVA+, eSUN) was printed using a desktop 3D printer (Adventurer 4, Zhejiang Flashforge 3D Technology Co., Ltd., Hangzhou, China). The printing step was performed in a standard laboratory environment for research-scale material fabrication rather than in a certified medical-device cleanroom. Settings were: nozzle 200 °C, bed 55 °C, nozzle diameter 0.4 mm, and layer height 0.2 mm. A double-layer grid (40 mm × 40 mm × 0.4 mm) was printed. Grid spacing was set to 0.1–0.5 mm in both directions.
Composite formation: PGD prepolymer was melted at 120 °C and poured into a PTFE dish. The printed grid was placed into the melt and vacuum was applied in a vacuum oven (DP310C, Yamato Scientific Co., Ltd., Tokyo, Japan) to improve infiltration. Samples were cured at 120 °C for 3 days to obtain PGD–PVA composite membranes. Final membrane thickness was 0.5, 1.0, or 1.5 mm. For the composite design used in the present study, the printed PVA grid was laser-cut to the target geometry before embedding, and the PGD matrix was prepared with a slightly larger lateral size so that the cut PVA edge remained fully encapsulated after curing.
Naming: composites are referred to as PGD–PVA. Samples are named PVA-0.1 to PVA-0.5, where the number indicates grid spacing (mm). Unless stated, “PVA-x” refers to the corresponding PGD–PVA composite.
2.2. Fourier Transform Infrared Spectroscopy (FTIR)
FTIR (7600, Lambda Scientific Systems, Inc., Miami, FL, USA) was used to characterize PGD and PGD–PVA. Samples were ground with KBr and pressed into pellets. Spectra were collected from 4000 to 400 cm−1. FTIR was used as a qualitative structural characterization; because no strict replicate number was predefined for this assay, representative spectra are shown for each group.
2.3. Differential Scanning Calorimetry (DSC)
DSC (DSC 8000, PerkinElmer, Inc., Waltham, MA, USA)) was used to measure glass transition temperature (Tg). Samples were cooled from 25 °C to 0 °C at 10 °C/min, held for 3 min, then heated to 60 °C at 10 °C/min. Tg was taken from the heating scan (n = 5).
2.4. Temperature-Triggered Shape-Memory Test
Rectangular strips (30 mm × 15 mm) were cut from PGD and PGD–PVA. Samples were heated to 37 °C and bent in a U-shaped mold, then cooled to room temperature and removed. The fixed angle was recorded as θf. Samples were reheated to 37 °C until stable and the recovered angle was recorded as θr. Angles were measured using ImageJ (version 1.53c, National Institutes of Health, Bethesda, MD, USA) (three repeats per group).
Shape fixity (Rf) and shape recovery (Rr) were calculated as: Rf = (θf/θi) × 100% and Rr = [(θf − θr)/(θf − θ0)] × 100%, where θi is the mold angle during programming and θ0 is the original angle before programming (flat state).
2.5. Tensile Testing
Dog-bone specimens were cut by laser (VLS2.30, Universal Laser Systems, Inc., Scottsdale, AZ, USA) from PGD and PGD–PVA (overall length 115 mm; gauge width 6 mm; thickness matched each sample). Uniaxial tensile tests were performed in water at 37 ± 1 °C using a mechanical test system (IPBF-300S, Tianjin Kaier, Tianjin, China). Specimens were pre-soaked in 37 °C water for 24 h. Crosshead speed was 10 mm/min. Elastic modulus and fracture strength were calculated (n = 10).
2.6. Swelling Ratio
Samples were soaked in deionized water at 37 °C for 48 h (n = 3 per group). Wet mass (mw) was measured after gently removing surface water. Samples were then dried to constant mass (md). Swelling ratio was calculated as (mw − md)/md × 100%.
2.7. In Vitro Occlusion Performance Test
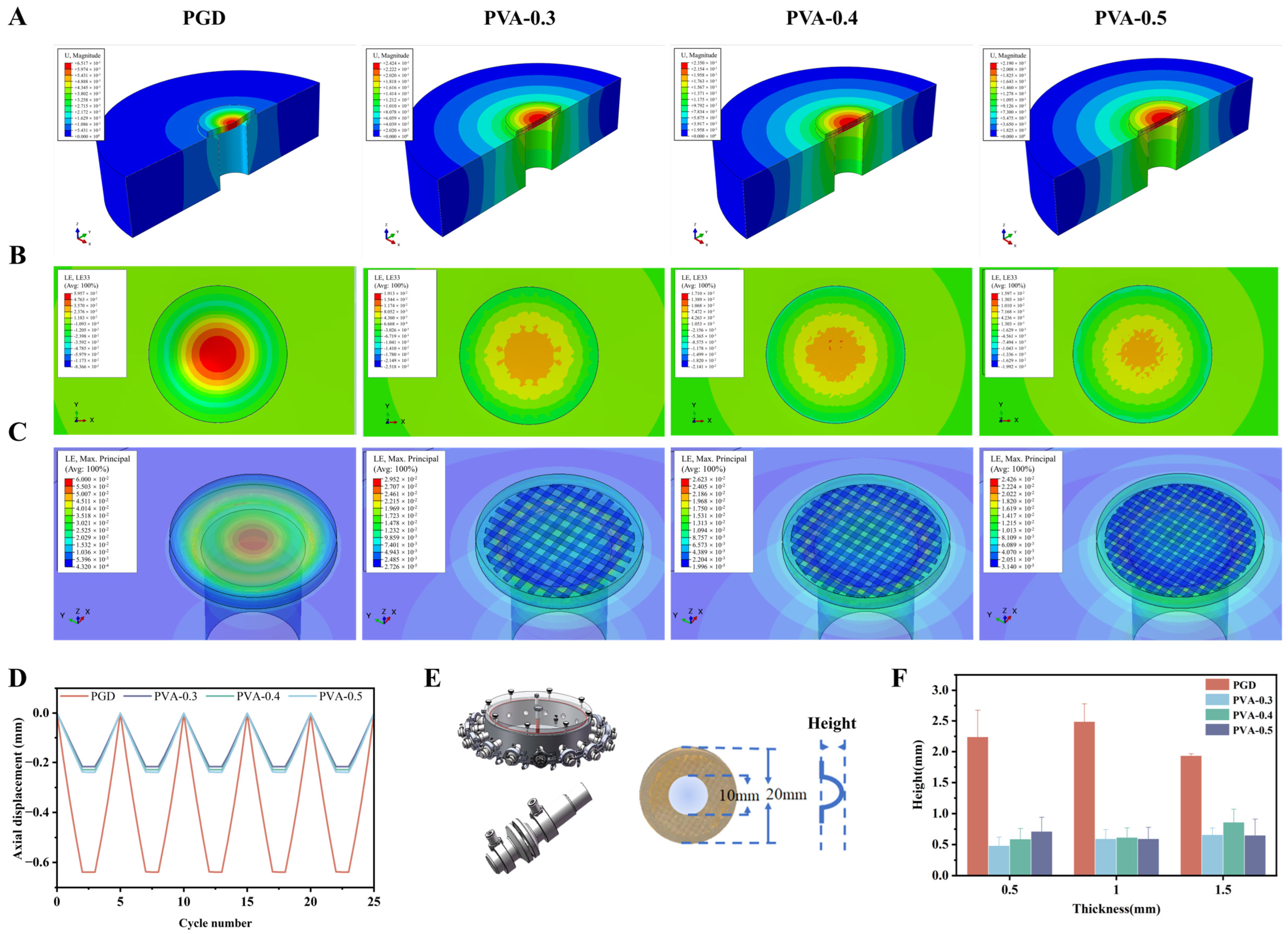
An in vitro occlusion setup was built. It included pump-driven loading, temperature control, and pressure monitoring. Phosphate-buffered saline (PBS; Beijing Solarbio Science & Technology Co., Ltd., Beijing, China) was driven to impact the sample from the defect opening, creating one-sided pulsatile pressure at 37 ± 1 °C. The defect opening diameter was 10 mm, and the effective clamping diameter was 20 mm.
Samples were fixed on the device and the system was filled with PBS and degassed. After reaching 37 °C, the pressure difference across the sample was set to 200 mmHg at 8 Hz for 10 min. The pump was then stopped while static pressure of 200 mmHg was maintained. While maintaining 200 mmHg, the setup was cooled to below 20 °C to fix the deformed shape. The sample was then removed and the maximum out-of-plane bulge height was measured. Each group was tested three times. This benchtop protocol was designed as a comparative short-term screen of deformation resistance rather than a long-term durability test.
2.8. Finite-Element Analysis (FEA)
FEA was performed in Abaqus 2020 to compare relative deformation and strain distribution between PGD and PGD–PVA. A tissue disk (40 mm diameter, 10 mm thickness) with a central defect (6 mm diameter) was used. The outer tissue boundary was fully constrained to provide a simplified reference boundary condition for comparing membrane designs, rather than to reproduce full physiologic myocardial motion. The PGD–PVA membrane was modeled as a cylinder (10 mm diameter, 1 mm height) covering the defect.
Material models: PGD was modeled with a one-term Ogden hyperelastic model (μ = 0.30 ± 0.12 MPa, α = 2.14 ± 0.50, D1 = 0.1333). In the present work, this parameter set was used as an effective material representation for comparative simulations of PGD deformation at 37 °C. The PVA grid was modeled as isotropic linear elastic (E = 2 × 10
9 Pa, ν = 0.46, ρ = 1230 kg/m
3) as a first-order representation of the printed reinforcement. The tissue was modeled as an incompressible Mooney–Rivlin hyperelastic material using parameters reported by Jernigan et al. [
24] (c10 = −5.84 × 10
4 Pa, c01 = 6.34 × 10
4 Pa, c20 = 1.60 × 10
7 Pa, c11 = −3.53 × 10
7 Pa, c02 = 1.97 × 10
7 Pa).
Loading: uniform pressure was applied with a cosine profile. Pressure increased from 0 to 8 kPa in 2 s, was held for 1 s, and then decreased to 0 in 2 s. After unloading, the model ran for another 2 s to capture rebound. The cycle was repeated for 25 cycles. Outputs included maximum principal strain and displacement fields, and axial displacement over cycles.
Note: The FEA geometry and loading were used for qualitative trend comparison rather than one-to-one quantitative prediction. In vitro occlusion testing followed the device geometry and loading described in
Section 2.7, so FEA and experiment were compared at the level of deformation trend rather than direct numerical equivalence.
2.9. Hemolysis Test
PGD and PGD–PVA (1 mm thick) were rinsed three times with ethanol and three times with PBS, then dried. Fresh anticoagulated rabbit blood (anticoagulant: blood = 1:9, v/v) was diluted with 0.9% saline at 4:5 (v/v). Samples were incubated in saline at 3 cm2/mL. Saline and distilled water served as negative and positive controls. After pre-incubation at 37 °C for 30 min, diluted blood (0.2 mL) was added to 10 mL saline and incubated at 37 °C for 1 h. After centrifugation in a high-speed centrifuge (ST8, Thermo Fisher Scientific, Waltham, MA, USA) at 3000 rpm for 5 min, the supernatant absorbance at 545 nm was measured using a microplate reader (Multiskan FC, Thermo Fisher Scientific, Waltham, MA, USA). Hemolysis was calculated as [(optical density (OD)sample − ODnegative)/(ODpositive − ODnegative)] × 100%. Each group was tested three times.
2.10. Plasma Recalcification Time
Platelet-poor plasma (PPP) was collected by sequential centrifugation of diluted anticoagulated rabbit blood using a high-speed centrifuge (ST8, Thermo Fisher Scientific, Waltham, MA, USA) (5 °C, 1000 rpm, 5 min; then 5 °C, 3000 rpm, 10 min). Washed and dried samples were ground into particles and added to 0.025 mol/L CaCl2 at 20% (w/v). CaCl2 in a siliconized tube served as a negative control, and CaCl2 in a regular glass tube served as a positive control. After incubating 100 μL CaCl2 solution at 37 °C for 30 min, 100 μL PPP was added. Starting 100 s after mixing, tubes were gently tilted every 2 s until clotting was observed. Clotting time was recorded (n = 3) and reported as a time ratio versus control.
2.11. Cytocompatibility
Sterilization: PGD and PGD–PVA (1 mm thick) were rinsed with ethanol (three times) and water (three times), dried, and exposed to UV light for 60 min. Extracts were prepared by soaking samples in human umbilical vein endothelial cell (HUVEC; Procell Life Science & Technology Co., Ltd., Wuhan, China) medium at 37 °C for 1, 3, or 5 days at 3 cm2/mL.
CCK-8 assay: HUVECs were seeded in 96-well plates at 3000 cells/well and cultured for 24 h. Medium was replaced with 100 μL extract (test) or fresh medium (control) (n = 5). Cells were cultured with extracts for 1 or 3 days. Then, 10 μL CCK-8 reagent was added and incubated for 1 h in the dark. Absorbance at 450 nm was measured using a microplate reader (Multiskan FC, Thermo Fisher Scientific).
Live/dead staining: HUVECs were seeded on sample surfaces at 1 × 106 cells/sample and cultured for 48 h. Acridine orange/propidium iodide (AO/PI) staining was performed following the kit instructions. Images were acquired with the same channels and exposure settings for all groups.
2.12. Statistics
Data are presented as mean ± standard deviation unless otherwise noted. Here, n denotes the number of independently tested specimens or biological replicates for each quantitative assay; for qualitative characterizations such as FTIR, representative data are shown instead of reporting a formal replicate number. Normality was tested with Shapiro–Wilk. One-way ANOVA with post hoc comparisons was used for normal data with equal variance; otherwise, the Kruskal–Wallis test was used. p < 0.05 was considered significant.
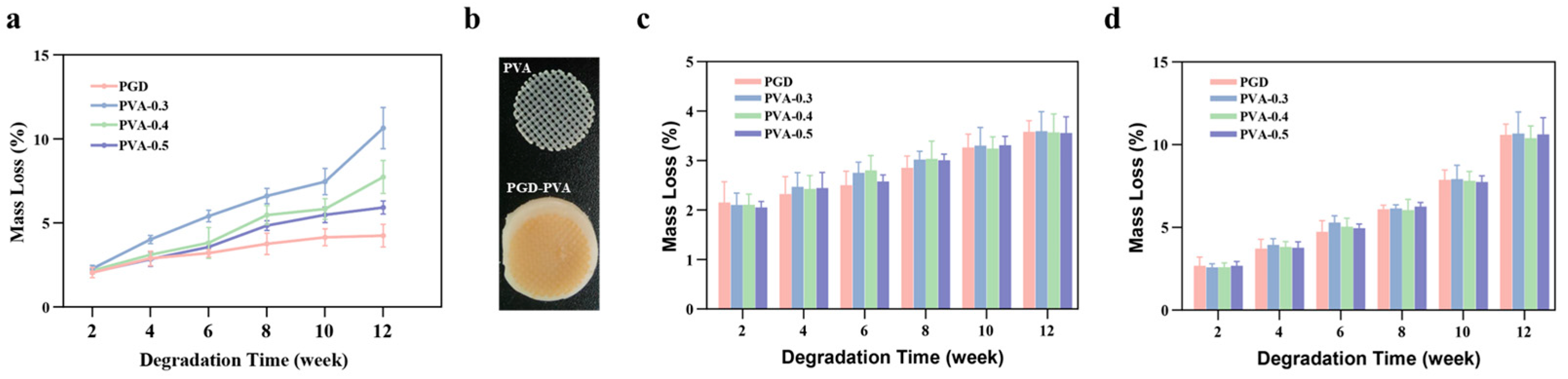
2.13. Pilot Degradation Study
A 12-week pilot degradation study was performed to determine whether the PVA scaffold changed mass-loss behavior and, more specifically, whether edge exposure was responsible for the faster degradation seen in the initial specimens. Two fabrication routes were compared. In the initial route, PGD–PVA composites were cured first and then laser-cut to final geometry, which left PVA cross-sections exposed at the specimen edge. In the revised route, the PVA grid was laser-cut to the target geometry before embedding, and the PGD matrix was prepared with a slightly larger lateral size so that the cut PVA edge was fully encapsulated after curing.
For degradation testing, PGD, PVA-0.3, PVA-0.4, and PVA-0.5 samples were cut with a CO2 laser into rectangular blocks (5 × 4 × 1 mm), weighed to obtain the initial mass, and immersed in PBS (n = 5 per group). Samples were maintained at 37 °C either under static immersion or on a shaker at 200 rpm. At 2, 4, 6, 8, 10, and 12 weeks, specimens were removed, rinsed with deionized water to remove residual deposits and medium, dried in a fume hood for at least 48 h, and reweighed. Mass loss was calculated as [(m0 − mt)/m0] × 100%. The shaking condition was used as a harsher aqueous screening condition, not as a direct physiological pressure-equivalence test.
4. Discussion
Ventricular Septal Defect (VSD) is a prevalent congenital heart anomaly that can severely impair physiological development and quality of life in infants [
1,
2]. Transcatheter closure using minimally invasive occluders is now widely practiced, with strong clinical evidence for both muscular and perimembranous defects [
4,
5]. Current commercial devices generally depend on permanent nitinol frameworks to maintain radial support and device geometry, but excessive stiffness and chronic device–tissue contact can contribute to conduction disturbance and late complications such as complete atrioventricular block [
5,
6,
7,
8]. These concerns have accelerated interest in degradable or polymer-dominant occluders that reduce permanent foreign material while maintaining adequate acute support [
9,
10,
11,
12]. Within that context, the present study focuses on the membrane material itself rather than a complete clinical device system, and therefore does not yet address anchoring, delivery-system compatibility, retrieval capability, or patient-specific interaction with septal anatomy.
FTIR did not show evidence of new covalent bonding between PGD and PVA. We therefore interpret the PGD–PVA interface mainly as physical interlocking of the printed grid within the cured PGD matrix, together with interfacial contact/friction. That appears sufficient for the short-term composite behavior reported here, but it does not resolve long-term resistance to interfacial damage under cyclic loading. The increase in Tg with membrane thickness is most plausibly related to thickness-dependent curing and network development in the PGD phase; local restriction of chain mobility by the embedded PVA may also contribute. Because DSC was performed on cured membranes of different thicknesses rather than on samples with systematically varied chemistry, this interpretation should be read as qualitative. Still, locating Tg near body temperature is advantageous for programming and recovery during catheter-based handling [
18,
19,
20].
Shape recovery remained dominated by the PGD phase. Once the temperature exceeded Tg, PGD provided the recovery force, whereas the PVA grid acted mainly as a passive reinforcement. This likely explains why changing grid density had only a modest effect on recovery under the present conditions, provided that PGD fully encapsulated the scaffold.
Under tensile loading in water at 37 °C, the PVA grid clearly reinforced the membrane, raising both modulus and fracture strength. The quantitative results are now reported directly as mean ± SD from ten specimens per group in
Table 1 and
Figure 2g. This is relevant because a membrane occluder must remain compliant while still resisting pressure-driven bulging [
3,
25,
26,
27,
28,
29,
30]. The trade-off is increased water uptake: the highest-swelling group reached ~18%, which could alter membrane thickness, local fit, or contact pressure in vivo if not controlled. In the present short-term occlusion test, however, this increase in swelling did not cancel the reinforcing effect, because PGD–PVA still showed markedly lower bulge height than PGD alone. Whether repeated swelling and cyclic deformation will promote debonding or sealing instability remains to be tested directly.
The finite-element model was used here as a comparative tool rather than a patient-specific predictor. The outer tissue boundary was fully constrained to provide a simple reference condition, and the PVA grid was modeled as a linear elastic support. Together with the absence of fluid–structure interaction and myocardial anisotropy, these assumptions limit the quantitative predictive value of the model. Even so, the simulations captured the same design trend observed experimentally: internal reinforcement reduced displacement and limited deformation localization.
Durability remains a major open question. A cardiac occluder experiences tens of millions of loading cycles each year. In the degradable occluder literature, this issue is usually addressed either by dedicated fatigue testing of the complete device or by longer implantation/follow-up studies focused on fixation, tissue ingrowth, and gradual absorption [
9,
11,
12]. Our study does not yet reach that stage. The 200 mmHg/8 Hz/10 min pulsatile test and the limited-cycle simulation were used as acute comparative screens for bulging and strain localization, not as lifetime qualification tests. We now state this boundary explicitly. A logical next step is accelerated fatigue testing in a 37 °C aqueous loop while tracking bulge height, stiffness, crack initiation, and damage at the PGD–PVA interface. Because FTIR points to physical interlocking rather than covalent coupling, future interface optimization may also require surface treatment of the PVA grid or other means of improving adhesion.
PGD is a hydrolytically degradable polyester elastomer, and prior work suggests mainly surface erosion with load-dependent modulation of mass loss [
16,
17,
31]. The pilot degradation study clarified one point that was unresolved in the original submission: the faster mass loss seen in the first PGD–PVA samples was largely an edge effect. When the composite was cut after curing, PVA cross-sections were exposed and the PVA-rich groups degraded faster under static immersion. In contrast,
Figure 5b shows the revised route, in which the PVA scaffold was trimmed to final geometry before composite formation and then fully covered by a slightly larger PGD matrix. After this change, the static degradation curves of PGD and PGD–PVA became very similar over 12 weeks, and under PBS at 37 °C with slow agitation at 200 rpm the fully encapsulated groups still remained close to one another.
Mass loss alone does not prove retention of mechanical support, but the present result is not obviously at odds with how degradable septal occluders are typically designed. Guo et al. reported a fully degradable VSD occluder intended to maintain robust fixation during the first 3 months and to disappear within 12 months [
11]. Li et al. reported a PDO-based VSD occluder in which endothelialization outpaced degradation [
12]. Against that background, our 12-week result argues against premature disintegration after full encapsulation, while also making clear what is still missing: time-resolved testing that links mass loss to retained stiffness, cyclic durability, and tissue repair.
At the device level, future work will have to address disk geometry, anchoring, delivery through a sheath, and interaction with different septal anatomies. Imaging-based planning and computational modeling tools are already being used in structural heart interventions to guide sizing and placement [
32]. In this context, patient-specific anatomical reconstruction combined with computational simulation could enable virtual evaluation of occluder deployment, including deformation, contact behavior, and interaction with surrounding tissues. Such a framework may support the optimization of the present membrane design by linking material properties and structural configuration to device-level performance under realistic anatomical and loading conditions, once the membrane concept is extended to a complete occluder geometry.
Finally, implantable cardiovascular devices require rigorous biological safety. Our hemolysis and plasma recalcification tests, together with HUVEC extract assays, indicate favorable initial hemocompatibility and cytocompatibility of PGD–PVA. The observed hemolysis below 1% falls within the nonhemolytic range commonly used for blood-contacting biomaterials, and the maintained endothelial cell viability is consistent with the baseline biocompatibility expected for polymeric occluder candidates [
33,
34,
35,
36,
37,
38]. Although direct cross-study comparison is limited because reported polymer occluder studies use different assay formats and endpoints, this initial profile is directionally consistent with the favorable in vitro biocompatibility generally reported for other polymer-based or bioresorbable VSD occluder candidates [
9,
10,
11,
12]. These assays do not replace long-term in vivo evaluation, but they support the feasibility of PGD–PVA as a candidate membrane platform for next-generation VSD occluders.