Author Contributions
Conceptualization, Q.W.; Methodology, Q.W. and H.G.; Validation, Q.W., L.L., X.X., H.Z. and W.W.; Investigation, L.L.; Resources, S.W.; Writing – original draft, Q.W.; Writing – review & editing, S.W. and H.G.; Visualization, L.L., X.X., H.Z. and H.G.; Supervision, S.W., H.Z., W.W. and H.G.; Funding acquisition, S.W. and H.G. All authors have read and agreed to the published version of the manuscript.
Figure 1.
Schematic diagram of segmentation results for clinical patient liver CT images.
Figure 1.
Schematic diagram of segmentation results for clinical patient liver CT images.
Figure 2.
Schematic diagram of three-dimensional liver simulation models. Figures (A–C) respectively show the segmentation reconstruction results, COMSOL solid simulation model, and mesh generation results for patients 50, 88, and 90 from the LiTS2017 dataset. The numbers on each coordinate axis in the figure (unit: mm) represent the relative position and size range of the liver 3D reconstruction model in space.
Figure 2.
Schematic diagram of three-dimensional liver simulation models. Figures (A–C) respectively show the segmentation reconstruction results, COMSOL solid simulation model, and mesh generation results for patients 50, 88, and 90 from the LiTS2017 dataset. The numbers on each coordinate axis in the figure (unit: mm) represent the relative position and size range of the liver 3D reconstruction model in space.
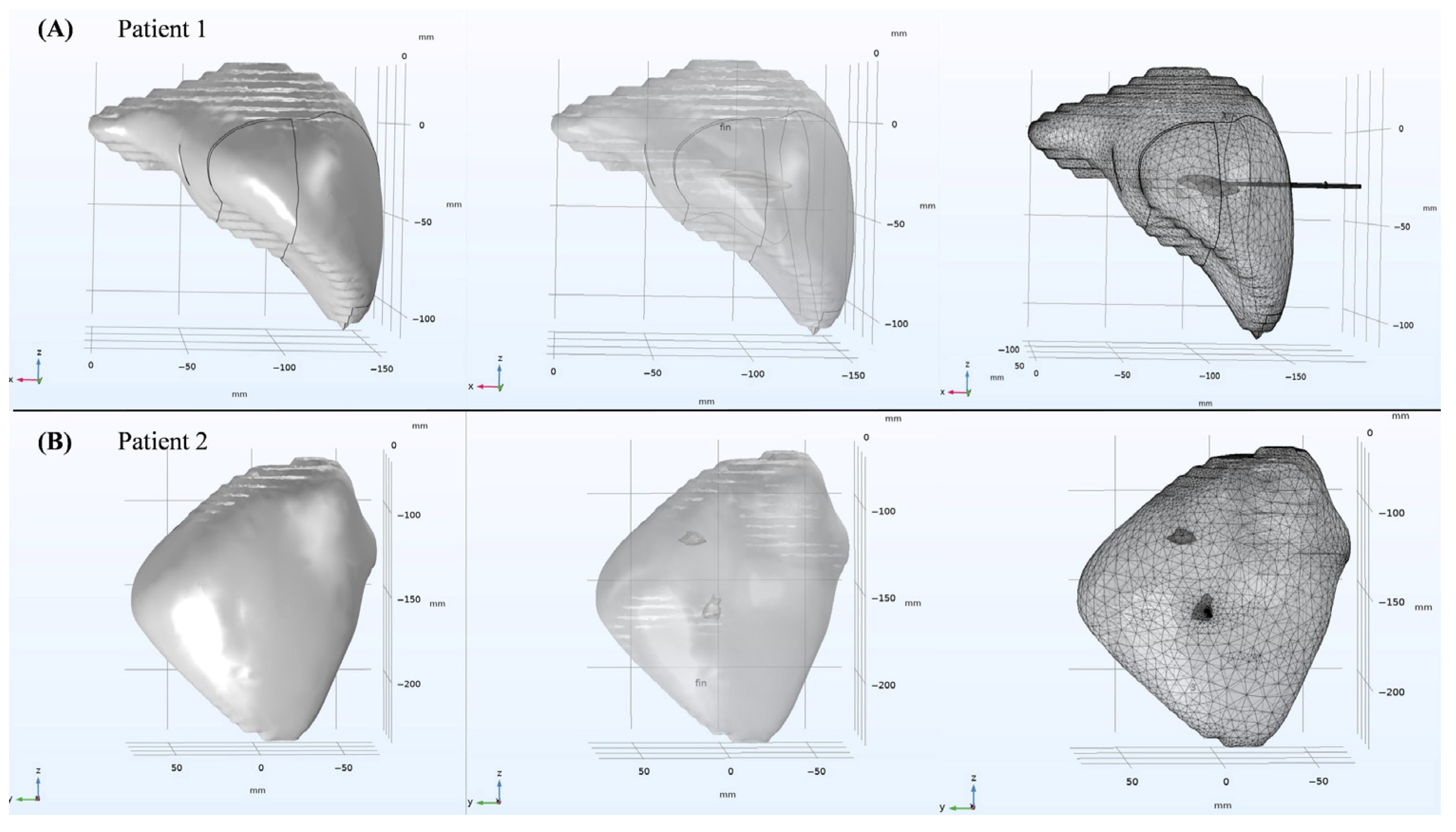
Figure 3.
Schematic diagram of three-dimensional simulation models for clinical patients. Figures (A,B) respectively represent the segmentation reconstruction results, the COMSOL solid simulation model, and the mesh division results for clinical patient 1 and clinical patient 2.
Figure 3.
Schematic diagram of three-dimensional simulation models for clinical patients. Figures (A,B) respectively represent the segmentation reconstruction results, the COMSOL solid simulation model, and the mesh division results for clinical patient 1 and clinical patient 2.
Figure 4.
Schematic diagram of the threshold coagulation zone at the 54 °C isotherm.
Figure 4.
Schematic diagram of the threshold coagulation zone at the 54 °C isotherm.
Figure 5.
Three-dimensional coagulation zones formed under different ablation strategies in the LiTS2017 case. Figures (A–C) show the three-dimensional coagulation zones formed in patients 50, 88, and 90 of the LiTS2017 dataset, respectively, when implementing overlapping MWA and single-needle ablation strategies. Dark green grids indicate tumors, light green grids represent liver tissue, and differently colored ellipses denote coagulation zones formed by varying ablation frequencies. Yellow ellipses indicate coagulation zones generated by the single-needle approach. In the overlapping MWA strategy, the differently colored ellipses (e.g., blue, green, and orange) represent the size of the coagulation zone formed by each ablation, while the combined overlapping region illustrates the cumulative ablation coverage achieved by the multi-needle strategy.
Figure 5.
Three-dimensional coagulation zones formed under different ablation strategies in the LiTS2017 case. Figures (A–C) show the three-dimensional coagulation zones formed in patients 50, 88, and 90 of the LiTS2017 dataset, respectively, when implementing overlapping MWA and single-needle ablation strategies. Dark green grids indicate tumors, light green grids represent liver tissue, and differently colored ellipses denote coagulation zones formed by varying ablation frequencies. Yellow ellipses indicate coagulation zones generated by the single-needle approach. In the overlapping MWA strategy, the differently colored ellipses (e.g., blue, green, and orange) represent the size of the coagulation zone formed by each ablation, while the combined overlapping region illustrates the cumulative ablation coverage achieved by the multi-needle strategy.
Figure 6.
Temperature distribution results generated by the LiTS2017 case under different ablation strategies. Figures (A–C), respectively, depict the two-dimensional coagulation zones formed in patients 50, 88, and 90 from the LiTS2017 dataset when implementing OMWA strategies and single-needle ablation strategies. The colored vertical bars indicate temperature distribution. 54 °C represents the critical temperature for achieving complete tumor necrosis within the coagulation region.
Figure 6.
Temperature distribution results generated by the LiTS2017 case under different ablation strategies. Figures (A–C), respectively, depict the two-dimensional coagulation zones formed in patients 50, 88, and 90 from the LiTS2017 dataset when implementing OMWA strategies and single-needle ablation strategies. The colored vertical bars indicate temperature distribution. 54 °C represents the critical temperature for achieving complete tumor necrosis within the coagulation region.
Figure 7.
Comparison of three-dimensional coagulation zones between overlapping and single-needle MWA in clinical patients. Three-dimensional coagulation zones in clinical data. Figures (A,B) show the sizes of the three-dimensional coagulation zones (indicated by the 54 °C isotherm) for OMWA and single-needle ablation in patient 1 and patient 2, respectively.
Figure 7.
Comparison of three-dimensional coagulation zones between overlapping and single-needle MWA in clinical patients. Three-dimensional coagulation zones in clinical data. Figures (A,B) show the sizes of the three-dimensional coagulation zones (indicated by the 54 °C isotherm) for OMWA and single-needle ablation in patient 1 and patient 2, respectively.
Figure 8.
Two-dimensional coagulation zones and temperature distribution in patients 1 and 2 after single-needle ablation and OMWA. Figures (A–D) represent patient 1, and Figures (E–G) represent patient 2. Figures (A,B,E,F) represent the results of OMWA treatment. Figures (C,D,G,H) represent the results of single-needle treatment. The color bar represents the temperature gradient (°C). 54°C represents the temperature at which tissue reaches necrosis. The 54 °C curve in the figure represents the range of ablation necrosis at each stage.
Figure 8.
Two-dimensional coagulation zones and temperature distribution in patients 1 and 2 after single-needle ablation and OMWA. Figures (A–D) represent patient 1, and Figures (E–G) represent patient 2. Figures (A,B,E,F) represent the results of OMWA treatment. Figures (C,D,G,H) represent the results of single-needle treatment. The color bar represents the temperature gradient (°C). 54°C represents the temperature at which tissue reaches necrosis. The 54 °C curve in the figure represents the range of ablation necrosis at each stage.
Figure 9.
Comparison of tumor CT images before and after clinical surgical treatment. (A–C) shows the preoperative tumor size. (A) represents the CT image of patient 1, and (B,C) represent the CT images of different lesions in patient 2. (D–F) correspond to the immediate postoperative images at the aforementioned locations. The crosshairs in the images mark the long-axis and short-axis (cm) of the lesion and the postoperative coagulation zone. The comparison shows that the postoperative coagulation necrosis zone significantly exceeds the original tumor boundary, and both its long and short axis measurements are greater than the preoperative tumor morphology, confirming that the coagulation zone completely and effectively covered the target tumor. The green crosshairs indicate the measured axes of the lesion or coagulation zone, and the red markers denote the endpoints of the measurements.
Figure 9.
Comparison of tumor CT images before and after clinical surgical treatment. (A–C) shows the preoperative tumor size. (A) represents the CT image of patient 1, and (B,C) represent the CT images of different lesions in patient 2. (D–F) correspond to the immediate postoperative images at the aforementioned locations. The crosshairs in the images mark the long-axis and short-axis (cm) of the lesion and the postoperative coagulation zone. The comparison shows that the postoperative coagulation necrosis zone significantly exceeds the original tumor boundary, and both its long and short axis measurements are greater than the preoperative tumor morphology, confirming that the coagulation zone completely and effectively covered the target tumor. The green crosshairs indicate the measured axes of the lesion or coagulation zone, and the red markers denote the endpoints of the measurements.
Table 1.
Physical characteristics of some cases from the LiTS2017 dataset.
Table 1.
Physical characteristics of some cases from the LiTS2017 dataset.
| Tissue | Liver-50 | Tumor-50 | Liver-88 | Tumor-88 | Liver-90 | Tumor-90 |
|---|
| Volume (mm3) | 713,348.00 | 3077.00 | 1,558,027.20 | 116,660.55 | 1,717,439.97 | 123,061.00 |
| Number | 1 | 1 | 1 | 2 | 1 | 3 |
| Shape | Flat and long | Regular shape | Normal | Giant multiple | Normal | Multiple irregular |
Table 2.
Physical characteristics of some cases from clinical data.
Table 2.
Physical characteristics of some cases from clinical data.
| Tissue | Liver-1 | Tumor-1 | Liver-2 | Tumor-2 |
|---|
| Volume (mm3) | 954,980.00 | 2983.76 | 1,238,650.00 | 2873.64 |
| Number | 1 | 1 | 1 | 2 |
| Shape | Slightly smaller | Irregular | Normal | Multiple and irregular |
Table 3.
Dimensions of the 2450 MHz microwave antenna.
Table 3.
Dimensions of the 2450 MHz microwave antenna.
| Material | Dimension (mm) |
|---|
| Antenna outer diameter | 1.9 |
| Antenna inner diameter | 1.3 |
| Distance from the transmitting point to the antenna tip | 11.5 |
| Distance from the transmitting point to the antenna bottom | 90 |
| Total length | 101.5 |
Table 4.
Tissue electrical and thermal parameters used in the model.
Table 4.
Tissue electrical and thermal parameters used in the model.
| Parameters | Description | Units | Corresponding Formula |
|---|
| (T) | Specific heat capacity | J/(kg·°C) | Equations (4) and (5) |
| Water content | Dimensionless | Equation (5) |
| Tissue density | kg/m3 | |
| Blood perfusion rate | 1/s | = 0.0004 [31] |
| Specific heat capacity of blood | J/(kg·°C) | = 3600 |
| Initial tissue temperature | °C | = 37°C |
| Metabolism | W/m3 | = 33,800 [32] |
| ε(T) | Relative permittivity | Dimensionless | Equation (6) |
| σ(T) | Electrical conductivity | S/m | Equation (7) |
| Thermal conductivity | W/(m·°C) | Equation (8) |
| Relative magnetic permeability | Dimensionless | = 1 |
| Free space wavenumber | 1/m | = |
| Permittivity of vacuum | F/m | = 8.854 |
Table 5.
Tissue parameters of tumors.
Table 5.
Tissue parameters of tumors.
| Tissue | (J/(kg·°C)) | (kg/m3) | (T) | (T) (S/m) | (W/(m·°C)) |
|---|
| Tumor | 3960 | 1040 | Equation (6) | Equation (7) | Equation (8) |
Table 6.
OMWA strategy for different cases.
Table 6.
OMWA strategy for different cases.
| Ablation Strategy | Case 50 (s) | Case 88 (s) | Case 90 (s) | Patient 1 (s) | Patient 2 (s) |
|---|
| First ablation | 80 | 180 | 50 | 30 | 10 |
| Surgical planning | 40 | 40 | 40 | 40 | 10 |
| Second ablation | 30 | 1020 | 660 | 70 | 60 |
| Surgical planning | * | 40 | 40 | * | * |
| Third ablation | * | 1200 | 10 | * | * |
| Surgical planning | * | 40 | * | * | * |
| Fourth ablation | * | 920 | * | * | * |
| Total time | 150 | 3400 | 800 | 140 | 80 |
Table 7.
Morphological characteristics of the coagulation zone in LiTS2017.
Table 7.
Morphological characteristics of the coagulation zone in LiTS2017.
| Morphological Characteristics | Long-Axis (mm) | Short-Axis (mm) |
|---|
| Case 50 | Overlapping | 35.58 | 31.04 |
| Single needle | 44.04 | 31.57 |
| Tumor | 26.79 | 20.73 |
| Case 88 | Overlapping | 75.76 | 71.35 |
| Single needle | 80.78 | 75.25 |
| Tumor | 72.76 | 68.66 |
| Case 90 | Overlapping | 45.06 | 41.64 |
| Single needle | 58.32 | 47.78 |
| Tumor | 40.91 | 37.61 |
Table 8.
Clinical data: two-dimensional coagulation zone size.
Table 8.
Clinical data: two-dimensional coagulation zone size.
| Morphological Characteristics | Long-Axis (mm) | Short-Axis (mm) |
|---|
| Patient1 | Overlapping | 41.28 | 21.75 |
| Single needle | 41.55 | 27.63 |
| Tumor | 38.37 | 18.35 |
| Patient2 | Overlapping | 42.63 | 21.22 |
| Single needle | 45.70 | 32.15 |
| Tumor | 38.91 | 16.32 |
Table 9.
Comparison of simulated ablation performance indicators between OMWA and single-needle strategies.
Table 9.
Comparison of simulated ablation performance indicators between OMWA and single-needle strategies.
| Case | Safety Margin (mm) | Ablation Volume (mm3) | HTDV (mm3) | HTDR (mm) | Tumor Volume (mm3) |
|---|
| Long Axis | Short Axis |
|---|
| Case 50 | Overlapping | 8.79 | 10.31 | 11,941 | 8864 | 0.74 | 3077.00 |
| Single needle | 17.25 | 10.84 | 24,582 | 21,505 | 0.87 | 3077.00 |
| Case 88 | Overlapping | 3.00 | 2.69 | 169,316 | 52,655.45 | 0.31 | 116,660.55 |
| Single needle | 10.16 | 6.59 | 242,558 | 125,897.45 | 0.52 | 116,660.55 |
| Case 90 | Overlapping | 4.15 | 4.03 | 49,563 | 8542.67 | 0.17 | 41,020.33 |
| Single needle | 17.41 | 10.17 | 81,770 | 40,749.67 | 0.50 | 41,020.33 |
| Patient 1 | Overlapping | 3.91 | 3.40 | 9861.9 | 6878.14 | 0.70 | 2983.76 |
| Single needle | 3.18 | 9.28 | 16,215 | 13,231.24 | 0.82 | 2983.76 |
| Patient 2 | Overlapping | 3.72 | 4.90 | 8399 | 5525.36 | 0.66 | 2873.64 |
| Single needle | 6.79 | 15.83 | 23,040 | 20,166.36 | 0.88 | 2873.64 |