Ultrasound-Guided Targeted Injection to the Anterior Labral–Ligamentous Complex of the Shoulder: A Cadaveric Feasibility Study for Regenerative Therapy
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Specimen
2.2. Ultrasound Equipment and Imaging Parameters
2.3. Positioning
2.4. Ultrasound-Guided Targeted Injection
2.5. Anatomical Dissection and Outcome Assessment
2.6. Outcome Definition
- Grossly visible focal localization of red filler within the ALLC inside the joint capsule
- Absence of extra-capsular spread
- Absence of diffuse intra-articular pooling within the glenohumeral joint cavity
- No staining of adjacent neurovascular structures
2.7. Ethics Statement
3. Results
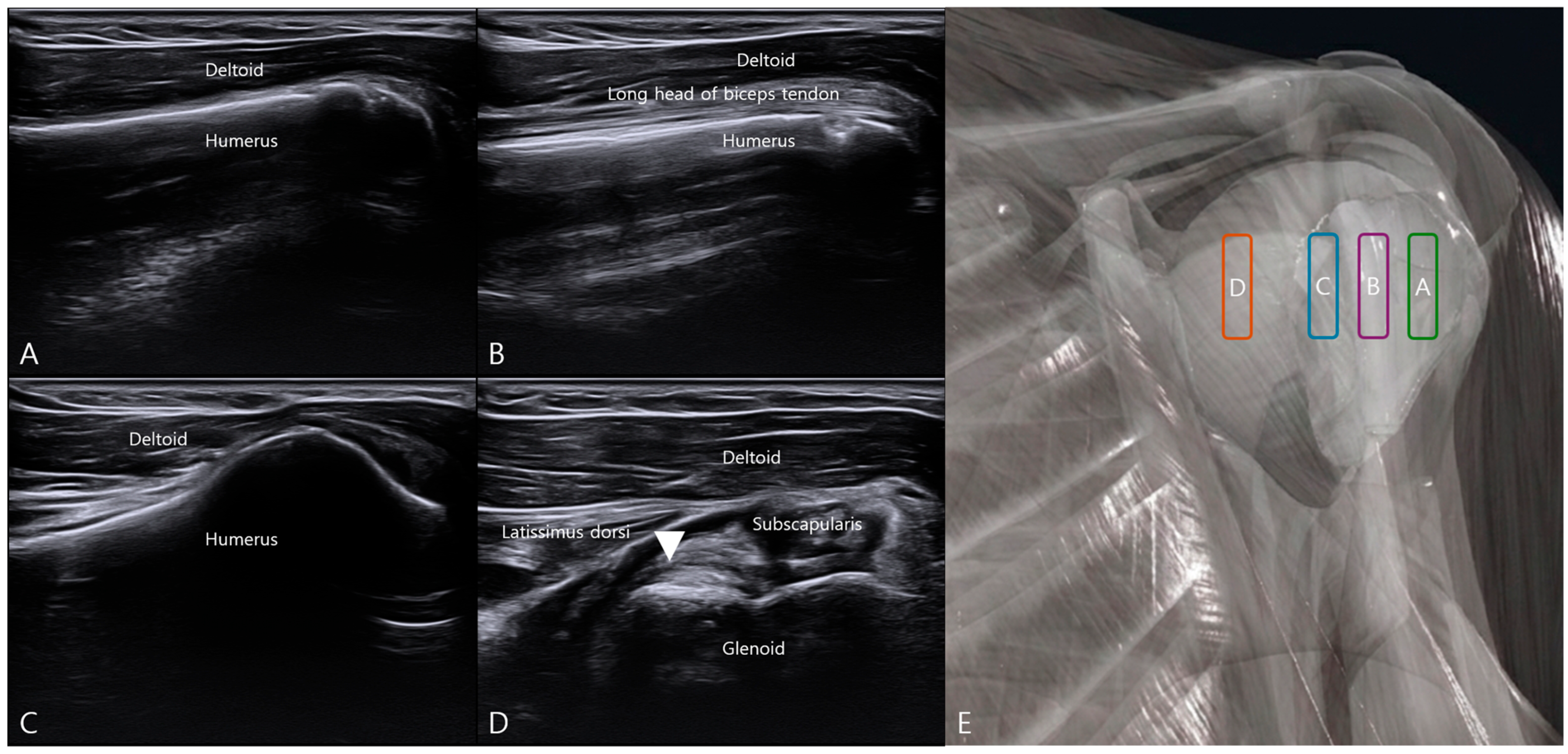
3.1. Ultrasound Landmark Identification and Localization of the ALLC
3.2. Ultrasound-Guided In-Plane Injection
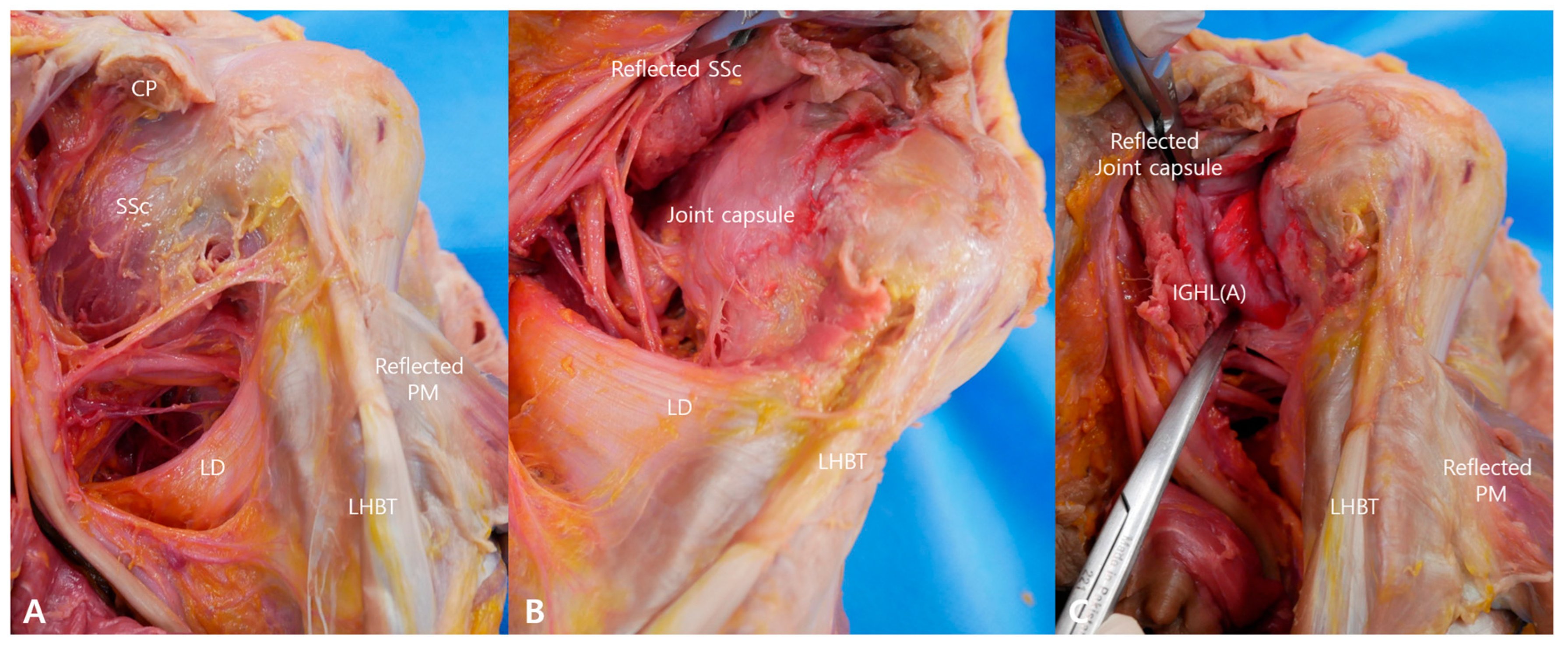
3.3. Cadaveric Dissection Confirmation of Injectate Localization (Bilateral Shoulders)
4. Discussion
4.1. Biomechanical Significance of the Anterior Labral–Ligamentous Complex
4.2. Rationale for Interface-Level Regenerative Delivery
4.3. Technical Considerations
4.4. Role of Ultrasound Guidance and Feasibility of Targeted ALLC Injection
4.5. Clinical Implications
4.6. Limitations
5. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- O’Brien, S.J.; Neves, M.C.; Arnoczky, S.P.; Rozbruck, S.R.; Dicarlo, E.F.; Warren, R.F.; Schwartz, R.; Wickiewicz, T.L. The anatomy and histology of the inferior glenohumeral ligament complex of the shoulder. Am. J. Sports Med. 1990, 18, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Turkel, S.J.; Panio, M.W.; Marshall, J.L.; Girgis, F.G. Stabilizing mechanisms preventing anterior dislocation of the glenohumeral joint. J. Bone Jt. Surg. 1981, 63, 1208–1217. [Google Scholar] [CrossRef]
- Burkart, A.C.; Debski, R.E. Anatomy and function of the glenohumeral ligaments in anterior shoulder instability. Clin. Orthop. Relat. Res. 2002, 400, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Palmer, W.E.; Brown, J.H.; Rosenthal, D.I. Labral–ligamentous complex of the shoulder: Evaluation with MR arthrography. Radiology 1994, 190, 645–651. [Google Scholar] [CrossRef]
- Park, Y.H.; Lee, J.Y.; Moon, S.H.; Mo, J.H.; Yang, B.K.; Hahn, S.H.; Resnick, D. MR Arthrography of the Labral Capsular Ligamentous Complex in the Shoulder. Am. J. Roentgenol. 2000, 175, 667–672. [Google Scholar] [CrossRef]
- Itoigawa, Y.; Itoi, E. Anatomy of the capsulolabral complex and rotator interval related to glenohumeral instability. Knee Surg. Sports Traumatol. Arthrosc. 2016, 24, 343–349. [Google Scholar] [CrossRef]
- Chen, J.; Chen, S.; Li, Y.; Hua, Y.; Li, H. Is the extended release of the inferior glenohumeral ligament necessary for frozen shoulder? Arthroscopy 2010, 26, 529–535. [Google Scholar] [CrossRef]
- Inada, T.; Tsutsumi, M.; Ikezu, M.; Iizuka, Y.; Nagano, T.; Kudo, S. Inferior glenohumeral joint capsule thickness in frozen shoulder via ultrasonography. JSES Int. 2024, 8, 1033–1038. [Google Scholar] [CrossRef]
- Enriquez, R.; Jones, E.; Özçakar, L.; Jain, N.B. Accuracy and Efficacy of Ultrasound-Guided Glenohumeral Joint Injections: A Systematic Review. Am. J. Phys. Med. Rehabil. 2025, 104, 1071–1085. [Google Scholar] [CrossRef]
- Kuratani, K.; Tanaka, M.; Hanai, H.; Hayashida, K. Accuracy of shoulder joint injections with ultrasound guidance: Confirmed by magnetic resonance arthrography. World J. Orthop. 2022, 13, 259–266. [Google Scholar] [CrossRef]
- Krzyżanowski, W. The use of ultrasound in the assessment of the glenoid labrum of the glenohumeral joint. Part I: Ultrasound anatomy and examination technique. J. Ultrason. 2012, 12, 164–177. [Google Scholar]
- Wu, W.-T.; Chang, K.-V.; Özçakar, L. Dynamic Ultrasound Imaging for the Diagnosis of Superior Labrum Anterior to Posterior Lesion. Am. J. Phys. Med. Rehabil. 2019, 98, e130–e131. [Google Scholar] [CrossRef] [PubMed]
- Mitsui, Y.; Funakoshi, T.; Hara, K.; Higuchi, K.; Miyamoto, A.; Nakamura, H.; Gotoh, M. Dynamic Anterior Glenohumeral Capsular Ligament Tensioning During Arthroscopic Shoulder Stabilization in Overhead-Throwing Athletes. Arthrosc. Tech. 2024, 13, 103069. [Google Scholar] [CrossRef]
- Passanante, G.J.; Skalski, M.R.; Patel, D.B.; White, E.A.; Schein, A.J.; Gottsegen, C.J.; Matcuk, G.R. Inferior glenohumeral ligament (IGHL) complex: Anatomy, injuries, imaging features, and treatment options. Emerg. Radiol. 2017, 24, 65–71. [Google Scholar] [CrossRef]
- Goulian, A.J.; Goldstein, B.; Saad, M.A. Advancements in Regenerative Therapies for Orthopedics: A Comprehensive Review of Platelet-Rich Plasma, Mesenchymal Stem Cells, Peptide Therapies, and Biomimetic Applications. J. Clin. Med. 2025, 14, 2061. [Google Scholar] [CrossRef] [PubMed]
- Reeves, K.D.; Sit, R.W.; Rabago, D.P. Dextrose Prolotherapy: A Narrative Review of Basic Science, Clinical Research, and Best Treatment Recommendations. Phys. Med. Rehabil. Clin. 2016, 27, 783–823. [Google Scholar] [CrossRef]
- Rosso, C.; Morrey, M.E.; Schär, M.O.; Grezda, K. The role of platelet-rich plasma in shoulder pathologies: A critical review of the literature. EFORT Open Rev. 2023, 8, 213–222. [Google Scholar] [CrossRef]
- Hauser, R.A.; Lackner, J.B.; Steilen-Matias, D.; Harris, D.K. A Systematic Review of Dextrose Prolotherapy for Chronic Musculoskeletal Pain. Clin. Med. Insights Arthritis Musculoskelet. Disord. 2016, 9, 139–159. [Google Scholar]
- Yoon, Y.; Sung, D.; Lee, J.; Lam, K.H.S. A Case Report of Deep Gluteal Syndrome Secondary to Piriformis Enthesopathy Successfully Treated With Ultrasound-Guided Dextrose Prolotherapy. Cureus 2026, 18, e102370. [Google Scholar] [CrossRef]
- Benjamin, M.; McGonagle, D. Entheses: Tendon and ligament attachment sites. Scand. J. Med. Sci. Sports 2009, 19, 520–527. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, M.; Toumi, H.; Ralphs, J.R.; Bydder, G.; Best, T.M.; Milz, S. Where tendons and ligaments meet bone: Attachment sites (‘entheses’) in relation to exercise and/or mechanical load. J. Anat. 2006, 208, 471–490. [Google Scholar] [CrossRef] [PubMed]
- Apaydin, N.; Uz, A.; Bozkurt, M.; Elhan, A. The anatomic relationships of the axillary nerve and surgical landmarks for its localization from the anterior aspect of the shoulder. Clin. Anat. 2007, 20, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Burkhead, W.Z., Jr.; Scheinberg, R.R.; Box, G. Surgical anatomy of the axillary nerve. J. Shoulder Elb. Surg. 1992, 1, 31–36. [Google Scholar] [CrossRef]
- Stecco, C.; Gagliano, G.; Lancerotto, L.; Tiengo, C.; Macchi, V.; Porzionato, A.; De Caro, R.; Aldegheri, R. Surgical anatomy of the axillary nerve and its implication in the transdeltoid approaches to the shoulder. J. Shoulder Elb. Surg. 2010, 19, 1166–1174. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, J.A. Fundamentals of Musculoskeletal Ultrasound; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Bianchi, S.; Martinoli, C. Ultrasound of the Musculoskeletal System, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kim, S.-H.; Lee, U.-Y.; Yoon, Y.; Hwang, J.; Kim, J.; Na, Y.; Kim, S.; Lam, K.H.S.; de Castro, J.C.; Suryadi, T.; et al. Ultrasound-Guided Targeted Injection to the Anterior Labral–Ligamentous Complex of the Shoulder: A Cadaveric Feasibility Study for Regenerative Therapy. Bioengineering 2026, 13, 418. https://doi.org/10.3390/bioengineering13040418
Kim S-H, Lee U-Y, Yoon Y, Hwang J, Kim J, Na Y, Kim S, Lam KHS, de Castro JC, Suryadi T, et al. Ultrasound-Guided Targeted Injection to the Anterior Labral–Ligamentous Complex of the Shoulder: A Cadaveric Feasibility Study for Regenerative Therapy. Bioengineering. 2026; 13(4):418. https://doi.org/10.3390/bioengineering13040418
Chicago/Turabian StyleKim, Sang-Hyun, U-Young Lee, Yonghyun Yoon, Jihyo Hwang, Jungyoun Kim, Yoonju Na, Seungbeom Kim, King Hei Stanley Lam, Jeimylo C. de Castro, Teinny Suryadi, and et al. 2026. "Ultrasound-Guided Targeted Injection to the Anterior Labral–Ligamentous Complex of the Shoulder: A Cadaveric Feasibility Study for Regenerative Therapy" Bioengineering 13, no. 4: 418. https://doi.org/10.3390/bioengineering13040418
APA StyleKim, S.-H., Lee, U.-Y., Yoon, Y., Hwang, J., Kim, J., Na, Y., Kim, S., Lam, K. H. S., de Castro, J. C., Suryadi, T., & Suhaimi, A. (2026). Ultrasound-Guided Targeted Injection to the Anterior Labral–Ligamentous Complex of the Shoulder: A Cadaveric Feasibility Study for Regenerative Therapy. Bioengineering, 13(4), 418. https://doi.org/10.3390/bioengineering13040418









