HIV-Associated Microstructural Abnormalities in Default Mode, Executive Control, and Salience Networks: Insights from Tensor-Valued Diffusion Encoding
Abstract
1. Introduction
2. Materials and Methods
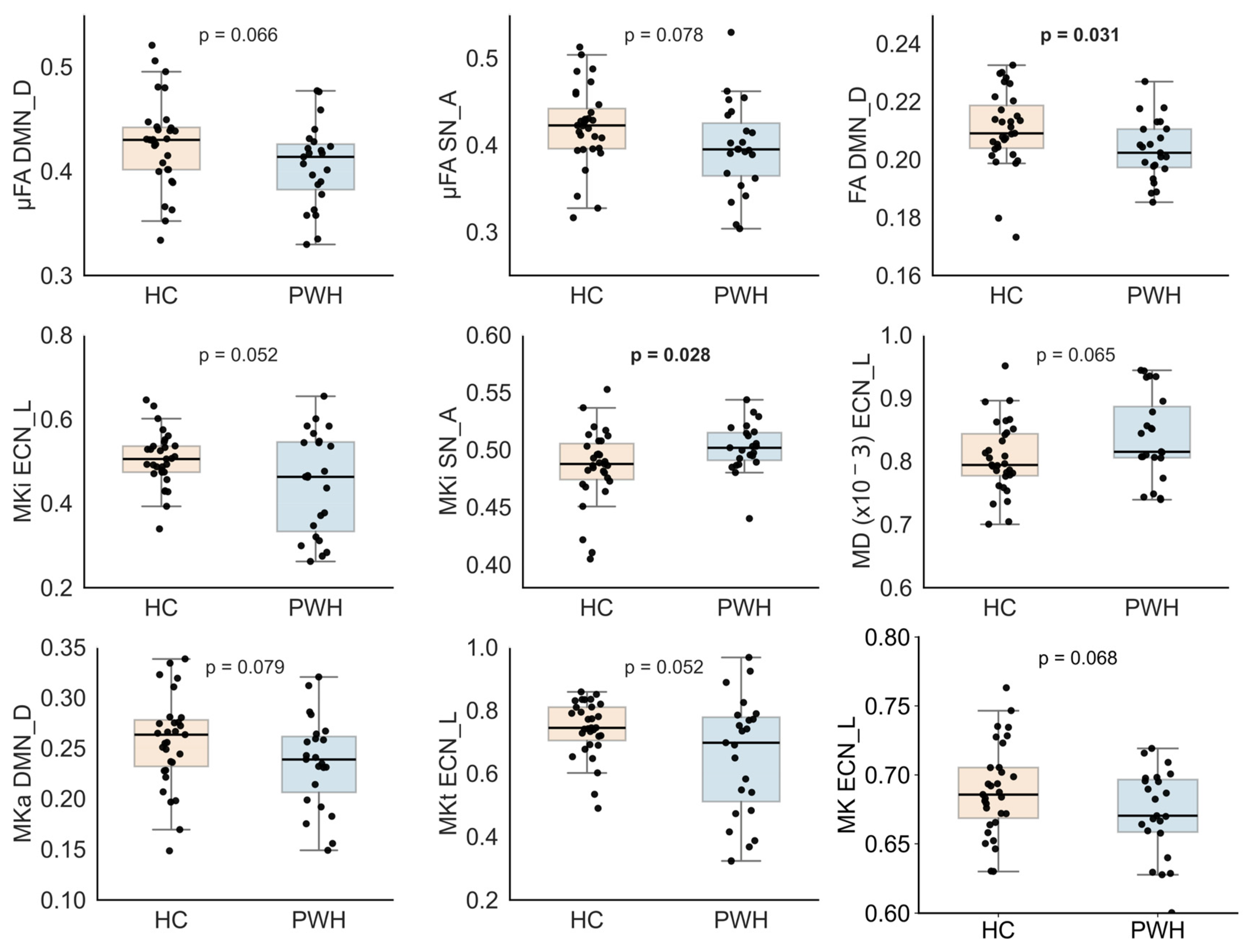
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PWH | People with HIV |
| cART | Combination antiretroviral therapy |
| ARV | Approved antiretroviral |
| NRTI | Nucleoside/Nucleotide Reverse Transcriptase Inhibitor |
| NNRTI | Non-Nucleoside Reverse Transcriptase Inhibitor |
| PI | Protease inhibitor |
| SD | Standard deviation |
| dMRI | Diffusion magnetic resonance imaging |
| DTI | Diffusion tensor imaging |
| FA | Fractional anisotropy |
| MD | Mean diffusivity |
| DKI | Diffusion kurtosis imaging |
| MK | Mean Kurtosis |
| MKa | Anisotropic mean kurtosis |
| MKi | Isotropic mean kurtosis |
| MKt | Total mean kurtosis |
| µFA | microscopic fractional anisotropy |
| ROI | Region of interest |
| RSRB | Institutional Research Subjects Review Board |
| CalCAP | California Computerized Assessment Package |
| T1w | T1-weighted |
| MPRAGE | Magnetization-prepared rapid acquisition gradient-echo |
| TI | Inversion time |
| TE | Echo time |
| TR | Repetition time |
| GRAPPA | GeneRalized Autocalibrating Partial Parallel Acquisition |
| FOV | Field of view |
| DWI | Diffusion weighted imaging |
| FWF | Free waveform |
| STE | Spherical tensor encoding |
| LTE | Linear tensor encoding |
| FSL | FMRIB Software Library |
| ANTs | Advanced Normalization Tools |
| BET | Brain extraction tool |
| FAST | FMRIB’s Automated Segmentation Tool |
| FIRST | FMRIB’s Integrated Registration and Segmentation Tool |
| FLIRT | FMRIB’s Linear Image Registration Tool |
| FNIRT | FMRIB’s Nonlinear Image Registration Tool |
| DMN_D | Dorsal Default Mode Network |
| DMN_V | Ventral Default Mode Network |
| ECN_L | Left Executive Control Network |
| ECN_R | Right Executive Control Network |
| SN_A | Anterior Salience Network |
| SN_P | Posterior Salience Network |
| Cog | Cognitive score |
| OLS | Ordinary least square |
| OVERALL | Total cognitive score |
| LEA | Learning |
| LAN | Language |
| MEM | Memory |
| EXE | Executive function |
| SPE | Processing speed |
| MOT | Motor control |
References
- Woods, S.P.; Moore, D.J.; Weber, E.; Grant, I. Cognitive neuropsychology of HIV-associated neurocognitive disorders. Neuropsychol. Rev. 2009, 19, 152–168. [Google Scholar] [CrossRef]
- Antinori, A.; Arendt, G.; Becker, J.; Brew, B.; Byrd, D.; Cherner, M.; Clifford, D.; Cinque, P.; Epstein, L.; Goodkin, K. Updated research nosology for HIV-associated neurocognitive disorders. Neurology 2007, 69, 1789–1799. [Google Scholar] [CrossRef]
- Heaton, R.; Clifford, D.; Franklin, D.; Woods, S.; Ake, C.; Vaida, F.; Ellis, R.; Letendre, S.; Marcotte, T.; Atkinson, J. HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy: CHARTER Study. Neurology 2010, 75, 2087–2096. [Google Scholar]
- Saylor, D.; Dickens, A.M.; Sacktor, N.; Haughey, N.; Slusher, B.; Pletnikov, M.; Mankowski, J.L.; Brown, A.; Volsky, D.J.; McArthur, J.C. HIV-associated neurocognitive disorder—Pathogenesis and prospects for treatment. Nat. Rev. Neurol. 2016, 12, 234–248. [Google Scholar]
- Hong, S.; Banks, W.A. Role of the immune system in HIV-associated neuroinflammation and neurocognitive implications. Brain Behav. Immun. 2015, 45, 1–12. [Google Scholar]
- Underwood, J.; Robertson, K.R.; Winston, A. Could antiretroviral neurotoxicity play a role in the pathogenesis of cognitive impairment in treated HIV disease? AIDS 2015, 29, 253–261. [Google Scholar]
- Chenevert, T.L.; Stegman, L.D.; Taylor, J.M.; Robertson, P.L.; Greenberg, H.S.; Rehemtulla, A.; Ross, B.D. Diffusion magnetic resonance imaging: An early surrogate marker of therapeutic efficacy in brain tumors. JNCI J. Natl. Cancer Inst. 2000, 92, 2029–2036. [Google Scholar] [CrossRef]
- Horsfield, M.A.; Jones, D.K. Applications of diffusion-weighted and diffusion tensor MRI to white matter diseases—A review. NMR Biomed. Int. J. Devoted Dev. Appl. Magn. Reson. In Vivo 2002, 15, 570–577. [Google Scholar]
- Lebel, C.; Walker, L.; Leemans, A.; Phillips, L.; Beaulieu, C. Microstructural maturation of the human brain from childhood to adulthood. NeuroImage 2008, 40, 1044–1055. [Google Scholar]
- Figley, C.R.; Uddin, M.N.; Wong, K.; Kornelsen, J.; Puig, J.; Figley, T.D. Potential pitfalls of using fractional anisotropy, axial diffusivity, and radial diffusivity as biomarkers of cerebral white matter microstructure. Front. Neurosci. 2022, 15, 799576. [Google Scholar] [CrossRef]
- Assaf, Y.; Johansen-Berg, H.; Thiebaut de Schotten, M. The role of diffusion MRI in neuroscience. NMR Biomed. 2019, 32, e3762. [Google Scholar] [CrossRef]
- Basser, P.J.; Pierpaoli, C. Microstructural and physiological features of tissues elucidated by quantitative-diffusion-tensor MRI. J. Magn. Reson. 2011, 213, 560–570. [Google Scholar] [CrossRef]
- Beaulieu, C. The basis of anisotropic water diffusion in the nervous system–a technical review. NMR Biomed. Int. J. Devoted Dev. Appl. Magn. Reson. In Vivo 2002, 15, 435–455. [Google Scholar]
- Uddin, M.N.; Faiyaz, A.; Wang, L.; Zhuang, Y.; Murray, K.D.; Descoteaux, M.; Tivarus, M.E.; Weber, M.T.; Zhong, J.; Qiu, X. A longitudinal analysis of brain extracellular free water in HIV infected individuals. Sci. Rep. 2021, 11, 1–12. [Google Scholar] [CrossRef]
- Jensen, J.H.; Helpern, J.A.; Ramani, A.; Lu, H.; Kaczynski, K. Diffusional kurtosis imaging: The quantification of non-gaussian water diffusion by means of magnetic resonance imaging. Magn. Reson. Med. Off. J. Int. Soc. Magn. Reson. Med. 2005, 53, 1432–1440. [Google Scholar] [CrossRef]
- Jensen, J.H.; Helpern, J.A. MRI quantification of non-Gaussian water diffusion by kurtosis analysis. NMR Biomed. 2010, 23, 698–710. [Google Scholar] [CrossRef]
- Minosse, S.; Picchi, E.; Conti, A.; di Giuliano, F.; di Ciò, F.; Sarmati, L.; Teti, E.; De Santis, S.; Andreoni, M.; Floris, R. Multishell diffusion MRI reveals whole-brain white matter changes in HIV. Hum. Brain Mapp. 2023, 44, 5113–5124. [Google Scholar] [CrossRef]
- Lampinen, B.; Szczepankiewicz, F.; Lätt, J.; Knutsson, L.; Mårtensson, J.; Björkman-Burtscher, I.M.; van Westen, D.; Sundgren, P.C.; Ståhlberg, F.; Nilsson, M. Probing brain tissue microstructure with MRI: Principles, challenges, and the role of multidimensional diffusion-relaxation encoding. NeuroImage 2023, 282, 120338. [Google Scholar] [CrossRef]
- Szczepankiewicz, F.; Sjölund, J.; Ståhlberg, F.; Lätt, J.; Nilsson, M. Tensor-valued diffusion encoding for diffusional variance decomposition (DIVIDE): Technical feasibility in clinical MRI systems. PLoS ONE 2019, 14, e0214238. [Google Scholar] [CrossRef]
- Nilsson, M.; Szczepankiewicz, F.; Brabec, J.; Taylor, M.; Westin, C.-F.; Golby, A.; van Westen, D.; Sundgren, P.C. Tensor-valued diffusion MRI in under 3 minutes: An initial survey of microscopic anisotropy and tissue heterogeneity in intracranial tumors. Magn. Reson. Med. 2020, 83, 608–620. [Google Scholar] [CrossRef]
- Szczepankiewicz, F.; Hoge, S.; Westin, C.-F. Linear, planar and spherical tensor-valued diffusion MRI data by free waveform encoding in healthy brain, water, oil and liquid crystals. Data Brief 2019, 25, 104208. [Google Scholar] [CrossRef]
- Nery, F.; Szczepankiewicz, F.; Kerkelä, L.; Hall, M.G.; Kaden, E.; Gordon, I.; Thomas, D.L.; Clark, C.A. In vivo demonstration of microscopic anisotropy in the human kidney using multidimensional diffusion MRI. Magn. Reson. Med. 2019, 82, 2160–2168. [Google Scholar] [CrossRef]
- Lasič, S.; Szczepankiewicz, F.; Dall’Armellina, E.; Das, A.; Kelly, C.; Plein, S.; Schneider, J.E.; Nilsson, M.; Teh, I. Motion-compensated b-tensor encoding for in vivo cardiac diffusion-weighted imaging. NMR Biomed. 2020, 33, e4213. [Google Scholar] [CrossRef]
- Gongvatana, A.; Schweinsburg, B.C.; Taylor, M.J.; Theilmann, R.J.; Letendre, S.L.; Alhassoon, O.M.; Jacobus, J.; Woods, S.P.; Jernigan, T.L.; Ellis, R.J. White matter tract injury and cognitive impairment in human immunodeficiency virusinfected individuals. J. Neurovirology 2009, 15, 187–195. [Google Scholar] [CrossRef]
- Cysique, L.A.; Soares, J.R.; Geng, G.; Scarpetta, M.; Moffat, K.; Green, M.; Brew, B.J.; Henry, R.G.; Rae, C. White matter measures are near normal in controlled HIV infection except in those with cognitive impairment and longer HIV duration. J. Neurovirology 2017, 23, 539–547. [Google Scholar] [CrossRef]
- Watson, C.; Busovaca, E.; Foley, J.M.; Allen, I.E.; Schwarz, C.G.; Jahanshad, N.; Nir, T.M.; Esmaeili-Firidouni, P.; Milanini, B.; Rosen, H. White matter hyperintensities correlate to cognition and fiber tract integrity in older adults with HIV. J. Neurovirology 2017, 23, 422–429. [Google Scholar] [CrossRef]
- Underwood, J.; Cole, J.H.; Caan, M.; De Francesco, D.; Leech, R.; Van Zoest, R.A.; Su, T.; Geurtsen, G.J.; Schmand, B.A.; Portegies, P. Gray and white matter abnormalities in treated human immunodeficiency virus disease and their relationship to cognitive function. Clin. Infect. Dis. 2017, 65, 422–432. [Google Scholar] [CrossRef]
- Su, T.; Caan, M.W.; Wit, F.W.; Schouten, J.; Geurtsen, G.J.; Cole, J.H.; Sharp, D.J.; Vos, F.M.; Prins, M.; Portegies, P. White matter structure alterations in HIV-1-infected men with sustained suppression of viraemia on treatment. AIDS 2016, 30, 311–322. [Google Scholar] [CrossRef]
- Figley, T.D.; Bhullar, N.; Courtney, S.M.; Figley, C.R. Probabilistic atlases of default mode, executive control and salience network white matter tracts: An fMRI-guided diffusion tensor imaging and tractography study. Front. Hum. Neurosci. 2015, 9, 585. [Google Scholar] [CrossRef]
- Figley, T.D.; Mortazavi Moghadam, B.; Bhullar, N.; Kornelsen, J.; Courtney, S.M.; Figley, C.R. Probabilistic white matter atlases of human auditory, basal ganglia, language, precuneus, sensorimotor, visual and visuospatial networks. Front. Hum. Neurosci. 2017, 11, 306. [Google Scholar] [CrossRef]
- Shirer, W.R.; Ryali, S.; Rykhlevskaia, E.; Menon, V.; Greicius, M.D. Decoding subject-driven cognitive states with whole-brain connectivity patterns. Cereb. Cortex 2012, 22, 158–165. [Google Scholar] [CrossRef]
- Uddin, M.N.; Singh, M.V.; Faiyaz, A.; Szczepankiewicz, F.; Nilsson, M.; Boodoo, Z.D.; Sutton, K.R.; Tivarus, M.E.; Zhong, J.; Wang, L. Tensor-valued diffusion MRI detects brain microstructural abnormalities in HIV infected individuals with cognitive impairment. Sci. Rep. 2024, 14, 28839. [Google Scholar] [CrossRef]
- Murray, K.D.; Singh, M.V.; Zhuang, Y.; Uddin, M.N.; Qiu, X.; Weber, M.T.; Tivarus, M.E.; Wang, H.Z.; Sahin, B.; Zhong, J.; et al. Pathomechanisms of HIV-Associated Cerebral Small Vessel Disease: A Comprehensive Clinical and Neuroimaging Protocol and Analysis Pipeline. Front. Neurol. 2020, 11, 595463. [Google Scholar] [CrossRef]
- Szczepankiewicz, F.; Westin, C.-F.; Nilsson, M. Gradient waveform design for tensor-valued encoding in diffusion MRI. J. Neurosci. Methods 2021, 348, 109007. [Google Scholar] [CrossRef]
- Jenkinson, M.; Beckmann, C.F.; Behrens, T.E.; Woolrich, M.W.; Smith, S.M. Fsl. NeuroImage 2012, 62, 782–790. [Google Scholar]
- Avants, B.B.; Tustison, N.; Song, G. Advanced normalization tools (ANTS). Insight J. 2009, 2, 1–35. [Google Scholar]
- Smith, S.M.; Jenkinson, M.; Woolrich, M.W.; Beckmann, C.F.; Behrens, T.E.; Johansen-Berg, H.; Bannister, P.R.; De Luca, M.; Drobnjak, I.; Flitney, D.E. Advances in functional and structural MR image analysis and implementation as FSL. NeuroImage 2004, 23, S208–S219. [Google Scholar] [CrossRef]
- Smith, S.M. Fast robust automated brain extraction. Hum. Brain Mapp. 2002, 17, 143–155. [Google Scholar] [CrossRef]
- Nilsson, M.; Szczepankiewicz, F.; Lampinen, B.; Ahlgren, A.; De Almeida Martins, J.P.; Lasic, S.; Westin, C.-F.; Topgaard, D. An open-source framework for analysis of multidimensional diffusion MRI data implemented in MATLAB. Proc. Intl. Soc. Mag. Reson. Med. 2018, 26, 5355. [Google Scholar]
- Nilsson, M.; Szczepankiewicz, F.; van Westen, D.; Hansson, O. Extrapolation-based references improve motion and eddy-current correction of high b-value DWI data: Application in Parkinson’s disease dementia. PLoS ONE 2015, 10, e0141825. [Google Scholar] [CrossRef]
- Klein, S.; Staring, M.; Murphy, K.; Viergever, M.A.; Pluim, J.P. Elastix: A toolbox for intensity-based medical image registration. IEEE Trans. Med. Imaging 2009, 29, 196–205. [Google Scholar]
- Westin, C.-F.; Knutsson, H.; Pasternak, O.; Szczepankiewicz, F.; Özarslan, E.; van Westen, D.; Mattisson, C.; Bogren, M.; O’Donnell, L.J.; Kubicki, M. Q-space trajectory imaging for multidimensional diffusion MRI of the human brain. NeuroImage 2016, 135, 345–362. [Google Scholar] [CrossRef]
- Avants, B.B.; Tustison, N.J.; Song, G.; Cook, P.A.; Klein, A.; Gee, J.C. A reproducible evaluation of ANTs similarity metric performance in brain image registration. NeuroImage 2011, 54, 2033–2044. [Google Scholar]
- Thomas, J.B.; Brier, M.R.; Snyder, A.Z.; Vaida, F.F.; Ances, B.M. Pathways to neurodegeneration: Effects of HIV and aging on resting-state functional connectivity. Neurology 2013, 80, 1186–1193. [Google Scholar]
- Zhou, Z.; Wang, W.; Li, H.; Shi, Y.; Zhao, L.; Lu, Y.; Wei, X.; Li, H. Decoding HIV-associated neurocognitive disorders: A new perspective from multimodal connectomics. Front. Neurol. 2025, 16, 1467175. [Google Scholar] [CrossRef]
- Andersen, K.W.; Lasič, S.; Lundell, H.; Nilsson, M.; Topgaard, D.; Sellebjerg, F.; Szczepankiewicz, F.; Siebner, H.R.; Blinkenberg, M.; Dyrby, T.B. Disentangling white-matter damage from physiological fibre orientation dispersion in multiple sclerosis. Brain Commun. 2020, 2, fcaa077. [Google Scholar] [CrossRef]
- Boito, D.; Eklund, A.; Tisell, A.; Levi, R.; Özarslan, E.; Blystad, I. MRI with generalized diffusion encoding reveals damaged white matter in patients previously hospitalized for COVID-19 and with persisting symptoms at follow-up. Brain Commun. 2023, 5, fcad284. [Google Scholar] [CrossRef]
- Li, X.; Sawamura, D.; Hamaguchi, H.; Urushibata, Y.; Feiweier, T.; Ogawa, K.; Tha, K.K. Microscopic fractional anisotropy detects cognitive training-induced microstructural brain changes. Tomography 2022, 8, 33–44. [Google Scholar] [CrossRef]
- Kamiya, K.; Kamagata, K.; Ogaki, K.; Hatano, T.; Ogawa, T.; Takeshige-Amano, H.; Murata, S.; Andica, C.; Murata, K.; Feiweier, T. Brain white-matter degeneration due to aging and Parkinson disease as revealed by double diffusion encoding. Front. Neurosci. 2020, 14, 584510. [Google Scholar] [CrossRef]
- Wang, H.; Jiu, X.; Wang, Z.; Zhang, Y. Neuroimaging advances in neurocognitive disorders among HIV-infected individuals. Front. Neurol. 2025, 16, 1479183. [Google Scholar] [CrossRef]
- O’Connor, E.E.; Jaillard, A.; Renard, F.; Zeffiro, T.A. Reliability of white matter microstructural changes in HIV infection: Meta-analysis and confirmation. Am. J. Neuroradiol. 2017, 38, 1510–1519. [Google Scholar] [CrossRef]
- Ma, J.; Yang, X.; Xu, F.; Li, H. Application of diffusion tensor imaging (DTI) in the diagnosis of HIV-associated neurocognitive disorder (HAND): A meta-analysis and a system review. Front. Neurol. 2022, 13, 898191. [Google Scholar] [CrossRef]
- Sui, J.; Li, X.; Bell, R.P.; Towe, S.L.; Gadde, S.; Chen, N.-K.; Meade, C.S. Structural and functional brain abnormalities in human immunodeficiency virus disease revealed by multimodal magnetic resonance imaging fusion: Association with cognitive function. Clin. Infect. Dis. 2021, 73, e2287–e2293. [Google Scholar] [CrossRef]
- Kanmogne, G.; Fonsah, J.; Tang, B.; Doh, R.; Kengne, A.; Umlauf, A. Effects of HIV on executive function and verbal fluency in Cameroon. Sci. Rep. 2018, 8, 17794. [Google Scholar] [CrossRef]
- Chen, C.; Song, F.; Peng, C.; Shi, Y.; Cai, D.-C. Attention or memory deficits? An HIV study in Shanghai using local and norm-based controls. Front. Neurol. 2025, 16, 1555378. [Google Scholar] [CrossRef]
- Walker, K.A.; Brown, G.G. HIV-associated executive dysfunction in the era of modern antiretroviral therapy: A systematic review and meta-analysis. J. Clin. Exp. Neuropsychol. 2018, 40, 357–376. [Google Scholar] [CrossRef]
- Abidin, A.Z.; DSouza, A.M.; Nagarajan, M.B.; Wang, L.; Qiu, X.; Schifitto, G.; Wismüller, A. Alteration of brain network topology in HIV-associated neurocognitive disorder: A novel functional connectivity perspective. NeuroImage Clin. 2018, 17, 768–777. [Google Scholar] [CrossRef]
- Chaganti, J.; Heinecke, A.; Gates, T.; Moffat, K.; Brew, B. Functional connectivity in virally suppressed patients with HIV-associated neurocognitive disorder: A resting-state analysis. Am. J. Neuroradiol. 2017, 38, 1623–1629. [Google Scholar] [CrossRef]
- Diaz, M.M.; Harris, M.G.; Koble, J.M.; Copperthite, K.; Jimenez, J.; Dayan, E. Association between processing speed and segregation/integration of large-scale functional networks in middle-aged and older people living with HIV. BMC Neurol. 2026, 26, 207. [Google Scholar] [CrossRef]
- Kanmogne, G.D.; Fonsah, J.Y.; Umlauf, A.; Moul, J.; Doh, R.F.; Kengne, A.M.; Tang, B.; Tagny, C.T.; Nchindap, E.; Kenmogne, L. Effects of HIV infection, antiretroviral therapy, and immune status on the speed of information processing and complex motor functions in adult Cameroonians. Sci. Rep. 2020, 10, 14016. [Google Scholar] [CrossRef]
- Chen, J.; Wang, Z.; Shi, M.; Shi, Z.; Shi, W. Large-scale functional network connectivity mediate the associations of white matter lesions with executive functions and information processing speed in asymptomatic cerebral small vessels diseases. NeuroImage Clin. 2025, 46, 103773. [Google Scholar] [CrossRef] [PubMed]
- Savini, G.; Pardini, M.; Castellazzi, G.; Lascialfari, A.; Chard, D.; D’Angelo, E.; Gandini Wheeler-Kingshott, C.A. Default mode network structural integrity and cerebellar connectivity predict information processing speed deficit in multiple sclerosis. Front. Cell. Neurosci. 2019, 13, 21. [Google Scholar] [CrossRef] [PubMed]
- Mazerolle, E.L.; Wojtowicz, M.A.; Omisade, A.; Fisk, J.D. Intra-individual variability in information processing speed reflects white matter microstructure in multiple sclerosis. NeuroImage Clin. 2013, 2, 894–902. [Google Scholar] [CrossRef]
- Devlin, K.N.; Giovannetti, T. Heterogeneity of neuropsychological impairment in HIV infection: Contributions from mild cognitive impairment. Neuropsychol. Rev. 2017, 27, 101–123. [Google Scholar] [CrossRef]
- Cysique, L.A.; Maruff, P.; Brew, B.J. Prevalence and pattern of neuropsychological impairment in human immunodeficiency virus-infected/acquired immunodeficiency syndrome (HIV/AIDS) patients across pre-and post-highly active antiretroviral therapy eras: A combined study of two cohorts. J. Neurovirology 2004, 10, 350–357. [Google Scholar] [CrossRef]
- Nightingale, S.; Ances, B.; Cinque, P.; Dravid, A.; Dreyer, A.J.; Gisslén, M.; Joska, J.A.; Kwasa, J.; Meyer, A.-C.; Mpongo, N. Cognitive impairment in people living with HIV: Consensus recommendations for a new approach. Nat. Rev. Neurol. 2023, 19, 424–433. [Google Scholar] [CrossRef]
- Cysique, L.A.; Brew, B.J.; Bruning, J.; Byrd, D.; Costello, J.; Daken, K.; Ellis, R.J.; Fazeli, P.L.; Goodkin, K.; Gouse, H. Cognitive criteria in HIV: Greater consensus is needed. Nat. Rev. Neurol. 2024, 20, 127–128. [Google Scholar] [CrossRef] [PubMed]




| Characteristics | PWH (n = 24) | HC (n = 31) | p-Value | |
|---|---|---|---|---|
| Age, mean ± SD | 55 ± 10 | 55 ± 15 | 0.947 | |
| Sex, n | 0.443 | |||
| Female | 7 | 7 | ||
| Male | 17 | 24 | ||
| Ethnicity, n | 0.847 | |||
| Hispanic or Latino | 0 | 2 | ||
| Not Hispanic or Latino | 23 | 29 | ||
| Other | 1 | 0 | ||
| Race, n | <0.001 | |||
| Caucasian | 16 | 27 | ||
| Black African American | 6 | 4 | ||
| Other | 1 | 0 | ||
| Missing | 1 | 0 | ||
| Education, n | <0.001 | |||
| ≤12 Years | 4 | 1 | ||
| >12 Years | 20 | 30 | ||
| Time since HIV diagnosis | ||||
| CD4+ T-cell count, cell/mm3, mean ± SD | 728 ± 288 | NA | NA | |
| Viral load copies/mL, mean ± SD | 7 ± 13 | NA | NA | |
| ARV drug class | ||||
| NRTI | 4 | NA | NA | |
| NNRTI | 3 | NA | NA | |
| PI | 16 | NA | NA | |
| Network | Cohort | µFA vs. EXE | MKi vs. EXE | MKa vs. EXE | MD vs. EXE | µFA vs. ATT | MKi vs. ATT | MKa vs. ATT | MD vs. ATT | MKi vs. SPE | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| DMN_D | HC | r (p) | 0.25 (0.185) | 0.15 (0.419) | 0.30 (0.113) | −0.21 (0.267) | 0.49 (0.006) | −0.07 (0.698) | 0.51 (0.004) | −0.48 (0.005) | 0.11 (0.612) |
| d | 0.52 | 0.30 | 0.63 | −0.43 | 1.12 | −0.14 | 1.19 | −1.09 | 0.22 | ||
| PWH | r (p) | 0.51 (0.016) | −0.56 (0.006) | 0.52 (0.014) | −0.54 (0.016) | 0.23 (0.298) | −0.53 (0.012) | 0.34 (0.119) | −0.09 (0.738) | −0.62 (0.002) | |
| d | 1.19 | −1.35 | 1.22 | −1.28 | 0.47 | −1.25 | 0.72 | −0.18 | −1.58 | ||
| DMN_V | HC | r (p) | 0.18 (0.348) | 0.21 (0.266) | 0.19 (0.314) | −0.07 (0.911) | 0.44 (0.016) | −0.12 (0.528) | 0.40 (0.029) | −0.34 (0.051) | 0.12 (0.528) |
| d | 0.37 | 0.43 | 0.39 | −0.14 | 0.98 | −0.24 | 0.87 | −0.72 | 0.24 | ||
| PWH | r (p) | 0.49 (0.020) | −0.33 (0.129) | 0.43 (0.048) | −0.25 (0.260) | 0.21 (0.353) | −0.28 (0.210) | 0.34 (0.119) | −0.10 (0.629) | −0.62 (0.002) | |
| d | 1.12 | −0.7 | 0.95 | −0.52 | 0.43 | −0.58 | 0.72 | −0.20 | −1.58 | ||
| ECN_L | HC | r (p) | 0.16 (0.391) | 0.23 (0.222) | 0.25 (0.181) | −0.03 (0.884) | 0.37 (0.046) | 0.19 (0.323) | 0.46 (0.011) | −0.34 (0.066) | 0.05 (0.867) |
| d | 0.32 | 0.47 | 0.52 | −0.06 | 0.80 | 0.39 | 1.04 | −0.72 | 0.1 | ||
| PWH | r (p) | 0.49 (0.022) | −0.31 (0.161) | 0.32 (0.152) | −0.33 (0.094) | 0.22 (0.331) | −0.34 (0.122) | 0.24 (0.287) | −0.11 (0.611) | −0.37 (0.088) | |
| d | 1.12 | −0.65 | 0.68 | −0.70 | 0.45 | −0.72 | 0.49 | −0.22 | −0.8 | ||
| ECN_R | HC | r (p) | 0.21 (0.273) | 0.19 (0.315) | 0.19 (0.315) | −0.20 (0.290) | 0.52 (0.003) | 0.06 (0.769) | 0.50 (0.005) | −0.42 (0.021) | 0.18 (0.340) |
| d | 0.43 | 0.39 | 0.39 | −0.41 | 1.22 | 0.12 | 1.15 | −0.93 | 0.37 | ||
| PWH | r (p) | 0.48 (0.023) | −0.43 (0.048) | 0.43 (0.043) | −0.49 (0.019) | 0.26 (0.245) | −0.39 (0.078) | 0.30 (0.178) | −0.03 (0.736) | −0.44 (0.056) | |
| d | 1.09 | −0.95 | 0.95 | −1.12 | 0.54 | −0.85 | 0.63 | −0.06 | −0.98 | ||
| SN_A | HC | r (p) | 0.17 (0.373) | 0.19 (0.304) | 0.09 (0.620) | −0.16 (0.465) | 0.49 (0.006) | −0.00 (0.993) | 0.46 (0.010) | −0.50 (0.005) | 0.18 (0.340) |
| d | 0.35 | 0.39 | 0.18 | −0.32 | 1.12 | 0.00 | 1.04 | −1.15 | 0.37 | ||
| PWH | r (p) | 0.43 (0.046) | −0.44 (0.038) | 0.48 (0.023) | −0.48 (0.016) | 0.15 (0.513) | −0.39 (0.074) | 0.29 (0.187) | −0.04 (0.832) | −0.50 (0.019) | |
| d | 0.95 | −0.98 | 1.09 | −1.09 | 0.30 | −0.85 | 0.61 | −0.08 | −1.15 | ||
| SN_P | HC | r (p) | 0.12 (0.511) | 0.23 (0.2170) | 0.12 (0.540) | −0.16 (0.400) | 0.42 (0.022) | 0.00 (0.984) | 0.43 (0.018) | −0.44 (0.015) | −0.01 (0.977) |
| d | 0.24 | 0.47 | 0.24 | −0.32 | 0.93 | 0.00 | 0.95 | −0.98 | −0.02 | ||
| PWH | r (p) | 0.26 (0.236) | −0.23 (0.310) | 0.23 (0.301) | −0.29 (0.191) | 0.15 (0.513) | 0.00 (0.987) | 0.12 (0.606) | −0.26 (0.097) | −0.32 (0.140) | |
| d | 0.54 | −0.47 | 0.47 | −0.61 | 0.30 | 0.00 | 0.24 | −0.54 | −0.68 | ||
| Cognition | Effect | Estimate | Std. Error | t Value | Cohen’s d | Pr (>|t|) |
|---|---|---|---|---|---|---|
| EXE | MKi DMN_D | 2.2 | 5.6 | 0.39 | 0.10 | 0.696 |
| HIV Status | 10.99 | 4.4 | 2.5 | 0.67 | 0.016 | |
| HIV × MKi DMN_D | −21.65 | 8.84 | −2.45 | −0.65 | 0.018 | |
| EXE | ECN_R | 6.66 | 4.40 | 1.51 | 0.40 | 0.136 |
| HIV Status | 5.72 | 2.54 | 2.25 | 0.60 | 0.029 | |
| HIV × MKi ECN_R | −11.29 | 5.09 | −2.22 | −0.59 | 0.031 | |
| ATT | ECN_L | 2.55 | 1.65 | 1.55 | 0.41 | 0.128 |
| HIV Status | 2.10 | 0.97 | 2.18 | 0.58 | 0.034 | |
| HIV × MKi ECN_L | −4.17 | 1.95 | −2.14 | −0.57 | 0.037 | |
| ATT | ECN_L | 2.55 | 1.65 | 1.55 | 0.41 | 0.128 |
| HIV Status | 2.10 | 0.97 | 2.18 | 0.58 | 0.034 | |
| HIV × MKi ECN_L | −4.17 | 1.95 | −2.14 | −0.57 | 0.037 | |
| ATT | ECN_L | 3.86 | 1.13 | 3.43 | 0.92 | 0.001 |
| HIV Status | 3.39 | 0.94 | 3.60 | 0.96 | 0.001 | |
| HIV × MKt ECN_L | −4.53 | 1.29 | −3.51 | −0.94 | 0.001 | |
| ATT | ECN_R | 4.07 | 1.36 | 3.00 | 0.8 | 0.004 |
| HIV Status | 3.89 | 1.25 | 3.12 | 0.83 | 0.003 | |
| HIV × MKt ECN_R | −5.05 | 1.65 | −3.06 | −0.82 | 0.004 | |
| ATT | ECN_R | −5.56 | 2.05 | −2.71 | −0.72 | 0.009 |
| HIV Status | −4.53 | 2.30 | −1.97 | −0.53 | 0.055 | |
| HIV × MD ECN_R | 6.06 | 2.98 | 2.03 | 0.54 | 0.047 | |
| ATT | SN_A | −5.98 | 1.96 | −3.06 | −0.82 | 0.004 |
| HIV Status | −5.82 | 2.47 | −2.36 | −0.63 | 0.022 | |
| HIV × MD SN_A | 7.13 | 2.93 | 2.43 | 0.65 | 0.019 | |
| ATT | SN_A | 7.34 | 2.21 | 3.32 | 0.89 | 0.002 |
| HIV Status | 2.87 | 1.31 | 2.20 | 0.59 | 0.033 | |
| HIV × μFA SN_A | −6.55 | 3.17 | −2.07 | −0.55 | 0.044 | |
| ATT | SN_A | 26.01 | 8.94 | 2.91 | 0.78 | 0.005 |
| HIV Status | 5.77 | 2.37 | 2.44 | 0.65 | 0.019 | |
| HIV × FA SN_A | −28.81 | 12.09 | −2.38 | −0.63 | 0.021 | |
| SPE | DMN_D | 4.18 | 4.85 | 0.86 | 0.23 | 0.393 |
| HIV Status | 11.69 | 3.81 | 3.07 | 0.82 | 0.003 | |
| HIV × MKi DMN_D | −23.77 | 7.67 | −3.10 | −0.83 | 0.003 | |
| SPE | ECN_R | 5.24 | 3.87 | 1.35 | 0.36 | 0.182 |
| HIV Status | 4.93 | 2.23 | 2.21 | 0.59 | 0.032 | |
| HIV × MKi ECN_R | −10.40 | 4.48 | −2.32 | −0.62 | 0.024 | |
| SPE | SN_A | 9.52 | 4.82 | 1.98 | 0.53 | 0.054 |
| HIV_Status | 12.52 | 4.89 | 2.56 | 0.68 | 0.013 | |
| HIV × MKi SN_A | −25.62 | 9.80 | −2.62 | −0.7 | 0.012 | |
| LAN | SN_A | 4.15 | 5.32 | 0.78 | 0.21 | 0.439 |
| HIV_Status | 12.61 | 5.40 | 2.34 | 0.63 | 0.024 | |
| HIV × MKi SN_A | −26.54 | 10.82 | −2.45 | −0.65 | 0.018 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Uddin, M.N.; Faiyaz, A.; Figley, C.R.; Qiu, X.; Weber, M.T.; Schifitto, G. HIV-Associated Microstructural Abnormalities in Default Mode, Executive Control, and Salience Networks: Insights from Tensor-Valued Diffusion Encoding. Bioengineering 2026, 13, 413. https://doi.org/10.3390/bioengineering13040413
Uddin MN, Faiyaz A, Figley CR, Qiu X, Weber MT, Schifitto G. HIV-Associated Microstructural Abnormalities in Default Mode, Executive Control, and Salience Networks: Insights from Tensor-Valued Diffusion Encoding. Bioengineering. 2026; 13(4):413. https://doi.org/10.3390/bioengineering13040413
Chicago/Turabian StyleUddin, Md Nasir, Abrar Faiyaz, Chase R. Figley, Xing Qiu, Miriam T. Weber, and Giovanni Schifitto. 2026. "HIV-Associated Microstructural Abnormalities in Default Mode, Executive Control, and Salience Networks: Insights from Tensor-Valued Diffusion Encoding" Bioengineering 13, no. 4: 413. https://doi.org/10.3390/bioengineering13040413
APA StyleUddin, M. N., Faiyaz, A., Figley, C. R., Qiu, X., Weber, M. T., & Schifitto, G. (2026). HIV-Associated Microstructural Abnormalities in Default Mode, Executive Control, and Salience Networks: Insights from Tensor-Valued Diffusion Encoding. Bioengineering, 13(4), 413. https://doi.org/10.3390/bioengineering13040413







