A Multi-Channel AM-TMAS Driving System Based on Amplitude-Modulated Sine Waves
Abstract
1. Introduction
- TMAS technology is combined with AM to design and implement a multi-channel AM-TMAS driving system based on phased arrays, enabling a single device to control multiple channels and generate AM waveforms with high carrier frequencies and low-frequency envelopes corresponding to brain rhythms.
- A DDS/LUT-based digital synthesis method is implemented on an FPGA + high-speed DAC platform, allowing the system to maintain the inherent advantages of high-frequency penetration and focal control while delivering controllable stimulation at specific rhythmic frequency bands.
- The proposed system is validated through electrical output measurements and magneto-acoustic electrical experiments in a saline medium under a static magnetic field, confirming waveform fidelity, multi-channel synchronization, and rhythmic modulation characteristics consistent with the stimulation envelope.
2. Materials and Methods
2.1. Basic Principle of Tmas
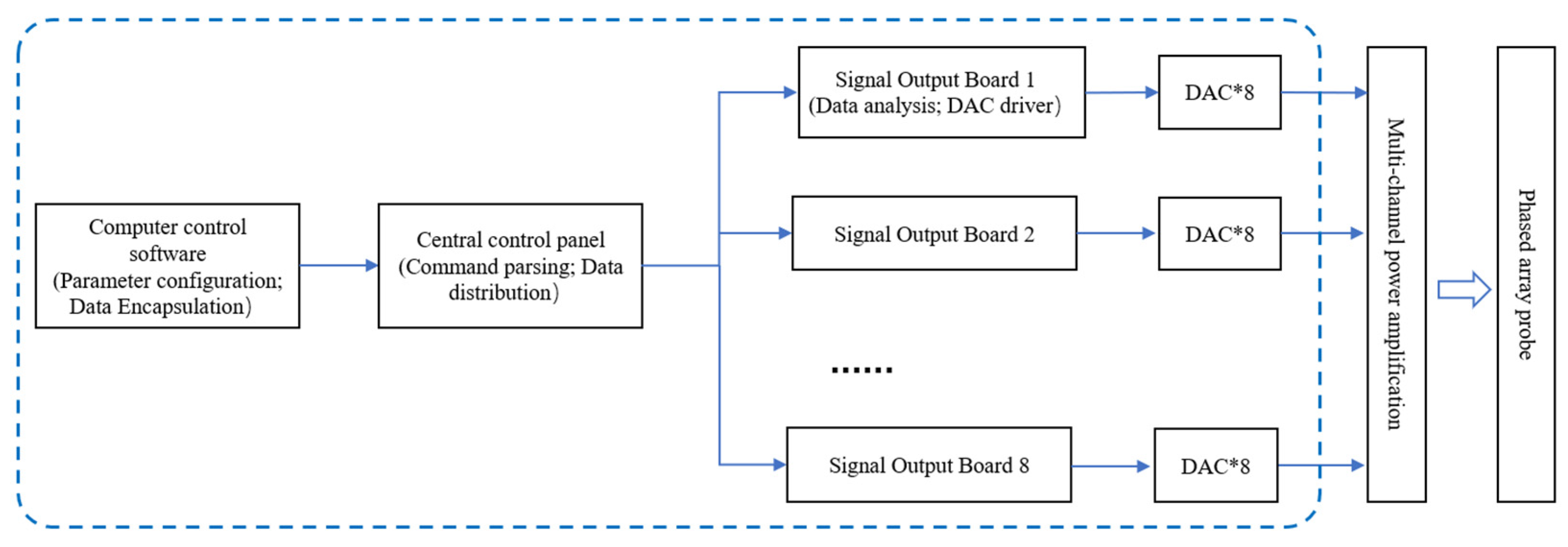
2.2. Am Hardware Architecture of the Am Module
2.3. Software Design of the Am Module
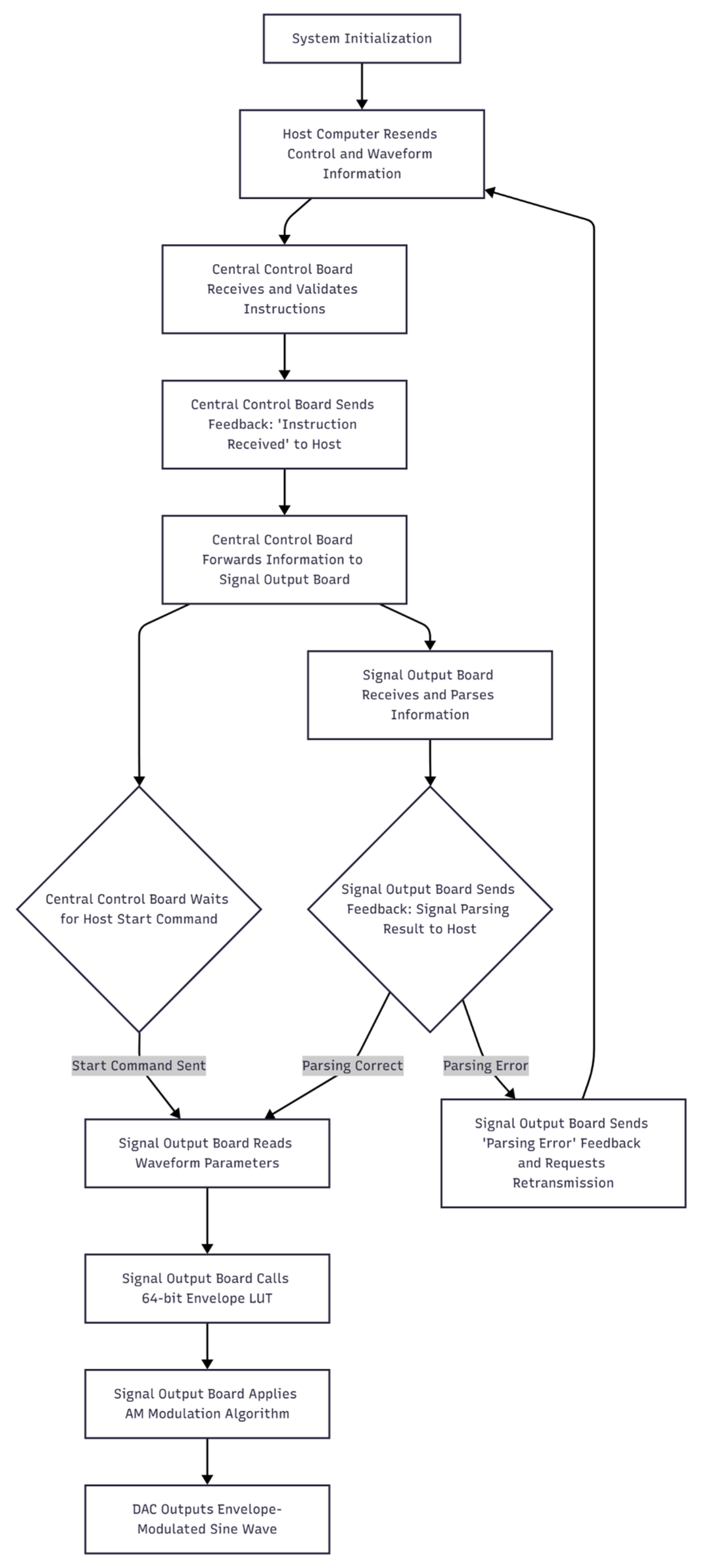
2.4. Implementation of Amplitude-Modulated Sine Waves
2.4.1. AM Algorithm
2.4.2. Implementation of the AM Algorithm on the Signal-Output Board
2.5. System Testing Methods
2.5.1. Modulated Signal-Output Test
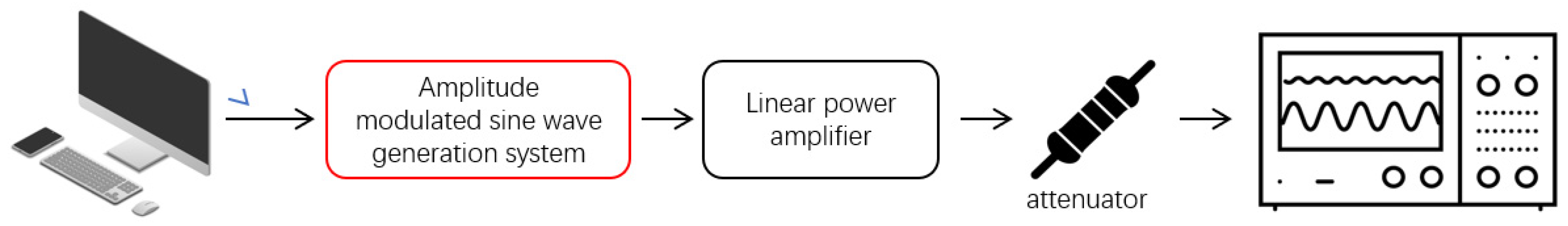
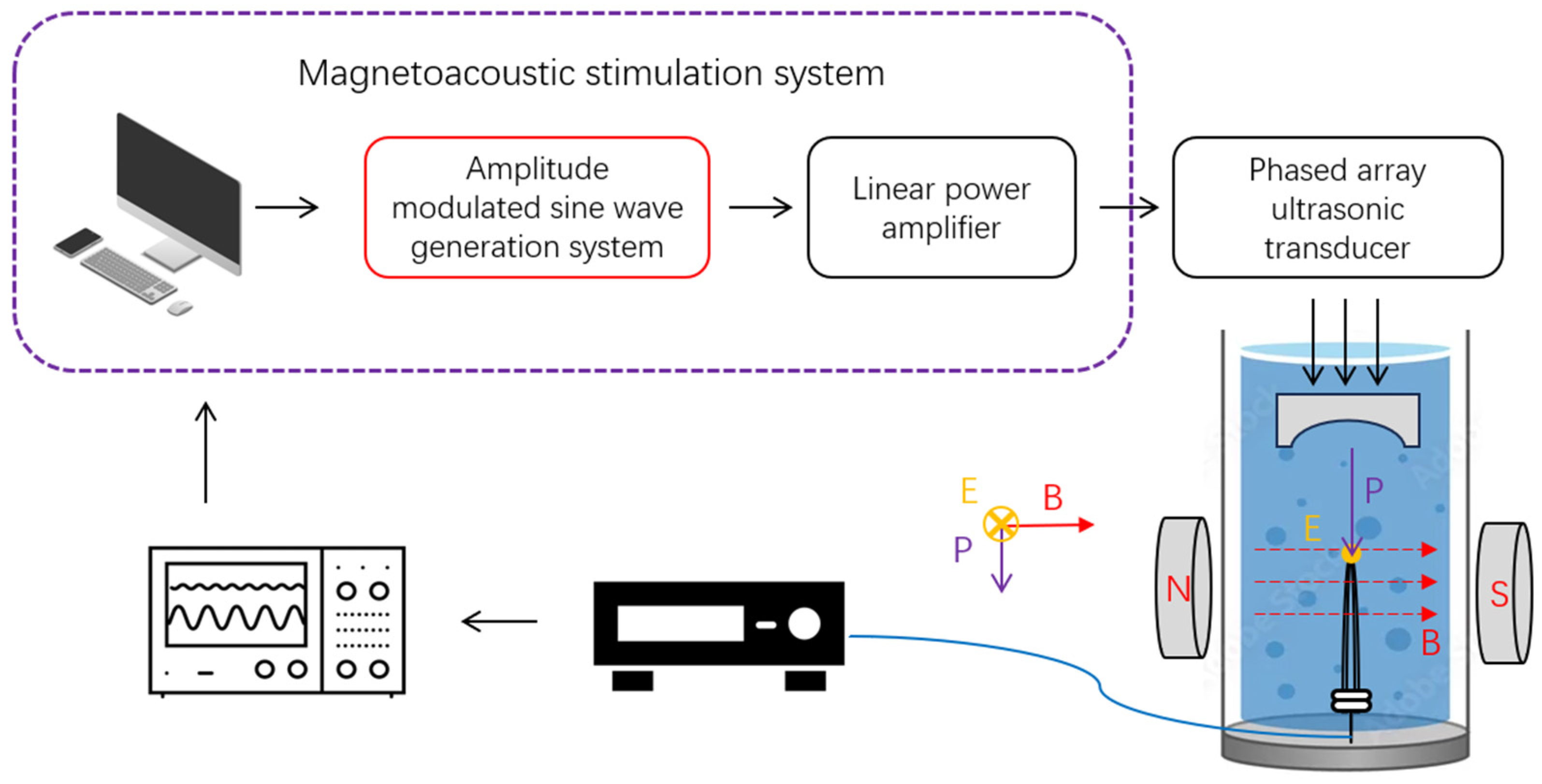
2.5.2. Magneto-Acoustic Electrical Measurement of the AM-TMAS System
3. Results
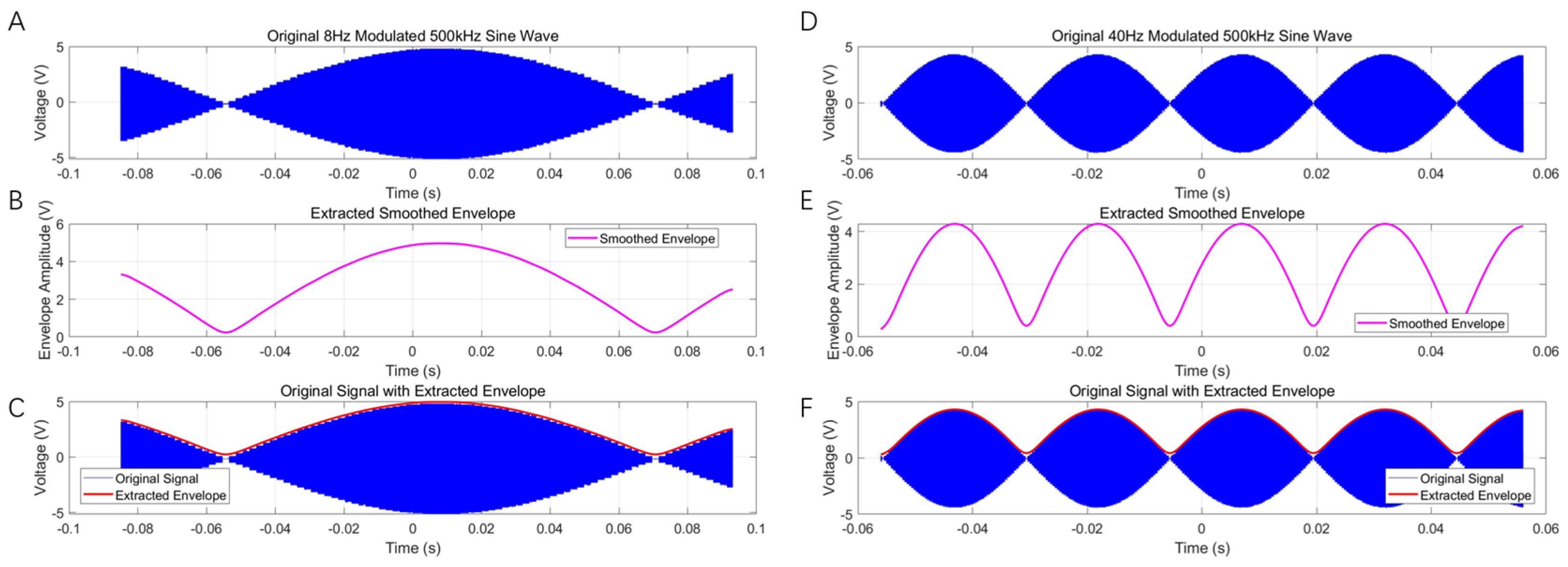
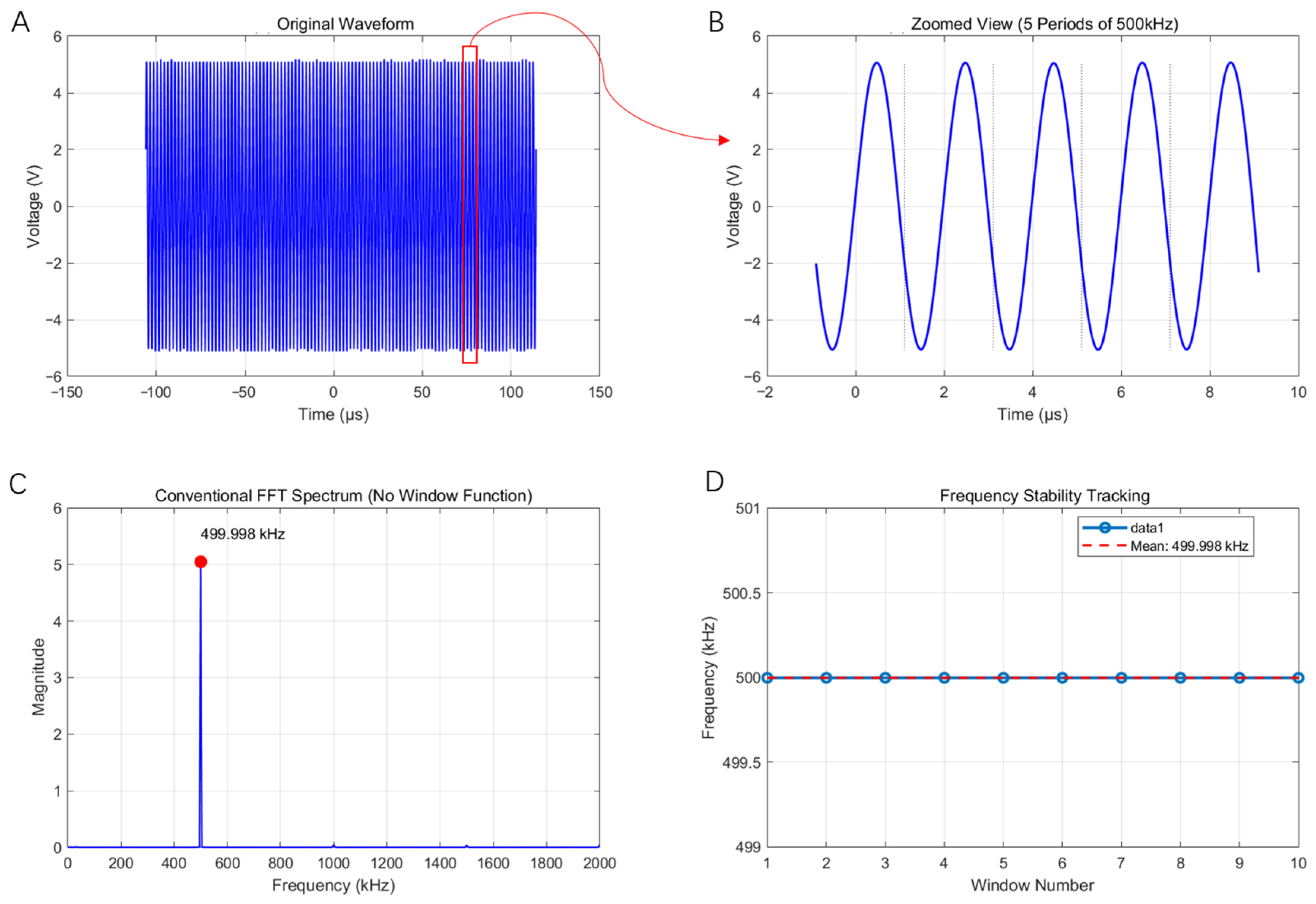
3.1. Multi-Channel AM Signal-Output Tests


3.2. AM Magneto-Acoustic-Coupled Electrical Signal Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Klimesch, W. The frequency architecture of brain and brain-body oscillations: An analysis. Eur. J. Neurosci. 2018, 48, 2431–2453. [Google Scholar] [CrossRef]
- Başar, E.; Başar Eroğlu, C.; Güntekin, B.; Yener, G.G. Brain’s alpha, beta, gamma, delta, and theta oscillations in neuropsychiatric diseases: Proposal for biomarker strategies. In Supplements to Clinical Neurophysiology; Elsevier: Amsterdam, The Netherlands, 2013; Volume 62, pp. 19–54. [Google Scholar] [CrossRef]
- Başar, E.; Güntekin, B. Review of delta, theta, alpha, beta and gamma response oscillations in neuropsychiatric disorders. In Supplements to Clinical Neurophysiology; Elsevier: Amsterdam, The Netherlands, 2013; Volume 62, pp. 303–341. [Google Scholar] [CrossRef]
- Santhosh, K.; Dev, P.P.; Jose, B.A.; Lynton, Z.; Das, P.; Ghaderpour, E. A modified Gray Wolf Optimization algorithm for early detection of Parkinson’s Disease. Biomed. Signal Process. Control 2025, 109, 108061. [Google Scholar] [CrossRef]
- Bulut Arikan, F.; Cetintas, D.; Aksoy, A.; Yildirim, M. A Deep Learning Approach to Alzheimer’s Diagnosis Using EEG Data: Dual-Attention and Optuna-Optimized SVM. Biomedicines 2025, 13, 2017. [Google Scholar] [CrossRef]
- Assenza, G.; Pellicciari, M.C.; Di Lazzaro, V. Oscillatory activities in neurological disorders of elderly: Biomarkers to target for neuromodulation. Front. Aging Neurosci. 2017, 9, 189. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.; Suk, H.; Jackson, B.; Milman, N.P.; Stark, D.; Beach, S.D.; Tsai, L. Induction of specific brain oscillations may restore neural circuits and be used for the treatment of Alzheimer’s disease. J. Intern. Med. 2021, 290, 993–1009. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, Y.; Berényi, A. Oscillotherapeutics—Time-targeted interventions in epilepsy and beyond. Neurosci. Res. 2020, 152, 87–107. [Google Scholar] [CrossRef]
- Rossini, P.M.; Cole, J.; Paulus, W.; Ziemann, U.; Chen, R. 1924–2024: First centennial of EEG. Clin. Neurophysiol. 2025, 170, 132–135. [Google Scholar] [CrossRef]
- Pascucci, D.; Menétrey, M.Q.; Passarotto, E.; Luo, J.; Paramento, M.; Rubega, M. EEG brain waves and alpha rhythms: Past, current and future direction. Neurosci. Biobehav. Rev. 2025, 176, 106288. [Google Scholar] [CrossRef]
- Omae, E.; Shima, A.; Tanaka, K.; Yamada, M.; Cao, Y.; Nakamura, T.; Hoshiai, H.; Chiba, Y.; Irisawa, H.; Mizushima, T.; et al. Case Report: An N-of-1 study using amplitude-modulated transcranial alternating current stimulation between Broca’s area and the right homotopic area to improve post-stroke aphasia with increased inter-regional synchrony. Front. Hum. Neurosci. 2024, 18, 1297683. [Google Scholar] [CrossRef]
- Haslacher, D.; Narang, A.; Sokoliuk, R.; Cavallo, A.; Reber, P.; Nasr, K.; Santarnecchi, E.; Soekadar, S.R. In vivo phase-dependent enhancement and suppression of human brain oscillations by transcranial alternating current stimulation (tACS). Neuroimage 2023, 275, 120187. [Google Scholar] [CrossRef]
- Haslacher, D.; Cavallo, A.; Reber, P.; Kattein, A.; Thiele, M.; Nasr, K.; Hashemi, K.; Sokoliuk, R.; Thut, G.; Soekadar, S.R. Working memory enhancement using real-time phase-tuned transcranial alternating current stimulation. Brain Stimul. 2024, 17, 850–859. [Google Scholar] [CrossRef]
- Grossman, N.; Bono, D.; Dedic, N.; Kodandaramaiah, S.B.; Rudenko, A.; Suk, H.-J.; Cassara, A.M.; Neufeld, E.; Kuster, N.; Tsai, L.-H.; et al. Noninvasive deep brain stimulation via temporally interfering electric fields. Cell 2017, 169, 1029–1041.e16. [Google Scholar] [CrossRef]
- Violante, I.R.; Alania, K.; Cassarà, A.M.; Neufeld, E.; Acerbo, E.; Carron, R.; Williamson, A.; Kurtin, D.L.; Rhodes, E.; Hampshire, A.; et al. Non invasive temporal interference electrical stimulation of the human hippocampus. Nat. Neurosci. 2023, 26, 1994–2004. [Google Scholar] [CrossRef]
- Wang, M.; Song, S.; Li, D.; Zhao, G.; Luo, Y.; Tian, Y.; Zhang, J.; Liu, Q.; Wei, P. Transcranial temporal interference stimulation precisely targets deep brain regions to regulate eye movements. Neurosci. Bull. 2025, 41, 1390–1402. [Google Scholar] [CrossRef]
- Wang, H.; Sun, W.; Zhang, J.; Yan, Z.; Wang, C.; Wang, L.; Liu, T.; Li, C.; Chen, D.; Shintaro, F.; et al. Influence of layered skull modeling on the frequency sensitivity and target accuracy in simulations of transcranial current stimulation. Hum. Brain Mapp. 2021, 42, 5345–5356. [Google Scholar] [CrossRef]
- Esmaeilpour, Z.; Kronberg, G.; Reato, D.; Parra, L.C.; Bikson, M. Temporal interference stimulation targets deep brain regions by modulating neural oscillations. Brain Stimul. 2021, 14, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Li, H.; Zhang, Y. Theoretical analysis of effects of transcranial magneto-acoustical stimulation. BMC Neurosci. 2022, 23, 26. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, X.; Cui, D.; Liu, R.; Tan, R.; Wang, X.; Liu, Z.; Yin, T. Comparative study of transcranial magneto-acoustic stimulation and transcranial ultrasound stimulation of motor cortex. Front. Behav. Neurosci. 2019, 13, 241. [Google Scholar] [CrossRef]
- Zhang, S.; Guo, Z.; Xu, Y.; Mi, J.; Liu, J.; Li, Z.; Xie, X.; Xu, G. Transcranial magneto-acoustic stimulation improves spatial memory and modulates hippocampal neural oscillations in a mouse model of Alzheimer’s disease. Front. Neurosci. 2024, 18, 1313639. [Google Scholar] [CrossRef]
- Chu, F.; Tan, R.; Wang, X.; Zhou, X.; Ma, R.; Ma, X.; Li, Y.; Liu, R.; Zhang, C.; Liu, X.; et al. Transcranial magneto-acoustic stimulation attenuates synaptic plasticity impairment through activation of Piezo1 in Alzheimer’s disease mouse model. Research 2023, 6, 0130. [Google Scholar] [CrossRef]
- Zhang, C.; Tan, R.; Zhou, X.; Wang, R.; Wang, X.; Ma, R.; Chu, F.; Li, Y.; Yin, T.; Liu, Z. Transcranial Magneto-Acoustic Stimulation protects synaptic rehabilitation from amyloid-beta plaques via regulation of microglial functions. Int. J. Mol. Sci. 2024, 25, 4651. [Google Scholar] [CrossRef]
- Zhang, T.; Pan, N.; Wang, Y.; Liu, C.; Hu, S. Transcranial focused ultrasound neuromodulation: A review of the excitatory and inhibitory effects on brain activity in human and animals. Front. Hum. Neurosci. 2021, 15, 749162. [Google Scholar] [CrossRef]
- Liu, R.; Ma, R.; Liu, X.; Zhou, X.; Wang, X.; Yin, T.; Liu, Z. A noninvasive deep brain stimulation method via temporal-spatial interference magneto-acoustic effect: Simulation and experimental validation. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2022, 69, 2474–2483. [Google Scholar] [CrossRef]
- Yuan, Y.; Li, X.; Wang, Z. Theoretical analysis of transcranial magneto acoustical stimulation. Front. Comput. Neurosci. 2016, 10, 35. [Google Scholar] [CrossRef]
- Mariappan, L.; Hu, G.; He, B. Magnetoacoustic tomography with magnetic induction: Bioimpedance reconstruction through vector source imaging. IEEE Trans. Med. Imaging 2013, 32, 619–627. [Google Scholar] [CrossRef]
- Zhu, K.; Zhou, X.; Liu, X.; Ma, R.; Wang, M.; Zhang, S.; Yin, T.; Liu, Z. The simulation and experimental validation of a novel noninvasive multi-target electrical stimulation method. Sci. Rep. 2025, 15, 12416. [Google Scholar] [CrossRef] [PubMed]
- Choi, H. Power Amplifier Design for Ultrasound Applications. Micromachines 2023, 14, 1342. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ma, R.; Wang, M.; Liu, R.; Wang, R.; Yin, T.; Zhou, X.; Liu, Z. A phased array ultrasonic transducer linear sinusoidal driving system for ultrasound neuromodulation. Appl. Acoust. 2024, 222, 109995. [Google Scholar] [CrossRef]
- Foster, P.; Huang, J.; Serb, A.; Stathopoulos, S.; Papavassiliou, C.; Prodromakis, T. An FPGA based system for generalised electron devices testing. Sci. Rep. 2022, 12, 13912. [Google Scholar] [CrossRef]
- Ren, L.M.; Xue, X. The design of high precision arbitrary waveform generator based on DDS technology and FPGA. J. Phys. Conf. Ser. 2021, 1820, 012010. [Google Scholar] [CrossRef]
- Guzmán Miranda, H.; Barriga Rivera, A. An arbitrary waveform neurostimulator for preclinical studies: Design and verification. Med. Biol. Eng. Comput. 2024, 63, 1143–1159. [Google Scholar] [CrossRef]
- Assef, A.; Maia, J.M.; Schneider, F.K.; Button, V.L.; Costa, E.T. A reconfigurable arbitrary waveform generator using PWM modulation for ultrasound research. Biomed. Eng. Online 2013, 12, 24. [Google Scholar] [CrossRef] [PubMed]
- Baig, M.T.; Johanning, M.; Wiese, A.; Heidbrink, S.; Ziolkowski, M.; Wunderlich, C. A scalable, fast, and multichannel arbitrary waveform generator. Rev. Sci. Instrum. 2013, 84, 124701. [Google Scholar] [CrossRef]
- Lathi, B.P.; Ding, Z. Modern Digital and Analog Communication Systems, 5th ed.; Oxford University Press: Oxford, UK, 2019. [Google Scholar]
- Bowler, R.; Warring, U.; Britton, J.W.; Sawyer, B.C.; Amini, J.M. Arbitrary waveform generator for quantum information processing with trapped ions. Rev. Sci. Instrum. 2013, 84, 033108. [Google Scholar] [CrossRef]
- Xie, Y.; Li, Z.; Diaconu, D.; Handagala, S.; Leeser, M.; Lin, X. LUTMUL: Exceed Conventional FPGA Roofline Limit by LUT-based efficient multiplication for neural network inference. In Proceedings of the ASP-DAC, Tokyo Odaiba Miraikan, Japan, 20–23 January 2025; pp. 713–719. [Google Scholar]
- Hao, X.; Fang, H.; Luo, W.; Zhang, B. Pulse Compression Shape-Based ADC/DAC Chain Synchronization Measurement Algorithm with Sub-Sampling Resolution. Sensors 2024, 24, 2831. [Google Scholar] [CrossRef]
- Hammond, C.; Bergman, H.; Brown, P. Pathological synchronization in Parkinson’s disease: Networks, models and treatments. Trends Neurosci. 2007, 30, 357–364. [Google Scholar] [CrossRef]
- Jiruska, P.; de Curtis, M.; Jefferys, J.G.R.; Schevon, C.A.; Schiff, S.J.; Schindler, K. Synchronization and desynchronization in epilepsy: Controversies and hypotheses. J. Physiol. 2013, 591, 787–797. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, S.; Wang, Y.; Yin, T.; Yang, Z.; Liu, Z. High-resolution transcranial electrical simulation for living mice based on magneto-acoustic effect. Front. Neurosci. 2019, 13, 1342. [Google Scholar] [CrossRef]
- Blackmore, J.; Shrivastava, S.; Sallet, J.; Butler, C.R.; Cleveland, R.O. Ultrasound neuromodulation: A review of results, mechanisms and safety. Ultrasound Med. Biol. 2019, 45, 1509–1536. [Google Scholar] [CrossRef]
- Prieto, M.L.; Oralkan, Ö.; Khuri-Yakub, B.T.; Maduke, M.C. Dynamic response of model lipid membranes to ultrasonic radiation force. PLoS ONE 2013, 8, e77115. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Liu, Y.; Qu, X.; Shi, B.; Zheng, Q.; Lin, X.; Chao, S.; Wang, C.; Zhou, J.; Sun, Y.; et al. Ultra-Stretchable and Fast Self-Healing Ionic Hydrogel in Cryogenic Environments for Artificial Nerve Fiber. Adv. Mater. 2022, 34, 2105416. [Google Scholar] [CrossRef] [PubMed]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Shi, Y.; Li, Z.; Liu, R.; Zhang, X.; Wang, M.; Ma, R.; Yin, T.; Zhou, X.; Liu, Z. A Multi-Channel AM-TMAS Driving System Based on Amplitude-Modulated Sine Waves. Bioengineering 2026, 13, 405. https://doi.org/10.3390/bioengineering13040405
Shi Y, Li Z, Liu R, Zhang X, Wang M, Ma R, Yin T, Zhou X, Liu Z. A Multi-Channel AM-TMAS Driving System Based on Amplitude-Modulated Sine Waves. Bioengineering. 2026; 13(4):405. https://doi.org/10.3390/bioengineering13040405
Chicago/Turabian StyleShi, Yiheng, Ze Li, Ruixu Liu, Xiyang Zhang, Mingpeng Wang, Ren Ma, Tao Yin, Xiaoqing Zhou, and Zhipeng Liu. 2026. "A Multi-Channel AM-TMAS Driving System Based on Amplitude-Modulated Sine Waves" Bioengineering 13, no. 4: 405. https://doi.org/10.3390/bioengineering13040405
APA StyleShi, Y., Li, Z., Liu, R., Zhang, X., Wang, M., Ma, R., Yin, T., Zhou, X., & Liu, Z. (2026). A Multi-Channel AM-TMAS Driving System Based on Amplitude-Modulated Sine Waves. Bioengineering, 13(4), 405. https://doi.org/10.3390/bioengineering13040405






