Viscoelastic Properties of Porcine Pericardium Under Biaxial Tensile Creep and Stress Relaxation: Application for Novel Aortic Valve Bioprosthesis Design
Abstract
1. Introduction
2. Materials and Methods
2.1. Tissue Acquisition
2.2. Experimental Equipment
Cellscale Biotester 5000
2.3. Biaxial Creep Testing
2.4. Biaxial Stress Relaxation
2.5. Viscoelastic Testing Analysis
2.5.1. Biaxial Tensile Creep Analysis
2.5.2. Biaxial Tensile Stress Relaxation Analysis
2.5.3. Error Terms
2.5.4. Statistical Analysis
3. Results
3.1. Viscoelastic Tests (Creep and Stress Relaxation)
3.1.1. Biaxial Creep Tests
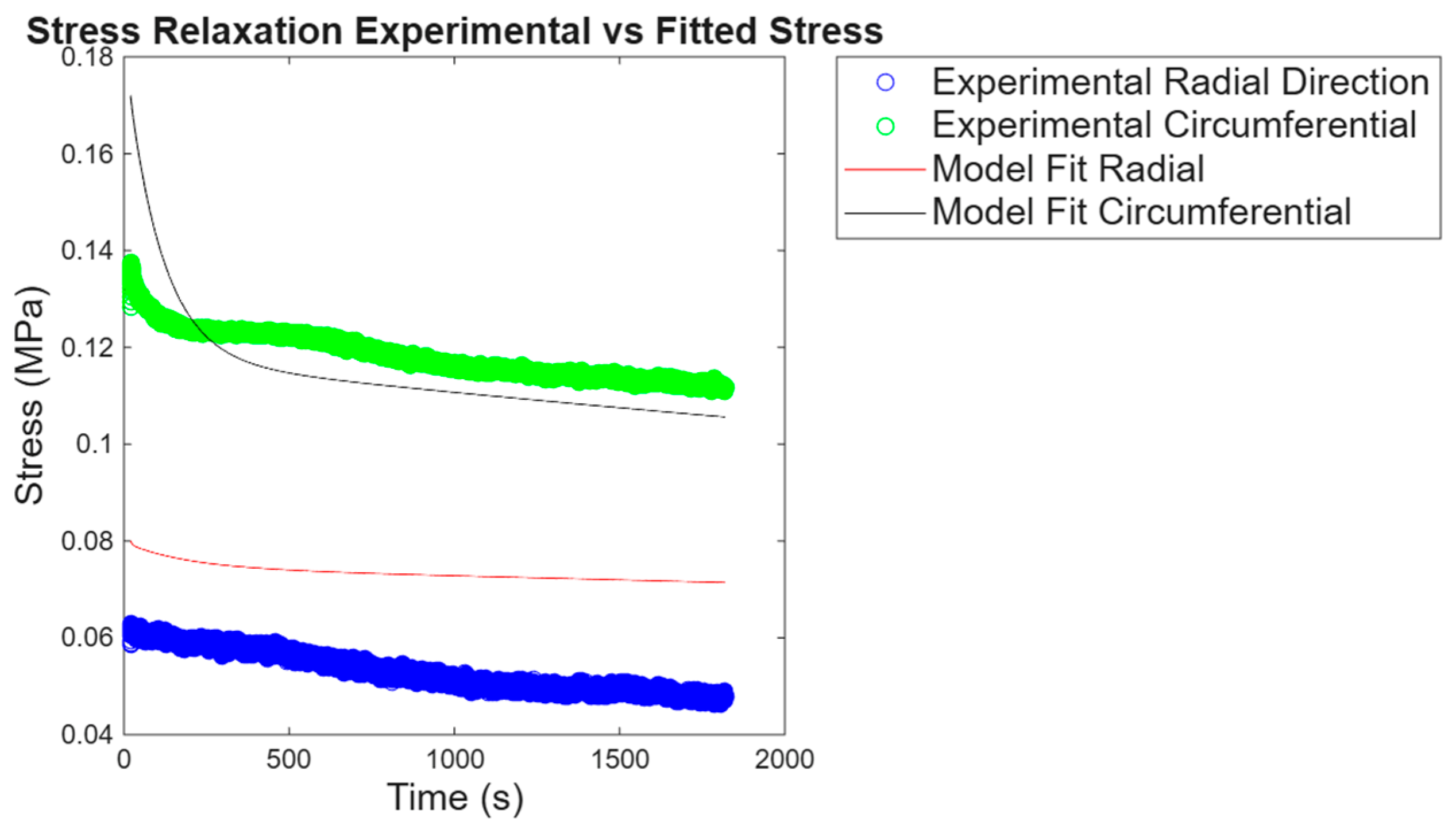
3.1.2. Biaxial Stress Relaxation
3.2. Viscoelastic Material Parameters
Generalized Kelvin–Voigt Viscoelastic Model Used for Creep Test Results, and Generalized Maxwell Model Used for Stress Relaxation
3.3. Statistical Analysis
3.3.1. Creep Test Statistical Analysis
3.3.2. Stress Relaxation Statistical Analysis
4. Discussion
4.1. Biaxial Tensile Creep
4.2. Biaxial Tensile Stress Relaxation
4.3. Clinical Significance and Implications
| Study Focus | Key Finding on Porcine Valves | Patient Population/Follow-Up | Citation |
|---|---|---|---|
| 20-Year Outcomes (Porcine vs. Pericardial) | Lower reintervention rate for porcine valves (19% vs. 26% at 20 years). Equivalent long-term survival. | A total of 1306 SAVR patients. Mean age 68. Twenty-year follow-up. | [50] |
| Long-Term Performance (Porcine vs. Pericardial) | Significantly better freedom from SVD for porcine valves at 10 years (98.0% vs. 96.3%). | A total of 3983 SAVR patients. Median follow-up of 10.4 years. | [51] |
| Pulmonic Position Durability (Porcine vs. Pericardial) | Porcine valves had lower reoperation rates and better long-term valve function. | A total of 258 cases (pulmonic position). Mean age 14.9. Median follow-up 10.5 years. | [44] |
| New Valve Feasibility (α-Gal-Free Porcine Pericardium) | New porcine pericardial valve (Pulsta) showed good short-term effectiveness and safety. | Ten patients (pulmonic position). Six-month follow-up. | [52] |
| Mechanical vs. Biological (General) | Biological valves, in general, are increasingly used and show good long-term outcomes, avoiding anticoagulation risks. | A total of 45,639 AVR/MVR patients. Up to 15-year follow-up. | [53] |
4.4. Future Application of Results
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kwiecinski, J.; Tzolos, E.; Cartlidge, T.R.; Fletcher, A.; Doris, M.K.; Bing, R.; Tarkin, J.M.; Seidman, M.A.; Gulsin, G.S.; Cruden, N.L.; et al. Native Aortic Valve Disease Progression and Bioprosthetic Valve Degeneration in Patients with Transcatheter Aortic Valve Implantation. Circulation 2021, 144, 1396–1408. [Google Scholar] [CrossRef]
- Kuchumov, A.G.; Makashova, A.; Vladimirov, S.; Borodin, V.; Dokuchaeva, A. Fluid–Structure Interaction Aortic Valve Surgery Simulation: A Review. Fluids 2023, 8, 295. [Google Scholar] [CrossRef]
- Mylonas, K.S.; Angouras, D.C. Bioprosthetic Valves for Lifetime Management of Aortic Stenosis: Pearls and Pitfalls. J. Clin. Med. 2023, 12, 7063. [Google Scholar] [CrossRef]
- Rahmani, B.; McGregor, C.; Byrne, G.; Burriesci, G. A Durable Porcine Pericardial Surgical Bioprosthetic Heart Valve: A Proof of Concept. J. Cardiovasc. Transl. Res. 2019, 12, 331–337. [Google Scholar] [CrossRef]
- Williams, D.F.; Bezuidenhout, D.; de Villiers, J.; Human, P.; Zilla, P. Long-Term Stability and Biocompatibility of Pericardial Bioprosthetic Heart Valves. Front. Cardiovasc. Med. 2021, 8, 728577. [Google Scholar] [CrossRef]
- Al-Atassi, T.; Toeg, H.; Ruel, M. Should we anticoagulate after bioprosthetic aortic valve replacement? Expert Rev. Cardiovasc. Ther. 2013, 11, 1649–1657. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, H.C.; Pibarot, P.; Wu, C.; Hahn, R.T.; Tang, G.H.; Abbas, A.E.; Playford, D.; Ruel, M.; Jilaihawi, H.; Sathananthan, J.; et al. Bioprosthetic Aortic Valve Hemodynamics: Definitions, Outcomes, and Evidence Gaps. J. Am. Coll. Cardiol. 2022, 80, 527–544. [Google Scholar] [CrossRef]
- Huang, X.; Wei, B.; Chen, L.; Yang, L.; Zheng, C.; Wang, Y. Degeneration mechanisms and advancements in optimization for preparation and crosslinking strategy of pericardium-based bioprosthetic heart valves. Acta Biomater. 2025, 201, 51–74. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Li, S.; Lei, Y.; Sun, M.; Wu, C.; Wang, X.; Xiong, S.; Wang, Q.; Kang, S.; Chen, S.; et al. Advancements and Perspectives in the Bioprosthetic Heart Valve: A Comprehensive Review on Biomaterial Processing and Emerging Polymeric Materials. J. Am. Heart Assoc. 2025, 14, e043061. [Google Scholar] [CrossRef] [PubMed]
- Alhadrami, H.A.; Syed, R.u.R.; Zahid, A.A.; Ahmed, R.; Hasan, S.; Hasan, A. Structure and Rheological Properties of Bovine Aortic Heart Valve and Pericardium Tissue: Implications in Bioprosthetic and Tissue-Engineered Heart Valves. J. Healthc. Eng. 2019, 2019, 290370. [Google Scholar] [CrossRef]
- Pil, N.; Kuchumov, A.G. Algorithmic Generation of Parameterized Geometric Models of the Aortic Valve and Left Ventricle. Sensors 2024, 25, 11. [Google Scholar] [CrossRef]
- Zhang, J.; He, R.; Wu, J.; Fan, Z.; Liu, D.; Gleadall, A.; Zhao, L.; Li, S. Lesion-specific design and oversizing as strategies for TAVI optimization: A patient-specific finite element study. Mater. Des. 2026, 262, 115476. [Google Scholar] [CrossRef]
- Kuchumov, A.G.; Khoroshev, D.V.; Selivanov, G.D.; Seleznev, M.A.; Tsilibin, V.K.; Kutuzova, A.M.; Barbashina, E.A.; Shilova, E.K.; Krestyaninov, O.V.; Baranov, A.A. Study of Bending Stiffness of Transcatheter Aortic Valve Stents. Mech. Solids 2026, 1–38. [Google Scholar] [CrossRef]
- Patel, S.P.; Garcia, S.; Sathananthan, J.; Tang, G.H.; Albaghdadi, M.S.; Pibarot, P.; Cubeddu, R.J. Structural Valve Deterioration in Transcatheter Aortic Bioprostheses: Diagnosis, Pathogenesis, and Treatment. Struct. Heart 2023, 7, 100155. [Google Scholar] [CrossRef] [PubMed]
- Onishchenko, P.; Klyshnikov, K.; Ovcharenko, E. Optimization of biological heart valve prosthesis ‘UniLine’: New tools for improving function. Russ. J. Biomech. 2024, 28, 6–16. [Google Scholar] [CrossRef]
- Pfaller, M.R.; Hörmann, J.M.; Weigl, M.; Nagler, A.; Chabiniok, R.; Bertoglio, C.; Wall, W.A. The importance of the pericardium for cardiac biomechanics: From physiology to computational modeling. Biomech. Model. Mechanobiol. 2019, 18, 503–529. [Google Scholar] [CrossRef]
- Mao, Y.; Liu, F.; Chen, X.; Liu, Y.; Tang, J.; Yang, J. Innovation and Development in Polymer Heart Valves: A New Era of Transcatheter Aortic Valve Replacement. Eur. Cardiol. Rev. 2025, 20, e29. [Google Scholar] [CrossRef] [PubMed]
- Hughes, C.; Whelan, A.; O’Reilly, D.; Campbell, E.M.; Lally, C. Aortic valve leaflet assessment to inform novel bioinspired materials: Understanding the impact of collagen fibres on the tissue’s mechanical behaviour. J. Mech. Behav. Biomed. Mater. 2025, 163, 106881. [Google Scholar] [CrossRef]
- Tornifoglio, B.; Hughes, C.; Digeronimo, F.; Guendouz, Y.; Johnston, R.D.; Lally, C. Imaging the microstructure of the arterial wall—Ex Vivo to In Vivo potential. Acta Biomater. 2025, 199, 18–34. [Google Scholar] [CrossRef]
- Armfield, D.; Boxwell, S.; McNamara, L.; Cook, S.; Conway, S.; Celikin, M.; Cardiff, P. Effect of bioprosthetic leaflet anisotropy on stent dynamics of Transcatheter Aortic Valve Replacement devices. J. Mech. Behav. Biomed. Mater. 2024, 157, 106650. [Google Scholar] [CrossRef]
- Suárez, S.; López-Campos, J.A.; Segade, A.; Veiga, C.G.; Jiménez, V.A. An study on the influence of collagen fiber directions in TAVs performance using FEM. J. Mech. Behav. Biomed. Mater. 2022, 126, 104969. [Google Scholar] [CrossRef]
- Rassoli, A.; Fatouraee, N.; Guidoin, R. Structural Model for Viscoelastic Properties of Pericardial Bioprosthetic Valves. Artif. Organs 2018, 42, 630–639. [Google Scholar] [CrossRef]
- Masoumi, S.F.; Rassoli, A.; Changizi, S.; Ravaghi, S.; Fatouraee, N. Comparative analysis of ovine and human aortic valve tissue for bioprosthetic valve development using relaxation tests and numerical simulation. Sci. Rep. 2026, 16, 7315. [Google Scholar] [CrossRef]
- Vorotnikov, G.S.; Rovinskii, B.M. Stress relaxation, creep, and uniaxial strain: General and special features. J. Appl. Mech. Tech. Phys. 1971, 7, 13–17. [Google Scholar] [CrossRef]
- Stella, J.A.; Liao, J.; Sacks, M.S. Time-dependent biaxial mechanical behavior of the aortic heart valve leaflet. J. Biomech. 2007, 40, 3169–3177. [Google Scholar] [CrossRef] [PubMed]
- Laville, C.; Acosta-Santamaria, V.; Trabelsi, O.; Avril, S.; Tillier, Y. Mechanical characterization of aortic valve tissues using an inverse analysis approach. Comput. Methods Biomech. Biomed. Eng. 2015, 18, 1976–1977. [Google Scholar] [CrossRef] [PubMed]
- Eckert, C.E.; Fan, R.; Mikulis, B.; Barron, M.; Carruthers, C.A.; Friebe, V.M.; Vyavahare, N.R.; Sacks, M.S. On the biomechanical role of glycosaminoglycans in the aortic heart valve leaflet. Acta Biomater. 2013, 9, 4653–4660. [Google Scholar] [CrossRef] [PubMed]
- Converse, G.L.; Armstrong, M.; Quinn, R.W.; Buse, E.E.; Cromwell, M.L.; Moriarty, S.J.; Lofland, G.K.; Hilbert, S.L.; Hopkins, R.A. Effects of cryopreservation, decellularization and novel extracellular matrix conditioning on the quasi-static and time-dependent properties of the pulmonary valve leaflet. Acta Biomater. 2012, 8, 2722–2729. [Google Scholar] [CrossRef]
- Anssari-Benam, A.; Tseng, Y.T.; Holzapfel, G.A.; Bucchi, A. Rate-dependency of the mechanical behaviour of semilunar heart valves under biaxial deformation. Acta Biomater. 2019, 88, 120–130. [Google Scholar] [CrossRef]
- Mira, S.E.; García-Briega, M.I.; Ribelles, J.L.G.; Munera, L.M.R. Viscoelastic Properties of Acellular Matrices of Porcine Esophageal Mucosa and Comparison with Acellular Matrices of Porcine Small Intestine Submucosa and Bovine Pericardium. Materials 2024, 17, 134. [Google Scholar] [CrossRef]
- Pandelani, T.; Semakane, L.; Msibi, M.; Kuchumov, A.G.; Nemavhola, F. Passive biaxial mechanical properties of sheep myocardium. Front. Bioeng. Biotechnol. 2025, 13, 1549829. [Google Scholar] [CrossRef]
- Nemavhola, F.; Ngwangwa, H.; Pandelani, T. Experimental analysis and biaxial biomechanical behaviour of ex-vivo sheep trachea. bioRxiv 2021. [Google Scholar] [CrossRef]
- Pierlot, C.M.; Moeller, A.D.; Lee, J.M.; Wells, S.M. Biaxial Creep Resistance and Structural Remodeling of the Aortic and Mitral Valves in Pregnancy. Ann. Biomed. Eng. 2015, 43, 1772–1785. [Google Scholar] [CrossRef]
- Ngwangwa, H.; Pandelani, T.; Msibi, M.; Mabuda, I.; Semakane, L.; Nemavhola, F. Biomechanical analysis of sheep oesophagus subjected to biaxial testing including hyperelastic constitutive model fitting. Heliyon 2022, 8, e09312. [Google Scholar] [CrossRef] [PubMed]
- Robinson, P.S.; Tranquillo, R.T. Planar Biaxial Behavior of Fibrin-Based Tissue-Engineered Heart Valve Leaflets. Tissue Eng. Part A Res. Adv. 2009, 15, 2763–2772. [Google Scholar] [CrossRef] [PubMed]
- Serra-Aguila, A.; Puigoriol-Forcada, J.M.; Reyes, G.; Menacho, J. Viscoelastic models revisited: Characteristics and interconversion formulas for generalized Kelvin–Voigt and Maxwell models. Acta Mech. Sin./Lixue Xuebao 2019, 35, 1191–1209. [Google Scholar] [CrossRef]
- Anssari-Benam, A.; Bucchi, A.; Bader, D.L. Unified viscoelasticity: Applying discrete element models to soft tissues with two characteristic times. J. Biomech. 2015, 48, 3128–3134. [Google Scholar] [CrossRef][Green Version]
- Anssari-Benam, A.; Bader, D.L.; Screen, H.R.C. Anisotropic time-dependant behaviour of the aortic valve. J. Mech. Behav. Biomed. Mater. 2011, 4, 1603–1610. [Google Scholar] [CrossRef]
- Tuladhar, S.R.; Mulderrig, S.; Della Barbera, M.; Vedovelli, L.; Bottigliengo, D.; Tessari, C.; Jockenhoevel, S.; Gregori, D.; Thiene, G.; Korossis, S.; et al. Bioengineered percutaneous heart valves for transcatheter aortic valve replacement: A comparative evaluation of decellularised bovine and porcine pericardia. Mater. Sci. Eng. C 2021, 123, 111936. [Google Scholar] [CrossRef]
- Noble, C.; Kamykowski, M.; Lerman, A.; Young, M. Rate-dependent and relaxation properties of porcine aortic heart valve biomaterials. IEEE Open J. Eng. Med. Biol. 2020, 1, 197–202. [Google Scholar] [CrossRef]
- Anssari-Benam, A.; Screen, H.R.C.; Bucchi, A. Insights into the micromechanics of stress-relaxation and creep behaviours in the aortic valve. J. Mech. Behav. Biomed. Mater. 2019, 93, 230–245. [Google Scholar] [CrossRef] [PubMed]
- Grashow, J.S.; Sacks, M.S.; Liao, J.; Yoganathan, A.P. Planar biaxial creep and stress relaxation of the mitral valve anterior leaflet. Ann. Biomed. Eng. 2006, 34, 1509–1518. [Google Scholar] [CrossRef]
- Mazine, A.; Chu, A.; Rocha, R.; Lee, D.S.; Rao, V.; Yau, T.M.; David, T.E.; Ouzounian, M. 2025 Heart Valve Society Annual Meeting Abstracts. J. Heart Valve Soc. 2025, 2, AC10. [Google Scholar]
- Kwak, J.G.; Bang, J.H.; Cho, S.; Kim, E.R.; Shih, B.C.H.; Kim, W.H. Long-term durability of bioprosthetic valves in pulmonary position: Pericardial versus porcine valves. J. Thorac. Cardiovasc. Surg. 2020, 160, 476–484. [Google Scholar] [CrossRef]
- Côté, N.; Pibarot, P.; Clavel, M.-A. Incidence, risk factors, clinical impact, and management of bioprosthesis structural valve degeneration. Curr. Opin. Cardiol. 2017, 32, 123–129. [Google Scholar] [CrossRef]
- Tan, T.; Cholewa, N.M.; Case, S.W.; De Vita, R. Micro-structural and Biaxial Creep Properties of the Swine Uterosacral–Cardinal Ligament Complex. Ann. Biomed. Eng. 2016, 44, 3225–3237. [Google Scholar] [CrossRef]
- Pil, N.; Kuchumov, A.G.; Nemavhola, F.; Pandelani, T.; Ha, T.S. Mechano-electrical-fluid interaction left-ventricle model for numerical evaluation of aortic valve hemodynamics. Front. Bioeng. Biotechnol. 2026, 13, 1713023. [Google Scholar] [CrossRef] [PubMed]
- Fedotova, P.; Pil, N.; Kuchumov, A.G.; Barbashina, E.; Tsilibin, V.; Nemavhola, F.; Pandelani, T.; Kadyraliev, B.; Ha, T.S. Numerical Simulation of Aortic Valve Leaflets Calcification Influence on Hemodynamic Performance Using Fluid–Structure Interaction Approach. Processes 2025, 13, 3750. [Google Scholar] [CrossRef]
- Pil, N.; Kuchumov, A.G.; Kadyraliev, B.; Arutunyan, V. Influence of Aortic Valve Leaflet Material Model on Hemodynamic Features in Healthy and Pathological States. Mathematics 2023, 11, 428. [Google Scholar] [CrossRef]
- Mazine, A.; Ouzounian, M. Optimizing Aortic Valve Replacement Through the Hawthorne Effect. Ann. Thorac. Surg. 2024, 117, 1162–1163. [Google Scholar] [CrossRef]
- Anselmi, A.; Daniel, M.; Aymami, M.; Chabanne, C.; Rosier, S.; Mancini, J.; Verhoye, J.P. Comparison of Long-Term Performance of Porcine versus Pericardial Bioprostheses. Thorac. Cardiovasc. Surg. 2025, 74, 181–190. [Google Scholar] [CrossRef]
- Kim, G.B.; Song, M.K.; Bae, E.J.; Park, E.A.; Lee, W.; Lim, H.G.; Kim, Y.J. Successful Feasibility Human Trial of a New Self-Expandable Percutaneous Pulmonary Valve (Pulsta Valve) Implantation Using Knitted Nitinol Wire Backbone and Trileaflet α-Gal–Free Porcine Pericardial Valve in the Native Right Ventricular Outflow Tract. Circ. Cardiovasc. Interv. 2018, 11, e006494. [Google Scholar] [CrossRef] [PubMed]
- Goldstone, A.B.; Chiu, P.; Baiocchi, M.; Lingala, B.; Patrick, W.L.; Fischbein, M.P.; Woo, Y.J. Mechanical or Biologic Prostheses for Aortic-Valve and Mitral-Valve Replacement. N. Engl. J. Med. 2017, 377, 1847–1857. [Google Scholar] [CrossRef] [PubMed]
- Fedorova, N.V.; Kosinov, A.M. Determination Mechanical Properties and Permeability of Porous Titanium Alloy Bone Implants, Including Under Conditions of Their Interaction with Biological Fluids. Russ. J. Biomech. 2024, 28, 45–56. [Google Scholar] [CrossRef]




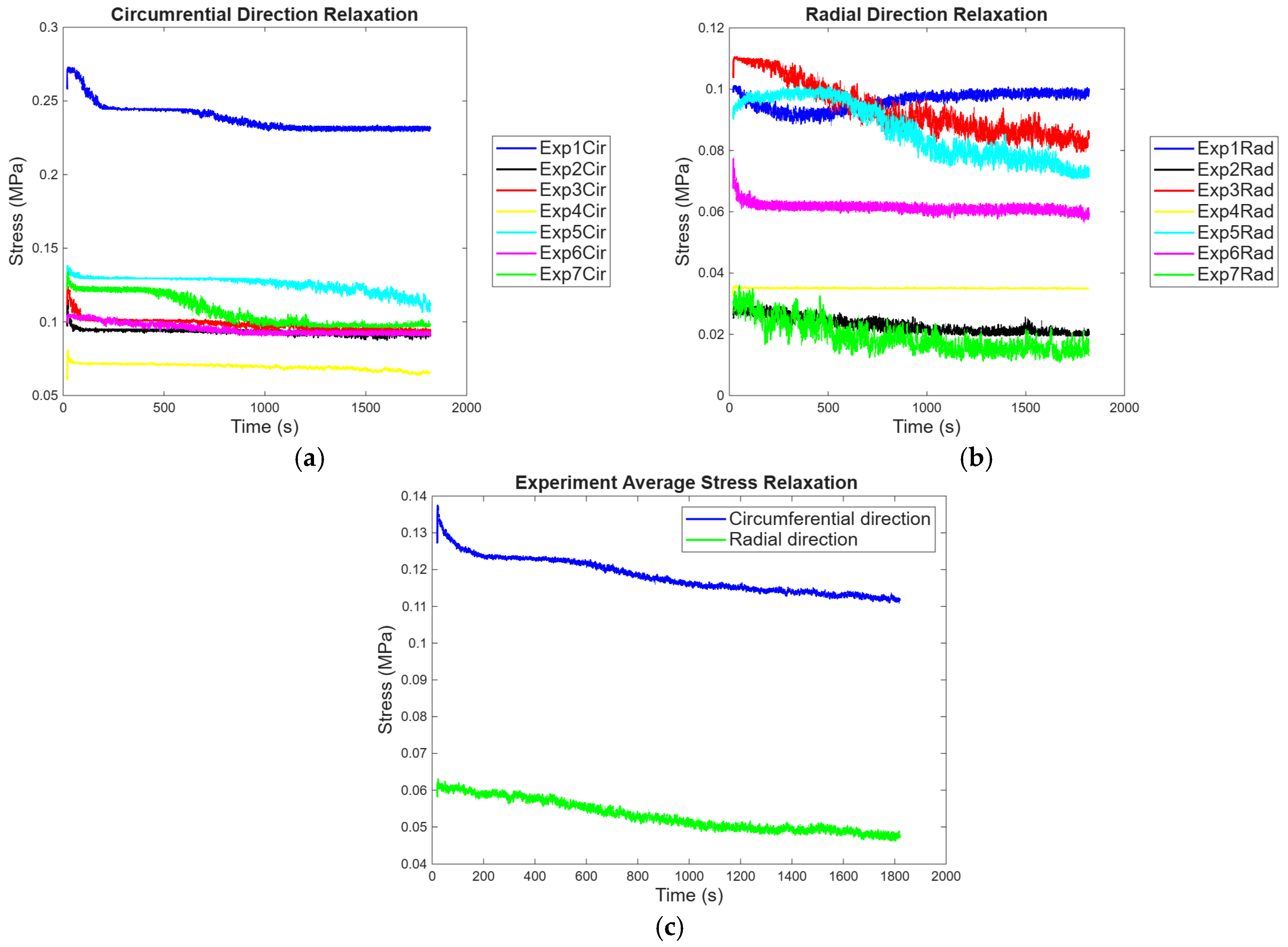
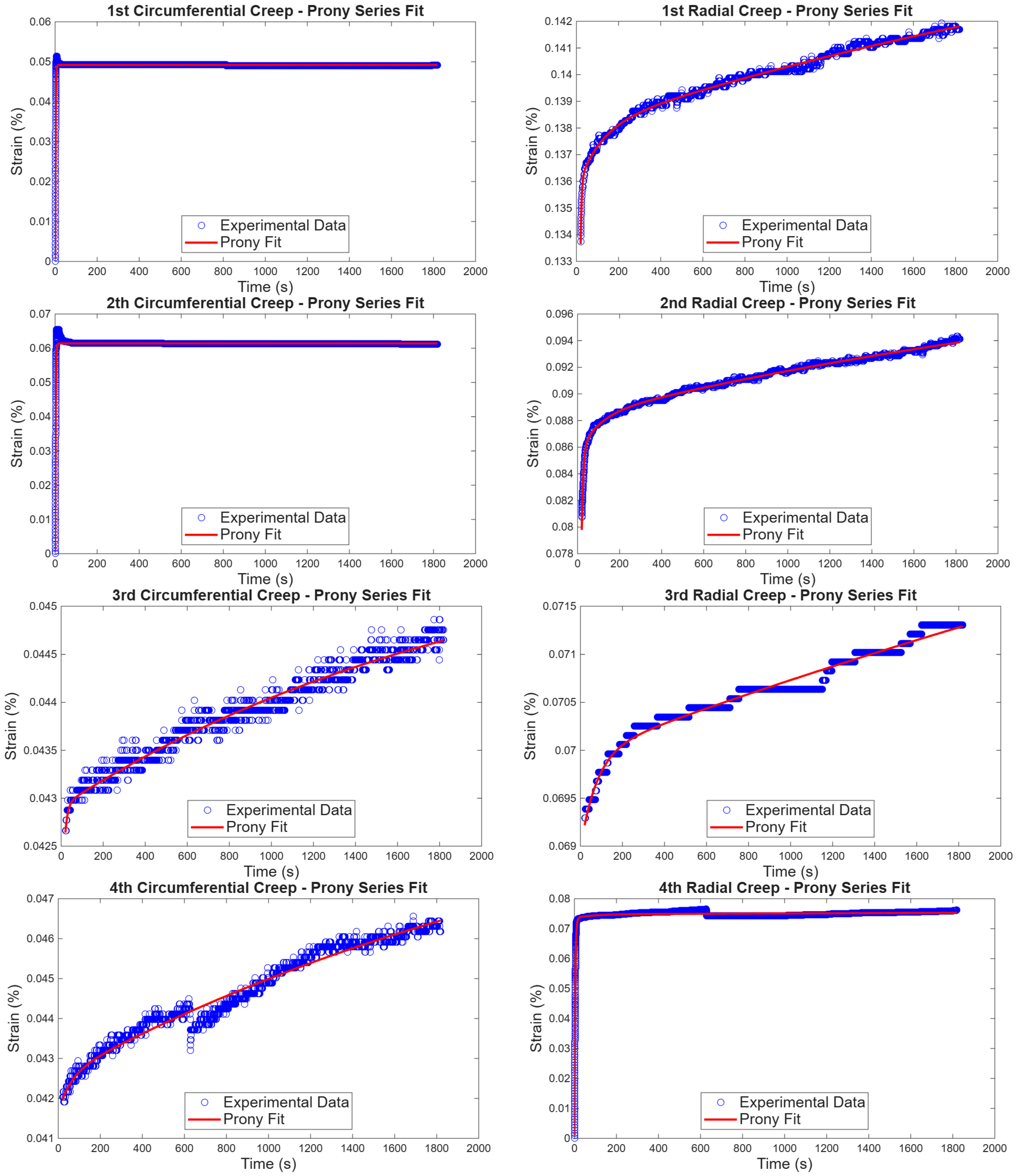

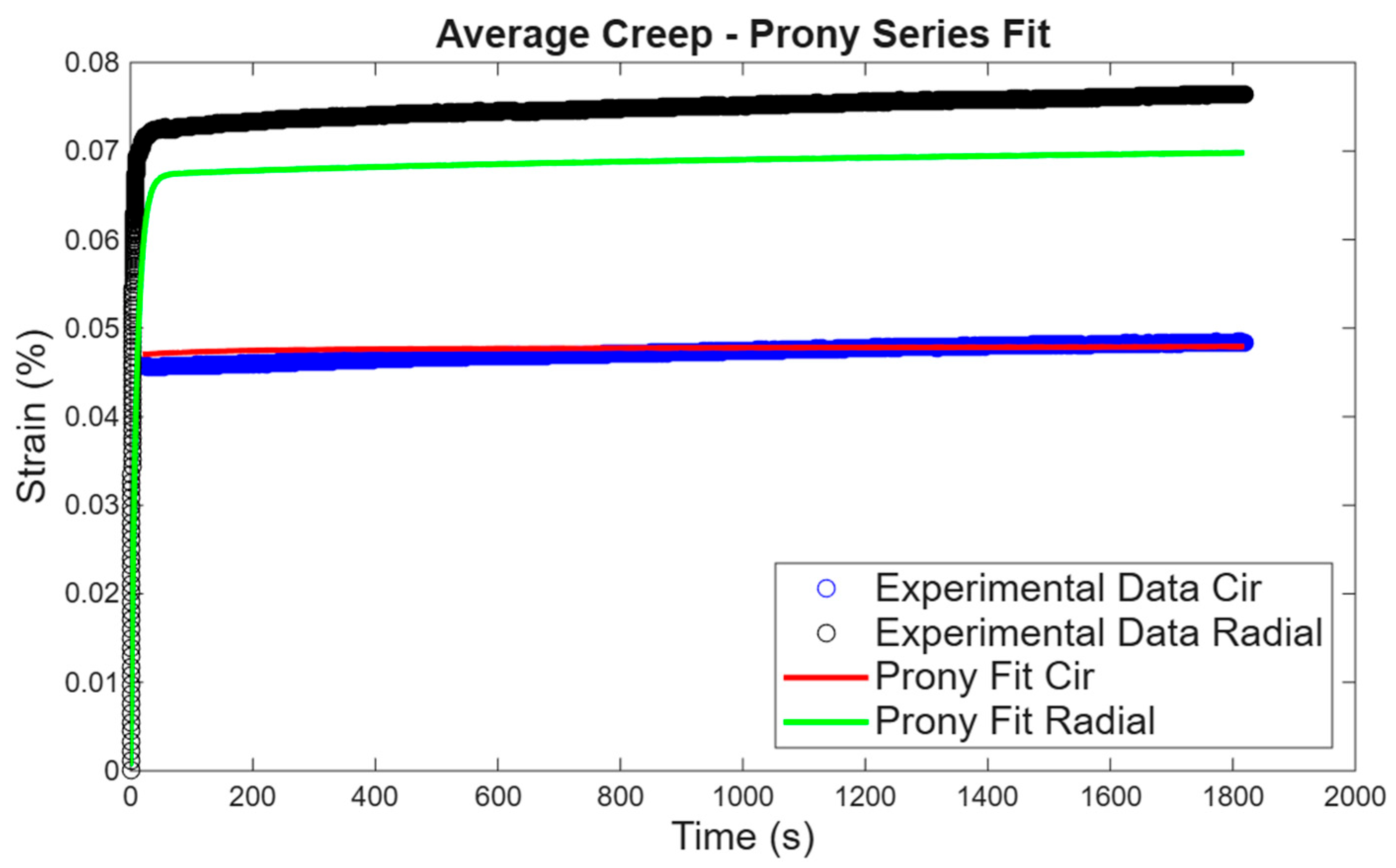

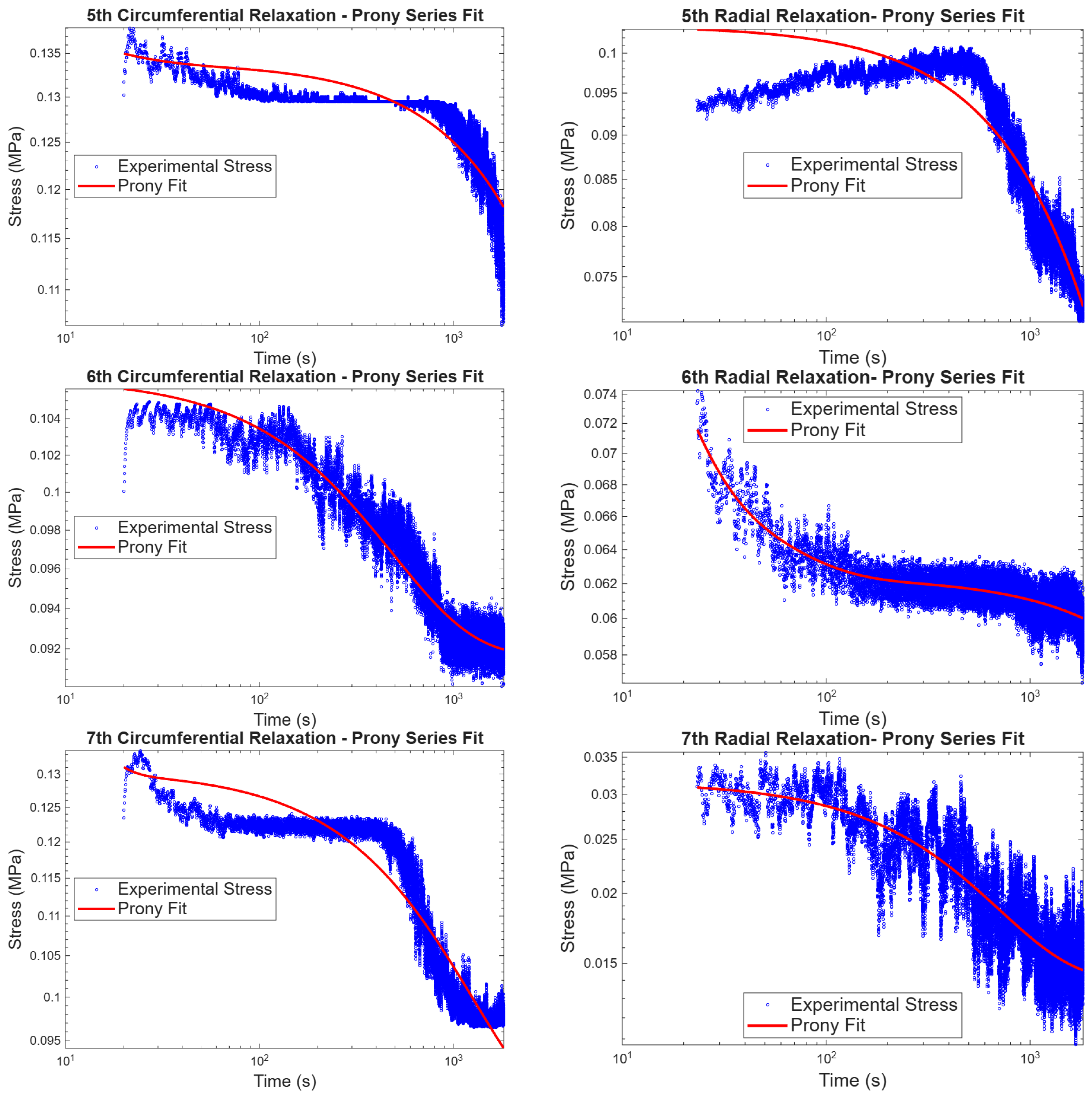
| Creep_1 | Creep_2 | Creep_3 | Creep_4 | Creep_5 | Creep_6 | Creep_7 | Average | |
|---|---|---|---|---|---|---|---|---|
| 2 × 103 | 1 × 103 | 2 × 103 | 2 × 103 | 1 × 103 | 7 × 102 | 1 × 103 | 1 × 103 | |
| 78.13 | 83.95 | 79.54 | 63.40 | 59.51 | 27.88 | 45.08 | 65.40 | |
| 2.01 | 2.36 | 0.56 | 0.61 | 2.98 | 3.18 | 0.78 | 1.95 | |
| 0.00 | 0.00 | 3.06 | 1.37 | 0.00 | 1.09 | 0.13 | 0.92 | |
| 80.56 | 97.25 | 12.22 | 57.79 | 476.73 | 152.74 | 40.63 | 146.22 | |
| 0.02 | 0.11 | 5.79 | 12.92 | 4.73 | 49.94 | 4.82 | 12.25 | |
| 2 × 105 | 1 × 105 | 2 × 103 | 3 × 103 | 9 × 102 | 2 × 104 | 3 × 103 | 5 × 104 | |
| R2 | 0.94 | 0.90 | 0.97 | 0.97 | 0.99 | 1.00 | 0.96 | 0.96 |
| r | 0.97 | 0.95 | 0.98 | 0.99 | 1.00 | 1.00 | 0.98 | 0.98 |
| ASE | 3 × 10−3 | 9 × 10−3 | 2 × 10−4 | 1 × 10−3 | 2 × 10−4 | 3 × 10−4 | 3 × 10−4 | 2 × 10−3 |
| NRMSE | 0.01 | 0.01 | 0.04 | 0.04 | 0.03 | 0.01 | 0.07 | 0.03 |
| Creep_1 | Creep_2 | Creep_3 | Creep_4 | Creep_5 | Creep_6 | Creep_7 | Average | |
|---|---|---|---|---|---|---|---|---|
| 9 × 102 | 8 × 102 | 7 × 102 | 8 × 102 | 8 × 103 | 1 × 103 | 8 × 102 | 2 × 103 | |
| 123.88 | 63.64 | 50.37 | 58.20 | 467.10 | 51.61 | 42.88 | 122.53 | |
| 5.00 | 7.67 | 0.12 | 3.29 | 47.34 | 0.46 | 4.40 | 9.76 | |
| 2.12 | 2.26 | 0.74 | 0.72 | 0.01 | 3.06 | 1.02 | 1.42 | |
| 1 × 102 | 9 × 101 | 8 × 101 | 3 × 101 | 2 × 103 | 2 × 101 | 2 × 102 | 4 × 102 | |
| 9.90 | 10.19 | 5.05 | 4.26 | 29.33 | 7.87 | 3.36 | 9.99 | |
| 4 × 103 | 3 × 103 | 8 × 103 | 1 × 103 | 1 × 104 | 4 × 103 | 2 × 103 | 5 × 103 | |
| R2 | 0.99 | 0.99 | 0.98 | 0.99 | 0.90 | 0.99 | 0.99 | 0.98 |
| r | 1.00 | 1.00 | 0.99 | 0.99 | 0.95 | 0.99 | 0.99 | 0.99 |
| ASE | 4 × 10−4 | 5 × 10−4 | 1 × 10−4 | 3 × 10−5 | 2 × 10−3 | 3 × 10−4 | 3 × 10−4 | 5 × 10−4 |
| NRMSE | 0.01 | 0.01 | 0.03 | 0.03 | 0.07 | 0.03 | 0.02 | 0.03 |
| Rela_1 | Rela_2 | Rela_3 | Rela_4 | Rela_5 | Rela_6 | Rela_7 | Average | |
|---|---|---|---|---|---|---|---|---|
| 0.22 | 0.02 | 0.00 | 0.01 | 0.00 | 0.09 | 0.09 | 0.06 | |
| 4 × 10−3 | 3 × 10−4 | 4 × 10−2 | 2 × 10−1 | 5 × 10−3 | 1 × 10−4 | 1 × 10−3 | 4 × 10−2 | |
| 0.41 | 0.32 | 28.64 | 0.82 | 5.54 | 0.04 | 1.28 | 5.29 | |
| 0.04 | 0.00 | 0.00 | 0.07 | 0.01 | 0.00 | 0.33 | 0.06 | |
| 66.44 | 615.13 | 33.01 | 8.79 | 12.87 | 14.89 | 3.62 | 107.82 | |
| 0.03 | 0.01 | 0.10 | 0.06 | 0.13 | 0.01 | 0.04 | 0.06 | |
| 1 × 103 | 9 × 102 | 2 × 104 | 2 × 104 | 1 × 104 | 5 × 102 | 1 × 103 | 7 × 103 | |
| R2 | 0.94 | 0.89 | 0.93 | 0.88 | 0.77 | 0.93 | 0.91 | 0.89 |
| r | 0.97 | 0.94 | 0.97 | 0.94 | 0.88 | 0.96 | 0.95 | 0.94 |
| ASE | 0.13 | 0.02 | 0.03 | 0.01 | 0.17 | 0.03 | 0.29 | 0.10 |
| NRMSE | 0.05 | 0.08 | 0.03 | 0.04 | 0.08 | 0.07 | 0.09 | 0.06 |
| Rela_1 | Rela_2 | Rela_3 | Rela_4 | Rela_5 | Rela_6 | Rela_7 | Average | |
|---|---|---|---|---|---|---|---|---|
| 0.08 | 0.00 | 0.08 | 0.03 | 0.00 | 0.01 | 0.01 | 0.03 | |
| 0.00 | 8 × 10−2 | 4 × 10−6 | 1 × 10−3 | 2 × 10−3 | 2 × 10−1 | 6 × 10−3 | 4 × 10−2 | |
| 5.60 | 12.16 | 0.02 | 10.53 | 0.48 | 6.36 | 1.17 | 5.19 | |
| 1 × 10−2 | 3 × 10−3 | 0.E+00 | 3 × 10−4 | 0.00 | 9 × 10−3 | 1 × 10−2 | 5 × 10−3 | |
| 108.00 | 14.43 | 517.19 | 60.23 | 5.76 | 42.74 | 541.09 | 184.20 | |
| 0.01 | 0.09 | 0.03 | 0.00 | 0.10 | 0.06 | 0.01 | 0.04 | |
| 6 × 104 | 3 × 104 | 8 × 102 | 3 × 104 | 5 × 103 | 4 × 104 | 6 × 102 | 2 × 104 | |
| R2 | 0.87 | 0.83 | 0.95 | 0.23 | 0.89 | 0.72 | 0.79 | 0.75 |
| r | 0.94 | 0.91 | 0.97 | 0.48 | 0.94 | 0.85 | 0.89 | 0.85 |
| ASE | 0.01 | 0.02 | 0.11 | 0.00 | 0.27 | 0.02 | 0.14 | 0.08 |
| NRMSE | 0.08 | 0.03 | 0.06 | 0.06 | 0.11 | 0.04 | 0.09 | 0.07 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Matjeka, E.; Kuchumov, A.G.; Ngwangwa, H.M.; Pandelani, T.; Nemavhola, F. Viscoelastic Properties of Porcine Pericardium Under Biaxial Tensile Creep and Stress Relaxation: Application for Novel Aortic Valve Bioprosthesis Design. Bioengineering 2026, 13, 401. https://doi.org/10.3390/bioengineering13040401
Matjeka E, Kuchumov AG, Ngwangwa HM, Pandelani T, Nemavhola F. Viscoelastic Properties of Porcine Pericardium Under Biaxial Tensile Creep and Stress Relaxation: Application for Novel Aortic Valve Bioprosthesis Design. Bioengineering. 2026; 13(4):401. https://doi.org/10.3390/bioengineering13040401
Chicago/Turabian StyleMatjeka, Edward, Alex G. Kuchumov, Harry M. Ngwangwa, Thanyani Pandelani, and Fulufhelo Nemavhola. 2026. "Viscoelastic Properties of Porcine Pericardium Under Biaxial Tensile Creep and Stress Relaxation: Application for Novel Aortic Valve Bioprosthesis Design" Bioengineering 13, no. 4: 401. https://doi.org/10.3390/bioengineering13040401
APA StyleMatjeka, E., Kuchumov, A. G., Ngwangwa, H. M., Pandelani, T., & Nemavhola, F. (2026). Viscoelastic Properties of Porcine Pericardium Under Biaxial Tensile Creep and Stress Relaxation: Application for Novel Aortic Valve Bioprosthesis Design. Bioengineering, 13(4), 401. https://doi.org/10.3390/bioengineering13040401








