Demonstration of Alpha-Band Entrainment via Low-Field Magnetic Stimulation: A Simulation-Driven Proof of Concept
Abstract
1. Introduction
2. Materials and Methods
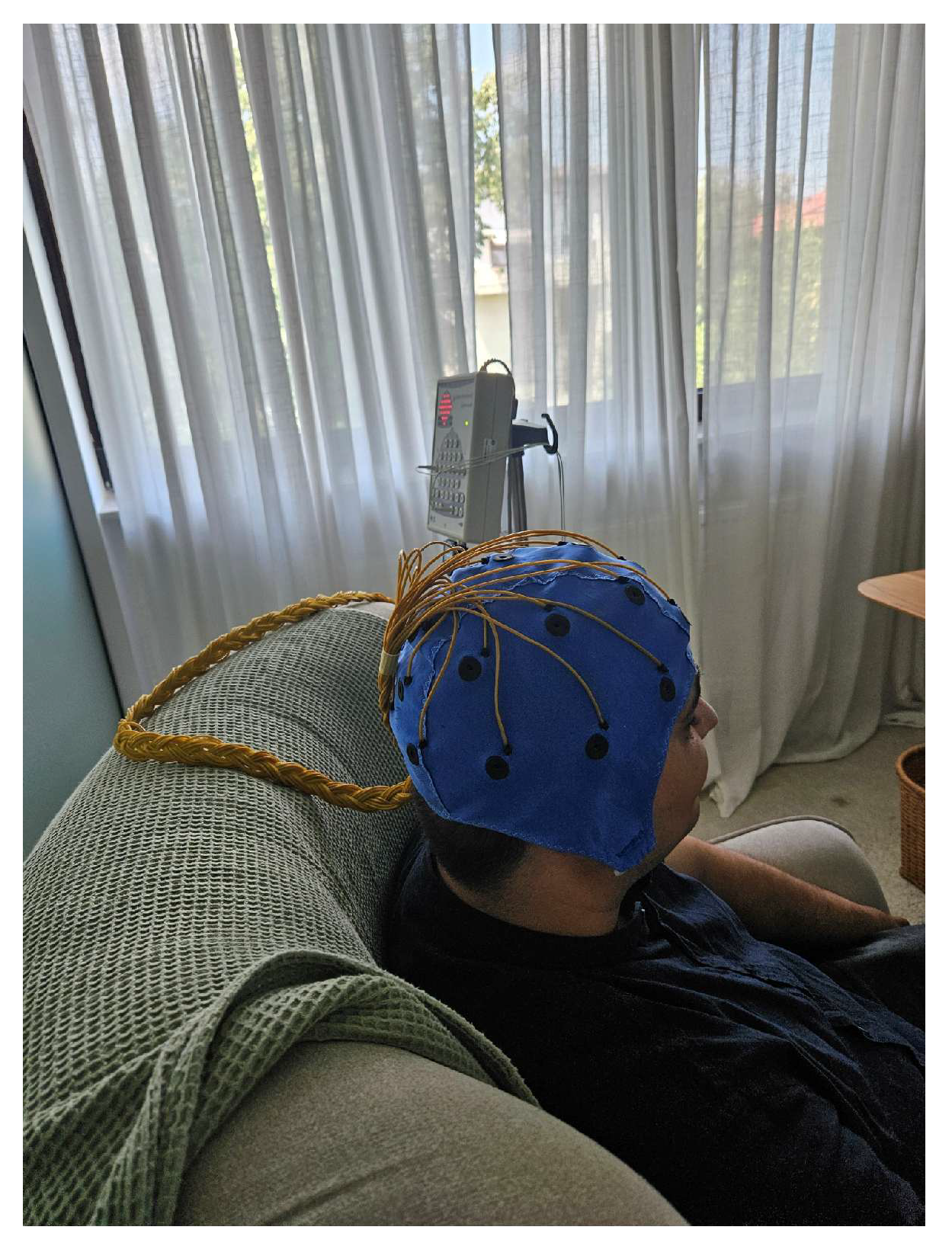
2.1. Neuromodulation Device Description
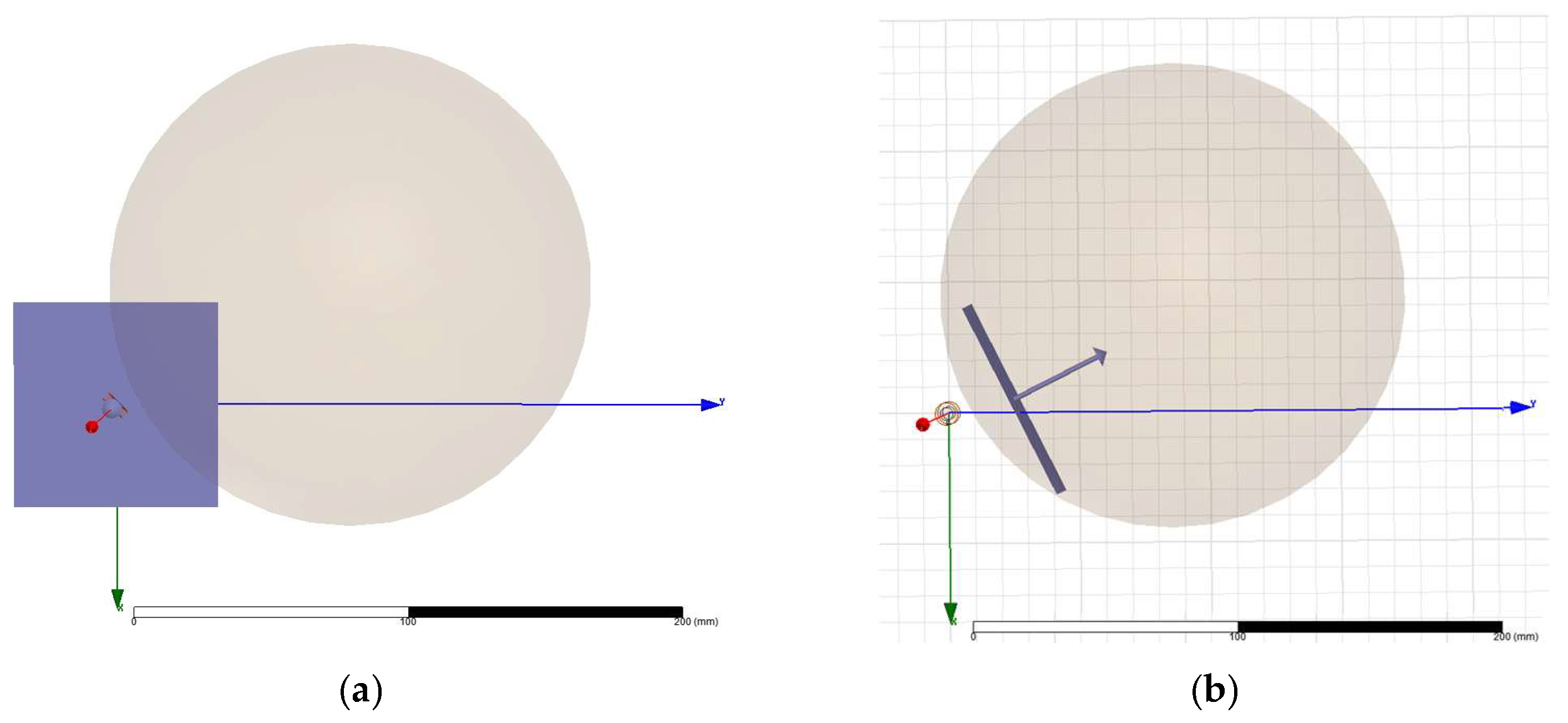
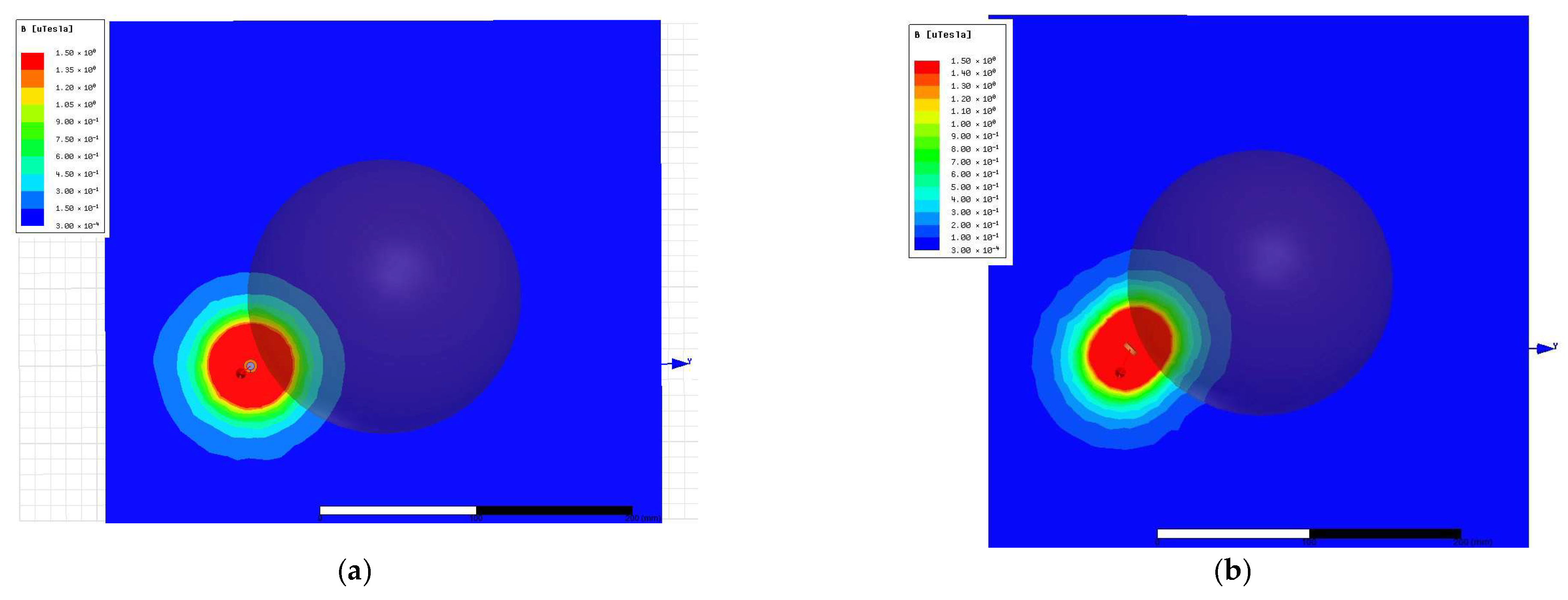
2.2. Magnetic Stimulation Transducer—Cylindrical vs. Figure-of-Eight Designs
2.3. Magnetic Stimulation of Brain Tissue
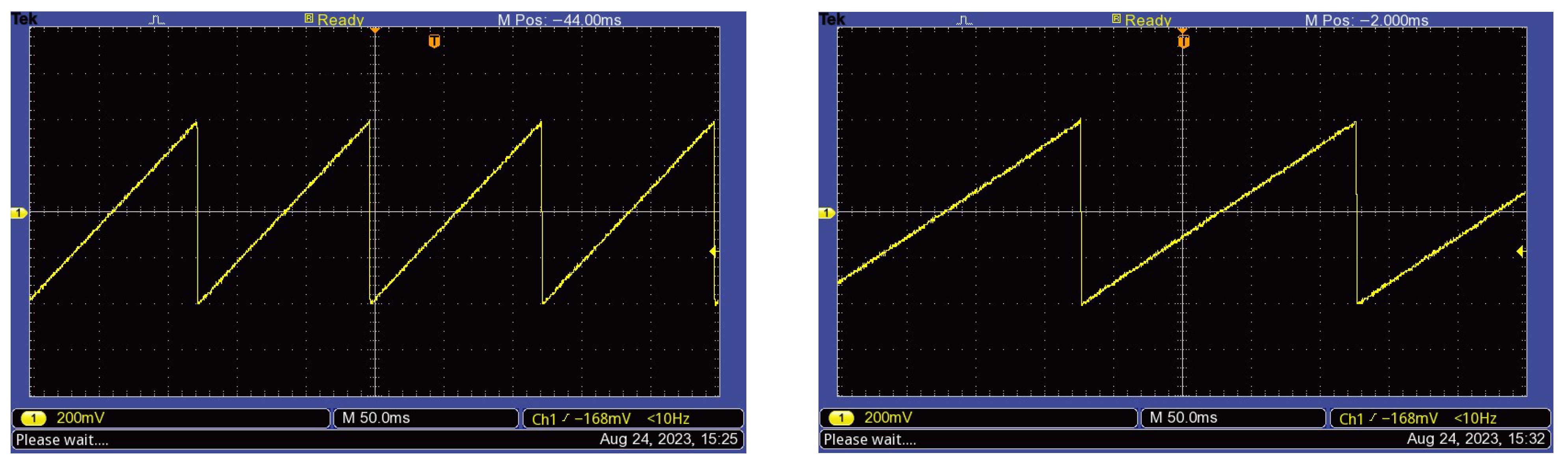
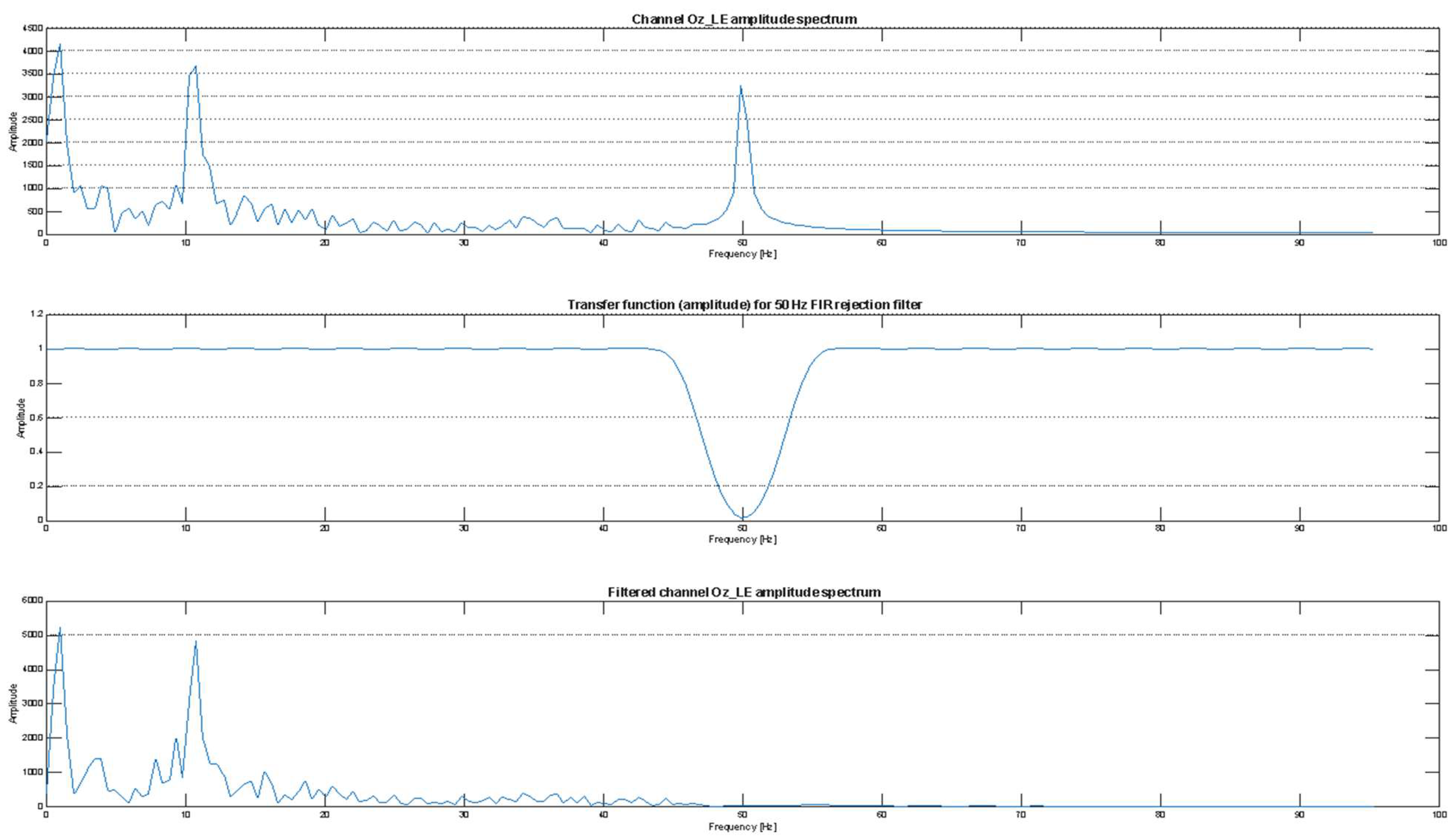
2.4. Testing Protocol and Signal Processing
3. Optimization Results
3.1. Magnetic Stimulation Transducer Analysis and Optimization
3.2. Brainwave Entrainment EEG Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Doelling, K.A.M. Neural oscillations are a start toward understanding brain activity rather than the end. PLoS Biol. 2021, 19, e3001234. [Google Scholar] [CrossRef]
- Marcantoni, I.; Assogna, R.; Del Borrello, G.; Di Stefano, M.; Morano, M.; Romagnoli, S.; Leoni, C.; Bruschi, G.; Sbrollini, A.; Morettini, M.; et al. Ratio Indexes Based on Spectral Electroencephalographic Brainwaves for Assessment of Mental Involvement: A Systematic Review. Sensors 2023, 23, 5968. [Google Scholar] [CrossRef]
- Gozali, A.A.; Nurhayati, I.K.; Rahmasari, G. EEG-Based Biosignal Analytics for Measuring English Speaking Anxiety. In Proceedings of the 2025 5th International Conference on Intelligent Cybernetics Technology & Applications (ICICyTA), Yogyakarta, Indonesia, 17–19 December 2025. [Google Scholar]
- Ippolito, G.; Bertaccini, R.; Tarasi, L.; Di Gregorio, F.; Trajkovic, J.; Battaglia, S.; Romei, V. The Role of Alpha Oscillations among the Main Neuropsychiatric Disorders in the Adult and Developing Human Brain: Evidence from the Last 10 Years of Research. Biomedicines 2022, 10, 3189. [Google Scholar] [CrossRef] [PubMed]
- Jensen, O.; Bonnefond, M. The alpha rhythm: From physiology to behaviour. Physiol. Rev. 2025, 106, 1123–1159. [Google Scholar] [CrossRef] [PubMed]
- Coffey, E.; Nicol, T.; White-Schwoch, T.; Chandrasekaran, B.; Krizman, J.; Skoe, E.; Zatorre, R.; Kraus, N. Evolving perspectives on the sources of the frequency-following response. Nat. Commun. 2019, 10, 5036. [Google Scholar] [CrossRef] [PubMed]
- Yadav, G.; Cidral-Filho, F.; Iyer, R. Using Heartfulness Meditation and Brainwave Entrainment to Improve Teenage Mental Wellbeing. Front. Psychol. 2021, 12, 742892. [Google Scholar] [CrossRef]
- Goodin, P.; Ciorciari, J.; Baker, K.; Carey, A.; Harper, M.; Kaufman, J. A high-density EEG investigation into steady state binaural beat stimulation. PLoS ONE 2012, 7, e34789. [Google Scholar] [CrossRef]
- Basu, S.; Banerjee, B. Exploring the impact of visual brainwave entrainment on working memory in pediatric ADHD. J. Neurol. Sci. 2025, 480, 124043. [Google Scholar] [CrossRef]
- Vöröslakos, M.; Takeuchi, Y.; Brinyiczki, K.; Zombori, T.; Oliva, A.; Fernández-Ruiz, A.; Kozák, G.; Kincses, Z.T.; Iványi, B.; Buzsáki, G. Direct effects of transcranial electric stimulation on brain circuits in rats and humans. Nat. Commun. 2018, 9, 483. [Google Scholar] [CrossRef]
- Foldes, S.; Jensen, A.; Jacobson, A.; Vassall, S.; Foldes, E.; Guthery, A.; Brown, D.; Levine, T.; Tyler, W.J.; Frye, R.E. Transdermal Electrical Neuromodulation for Anxiety and Sleep Problems in High-Functioning Autism Spectrum Disorder: Feasibility and Preliminary Findings. J. Pers. Med. 2021, 11, 1307. [Google Scholar] [CrossRef]
- Jackson, M.; Rahman, A.; Lafon, B.; Kronberg, G.; Ling, D.; Parra, L.; Bikson, M. Animal models of transcranial direct current stimulation: Methods and mechanisms. Clin. Neurophysiol. 2016, 127, 3425–3454. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Majdi, A.; Asamoah, B.; Laughlin, M.M. Neural Mechanisms of tDCS: Insights from an In-Vivo Rodent Model Across a Range of Electric Field Strengths. Brain Stimul. 2026, 19, 103053. [Google Scholar] [CrossRef]
- Zhang, S.; Qin, Y.; Wang, J.; Yu, Y.; Wu, L.; Zhang, T. Noninvasive Electrical Stimulation Neuromodulation and Digital Brain Technology: A Review. Biomedicines 2023, 11, 1513. [Google Scholar] [CrossRef]
- Siebner, H.; Funke, K.; Aberra, A.; Antal, A.; Bestmann, S.; Chen, R.; Classen, J.; Davare, M.; Di Lazzaro, V.; Fox, P.T.; et al. Transcranial magnetic stimulation of the brain: What is stimulated?—A consensus and critical position paper. Clin. Neurophysiol. 2022, 140, 59–97. [Google Scholar] [CrossRef]
- Chail, A.; Saini, R.; Bhat, P.; Srivastava, K.; Chauhan, V. Transcranial magnetic stimulation: A review of its evolution and current applications. Ind. Psychiatry J. 2018, 27, 172–180. [Google Scholar] [CrossRef]
- Larsen, E.; Licht, R.; Nielsen, R.; Lolk, A.; Borck, B.; Sørensen, C.; Christensen, E.M.; Bizik, G.; Ravn, J.; Martiny, K.; et al. Transcranial pulsed electromagnetic fields for treatment-resistant depression: A multicenter 8-week single-arm cohort study. Eur. Psychiatry 2020, 63, e18. [Google Scholar] [CrossRef]
- Rohan, M.; Yamamoto, R.; Ravichandran, C.; Cayetano, K.R.; Morales, O.G.; Olson, D.P.; Vitaliano, G.; Paul, S.M.; Cohen, B.M. Rapid mood-elevating effects of low field magnetic stimulation in depression. Biol. Psychiatry 2014, 76, 186–193. [Google Scholar] [CrossRef]
- Leuchter, A.; Cook, I.; Feifel, D.; Goethe, J.W.; Husain, M.; Carpenter, L.L.; Thase, M.E.; Krystal, A.D.; Philip, N.S.; Bhati, M.T.; et al. Efficacy and Safety of Low-field Synchronized Transcranial Magnetic Stimulation (sTMS) for Treatment of Major Depression. Brain Stimul. 2015, 8, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Messina, G.; Monda, A.; Messina, A.; Di Maio, G.; Monda, V.; Limone, P.; Dipace, A.; Monda, M.; Polito, R.; Moscatelli, F. Relationship between Non-Invasive Brain Stimulation and Autonomic Nervous System. Biomedicines 2024, 12, 972. [Google Scholar] [CrossRef]
- Reato, D.; Rahman, A.; Bikson, M.; Parra, L. Low-intensity electrical stimulation affects network dynamics by modulating population rate and spike timing. J. Neurosci. 2010, 30, 15067–15079. [Google Scholar] [CrossRef] [PubMed]
- Ladenbauer, J.; Obermayer, K. Weak electric fields promote resonance in neuronal spiking activity: Analytical results from two-compartment cell and network models. PLoS Comput. Biol. 2019, 15, e1006974. [Google Scholar] [CrossRef]
- Guidetti, M.; Arlotti, M.; Bocci, T.; Bianchi, A.; Parazzini, M.; Ferrucci, R.; Priori, A. Electric Fields Induced in the Brain by Transcranial Electric Stimulation: A Review of In Vivo Recordings. Biomedicines 2022, 10, 2333. [Google Scholar] [CrossRef]
- Gomez-Tames, J.; Hamasaka, A.; Hirata, A.; Laakso, I.; Lu, M.; Ueno, S. Group-level analysis of induced electric field in deep brain regions by different TMS coils. Phys. Med. Biol. 2020, 65, 025007. [Google Scholar] [CrossRef]
- Thielscher, A.; Kammer, T. Electric field properties of two commercial figure-8 coils in TMS: Calculation of focality and efficiency. Clin. Neurophysiol. 2004, 115, 1697–1708. [Google Scholar] [CrossRef]
- Ueno, S.; Sekino, M. Figure-Eight Coils for Magnetic Stimulation: From Focal Stimulation to Deep Stimulation. Front. Hum. Neurosci. 2021, 15, 805971. [Google Scholar] [CrossRef]
- Zhang, Z.; Ai, W.; Deng, B.; Han, W.; Wang, J. Improved Figure-of-Eight Coil for Transcranial Magnetic Stimulation Using Magnetic Resonant Coupling. IEEE Trans. Magn. 2017, 53, 119100605. [Google Scholar] [CrossRef]
- Yu, L.; Xiong, W.; Zhang, H.; Zhao, W.; Yuan, Y.; Liang, Z. Paramatic design of transranial magnetic stimulation coil based on VB and Ansoft Maxwell. In Proceedings of the 2017 Chinese Automation Congress (CAC), Jinan, China, 20–22 October 2017; pp. 4484–4488. [Google Scholar] [CrossRef]
- Liu, S.; Kuwahata, A.; Sekino, M. Design of a Multi-Locus Transcranial Magnetic Stimulation Coil With a Single Driver. URSI Radio Sci. Lett. 2020, 2, 1–5. [Google Scholar] [CrossRef]
- Lazutkin, D.; Husar, P. Modeling of electromagnetic stimulation of the human brain. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology, Buenos Aires, Argentina, 31 August–4 September 2010; pp. 581–584. [Google Scholar] [CrossRef]
- Herwig, U.; Satrapi, P.; Schönfeldt-Lecuona, C. Using the International 10-20 EEG System for Positioning of Transcranial Magnetic Stimulation. Brain Topogr. 2003, 16, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Dowsett, J.; Herrmann, C. Transcranial Alternating Current Stimulation with Sawtooth Waves: Simultaneous Stimulation and EEG Recording. Front. Hum. Neurosci. 2016, 10, 135. [Google Scholar] [CrossRef]
- Dowsett, J. Enhancing Neural Oscillations with Sawtooth Wave Transcranial Alternating Current Stimulation; Carl von Ossietzky University: Oldenburg, Germany, 2018. [Google Scholar]
- Duyn, J.; Schenck, J. Contributions to magnetic susceptibility of brain tissue. NMR Biomed. 2017, 30, e3546. [Google Scholar] [CrossRef] [PubMed]
- Klohs, J.; Hirt, A. Investigation of the magnetic susceptibility properties of fresh and fixed mouse heart, liver, skeletal muscle and brain tissue. Phys. Med. 2021, 88, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Abhang, P.; Gawali, B.W.; Mehrotra, S.C. Technological Basics of EEG Recording and Operation of Apparatus. In Introduction to EEG- and Speech-Based Emotion Recognition; Academic Press: Cambridge, MA, USA, 2016; pp. 19–50. [Google Scholar] [CrossRef]
- Medisains, D. Bellabee. Available online: https://www.micaelasolution.com/product/63 (accessed on 15 May 2024).









| Electrical Parameter | Original Cylindrical Coil | Figure-of-Eight Coil |
|---|---|---|
| Inductance Ls | 434.8 μH | 579.6 μH |
| Resistance Rs | 2.24 Ω | 2.37 Ω |
| Freq. Band/Channel | Delta | Theta | Alpha | Beta | Gamma |
|---|---|---|---|---|---|
| O1_LE | 66.35 | 4.71 | 20.12 | 7.11 | 1.71 |
| O2_LE | 71.03 | 3.99 | 17.12 | 6.05 | 1.8 |
| Oz_LE | 70.58 | 4.67 | 16.24 | 6.72 | 1.8 |
| Freq. Band/Channel | Delta | Theta | Alpha | Beta | Gamma |
|---|---|---|---|---|---|
| O1_LE | 22.93 | 7.6 | 49.53 | 15.74 | 4.19 |
| O2_LE | 23.5 | 7.14 | 50.47 | 14.9 | 3.98 |
| Oz_LE | 25.75 | 7.1 | 48.82 | 14.96 | 3.36 |
| Freq. Band/Channel | Delta | Theta | Alpha | Beta | Gamma |
|---|---|---|---|---|---|
| O1_LE | −65.44 | 61.36 | 146.17 | 121.38 | 145.03 |
| O2_LE | −66.92 | 78.95 | 194.8 | 146.28 | 121.11 |
| Oz_LE | −63.52 | 52.03 | 200.62 | 122.62 | 86.67 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Dămășaru, C.; Roșu, G.; Tuță, L.; Cernian, A.; Rus, M. Demonstration of Alpha-Band Entrainment via Low-Field Magnetic Stimulation: A Simulation-Driven Proof of Concept. Bioengineering 2026, 13, 395. https://doi.org/10.3390/bioengineering13040395
Dămășaru C, Roșu G, Tuță L, Cernian A, Rus M. Demonstration of Alpha-Band Entrainment via Low-Field Magnetic Stimulation: A Simulation-Driven Proof of Concept. Bioengineering. 2026; 13(4):395. https://doi.org/10.3390/bioengineering13040395
Chicago/Turabian StyleDămășaru, Costin, Georgiana Roșu, Leontin Tuță, Alexandra Cernian, and Mihaela Rus. 2026. "Demonstration of Alpha-Band Entrainment via Low-Field Magnetic Stimulation: A Simulation-Driven Proof of Concept" Bioengineering 13, no. 4: 395. https://doi.org/10.3390/bioengineering13040395
APA StyleDămășaru, C., Roșu, G., Tuță, L., Cernian, A., & Rus, M. (2026). Demonstration of Alpha-Band Entrainment via Low-Field Magnetic Stimulation: A Simulation-Driven Proof of Concept. Bioengineering, 13(4), 395. https://doi.org/10.3390/bioengineering13040395








