Performance Evaluation of Chlorococcum sp. in Various Photobioreactor Designs: Impact on Biomass Production and Nutrient Removal
Abstract
1. Introduction
2. Materials and Methods
2.1. PBR Design and Experimental Setup
2.2. Cultivation Medium and Inoculation
2.3. Monitoring and Analytical Procedures
2.4. Geometric and Productivity Calculations
2.4.1. Surface-to-Volume Ratio (S/V)
2.4.2. Volumetric Productivity
2.4.3. Specific Electrical Energy Consumption per Biomass
2.5. Statistical Analysis
3. Results and Discussion
3.1. Biomass Growth Kinetics
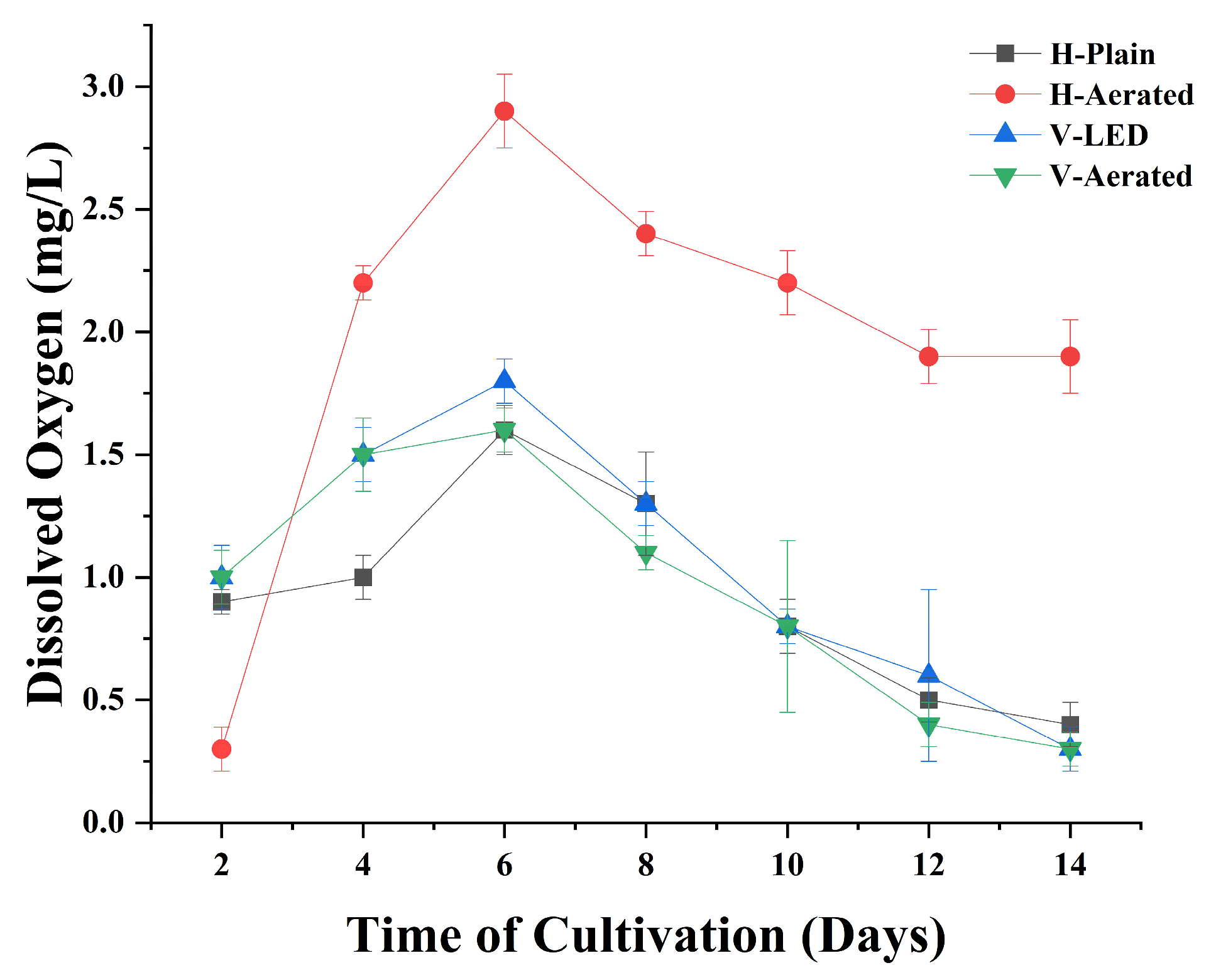
3.2. Dissolved Oxygen
3.3. Nutrient Removal
3.4. PBR Geometry Impact
3.5. Impact of PBR Materials on Chlorococcum sp. Cultivation Efficiency
3.6. Volumetric Productivity and Electrical Energy Consumption
3.7. Implications for Integrated Test Strategy and Scale-Up
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of variance |
| COD | Chemical oxygen demand |
| DO | Dissolved oxygen |
| H-Aerated | Horizontal photobioreactor with aeration |
| H-Plain | Horizontal photobioreactor without aeration |
| LED | Light-emitting diode |
| LEDs | Light-emitting diodes |
| NO3− | Nitrate ion |
| PBR | Photobioreactor |
| PBRs | Photobioreactors |
| PO43− | Phosphate ion |
| S/V | Surface-area-to-volume ratio |
| V-Aerated | Vertical photobioreactor with aeration |
| V-LED | Vertical photobioreactor with aeration and red LED lighting |
| kWh | Kilowatt-hour |
References
- Morsi, H.H.; El-Sheekh, M.M.; Eladel, H.; Al-Tuwaijri, M.M.; El-Sabbagh, S.M.; Maher, A.; Saber, A.A. Screening the Pollution-Tolerant Chlorococcum sp. (Chlorophyceae) Grown in Municipal Wastewater for Simultaneous Nutrient Removal and Biodiesel Production. Water 2023, 15, 1723. [Google Scholar] [CrossRef]
- Oyelami, S.; Azeez, N.A.; Adekanmi, A.A.; Adeleke, K.M.; Oyewo, A.T.; Adeyi, A.J. Production and Characterization of Biodiesel from Chlorococcum sp.: A Green Microalgae. Environ. Qual. Mgmt. 2023, 33, 387–396. [Google Scholar] [CrossRef]
- Chuechomsuk, S.; Thumthanaruk, B.; Vatanyoopaisarn, S.; Rungsardthong, V.; Kunyalung, W.; Mohamadnia, S.; Angelidaki, I. Enhancement of β-Cryptoxanthin Production in Three Different Green Microalgae Species Using an Innovative Red LED Wavelength Shift Approach. Appl. Sci. Eng. Prog. 2025, 18, 7707. [Google Scholar] [CrossRef]
- Laje, K.; Seger, M.; Dungan, B.; Cooke, P.; Polle, J.; Holguin, F.O. Phytoene Accumulation in the Novel Microalga Chlorococcum sp. Using the Pigment Synthesis Inhibitor Fluridone. Mar. Drugs 2019, 17, 187. [Google Scholar] [CrossRef]
- Menguy, E.; Dumontet, V.; Coulombier, N.; Meriot, V.; Déan, L.L.; Barthelemy, V.; Jauffrais, T. A Method to Assess Algicidal Activity of Microalgal Extracts Coupling Microalgae Produced in Stirred Closed Photobioreactor Operating in Continuous with Pulse Amplitude Modulated (PAM) Fluorometry. MethodsX 2020, 7, 101037. [Google Scholar] [CrossRef]
- Rifai, A.; Jamilah, S.; Admirasari, R.; Prayitno, J. Alternative Source of Nutrients for Microalgae Cultivation in a Photobioreactor System. IOP Conf. Ser. Earth Environ. Sci. 2022, 1034, 012005. [Google Scholar] [CrossRef]
- Trentin, G.; Barbera, E.; Bertucco, A.; Sforza, E. Role of Oxygen in Tubular Photobioreactors: Model-Based Design and Operating Conditions to Minimize Productivity Losses. Chem. Eng. Process. Process Intensif. 2020, 157, 108151. [Google Scholar] [CrossRef]
- Magnabosco, C.; Santaniello, G.; Romano, G. Microalgae: A Promising Source of Bioactive Polysaccharides for Biotechnological Applications. Molecules 2025, 30, 2055. [Google Scholar] [CrossRef] [PubMed]
- Hwang, T.G.; Kim, G.-Y.; Han, J.-I.; Kim, S.; Kim, J.P. Enhancement of Lipid Productivity of Chlorella sp. Using Light-Converting Red Fluorescent Films Based on Aggregation-Induced Emission. ACS Sustain. Chem. Eng. 2020, 8, 15888–15897. [Google Scholar] [CrossRef]
- Jung, C.H.G.; Waldeck, P.; Sykora, S.; Braune, S.; Petrick, I.; Küpper, J.-H.; Jung, F. Influence of Different Light-Emitting Diode Colors on Growth and Phycobiliprotein Generation of Arthrospira Platensis. Life 2022, 12, 895. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Purton, S.; Baganz, F. Characterisation of a Simple ‘Hanging Bag’ Photobioreactor for Low-cost Cultivation of Microalgae. J. Chem. Technol. Biotechnol. 2022, 97, 608–619. [Google Scholar] [CrossRef]
- Mathivanan, K.; Ameen, F.; Zhang, R.; Ravi, G.; Beduru, S. Bubble Column Photobioreactor (BCPR) for Cultivating Microalgae and Microalgal Consortium (Co-CC) with Additional CO2 Supply for Enhancing Biomass, Lipid, and Preferable Fatty Acids Production. Environ. Res. 2023, 238, 117284. [Google Scholar] [CrossRef] [PubMed]
- Mora-Sánchez, J.F.; Ribes, J.; González-Camejo, J.; Seco, A.; Ruano, M.V. Towards Optimisation of Microalgae Cultivation through Monitoring and Control in Membrane Photobioreactor Systems. Water 2023, 16, 155. [Google Scholar] [CrossRef]
- Hawrot-Paw, M.; Ratomski, P. Efficient Production of Microalgal Biomass—Step by Step to Industrial Scale. Energies 2024, 17, 944. [Google Scholar] [CrossRef]
- Omar, A.; Almomani, F.; Qiblawey, H.; Rasool, K. Advances in Nitrogen-Rich Wastewater Treatment: A Comprehensive Review of Modern Technologies. Sustainability 2024, 16, 2112. [Google Scholar] [CrossRef]
- Hashmi, Z.; Zaini, J.; Abdullah, R.; Abu Bakar, M.S.; Bilad, M.R. Effects of Light Color on Growth, Nutrient Uptake, and Harvesting of the Indigenous Strain of Chlorococcum sp. Bioresour. Technol. Rep. 2024, 28, 101966. [Google Scholar] [CrossRef]
- Hashmi, Z.; Idriss, I.M.; Zaini, J.; Abu Bakar, M.S.; Wibisono, Y.; Abdullah, R.; Bilad, M.R. Advancements in Membrane Modifications for Enhanced Microalgae Harvesting: A Comprehensive Review. Sep. Purif. Technol. 2025, 360, 131012. [Google Scholar] [CrossRef]
- Singh, R.S.; Chauhan, K.; Pandey, A. Influence of Aeration, Agitation and Process Duration on Fungal Inulinase Production from Paneer Whey in a Stirred Tank Reactor. Bioresour. Technol. Rep. 2019, 8, 100343. [Google Scholar] [CrossRef]
- Singh, R.N.; Sharma, S.; Singh, A.K.; Srivastava, N. Design and Development of a Simple Stirred Tank Photobioreactor for Algal Production. J. Solar Eneg. Res. Updat. 2021, 2, 24–26. [Google Scholar] [CrossRef]
- Hashmi, Z.; Idriss, I.M.; Taha, H.; Zaini, J.; Bakar, M.S.A.; Abdullah, R.; Nandianto, A.B.D.; Bilad, M.R. Cultivation of Indigenous Chlorococcum sp. in Aquaculture Wastewater under Various Light Color and Biomass Harvesting Using Membrane Filtration: A Simultaneous Wastewater Treatment and Biomass Production. Clean. Waste Syst. 2025, 10, 100209. [Google Scholar] [CrossRef]
- Romesburg, H.C. Pedagogical Positives of the Surface-to-Volume Ratio. Am. Biol. Teach. 2011, 73, 260–264. [Google Scholar] [CrossRef]
- Cornet, J.; Dussap, C. A Simple and Reliable Formula for Assessment of Maximum Volumetric Productivities in Photobioreactors. Biotechnol. Prog. 2009, 25, 424–435. [Google Scholar] [CrossRef]
- Sharma, V.; Abbas, R.; Sodré, J.R.; Ayad, S.M.M.E.; Belchior, C. Model for Energy Consumption and Costs of Bioethanol Production from Wastepaper. J. Sustain. Dev. Energy Water Environ. Syst. 2022, 10, 1100431. [Google Scholar] [CrossRef]
- Chunzhuk, E.A.; Grigorenko, A.V.; Kiseleva, S.V.; Chernova, N.I.; Vlaskin, M.S.; Ryndin, K.G.; Butyrin, A.V.; Ambaryan, G.N.; Dudoladov, A.O. Effects of Light Intensity on the Growth and Biochemical Composition in Various Microalgae Grown at High CO2 Concentrations. Plants 2023, 12, 3876. [Google Scholar] [CrossRef]
- Maltsev, Y.; Maltseva, K.; Kulikovskiy, M.; Maltseva, S. Influence of Light Conditions on Microalgae Growth and Content of Lipids, Carotenoids, and Fatty Acid Composition. Biology 2021, 10, 1060. [Google Scholar] [CrossRef]
- Nzayisenga, J.C.; Farge, X.; Groll, S.L.; Sellstedt, A. Effects of Light Intensity on Growth and Lipid Production in Microalgae Grown in Wastewater. Biotechnol. Biofuels 2020, 13, 4. [Google Scholar] [CrossRef] [PubMed]
- Ortiz Tena, F.; Ranglová, K.; Kubač, D.; Steinweg, C.; Thomson, C.; Masojidek, J.; Posten, C. Characterization of an Aerated Submerged Hollow Fiber Ultrafiltration Device for Efficient Microalgae Harvesting. Eng. Life Sci. 2021, 21, 607–622. [Google Scholar] [CrossRef] [PubMed]
- Almeida, C.J.; Carneiro, D.D.C.; Sales, E.A.; Lima, S.T.D.C. Biotechnological Assessment of Culture Conditions on the Stress-Induced Carotenoid Production of Dunaliella Salina and Growth Kinetics of Chlorophyceae Microalgae Strains. Braz. Arch. Biol. Technol. 2020, 63, e20190204. [Google Scholar] [CrossRef]
- Loomba, V.; Huber, G.; Von Lieres, E. Single-Cell Computational Analysis of Light Harvesting in a Flat-Panel Photo-Bioreactor. Biotechnol. Biofuels 2018, 11, 149. [Google Scholar] [CrossRef]
- Plouviez, M.; Bolot, P.; Shilton, A.; Guieysse, B. Phosphorus Uptake and Accumulation in Chlamydomonas Reinhardtii: Influence of Biomass Concentration, Phosphate Concentration, Phosphorus Depletion Time, and Light Supply. Algal Res. 2023, 71, 103085. [Google Scholar] [CrossRef]
- Wang, L.; Jia, X.; Xu, L.; Yu, J.; Ren, S.; Yang, Y.; Wang, K.; López-Arredondo, D.; Herrera-Estrella, L.; Lambers, H.; et al. Engineering Microalgae for Water Phosphorus Recovery to Close the Phosphorus Cycle. Plant Biotechnol. J. 2023, 21, 1373–1382. [Google Scholar] [CrossRef]
- Hashmi, Z.; Idriss, I.M.; Zaini, J.; Abu Bakar, M.S.; Bilad, M.R. Advancements in Photobioreactor Systems: Optimizing Operations for Enhanced Microalgal Growth and Bioremediation. Algal Res. 2025, 91, 104282. [Google Scholar] [CrossRef]
- Hu, H.; Wang, H.; Li, J.; Ma, L.; Shen, X.; Zeng, R.J. Evaluation of the Effect of Agitation Speed on the Growth and High-value LC-PUFA Formation of Porphyridium cruentum Based on Basic Rheological Analysis. J. Chem. Technol. Biotechnol. 2019, 94, 2158–2166. [Google Scholar] [CrossRef]
- Özer Uyar, G.E.; Mısmıl, N. Symbiotic Association of Microalgae and Plants in a Deep Water Culture System. PeerJ 2022, 10, e14536. [Google Scholar] [CrossRef]
- Samer, M. Photobiostimulation of Chlorella Sorokiniana Using Light Emitting Diodes (LEDs) for Increasing Lipid and Biodiesel Production. Egypt. J. Chem. 2021, 64, 5575–5583. [Google Scholar] [CrossRef]
- Riihiaho, K.A.; Lind, L.; Calderini, M.L.; Halonen, V.; Pölönen, I.; Salmi, P. Photobioreactor Design Utilizing Procedural Three-Dimensional Modelling and Ray Tracing. J. R. Soc. Interface 2025, 22, 20240451. [Google Scholar] [CrossRef] [PubMed]
- La Bella, E.L.; Occhipinti, P.S.; Puglisi, I.; Fragalà, F.; Saccone, R.; Russo, N.; Randazzo, C.L.; Caggia, C.; Baglieri, A. Comparative Phycoremediation Performance of Three Microalgae Species in Two Different Magnitude of Pollutants in Wastewater from Farmhouse. Sustainability 2023, 15, 11644. [Google Scholar] [CrossRef]
- Mao, Y.; Xiong, R.; Gao, X.; Jiang, L.; Peng, Y.; Xue, Y. Analysis of the Status and Improvement of Microalgal Phosphorus Removal from Municipal Wastewater. Processes 2021, 9, 1486. [Google Scholar] [CrossRef]
- Tian, X.; Lin, X.; Xie, Q.; Liu, J.; Luo, L. Effects of Temperature and Light on Microalgal Growth and Nutrient Removal in Turtle Aquaculture Wastewater. Biology 2024, 13, 901. [Google Scholar] [CrossRef]
- Goh, P.S.; Ahmad, N.A.; Lim, J.W.; Liang, Y.Y.; Kang, H.S.; Ismail, A.F.; Arthanareeswaran, G. Microalgae-Enabled Wastewater Remediation and Nutrient Recovery through Membrane Photobioreactors: Recent Achievements and Future Perspective. Membranes 2022, 12, 1094. [Google Scholar] [CrossRef] [PubMed]
- Ak, B.; Atak, E.; Köse, M.D.; Bayraktar, O. Production of Chlorella sp. in a Designed Photobioreactor. Celal Bayar Üniversitesi Fen Bilim. Derg. 2019, 15, 377–383. [Google Scholar] [CrossRef][Green Version]
- Labbafi, S.; Astaraei, F.R. Experimental and Numerical Parametric Study of Horizontal Photobioreactor on the Growth Rate of Chlorella Vulgaris Microalgae. Res. Sq. 2025. [Google Scholar] [CrossRef]
- Abdel-Baset, A.; Matter, I.A.; Ali, M.A. Enhanced Scenedesmus Obliquus Cultivation in Plastic-Type Flat Panel Photobioreactor for Biodiesel Production. Sustainability 2024, 16, 3148. [Google Scholar] [CrossRef]
- Zeriouh, O.; Marco-Rocamora, A.; Reinoso-Moreno, J.V.; López-Rosales, L.; García-Camacho, F.; Molina-Grima, E. New Insights into Developing Antibiofouling Surfaces for Industrial Photobioreactors. Biotechnol. Bioeng. 2019, 116, 2212–2222. [Google Scholar] [CrossRef]
- Mamman, R.O.; Johnson, T.; Weerakkody, T.; Lamuta, C. Fouling Release Mechanism of an Octopus-Inspired Smart Skin. Adv. Funct. Mater. 2024, 34, 2406405. [Google Scholar] [CrossRef]
- Sánchez-Zurano, A.; Lafarga, T.; Morales-Amaral, M.D.M.; Gómez-Serrano, C.; Fernández-Sevilla, J.M.; Acién-Fernández, F.G.; Molina-Grima, E. Wastewater Treatment Using Scenedesmus Almeriensis: Effect of Operational Conditions on the Composition of the Microalgae-Bacteria Consortia. J. Appl. Phycol. 2021, 33, 3885–3897. [Google Scholar] [CrossRef]
- Zhu, J.; Yang, L.; Ding, L.; Yang, Z.; Yin, Y.; Wakisaka, M.; Ashouri, S.; Jazini, M.; Fang, W. Enhanced Recovery of Food-Grade Euglena Gracilis Biomass Through Synergistic pH-Modified Chitosan Flocculation and Green Light Stimulation. Microorganisms 2025, 13, 303. [Google Scholar] [CrossRef]
- Gurreri, L.; Calanni Rindina, M.; Luciano, A.; Falqui, L.; Fino, D.; Mancini, G. Microalgae Production in an Industrial-Scale Photobioreactors Plant: A Comprehensive Life Cycle Assessment. Sustain. Chem. Pharm. 2024, 39, 101598. [Google Scholar] [CrossRef]
- Endres, C.H.; Roth, A.; Brück, T.B. Modeling Microalgae Productivity in Industrial-Scale Vertical Flat Panel Photobioreactors. Environ. Sci. Technol. 2018, 52, 5490–5498. [Google Scholar] [CrossRef] [PubMed]
- Achinas, S.; Heins, J.-I.; Krooneman, J.; Euverink, G.J.W. Miniaturization and 3D Printing of Bioreactors: A Technological Mini Review. Micromachines 2020, 11, 853. [Google Scholar] [CrossRef] [PubMed]
- Strotmann, U.J.; Eismann, F.; Hauth, B.; Bias, W.R. An Integrated Test Strategy for the Assessment of Anaerobic Biodegradability of Wastewaters. Chemosphere 1993, 26, 2241–2254. [Google Scholar] [CrossRef]





| Setup | Surface Area (cm2) | Volume (L) | Surface-to-Volume Ratio (cm−1) | Aeration (L/min) | Lighting (µmol Photons m−2 s−1) |
|---|---|---|---|---|---|
| H-Plain | 4460 | 58 | 7.69 | None | 30 |
| H-Aerated | 4460 | 58 | 7.69 | 8 | 30 |
| V-LED | 4241 | 13.8 | 30.73 | 4 | 53 |
| V-Aerated | 4241 | 13.8 | 30.73 | 4 | 30 |
| Setup | Peak Biomass Concentration (g L−1) | Volumetric Productivity (g L−1/day) | Total Electrical Energy (kWh/day) | Daily Electrical Energy per Biomass (kWh/(g L−1 day−1)) |
|---|---|---|---|---|
| H-Plain | 0.38 ± 0.012 | 0.0475 ± 0.001 | 0 | 0 |
| H-Aerated | 0.47 ± 0.025 | 0.059 ± 0.003 | 0.24 | 4.067 |
| V-LED | 0.5 ± 0.053 | 0.063 ± 0.007 | 1.08 | 17.28 |
| V-Aerated | 0.42 ± 0.042 | 0.053 ± 0.003 | 0.12 | 2.286 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Aldio, R.Z.; Donglah, N.A.; Hashmi, Z.; Zaini, J.; Abu Bakar, M.S.; Bilad, M.R. Performance Evaluation of Chlorococcum sp. in Various Photobioreactor Designs: Impact on Biomass Production and Nutrient Removal. Bioengineering 2026, 13, 388. https://doi.org/10.3390/bioengineering13040388
Aldio RZ, Donglah NA, Hashmi Z, Zaini J, Abu Bakar MS, Bilad MR. Performance Evaluation of Chlorococcum sp. in Various Photobioreactor Designs: Impact on Biomass Production and Nutrient Removal. Bioengineering. 2026; 13(4):388. https://doi.org/10.3390/bioengineering13040388
Chicago/Turabian StyleAldio, Rieza Zulrian, Nur Aqidah Donglah, Zubair Hashmi, Juliana Zaini, Muhammad Saifullah Abu Bakar, and Muhammad Roil Bilad. 2026. "Performance Evaluation of Chlorococcum sp. in Various Photobioreactor Designs: Impact on Biomass Production and Nutrient Removal" Bioengineering 13, no. 4: 388. https://doi.org/10.3390/bioengineering13040388
APA StyleAldio, R. Z., Donglah, N. A., Hashmi, Z., Zaini, J., Abu Bakar, M. S., & Bilad, M. R. (2026). Performance Evaluation of Chlorococcum sp. in Various Photobioreactor Designs: Impact on Biomass Production and Nutrient Removal. Bioengineering, 13(4), 388. https://doi.org/10.3390/bioengineering13040388









