1. Introduction
The challenges of renewable energy sources like wind and solar highlight their output variability, driving increased focus on energy storage solutions. While batteries [
1] and flywheels [
2] are promising options, they are not yet cost-effective [
3]. An alternative solution is to store electricity as chemical energy, specifically in the form of H
2, CH
4 via biomethanation, or NH
3. However, NH
3 storage poses significant safety challenges: NH
3 is hazardous and should be handled with caution. H
2 storage, particularly, is challenged by its low energy density, safety risks, and high transportation costs [
4]. Biomethanation offers a promising solution by converting H
2 and CO
2 into CH
4, which has a higher energy density than H
2 and is easier to store and transport.
Biomethanation is a biological process that converts CO
2 and H
2 into CH
4 through the activity of hydrogenotrophic methanogens according to the reaction CO
2 + 4H
2 → CH
4 + 2H
2O (ΔH = −165.0 kJ/mol) [
5]. The CO
2 in this process can be obtained from various sources, such as biogas [
6]. With H
2 sourced from renewable energy sources such as solar or wind, biomethanation can be particularly interesting for both renewable energy storage and biogas upgrading, as it converts the CO
2 fraction of biogas (approximately 40%) into CH
4 while utilising H
2 generated from surplus renewable electricity.
Alternative biogas upgrading technologies are commercially available, but most focus exclusively on physical CO
2 removal rather than CO
2 conversion [
7]. Established technologies, including absorption [
8], adsorption [
9], membrane separation [
10], and cryogenic separation [
11,
12], effectively increase CH
4 concentration and are well documented at an industrial scale. However, these approaches do not enhance the energetic content of biogas beyond concentration effects.
In contrast, biological methanation is fundamentally constrained by H
2 mass transfer. Due to the low solubility of H
2 in water, gas–liquid mass transfer rates are limited, thereby restricting H
2 availability to hydrogenotrophic methanogens and reducing overall process efficiency [
13].
Conventional reactor configurations used for biological methanation exhibit inherent limitations when applied to H
2-driven CO
2 conversion. In continuously stirred tank reactors (CSTRs), the low solubility of H
2 in water results in severe gas–liquid mass transfer limitations, limiting H
2 availability to hydrogenotrophic methanogens and consequently the achievable CH
4 formation rates. Upflow anaerobic sludge blanket (UASB) reactors offer improved biomass retention; however, their applicability for ex situ biomethanation is constrained by limited control over gas distribution, challenges in handling high H
2 gas flows, and the risk of gas channelling, which can compromise process stability [
14]. Membrane-assisted systems have been proposed to enhance H
2 delivery, yet these configurations introduce additional system complexity, increased capital and operational costs, and membrane fouling risks, which hinder large-scale implementation.
In contrast, trickle-bed reactors (TBRs) combine immobilised biomass with a high specific gas–liquid interfacial area, enabling efficient H2 transfer to methanogenic biofilms while maintaining operational stability. The fixed-bed configuration allows decoupling of hydraulic and gas residence times, facilitating independent optimisation of nutrient supply and gas conversion. This makes TBRs particularly suitable for high-rate ex situ biomethanation, where H2 mass transfer and microbial retention are critical performance determinants. Consequently, TBR technology represents a relevant reactor concept for achieving high CH4 formation rates under thermophilic biomethanation conditions.
In view of the mass transfer and gas-handling limitations associated with conventional reactor configurations, this study focuses on the application of a trickle-bed reactor (TBR) for ex situ biomethanation, which provides a high specific gas–liquid interfacial area and enables effective immobilisation of hydrogenotrophic methanogens [
15]. The performance of a TBR can be evaluated using the Methane Formation Rate (MFR, m
3 CH
4/m
3 reactor/day), a critical parameter for assessing the efficiency of biogas production systems [
15].
Previous studies have reported that TBRs achieve a high specific CH
4 production, reaching up to ~15 m
3 per m
3 of reactor volume per day, which is considerably higher than other configurations, such as CSTRs at ~3.7 m
3/(m
3·d) and up-flow reactors at ~0.25 m
3/(m
3·d) ([
16,
17,
18]). Moreover, the biomethane produced meets the quality standards for direct injection into the gas grid, as specified in the European standard, which outlines the specifications for biomethane for injection in the natural gas network [
19].
This study investigates the performance of a thermophilic trickle-bed reactor (TBR) for ex situ biomethanation of CO2 via hydrogenotrophic methanogenesis. The objectives are to (i) quantify CH4 formation rates and H2 conversion efficiency under controlled operating conditions, (ii) assess reactor stability and operational robustness, including restart after prolonged idle periods, (iii) evaluate reactor performance using both pure CO2 and biogas as feed gas, and (iv) examine the applicability of wastewater as a trickling medium. The study focuses on experimental reactor performance and operational behaviour; detailed techno-economic assessment and scale-up modelling are beyond the scope of this work.
3. Results
The performance of the trickle-bed reactor was evaluated based on the MFR and the composition of the reactor off-gas. Experiments were conducted using H2 and CO2 as substrates, as well as biogas containing both CH4 and CO2. Reactor performance was monitored over time by measuring gas flow rates, CH4 production, and H2 conversion under the applied operating conditions.
3.1. Characterisation of Siporax™ Carrier Material
The physical characteristics of Siporax™ (Hornbach, Groningerweg 45/2, 9738 AB Groningen) carrier material were quantified using a laboratory-scale column. Characterisation experiments were conducted in a reactor volume of 700 mL, which was adjusted to 1000 mL using a combination of whole and quarter Siporax™ rings. A fully packed column contained 273 g of whole rings or 387 g of quarter rings, corresponding to total specific surface areas of 270 m2 L−1 and 382 m2 L−1, respectively.
The pore volume of the carrier material was determined to be 185 mL L−1 for whole rings and 277 mL L−1 for quarter rings. The total volume occupied by the carrier material was measured by filling the column with water-saturated Siporax™ and subsequently adding water until the column was filled. The void volume was calculated from the additional volume of water required to fill the column and amounted to 314 mL L−1 for whole rings and 457 mL L−1 for quarter rings.
Based on the measured surface area and void volume, area-to-void-volume ratios of 859 m
2 L
−1 and 835 m
2 L
−1 were calculated for whole and quarter rings, respectively. The volume of open space (VOS), defined as the reactor volume minus the volume of the carrier material, was used to calculate the gas residence time (GRT). The gas residence time (GRT) was calculated as the ratio between the gas-filled volume (V
gas) and the inlet gas flow rate (Q
gas). At a gas flow rate of 10 mL min
−1, the GRT was 31 min for whole rings and 46 min for quarter rings. When applying a H₂/CO₂ molar ratio of 3.7:1 (and assuming complete hydrogen conversion with H₂ as the limiting reactant), the gas volume decreases by a factor of 4.7, resulting in corresponding equivalent gas residence times of 147 min and 214 min for whole and quarter rings, respectively. Detailed calculations and underlying assumptions are provided in the
Supporting Information (Section S1).
An overview of the measured and calculated characteristics of the Siporax™ carrier material is provided in
Table 1.
3.2. Optimisation of CH4 Production in Laboratory-Scale TBRs
A series of laboratory-scale experiments was conducted to evaluate CH4 production performance under varying operational conditions. Experiments were performed in 1 L trickle-bed reactors filled with whole Siporax™ rings and inoculated with anaerobic sludge. The reactors were supplied with H2 and CO2, and CH4 production was monitored over time.
During the initial operation, CH4 production increased gradually and reached approximately 0.1 L CH4 L−1 d−1. After 25 days of operation, a maximum (MFR) of 4.3 L-CH4 L−1 d−1 was observed, after which the MFR stabilised at approximately 3 L-CH4 L−1 d−1. During this period, the circulating trickling liquid was refreshed weekly to maintain reactor operation.
Following adjustments to the reactor configuration and operating conditions, a substantial increase in CH4 production was observed. Under the modified conditions, the reactor reached a maximum MFR of 15 L-CH4 L−1 d−1 and subsequently stabilised at approximately 13 L-CH4 L−1 d−1. Similar conditions were applied in subsequent experiments.
Reactor performance was progressively optimised through three targeted modifications: (i) replacement of the original wastewater treatment inoculum with pre-enriched methanogenic granular sludge, (ii) increasing the operational temperature from 40 °C to 55 °C [
22], and (iii) enhancing the specific surface area of the carrier material by using fragmented Siporax rings instead of intact elements [
20].
Collectively, these adjustments resulted in a substantial improvement in CH4 formation rate (MFR), which increased from 0.4 to 13 L CH4 L−1 d−1, corresponding to an approximately fourfold enhancement relative to the stabilised baseline performance.
3.3. Performance of a 15 L Trickle-Bed Reactor
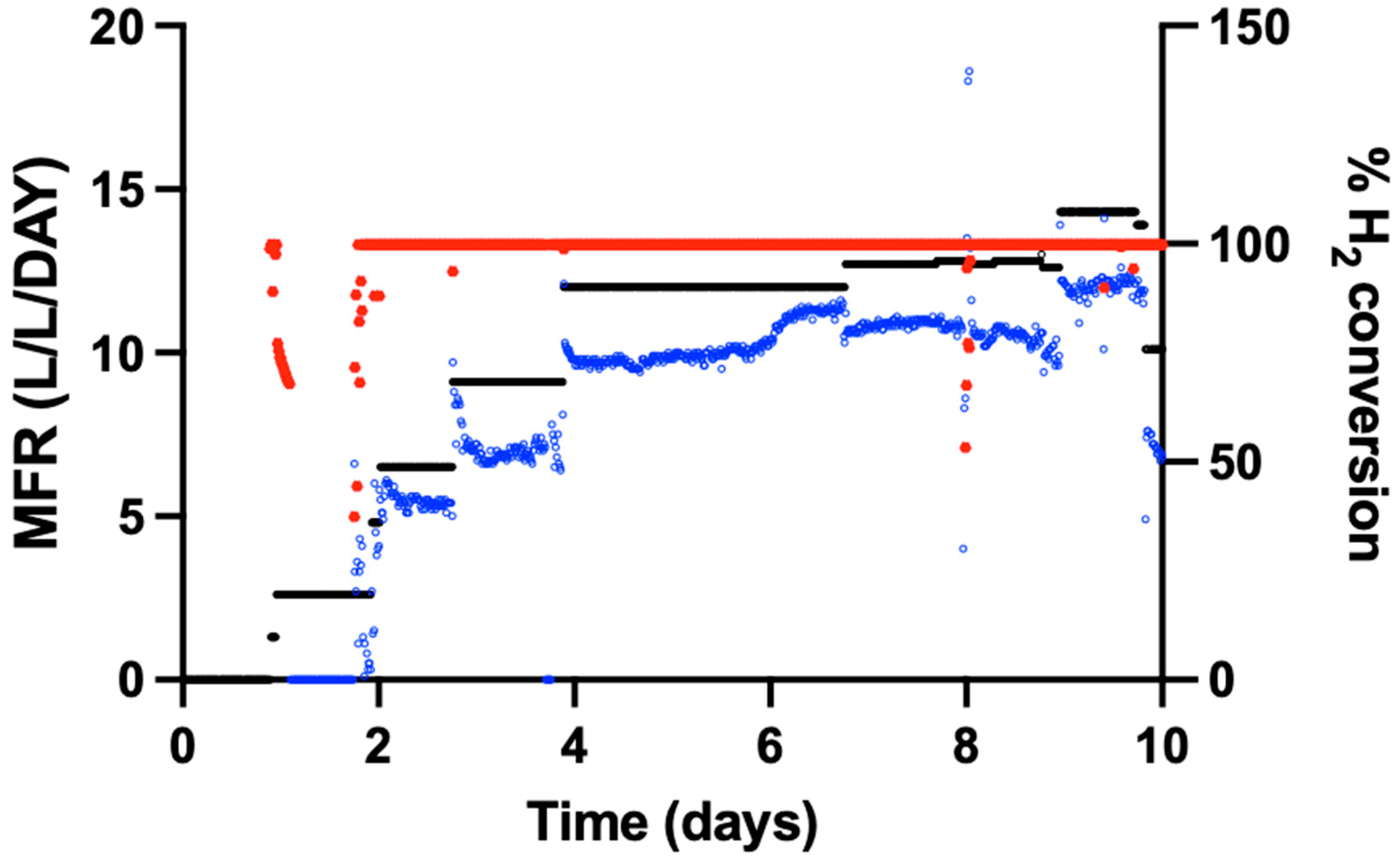
The performance of a 15 L trickle-bed reactor (TBR) filled with Siporax™ was evaluated over a 45-day continuous operation period. Reactor performance was assessed based on the MFR and H
2 conversion.
Figure 2 shows the experimentally determined MFR, the modelled MFR, and the corresponding H
2 conversion over time.
During reactor operation, periods were observed in which H
2 and CO
2 were detected in the reactor off-gas. The methane formation rate (MFR) was quantified from the measured CH
4 concentration in the effluent gas. When total gas production exceeded the CH
4 volume predicted by the stoichiometric model, the difference was interpreted as residual H
2 and CO
2. This non-methanized fraction was not included in the reported MFR. The experimental MFR values were compared with the modelled MFR, which was calculated from the inlet gas composition assuming stoichiometric conversion of H
2 and CO
2 according to:
where
H2-input = volumetric H2 supply rate (m3 H2 d−1)
Vreactor = working reactor volume (m3)
4 = stoichiometric conversion factor (4 mol H2 → 1 mol CH4)
Throughout the experiment, H
2 conversion remained high, reaching 100% in most of the operational period (
Figure 2). Temporal variability was observed in the experimentally determined MFR, particularly between days 10 and 30, whereas the modelled MFR remained relatively constant. Periods in which the experimentally calculated MFR exceeded the modelled MFR coincided with a reduction in the measured H
2 conversion.
Periodic gas chromatography measurements confirmed the presence of H
2 in the reactor off-gas during these periods. An overview of the MFR and H
2 conversion trends is provided in
Figure 2.
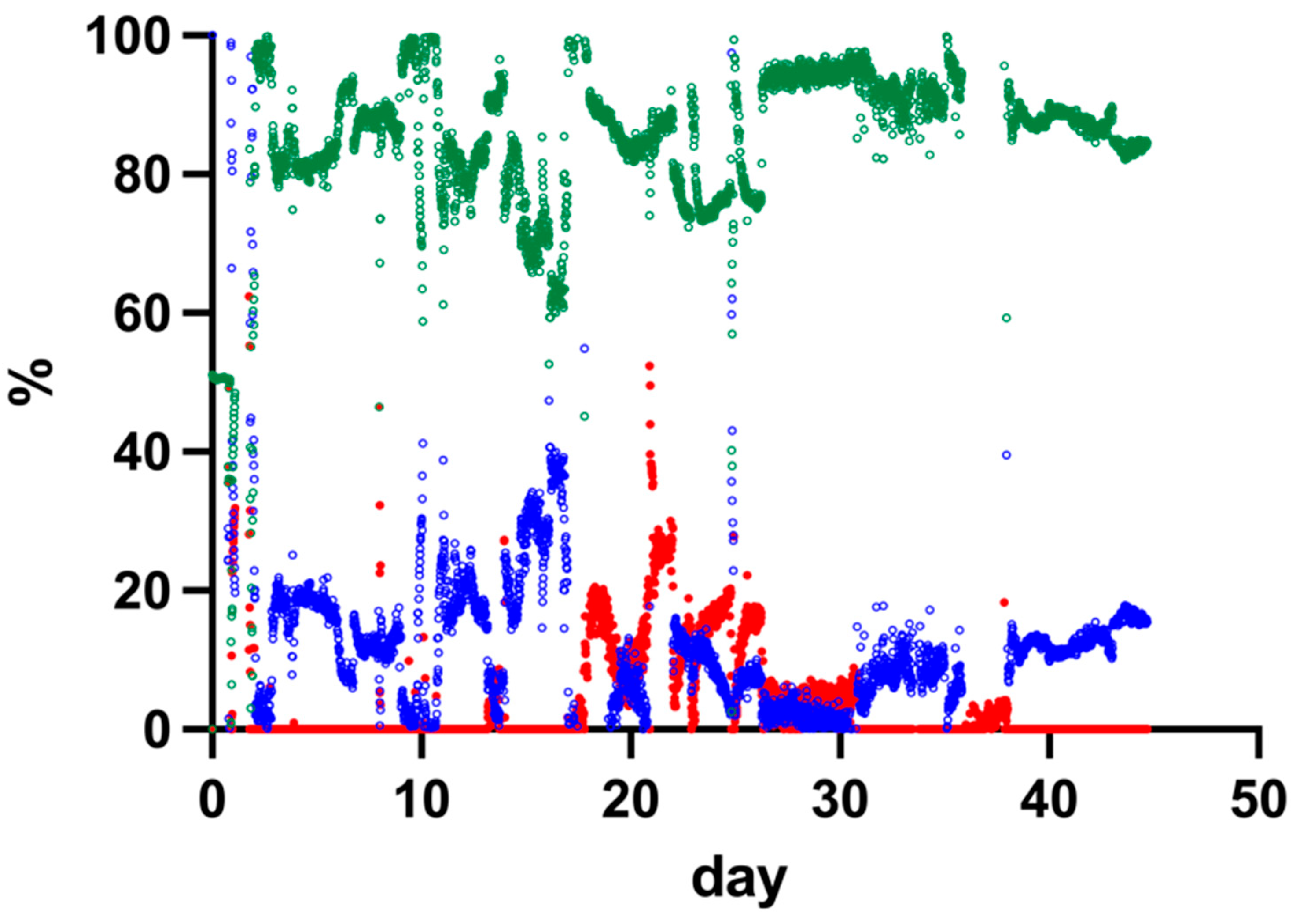
3.4. Gas Composition in the Trickle-Bed Reactor Performance over Time
Temporal changes in reactor off-gas composition were monitored during operation of the 15 L trickle-bed reactor. The concentrations of CH
4, CO
2, and H
2 in the off-gas were determined over time.
Figure 3 presents the measured gas composition profiles throughout the experimental period.
During reactor operation, fluctuations in gas composition were observed. After the initial start-up phase, CH4 concentrations stabilised, while H2 concentrations in the off-gas decreased to near-zero values after day 10. CO2 concentrations varied throughout the experiment.
Based on the stabilised operational period, the average CH4 concentration in the off-gas was calculated from day 26 to the end of the experiment and was 87.0 ± 4.0%. Variations in gas composition were observed between days 18 and 25, during which changes in the relative concentrations of CO2 and H2 occurred. The gas composition values represent relative volumetric percentages of the total measured off-gas.
3.5. Reactor Reactivation After Prolonged Inactivity
Following an extended idle period, the 15 L trickle-bed reactor was evaluated for reactivation performance. The reactor was stored at room temperature for 8 weeks after flushing with nitrogen gas. After the idle period, the reactor was reactivated by trickling with fresh inoculum and flushing with CO
2. H
2 was subsequently introduced. Once an H
2 breakthrough is observed, the previous stable flow rate is considered the maximum biological conversion capacity. During the initial reactivation phase, minor H
2 concentrations were detected in the off-gas. Within 24 h of H
2 introduction, CH
4 production resumed. The reactor reached CH
4 formation rates in the range of approximately 10–12 L-CH
4 L
−1 reactor d
−1 without additional mechanical or structural modification (
Figure 4). CH
4 production stabilised after this reactivation period .
Figure 4.
Start-up phase of the reactor after an 8-week storage period. The left y-axis represents the Methane Formation Rate (MFR, blue), and the right y-axis represents the H2 conversion (red) percentage. The x-axis indicates time. The black line shows the theoretical MFR based upon the H2 feed.
Figure 4.
Start-up phase of the reactor after an 8-week storage period. The left y-axis represents the Methane Formation Rate (MFR, blue), and the right y-axis represents the H2 conversion (red) percentage. The x-axis indicates time. The black line shows the theoretical MFR based upon the H2 feed.
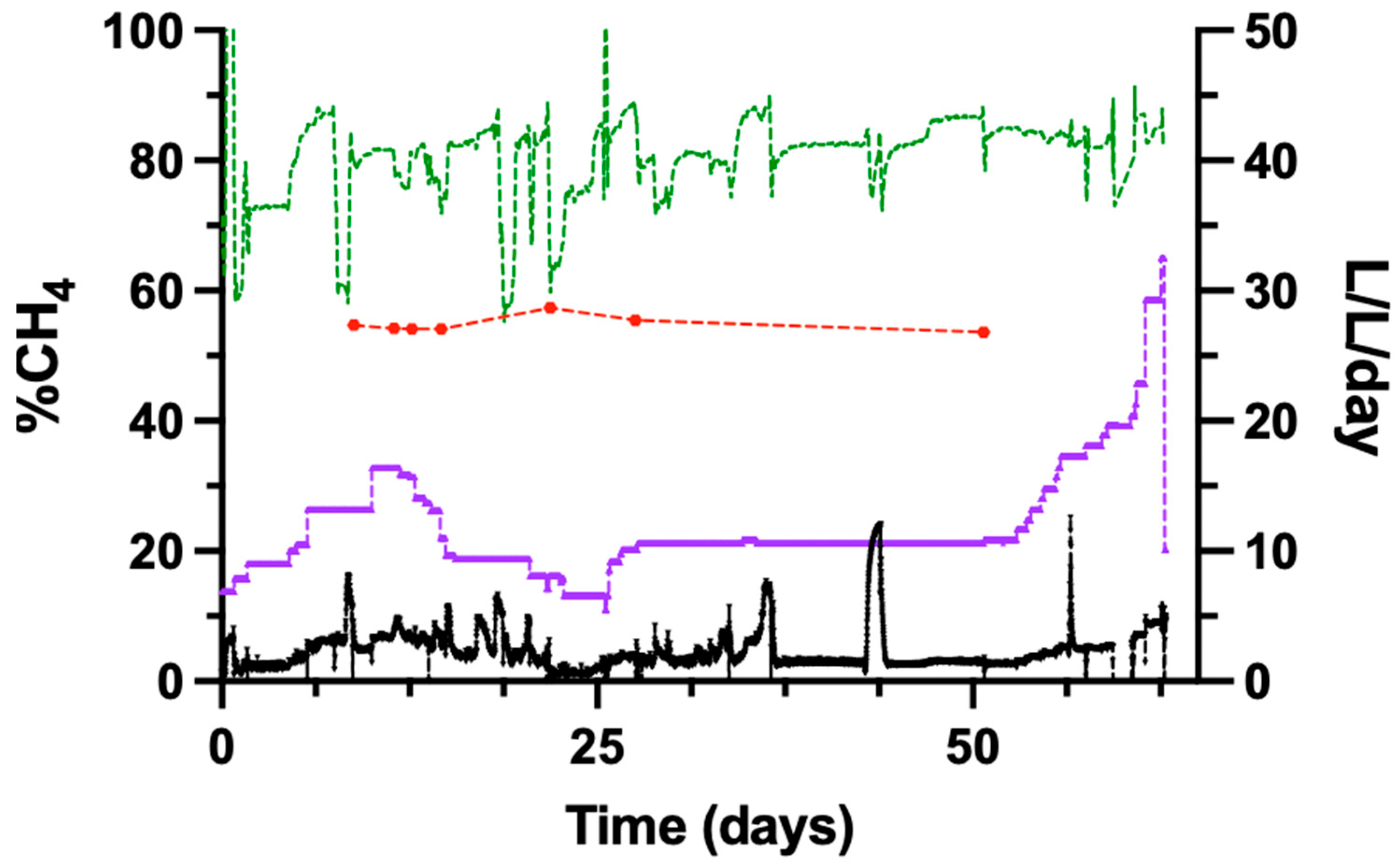
3.6. Operation of the Trickle-Bed Reactor with Biogas as Feed Gas
The performance of the trickle-bed reactor was evaluated using biogas as the inlet gas. The biogas feed consisted of 58% CH4, 38% CO2, and approximately 4% N2, O2, and water vapour. The reactor performance was assessed based on MFR, methane evolution rate (MER), and off-gas composition.
Following the switch from pure gases to biogas, the CH4 concentration in the reactor off-gas remained approximately 80% despite an increased biogas feed rate. The CH4 evolution rate, expressed as L-CH4 L−1 reactor d−1, increased with increasing biogas supply and represents the total CH4 flux through the reactor column.
The MFR, defined as the volume of newly produced CH
4 produced from the conversion of CO
2 and H
2, was lower during biogas operation than during experiments using pure CO
2 and H
2 as substrates.
Figure 5 shows the temporal evolution of CH
4 content in the biogas feed and the upgraded off-gas. Measurements of the biogas inlet composition, obtained from a bypass of the feed stream, showed an average CH
4 content of 54.8 ± 1.2%. The average CH
4 concentration in the upgraded off-gas during the experimental period was 79.4 ± 12.9%.
Fluctuations in off-gas CH4 concentration were observed throughout the 52-day experiment.
3.7. Comparison Between Experimental and Theoretical Methane Formation Rates
The experimentally determined MFR-exp was compared with the theoretical MFR-model calculated from the inlet gas composition.
Figure 6 presents the temporal evolution of both MFR values during reactor operation.
Throughout the experimental period, the experimentally determined methane formation rate (MFR-exp) fluctuated despite stable inlet gas flow rates. These deviations likely reflect transient mass transfer limitations and dynamic biofilm activity rather than changes in substrate supply.
H2 conversion showed temporal variation during reactor operation. During the initial phase of operation, H2 conversion values approached 100%. Subsequently, periods of reduced H2 conversion were observed, followed by a later phase in which H2 conversion and MFR-exp increased and became more stable. Around day 30, both H2 conversion and MFR-exp showed a general upward trend.
An overview of the experimental and theoretical MFR values and the corresponding H
2 conversion is shown in
Figure 6.
3.8. Gas Residence Time During Operation of the Trickle-Bed Reactor
The gas residence time (GRT) in the trickle-bed reactor was calculated under different gas feed compositions. When operating with pure H2 and CO2, the calculated GRT was 101.1 min at an MFR of 10 L-CH4 L−1 reactor d−1. Under biogas operation, where CH4 was present in the inlet gas, the calculated GRT decreased to 52.3 min at the same MFR.
The reduction in GRT reflects the increased total gas flow rate due to CH4 in the inlet gas. Calculated GRT values were used to compare reactor operation under different feed gas compositions.
4. Discussion
A trickle-bed reactor provides a favourable configuration for ex situ biomethanation by combining counter-current gas–liquid flow with immobilised biomass on a fixed bed. This configuration enables high biomass retention and enhanced gas–liquid interfacial area, both of which are critical for hydrogenotrophic methanogenesis. However, reactor performance is strongly influenced by the interaction between carrier material properties, hydrodynamics, and operational conditions [
23].
In this study, Siporax™ was selected as a carrier material based on its exceptionally high specific surface area and three-dimensional pore structure [
20]. Compared to commonly used carrier materials reported in the literature, Siporax™ provides almost two orders of magnitude higher surface area per unit volume, which is expected to support extensive microbial attachment and biofilm development [
24]. The characterisation experiments demonstrated that both whole and fragmented Siporax rings exhibit high surface-area densities (270–382 m
2 L
−1), confirming their suitability as biofilm carriers in TBR systems.
At the same time, the results highlight that increased surface area is accompanied by trade-offs related to packing behaviour and hydrodynamics. Fragmentation of Siporax rings resulted in a higher measured void volume compared to whole rings, despite the smaller particle size. This counterintuitive effect can be attributed to irregular particle shape and less efficient packing of the carrier material, which increases the open space between particles. These differences in void volume directly affect the distribution of gas flow and gas residence time, both of which are critical parameters in H2-driven biomethanation.
The area-to-void-volume ratio provides a useful metric for balancing biofilm support and gas–liquid interactions. Although whole and fragmented rings exhibited similar ratios, the more complex carrier-material arrangement in fragmented Siporax is expected to promote greater turbulence and local mixing. Such effects can enhance gas–liquid mass transfer, particularly when H
2 availability limits methane formation. However, increased turbulence and surface area may also increase the risk of excessive biofilm growth, clogging, or uneven flow distribution, emphasising the need for careful operational control [
16].
Gas residence time emerged as a key factor linking carrier material choice to reactor performance [
25]. Calculated gas residence times differed substantially between whole and fragmented rings and were further reduced during biogas operation due to the presence of CH
4 in the inlet gas. While CH
4 does not participate in the methanation reaction, it increases total gas flow and thereby reduces the effective residence time available for CO
2 and H
2 conversion [
26]. This effect explains the observed reduction in methane formation rate during biogas operation compared to experiments using pure CO
2 and H
2, despite maintaining high H
2 conversion.
The use of a practical H
2:CO
2 molar ratio of 3.7:1, rather than the stoichiometric 4:1, further illustrates the interaction between microbial activity and reactor design. This deviation accounts for CO
2 assimilation into microbial biomass and reflects realistic operating conditions in biological systems. Under these conditions, effective gas residence time increases substantially, partially compensating for the dilution effects introduced by CH
4 in the feed gas. Consequently, H
2 and CO
2 are supplied at a lower flux relative to the available biofilm surface, partially mitigating dilution effects caused by CH
4 in the feed gas [
27].
Overall, the results demonstrate that the selection of carrier material in TBRs cannot be optimised solely on the basis of surface area. Instead, reactor performance is governed by a balance between surface area, void volume, gas residence time, and flow distribution. Siporax™ proved to be an effective carrier material for high-rate biomethanation. In practice, the use of Siporax™ in trickle-bed biomethanation reactors requires tight control of gas flow, gas recirculation, and liquid distribution to ensure sufficient gas–biofilm contact while avoiding channelling, excessive pressure drop, and biofilm instability [
14,
28,
29]. These findings underline the importance of integrating carrier material properties with reactor design and process control strategies when scaling up TBR technology for biogas upgrading and power-to-gas applications [
30]. From an economic perspective, ex situ biomethanation is primarily constrained by H
2 supply costs, with electricity price and electrolyser CAPEX dominating total system costs [
14]. The bioreactor affects economics indirectly by determining volumetric CH
4 productivity (MFR) and, in turn, the required reactor volume. Conventional wet reactors, such as CSTRs or UASB-type systems, benefit from technological maturity but are inherently limited by H
2 gas–liquid mass transfer, resulting in large reactor volumes at high H
2 loading rates. Reactor concepts that enhance the gas–liquid interfacial area, such as trickle-bed reactors, are therefore frequently considered economically promising, as they enable more compact designs with relatively low auxiliary energy demand [
31]. A detailed techno-economic analysis is outside the scope of this study; the results are intended to position the reactor concept within qualitative economic trade-offs.
5. Conclusions
This study demonstrates that thermophilic trickle-bed reactors (TBRs) can achieve high-rate ex situ biomethanation under laboratory-scale conditions . Methane formation rates were consistently high, with CO2 conversion approaching 90% and H2 conversion exceeding 99% during stable operation using pure CO2 and H2 as feed gases. These results confirm the suitability of TBR configurations for hydrogenotrophic biomethanation, provided that sufficient H2 is supplied relative to CO2.
The experimental results indicate that CO2 conversion is strongly constrained by H2 availability and reactor operating conditions. While near-complete H2 conversion was achieved, CO2 conversion remained below full stoichiometric conversion, highlighting the importance of H2 supply control and gas–liquid mass transfer in determining overall reactor performance.
The use of wastewater as a trickling medium enabled stable reactor operation while simplifying nutrient supply, although it introduced variability that may affect reproducibility. Despite this variability, the reactor maintained stable performance during extended operation and recovered rapidly after prolonged inactivity, indicating a high degree of operational robustness.
Overall, the results show that trickle-bed reactors represent a promising platform for high-rate biomethanation and biogas upgrading. Further work is required to evaluate long-term stability and performance under biogas feed conditions and to assess scalability under industrially relevant operating regimes.