Electrogastrography-Derived Mean Power Ratio as an Exploratory Objective Measure of Feeding Intolerance in Preterm Infants
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
Feeding Intolerance Cohorts
2.2. Experimental Setup
2.3. EGG Acquisition
2.4. Data Processing
2.4.1. EGG Data Preprocessing
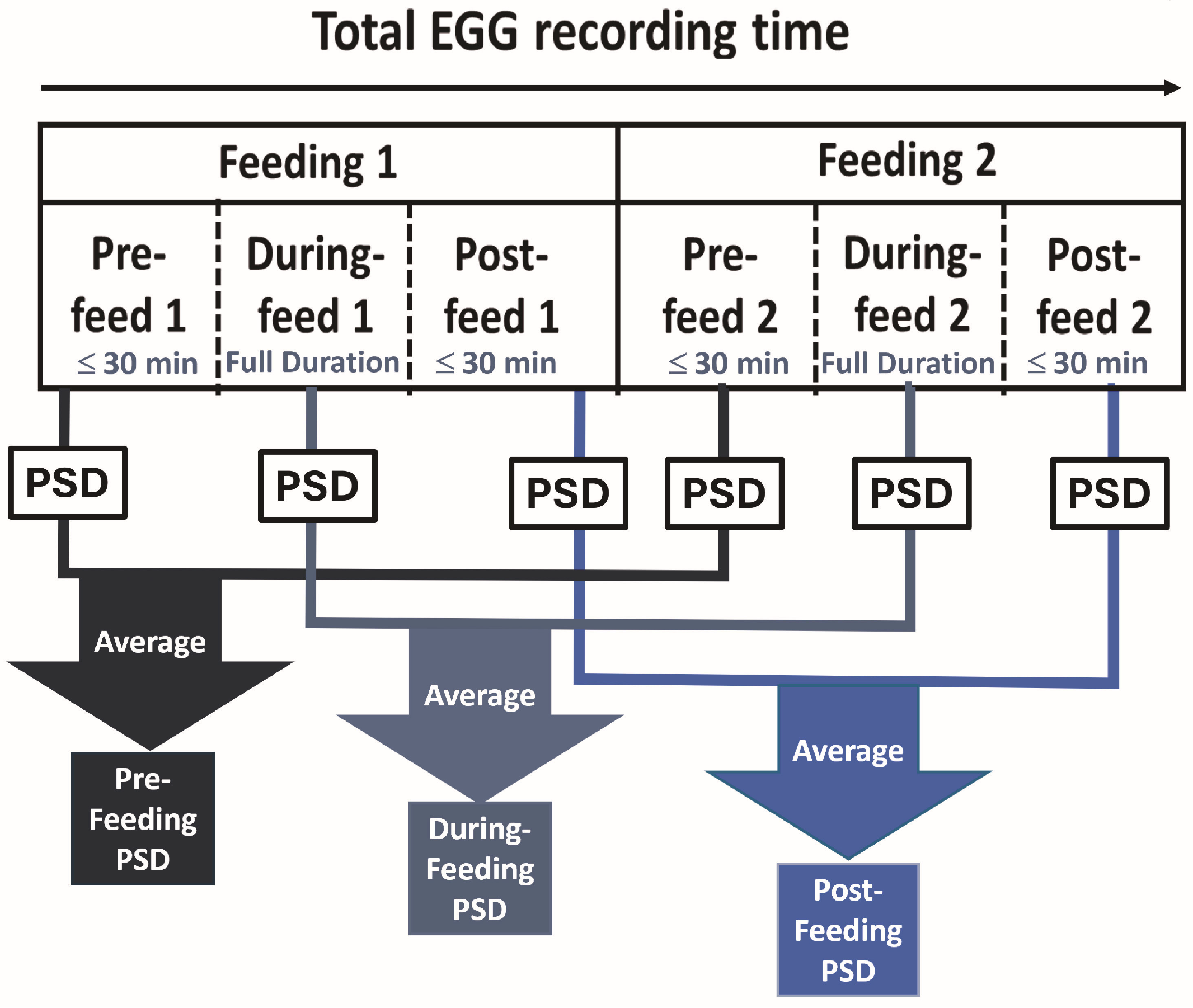
2.4.2. Segmentation into Feeding Periods
2.4.3. Power Spectral Density (PSD) Calculation and Averaging Across Segments
2.4.4. Group-Level PSD Curves
2.4.5. Power Spectral Density Ratio (PSDR) Curves
2.5. Statistical Analysis
2.5.1. Baseline Comparison
2.5.2. Calculations and Comparisons of Mean Power Ratio (mPR) Metrics
3. Results
3.1. Demographics of the Study Cohort
3.2. Group-Averaged PSD Curves Across Feeding Periods
3.3. Group-Averaged PSD Ratio Curves with Respect to the Pre-Feeding PSD
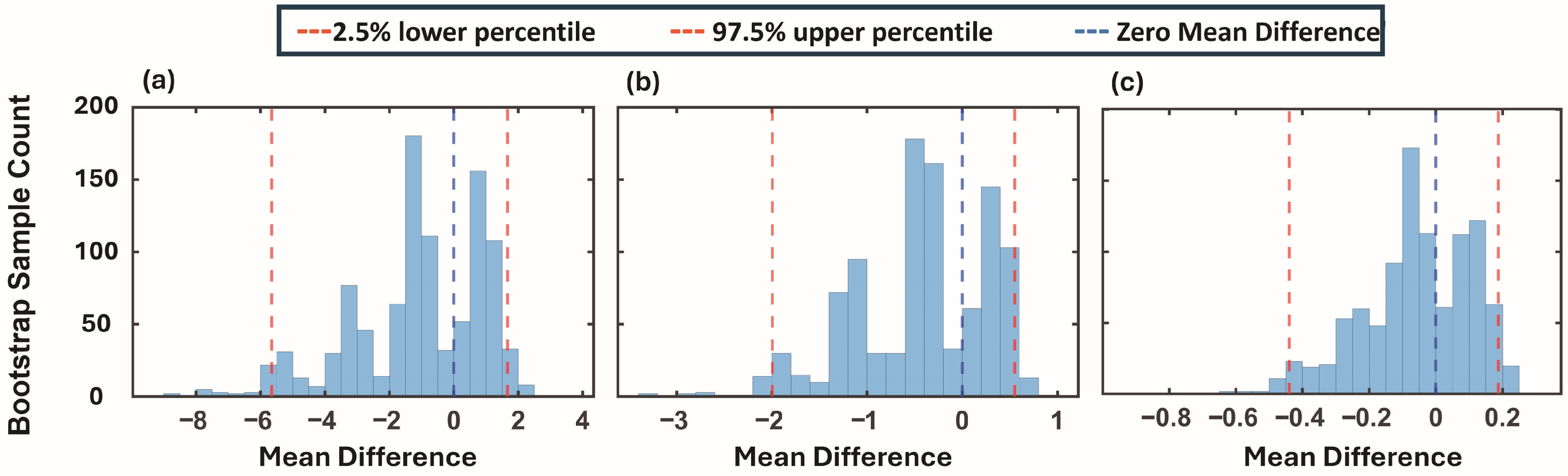
3.4. Baseline Gastric Activity: Bootstrapping Analysis of mPSD Values
3.5. Gastric Activity Ratios: Bootstrapping Analysis of mPR Values
4. Discussion
4.1. Key Findings of the Study
4.2. Novel Analytical Development of the Study
4.3. Clinical Implications of the Study
4.4. Limitations and Future Work of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EGG | Electrogastrography |
| FI | Feeding intolerance |
| NFI | No feeding intolerance |
| DFI | Developmental feeding intolerance |
| PFI | Pathological feeding intolerance |
| NEC | Necrotizing enterocolitis |
| GA | Gestational age |
| PMA | Postmenstrual age |
| NICU | Neonatal intensive care unit |
| IRB | Institutional Review Board |
| NPO | Nil per os (nothing by mouth) |
| SFT | Sustained feeding tolerance |
| GR | Gastric rhythm |
| PSD | Power spectral density |
| mPSD | Mean power spectral density |
| mPSDbrady | Mean power spectral density in bradygastria |
| mPSDnormo | Mean power spectral density in normogastria |
| mPSDtachy | Mean power spectral density in tachygastria |
| PSDR | Power spectral density ratio |
| PSDRDur/Pre | During-feeding/pre-feeding power spectral density ratio |
| PSDRPost/Pre | Post-feeding/pre-feeding power spectral density ratio |
| mPR | Mean power ratio |
| mPRDur/Pre | During-feeding/pre-feeding mean power ratio |
| mPRDur/Pre_b | During-feeding/pre-feeding mean power ratio in bradygastria |
| mPRDur/Pre_n | During-feeding/pre-feeding mean power ratio in normogastria |
| mPRDur/Pre_t | During-feeding/pre-feeding mean power ratio in tachygastria |
| mPRPost/Pre | Post-feeding/pre-feeding mean power ratio |
| mPRPost/Pre_b | Post-feeding/pre-feeding mean power ratio in bradygastria |
| mPRPost/Pre_n | Post-feeding/pre-feeding mean power ratio in normogastria |
| mPRPost/Pre_t | Post-feeding/pre-feeding mean power ratio in tachygastria |
| cpm | Cycles per minute |
| Hz | Hertz |
| Kg | Kilogram |
| g | Grams |
| mL | Milliliters |
| CI | Confidence interval |
Appendix A
| Conventional EGG Metrics | No Feeding Intolerance (NFI), n = 75 | Feeding Intolerance (FI), n = 9 | p Value * |
|---|---|---|---|
| % Bradygastria, <2 cpm | |||
| Pre-prandial | 23 (16–31) | 24 (18–32) | 0.36 |
| Post-prandial | 23 (14–28) | 25(17–26) | 0.61 |
| % Normogastria, >2 to ≤4 cpm | |||
| Pre-prandial | 22 (16–28) | 23 (18–26) | 0.32 |
| Post-prandial | 22 (17–27) | 23 (19–25) | 0.89 |
| % Tachygastria, >4 to ≤9 cpm | |||
| Pre-prandial | 40 (30–46) | 41 (31–47) | 0.90 |
| Post-prandial | 40 (36–45) | 40 (38–45) | 0.66 |
| Dominant Frequency, cpm | |||
| Pre-prandial | 1.8 (0.6–5) | 0.9 (0.6–3.6) | 0.36 |
| Post-prandial | 1.5 (0.6–4.5) | 1.2 (0.6–3.6) | 0.35 |
| Dominant power, mV2/Hz | |||
| Pre-prandial | 0.3 (0–0.6) | 0 (0–1) | 0.12 |
| Post-prandial | 0.1 (0.1–0.6) | 0 (0–0.1) | 0.27 |
| Power ratio | 1.9 (0.2–57) | 2 (0.3–4.6) | 0.72 |
| Instability coefficient | |||
| Pre-prandial | 0.7 (0.4–0.9) | 0.7 (0.5–0.9) | 0.19 |
| Post-prandial | 0.6 (0.4–0.9) | 0.7 (0.5–0.8) | 0.63 |
References
- Moore, T.A.; Pickler, R.H. Feeding intolerance, inflammation, and neurobehaviors in preterm infants. J. Neonatal Nurs. 2017, 23, 134–141. [Google Scholar] [CrossRef]
- Neu, J.; Zhang, L. Feeding intolerance in very-low-birthweight infants: What is it and what can we do about it? Acta Paediatr. 2005, 94, 93–99. [Google Scholar] [CrossRef]
- Ortigoza, E.B. Feeding intolerance. Early Hum. Dev. 2022, 171, 105601. [Google Scholar] [CrossRef]
- Moore, T.A.; Wilson, M.E. Feeding intolerance: A concept analysis. Adv. Neonatal Care 2011, 11, 149–154. [Google Scholar] [CrossRef]
- Fanaro, S. Feeding intolerance in the preterm infant. Early Hum. Dev. 2013, 89, S13–S20. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Chen, J.D. Electrogastrography: Methodology, validation and applications. J. Neurogastroenterol. Motil. 2013, 19, 5–17. [Google Scholar] [CrossRef]
- Chen, J.D.; Schirmer, B.D.; McCallum, R.W. Serosal and cutaneous recordings of gastric myoelectrical activity in patients with gastroparesis. Am. J. Physiol. 1994, 266, G90–G98. [Google Scholar] [CrossRef]
- Chen, J.D.; Zou, X.; Lin, X.; Ouyang, S.; Liang, J. Detection of gastric slow wave propagation from the cutaneous electrogastrogram. Am. J. Physiol. 1999, 277, G424–G430. [Google Scholar] [CrossRef] [PubMed]
- Riezzo, G.; Russo, F.; Indrio, F. Electrogastrography in adults and children: The strength, pitfalls, and clinical significance of the cutaneous recording of the gastric electrical activity. Biomed. Res. Int. 2013, 2013, 282757. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.D.; McCallum, R.W. Clinical applications of electrogastrography. Am. J. Gastroenterol. 1993, 88, 1324–1336. [Google Scholar]
- Chen, J.-D. Spectral analysis of electrogastrogram and its clinical significance. World J. Gastroenterol. 1996, 2, 9–11. [Google Scholar]
- Chen, J. A computerized data analysis system for electrogastrogram. Comput. Biol. Med. 1992, 22, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.D.; Lin, Z.; Pan, J.; McCallum, R.W. Abnormal gastric myoelectrical activity and delayed gastric emptying in patients with symptoms suggestive of gastroparesis. Dig. Dis. Sci. 1996, 41, 1538–1545. [Google Scholar] [CrossRef] [PubMed]
- Pfaffenbach, B.; Adamek, R.J.; Bartholomaus, C.; Wegener, M. Gastric dysrhythmias and delayed gastric emptying in patients with functional dyspepsia. Dig. Dis. Sci. 1997, 42, 2094–2099. [Google Scholar] [CrossRef]
- Precioso, A.R.; Pereira, G.R.; Vaz, F.A. Gastric myoelectrical activity in neonates of different gestational ages by means of electrogastrography. Rev. Hosp. Clin. Fac. Med. Sao Paulo 2003, 58, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, A.; Wang, X.; Roblyer, L.; Sanghavi, R.; Liu, H.; Ortigoza, E.B. Frequency-specific electrogastrography as a non-invasive tool to measure gastrointestinal maturity in preterm infants. Sci. Rep. 2022, 12, 20728. [Google Scholar] [CrossRef]
- Liang, J.; Co, E.; Zhang, M.; Pineda, J.; Chen, J.D. Development of gastric slow waves in preterm infants measured by electrogastrography. Am. J. Physiol. 1998, 274, G503–G508. [Google Scholar] [CrossRef]
- Riezzo, G.; Chiloiro, M.; Guerra, V.; Borrelli, O.; Salvia, G.; Cucchiara, S. Comparison of gastric electrical activity and gastric emptying in healthy and dyspeptic children. Dig. Dis. Sci. 2000, 45, 517–524. [Google Scholar] [CrossRef]
- Chen, J.D.; Lin, X.; Zhang, M.; Torres-Pinedo, R.B.; Orr, W.C. Gastric myoelectrical activity in healthy children and children with functional dyspepsia. Dig. Dis. Sci. 1998, 43, 2384–2391. [Google Scholar] [CrossRef]
- Siegl, A.; Mayr, J.; Huber, A.; Uray, E. Postprandial tachygastria is frequent in infants with gastroesophageal reflux. Pediatr. Surg. Int. 1998, 13, 569–571. [Google Scholar] [CrossRef]
- Sadder, L.S.; Brown, L.S.; Roblyer, L.; Sanghavi, R.; Ortigoza, E.B. Antibiotic duration and gastric dysmotility in preterm neonates. J. Pediatr. Gastroenterol. Nutr. 2024, 79, 533–540. [Google Scholar] [CrossRef]
- Ortigoza, E.B.; Cagle, J.; Brown, L.S.; Mansi, S.; Gosser, S.P.; Montgomery, A.D.; Foresman, Z.; Boren, M.L.; Pettit, P.S.; Thompson, T.D.; et al. Tachygastria in Preterm Infants: A Longitudinal Cohort Study. J. Pediatr. Gastroenterol. Nutr. 2022, 75, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Ortigoza, E.B.; Cagle, J.; Chien, J.H.; Oh, S.; Brown, L.S.; Neu, J. Electrogastrography, Near-infrared Spectroscopy, and Acoustics to Measure Gastrointestinal Development in Preterm Babies. J. Pediatr. Gastroenterol. Nutr. 2018, 66, e146–e152. [Google Scholar] [CrossRef]
- Welch, P. The use of fast Fourier transform for the estimation of power spectra: A method based on time averaging over short, modified periodograms. IEEE Trans. Audio Electroacoust. 1967, 15, 70–73. [Google Scholar] [CrossRef]
- Curran-Everett, D. Explorations in statistics: The analysis of ratios and normalized data. Adv. Physiol. Educ. 2013, 37, 213–219. [Google Scholar] [CrossRef]
- Efron, B.; Tibshirani, R.J. An Introduction to the Bootstrap, 1st ed.; Chapman and Hall/CRC: New York, NY, USA, 1994. [Google Scholar]
- Zoubir, A.M.; Boashash, B. The bootstrap and its application in signal processing. IEEE Signal Process. Mag. 1998, 15, 56–76. [Google Scholar] [CrossRef]
- Dwivedi, A.K.; Mallawaarachchi, I.; Alvarado, L.A. Analysis of small sample size studies using nonparametric bootstrap test with pooled resampling method. Stat. Med. 2017, 36, 2187–2205. [Google Scholar] [CrossRef] [PubMed]
- Verhagen, M.A.; Van Schelven, L.J.; Samsom, M.; Smout, A.J. Pitfalls in the analysis of electrogastrographic recordings. Gastroenterology 1999, 117, 453–460. [Google Scholar] [CrossRef]
- Chang, F.Y. Electrogastrography: Basic knowledge, recording, processing and its clinical applications. J. Gastroenterol. Hepatol. 2005, 20, 502–516. [Google Scholar] [CrossRef] [PubMed]
- Neu, J. Neonatal necrotizing enterocolitis: An update. Acta Paediatr. 2005, 94, 100–105. [Google Scholar] [CrossRef]




| Number (n) of Patients | NFI (n = 75) | DFI (n = 5) | PFI (n = 4) |
|---|---|---|---|
| Gestational Age (weeks) | 29 [24–39] | 30 [26–33] | 27 [25–28] |
| Birthweight (g) | 1391 [450–4245] | 1350 [810–2000] | 895 [710–1150] |
| Sex | |||
| Female | 41 [55%] | 3 [60%] | 2 [50%] |
| Male | 34 [45%] | 2 [40%] | 2 [50%] |
| Race/Ethnicity | |||
| Black Non-Hispanic/Latino | 20 [27%] | 3 [60%] | 3 [75%] |
| White Non-Hispanic/Latino | 2 [3%] | 0 [0] | 0 [0%] |
| Hispanic/Latino | 50 [67%] | 2 [40%] | 1 [25%] |
| Asian | 1 [1%] | 0 [0] | 0 [0] |
| Unknown/Not Reported | 2 [3%] | 0 [0] | 0 [0] |
| Feeding Type | |||
| Mother’s Own Milk (MoM) | 45 [60%] | 3 [60%] | 4 [100%] |
| Donor Human Milk (DHM) | 11 [15%] | 1 [20%] | 0 [0%] |
| Formula | 18 [25%] | 1 [20%] | 0 [0%] |
| Mixture of MoM and DHM | 0 [0%] | 0 [0%] | 0 [0%] |
| Mixture of MoM and Formula | 1 [1%] | 0 [0%] | 0 [0%] |
| Feeding Volume (mL/Kg) | 11.7 [1.6–20.4] | 9.5 [2.5–15.4] | 13.1 [9.1–17.2] |
| Age of First Feed (days) | 2 [1–4] | 2 [1–3] | 2 [2–3] |
| Days of Hospitalization | 68 [3–197] | 62 [15–151] | 90 [39–134] |
| Days on Parenteral Nutrition | 11 [0–35] | 10 [0–17] | 60 [32–103] |
| Central Line Days | 10 [0–38] | 8 [0–13] | 40 [8–105] |
| Days to Full Feeds | 9 [2–35] | 8 [6–9] | 20 [6–34] |
| Days to Sustained Feeding Tolerance (SFT) * | 10 [4–34] | 8 [6–10] | 25 [7–53] |
| Number of Times NPO | 2 [0–10] | 1 [0–3] | 6 [3–14] |
| Number of Days NPO | 2 [0–11] | 1 [0–3] | 16 [9–29] |
| Growth Velocity, g/Kg/day | 15 [10–29] | 9 [8–10] | 5 [−11–14] |
| NEC Stage ≥ IIa | |||
| Medical | 0 [0%] | 0 [0%] | 1 [25%] |
| Surgical or Death | 0 [0%] | 0 [0%] | 2 [50%] |
| Bowel Obstruction (Volvulus) | 0 [0%] | 0 [0%] | 1 [25%] |
| Death | 0 [0%] | 0 [0%] | 1 [25%] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Norasteh, S.; Roblyer, L.; Sanghavi, R.; Liu, H.; Ortigoza, E.B. Electrogastrography-Derived Mean Power Ratio as an Exploratory Objective Measure of Feeding Intolerance in Preterm Infants. Bioengineering 2026, 13, 342. https://doi.org/10.3390/bioengineering13030342
Norasteh S, Roblyer L, Sanghavi R, Liu H, Ortigoza EB. Electrogastrography-Derived Mean Power Ratio as an Exploratory Objective Measure of Feeding Intolerance in Preterm Infants. Bioengineering. 2026; 13(3):342. https://doi.org/10.3390/bioengineering13030342
Chicago/Turabian StyleNorasteh, Soheila, Lindsay Roblyer, Rinarani Sanghavi, Hanli Liu, and Eric B. Ortigoza. 2026. "Electrogastrography-Derived Mean Power Ratio as an Exploratory Objective Measure of Feeding Intolerance in Preterm Infants" Bioengineering 13, no. 3: 342. https://doi.org/10.3390/bioengineering13030342
APA StyleNorasteh, S., Roblyer, L., Sanghavi, R., Liu, H., & Ortigoza, E. B. (2026). Electrogastrography-Derived Mean Power Ratio as an Exploratory Objective Measure of Feeding Intolerance in Preterm Infants. Bioengineering, 13(3), 342. https://doi.org/10.3390/bioengineering13030342







