Degeneration-Driven and Load-Modulated Fluid-Driven Viscoelasticity of the Human Intervertebral Disc: A Probabilistic Biphasic Swelling Modeling Study
Abstract
1. Introduction
2. Methods
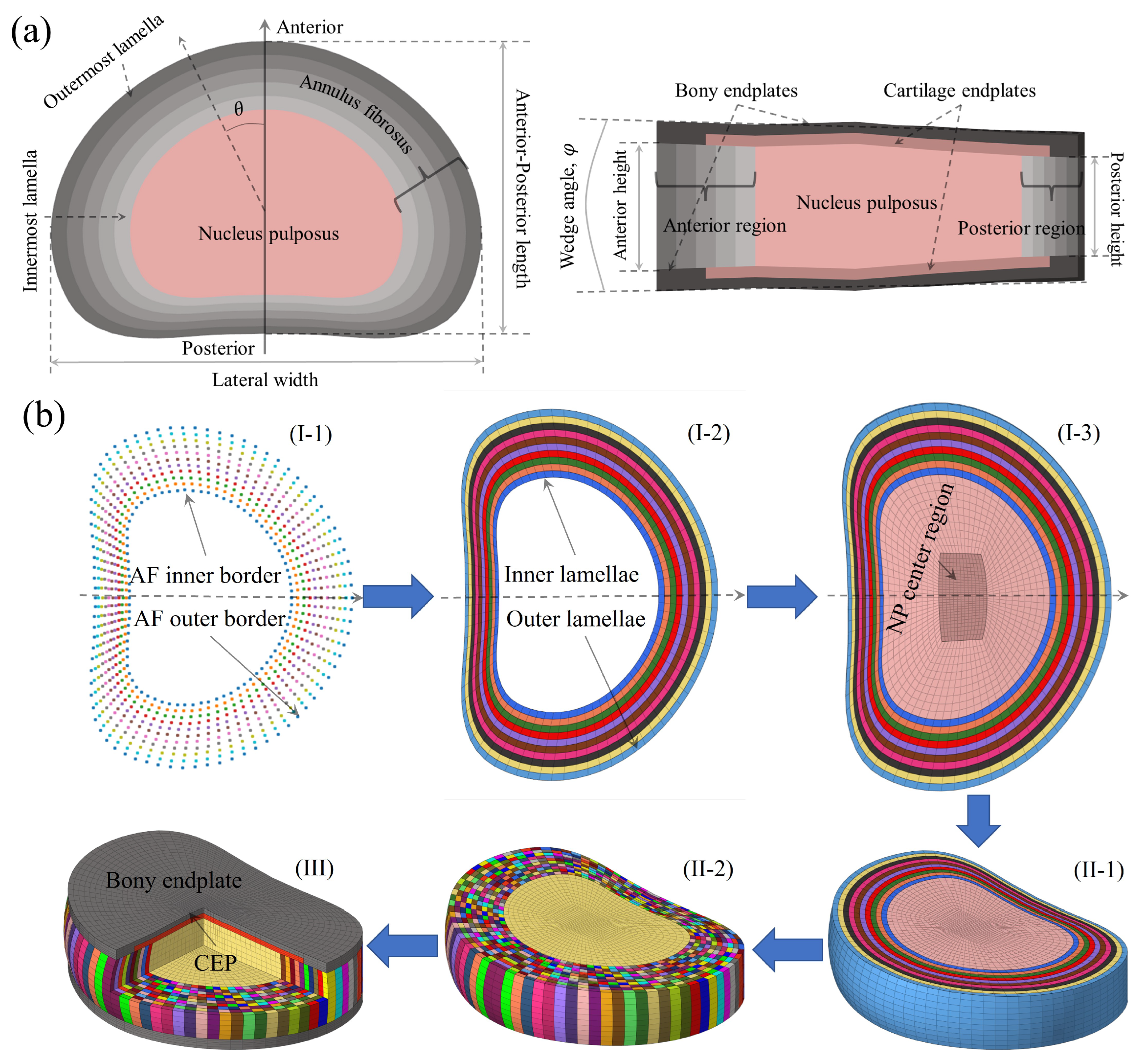
2.1. Geometric Parameterization and Meshing Process
2.2. Sampling of Intervertebral Discs
2.3. Constitutive Models and Characteristics of Degeneration
2.4. Loading Protocols
2.5. Data Analysis Methods
3. Results
3.1. Swelling Tests
3.2. Creep Tests
3.3. Relaxation Tests
3.4. p-Values Between Different Loading Levels
4. Discussion
4.1. Selections of Rheological Model
4.2. Effect of Degeneration
4.3. Effect of Loading Magnitudes
4.4. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Martin, J.T.; Oldweiler, A.B.; Spritzer, C.E.; Soher, B.J.; Erickson, M.M.; Goode, A.P.; DeFrate, L.E. A magnetic resonance imaging framework for quantifying intervertebral disc deformation in vivo: Reliability and application to diurnal variations in lumbar disc shape. J. Biomech. 2018, 71, 291–295. [Google Scholar] [CrossRef]
- Broberg, K.B. Slow deformation of intervertebral discs. J. Biomech. 1993, 26, 501–512. [Google Scholar] [CrossRef]
- O’Connell, G.D.; Jacobs, N.T.; Sen, S.; Vresilovic, E.J.; Elliott, D.M. Axial creep loading and unloaded recovery of the human intervertebral disc and the effect of degeneration. J. Mech. Behav. Biomed. Mater. 2011, 4, 933–942. [Google Scholar] [CrossRef]
- Feki, F.; Zaïri, F.; Tamoud, A.; Moulart, M.; Taktak, R.; Haddar, N.; Zaïri, F. Understanding the recovery of the intervertebral disc: A comprehensive review of in vivo and in vitro studies. J. Bionic. Eng. 2024, 21, 1919–1948. [Google Scholar] [CrossRef]
- Feki, F.; Taktak, R.; Kandil, K.; Derrouiche, A.; Moulart, M.; Haddar, N.; Zaïri, F.; Zaïri, F. How Osmoviscoelastic Coupling Affects Recovery of Cyclically Compressed Intervertebral Disc. Spine 2020, 45, E1376–E1385. [Google Scholar] [CrossRef]
- Sun, Z.; Pan, J.; Liang, W.; Mi, C.; Yin, P. Lumbar intervertebral disc biomechanics: Geometry and collagen fiber orientation configurations effects. Biocybern. Biomed. Eng. 2025, 45, 496-–506. [Google Scholar] [CrossRef]
- Pollintine, P.; van Tunen, M.S.; Luo, J.; Brown, M.D.; Dolan, P.; Adams, M.A. Time-dependent compressive deformation of the ageing spine: Relevance to spinal stenosis. Spine 2010, 35, 386–394. [Google Scholar] [CrossRef]
- Azril; Huang, K.Y.; Hobley, J.; Rouhani, M.; Liu, W.L.; Jeng, Y.R. Correlation of the degenerative stage of a disc with magnetic resonance imaging, chemical content, and biomechanical properties of the nucleus pulposus. J. Biomed. Mater. Res. A 2023, 111, 1054–1066. [Google Scholar] [CrossRef] [PubMed]
- Crump, K.B.; Alminnawi, A.; Bermudez-Lekerika, P.; Compte, R.; Gualdi, F.; McSweeney, T.; Muñoz-Moya, E.; Nüesch, A.; Geris, L.; Dudli, S.; et al. Cartilaginous endplates: A comprehensive review on a neglected structure in intervertebral disc research. JOR Spine 2023, 6, e1294. [Google Scholar] [CrossRef] [PubMed]
- DeLucca, J.F.; Cortes, D.H.; Jacobs, N.T.; Vresilovic, E.J.; Duncan, R.L.; Elliott, D.M. Human cartilage endplate permeability varies with degeneration and intervertebral disc site. J. Biomech. 2016, 49, 550–557. [Google Scholar] [CrossRef]
- Yang, M.; Cui, Y.; Zhang, Y.; Wu, H.; Hu, B.; Wang, S.; Liu, W. Quantitative characterization of the elasticity, net creep, and swelling of the intervertebral disc: An in vitro experiment. J. Bionic. Eng. 2022, 19, 1077-–1086. [Google Scholar] [CrossRef]
- Vergroesen, P.A.; Emanuel, K.S.; Peeters, M.; Kingma, I.; Smit, T.H. Are axial intervertebral disc biomechanics determined by osmosis? J. Biomech. 2018, 70, 4–9. [Google Scholar] [CrossRef]
- Sciortino, V.; Jansen, J.U.; Cerniglia, D.; Ingrassia, T.; Wilke, H.-J. Intervertebral disc creep behaviour through viscoelastic models: An in-vitro study. Discov. Appl. Sci. 2024, 6, 392. [Google Scholar] [CrossRef]
- Vergroesen, P.A.; van der Veen, A.J.; Emanuel, K.S.; van Dieën, J.H.; Smit, T.H. The poro-elastic behaviour of the intervertebral disc: A new perspective on diurnal fluid flow. J. Biomech. 2016, 49, 857–863. [Google Scholar] [CrossRef] [PubMed]
- Morino, C.; Middleton, S.; Op’t, Eynde, J.; Dimbath, E.; Kait, J.; Luck, J.; Bass, C. Primary Creep Characterization in Porcine Lumbar Spine Subject to Repeated Loading. Ann. Biomed. Eng. 2024, 1–14, Correction in Ann. Biomed. Eng. 2024. https://doi.org/10.1007/s10439-024-03579-w. [Google Scholar] [CrossRef]
- Feki, F.; Taktak, R.; Haddar, N.; Moulart, M.; Zaïri, F.; Zaïri, F. Overloading effect on the osmo-viscoelastic and recovery behavior of the intervertebral disc. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2024, 238, 430–437. [Google Scholar] [CrossRef]
- Schmidt, H.; Schilling, C.; Reyna, A.L.P.; Shirazi-Adl, A.; Dreischarf, M. Fluid-flow dependent response of intervertebral discs under cyclic loading: On the role of specimen preparation and preconditioning. J. Biomech. 2016, 49, 846–856. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Wei, L.; Wang, G.; Pugno, N.M.; Chen, Q.; Li, Z. In silico evaluation of in vivo degradation kinetics of poly(lactic acid) vascular stent devices. J. Funct. Biomater. 2024, 15, 135. [Google Scholar] [CrossRef]
- Sun, Z.; Mi, C. On the identification of the ultra-structural organization of elastic fibers and their effects on the integrity of annulus fibrosus. J. Biomech. 2023, 157, 111728. [Google Scholar] [CrossRef]
- Sun, Z.; Sun, Y.; Mi, C. Comprehensive modeling of annulus fibrosus: From biphasic refined characterization to damage accumulation under viscous loading. Acta Biomater. 2024, 174, 228–244. [Google Scholar] [CrossRef]
- Sun, Z.; Sun, Y.; Lu, T.; Li, J.; Mi, C. A swelling-based biphasic analysis on the quasi-static biomechanical behaviors of healthy and degenerative intervertebral discs. Comput. Methods Programs Biomed. 2023, 235, 107513. [Google Scholar] [CrossRef]
- Showalter, B.L.; DeLucca, J.F.; Peloquin, J.M.; Cortes, D.H.; Yoder, J.H.; Jacobs, N.T.; Wright, A.C.; Gee, J.C.; Vresilovic, E.J.; Elliott, D.M. Novel human intervertebral disc strain template to quantify regional three-dimensional strains in a population and compare to internal strains predicted by a finite element model. J. Orthop. Res. 2016, 34, 1264–1273. [Google Scholar] [CrossRef]
- Yoder, J.H.; Peloquin, J.M.; Song, G.; Tustison, N.J.; Moon, S.M.; Wright, A.C.; Vresilovic, E.J.; Gee, J.C.; Elliott, D.M. Internal three-dimensional strains in human intervertebral discs under axial compression quantified noninvasively by magnetic resonance imaging and image registration. J. Biomech. Eng. 2014, 136, 111008. [Google Scholar] [CrossRef]
- Bezci, S.E.; Lim, S.; O’Connell, G.D. Nonlinear stress-dependent recovery behavior of the intervertebral disc. J. Mech. Behav. Biomed. Mater. 2020, 110, 103881. [Google Scholar] [CrossRef]
- Tamoud, A.; Zaïri, F.; Zaïri, F. A data-driven microstructure-based model for predicting circumferential behavior and failure in degenerated human annulus fibrosus. Acta Biomater. 2024, 190, 379–397. [Google Scholar] [CrossRef]
- Newell, N.; Little, J.P.; Christou, A.; Adams, M.A.; Adam, C.J.; Masouros, S.D. Biomechanics of the human intervertebral disc: A review of testing techniques and results. J. Mech. Behav. Biomed. Mater. 2017, 69, 420–434. [Google Scholar] [CrossRef] [PubMed]
- Maas, S.A.; Ellis, B.J.; Ateshian, G.A.; Weiss, J.A. FEBio: Finite elements for biomechanics. J. Biomech. Eng. 2012, 134, 011005. [Google Scholar] [CrossRef]
- Sun, Z.; Mi, C. Biomechanics of annulus fibrosus: Elastic fiber simplification and degenerative impact on damage initiation and propagation. J. Mech. Behav. Biomed Mater. 2024, 157, 106628. [Google Scholar] [CrossRef] [PubMed]
- Żak, M.; Szotek, S. Effect of hydration time in saline on the swelling and uniaxial tensile response of annulus fibrosus of the intervertebral disc. J. Funct. Biomater. 2025, 16, 365. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; O’Connell, G.D. Intervertebral disc swelling maintains strain homeostasis throughout the annulus fibrosus: A finite element analysis of healthy and degenerated discs. Acta. Biomater. 2019, 100, 61–74. [Google Scholar] [CrossRef]
- Nguyen, A.M.; Johannessen, W.; Yoder, J.H.; Wheaton, A.J.; Vresilovic, E.J.; Borthakur, A.; Elliott, D.M. Noninvasive quantification of human nucleus pulposus pressure with use of T1rho-weighted magnetic resonance imaging. J. Bone Jt. Surg. Am. 2008, 90, 796–802. [Google Scholar] [CrossRef]
- Sato, K.; Kikuchi, S.; Yonezawa, T. In vivo intradiscal pressure measurement in healthy individuals and in patients with ongoing back problems. Spine 1999, 24, 2468–2474. [Google Scholar] [CrossRef] [PubMed]
- Newman, H.R.; DeLucca, J.F.; Peloquin, J.M.; Vresilovic, E.J.; Elliott, D.M. Multiaxial validation of a finite element model of the intervertebral disc with multigenerational fibers to establish residual strain. JOR Spine 2021, 4, e1145. [Google Scholar] [CrossRef] [PubMed]
- Bezci, S.E.; O’Connell, G.D. Osmotic pressure alters time-dependent recovery behavior of the intervertebral disc. Spine 2018, 43, E334–E340. [Google Scholar] [CrossRef] [PubMed]
- Raftery, K.; Rahman, T.; Smith, N.; Schaer, T.; Newell, N. The role of the nucleus pulposus in intervertebral disc recovery: Towards improved specifications for nucleus replacement devices. J. Biomech. 2024, 166, 111990. [Google Scholar] [CrossRef]
- Vresilovic, E.J.; Johannessen, W.; Elliott, D.M. Disc mechanics with trans-endplate partial nucleotomy are not fully restored following cyclic compressive loading and unloaded recovery. J. Biomech. Eng. 2006, 128, 823–829. [Google Scholar] [CrossRef]
- Cachot, U.; Kandil, K.; Zaïri, F.; Zaïri, F. A multiscale finite element model of fluid-microstructure interactions in human intervertebral disc compression. Acta Biomater. 2025, 207, 398–413. [Google Scholar] [CrossRef]



| Parameters | Disc Height, mm | Anterior-Posterior Length, mm | Lateral Width, mm | Nucleus Pulposus Volume Ratio, % | Wedge Angle, ° |
|---|---|---|---|---|---|
| Mean values | 10.21 | 37.5 | 55.5 | 39.3 | 7.82 |
| Standard deviation | 1.8 | 6.21 | 6.3 | 5.5 | 6.07 |
| Property | BEPs | CEPs (H) | CEPs (D) | NP (H) | NP (D) | AF (H) | AF (D) |
|---|---|---|---|---|---|---|---|
| (MPa) | 12,000 | 0.25 | 0.328 | 0.065 | 0.065 | 0.027 | 0.027 |
| 0.3 | 0.16 | 0.25 | 0.24 | 0.24 | 0.16 | 0.16 | |
| N.A. | 1 | 1.06 | 0.95 | 0.95 | 0.09 | 0.09 | |
| M | N.A. | 4.9 | 3.9 | 1.9 | 1.9 | 4.8 | 4.8 |
| ( mm4/Ns) | 5.56 | 2.5 | 5.5 | 5.5 | 16 (47) a | 16 (47) a | |
| 0.6 | 0.6 | 0.6 | 0.8 | 0.8 | 0.8 (0.7) a | 0.8 (0.7) a | |
| (mmol/L) | N.A. | −326 | −365 | −300 | −100 | −300 (−100) a | −150 (−100) a |
| N.A. | 1 | 1 | 1 | 1 | 1 | 1 |
| Property | Collagen Fibers | Elastic Fibers () | Cartilage Endplate () | |||
|---|---|---|---|---|---|---|
| AO | AI | PO | PI | |||
| Modulus, , MPa | 123.98 | 52.85 | 98.7 | 20.17 | 0.62 | 7.01 |
| Rate of fiber stiffening, | 4.3 | 3.92 | 3.82 | 6.03 | 2.68 | 2.88 |
| Critical stretch square, | 1.04 | 1.16 | 1.21 | 1.39 | 1.93 | \ |
| Test | Preload | Variable | Protocol | Comparative Experiment |
|---|---|---|---|---|
| Swelling | Non | Fixed charge density | Load from 0 to full in 1 s and then hold constant for 30 h | [30] |
| Pre-compression 20 N | Swelling | Compressive force | Load from 0 to 20 N under the rate of 1 N/s then hold constant for 30 h | [3] |
| Pre-compression 50 N | Swelling | Compressive force | Load from 0 to 50 N under the rate of 1 N/s then hold constant for 30 h | [22,23] |
| Creep | Pre-compression 20 N | Compressive force | Load from 20 N to 500, 1000, or 1500 N in 1 s, 2 s, or 3 s, respectively, and then hold constant for 30 h | [3] |
| Stress-relaxation | Pre-compression 50 N | Compressive strain | Load from 0% to 5%, 10% and 15% in 5 s, 10 s and 15 s, respectively, and then hold for 30 h. | [22,23] |
| Group | Indicators | 500 N vs. 1000 N | 500 N vs. 1500 N | 1000 N vs. 1500 N |
|---|---|---|---|---|
| Healthy | Elastic-term displacement | 0.000 | 0.000 | 0.023 |
| Shot-term displacement | 0.000 | 0.000 | 0.004 | |
| Long-term displacement | 0.000 | 0.000 | 0.000 | |
| Total displacement | 0.000 | 0.000 | 0.001 | |
| Short-term time | 0.021 | 0.008 | 0.673 | |
| Long-term time | 0.000 | 0.000 | 0.054 | |
| Equilibrium time | 0.000 | 0.000 | 0.027 | |
| Degenerative | Elastic-term displacement | 0.002 | 0.000 | 0.060 |
| Shot-term displacement | 0.000 | 0.000 | 0.322 | |
| Long-term displacement | 0.000 | 0.000 | 0.020 | |
| Total displacement | 0.000 | 0.000 | 0.060 | |
| Short-term time | 0.000 | 0.000 | 0.001 | |
| Long-term time | 0.060 | 0.000 | 0.074 | |
| Equilibrium time | 0.027 | 0.000 | 0.038 |
| Group | Biomechanical Indicator | 5% vs. 10% | 5% vs. 15% | 10% vs. 15% |
|---|---|---|---|---|
| Healthy | Residual force | 0.000 | 0.000 | 0.000 |
| Short-term force | 0.000 | 0.000 | 0.000 | |
| Long-term force | 0.000 | 0.000 | 0.000 | |
| Short-term time | 0.214 | 0.000 | 0.000 | |
| Long-term time | 0.023 | 0.022 | 0.923 | |
| Equilibrium time | 0.001 | 0.000 | 0.003 | |
| Degenerative | Residual force | 0.000 | 0.000 | 0.000 |
| Short-term force | 0.000 | 0.000 | 0.000 | |
| Long-term force | 0.000 | 0.000 | 0.000 | |
| Short-term time | 0.000 | 0.000 | 0.007 | |
| Long-term time | 0.000 | 0.000 | 0.086 | |
| Equilibrium time | 0.000 | 0.000 | 0.482 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sun, Z.; Dang, Y.; Mi, C.; Gu, J.; Pan, J. Degeneration-Driven and Load-Modulated Fluid-Driven Viscoelasticity of the Human Intervertebral Disc: A Probabilistic Biphasic Swelling Modeling Study. Bioengineering 2026, 13, 312. https://doi.org/10.3390/bioengineering13030312
Sun Z, Dang Y, Mi C, Gu J, Pan J. Degeneration-Driven and Load-Modulated Fluid-Driven Viscoelasticity of the Human Intervertebral Disc: A Probabilistic Biphasic Swelling Modeling Study. Bioengineering. 2026; 13(3):312. https://doi.org/10.3390/bioengineering13030312
Chicago/Turabian StyleSun, Zhongwei, Yixuan Dang, Changwen Mi, Jie Gu, and Jiabao Pan. 2026. "Degeneration-Driven and Load-Modulated Fluid-Driven Viscoelasticity of the Human Intervertebral Disc: A Probabilistic Biphasic Swelling Modeling Study" Bioengineering 13, no. 3: 312. https://doi.org/10.3390/bioengineering13030312
APA StyleSun, Z., Dang, Y., Mi, C., Gu, J., & Pan, J. (2026). Degeneration-Driven and Load-Modulated Fluid-Driven Viscoelasticity of the Human Intervertebral Disc: A Probabilistic Biphasic Swelling Modeling Study. Bioengineering, 13(3), 312. https://doi.org/10.3390/bioengineering13030312






