Pareto-Based Diagnostics and Selection for Mechanics–Synergy Trade-Offs in Unmeasured Muscle Activation Reconstruction
Abstract
1. Introduction
- A two-stage reconstruction framework combining a synergy reconstruction prior (SynRc) and a mechanics-constrained multi-objective inverse problem with optional smoothness refinement;
- A Pareto-based diagnostic and selection workflow (utopia-closest + neighborhood sensitivity) and a practical reporting template for applications without ground truth;
- Synthetic verification and stress tests illustrating how trade-off geometry changes across observability regimes (i.e., how sensitive joint moments are to unmeasured activations) and under noise/mismatch;
- A minimal reduced-dimension parameterization demo that motivates scalable implementations.
2. Methods
2.1. Nomenclature
- muscle activation vector at time t (bounded in ).
- measured and unmeasured activations.
- measured-channel SynRc reconstruction error.
- synergy excitation at time t (bounded in ).
- joint-moment-consistency, neural-consistency, and smoothness objectives.
- Observability regime: The degree to which joint moments are informative about unmeasured activations under fixed kinematics. High observability means joint moments are sensitive to unmeasured channels; low observability means many unmeasured-activation patterns produce similar joint moments.
- M number of muscles.
- number of joint degrees of freedom (dimension of the joint-moment vector).
- r synergy dimension (rank) in NMF.
- T number of time samples.
- synergy weight matrix.
- Euclidean norm.
- weights for and in local refinement.
- inverse-dynamics and forward-computed joint moments.
2.2. Notation and Problem Setup
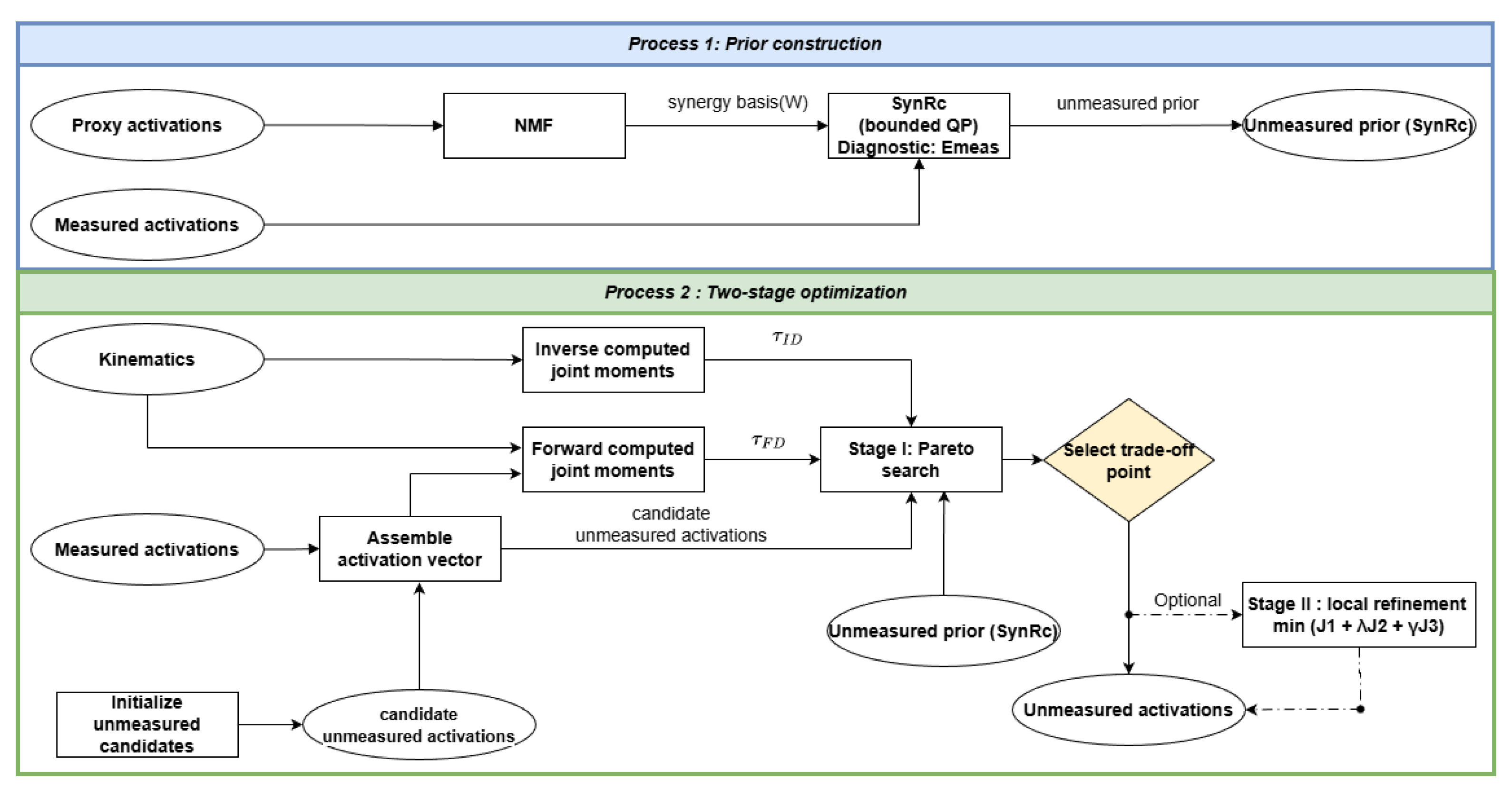
2.3. Algorithm Overview
- NMF: Identify a synergy basis from proxy activations using NMF (Section 2.4).
- SynRc: Estimate synergy excitations from the measured channels via a bounded quadratic program (QP) to obtain a synergy reconstruction prior (SynRc) for unmeasured activations (Section 2.5).
- Stage I: Approximate a stage-I Pareto set for over the unmeasured activation time series using derivative-free multi-objective search (Section 2.8).
- Select trade-off point: Select a stage-I point using the recommended heuristics and use-case guidance (Section 3.6).
- Stage II: Perform local refinement using a scalarized objective with an additional smoothness penalty (Section 2.8).
2.4. Synergy Basis Identification from Proxy Activations
Practical Sources of Proxy Activations
2.5. Synergy Reconstruction Prior from Sparse Observations (SynRc)
2.6. Forward Computation of Muscle-Induced Joint Moments
2.7. Multi-Objective Inverse Problem for Unmeasured Activations
Why a Second-Stage Optimization?
- Joint-moment-consistency objective:Normalization makes scale-invariant across tasks and facilitates comparison across Pareto solutions.Interpretation note: compares the inverse-dynamics target , which represents net joint moments, against the muscle-induced forward moments produced by OpenSim under fixed kinematics. Therefore, even the synthetic computed muscle control (CMC) reference is not guaranteed to minimize , and local refinement can achieve a lower by fitting residual mismatch between and . We accordingly interpret as a joint-moment-consistency diagnostic rather than a guarantee of physiological correctness.
- Neural-consistency objective (deviation from SynRc prior):
- Smoothness penalty (used in local refinement):
2.8. Two-Stage Solver: Pareto Front Search and Local Refinement
2.9. Synthetic Benchmark Design and Implementation Details
2.9.1. Case 2: Alternative Measured Set(Setup)
2.9.2. Case 3: Different Model and Task(Setup) (gait10dof18musc Walking)
2.10. Evaluation Metrics and Baselines
- SynRc-only baseline: The SynRc prior for the unmeasured activations (no joint-moment-consistency optimization).
- Joint-moment-only + smoothness baseline: A joint-moment-tracking reconstruction without a synergy prior, minimizing under a reduced-dimension parameterization.
- Proposed method: Two-stage multi-objective optimization with different stage-I selections and smoothness weights .
3. Results
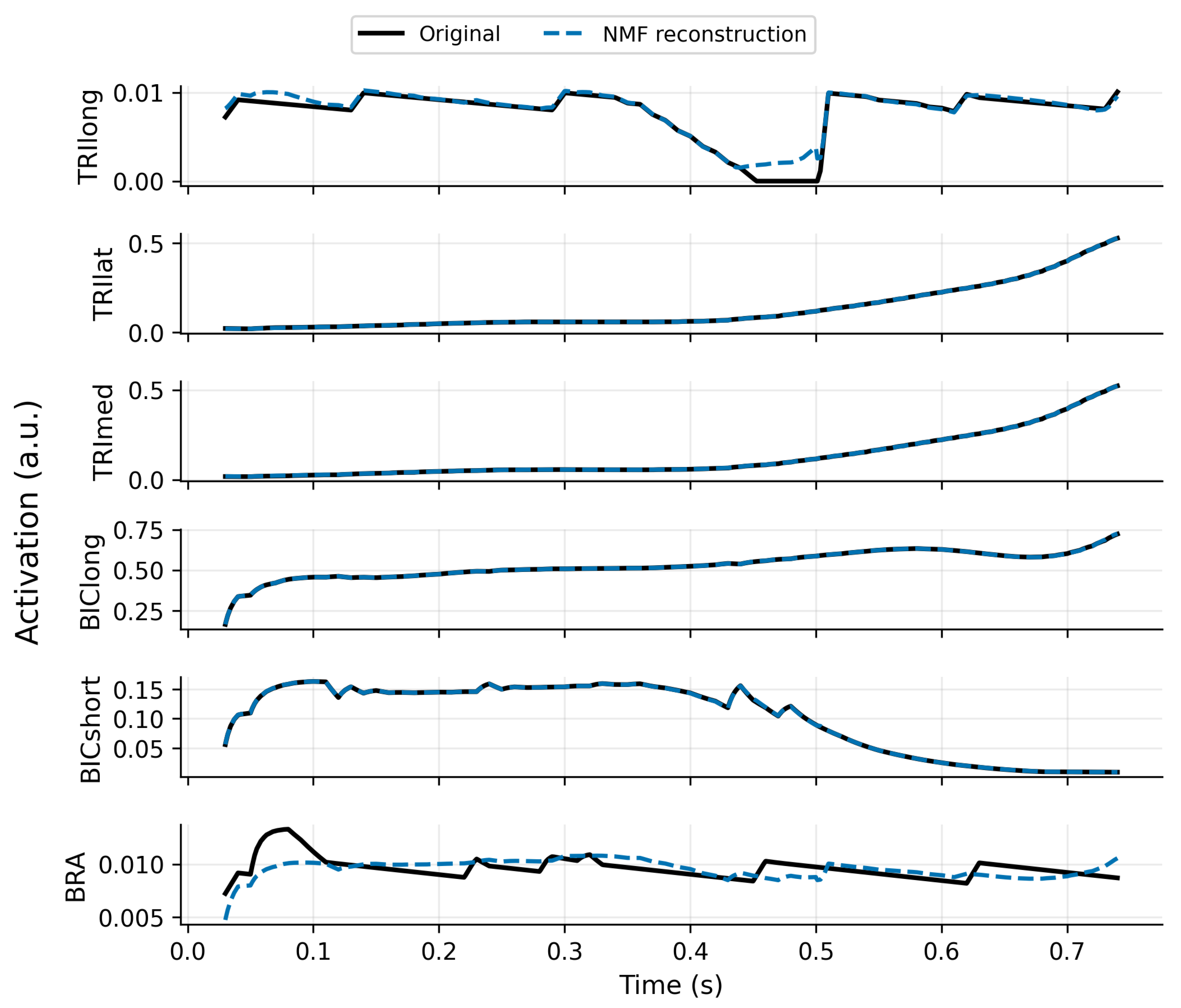
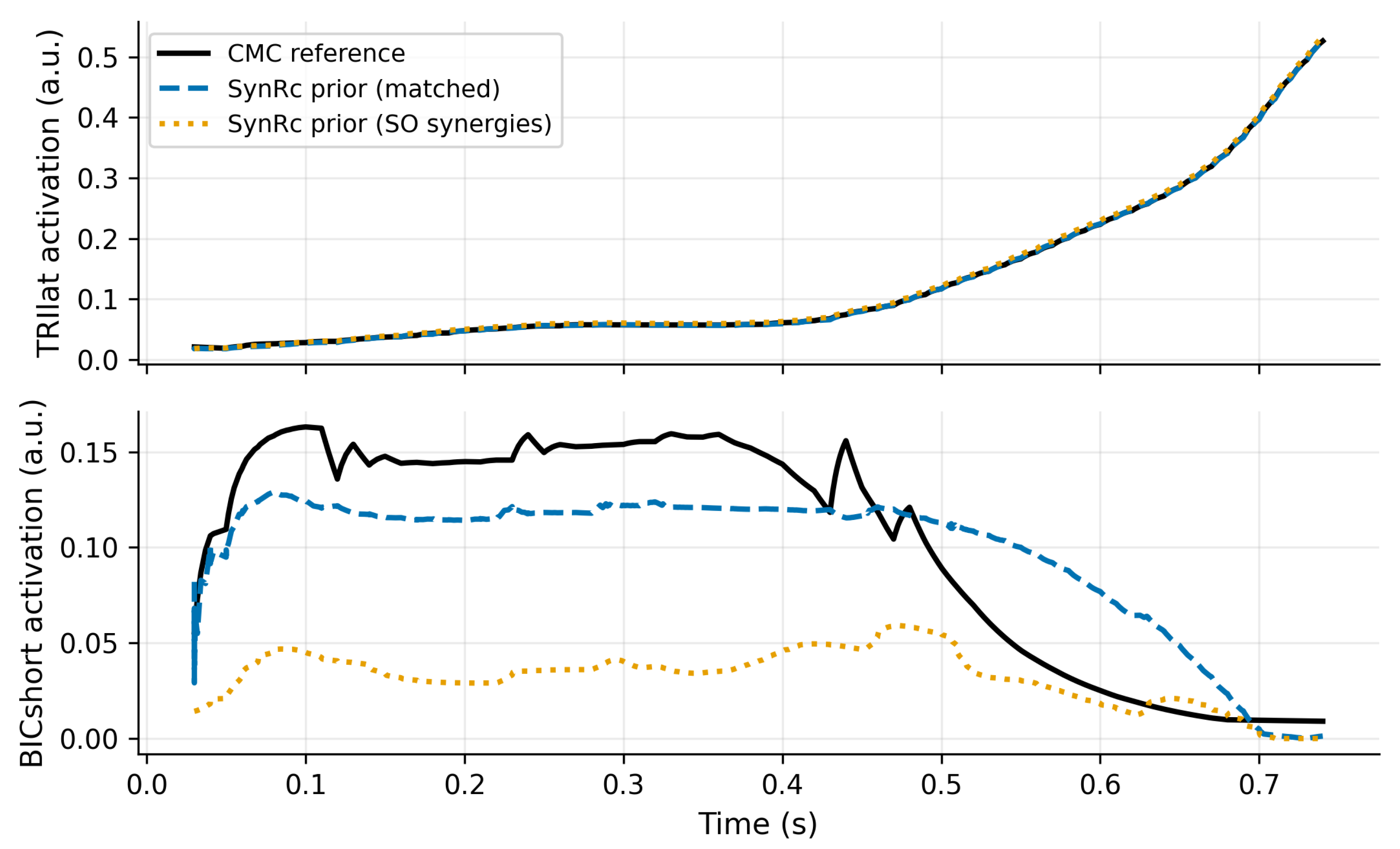
3.1. Synergy Identification and SynRc Prior Quality
Robustness to Proxy-Synergy Mismatch
3.2. Case 2: Alternative Measured Set
3.3. Case 3: Different Model and Task (gait10dof18musc Walking)
3.4. Synthetic Robustness Tests Under Noise and Mismatch
3.5. Multi-Case Synthetic Study: Observability Regimes
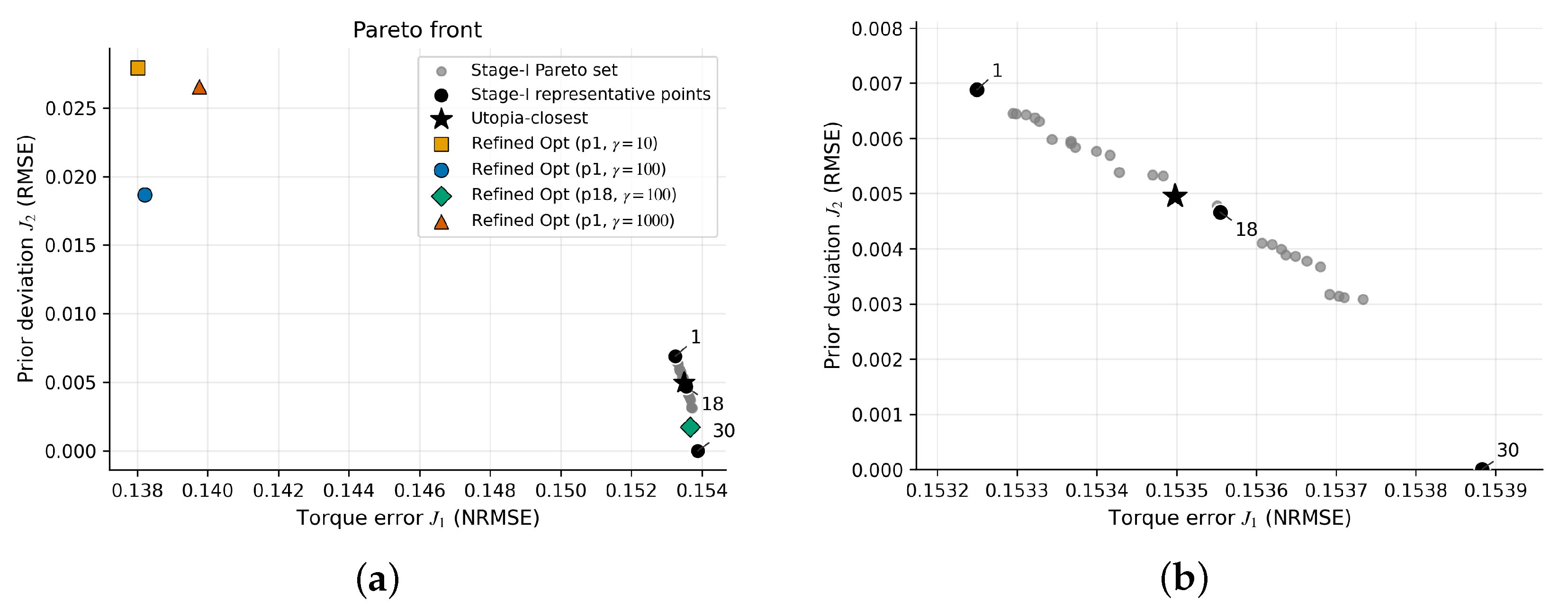
3.6. Pareto Front and Task-Dependent Solution Selection
3.6.1. Neighborhood Sensitivity
3.6.2. Scalarization Baseline
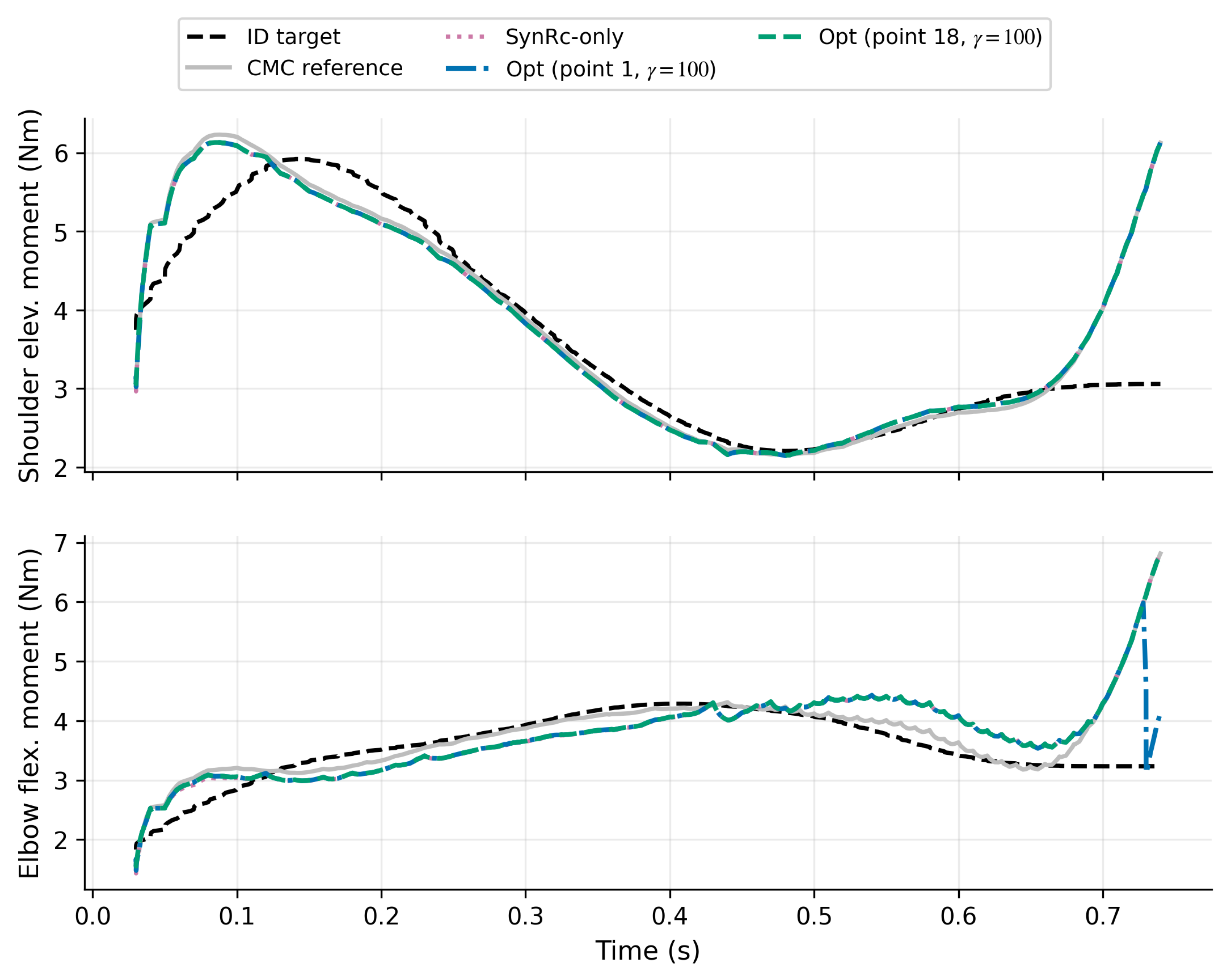
3.7. Unmeasured Activation Reconstruction Results
4. Discussion
4.1. Selection Workflow Without Ground Truth
- Report the stage-I Pareto set and the SynRc diagnostic (Equation (5)) as a proxy-transfer indicator;
- Report ID-target quality diagnostics (e.g., high-frequency ratio, spike score, estimated lag under synchronization checks, and the minimum achievable within the stage-I set) to contextualize how strongly should be trusted;
- State the selection rationale (e.g., endpoint toward low or low , or a compromise rule such as utopia-closest) and report the selected index and ;
- Report sensitivity by evaluating a small neighborhood on the stage-I set and, when refinement is used, a small sweep.
4.2. Relation to Prior Work
4.3. Limitations and Future Directions
4.4. Computational Considerations
4.5. Minimal Extensibility Demo: Reduced-Dimension Parameterization
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Delp, S.L.; Anderson, F.C.; Arnold, A.S.; Loan, P.; Habib, A.; John, C.T.; Guendelman, E.; Thelen, D.G. OpenSim: Open-source software to create and analyze dynamic simulations of movement. IEEE Trans. Bio-Med. Eng. 2007, 54, 1940–1950. [Google Scholar] [CrossRef]
- Seth, A.; Hicks, J.L.; Uchida, T.K.; Habib, A.; Dembia, C.L.; Dunne, J.J.; Ong, C.F.; DeMers, M.S.; Rajagopal, A.; Millard, M.; et al. OpenSim: Simulating musculoskeletal dynamics and neuromuscular control to study human and animal movement. PLoS Comput. Biol. 2018, 14, e1006223. [Google Scholar] [CrossRef]
- Anderson, F.C.; Pandy, M.G. Static and dynamic optimization solutions for gait are practically equivalent. J. Biomech. 2001, 34, 153–161. [Google Scholar] [CrossRef]
- De Groote, F.; Kinney, A.L.; Rao, A.V.; Geijtenbeek, T.; Blake, J.B.; Delp, S.L. Evaluation of Direct Collocation Optimal Control Problem Formulations for Solving the Muscle Redundancy Problem. Ann. Biomed. Eng. 2016, 44, 2922–2936. [Google Scholar] [CrossRef] [PubMed]
- Falisse, A.; Serrancolí, G.; Dembia, C.L.; Gillis, J.; Jonkers, I.; De Groote, F. Rapid predictive simulations with complex musculoskeletal models suggest that diverse healthy and pathological human gaits can emerge from similar control strategies. J. R. Soc. Interface 2019, 16, 20190402. [Google Scholar] [CrossRef]
- Dembia, C.L.; Bianco, N.A.; Falisse, A.; Hicks, J.L.; Delp, S.L. OpenSim Moco: Musculoskeletal optimal control. PLoS Comput. Biol. 2020, 16, e1008493. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, D.G.; Besier, T.F. An EMG-driven musculoskeletal model to estimate muscle forces and knee joint moments in vivo. J. Biomech. 2003, 36, 765–776. [Google Scholar] [CrossRef] [PubMed]
- Sartori, M.; Reggiani, M.; Farina, D.; Lloyd, D.G. EMG-Driven Forward-Dynamic Estimation of Muscle Force and Joint Moment about Multiple Degrees of Freedom in the Human Lower Extremity. PLoS ONE 2012, 7, e52618. [Google Scholar] [CrossRef]
- Sartori, M.; Farina, D.; Lloyd, D.G. Hybrid neuromusculoskeletal modeling to best track joint moments using a balance between muscle excitations derived from electromyograms and optimization. J. Biomech. 2014, 47, 3613–3621. [Google Scholar] [CrossRef]
- Ting, L.H.; McKay, J.L. Neuromechanics of muscle synergies for posture and movement. Curr. Opin. Neurobiol. 2007, 17, 622–628. [Google Scholar] [CrossRef]
- Tresch, M.C.; Jarc, A. The case for and against muscle synergies. Curr. Opin. Neurobiol. 2009, 19, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Cheung, V.C.K.; Turolla, A.; Agostini, M.; Silvoni, S.; Bennis, C.; Kasi, P.; Paganoni, S.; Bonato, P.; Bizzi, E. Muscle synergy patterns as physiological markers of motor cortical damage. Proc. Natl. Acad. Sci. USA 2012, 109, 14652–14656. [Google Scholar] [CrossRef]
- Ivanenko, Y.P.; Poppele, R.E.; Lacquaniti, F. Five basic muscle activation patterns account for muscle activity during human locomotion. J. Physiol. 2004, 556, 267–282. [Google Scholar] [CrossRef] [PubMed]
- Torres-Oviedo, G.; Ting, L.H. Muscle Synergies Characterizing Human Postural Responses. J. Neurophysiol. 2007, 98, 2144–2156. [Google Scholar] [CrossRef]
- Safavynia, S.A.; Torres-Oviedo, G.; Ting, L.H. Muscle Synergies: Implications for Clinical Evaluation and Rehabilitation of Movement. Top. Spinal Cord Inj. Rehabil. 2011, 17, 16–24. [Google Scholar] [CrossRef]
- Bianco, N.A.; Patten, C.; Fregly, B.J. Can Measured Synergy Excitations Accurately Construct Unmeasured Muscle Excitations? J. Biomech. Eng. 2018, 140, 011011. [Google Scholar] [CrossRef]
- Ao, D.; Shourijeh, M.S.; Patten, C.; Fregly, B.J. Evaluation of Synergy Extrapolation for Predicting Unmeasured Muscle Excitations from Measured Muscle Synergies. Front. Comput. Neurosci. 2020, 14, 588943. [Google Scholar] [CrossRef]
- Ao, D.; Fregly, B.J. Comparison of synergy extrapolation and static optimization for estimating multiple unmeasured muscle activations during walking. J. Neuroeng. Rehabil. 2024, 21, 194. [Google Scholar] [CrossRef]
- Hicks, J.L.; Uchida, T.K.; Seth, A.; Rajagopal, A.; Delp, S.L. Is My Model Good Enough? Best Practices for Verification and Validation of Musculoskeletal Models and Simulations of Movement. J. Biomech. Eng. 2015, 137, 020905. [Google Scholar] [CrossRef]
- Lee, D.D.; Seung, H.S. Learning the parts of objects by non-negative matrix factorization. Nature 1999, 401, 788–791. [Google Scholar] [CrossRef] [PubMed]
- Thelen, D.G. Adjustment of Muscle Mechanics Model Parameters to Simulate Dynamic Contractions in Older Adults. J. Biomech. Eng. 2003, 125, 70–77. [Google Scholar] [CrossRef]
- Audet, C.; Dennis, J.E. Mesh Adaptive Direct Search Algorithms for Constrained Optimization. SIAM J. Optim. 2006, 17, 188–217. [Google Scholar] [CrossRef]
- Holzbaur, K.R.S.; Murray, W.M.; Delp, S.L. A model of the upper extremity for simulating musculoskeletal surgery and analyzing neuromuscular control. Ann. Biomed. Eng. 2005, 33, 829–840. [Google Scholar] [CrossRef]
- Thelen, D.G.; Anderson, F.C.; Delp, S.L. Generating dynamic simulations of movement using computed muscle control. J. Biomech. 2003, 36, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Thelen, D.G.; Anderson, F.C. Using computed muscle control to generate forward dynamic simulations of human walking from experimental data. J. Biomech. 2006, 39, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Ao, D.; Vega, M.M.; Shourijeh, M.S.; Patten, C.; Fregly, B.J. EMG-driven musculoskeletal model calibration with estimation of unmeasured muscle excitations via synergy extrapolation. Front. Bioeng. Biotechnol. 2022, 10, 962959. [Google Scholar] [CrossRef] [PubMed]






| Item | Value | Description |
|---|---|---|
| Model | Arm26 (2 DOF, 6 muscles) | OpenSim upper-extremity benchmark (Section 2.9). |
| Time horizon | ( s) | Activation grid used for all objectives. |
| Measured set | TRIlong, TRImed, BIClong, BRA | “Observed” channels fixed during reconstruction. |
| Unmeasured set | TRIlat, BICshort | Decision variables in the inverse problem. |
| Synergy rank | NMF synergy dimension. | |
| SynRc QP weights | , , | Measured-channel fit, Tikhonov regularization, and temporal excitation smoothing (Equation (4); distinct from the refinement weights and in Equation (13)). |
| ID preprocessing | 4th-order Butterworth, 5 Hz, zero-phase | Low-pass filter applied to (Section 2.9). |
| Stage-I solver | paretosearch | ParetoSetSize=30, MaxFunctionEvaluations=90,000, MeshTolerance=0.2. |
| Stage-II solver | patternsearch | MaxIterations=60, FunctionTolerance=; refinement objective in Equation (13). |
| Smoothness weights | Representative sweep in Supplementary Figure S3. | |
| Joint-moment-only baseline | , , | Reduced-dimension knot parameterization used to solve without a synergy prior (reported later in the Section 3). |
| Method | (NRMSE) | TRImed RMSE | BRA RMSE |
|---|---|---|---|
| SO | 0.2914 | 0.07553 | 0.001286 |
| SynRc-only | 0.1414 | 0.0009898 | 0.00122 |
| Stage-I () | 0.1414 | 0.0009898 | 0.001277 |
| Opt () | 0.1414 | 0.0009898 | 0.001277 |
| Method | (NRMSE) | hamstrings_r RMSE | gastroc_r RMSE |
|---|---|---|---|
| SynRc-only | 0.1454 | 0.03085 | 0.03126 |
| Joint-moment-only | 0.1463 | 0.03077 | 0.03126 |
| Stage-I () | 0.1432 | 0.03029 | 0.03126 |
| Opt () | 0.1465 | 0.03089 | 0.03126 |
| Scenario | Scale | range | TRIlat RMSE | BICshort RMSE | ||
|---|---|---|---|---|---|---|
| Unmeasured strength scale ×0.2 | 0.2 | 22 | [0.4250, 0.4251] | 0.4251 | 0.0009 | 0.0332 |
| Unmeasured strength scale ×1.0 | 1.0 | 16 | [0.1532, 0.1539] | 0.1535 | 0.0009 | 0.0335 |
| Unmeasured strength scale ×2.0 | 2.0 | 30 | [0.3319, 0.3337] | 0.3327 | 0.0009 | 0.0329 |
| Method/Setting | (NRMSE) | (RMSE) | (a.u.) |
|---|---|---|---|
| CMC reference | 0.1414 | 0.0233 | 0.00205 |
| SynRc-only baseline | 0.1542 | 0 | 0.00316 |
| Joint-moment-only + smoothness (P = 10 knots) | 0.1449 | 0.0211 | 0.000918 |
| Opt (stage-I point 1, ) | 0.1380 | 0.0280 | 0.0116 |
| Opt (stage-I point 1, ) | 0.1382 | 0.0187 | 0.00607 |
| Opt (stage-I point 18, ) | 0.1537 | 0.00174 | 0.00204 |
| Opt (stage-I point 1, ) | 0.1398 | 0.0266 | 0.00720 |
| Method/Setting | TRIlat RMSE | BICshort RMSE |
|---|---|---|
| Static optimization (SO) | 0.0754 | 0.0418 |
| SynRc-only prior | 0.0009 | 0.0329 |
| Joint-moment-only + smoothness (P = 10 knots) | 0.0284 | 0.0324 |
| Opt (stage-I point 1, ) | 0.0395 | 0.0329 |
| Opt (stage-I point 1, ) | 0.0262 | 0.0329 |
| Opt (stage-I point 18, ) | 0.0010 | 0.0329 |
| Opt (stage-I point 1, ) | 0.0374 | 0.0329 |
| Item | Report (with Heuristic Flags) |
|---|---|
| (SynRc diagnostic) | Mean measured-channel RMSE Equation (5); interpret relatively across proxy-library choices. Heuristic: a large increase (e.g., the lowest among candidate priors) often indicates proxy mismatch, motivating a shift toward lower . |
| ID quality | Report HF ratio, spike score, and estimated lag (Supplementary Table S3), plus (minimum achievable within the stage-I set). Heuristic: HF ratio , spike score , or ms are warning signs of potential ID artifacts, motivating a shift toward lower . |
| Selection | Report the selection rule (endpoint, knee/utopia-closest) and the selected index with ; also report the endpoints and on the stage-I set for context. |
| sensitivity | Report a small neighborhood (e.g., ) with (Supplementary Table S4); if stage-II refinement is used, also report a small sweep (Supplementary Figure S3) to show whether refinement conclusions are stable. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Jiang, P.-H.; Chan, K.-Y. Pareto-Based Diagnostics and Selection for Mechanics–Synergy Trade-Offs in Unmeasured Muscle Activation Reconstruction. Bioengineering 2026, 13, 293. https://doi.org/10.3390/bioengineering13030293
Jiang P-H, Chan K-Y. Pareto-Based Diagnostics and Selection for Mechanics–Synergy Trade-Offs in Unmeasured Muscle Activation Reconstruction. Bioengineering. 2026; 13(3):293. https://doi.org/10.3390/bioengineering13030293
Chicago/Turabian StyleJiang, Po-Hsien, and Kuei-Yuan Chan. 2026. "Pareto-Based Diagnostics and Selection for Mechanics–Synergy Trade-Offs in Unmeasured Muscle Activation Reconstruction" Bioengineering 13, no. 3: 293. https://doi.org/10.3390/bioengineering13030293
APA StyleJiang, P.-H., & Chan, K.-Y. (2026). Pareto-Based Diagnostics and Selection for Mechanics–Synergy Trade-Offs in Unmeasured Muscle Activation Reconstruction. Bioengineering, 13(3), 293. https://doi.org/10.3390/bioengineering13030293






