Influence of Hydrolysis Degree and Molecular Weight on the Structure and Absorption Properties of Polyvinyl Alcohol Freeze-Dried Porous Polymer
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation and Characterization of Freeze-Dried Porous Polymer
2.3. Characterization of Freeze-Dried Porous Polymer
2.3.1. SEM
2.3.2. Porosity
2.3.3. Density
2.3.4. X-Ray Diffraction (XRD)
2.3.5. Fourier Transform Infrared Spectroscopy (FTIR)
2.3.6. Thermal Gravimetric Analyzer
2.3.7. Water Absorption Capacity (WAC)
2.3.8. Water Absorption Dynamic Curve
2.4. Statistical Analysis
3. Results
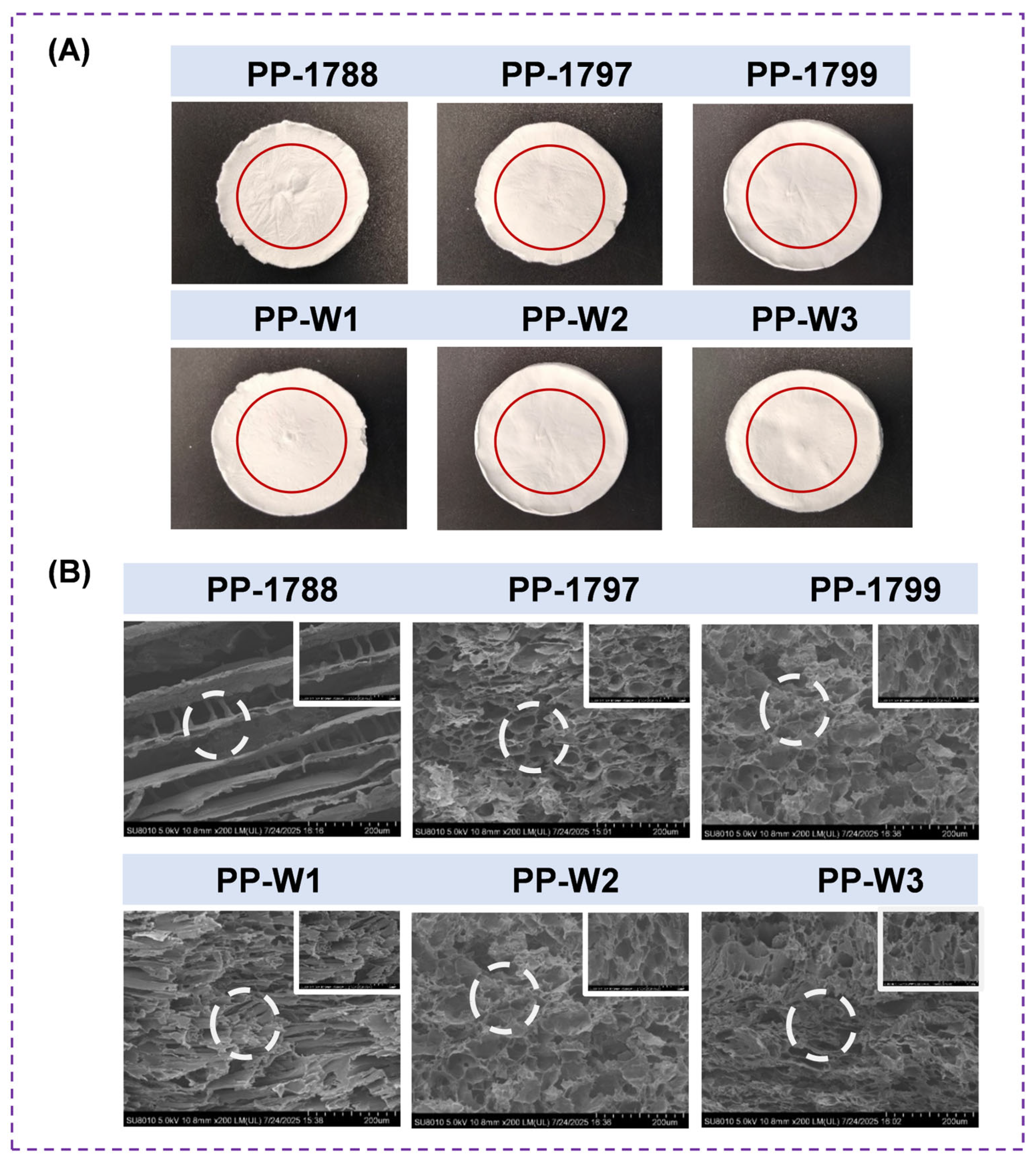
3.1. Morphology of Freeze-Dried Porous Polymer
3.2. Porosity and Density
3.3. XRD
3.4. FTIR
3.5. Thermal Gravimetric Analyzer
3.6. WAC
3.7. Water Absorption Kinetics
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PVA | Polyvinyl alcohol |
| PAAS | Sodium polyacrylate |
| PP-1788 | Mw (72,000–81,000) hydrolysis (87–89%) |
| PP-1797 | Mw (72,000–81,000) hydrolysis (96–98%) |
| PP-1799 | Mw (72,000–81,000) hydrolysis (98–99%) |
| PP-W1 | Hydrolysis (98–99%) Mw (31,000–50,000) |
| PP-W2 | Hydrolysis (98–99%) Mw (72,000–81,000 |
| PP-W3 | Hydrolysis (98–99%) Mw (190,000) |
References
- Li, C.; Wang, F.; Xiang, P.; Jiang, F.; Xiao, M. High-absorbent antibacterial konjac glucomannan/xanthan gum/bamboo fiber aerogel with carvacrol for chilled pork preservation. Int. J. Biol. Macromol. 2025, 306, 141624. [Google Scholar] [CrossRef]
- Song, X.; Wang, Z.; Zhou, M.; Li, Y.; Wei, Y.; Luo, Y.; Li, J.; Xu, B.; Shang, B.; Duan, Q. Humidity-responsive PCL/ICs/PVA pad with directional water transport for fruit preservation. Chem. Eng. J. 2025, 524, 169596. [Google Scholar] [CrossRef]
- Selvasekaran, P.; Chidambaram, R. Bioaerogels as food materials: A state-of-the-art on production and application in micronutrient fortification and active packaging of foods. Food Hydrocoll. 2022, 131, 107760. [Google Scholar] [CrossRef]
- De Azeredo, H.M.C. Antimicrobial nanostructures in food packaging. Trends Food Sci. Technol. 2013, 30, 56–69. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, X.; Chen, L.; Zhou, X.; Fan, X.; Hu, Y.; Niu, X.; Xu, X.; Zhou, G.; Ullah, N.; et al. Antibacterial aerogels with nano-silver reduced in situ by carboxymethyl cellulose for fresh meat preservation. Int. J. Biol. Macromol. 2022, 213, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Zheng, D.; He, J.; Liu, X.; Liu, T. Preparation and characterization of a thymol nanoemulsion-loaded multifunctional sustained-release corn straw cellulose nanocrystal/acetylated starch-based aerogel and its application in chilled meat preservation. Carbohydr. Polym. 2025, 348, 122758. [Google Scholar] [CrossRef] [PubMed]
- Ramli, R.A. A critical review of biodegradable polymers for food packaging: Enhancing functionality and environmental sustainability. Food Res. Int. 2026, 229, 118497. [Google Scholar] [CrossRef]
- Otoni, C.G.; Espitia, P.J.P.; Avena-Bustillos, R.J.; McHugh, T.H. Trends in antimicrobial food packaging systems: Emitting sachets and absorbent pads. Food Res. Int. 2016, 83, 60–73. [Google Scholar] [CrossRef]
- Prus-Walendziak, W.; Kozlowska, J. Lyophilized emulsions in the form of 3D porous matrices as a novel material for topical application. Materials 2021, 14, 950. [Google Scholar] [CrossRef]
- Qian, L.; Zhang, H. Controlled freezing and freeze drying: A versatile route for porous and micro-/nano-structured materials. J. Chem. Technol. Biotechnol. 2010, 86, 172–184. [Google Scholar] [CrossRef]
- Ben Rejeb, Z.; Abidli, A.; Zaoui, A.; Fashandi, M.; Selka, A.; Naguib, H.E.; Park, C.B. One-pot synthesis of rationally-designed flexible, robust, and hydrophobic ambient-dried molecularly-bridged silica aerogels with efficient and versatile oil/water separation applications. Adv. Compos. Hybrid Mater. 2024, 7, 188. [Google Scholar] [CrossRef]
- Xiao, M.; Tan, M.; Peng, C.; Jiang, F.; Wu, K.; Liu, N.; Li, D.; Yao, X. Soft and flexible polyvinyl alcohol/pullulan aerogels with fast and high-water absorption capacity for facial mask substrates. Int. J. Biol. Macromol. 2024, 264, 130469. [Google Scholar] [CrossRef] [PubMed]
- Rincón, E.; Espinosa, E.; Pinillos, M.; Serrano, L. Bioactive Absorbent Chitosan Aerogels Reinforced with Bay Tree Pruning Waste Nanocellulose with Antioxidant Properties for Burger Meat Preservation. Polymers 2023, 15, 866. [Google Scholar] [CrossRef]
- Long, L.Y.; Weng, Y.X.; Wang, Y.Z. Cellulose Aerogels: Synthesis, Applications, and Prospects. Polymers 2018, 10, 623. [Google Scholar] [CrossRef]
- Gosden, D.; Studley, M.; Rossiter, J. Material extrusion of sodium polyacrylate superabsorbent polymer. Addit. Manuf. 2023, 78, 103886. [Google Scholar] [CrossRef]
- Deng, H.; Su, J.; Zhang, W.; Khan, A.; Sani, M.A.; Goksen, G.; Kashyap, P.; Ezati, P.; Rhim, J.-W. A review of starch/polyvinyl alcohol (PVA) blend film: A potential replacement for traditional plastic-based food packaging film. Int. J. Biol. Macromol. 2024, 273, 132926. [Google Scholar] [CrossRef]
- Monjazeb Marvdashti, L.; Koocheki, A.; Yavarmanesh, M. Alyssum homolocarpum seed gum-polyvinyl alcohol biodegradable composite film: Physicochemical, mechanical, thermal and barrier properties. Carbohydr. Polym. 2017, 155, 280–293. [Google Scholar] [CrossRef]
- Chang, L.; Kong, B.; Liu, Q.; Du, X.; Xia, X. Effect of freeze-thaw processes on the water-absorption ability and mechanical properties of hydrogel pads with chitosan/poly(vinyl alcohol) based on citrate crosslinks. Food Chem. 2025, 471, 142785. [Google Scholar] [CrossRef]
- Chi, S.; Liu, X.; Wu, J.; Feng, Q.; Wang, L.; Li, J.; Sun, T. Preparation of polyvinyl alcohol/sodium alginate/Artemisia sphaerocephala Krasch gum hydrogels with excellent water absorption and its application in the preservation of Lateolabrax Japonicus fillets. Int. J. Biol. Macromol. 2025, 308, 141824. [Google Scholar] [CrossRef] [PubMed]
- Aktürk, A.; Erol Taygun, M.; Karbancıoğlu Güler, F.; Goller, G.; Küçükbayrak, S. Fabrication of antibacterial polyvinylalcohol nanocomposite mats with soluble starch coated silver nanoparticles. Colloids Surf. A Physicochem. Eng. Asp. 2019, 562, 255–262. [Google Scholar] [CrossRef]
- Jiang, C.; Yu, G.; Wang, P.; Yao, Z.; Mu, L.; Li, Q.; Shi, Y. Preparation of delignified wood fibers based absorbent pad for tray package and its application in preservation of cold and fresh pork. Ind. Crops Prod. 2024, 219, 119097. [Google Scholar] [CrossRef]
- Asadpour, S.; Raeisi Vanani, A.; Kooravand, M.; Asfaram, A. A review on zinc oxide/poly(vinyl alcohol) nanocomposites: Synthesis, characterization and applications. J. Clean. Prod. 2022, 362, 132297. [Google Scholar] [CrossRef]
- Mujahid, M.; Zubair, M.; Yaqoob, A.; Shahzad, S.; Ullah, A. Formulation and Evaluation of Licorice-Extract-Enhanced Chitosan, PVA, and Gelatin-Derived Hydrogels for Wound Dressing. Bioengineering 2025, 12, 439. [Google Scholar] [CrossRef]
- Ren, J.; Hasuo, K.; Wei, Y.; Tabata, I.; Hori, T.; Hirogaki, K. Investigation of the influences of the molecular weights and dosage ratios of polyvinyl alcohol on para-aramid nanofibers/polyvinyl alcohol composite aerogels. J. Appl. Polym. Sci. 2022, 139, e52960. [Google Scholar] [CrossRef]
- Arabkhani, P.; Asfaram, A. Development of a novel three-dimensional magnetic polymer aerogel as an efficient adsorbent for malachite green removal. J. Hazard. Mater. 2020, 384, 121394. [Google Scholar] [CrossRef]
- Mansur, H.S.; Sadahira, C.M.; Souza, A.N.; Mansur, A.A.P. FTIR spectroscopy characterization of poly (vinyl alcohol) hydrogel with different hydrolysis degree and chemically crosslinked with glutaraldehyde. Mater. Sci. Eng. C 2008, 28, 539–548. [Google Scholar] [CrossRef]
- Wang, F.; Xu, Z.; Chen, L.; Qiao, Z.; Hu, Y.; Fan, X.; Liu, Y.; Kang, Z.; Huang, F.; Han, M.; et al. Super absorbent resilience antibacterial aerogel with curcumin for fresh pork preservation. Food Control 2024, 159, 110289. [Google Scholar] [CrossRef]
- Dilamian, M.; Noroozi, B. Rice straw agri-waste for water pollutant adsorption: Relevant mesoporous super hydrophobic cellulose aerogel. Carbohydr. Polym. 2021, 251, 117016. [Google Scholar] [CrossRef] [PubMed]
- Mirmoeini, S.S.; Hosseini, S.H.; Lotfi Javid, A.; Esmaeili Koutamehr, M.; Sharafi, H.; Molaei, R.; Moradi, M. Essential oil-loaded starch/cellulose aerogel: Preparation, characterization and application in cheese packaging. Int. J. Biol. Macromol. 2023, 244, 125356. [Google Scholar] [CrossRef]
- Herbanu, A.; Ana, I.D.; Ardhani, R.; Siswomihardjo, W. Fibrous PVA Matrix Containing Strontium-Substituted Hydroxyapatite Nanoparticles from Golden Apple Snail (Pomacea canaliculata L.) Shells for Bone Tissue Engineering. Bioengineering 2023, 10, 844. [Google Scholar] [CrossRef]
- Zhu, C.; Yang, Q.; Tian, M.; Yang, W.; Min, C.; Fan, S.; Wang, D.; Li, X.; Zhang, D.; Hou, C. Sustainable nanofiber films based on polylactic acid/modified cellulose nanocrystals containing various types of polyphenols, exhibiting antioxidant activity and high stability. Food Chem. 2025, 477, 143514. [Google Scholar] [CrossRef]
- Espinosa, E.; Filgueira, D.; Rodríguez, A.; Chinga-Carrasco, G. Nanocellulose-Based Inks—Effect of Alginate Content on the Water Absorption of 3D Printed Constructs. Bioengineering 2019, 6, 65. [Google Scholar] [CrossRef]
- Fontes-Candia, C.; Erboz, E.; Martínez-Abad, A.; López-Rubio, A.; Martínez-Sanz, M. Superabsorbent food packaging bioactive cellulose-based aerogels from Arundo donax waste biomass. Food Hydrocoll. 2019, 96, 151–160. [Google Scholar] [CrossRef]
- Zhao, B.; Gu, Z.; Cheng, L.; Hong, Y. Non-covalent sorption properties of porous starch materials: Enhancement strategy and applications. Food Hydrocoll. 2024, 154, 110105. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, F.; Yu, Y.; Guo, R.; You, X.; Xiang, X.; Fan, X.; Niu, Y.; Wang, X.; Feng, X.; et al. Muti-network active aerogel based on PVA, bacterial cellulose, and sodium alginate: Characterization, kinetics studies, and application for mutton preservation. Food Hydrocoll. 2025, 163, 111033. [Google Scholar] [CrossRef]
- Liu, F.; Zhang, Y.; Xiao, X.; Cao, Y.; Jiao, W.; Bai, H.; Yu, L.; Duan, Q. Effects of polyvinyl alcohol content and hydrolysis degree on the structure and properties of extruded starch-based foams. Chem. Eng. J. 2023, 472, 144959. [Google Scholar] [CrossRef]
- Zanela, J.; Casagrande, M.; Reis, M.O.; Grossmann, M.V.E.; Yamashita, F. Biodegradable Sheets of Starch/Polyvinyl Alcohol (PVA): Effects of PVA Molecular Weight and Hydrolysis Degree. Waste Biomass Valor. 2019, 10, 319–326. [Google Scholar] [CrossRef]
- Jayasekara, R.; Harding, I.; Bowater, I.; Christie, G.B.Y.; Lonergan, G.T. Preparation, surface modification and characterization of solution cast starch PVA blended films. Polym. Test. 2004, 23, 17–27. [Google Scholar] [CrossRef]
- Lou, C.W.; Tian, F.; Zhou, X.; Zhang, L.; Li, S.; Lin, J.H.; Li, T.T. Dual-network PVA fiber-doped cellulose aerogel with labyrinth structure for low and medium frequency acoustic and flame retardant applications. Cellulose 2024, 31, 5795–5807. [Google Scholar] [CrossRef]






| Sample | Molecular Weight | Degree of Hydrolysis |
|---|---|---|
| PP-1788 | 72,000~81,000 | 87~89% |
| PP-1797 | 72,000~81,000 | 96~98% |
| PP-1799 | 72,000~81,000 | 98~99% |
| PP-W1 | 31,000~50,000 | 98~99% |
| PP-W2 | 72,000~81,000 | 98~99% |
| PP-W3 | 190,000 | 98~99% |
| Samples | Model | Curve-Fitted Equation | R2 | Kinetic Data (g/g/min) |
|---|---|---|---|---|
| PP-1797 | Pseudo-first-order kinetic | y= 0.350x − 0.001 | 0.98 | k1 0.35 |
| Pseudo-second-order kinetic | y = 1.967x + 0.002 | 0.78 | k2 1.97 | |
| PP-1799\PP-W2 | Pseudo-first-order kinetic | y = 0.409x − 0.234 | 0.92 | k1 0.41 |
| Pseudo-second-order kinetic | y = 1.392x + 0.966 | 0.76 | k2 1.40 | |
| PP-W3 | Pseudo-first-order kinetic | y = 0.452x − 0.265 | 0.98 | k1 0.45 |
| Pseudo-second-order kinetic | y = 1.814x + 1.007 | 0.63 | k2 1.81 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tian, M.; Zhu, C.; Yang, Q.; Fan, S.; Pang, J.; Liu, L.; Wang, D.; Zhang, D.; Li, X.; Hou, C. Influence of Hydrolysis Degree and Molecular Weight on the Structure and Absorption Properties of Polyvinyl Alcohol Freeze-Dried Porous Polymer. Bioengineering 2026, 13, 259. https://doi.org/10.3390/bioengineering13030259
Tian M, Zhu C, Yang Q, Fan S, Pang J, Liu L, Wang D, Zhang D, Li X, Hou C. Influence of Hydrolysis Degree and Molecular Weight on the Structure and Absorption Properties of Polyvinyl Alcohol Freeze-Dried Porous Polymer. Bioengineering. 2026; 13(3):259. https://doi.org/10.3390/bioengineering13030259
Chicago/Turabian StyleTian, Ming, Chaoqiao Zhu, Qingfeng Yang, Simin Fan, Jinkai Pang, Le Liu, Debao Wang, Dequan Zhang, Xin Li, and Chengli Hou. 2026. "Influence of Hydrolysis Degree and Molecular Weight on the Structure and Absorption Properties of Polyvinyl Alcohol Freeze-Dried Porous Polymer" Bioengineering 13, no. 3: 259. https://doi.org/10.3390/bioengineering13030259
APA StyleTian, M., Zhu, C., Yang, Q., Fan, S., Pang, J., Liu, L., Wang, D., Zhang, D., Li, X., & Hou, C. (2026). Influence of Hydrolysis Degree and Molecular Weight on the Structure and Absorption Properties of Polyvinyl Alcohol Freeze-Dried Porous Polymer. Bioengineering, 13(3), 259. https://doi.org/10.3390/bioengineering13030259









