Volumetric Modulated Arc Therapy Versus Dynamic Conformal Arc Therapy for Single Isocenter Stereotactic Radiotherapy of Multiple Brain Metastases
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. HyperArc Treatment Planning
2.3. DCA Treatment Planning
2.4. Data Analysis
- TVPIV: Target volume covered by the isodose curve of the prescribed dose.
- PIV: Volume covered by the isodose corresponding to the prescribed dose.
- TV: Total volume of the target.
- V50%: The volume of tissue receiving 50% of the prescribed dose.
- V100%: The volume of tissue receiving 100% of the prescribed dose.
3. Results
3.1. Plan Quality and Dosimetric Comparison
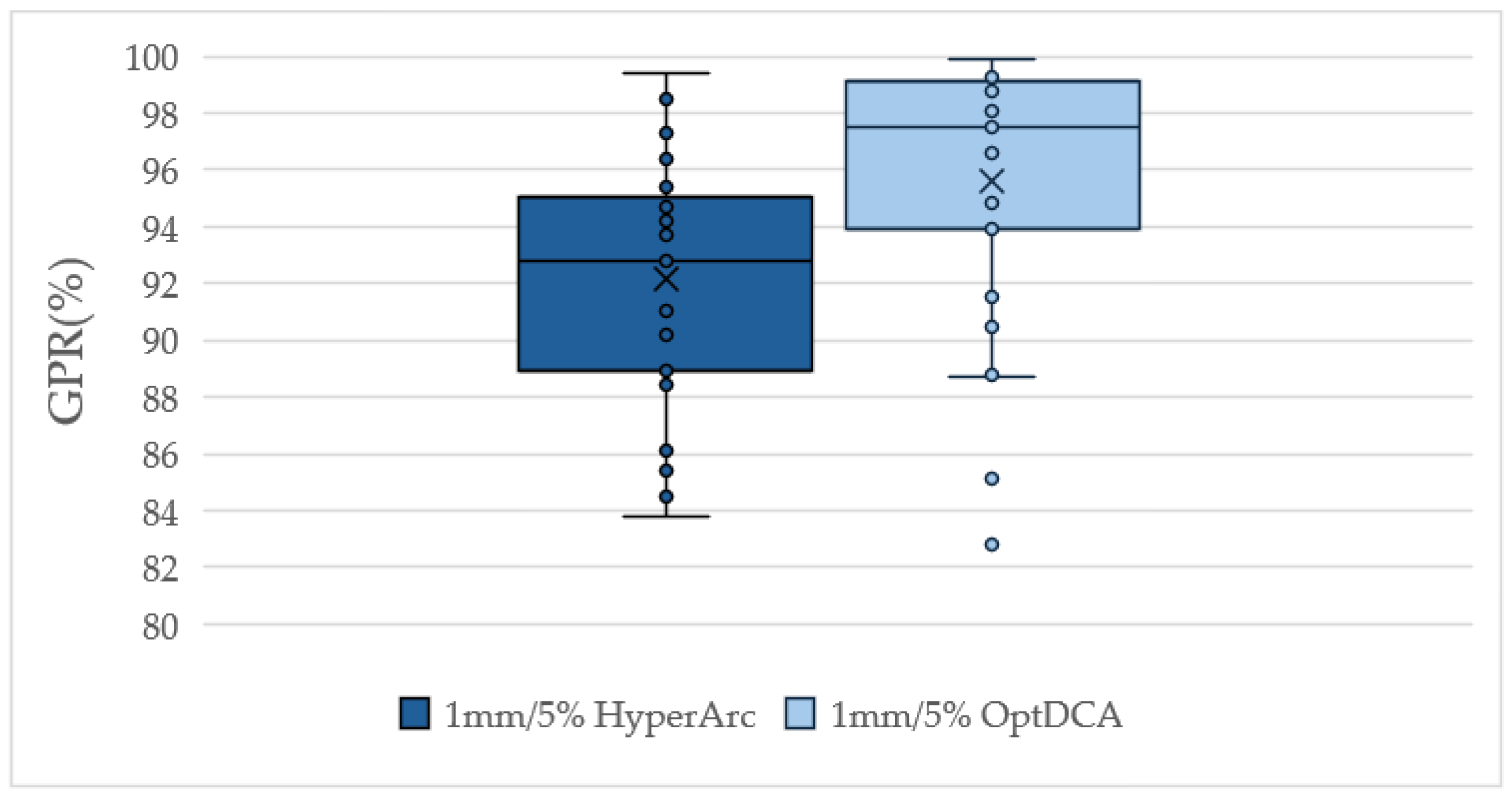
3.2. PSQA Results
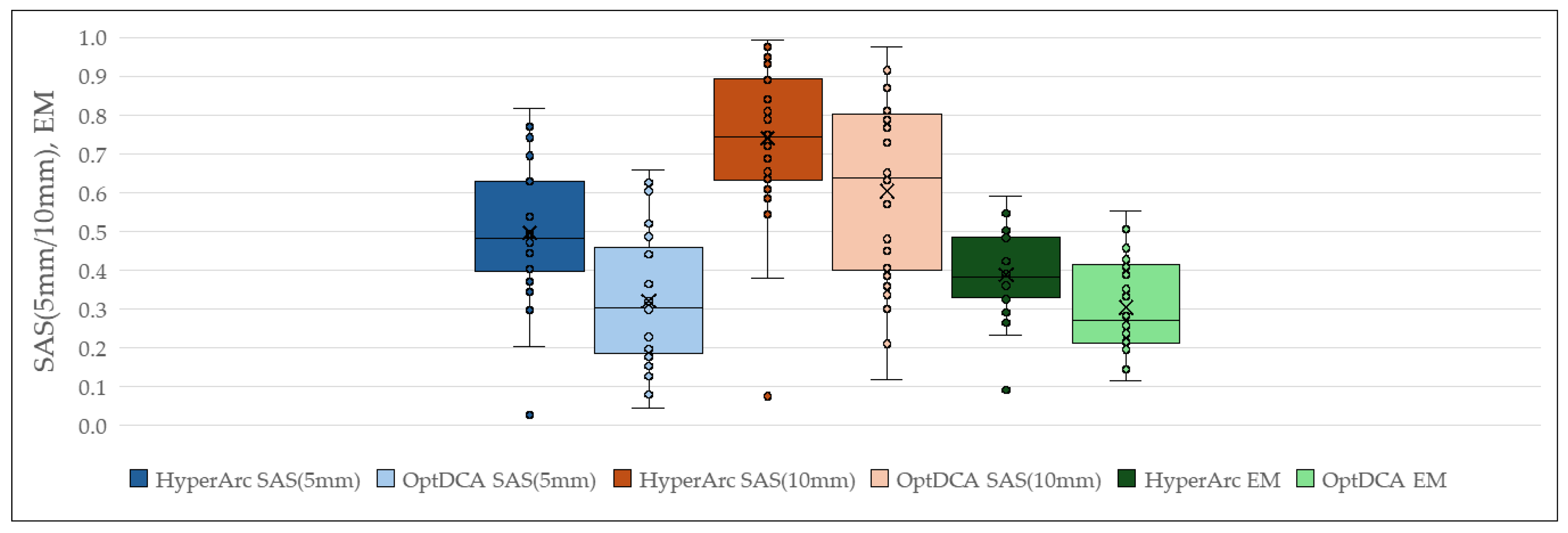
3.3. Plan Complexity Metrics
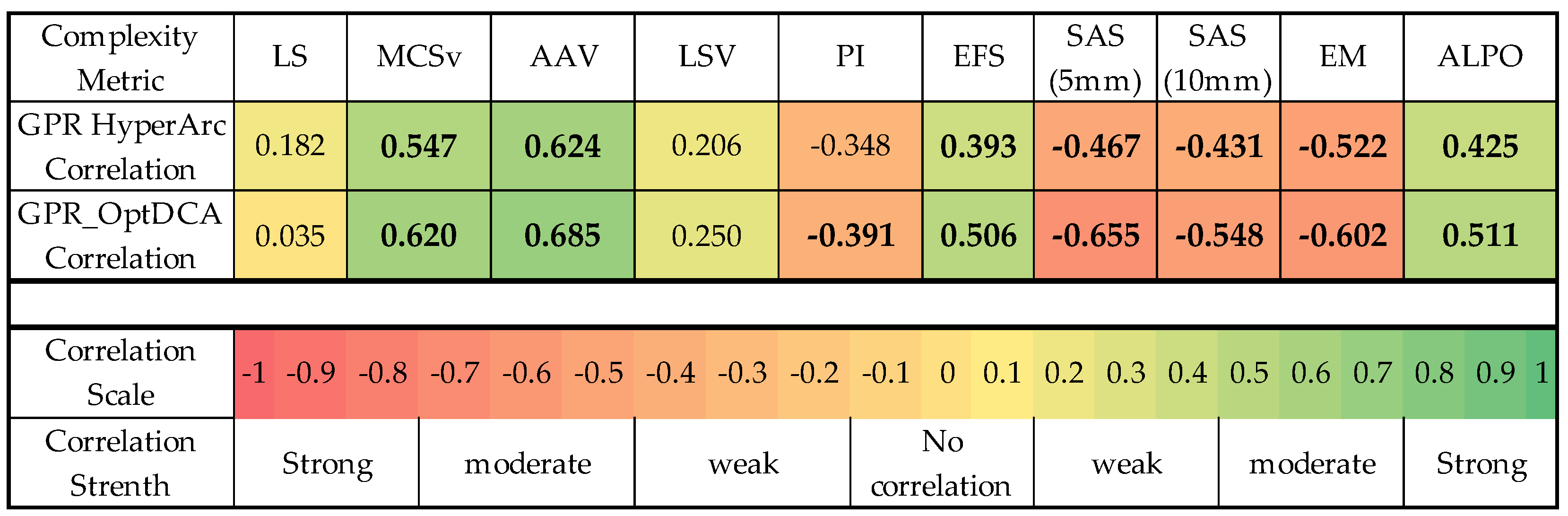
3.4. Correlation Between Plan Complexity and Deliverability
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| BM | Brain Metastases |
| SRS | Stereotactic Radiosurgery |
| LINAC | Linear Accelerators |
| MLC | Multileaf Collimator |
| VMAT | Volumetric Modulated Arc Therapy |
| DCA | Dynamic Conformal Arc |
| TPS | Treatment Patient System |
| FFF | Flattening Filter Free |
| PO | Photon Optimizer |
| MU | Monitor Unit |
| NTO | Normal Tissue Objective |
| PTV | Planning Target Volume |
| PCI | Paddick Conformity Index |
| GI | Gradient Index |
| GPR | Gamma Passing Rate |
| DVH | Dose Volume Histogram |
| PSQA | Patient Specific Quality Assurance |
| O4D | Octavius 4D |
| PI | Plan Irregularity |
| LS | Leaf Speed |
| AAV | Aperture Area Variability |
| LSV | Leaf Sequence Variability |
| MCS | Modulation Complexity Score |
| ALPO | Average Leaf Pair Opening |
| SAS | Small Aperture Score |
| EM | Edge Metrics |
| EFS | Equivalent Field Size |
Appendix A
| No. of Patients | No. of Lesions | Volume of Each Lesion (cc) |
|---|---|---|
| 1 | 3 | 2.73, 3.18, 0.76 |
| 2 | 5 | 0.1, 0.27, 0.36, 0.25, 0.05 |
| 3 | 2 | 0.26, 0.17 |
| 4 | 2 | 0.04, 0.02 |
| 5 | 10 | 0.6, 0.5, 6.4, 0.5, 0.4, 8.8, 0.4, 0.3, 0.7, 1 |
| 6 | 6 | 5.39, 0.05, 0.07, 2.37, 1.81, 5.28 |
| 7 | 3 | 3.41, 1.02, 3.24 |
| 8 | 4 | 0.03, 0.08, 0.06, 0.39 |
| 9 | 2 | 0.95, 0.1 |
| 10 | 3 | 5.15, 6.45, 0.1 |
| 11 | 2 | 1.45, 2.1 |
| 12 | 5 | 2.09, 4.73, 7.5, 1.05, 0.44 |
| 13 | 7 | 0.51, 0.48, 0.65, 0.33, 0.93, 0.26, 0.27 |
| 14 | 2 | 14.6, 8.5 |
| 15 | 5 | 1.51, 1.43, 0.09, 0.03, 0.05 |
| 16 | 2 | 4.6, 1.7 |
| 17 | 3 | 0.7, 2.18, 0.22 |
| 18 | 3 | 0.3, 1.2, 0.7 |
| 19 | 2 | 0.14, 0.12 |
| 20 | 2 | 4.8, 2.8 |
| 21 | 2 | 0.32, 0.62 |
| 22 | 3 | 8.08, 0.77, 0.51 |
| 23 | 4 | 2.37, 2.94, 4.48, 0.6 |
| 24 | 3 | 3.4, 2.5, 2.4 |
| 25 | 3 | 2.6, 14.1, 13.6 |
| 26 | 2 | 1.7, 0.3 |
| 27 | 2 | 0.13, 0.23 |
| 28 | 4 | 0.34, 0.06, 0.03, 0.04 |
| 29 | 3 | 4.06, 0.86, 0.55 |
| 30 | 3 | 2.85, 0.02, 0.02 |
| Category | Complexity Metrics | Formula |
|---|---|---|
| Deliverability | PI | |
| LS | ||
| AAV | ||
| LSV | ||
| MCS | ||
| ALPO | ||
| Accuracy | SAS | |
| EM | ||
| EFS |
References
- Lutz, W.; Winston, K.R.; Maleki, N. A system for stereotactic radiosurgery with a linear accelerator. Int. J. Radiat. Oncol. Biol. Phys. 1988, 14, 373–381. [Google Scholar] [CrossRef]
- Lobb, E.C.; Degnan, M. Comparison of VMAT complexity-reduction strategies for single-target cranial radiosurgery with the Eclipse treatment planning system. J. Appl. Clin. Med. Phys. 2020, 21, 97–108. [Google Scholar] [CrossRef]
- Ohira, S.; Ueda, Y.; Akino, Y.; Hashimoto, M.; Masaoka, A.; Hirata, T.; Miyazaki, M.; Koizumi, M.; Teshima, T. HyperArc VMAT planning for single and multiple brain metastases stereotactic radiosurgery: A new treatment planning approach. Radiat. Oncol. 2018, 13, 13. [Google Scholar] [CrossRef] [PubMed]
- Chambrelant, I.; Jarnet, D.; Bou-Gharios, J.; Le Fèvre, C.; Kuntz, L.; Antoni, D.; Jenny, C.; Noël, G. Stereotactic Radiation Therapy of Single Brain Metastases: A Literature Review of Dosimetric Studies. Cancers 2023, 15, 3937. [Google Scholar] [CrossRef]
- Kaal, E.C.A.; Niël, C.G.J.H.; Vecht, C.J. Therapeutic management of brain metastasis. Lancet Neurol. 2005, 4, 289–298. [Google Scholar] [CrossRef]
- Franchino, F.; Rudà, R.; Soffietti, R. Mechanisms and therapy for cancer metastasis to the brain. Front. Media 2018, 8, 161. [Google Scholar] [CrossRef]
- Gevaert, T.; Steenbeke, F.; Pellegri, L.; Engels, B.; Christian, N.; Hoornaert, M.T.; Verellen, D.; Mitine, C.; De Ridder, M. Evaluation of a dedicated brain metastases treatment planning optimization for radiosurgery: A new treatment paradigm? Radiat. Oncol. 2016, 11, 13. [Google Scholar] [CrossRef]
- Clark, G.M.; Popple, R.A.; Young, P.E.; Fiveash, J.B. Feasibility of Single-Isocenter Volumetric Modulated Arc Radiosurgery for Treatment of Multiple Brain Metastases. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, 296–302. [Google Scholar] [CrossRef] [PubMed]
- McNiven, A.L.; Sharpe, M.B.; Purdie, T.G. A new metric for assessing IMRT modulation complexity and plan deliverability. Med. Phys. 2010, 37, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Masi, L.; Doro, R.; Favuzza, V.; Cipressi, S.; Livi, L. Impact of plan parameters on the dosimetric accuracy of volumetric modulated arc therapy. Med. Phys. 2013, 40, 071718. [Google Scholar] [CrossRef]
- Younge, K.C.; Matuszak, M.M.; Moran, J.M.; McShan, D.L.; Fraass, B.A.; Roberts, D.A. Penalization of aperture complexity in inversely planned volumetric modulated arc therapy. Med. Phys. 2012, 39, 7160–7170. [Google Scholar] [CrossRef] [PubMed]
- Crowe, S.B.; Kairn, T.; Middlebrook, N.; Sutherland, B.; Hill, B.; Kenny, J.; Langton, C.M.; Trapp, J.V. Examination of the properties of IMRT and VMAT beams and evaluation against pre-treatment quality assurance results. Phys. Med. Biol. 2015, 60, 2587–2601. [Google Scholar] [CrossRef] [PubMed]
- Gaudreault, M.; Nguyen, P.H.; Lawford, C.; Franich, R.; Hardcastle, N. Implementations of modulation complexity scores and their correlations with treatment plan quality in stereotactic radiation therapy. Med. Phys. 2025, 52, e17961. [Google Scholar] [CrossRef]
- Xue, X.; Luan, S.; Ding, Y.; Li, X.; Li, D.; Wang, J.; Ma, C.; Jiang, M.; Wei, W.; Wang, X. Treatment plan complexity quantification for predicting gamma passing rates in patient-specific quality assurance for stereotactic volumetric modulated arc therapy. J. Appl. Clin. Med. Phys. 2024, 25, e14432. [Google Scholar] [CrossRef] [PubMed]
- Cavinato, S.; Amico, A.G.; Bettinelli, A.; Caricato, P.; Ceroni, P.; Khouzai, B.E.; Guida, F.; Paiusco, M.; Paronetto, C.; Pivato, N.; et al. Exploring plan quality: Using plan complexity to quantitatively analyse the tradeoff between clinical suitability and dosimetric accuracy. Phys. Medica 2025, 136, 105026. [Google Scholar] [CrossRef]
- Kondziolka, D.; Flickinger, J.C.; Lunsford, L.D. Radiosurgery for brain metastases. Prog. Neurol. Surg. 2012, 25, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Chin, K.; Robbins, J.R.; Kim, J.; Li, H.; Amro, H.; Chetty, I.J.; Gordon, J.; Ryu, S. Radiosurgery of multiple brain metastases with single-isocenter dynamic conformal arcs (SIDCA). Radiother. Oncol. 2014, 112, 128–132. [Google Scholar] [CrossRef]
- Vellayappan, B.A.; McGranahan, T.; Graber, J.; Taylor, L.; Venur, V.; Ellenbogen, R.; Sloan, A.E.; Redmond, K.J.; Foote, M.; Chao, S.T.; et al. Radiation Necrosis from Stereotactic Radiosurgery—How Do We Mitigate? Curr. Treat. Options Oncol. 2021, 22, 57. [Google Scholar] [CrossRef]
- Fogliata, A.; Thompson, S.; Stravato, A.; Tomatis, S.; Scorsetti, M.; Cozzi, L. On the gEUD biological optimization objective for organs at risk in Photon Optimizer of Eclipse treatment planning system. J. Appl. Clin. Med. Phys. 2018, 19, 106–114. [Google Scholar] [CrossRef]
- Pokhrel, D.; Palmiero, A.N.; Bernard, M.E.; Clair, W.S. Dynamic conformal arcs-based single-isocenter VMAT planning technique for radiosurgery of multiple brain metastases. Med. Dosim. 2021, 46, 195–200. [Google Scholar] [CrossRef]
- Craft, D.; Bortfeld, T. On the tradeoff between treatment time and plan quality in rotational arc radiation delivery. arXiv 2009. [Google Scholar] [CrossRef]
- Gloi, A.M. HyperArc multiple brain metastases report. Int. J. Res. Med. Sci. 2024, 12, 566–571. [Google Scholar] [CrossRef]
- Paddick, I.; Lippitz, B. A simple dose gradient measurement tool to complement the conformity index. J. Neurosurg. 2006, 105, 194–201. [Google Scholar] [CrossRef] [PubMed]
- McCulloch, J.; Pawlowski, J.; Kirby, N.; Rasmussen, K.; Shi, Z.; Myers, P.; Stathakis, S.; Papanikolaou, N.; Saenz, D.L. Patient-specific dose quality assurance of single-isocenter multiple brain metastasis stereotactic radiosurgery using PTW Octavius 4D. J. Appl. Clin. Med. Phys. 2020, 21, 107–115. [Google Scholar] [CrossRef]
- Cavinato, S.; Scaggion, A.; Paiusco, M. Technical note: A software tool to extract complexity metrics from radiotherapy treatment plans. Med. Phys. 2024, 51, 8602–8612. [Google Scholar] [CrossRef] [PubMed]
- Chiavassa, S.; Bessieres, I.; Edouard, M.; Mathot, M.; Moignier, A. Complexity metrics for IMRT and VMAT plans: A review of current literature and applications. Br. J. Radiol. 2019, 92, 20190270. [Google Scholar] [CrossRef]
- Stanley, J.; Breitman, K.; Dunscombe, P.; Spencer, D.P.; Lau, H. Evaluation of stereotactic radiosurgery conformity indices for 170 target volumes in patients with brain metastases. J. Appl. Clin. Med. Phys. 2011, 12, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Paddick, I. A simple scoring ratio to index the conformity of radiosurgical treatment plans. J. Neurosurg. 2000, 93, 219–222. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.J.; Ho, Y.W.; Lok, K.H.; Lu, Y.Y.; Zhu, C.R.; Cheng, H.C.Y. Dosimetric and radiobiological evaluation of stereotactic radiosurgery using volumetric modulated arc therapy and dynamic conformal arc therapy for multiple brain metastases. Sci. Rep. 2025, 15, 9118. [Google Scholar] [CrossRef]
- Hofmaier, J.; Bodensohn, R.; Garny, S.; Hadi, I.; Fleischmann, D.F.; Eder, M.; Dinc, Y.; Reiner, M.; Corradini, S.; Parodi, K.; et al. Single isocenter stereotactic radiosurgery for patients with multiple brain metastases: Dosimetric comparison of VMAT and a dedicated DCAT planning tool. Radiat. Oncol. 2019, 14, 103. [Google Scholar] [CrossRef]
- Chambrelant, I.; Jarnet, D.; Le Fèvre, C.; Kuntz, L.; Jacob, J.; Jenny, C.; Noël, G. Comparative study of dynamic conformal arc therapy and volumetric modulated arc therapy for treating single brain metastases: A retrospective analysis of dosimetric and clinical outcomes. Phys. Imaging Radiat. Oncol. 2024, 30, 100591. [Google Scholar] [CrossRef] [PubMed]
- Velten, C.; Kabarriti, R.; Garg, M.; Tomé, W.A. Single isocenter treatment planning techniques for stereotactic radiosurgery of multiple cranial metastases. Phys. Imaging Radiat. Oncol. 2021, 17, 47–52. [Google Scholar] [CrossRef]
- Bokrantz, R.; Wedenberg, M.; Sandwall, P. Dynamic conformal arcs for lung stereotactic body radiation therapy: A comparison with volumetric-modulated arc therapy. J. Appl. Clin. Med. Phys. 2020, 21, 103–109. [Google Scholar] [CrossRef]
- Saǧlam, Y. A novel weight optimized dynamic conformal arcs with TrueBeam. Linac for very small tumors (≤1 cc) with single isocenter of multiple brain metastases (2≤, ≥4) in stereotactic radiosurgery: A comparison with volumetric modulated arc therapy. J. Cancer Res. Ther. 2023, 19, 1297–1304. [Google Scholar] [CrossRef] [PubMed]
- Lan, M.; Wu, R.; Deng, G.; Yang, B.; Zhuang, Y.; Yi, W.; Xu, W.; Sun, J. Dosimetric comparison and evaluation of different convergence modes in nasopharyngeal carcinoma using VMAT treatment deliveries. PeerJ 2024, 12, e18773. [Google Scholar] [CrossRef] [PubMed]




| No. of Lesions | No. of Patients |
|---|---|
| 2 | 11 |
| 3 | 10 |
| 4 | 3 |
| 5 | 3 |
| 6 | 1 |
| 7 | 1 |
| 8 | - |
| 9 | - |
| 10 | 1 |
| Parameters | HyperArc | OptDCA | p-Value |
|---|---|---|---|
| PCI | 0.72 (0.41–0.9) | 0.71 (0.15–0.91) | 0.011 |
| GI | 4.09 (2.52–89.53) | 3.88 (1.4–57.67) | 0.181 |
| Target Coverage (%) | 98.09 (93.3–104.8) | 98.17 (87–108.23) | 0.357 |
| MUs | 5473 (1883.5–9497.7) | 4735 (1408.8–9562.6) | 0.007 |
| V12(cc) | 6.95 (3.18–17.4) | 9.33 (3.64–25.26) | <0.001 |
| V20(cc) | 2.56 (0.85–6.01) | 2.73 (0.88–8.1) | 0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Stroubinis, T.; Giannopoulou, M.; Stasinou, D.; Psarras, M.; Zygogianni, A.; Protopapa, M.; Kouloulias, V.; Platoni, K. Volumetric Modulated Arc Therapy Versus Dynamic Conformal Arc Therapy for Single Isocenter Stereotactic Radiotherapy of Multiple Brain Metastases. Bioengineering 2026, 13, 207. https://doi.org/10.3390/bioengineering13020207
Stroubinis T, Giannopoulou M, Stasinou D, Psarras M, Zygogianni A, Protopapa M, Kouloulias V, Platoni K. Volumetric Modulated Arc Therapy Versus Dynamic Conformal Arc Therapy for Single Isocenter Stereotactic Radiotherapy of Multiple Brain Metastases. Bioengineering. 2026; 13(2):207. https://doi.org/10.3390/bioengineering13020207
Chicago/Turabian StyleStroubinis, Theodoros, Maria Giannopoulou, Despoina Stasinou, Michalis Psarras, Anna Zygogianni, Maria Protopapa, Vassilis Kouloulias, and Kalliopi Platoni. 2026. "Volumetric Modulated Arc Therapy Versus Dynamic Conformal Arc Therapy for Single Isocenter Stereotactic Radiotherapy of Multiple Brain Metastases" Bioengineering 13, no. 2: 207. https://doi.org/10.3390/bioengineering13020207
APA StyleStroubinis, T., Giannopoulou, M., Stasinou, D., Psarras, M., Zygogianni, A., Protopapa, M., Kouloulias, V., & Platoni, K. (2026). Volumetric Modulated Arc Therapy Versus Dynamic Conformal Arc Therapy for Single Isocenter Stereotactic Radiotherapy of Multiple Brain Metastases. Bioengineering, 13(2), 207. https://doi.org/10.3390/bioengineering13020207








