Histopathological Evaluation of Bioactive Glass Wound Sites in a Swine Model
Abstract
1. Introduction
2. Materials and Methods
Histomorphometric Analysis
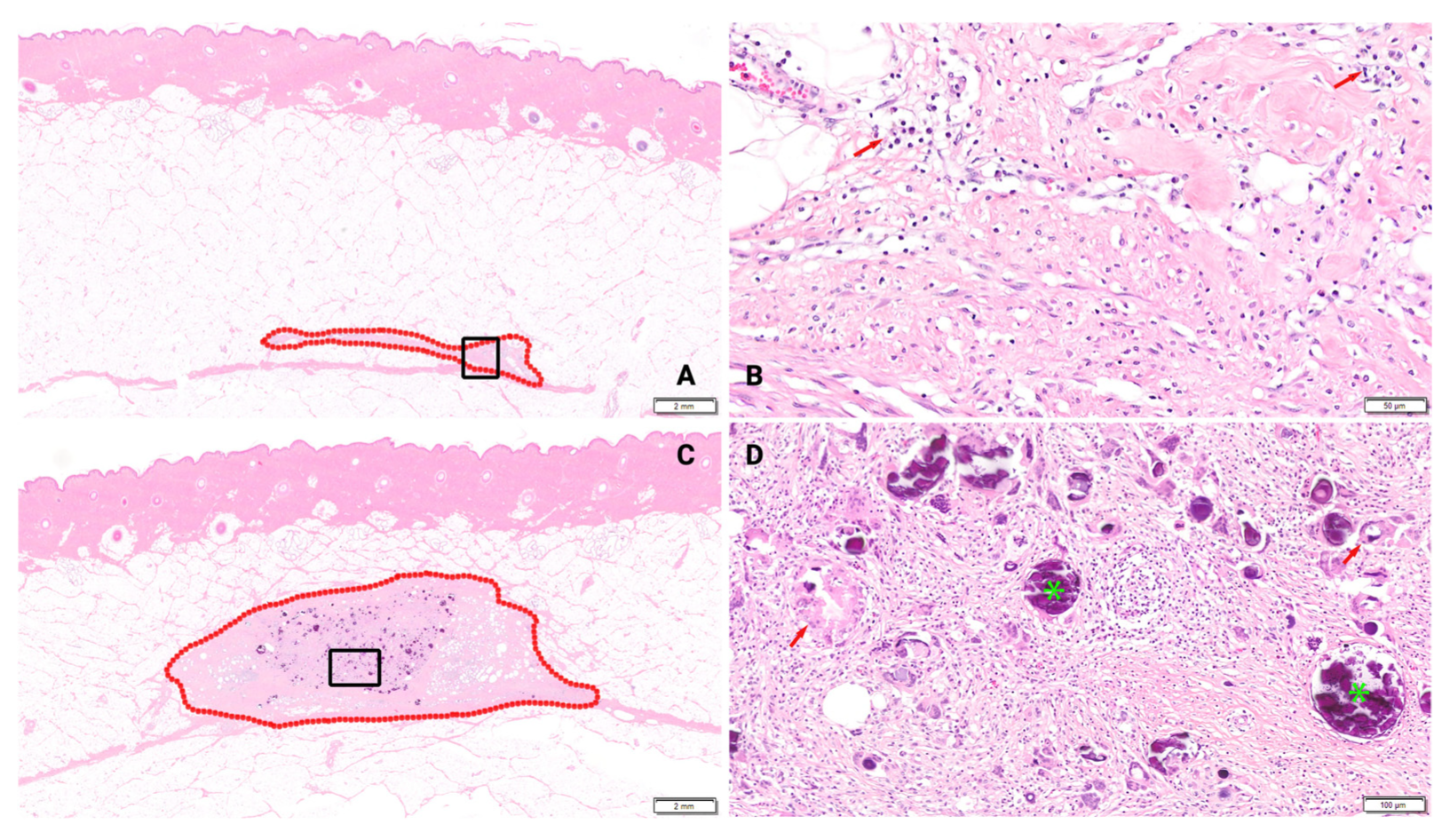
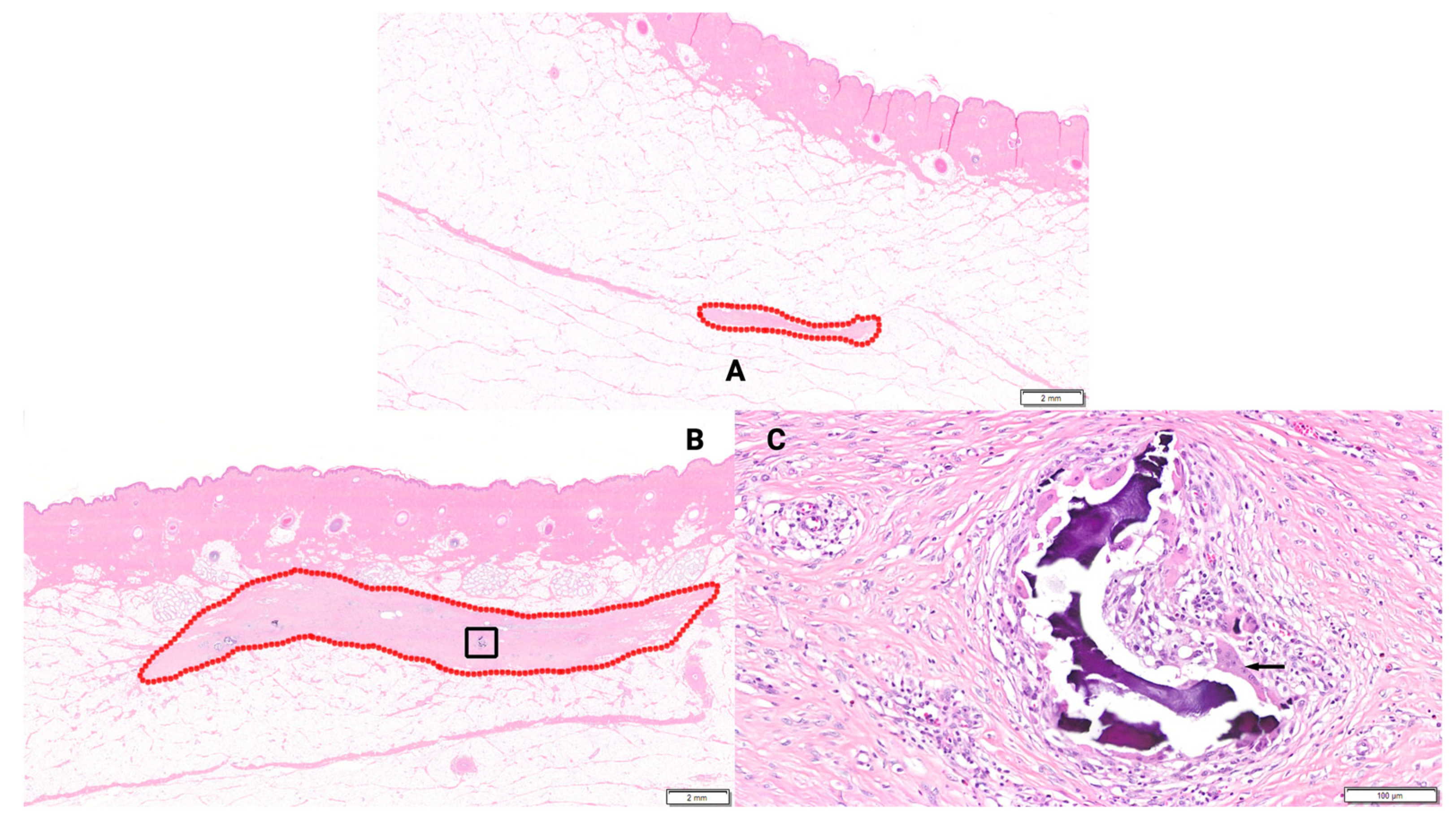
3. Results
3.1. Inflammation
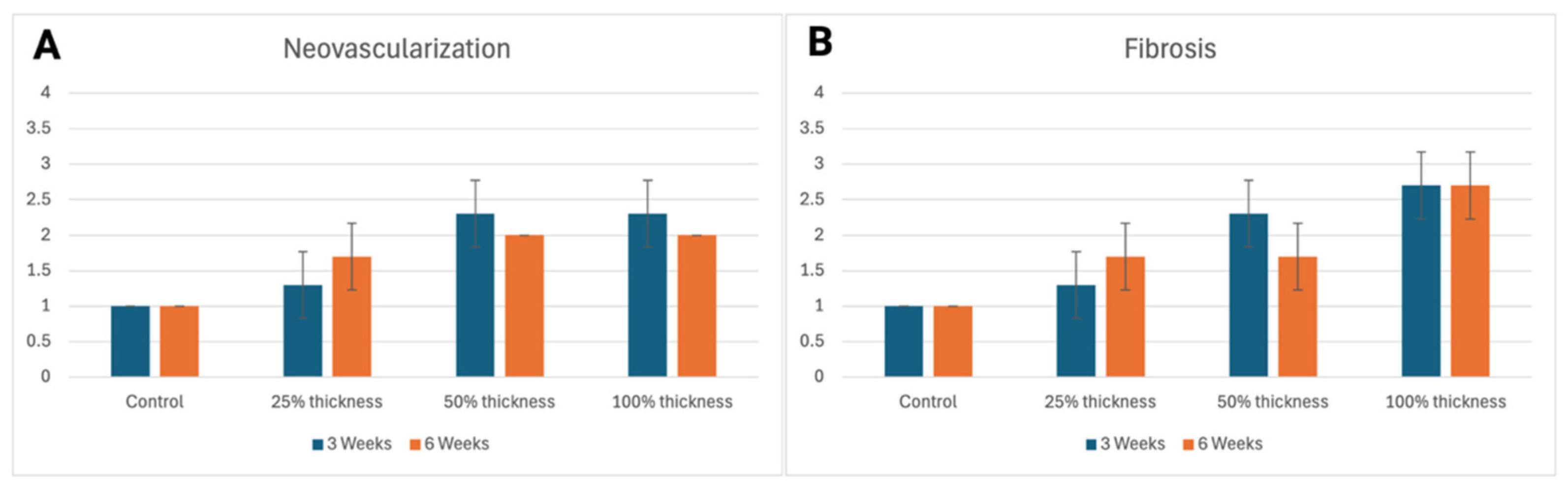
3.2. Neovascularization/Fibrosis
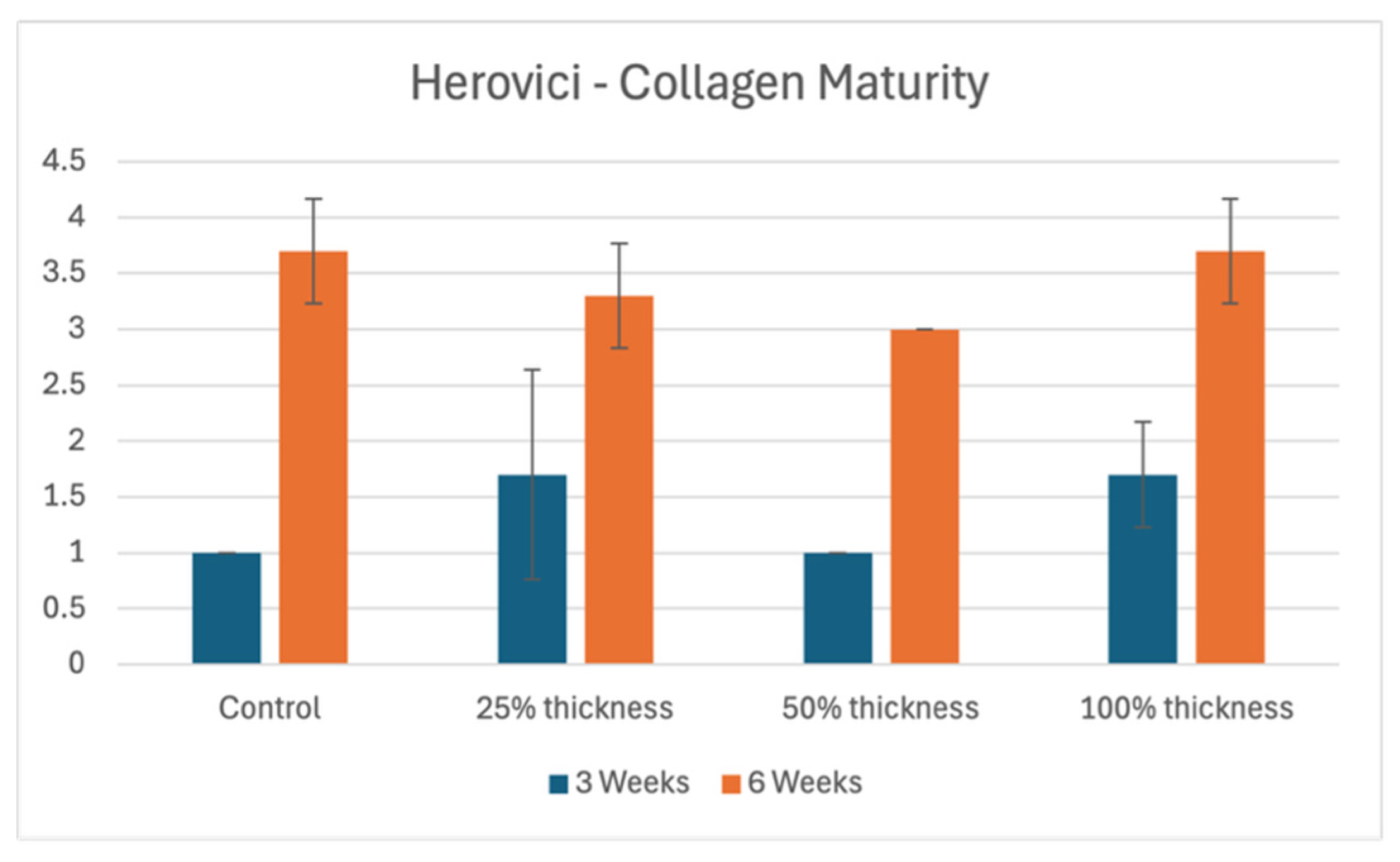
3.3. Herovici Stains
3.4. Immunohistochemistry Evaluation—Semi-Quantitative for CD31
3.5. Histomorphometry Evaluation
4. Discussion
4.1. Inflammation
4.2. Neovascularization
4.3. Collagen Deposition and Maturity
4.4. Scar Thickness and Vascular Density
4.5. Biodegradation and Material Persistence
4.6. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DFU | Diabetic Foot Ulcer |
| VLU | Venous Leg Ulcer |
| BBGFM | Borate-Based Bioactive Glass Fiber Matrix |
| MMP | Matrix Metalloproteinase |
| IL | Interleukin |
| TNF | Tissue Necrosis Factor |
| VEGF | Vascular Endothelial Growth Factor |
| FDA | Food and Drug Administration |
| SOP | Standard Operating Procedure |
| NBF | Neutral Buffered Formalin |
| H&E | Hematoxylin and Eosin |
| IHC | Immunohistochemistry |
References
- Armstrong, D.G.; Boulton, A.J.M.; Bus, S.A. Diabetic foot ulcers and their recurrence. N. Engl. J. Med. 2017, 376, 2367–2375. [Google Scholar] [CrossRef]
- Frykberg, R.G.; Banks, J. Challenges in the treatment of chronic wounds. Adv. Wound Care 2015, 4, 560–582. [Google Scholar] [CrossRef] [PubMed]
- Jeffcoate, W.J.; Harding, K.G. Diabetic foot ulcers. Lancet 2003, 361, 1545–1551. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.K.; Gordillo, G.M.; Roy, S.; Kirsner, R.; Lambert, L.; Hunt, T.K.; Gottrup, F.; Gurtner, G.C.; Longaker, M.T. Human skin wounds: A major and snowballing threat to public health and the economy. Wound Repair. Regen. 2009, 17, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Nussbaum, S.R.; Carter, M.J.; Fife, C.E.; DaVanzo, J.; Haught, R.; Nusgart, M.; Cartwright, D. An economic evaluation of the impact, cost, and Medicare policy implications of chronic nonhealing wounds. Value Health 2018, 21, 27–32. [Google Scholar] [CrossRef]
- Harding, K.G.; Morris, H.L.; Patel, G.K. Science, medicine and the future: Healing chronic wounds. BMJ 2002, 324, 160–163. [Google Scholar] [CrossRef]
- Almadani, Y.H.; Vorstenbosch, J.; Davison, P.G.; Murphy, A.M. Wound Healing: A Comprehensive Review. Semin. Plast. Surg. 2021, 35, 141–144. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rodrigues, M.; Kosaric, N.; Bonham, C.A.; Gurtner, G.C. Wound Healing: A Cellular Perspective. Physiol. Rev. 2019, 99, 665–706. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: A critical step during wound healing. Cell Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cialdai, F.; Risaliti, C.; Monici, M. Role of fibroblasts in wound healing and tissue remodeling on Earth and in space. Front. Bioeng. Biotechnol. 2022, 10, 958381. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mishra, A.; Kushare, A.; Gupta, M.N.; Ambre, P. Advanced Dressings for Chronic Wound Management. ACS Appl. Bio Mater. 2024, 7, 2660–2676. [Google Scholar] [CrossRef] [PubMed]
- Nur, M.G.; Rahman, M.; Dip, T.M.; Hossain, M.H.; Hossain, N.B.; Baratchi, S.; Padhye, R.; Houshyar, S. Recent advances in bioactive wound dressings. Wound Repair Regen. 2025, 33, e13233. [Google Scholar] [CrossRef] [PubMed]
- Aijaz, A.; Elloso, M.; Chen, Y.; Chogan, F.; Challagundla, B.; Rix, G.; Hota, S.; Matveev, A.; Jeschke, M.G. A bio-instructive, bioactive in situ polymerizable wound matrix promotes scar-free burn wound repair. iScience 2025, 28, 112471. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Thirumalaivasan, N.; Kanagaraj, K.; Nangan, S.; Pothu, R.; Rajendra, S.P.; Karuppiah, P.; Boddula, R. Bioactive hydrogels (Bio-HyGs): Emerging trends in drug delivery and wound healing applications. Polym. Adv. Technol. 2025, 36, e70132. [Google Scholar] [CrossRef]
- Lamberger, Z.; Lang, G. Engineering Extracellular Microenvironments: The Impact of Fibrous Materials on Cell Behavior. Adv. Healthc. Mater. 2025, 14, e01942. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Joyce, K.; Fabra, G.T.; Bozkurt, Y.; Pandit, A. Bioactive potential of natural biomaterials: Identification, retention and assessment of biological properties. Signal Transduct. Target. Ther. 2021, 6, 122. [Google Scholar] [CrossRef]
- Han, G.; Ceilley, R. Chronic wound healing: A review of current management and treatments. Adv. Ther. 2017, 343, 599–610. [Google Scholar] [CrossRef]
- Homaeigohar, S.; Li, M.; Boccaccini, A.R. Bioactive glass-based fibrous wound dressings. Burn. Trauma 2022, 10, tkac038. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhou, J.; Wang, H.; Zhao, S.; Zhou, N.; Li, L.; Huang, W.; Wang, D.; Zhang, C. In vivo and in vitro studies of borate based glass micro-fibers for dermal repairing. Mater. Sci. Eng. C 2016, 60, 437–445. [Google Scholar] [CrossRef]
- Ege, D.; Zheng, K.; Boccaccini, A.R. Borate Bioactive Glasses (BBG): Bone Regeneration, Wound Healing Applications, and Future Directions. ACS Appl. Bio Mater. 2022, 5, 3608–3622. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jung, S.; Day, T.; Boone, T.; Buziak, B.; Omar, A. Anti-biofilm activity of two novel, borate based, bioactive glass wound dressings. Biomed. Glas. 2019, 5, 67–75. [Google Scholar] [CrossRef]
- Jung, S.; Schultz, G.; Mafiz, A.; Bevels, E.; Jaskula, K.; Brownell, K.; Lantz, E.; Strickland, A. Antimicrobial effects of a borate-based bioactive glass wound matrix on wound-relevant pathogens. J. Wound Care 2023, 32, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Gharbi, A.; Oudadesse, H.; El Feki, H.; Cheikhrouhou-Koubaa, W.; Chatzistavrou, X.; VRau, J.; Heinämäki, J.; Antoniac, I.; Ashammakhi, N.; Derbel, N. High Boron Content Enhances Bioactive Glass Biodegradation. J. Funct. Biomater. 2023, 14, 364. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zelenetskii, A.N.; Uspenskii, S.; Zaboronok, A.; Cherkaev, G.; Shchegolihin, A.; Mathis, B.J.; Selyanin, M.; Yamamoto, T.; Matsumura, A. Polycomplexes of Hyaluronic Acid and Borates in a Solid State and Solution: Synthesis, Characterization and Perspectives of Application in Boron Neutron Capture Therapy. Polymers 2018, 10, 181. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Armstrong, D.G.; Orgill, D.P.; Galiano, R.D.; Lantis, J.; Glat, P.M.; Gitterle, M.; Carter, M.J.; Young, N.; Zelen, C.M. A Borate-Based Bioactive Glass Advances Wound Healing in Non-Healing Wagner Grade 1 Diabetic Foot Ulcers: A Randomised Controlled Clinical Trial. Int. Wound J. 2025, 22, e70763. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- US Food and Drug Administration. Classification of Absorbable Synthetic Wound Dressings: Executive Summary—Meeting of the General and Plastic Surgery Devices Panel of the Medical Devices Advisory Committee; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2022. Available online: https://www.fda.gov/media/162545/download (accessed on 12 December 2025).
- Schilrreff, P.; Alexiev, U. Chronic Inflammation in Non-Healing Skin Wounds and Promising Natural Bioactive Compounds Treatment. Int. J. Mol. Sci. 2022, 23, 4928. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Riaz, M.; Iqbal, M.Z.; Klar, A.S.; Biedermann, T. Immunomodulatory Mechanisms of Chronic Wound Healing: Translational and Clinical Relevance. MedComm 2025, 6, e70378. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Moreira, H.R.; Marques, A.P. Vascularization in skin wound healing: Where do we stand and where do we go? Curr. Opin. Biotechnol. 2022, 73, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Brem, H.; Tomic-Canic, M. Cellular molecular basis of wound healing in diabetes. J. Clin. Investig. 2007, 117, 1219–1222. [Google Scholar] [CrossRef]
- Mathew-Steiner, S.S.; Roy, S.; Sen, C.K. Collagen in Wound Healing. Bioengineering 2021, 8, 63. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Galiano, R.D.; Tepper, O.M.; Pelo, C.R.; Bhatt, K.A.; Callaghan, M.; Bastidas, N.; Bunting, S.; Steinmetz, H.G.; Gurtner, G.C. Topical vascular endothelial growth factor accelerates diabetic wound healing through increased angiogenesis and by mobilizing and recruiting bone marrow-derived cells. Am. J. Pathol. 2004, 164, 1935–1947. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Office of Device Evaluation Center for Devices Radiological Health, U.S. Food and Drug Administration. Summary of Safety and Effectiveness Data: P900033/S008—Integra Dermal Regeneration Template; FDA: Rockville, MD, USA, 2002.
- Shi, L.; Ronfard, V. Biochemical and biomechanical characterization of porcine small intestinal submucosa (SIS): A mini review. Int. J. Burn. Trauma 2013, 3, 173–179. [Google Scholar] [PubMed] [PubMed Central]
- Gierek, M.; Łabuś, W.; Kitala, D.; Lorek, A.; Ochała-Gierek, G.; Zagórska, K.M.; Waniczek, D.; Szyluk, K.; Niemiec, P. Human Acellular Dermal Matrix in Reconstructive Surgery-A Review. Biomedicines 2022, 10, 2870. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rajaram, R.; Zhang, M.; Premaratne, G.; Ng, S. Novosorb® BTM-history, production and application in challenging wounds. Front. Bioeng. Biotechnol. 2024, 12, 1450973. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ortega-Gómez, A.; Perretti, M.; Soehnlein, O. Resolution of inflammation: An integrated view. EMBO Mol. Med. 2013, 5, 661–674. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]




| (A) Extent of Collagen Deposition/Maturity | Score |
| No collagen deposition | 0 |
| Majority of wound composed of immature collagen fibers (~1% to <50%) | 1 |
| Approximately equal amounts of immature and mature collagen fibers within wound | 2 |
| The majority of wound composed of mature collagen fibers (~51% to <95%) | 3 |
| Essentially all of wound composed of mature collagen fibers (~>95%) | 4 |
| (B) Neutrophils, Eosinophils, Lymphocytes, Plasma Cells, Macrophages | Score |
| Absent | 0 |
| Rare, 1–5/hpf | 1 |
| 6–10/hpf | 2 |
| Moderate, heavy infiltrates | 3 |
| Packed | 4 |
| (C) Multinucleated Giant Cells | Score |
| Absent | 0 |
| Rare, 1–2/hpf | 1 |
| 3–5/hpf | 2 |
| Moderate, heavy infiltrates | 3 |
| Sheets | 4 |
| (D) General Scoring | Score |
| Absent | 0 |
| Minimal | 1 |
| Mild | 2 |
| Moderate | 3 |
| Marked/Severe | 4 |
| (E) Amount | Score |
| Negative, no reactivity | 0 |
| Very rare, estimated as less than 5% | 1 |
| Rare, estimated as 5–25% | 2 |
| Occasional, estimated as 26–50% | 3 |
| Frequent, estimated as 51–75% | 4 |
| Very frequent, estimated as 76–100% | 5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Rabin, D.A.; Chaudhry, A.S.; Adam, T.H.; Kozlowski, K.; Lopez, M.P.; Kim, T.; Green, S.; Galiano, R.D.; Manista, G.C.; Buck, D.W., II; et al. Histopathological Evaluation of Bioactive Glass Wound Sites in a Swine Model. Bioengineering 2026, 13, 200. https://doi.org/10.3390/bioengineering13020200
Rabin DA, Chaudhry AS, Adam TH, Kozlowski K, Lopez MP, Kim T, Green S, Galiano RD, Manista GC, Buck DW II, et al. Histopathological Evaluation of Bioactive Glass Wound Sites in a Swine Model. Bioengineering. 2026; 13(2):200. https://doi.org/10.3390/bioengineering13020200
Chicago/Turabian StyleRabin, Daniel A., Aneeq S. Chaudhry, Tarifa H. Adam, Katherine Kozlowski, Marlynn P. Lopez, Tiffany Kim, Spencer Green, Robert D. Galiano, Gregory C. Manista, Donald W. Buck, II, and et al. 2026. "Histopathological Evaluation of Bioactive Glass Wound Sites in a Swine Model" Bioengineering 13, no. 2: 200. https://doi.org/10.3390/bioengineering13020200
APA StyleRabin, D. A., Chaudhry, A. S., Adam, T. H., Kozlowski, K., Lopez, M. P., Kim, T., Green, S., Galiano, R. D., Manista, G. C., Buck, D. W., II, & Jung, S. (2026). Histopathological Evaluation of Bioactive Glass Wound Sites in a Swine Model. Bioengineering, 13(2), 200. https://doi.org/10.3390/bioengineering13020200






