Artificial Intelligence-Based Proximal Bone Shape Asymmetry Analysis and Clinical Correlation with Cartilage Relaxation Times and Functional Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Image Acquisition
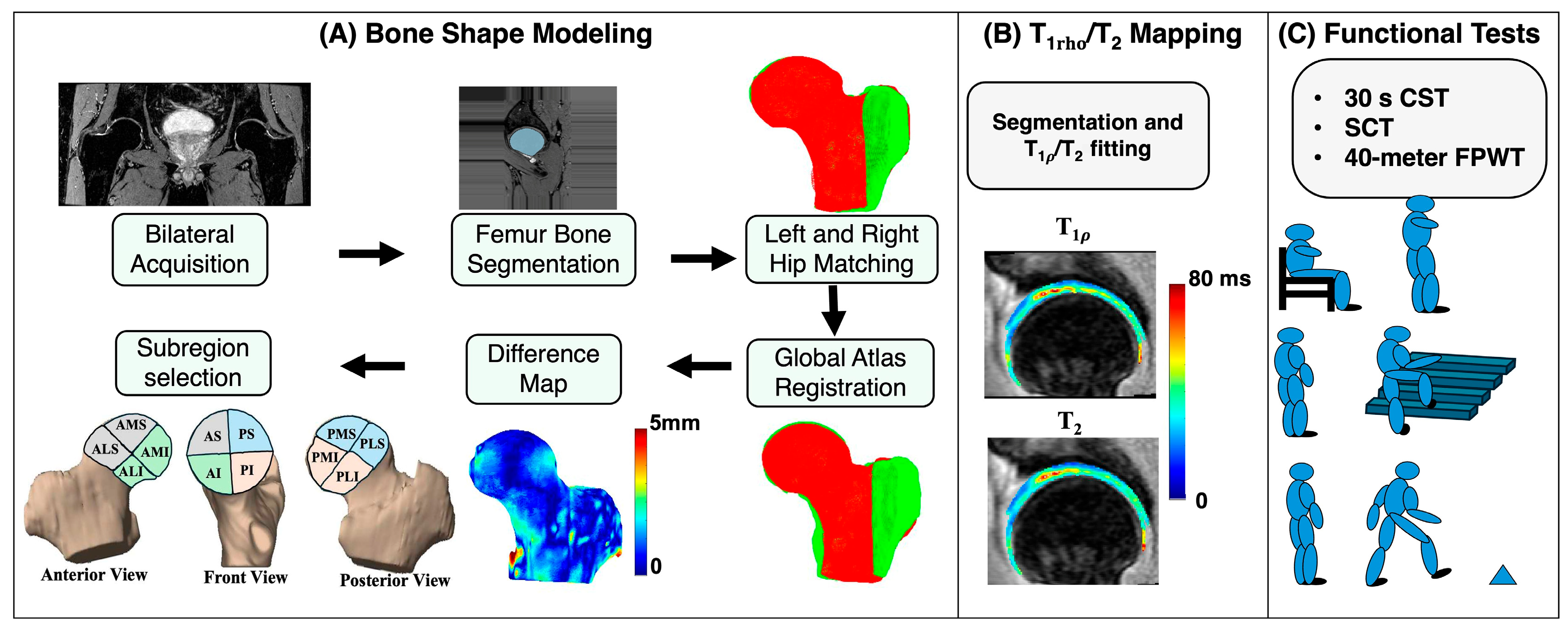
2.3. Postprocessing of MRI Data
2.3.1. Left and Right Hip Image Splitting
2.3.2. Image Preprocessing
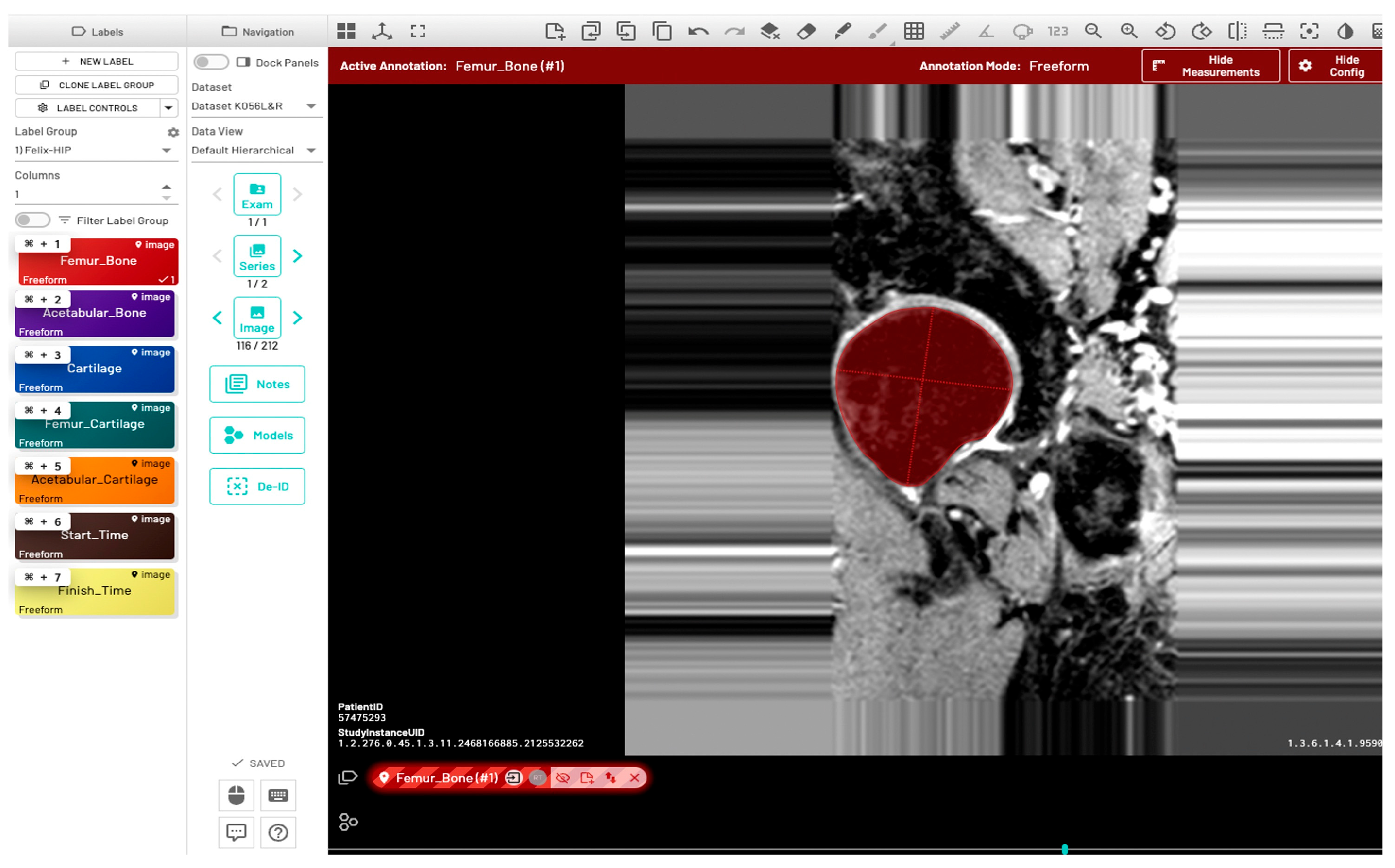
2.3.3. Bone Segmentation: Deep Learning-Based Approach
2.3.4. Bone Shape Analysis
2.3.5. Cartilage and Quantification
2.4. Functional Activity Test
2.5. Statistical Analysis
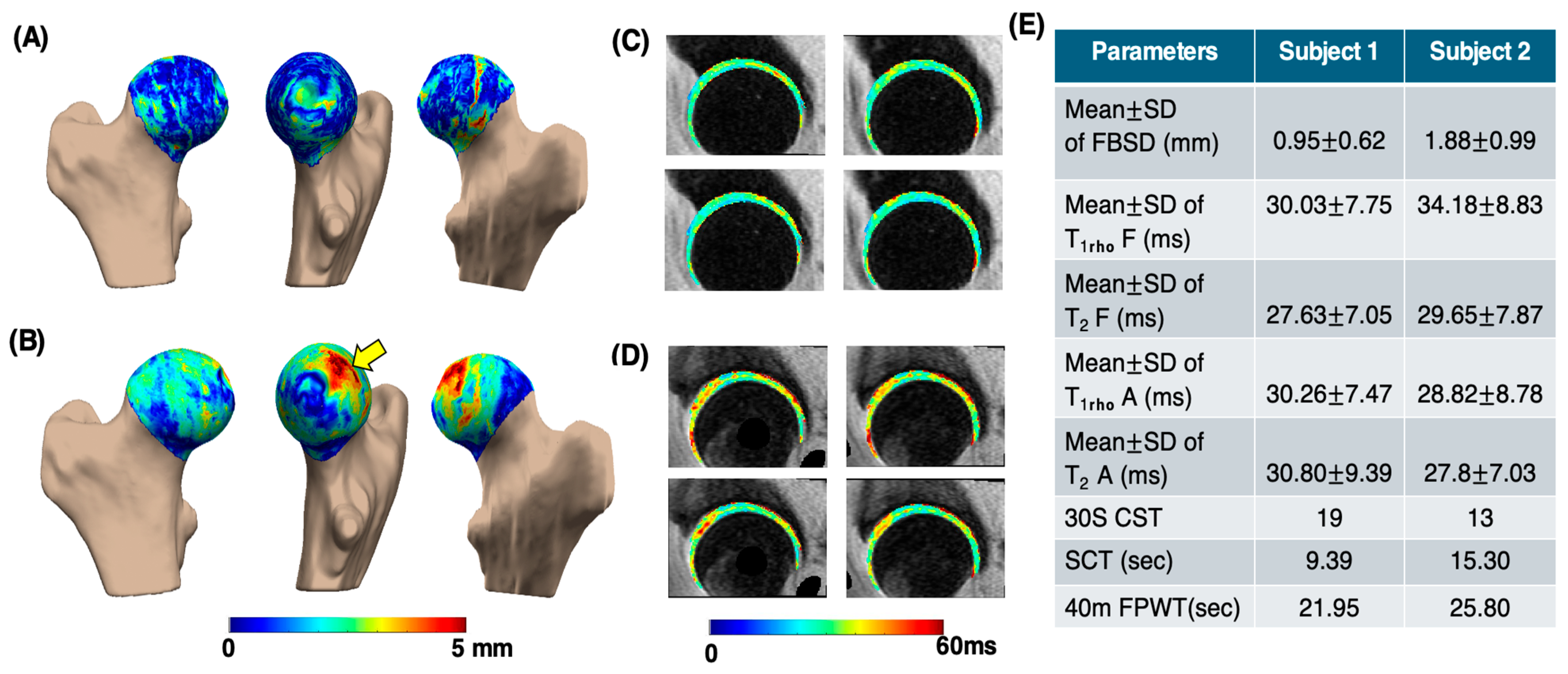
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lespasio, M.J.; Sultan, A.A.; Piuzzi, N.S.; Khlopas, A.; Husni, M.E.; Muschler, G.F.; Mont, M.A. Hip Osteoarthritis: A Primer. Perm. J. 2018, 22, 17–084. [Google Scholar] [CrossRef]
- Bennell, K. Physiotherapy management of hip osteoarthritis. J. Physiother. 2013, 59, 145–157. [Google Scholar] [CrossRef]
- Shi, X.; Mai, Y.; Fang, X.; Wang, Z.; Xue, S.; Chen, H.; Dang, Q.; Wang, X.; Tang, S.; Ding, C.; et al. Bone marrow lesions in osteoarthritis: From basic science to clinical implications. Bone Rep. 2023, 18, 101667. [Google Scholar] [CrossRef]
- Neogi, T. Clinical significance of bone changes in osteoarthritis. Ther. Adv. Musculoskelet. Dis. 2012, 4, 259–267. [Google Scholar] [CrossRef]
- Pedoia, V.; Samaan, M.A.; Inamdar, G.; Gallo, M.C.; Souza, R.B.; Majumdar, S. Study of the interactions between proximal femur 3d bone shape, cartilage health, and biomechanics in patients with hip Osteoarthritis. J. Orthop. Res. 2018, 36, 330–341. [Google Scholar] [CrossRef]
- Dudaric, L.; Dumic-Cule, I.; Divjak, E.; Cengic, T.; Brkljacic, B.; Ivanac, G. Bone Remodeling in Osteoarthritis—Biological and Radiological Aspects. Medicina 2023, 59, 1613. [Google Scholar] [CrossRef] [PubMed]
- Vissers, M.M.; Bussmann, J.B.J.; de Groot, I.B.; Verhaar, J.; Reijman, M. Walking and chair rising performed in the daily life situation before and after total hip arthroplasty. Osteoarthr. Cartil. 2011, 19, 1102–1107. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Ramirez, A.; Weenk, D.; Lecumberri, P.; Verdonschot, N.; Pakvis, D.; Veltink, P.H. Preoperative Ambulatory Measurement of Asymmetric Leg Loading During Sit-to-Stand in Hip Arthroplasty Patients. IEEE Trans. Neural Syst. Rehabil. Eng. 2014, 22, 585–592. [Google Scholar] [CrossRef]
- Eitzen, I.; Fernandes, L.; Nordsletten, L.; Snyder-Mackler, L.; Risberg, M.A. Weight-bearing asymmetries during Sit-to-Stand in patients with mild-to-moderate hip osteoarthritis. Gait Posture 2014, 39, 683–688. [Google Scholar] [CrossRef] [PubMed]
- Memiş, A.; Varlı, S.; Bilgili, F. A novel approach for computerized quantitative image analysis of proximal femur bone shape deformities based on the hip joint symmetry. Artif. Intell. Med. 2021, 115, 102057. [Google Scholar] [CrossRef]
- Harris, M.D.; Datar, M.; Whitaker, R.T.; Jurrus, E.R.; Peters, C.L.; Anderson, A.E. Statistical shape modeling of cam femoroacetabular impingement. J. Orthop. Res. 2013, 31, 1620–1626. [Google Scholar] [CrossRef] [PubMed]
- Ronneberger, O.; Fischer, P.; Brox, T. U-Net: Convolutional Networks for Biomedical Image Segmentation. arXiv 2015, arXiv:1505.04597. [Google Scholar] [CrossRef]
- Hess, M.; Allaire, B.; Gao, K.T.; Tibrewala, R.; Inamdar, G.; Bharadwaj, U.; Chin, C.; Pedoia, V.; Bouxsein, M.; Anderson, D.; et al. Deep Learning for Multi-Tissue Segmentation and Fully Automatic Personalized Biomechanical Models from BACPAC Clinical Lumbar Spine MRI. Pain Med. 2023, 24, S139–S148. [Google Scholar] [CrossRef] [PubMed]
- Deniz, C.M.; Xiang, S.; Hallyburton, R.S.; Welbeck, A.; Babb, J.S.; Honig, S.; Cho, K.; Chang, G. Segmentation of the Proximal Femur from MR Images using Deep Convolutional Neural Networks. Sci. Rep. 2018, 8, 16485. [Google Scholar] [CrossRef]
- Bonaldi, L.; Pretto, A.; Pirri, C.; Uccheddu, F.; Fontanella, C.G.; Stecco, C. Deep Learning-Based Medical Images Segmentation of Musculoskeletal Anatomical Structures: A Survey of Bottlenecks and Strategies. Bioengineering 2023, 10, 137. [Google Scholar] [CrossRef]
- Imani, M.; Buratto, J.; Dao, T.; Meijering, E.; Vogrin, S.; Kwok, T.C.Y.; Orwoll, E.S.; Cawthon, P.M.; Duque, G. Deep Learning Technique for Automatic Segmentation of Proximal Hip Musculoskeletal Tissues from CT Scan Images: A MrOS Study. J. Cachexia Sarcopenia Muscle 2025, 16, e13728. [Google Scholar] [CrossRef]
- Nelson, A.E.; Liu, F.; Lynch, J.A. Association of Incident Symptomatic Hip Osteoarthritis with Differences in Hip Shape by Active Shape Modeling: The Johnston County Osteoarthritis Project. Arthritis Care Res. 2014, 66, 74–81. [Google Scholar] [CrossRef]
- Wise, B.L.; Kritikos, L.; Lynch, J.A.; Liu, F.; Parimi, N.; Tileston, K.; Nevitt, M.; Lane, N. Proximal femur shape differs between subjects with lateral and medial knee osteoarthritis and controls: The Osteoarthritis Initiative. Osteoarthr. Cartil. 2014, 22, 2067–2073. [Google Scholar] [CrossRef][Green Version]
- Doherty, M.; Courtney, P.; Doherty, S.; Jenkins, W.; Maciewicz, R.A.; Muir, K.; Zhang, W. Nonspherical femoral head shape (pistol grip deformity), neck shaft angle, and risk of hip osteoarthritis: A case–control study. Arthritis Rheum. 2008, 58, 3172–3182. [Google Scholar] [CrossRef]
- Keating, T.C.; Leong, N.; Beck, E.C.; Nwachukwu, B.U.; Orías, A.A.E.; Qian, X.; Li, K.; Nho, S.J. Evaluation of Statistical Shape Modeling in Quantifying Femoral Morphologic Differences Between Symptomatic and Nonsymptomatic Hips in Patients with Unilateral Femoroacetabular Impingement Syndrome. Arthrosc. Sports Med. Rehabil. 2020, 2, e91–e95. [Google Scholar] [CrossRef]
- Fischer, M.; Walter, S.S.; Hepp, T.; Zimmer, M.; Notohamiprodjo, M.; Schick, F.; Yang, B. Automated Morphometric Analysis of the Hip Joint on MRI from the German National Cohort Study. Radiol. Artif. Intell. 2021, 3, e200213. [Google Scholar] [CrossRef]
- Neogi, T.; Bowes, M.A.; Niu, J.; De Souza, K.M.; Vincent, G.R.; Goggins, J.; Zhang, Y.; Felson, D.T. Magnetic Resonance Imaging–Based Three-Dimensional Bone Shape of the Knee Predicts Onset of Knee Osteoarthritis: Data from the Osteoarthritis Initiative. Arthritis Rheum. 2013, 65, 2048–2058. [Google Scholar] [CrossRef]
- Inamdar, G.; Pedoia, V.; Rossi-Devries, J.; Samaan, M.A.; Link, T.M.; Souza, R.B.; Majumdar, S. MR study of longitudinal variations in proximal femur 3D morphological shape and associations with cartilage health in hip osteoarthritis. J. Orthop. Res. 2019, 37, 161–170. [Google Scholar] [CrossRef]
- Siebelt, M.; Agricola, R.; Weinans, H.; Kim, Y. The role of imaging in early hip OA. Osteoarthr. Cartil. 2014, 22, 1470–1480. [Google Scholar] [CrossRef] [PubMed]
- Guermazi, A.; Alizai, H.; Crema, M.D.; Trattnig, S.; Regatte, R.; Roemer, F. Compositional MRI techniques for evaluation of cartilage degeneration in osteoarthritis. Osteoarthr. Cartil. 2015, 23, 1639–1653. [Google Scholar] [CrossRef]
- Matzat, S.J.; Tiel, J.; van Gold, G.E.; Oei, E.H.G. Quantitative MRI techniques of cartilage composition. Quant. Imaging Med. Surg. 2013, 3, 162–174. [Google Scholar]
- Adusumilli, G.; Joseph, S.E.; Samaan, M.A. iPhone Sensors in Tracking Outcome Variables of the 30-Second Chair Stand Test and Stair Climb Test to Evaluate Disability: Cross-Sectional Pilot Study. JMIR mHealth uHealth 2017, 5, e166. [Google Scholar] [CrossRef] [PubMed]
- Dobson, F.; Bennell, K.L.; Hinman, R.S.; Abbott, J.H.; Roos, E.M. OARSI recommended performance-based tests to assess physical function in people diagnosed with hip or knee osteoarthritis. Osteoarthr. Cartil. 2013, 21, 1042–1052. [Google Scholar] [CrossRef]
- Liao, T.-C.; Samaan, M.A.; Popovic, T.; Neumann, J.; Zhang, A.L.; Link, T.M.; Majumdar, S.; Souza, R.B. Abnormal Joint Loading During Gait in Persons with Hip Osteoarthritis Is Associated with Symptoms and Cartilage Lesions. J. Orthop. Sports Phys. Ther. 2019, 49, 917–924. [Google Scholar] [CrossRef] [PubMed]
- Zunair, H.; Hamza, A.B. Sharp U-Net: Depthwise convolutional network for biomedical image segmentation. Comput. Biol. Med. 2021, 136, 104699. [Google Scholar] [CrossRef]
- Tolpadi, A.A.; Bharadwaj, U.; Gao, K.T.; Bhattacharjee, R.; Gassert, F.G.; Luitjens, J.; Giesler, P.; Morshuis, J.N.; Fischer, P.; Hein, M.; et al. K2S Challenge: From Undersampled K-Space to Automatic Segmentation. Bioengineering 2023, 10, 267. [Google Scholar] [CrossRef]
- Lombaert, H.; Grady, L.; Polimeni, J.R.; Cheriet, F. Focusr: Feature Oriented Correspondence Using Spectral Regularization--A Method for Precise Surface Matching. IEEE Trans. Pattern Anal. Mach. Intell. 2013, 35, 2143–2160. [Google Scholar] [CrossRef]
- Mokrovic, H.; Komen, S.; Gulan, L.; Gulan, G. Radiographic analysis of the proximal femoral anatomy in the Croatian population. Int. Orthop. 2021, 45, 923–929. [Google Scholar] [CrossRef]
- Koff, M.F.; Amrami, K.K.; Kaufman, K.R. Clinical evaluation of T2 values of patellar cartilage in patients with osteoarthritis. Osteoarthr. Cartil. 2007, 15, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, R.; Thahakoya, R.; Luitjens, J.; Han, M.; Roach, K.E.; Jiang, F.; Souza, R.B.; Pedoia, V.; Majumdar, S. Effects of T1p Characteristics of Load-Bearing Hip Cartilage on Bilateral Knee Patellar Cartilage Subregions: Subjects with None to Moderate Radiographic Hip Osteoarthritis. J. Magn. Reson. Imaging 2024, 60, 186–202. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B Methodol. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, H.; Liu, C.; Liao, B.; Li, Y.; Zhu, Y.; Hu, Z.; Liao, J.; Liang, D. Recent advances in highly accelerated 3D MRI. Phys. Med. Biol. 2023, 68, 14TR01. [Google Scholar] [CrossRef] [PubMed]
- Roach, K.E.; Han, M.; Link, T.M.; Souza, R.; Majumdar, S.; Pedoia, V. Feasibility of simultaneous bilateral hip quantitative MRI. In Proceedings of the 2022 International Society for Magnetic Resonance in Medicine (ISMRM) Annual Meeting, London, UK, 7–12 May 2022; p. 2309. [Google Scholar]
- Greenwald, A.S.; Haynes, D.W. Weight-Bearing Areas in the Human Hip Joint. J. Bone Jt. Surg. Br. 1972, 54, 157–163. [Google Scholar] [CrossRef]
- Farkas, G.J.; Cvetanovich, G.L.; Rajan, K.B.; Espinoza Orías, A.A.; Nho, S.J. Impact of Femoroacetabular Impingement Morphology on Gait Assessment in Symptomatic Patients. Sports Health 2015, 7, 429–436. [Google Scholar] [CrossRef]
- Subburaj, K.; Valentinitsch, A.; Dillon, A.B.; Joseph, G.B.; Li, X.; Link, T.M.; Vail, T.P.; Majumdar, S. Regional variations in MR relaxation of hip joint cartilage in subjects with and without femoralacetabular impingement. Magn. Reson. Imaging 2013, 31, 1129–1136. [Google Scholar] [CrossRef]
- Youssefian, S.; Bressner, J.A.; Osanov, M.; Guest, J.K.; Zbijewski, W.B.; Levin, A.S. Sensitivity of the stress field of the proximal femur predicted by CT-based FE analysis to modeling uncertainties. J. Orthop. Res. 2022, 40, 1163–1173. [Google Scholar] [CrossRef] [PubMed]



| Parameters | ||
|---|---|---|
| Scanner Used | GE Signa Premier 3.0 T MR Scanner (GE Healthcare, Waukesha, WI, USA) | |
| Coils Used | 30-channel adaptive image receive (AIR) anterior array coil and 60-channel spine posterior array coil (GE Healthcare, Waukesha, WI, USA) | |
| Sequence Name | Hip MAPSS Sagittal | Fat-Suppressed 3D CUBE (Fast Spin Echo) Coronal |
| Acquisition Time | 16 min 30 s | 12 min 30 s |
| Acquisition Matrix | 256 × 128 | 200 × 400 |
| TR (per view) | 5.2 | 1200 |
| TSLs (ms) | 0, 15, 30, 45 | |
| TEs (ms) | 0, 10.4, 20.8, 41.6 | 20.62 |
| FOV (cm × cm) | 14 × 14 | 16 × 32 (S/I × R/L) |
| Slice Thickness (mm) | 4 | 0.8 |
| ARC Acceleration Factor | 2 × 2 (ky × kz) | (ky × kz) |
| Spin Lock Frequency | 300 Hz | |
| Number of Slices | 60 | 210–230 |
| BSDM (mm) (Mean ± SD) | Head | AMS | AMI | ALS | ALI | PMS | PMI | PLS | PLI |
|---|---|---|---|---|---|---|---|---|---|
| Control Group | 2.11 ± 0.79 | 2.12 ± 1.11 | 2.03 ± 0.77 | 2.04 ± 1.06 | 1.90 ± 0.84 | 2.03 ± 1.18 | 2.26 ± 1.77 | 2.30 ± 1.37 | 2.26 ± 1.53 |
| OA Group | 2.54 ± 0.88 | 2.43 ± 1.15 | 2.52 ± 1.11 | 2.49 ± 1.44 | 2.68 ± 1.73 | 2.92 ± 1.64 | 2.87 ± 1.31 | 2.40 ± 1.34 | 2.53 ± 1.48 |
| p-value | 0.097 | 0.371 | 0.079 | 0.235 | 0.043 * | 0.037 * | 0.221 | 0.811 | 0.548 |
| Parameters | p-Value | R/rho-Value | Correlation Type |
|---|---|---|---|
| Head vs. T2 femur R3 | 0.047 | 0.30 | Pearson |
| ALI vs. T2 acetabular R6 | 0.047 | 0.19 | Pearson |
| femur R2 | 0.039 | 0.32 | Spearman Rank |
| AMI vs. T2 femur R3 | 0.039 | 0.32 | Spearman Rank |
| ALS vs. T2 femur R3 | 0.039 | 0.31 | Spearman Rank |
| PMI vs. T2 femur R3 | 0.042 | 0.30 | Spearman Rank |
| acetabular R5 | 0.049 | 0.29 | Spearman Rank |
| acetabular R6 | 0.006 | 0.37 | Spearman Rank |
| PMS vs. T2 acetabular R6 | 0.042 | 0.30 | Spearman Rank |
| PMI vs. T2 acetabular R6 | 0.039 | 0.31 | Spearman Rank |
| AMS vs. CST | 0.039 | −0.31 | Spearman Rank |
| PMI vs. CST | 0.039 | −0.34 | Spearman Rank |
| PLS vs. CST | 0.006 | −0.41 | Spearman Rank |
| Head vs. SCT | 0.039 | 0.33 | Spearman Rank |
| AMS vs. SCT | 0.039 | 0.34 | Spearman Rank |
| PMI vs. SCT | 0.039 | 0.30 | Spearman Rank |
| Study | Anatomical Focus | Population | Assessment Method | Quantitative Metrics | Key Findings |
|---|---|---|---|---|---|
| Harris et al., 2012 [11] | Proximal femur and acetabulum | Healthy subjects | CT | Contact area, Load transfer and stress concentration | Hip contact stresses are concentrated in anterior–superior regions during weight bearing |
| Farkas et al., 2015 [40] | Proximal femur | FAI Patients | Radiographs and Gait analysis | Alpha angle, Cener edge angle Gait kinetics/kinematics | Cam morphology associated with altered hip kinematics |
| Valentina et al., 2016 [5] | Proximal femur and acetabulum | Healthy and OA subjects | MRI and T2 mapping | T1rho and T2 relaxation times | Early cartilage matrix degeneration detected using MRI prior to radiographic OA |
| Subburaj et al., 2013 [41] | Proximal femur and acetabulum | Healthy and FAI Patients | MRI and T2 mapping | T1rho and T2 relaxation times | Anterior-superior cartilage sub-region of patient were significantly different from controls |
| Youssefian et al., 2021 [42] | Proximal Femur | Cadaver | CT-based FE Models | Susceptibility of Stress | The model was more susceptible to element size and density–elasticity relationships |
| Proposed study | Proximal Femur | Health and OA subjects | MRI and T2 mapping, bone shape asymmetry and functional analysis | Bone shape difference measurements, T1rho and T2 relaxation times and functional activity parameters | Bone shape asymmetry correlated with biochemical characteristic changes of cartilage and functional activities |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Thahakoya, R.; Bhattacharjee, R.; Han, M.; Gassert, F.G.; Luitjens, J.; Pedoia, V.; Souza, R.B.; Majumdar, S. Artificial Intelligence-Based Proximal Bone Shape Asymmetry Analysis and Clinical Correlation with Cartilage Relaxation Times and Functional Activity. Bioengineering 2026, 13, 184. https://doi.org/10.3390/bioengineering13020184
Thahakoya R, Bhattacharjee R, Han M, Gassert FG, Luitjens J, Pedoia V, Souza RB, Majumdar S. Artificial Intelligence-Based Proximal Bone Shape Asymmetry Analysis and Clinical Correlation with Cartilage Relaxation Times and Functional Activity. Bioengineering. 2026; 13(2):184. https://doi.org/10.3390/bioengineering13020184
Chicago/Turabian StyleThahakoya, Rafeek, Rupsa Bhattacharjee, Misung Han, Felix Gerhard Gassert, Johanna Luitjens, Valentina Pedoia, Richard B. Souza, and Sharmila Majumdar. 2026. "Artificial Intelligence-Based Proximal Bone Shape Asymmetry Analysis and Clinical Correlation with Cartilage Relaxation Times and Functional Activity" Bioengineering 13, no. 2: 184. https://doi.org/10.3390/bioengineering13020184
APA StyleThahakoya, R., Bhattacharjee, R., Han, M., Gassert, F. G., Luitjens, J., Pedoia, V., Souza, R. B., & Majumdar, S. (2026). Artificial Intelligence-Based Proximal Bone Shape Asymmetry Analysis and Clinical Correlation with Cartilage Relaxation Times and Functional Activity. Bioengineering, 13(2), 184. https://doi.org/10.3390/bioengineering13020184








