Harnessing the Therapeutic Potential of Extracellular Vesicles for Oral Wound Healing
Abstract
1. Introduction
2. Mechanistic Differences Between Oral and Cutaneous Wound Healing
3. Clinical Complications of Poor Oral Wound Healing
3.1. Local Factors
3.2. Systemic Conditions and Comorbidities
3.3. Iatrogenic and Therapeutic Factors of Impaired Oral Wound Healing
3.4. Patient Related/Modifiable Risk Factors
3.5. Introduction to Extracellular Vesicles
3.6. Harvesting Extracellular Vesicles
3.7. Potential Sources of Extracellular Vesicles for Oral Wound Healing
3.8. Mesenchymal Stromal Cells
3.9. Macrophage
3.10. Fibroblasts
3.11. Saliva
3.12. Milk
3.13. Bacteria
3.14. Platelet
3.15. Dental Pulp Stem Cell
4. Engineering Extracellular Vesicles for Oral Wound Healing
4.1. Genetic Engineering of Parental Cells
4.2. Pharmacological and Environmental Preconditioning
4.3. Passive EV Loading
4.4. Active EV Loading
4.5. Cell Surface Engineering
4.6. Engineering Challenges
4.7. Advances in EV Delivery Strategies
4.8. Hydrogels
4.9. Microneedle Technology
4.10. Aerosols
4.11. Challenges and Opportunities
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAV | Adeno-associated virus |
| Alpha-SMA | Alpha-smooth muscle actin |
| ARG | Arginase |
| BEV | Bacterial extracellular vesicle |
| BMP2 | Bone morphogenetic protein 2 |
| CD | Cluster of differentiation |
| COL1A1 | Collagen type I alpha 1 chain |
| COL2A | Collagen type II alpha 1 chain |
| Cx43 | Connexin 43 |
| CXCL10 | Chemokine (C-X-C motif) ligand 10 |
| ECM | Extracellular matrix |
| EGF | Epidermal growth factor |
| EPL | Poly-e-L-lysine |
| EV | Extracellular vesicle |
| FEV | Fibroblast-derived extracellular vesicle |
| FGF2 | Fibroblast growth factor 2 |
| GelMA | Gelatin methacrylate |
| GAPDH | Glyceraldehyde-3-phosphate dehydrogenase |
| HbA1c | Hemoglobin A1c |
| HIF-1 | Hypoxia-inducible factor 1 |
| HIF1AN | Hypoxia-inducible factor 1 subunit alpha inhibitor |
| HUVEC | Human umbilical vein endothelial cell |
| IL-1β | Interleukin-1 beta |
| IL-4 | Interleukin-4 |
| IL-6 | Interleukin-6 |
| IL-8 | Interleukin-8 |
| IL-10 | Interleukin-10 |
| ISEV | International Society of Extracellular Vesicles |
| ITGB1 | Integrin subunit beta 1 |
| LIF | Leukemia inhibitory factor |
| LPS | Lipopolysaccharide |
| LRR | Leucine-rich repeat |
| MAP2K2 | Mitogen-activated protein kinase kinase 2 |
| MAP3K7 | Mitogen-activated protein kinase kinase kinase 7 |
| MISEV | Minimal Information for Studies of Extracellular Vesicles |
| MSC | Mesenchymal stromal cell |
| MRC1 | Mannose receptor C-type 1 |
| MMP1 | Matrix metallopeptidase 1 |
| MMP3 | Matrix metallopeptidase 3 |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NLRP3 | NLR family pyrin domain containing 3 |
| NSAID | Nonsteroidal anti-inflammatory drug |
| OHAMA | Oxidized methacrylate hyaluronic acid |
| ONF | Oronasal fistula |
| PCL | Polycaprolactone |
| PDCD6IP/ALIX | Programmed cell death 6 interacting protein |
| PDGF | Platelet-derived growth factor |
| PDLSC | Periodontal ligament stem cell |
| PEG | Polyethylene glycol |
| PI3K | Phosphatidylinositol 3-kinase |
| PITX | Paired-like homeodomain |
| PLA | Polylactic acid |
| Prx1+ | Paired-related homeobox-1 positive (fibroblast subtype) |
| PTEN | Phosphatase and tensin homolog |
| PYD | Pyrin domain |
| ROS | Reactive oxygen species |
| SASP | Senescence-associated secretory phenotype |
| SMAD6 | Mothers against decapentaplegic homolog 6 |
| SOX2 | Sex-determining region Y-box 2 |
| SILY | Collagen-binding peptide motif |
| STAT3 | Signal transducer and activator of transcription 3 |
| TAB2 | TGF-β activated kinase 1 binding protein 2 |
| TGF-β1 | Transforming growth factor beta 1 |
| TLR3 | Toll-like receptor 3 |
| TNF-α | Tumor necrosis factor alpha |
| Trem1 | Triggering Receptor expressed on myeloid cells-1 |
| tRNA-Met | Transfer RNA methionine |
| TSG101 | Tumor susceptibility gene 101 |
| UBE2O | Ubiquitin-conjugating enzyme E2O |
| VEGF | Vascular endothelial growth factor |
| VCL | Vinculin |
| VCAN | Versican |
| VH-EV | EVs loaded with VH298 |
| WNT5A | Wingless-related integration site 5A |
| YAP | Yes-associated protein |
References
- Wallace, H.A.; Basehore, B.M.; Zito, P.M. Wound Healing Phases. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2025. [Google Scholar]
- Toma, A.I.; Fuller, J.M.; Willett, N.J.; Goudy, S.L. Oral wound healing models and emerging regenerative therapies. Transl. Res. 2021, 236, 17–34. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.J. Physiology of wound healing and surgical wound care. Asaio J. 2000, 46, S2–S5. [Google Scholar] [CrossRef]
- Wilgus, T.A.; Roy, S.; McDaniel, J.C. Neutrophils and Wound Repair: Positive Actions and Negative Reactions. Adv. Wound Care 2013, 2, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Austermann, J.; Roth, J.; Barczyk-Kahlert, K. The Good and the Bad: Monocytes’ and Macrophages’ Diverse Functions in Inflammation. Cells 2022, 11, 1979. [Google Scholar] [CrossRef]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: A critical step during wound healing. Cell. Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar] [CrossRef]
- Pastar, I.; Stojadinovic, O.; Yin, N.C.; Ramirez, H.; Nusbaum, A.G.; Sawaya, A.; Patel, S.B.; Khalid, L.; Isseroff, R.R.; Tomic-Canic, M. Epithelialization in Wound Healing: A Comprehensive Review. Adv. Wound Care 2014, 3, 445–464. [Google Scholar] [CrossRef]
- El Ghalbzouri, A.; Hensbergen, P.; Gibbs, S.; Kempenaar, J.; van der Schors, R.; Ponec, M. Fibroblasts facilitate re-epithelialization in wounded human skin equivalents. Lab. Investig. 2004, 84, 102–112. [Google Scholar] [CrossRef]
- Irma, J.; Kartasasmita, A.S.; Kartiwa, A.; Irfani, I.; Rizki, S.A.; Onasis, S. From Growth Factors to Structure: PDGF and TGF-β in Granulation Tissue Formation. A Literature Review. J. Cell. Mol. Med. 2025, 29, e70374. [Google Scholar] [CrossRef] [PubMed]
- Waasdorp, M.; Krom, B.P.; Bikker, F.J.; van Zuijlen, P.P.M.; Niessen, F.B.; Gibbs, S. The Bigger Picture: Why Oral Mucosa Heals Better Than Skin. Biomolecules 2021, 11, 1165. [Google Scholar] [CrossRef]
- Rodrigues Neves, C.; Buskermolen, J.; Roffel, S.; Waaijman, T.; Thon, M.; Veerman, E.; Gibbs, S. Human saliva stimulates skin and oral wound healing in vitro. J. Tissue Eng. Regen. Med. 2019, 13, 1079–1092. [Google Scholar] [CrossRef]
- Brand, H.S.; Veerman, E.C. Saliva and wound healing. Chin. J. Dent. Res. 2013, 16, 7–12. [Google Scholar]
- Lei, X.; Cheng, L.; Lin, H.; Pang, M.; Yao, Z.; Chen, C.; Forouzanfar, T.; Bikker, F.J.; Wu, G.; Cheng, B. Human Salivary Histatin-1 Is More Efficacious in Promoting Acute Skin Wound Healing Than Acellular Dermal Matrix Paste. Front. Bioeng. Biotechnol. 2020, 8, 999. [Google Scholar] [CrossRef]
- Shi, L.; Xu, Y.; Li, J.; He, L.; Li, K.; Yin, S.; Nie, M.; Liu, X. Vascularized characteristics and functional regeneration of three-dimensional cell reconstruction of oral mucosa equivalents based on vascular homeostasis phenotypic modification. J. Tissue Eng. 2024, 15, 20417314241268912. [Google Scholar] [CrossRef] [PubMed]
- Kadwaikar, M.; Shinde, V. Healing with microbial Allies: Exploration of probiotics in wound management. Microb. Pathog. 2025, 207, 107906. [Google Scholar] [CrossRef]
- Ko, K.I.; DerGarabedian, B.P.; Chen, Z.; Debnath, R.; Ko, A.; Link, B.N.; Korostoff, J.M.; Graves, D.T. Distinct fibroblast progenitor subpopulation expedites regenerative mucosal healing by immunomodulation. J. Exp. Med. 2023, 220, e20221350. [Google Scholar] [CrossRef] [PubMed]
- Bogdan-Andreescu, C.F.; Bănățeanu, A.-M.; Botoacă, O.; Defta, C.L.; Poalelungi, C.-V.; Brăila, A.D.; Damian, C.M.; Brăila, M.G.; Dȋră, L.M. Oral Wound Healing in Aging Population. Surgeries 2024, 5, 956–969. [Google Scholar] [CrossRef]
- Politis, C.; Schoenaers, J.; Jacobs, R.; Agbaje, J.O. Wound Healing Problems in the Mouth. Front. Physiol. 2016, 7, 507. [Google Scholar] [CrossRef]
- Ko, K.I.; Sculean, A.; Graves, D.T. Diabetic wound healing in soft and hard oral tissues. Transl. Res. 2021, 236, 72–86. [Google Scholar] [CrossRef]
- Lionel, D.; Christophe, L.; Marc, A.; Jean-Luc, C. Oral mucositis induced by anticancer treatments: Physiopathology and treatments. Ther. Clin. Risk Manag. 2006, 2, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Bozdemir, E.; Yilmaz, H.H.; Orhan, H. Oral mucosal lesions and risk factors in elderly dental patients. J. Dent. Res. Dent. Clin. Dent. Prospect. 2019, 13, 24–30. [Google Scholar] [CrossRef]
- Richards, D. One billion people have experienced a traumatic dental injury. Evid. Based Dent. 2018, 19, 34–35. [Google Scholar] [CrossRef]
- Sadhu, P. Oronasal fistula in cleft palate surgery. Indian J. Plast. Surg. 2009, 42, S123–S128. [Google Scholar] [CrossRef]
- Xu, J.; Gou, L.; Zhang, P.; Li, H.; Qiu, S. Platelet-rich plasma and regenerative dentistry. Aust. Dent. J. 2020, 65, 131–142. [Google Scholar] [CrossRef]
- Kennedy, T.J.; Hall, J.E. A drug-free oral hydrogel wound dressing for pain management in immediate denture patients. Gen. Dent. 2009, 57, 420–427. [Google Scholar] [PubMed]
- Narauskaitė, D.; Vydmantaitė, G.; Rusteikaitė, J.; Sampath, R.; Rudaitytė, A.; Stašytė, G.; Aparicio Calvente, M.I.; Jekabsone, A. Extracellular Vesicles in Skin Wound Healing. Pharmaceuticals 2021, 14, 811. [Google Scholar] [CrossRef] [PubMed]
- Park, D.J.; Choi, W.; Sayeed, S.; Dorschner, R.A.; Rainaldi, J.; Ho, K.; Kezios, J.; Nolan, J.P.; Mali, P.; Costantini, T.; et al. Defining the activity of pro-reparative extracellular vesicles in wound healing based on miRNA payloads and cell type-specific lineage mapping. Mol. Ther. 2024, 32, 3059–3079. [Google Scholar] [CrossRef]
- Setua, S.; Thangaraju, K.; Dzieciatkowska, M.; Wilkerson, R.B.; Nemkov, T.; Lamb, D.R.; Tagaya, Y.; Boyer, T.; Rowden, T.; Doctor, A.; et al. Coagulation potential and the integrated omics of extracellular vesicles from COVID-19 positive patient plasma. Sci. Rep. 2022, 12, 22191. [Google Scholar] [CrossRef] [PubMed]
- Owens, A.P., 3rd; Mackman, N. Microparticles in hemostasis and thrombosis. Circ. Res. 2011, 108, 1284–1297. [Google Scholar] [CrossRef]
- Berckmans, R.J.; Nieuwland, R.; Böing, A.N.; Romijn, F.P.; Hack, C.E.; Sturk, A. Cell-derived microparticles circulate in healthy humans and support low grade thrombin generation. Thromb. Haemost. 2001, 85, 639–646. [Google Scholar] [CrossRef]
- Tao, S.C.; Guo, S.C.; Zhang, C.Q. Platelet-derived Extracellular Vesicles: An Emerging Therapeutic Approach. Int. J. Biol. Sci. 2017, 13, 828–834. [Google Scholar] [CrossRef]
- Aatonen, M.; Grönholm, M.; Siljander, P.R. Platelet-derived microvesicles: Multitalented participants in intercellular communication. Semin. Thromb. Hemost. 2012, 38, 102–113. [Google Scholar] [CrossRef]
- Ding, J.Y.; Chen, M.J.; Wu, L.F.; Shu, G.F.; Fang, S.J.; Li, Z.Y.; Chu, X.R.; Li, X.K.; Wang, Z.G.; Ji, J.S. Mesenchymal stem cell-derived extracellular vesicles in skin wound healing: Roles, opportunities and challenges. Mil. Med. Res. 2023, 10, 36. [Google Scholar] [CrossRef]
- Sun, J.; Wang, Z.; Liu, P.; Hu, Y.; Li, T.; Yang, J.; Gao, P.; Xu, Q. Exosomes Derived From Human Gingival Mesenchymal Stem Cells Attenuate the Inflammatory Response in Periodontal Ligament Stem Cells. Front. Chem. 2022, 10, 863364. [Google Scholar] [CrossRef]
- Li, X.; Liu, L.; Yang, J.; Yu, Y.; Chai, J.; Wang, L.; Ma, L.; Yin, H. Exosome Derived From Human Umbilical Cord Mesenchymal Stem Cell Mediates MiR-181c Attenuating Burn-induced Excessive Inflammation. EBioMedicine 2016, 8, 72–82. [Google Scholar] [CrossRef]
- Peng, B.; Wang, L.; Han, G.; Cheng, Y. Mesenchymal stem cell-derived exosomes: A potential cell-free therapy for orthodontic tooth stability management. Stem Cell Res. Ther. 2024, 15, 342. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.; He, Z. Mesenchymal stem cell-derived exosomes regulate the polarization and inflammatory response of macrophages via miR-21-5p to promote repair after myocardial reperfusion injury. Ann. Transl. Med. 2021, 9, 1323. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Guan, L.; Liu, Z.; Yan, F.; Fang, S.; Zhang, X.; Gao, C. Using extracellular vesicles derived from human umbilical cord mesenchymal stem cells for a topical coating promotes oral mucositis healing in rats. Ann. Transl. Med. 2022, 10, 290. [Google Scholar] [CrossRef]
- Shahsavandi, Y.; Banaeian, F.; Jafarinia, M.; Nasri, F.; Shapoori, S. miRNAs from mesenchymal-stem-cell-derived extracellular vesicles: Emerging players in regenerative medicine and disease therapy. Mol. Ther. Nucleic Acids 2025, 36, 102715. [Google Scholar] [CrossRef]
- Li, Q.; Zhou, L.; Li, W.; Zhao, W.; Chen, W.; AlQranei, M.S.; Bi, J.; Huang, P. GelMA hydrogel-loaded extracellular vesicles derived from keratinocytes promote skin microvasculature regeneration and wound healing in diabetic mice through activation of the PDGF-induced PI3K/AKT pathway. Cell Biol. Toxicol. 2025, 41, 103. [Google Scholar] [CrossRef] [PubMed]
- Ju, C.; Liu, D. Exosomal microRNAs from Mesenchymal Stem Cells: Novel Therapeutic Effect in Wound Healing. Tissue Eng. Regen. Med. 2023, 20, 647–660. [Google Scholar] [CrossRef]
- Zeng, Q.L.; Liu, D.W. Mesenchymal stem cell-derived exosomes: An emerging therapeutic strategy for normal and chronic wound healing. World J. Clin. Cases 2021, 9, 6218–6233. [Google Scholar] [CrossRef]
- Li, Z.; Bai, Y.; Wu, H.; Feng, Y.; Wang, X.; Zhao, C.; Wang, X. PTEN/PI3K/AKT pathway activation with hypoxia-induced human umbilical vein endothelial cell exosome for angiogenesis-based diabetic skin reconstruction. Mater. Today Bio 2025, 32, 101651. [Google Scholar] [CrossRef]
- Yuan, R.; Dai, X.; Li, Y.; Li, C.; Liu, L. Exosomes from miR-29a-modified adipose-derived mesenchymal stem cells reduce excessive scar formation by inhibiting TGF-β2/Smad3 signaling. Mol. Med. Rep. 2021, 24, 12398. [Google Scholar] [CrossRef]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef]
- Wang, L.; Hu, L.; Zhou, X.; Xiong, Z.; Zhang, C.; Shehada, H.M.A.; Hu, B.; Song, J.; Chen, L. Exosomes secreted by human adipose mesenchymal stem cells promote scarless cutaneous repair by regulating extracellular matrix remodelling. Sci. Rep. 2017, 7, 13321. [Google Scholar] [CrossRef]
- Ahmadpour, F.; Rasouli, H.R.; Talebi, S.; Golchin, D.; Esmailinejad, M.R.; Razie, A. Effects of exosomes derived from fibroblast cells on skin wound healing in Wistar rats. Burns 2023, 49, 1372–1381. [Google Scholar] [CrossRef]
- Oh, E.J.; Gangadaran, P.; Rajendran, R.L.; Kim, H.M.; Oh, J.M.; Choi, K.Y.; Chung, H.Y.; Ahn, B.C. Extracellular vesicles derived from fibroblasts promote wound healing by optimizing fibroblast and endothelial cellular functions. Stem Cells 2021, 39, 266–279. [Google Scholar] [CrossRef] [PubMed]
- Vila, T.; Rizk, A.M.; Sultan, A.S.; Jabra-Rizk, M.A. The power of saliva: Antimicrobial and beyond. PLoS Pathog. 2019, 15, e1008058. [Google Scholar] [CrossRef]
- Khurshid, Z.; Naseem, M.; Sheikh, Z.; Najeeb, S.; Shahab, S.; Zafar, M.S. Oral antimicrobial peptides: Types and role in the oral cavity. Saudi Pharm. J. 2016, 24, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Huang, L.; Clayton, J.M.; Nicholas, S.E.; Hefley, B.S.; Ma, J.X.; Karamichos, D. Exploring the Therapeutic Potential of Salivary Exosomes in Corneal Epithelial Wound Healing. Investig. Ophthalmol. Vis. Sci. 2025, 66, 8. [Google Scholar] [CrossRef]
- Cui, C.; Mei, L.; Wang, D.; Jia, P.; Zhou, Q.; Liu, W. A self-stabilized and water-responsive deliverable coenzyme-based polymer binary elastomer adhesive patch for treating oral ulcer. Nat. Commun. 2023, 14, 7707. [Google Scholar] [CrossRef] [PubMed]
- Prost-Squarcioni, C. Histology of skin and hair follicle. Med. Sci. 2006, 22, 131–137. [Google Scholar] [CrossRef]
- Bielecki, J.E.; Chen, R.J.; Gupta, V. Caustic Ingestions. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2025. [Google Scholar]
- Macartney, R.A.; Das, A.; Imaniyyah, A.G.; Fricker, A.T.; Smith, A.M.; Fedele, S.; Roy, I.; Kim, H.W.; Lee, D.; Knowles, J.C. In vitro and ex vivo models of the oral mucosa as platforms for the validation of novel drug delivery systems. J. Tissue Eng. 2025, 16, 20417314241313458. [Google Scholar] [CrossRef]
- Yousef, H.; Alhajj, M.; Fakoya, A.O.; Sharma, S. Anatomy, Skin (Integument), Epidermis. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2025. [Google Scholar]
- Chuhuaicura, P.; Rodríguez-Niklitschek, C.; Oporto, G.H.; Salazar, L.A. Distinct Molecular Mechanisms in Oral Mucosal Wound Healing: Translational Insights and Future Directions. Int. J. Mol. Sci. 2025, 26, 10660. [Google Scholar] [CrossRef]
- Overmiller, A.M.; Sawaya, A.P.; Hope, E.D.; Morasso, M.I. Intrinsic Networks Regulating Tissue Repair: Comparative Studies of Oral and Skin Wound Healing. Cold Spring Harb. Perspect. Biol. 2022, 14, a041244. [Google Scholar] [CrossRef] [PubMed]
- Szpaderska, A.M.; Walsh, C.G.; Steinberg, M.J.; DiPietro, L.A. Distinct patterns of angiogenesis in oral and skin wounds. J. Dent. Res. 2005, 84, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Iglesias-Bartolome, R.; Uchiyama, A.; Molinolo, A.A.; Abusleme, L.; Brooks, S.R.; Callejas-Valera, J.L.; Edwards, D.; Doci, C.; Asselin-Labat, M.-L.; Onaitis, M.W.; et al. Transcriptional signature primes human oral mucosa for rapid wound healing. Sci. Transl. Med. 2018, 10, eaap8798. [Google Scholar] [CrossRef]
- Han, C.; Zhang, Y.; Yuan, H.; Keiser, S.; Leonardo, T.; Chen, L.; Wietecha, M.; DiPietro, L.A. Transcriptomic Differences Between Immortalized Oral and Skin Keratinocytes. FASEB J. 2025, 39, e70744. [Google Scholar] [CrossRef]
- Xuanyuan, X.; Zhang, L.; Zheng, Y.; Jiang, R.; Ma, Y.; Liu, R.; Hou, P.; Lei, M.; Xu, H.; Zeng, H. SPRR1B+ keratinocytes prime oral mucosa for rapid wound healing via STAT3 activation. Commun. Biol. 2024, 7, 1155. [Google Scholar] [CrossRef]
- Xue, J.-D.; Gao, J.; Tang, A.-F.; Feng, C. Shaping the immune landscape: Multidimensional environmental stimuli refine macrophage polarization and foster revolutionary approaches in tissue regeneration. Heliyon 2024, 10, e37192. [Google Scholar] [CrossRef]
- Wang, Y.; Mao, J.; Wang, Y.; Wang, R.; Duan, D.; Liu, Z.; Hu, X.; Yu, Z.; Shi, X. Macrophage-induced immunomodulation in oral tissue repair and regeneration: Recent advances and future perspectives. J. Adv. Res 2025. [Google Scholar] [CrossRef]
- Roeslan, O.; Wulansari, S.; Tazkia, R. Why Wound Healing in the Oral Cavity Occurs Faster than in the Skin. J. Kedokt. Gigi Terpadu 2025, 7, 52–57. [Google Scholar] [CrossRef]
- El Ayadi, A.; Jay, J.W.; Prasai, A. Current Approaches Targeting the Wound Healing Phases to Attenuate Fibrosis and Scarring. Int. J. Mol. Sci. 2020, 21, 1105. [Google Scholar] [CrossRef]
- Li, J.; Wang, S.; Yuan, J.; Mao, X.; Wang, X.; Zhang, L.; Dong, Q.; Chen, Z.; Wang, Y.; Tang, N. Tissue regeneration: Unraveling strategies for resolving pathological fibrosis. Cell Stem Cell 2025, 32, 1639–1658. [Google Scholar] [CrossRef]
- Mak, K.; Manji, A.; Gallant-Behm, C.; Wiebe, C.; Hart, D.A.; Larjava, H.; Häkkinen, L. Scarless healing of oral mucosa is characterized by faster resolution of inflammation and control of myofibroblast action compared to skin wounds in the red Duroc pig model. J. Dermatol. Sci. 2009, 56, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, M.; Pawłowska, A.; Orzeł, A.; Sulej, L.; Muzyka-Placzyńska, K.; Baran, A.; Filipecka-Tyczka, D.; Pawłowska, P.; Nowińska, A.; Bogusławska, J.; et al. Wound Microbiota and Its Impact on Wound Healing. Int. J. Mol. Sci. 2023, 24, 17318. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.B.; Alimova, Y.; Ebersole, J.L. Macrophage polarization in response to oral commensals and pathogens. Pathog. Dis. 2016, 74, ftw011. [Google Scholar] [CrossRef]
- Shannon, D.B.; McKeown, S.T.; Lundy, F.T.; Irwin, C.R. Phenotypic differences between oral and skin fibroblasts in wound contraction and growth factor expression. Wound Repair Regen. 2006, 14, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Vijayashree, R.J.; Sivapathasundharam, B. The diverse role of oral fibroblasts in normal and disease. J. Oral Maxillofac. Pathol. 2022, 26, 6–13. [Google Scholar] [CrossRef]
- Schrementi, M.E.; Ferreira, A.M.; Zender, C.; DiPietro, L.A. Site-specific production of TGF-beta in oral mucosal and cutaneous wounds. Wound Repair Regen. 2008, 16, 80–86. [Google Scholar] [CrossRef]
- Willenborg, S.; Schönborn, K.; Sawant, M.; Bornikoel, A.; Yamane, T.; Zeinert, I.; Eckes, B.; Eming, S.A.; Krieg, T. Fibroblast-Derived TGFβ1 Regulates Skin Repair and Fibrosis. Wound Repair Regen. 2025, 33, e70065. [Google Scholar] [CrossRef] [PubMed]
- Demidova-Rice, T.N.; Hamblin, M.R.; Herman, I.M. Acute and impaired wound healing: Pathophysiology and current methods for drug delivery, part 1: Normal and chronic wounds: Biology, causes, and approaches to care. Adv. Ski. Wound Care 2012, 25, 304–314. [Google Scholar] [CrossRef]
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci. Transl. Med. 2014, 6, 265sr6. [Google Scholar] [CrossRef] [PubMed]
- Hom, D.B.; Davis, M.E. Reducing Risks for Poor Surgical Wound Healing. Facial Plast. Surg. Clin. N. Am. 2023, 31, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Han, N.; Jia, L.; Guo, L.; Su, Y.; Luo, Z.; Du, J.; Mei, S.; Liu, Y. Balanced oral pathogenic bacteria and probiotics promoted wound healing via maintaining mesenchymal stem cell homeostasis. Stem Cell Res. Ther. 2020, 11, 61. [Google Scholar] [CrossRef]
- Maier, T. Oral Microbiome in Health and Disease: Maintaining a Healthy, Balanced Ecosystem and Reversing Dysbiosis. Microorganisms 2023, 11, 1453. [Google Scholar] [CrossRef]
- Jiang, W.; Lv, H.; Wang, H.; Wang, D.; Sun, S.; Jia, Q.; Wang, P.; Song, B.; Ni, L. Activation of the NLRP3/caspase-1 inflammasome in human dental pulp tissue and human dental pulp fibroblasts. Cell Tissue Res. 2015, 361, 541–555. [Google Scholar] [CrossRef]
- Ding, P.H.; Yang, M.X.; Wang, N.N.; Jin, L.J.; Dong, Y.; Cai, X.; Chen, L.L. Porphyromonas gingivalis-Induced NLRP3 Inflammasome Activation and Its Downstream Interleukin-1β Release Depend on Caspase-4. Front. Microbiol. 2020, 11, 1881. [Google Scholar] [CrossRef]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef]
- Rajasekaran, J.J.; Krishnamurthy, H.K.; Bosco, J.; Jayaraman, V.; Krishna, K.; Wang, T.; Bei, K. Oral Microbiome: A Review of Its Impact on Oral and Systemic Health. Microorganisms 2024, 12, 1797. [Google Scholar] [CrossRef]
- Santonocito, S.; Giudice, A.; Polizzi, A.; Troiano, G.; Merlo, E.M.; Sclafani, R.; Grosso, G.; Isola, G. A Cross-Talk between Diet and the Oral Microbiome: Balance of Nutrition on Inflammation and Immune System’s Response during Periodontitis. Nutrients 2022, 14, 2426. [Google Scholar] [CrossRef]
- Gegout, P.Y.; Mary, B.; Stutz, C.; Hyenne, V.; Ginesin, O.; Zigdon-Giladi, H.; Huck, O. Porphyromonas gingivalis infection of oral keratinocytes drives the release of pro-inflammatory extracellular vesicles. Sci. Rep. 2025, 15, 24704. [Google Scholar] [CrossRef]
- Ma, R.Y.; Deng, Z.L.; Du, Q.Y.; Dai, M.Q.; Luo, Y.Y.; Liang, Y.E.; Dai, X.Z.; Guo, S.M.; Zhao, W.H. Enterococcus faecalis Extracellular Vesicles Promote Apical Periodontitis. J. Dent. Res. 2024, 103, 672–682. [Google Scholar] [CrossRef]
- Wu, R.; Cui, G.; Cao, Y.; Zhao, W.; Lin, H. Streptococcus Mutans Membrane Vesicles Enhance Candida albicans Pathogenicity and Carbohydrate Metabolism. Front. Cell. Infect. Microbiol. 2022, 12, 940602. [Google Scholar] [CrossRef]
- Chen, Y.; Huang, X.; Liu, A.; Fan, S.; Liu, S.; Li, Z.; Yang, X.; Guo, H.; Wu, M.; Liu, M.; et al. Lactobacillus reuteri Vesicles Regulate Mitochondrial Function of Macrophages to Promote Mucosal and Cutaneous Wound Healing. Adv. Sci. 2024, 11, e2309725. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, T.; Alberius, P.; Schmidtchen, A.; Reiss, K.; Schröder, J.M.; Sørensen, O.E. Saliva induces expression of antimicrobial peptides and promotes intracellular killing of bacteria in keratinocytes by epidermal growth factor receptor transactivation. Br. J. Dermatol. 2017, 176, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Pinna, R.; Campus, G.; Cumbo, E.; Mura, I.; Milia, E. Xerostomia induced by radiotherapy: An overview of the physiopathology, clinical evidence, and management of the oral damage. Ther. Clin. Risk Manag. 2015, 11, 171–188. [Google Scholar] [CrossRef] [PubMed]
- Negrato, C.A.; Tarzia, O. Buccal alterations in diabetes mellitus. Diabetol. Metab. Syndr. 2010, 2, 3. [Google Scholar] [CrossRef]
- Socransky, S.S.; Haffajee, A.D.; Cugini, M.A.; Smith, C.; Kent, R.L., Jr. Microbial complexes in subgingival plaque. J. Clin. Periodontol. 1998, 25, 134–144. [Google Scholar] [CrossRef]
- Canning, P.; Ruan, Q.; Schwerd, T.; Hrdinka, M.; Maki, J.L.; Saleh, D.; Suebsuwong, C.; Ray, S.; Brennan, P.E.; Cuny, G.D.; et al. Inflammatory Signaling by NOD-RIPK2 Is Inhibited by Clinically Relevant Type II Kinase Inhibitors. Chem. Biol. 2015, 22, 1174–1184. [Google Scholar] [CrossRef]
- Han, Y.W. Fusobacterium nucleatum: A commensal-turned pathogen. Curr. Opin. Microbiol. 2015, 23, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Engevik, M.A.; Danhof, H.A.; Ruan, W.; Engevik, A.C.; Chang-Graham, A.L.; Engevik, K.A.; Shi, Z.; Zhao, Y.; Brand, C.K.; Krystofiak, E.S.; et al. Fusobacterium nucleatum Secretes Outer Membrane Vesicles and Promotes Intestinal Inflammation. mBio 2021, 12, 10–1128. [Google Scholar] [CrossRef]
- Gudadappanavar, A.M.; Hombal, P.R.; Timashetti, S.S.; Javali, S.B. Influence of Lactobacillus acidophilus and Lactobacillus plantarum on wound healing in male Wistar rats—An experimental study. Int. J. Appl. Basic Med. Res. 2017, 7, 233–238. [Google Scholar] [CrossRef]
- Hosseini, M.S.; Sanaie, S.; Mahmoodpoor, A.; Jabbari Beyrami, S.; Jabbari Beyrami, H.; Fattahi, S.; Jahanshahlou, F.; Zarei, M.; Rahimi Mamaghani, A.; Kuchaki Rafsanjani, M. Cancer treatment-related xerostomia: Basics, therapeutics, and future perspectives. Eur. J. Med. Res. 2024, 29, 571. [Google Scholar] [CrossRef]
- Kapourani, A.; Kontogiannopoulos, K.N.; Manioudaki, A.-E.; Poulopoulos, A.K.; Tsalikis, L.; Assimopoulou, A.N.; Barmpalexis, P. A Review on Xerostomia and Its Various Management Strategies: The Role of Advanced Polymeric Materials in the Treatment Approaches. Polymers 2022, 14, 850. [Google Scholar] [CrossRef]
- Dumbrigue, H.B.; Sandow, P.L.; Nguyen, K.H.; Humphreys-Beher, M.G. Salivary epidermal growth factor levels decrease in patients receiving radiation therapy to the head and neck. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 2000, 89, 710–716. [Google Scholar] [CrossRef]
- Azuma, N.; Katada, Y.; Sano, H. Deterioration in saliva quality in patients with Sjögren’s syndrome: Impact of decrease in salivary epidermal growth factor on the severity of intraoral manifestations. Inflamm. Regen. 2018, 38, 6. [Google Scholar] [CrossRef] [PubMed]
- Al-Maskari, A.Y.; Al-Maskari, M.Y.; Al-Sudairy, S. Oral Manifestations and Complications of Diabetes Mellitus: A review. Sultan Qaboos Univ. Med. J. 2011, 11, 179–186. [Google Scholar] [CrossRef]
- Xiong, Y.; Knoedler, S.; Alfertshofer, M.; Kim, B.S.; Jiang, D.; Liu, G.; Rinkevich, Y.; Mi, B. Mechanisms and therapeutic opportunities in metabolic aberrations of diabetic wounds: A narrative review. Cell Death Dis. 2025, 16, 341. [Google Scholar] [CrossRef] [PubMed]
- Rask-Madsen, C.; King, G.L. Vascular complications of diabetes: Mechanisms of injury and protective factors. Cell Metab. 2013, 17, 20–33. [Google Scholar] [CrossRef] [PubMed]
- Urata, Y.; Yamaguchi, M.; Higashiyama, Y.; Ihara, Y.; Goto, S.; Kuwano, M.; Horiuchi, S.; Sumikawa, K.; Kondo, T. Reactive oxygen species accelerate production of vascular endothelial growth factor by advanced glycation end products in RAW264.7 mouse macrophages. Free Radic. Biol. Med. 2002, 32, 688–701. [Google Scholar] [CrossRef]
- González-Moles, M.; Ramos-García, P. State of Evidence on Oral Health Problems in Diabetic Patients: A Critical Review of the Literature. J. Clin. Med. 2021, 10, 5383. [Google Scholar] [CrossRef] [PubMed]
- López-Pintor, R.M.; Casañas, E.; González-Serrano, J.; Serrano, J.; Ramírez, L.; de Arriba, L.; Hernández, G. Xerostomia, Hyposalivation, and Salivary Flow in Diabetes Patients. J. Diabetes Res. 2016, 2016, 4372852. [Google Scholar] [CrossRef]
- Rodrigues, C.F.; Rodrigues, M.E.; Henriques, M. Candida sp. Infections in Patients with Diabetes Mellitus. J. Clin. Med. 2019, 8, 76. [Google Scholar] [CrossRef] [PubMed]
- Jegan, M.; Ahamed, A.S.; Lakshmi, G.V. The Influence of Glycemic Control Over Post-extraction Healing in Diabetic Patients. Cureus 2024, 16, e70998. [Google Scholar] [CrossRef]
- Preshaw, P.M.; Alba, A.L.; Herrera, D.; Jepsen, S.; Konstantinidis, A.; Makrilakis, K.; Taylor, R. Periodontitis and diabetes: A two-way relationship. Diabetologia 2012, 55, 21–31. [Google Scholar] [CrossRef]
- Păunică, I.; Giurgiu, M.; Dumitriu, A.S.; Păunică, S.; Pantea Stoian, A.M.; Martu, M.A.; Serafinceanu, C. The Bidirectional Relationship between Periodontal Disease and Diabetes Mellitus-A Review. Diagnostics 2023, 13, 681. [Google Scholar] [CrossRef]
- D’Aiuto, F.; Gkranias, N.; Bhowruth, D.; Khan, T.; Orlandi, M.; Suvan, J.; Masi, S.; Tsakos, G.; Hurel, S.; Hingorani, A.D.; et al. Systemic effects of periodontitis treatment in patients with type 2 diabetes: A 12 month, single-centre, investigator-masked, randomised trial. Lancet Diabetes Endocrinol. 2018, 6, 954–965. [Google Scholar] [CrossRef]
- Heron, S.E.; Elahi, S. HIV Infection and Compromised Mucosal Immunity: Oral Manifestations and Systemic Inflammation. Front. Immunol. 2017, 8, 241. [Google Scholar] [CrossRef]
- Nappalli, D.; Lingappa, A. Oral manifestations in transplant patients. Dent. Res. J. 2015, 12, 199–208. [Google Scholar]
- Słonimska, P.; Sachadyn, P.; Zieliński, J.; Skrzypski, M.; Pikuła, M. Chemotherapy-Mediated Complications of Wound Healing: An Understudied Side Effect. Adv. Wound Care 2024, 13, 187–199. [Google Scholar] [CrossRef]
- Xu, W.; Boer, K.; Hesselink, D.A.; Baan, C.C. Extracellular Vesicles and Immune Activation in Solid Organ Transplantation: The Impact of Immunosuppression. BioDrugs 2025, 39, 445–459. [Google Scholar] [CrossRef]
- Midwood, K.S.; Williams, L.V.; Schwarzbauer, J.E. Tissue repair and the dynamics of the extracellular matrix. Int. J. Biochem. Cell Biol. 2004, 36, 1031–1037. [Google Scholar] [CrossRef]
- Tachibana, K.; Kasahara, M.; Matsuura, N.; Ichinohe, T. Effects of Local Anesthetics With Vasoconstrictors on Dental Pulp Blood Flow and Oxygen Tension. Anesth. Prog. 2021, 68, 214–219. [Google Scholar] [CrossRef]
- Sroussi, H.Y.; Epstein, J.B.; Bensadoun, R.J.; Saunders, D.P.; Lalla, R.V.; Migliorati, C.A.; Heaivilin, N.; Zumsteg, Z.S. Common oral complications of head and neck cancer radiation therapy: Mucositis, infections, saliva change, fibrosis, sensory dysfunctions, dental caries, periodontal disease, and osteoradionecrosis. Cancer Med. 2017, 6, 2918–2931. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Wu, Y.; Brouwer, U.; van Vliet, T.; Wang, B.; Demaria, M.; Barazzuol, L.; Coppes, R.P. Cellular senescence contributes to radiation-induced hyposalivation by affecting the stem/progenitor cell niche. Cell Death Dis. 2020, 11, 854. [Google Scholar] [CrossRef] [PubMed]
- Adjemian, S.; Oltean, T.; Martens, S.; Wiernicki, B.; Goossens, V.; Vanden Berghe, T.; Cappe, B.; Ladik, M.; Riquet, F.B.; Heyndrickx, L.; et al. Ionizing radiation results in a mixture of cellular outcomes including mitotic catastrophe, senescence, methuosis, and iron-dependent cell death. Cell Death Dis. 2020, 11, 1003. [Google Scholar] [CrossRef]
- Yamaga, S.; Murao, A.; Ma, G.; Brenner, M.; Aziz, M.; Wang, P. Radiation upregulates macrophage TREM-1 expression to exacerbate injury in mice. Front. Immunol. 2023, 14, 1151250. [Google Scholar] [CrossRef] [PubMed]
- Teresa Pinto, A.; Laranjeiro Pinto, M.; Patrícia Cardoso, A.; Monteiro, C.; Teixeira Pinto, M.; Filipe Maia, A.; Castro, P.; Figueira, R.; Monteiro, A.; Marques, M.; et al. Ionizing radiation modulates human macrophages towards a pro-inflammatory phenotype preserving their pro-invasive and pro-angiogenic capacities. Sci. Rep. 2016, 6, 18765. [Google Scholar] [CrossRef]
- Jung, S.; Kim, J.; Park, J.H.; Kim, K.-Y.; Kim, H.J.; Park, W. A 5-year retrospective cohort study of denosumab induced medication related osteonecrosis of the jaw in osteoporosis patients. Sci. Rep. 2022, 12, 8641. [Google Scholar] [CrossRef]
- Lee, K.; Kim, K.; Kim, J.Y.; Kim, J.W.; Kang, Y.H.; Kim, Y.H.; Kim, S.J. Mechanisms Underlying Medication-Related Osteonecrosis of the Jaw. Oral. Dis. 2025, 31, 1073–1083. [Google Scholar] [CrossRef]
- He, L.; Sun, X.; Liu, Z.; Qiu, Y.; Niu, Y. Pathogenesis and multidisciplinary management of medication-related osteonecrosis of the jaw. Int. J. Oral. Sci. 2020, 12, 30. [Google Scholar] [CrossRef]
- Anastasilakis, A.D.; Pepe, J.; Napoli, N.; Palermo, A.; Magopoulos, C.; Khan, A.A.; Zillikens, M.C.; Body, J.J. Osteonecrosis of the Jaw and Antiresorptive Agents in Benign and Malignant Diseases: A Critical Review Organized by the ECTS. J. Clin. Endocrinol. Metab. 2022, 107, 1441–1460. [Google Scholar] [CrossRef] [PubMed]
- Su, W.H.; Cheng, M.H.; Lee, W.L.; Tsou, T.S.; Chang, W.H.; Chen, C.S.; Wang, P.H. Nonsteroidal anti-inflammatory drugs for wounds: Pain relief or excessive scar formation? Mediat. Inflamm. 2010, 2010, 413238. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Dipietro, L.A. Factors affecting wound healing. J. Dent. Res. 2010, 89, 219–229. [Google Scholar] [CrossRef]
- Wong, L.S.; Martins-Green, M. Firsthand cigarette smoke alters fibroblast migration and survival: Implications for impaired healing. Wound Repair Regen. 2004, 12, 471–484. [Google Scholar] [CrossRef]
- Silva, H. Tobacco Use and Periodontal Disease-The Role of Microvascular Dysfunction. Biology 2021, 10, 441. [Google Scholar] [CrossRef]
- Amerio, E.; Sparano, F.; Muñoz-Sanz, A.; Valles, C.; Nart, J.; Monje, A. Effects of Smoking on Macrophage Polarization in Peri-Implantitis Lesions. Clin. Oral Implant. Res. 2025, 36, 1017–1029. [Google Scholar] [CrossRef]
- Xiao, P.; Zhang, Y.; Zeng, Y.; Yang, D.; Mo, J.; Zheng, Z.; Wang, J.; Zhang, Y.; Zhou, Z.; Zhong, X.; et al. Impaired angiogenesis in ageing: The central role of the extracellular matrix. J. Transl. Med. 2023, 21, 457. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.A.; Wang, J.; Zinn, C.J.; Learmonth, M.; Lerman, L.O.; Wolfram, J.; Hickson, L.J. Fortifying the Diabetic Kidney Disease Treatment Armamentarium: Multitarget Senotherapeutic and Regenerative Strategies. J. Am. Soc. Nephrol. 2025, 36, 1655–1658. [Google Scholar] [CrossRef]
- Huang, W.; Hickson, L.J.; Eirin, A.; Kirkland, J.L.; Lerman, L.O. Cellular senescence: The good, the bad and the unknown. Nat. Rev. Nephrol. 2022, 18, 611–627. [Google Scholar] [CrossRef]
- Yan, J.; Chen, S.; Yi, Z.; Zhao, R.; Zhu, J.; Ding, S.; Wu, J. The role of p21 in cellular senescence and aging-related diseases. Mol. Cells 2024, 47, 100113. [Google Scholar] [CrossRef] [PubMed]
- Gasek, N.S.; Yan, P.; Zhu, J.; Purushothaman, K.R.; Kim, T.; Wang, L.; Wang, B.; Flynn, W.F.; Sun, M.; Guo, C.; et al. Clearance of p21 highly expressing senescent cells accelerates cutaneous wound healing. Nat. Aging 2025, 5, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Huang, X. Stem cell-based drug delivery strategy for skin regeneration and wound healing: Potential clinical applications. Inflamm. Regen. 2023, 43, 33. [Google Scholar] [CrossRef]
- Huelsboemer, L.; Knoedler, L.; Kochen, A.; Yu, C.T.; Hosseini, H.; Hollmann, K.S.; Choi, A.E.; Stögner, V.A.; Knoedler, S.; Hsia, H.C.; et al. Cellular therapeutics and immunotherapies in wound healing—On the pulse of time? Mil. Med. Res. 2024, 11, 23. [Google Scholar] [CrossRef]
- Sato, Y.; Bando, H.; Di Piazza, M.; Gowing, G.; Herberts, C.; Jackman, S.; Leoni, G.; Libertini, S.; MacLachlan, T.; McBlane, J.W.; et al. Tumorigenicity assessment of cell therapy products: The need for global consensus and points to consider. Cytotherapy 2019, 21, 1095–1111. [Google Scholar] [CrossRef]
- Che Shaffi, S.; Hairuddin, O.N.; Mansor, S.F.; Syafiq, T.M.F.; Yahaya, B.H. Unlocking the Potential of Extracellular Vesicles as the Next Generation Therapy: Challenges and Opportunities. Tissue Eng. Regen. Med. 2024, 21, 513–527. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.; Bao, C.; Chen, Q.; Gao, Q.; Wang, N.; Gao, Q.; Mao, L. Extracellular vesicles (EVs)’ journey in recipient cells: From recognition to cargo release. J. Zhejiang Univ. Sci. B 2024, 25, 633–655. [Google Scholar] [CrossRef]
- Clancy, J.W.; Schmidtmann, M.; D’Souza-Schorey, C. The ins and outs of microvesicles. FASEB BioAdvances 2021, 3, 399–406. [Google Scholar] [CrossRef]
- Lee, Y.J.; Shin, K.J.; Chae, Y.C. Regulation of cargo selection in exosome biogenesis and its biomedical applications in cancer. Exp. Mol. Med. 2024, 56, 877–889. [Google Scholar] [CrossRef]
- Welsh, J.A.; Goberdhan, D.C.I.; O’Driscoll, L.; Buzas, E.I.; Blenkiron, C.; Bussolati, B.; Cai, H.; Di Vizio, D.; Driedonks, T.A.P.; Erdbrügger, U.; et al. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. J. Extracell. Vesicles 2024, 13, e12404. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lan, M.; Chen, Y. Minimal Information for Studies of Extracellular Vesicles (MISEV): Ten-Year Evolution (2014–2023). Pharmaceutics 2024, 16, 1394. [Google Scholar] [CrossRef]
- Saint-Pol, J.; Culot, M. Minimum information for studies of extracellular vesicles (MISEV) as toolbox for rigorous, reproducible and homogeneous studies on extracellular vesicles. Toxicol. Vitr. 2025, 106, 106049. [Google Scholar] [CrossRef]
- Su, X.; Wang, H.; Li, Q.; Chen, Z. Extracellular Vesicles: A Review of Their Therapeutic Potentials, Sources, Biodistribution, and Administration Routes. Int. J. Nanomed. 2025, 20, 3175–3199. [Google Scholar] [CrossRef]
- Abels, E.R.; Breakefield, X.O. Introduction to Extracellular Vesicles: Biogenesis, RNA Cargo Selection, Content, Release, and Uptake. Cell. Mol. Neurobiol. 2016, 36, 301–312. [Google Scholar] [CrossRef]
- Liu, Y.-J.; Wang, C. A review of the regulatory mechanisms of extracellular vesicles-mediated intercellular communication. Cell Commun. Signal. 2023, 21, 77. [Google Scholar] [CrossRef]
- Kwok, Z.H.; Wang, C.; Jin, Y. Extracellular Vesicle Transportation and Uptake by Recipient Cells: A Critical Process to Regulate Human Diseases. Processes 2021, 9, 273. [Google Scholar] [CrossRef]
- Shimizu, Y.; Ntege, E.H.; Inoue, Y.; Matsuura, N.; Sunami, H.; Sowa, Y. Optimizing mesenchymal stem cell extracellular vesicles for chronic wound healing: Bioengineering, standardization, and safety. Regen. Ther. 2024, 26, 260–274. [Google Scholar] [CrossRef] [PubMed]
- Le, M.T.; Dao, H.H.; Nguyen, X.H.; Nguyen, T.V.; Nguyen, T.D.; Than, U.T.T. Extracellular vesicle microRNA cargoes: Candidates for diagnosis and targeted therapy of cutaneous wound healing. Ann. Med. 2025, 57, 2534851. [Google Scholar] [CrossRef] [PubMed]
- Kodam, S.P.; Ullah, M. Diagnostic and Therapeutic Potential of Extracellular Vesicles. Technol. Cancer Res. Treat. 2021, 20, 15330338211041203. [Google Scholar] [CrossRef]
- Cruz, C.G.; Sodawalla, H.M.; Mohanakumar, T.; Bansal, S. Extracellular Vesicles as Biomarkers in Infectious Diseases. Biology 2025, 14, 182. [Google Scholar] [CrossRef]
- Du, S.; Guan, Y.; Xie, A.; Yan, Z.; Gao, S.; Li, W.; Rao, L.; Chen, X.; Chen, T. Extracellular vesicles: A rising star for therapeutics and drug delivery. J. Nanobiotechnol. 2023, 21, 231. [Google Scholar] [CrossRef]
- Lu, S.; Lu, L.; Liu, Y.; Li, Z.; Fang, Y.; Chen, Z.; Zhou, J. Native and engineered extracellular vesicles for wound healing. Front. Bioeng. Biotechnol. 2022, 10, 1053217. [Google Scholar] [CrossRef]
- Li, R.; Wang, H.; Wang, X.; Yang, Y.; Zhong, K.; Zhang, X.; Li, H. MSC-EVs and UCB-EVs promote skin wound healing and spatial transcriptome analysis. Sci. Rep. 2025, 15, 4006. [Google Scholar] [CrossRef]
- Roefs, M.T.; Sluijter, J.P.G.; Vader, P. Extracellular Vesicle-Associated Proteins in Tissue Repair. Trends Cell Biol. 2020, 30, 990–1013. [Google Scholar] [CrossRef] [PubMed]
- Konoshenko, M.Y.; Lekchnov, E.A.; Vlassov, A.V.; Laktionov, P.P. Isolation of Extracellular Vesicles: General Methodologies and Latest Trends. BioMed Res. Int. 2018, 2018, 8545347. [Google Scholar] [CrossRef] [PubMed]
- Meggiolaro, A.; Moccia, V.; Brun, P.; Pierno, M.; Mistura, G.; Zappulli, V.; Ferraro, D. Microfluidic Strategies for Extracellular Vesicle Isolation: Towards Clinical Applications. Biosensors 2022, 13, 50. [Google Scholar] [CrossRef]
- Jia, Y.; Yu, L.; Ma, T.; Xu, W.; Qian, H.; Sun, Y.; Shi, H. Small extracellular vesicles isolation and separation: Current techniques, pending questions and clinical applications. Theranostics 2022, 12, 6548–6575. [Google Scholar] [CrossRef] [PubMed]
- Ansari, F.J.; Tafti, H.A.; Amanzadeh, A.; Rabbani, S.; Shokrgozar, M.A.; Heidari, R.; Behroozi, J.; Eyni, H.; Uversky, V.N.; Ghanbari, H. Comparison of the efficiency of ultrafiltration, precipitation, and ultracentrifugation methods for exosome isolation. Biochem. Biophys. Rep. 2024, 38, 101668. [Google Scholar] [CrossRef]
- Visan, K.S.; Lobb, R.J.; Ham, S.; Lima, L.G.; Palma, C.; Edna, C.P.Z.; Wu, L.Y.; Gowda, H.; Datta, K.K.; Hartel, G.; et al. Comparative analysis of tangential flow filtration and ultracentrifugation, both combined with subsequent size exclusion chromatography, for the isolation of small extracellular vesicles. J. Extracell. Vesicles 2022, 11, e12266. [Google Scholar] [CrossRef] [PubMed]
- Brennan, K.; Martin, K.; FitzGerald, S.P.; O’Sullivan, J.; Wu, Y.; Blanco, A.; Richardson, C.; Mc Gee, M.M. A comparison of methods for the isolation and separation of extracellular vesicles from protein and lipid particles in human serum. Sci. Rep. 2020, 10, 1039. [Google Scholar] [CrossRef] [PubMed]
- Khanabdali, R.; Mandrekar, M.; Grygiel, R.; Vo, P.A.; Palma, C.; Nikseresht, S.; Barton, S.; Shojaee, M.; Bhuiyan, S.; Asari, K.; et al. High-throughput surface epitope immunoaffinity isolation of extracellular vesicles and downstream analysis. Biol. Methods Protoc. 2024, 9, bpae032. [Google Scholar] [CrossRef]
- Hassanzadeh-Barforoushi, A.; Sango, X.; Johnston, E.L.; Haylock, D.; Wang, Y. Microfluidic Devices for Manufacture of Therapeutic Extracellular Vesicles: Advances and Opportunities. J. Extracell. Vesicles 2025, 14, e70132. [Google Scholar] [CrossRef] [PubMed]
- Cieślik, M.; Nazimek, K.; Bryniarski, K. Extracellular Vesicles-Oral Therapeutics of the Future. Int. J. Mol. Sci. 2022, 23, 7554. [Google Scholar] [CrossRef]
- Kim, H.; Kim, D.E.; Han, G.; Lim, N.R.; Kim, E.H.; Jang, Y.; Cho, H.; Jang, H.; Kim, K.H.; Kim, S.H.; et al. Harnessing the Natural Healing Power of Colostrum: Bovine Milk-Derived Extracellular Vesicles from Colostrum Facilitating the Transition from Inflammation to Tissue Regeneration for Accelerating Cutaneous Wound Healing. Adv. Healthc. Mater. 2022, 11, e2102027. [Google Scholar] [CrossRef]
- Wang, J.; Li, X.; Zhao, X.; Yuan, S.; Dou, H.; Cheng, T.; Huang, T.; Lv, Z.; Tu, Y.; Shi, Y.; et al. Lactobacillus rhamnosus GG-derived extracellular vesicles promote wound healing via miR-21-5p-mediated re-epithelization and angiogenesis. J. Nanobiotechnol. 2024, 22, 644. [Google Scholar] [CrossRef]
- Lin, S.; Lai, D.; Tian, Y.; Lai, F.; Long, M.; Ji, C.; Hao, G. MicroRNA hsa-let-7e-5p in hUC-MSC-EVs alleviates oral mucositis by targeting TAB2. Scand. J. Immunol. 2024, 100, e13399. [Google Scholar] [CrossRef]
- Zhang, Q.; He, P.; Shi, S.; Xu, Q.; Granquist, E.J.; Winkelstein, B.A.; Shanti, R.M.; Le, A.D. Secretome enriched with small extracellular vesicles derived from human gingiva-derived mesenchymal stem cells enhances rat tongue muscle regeneration. J. Nanobiotechnol. 2025, 23, 434. [Google Scholar] [CrossRef]
- Guo, X.; Huang, Z.; Wu, F.; Jiang, W.; Li, Y.; Wang, T.; Tran, S.D.; Lin, Z.; Su, X. Exosomes of human adipose stem cells mitigate irradiation injury to salivary glands by inhibiting epithelial-mesenchymal transition through miR-199a-3p targeting Twist1 and regulating TGFβ1/Smad3 pathway. Theranostics 2025, 15, 1622–1641. [Google Scholar] [CrossRef]
- Li, D.; Liu, Y.; Lyu, X.; Hu, C.; Yan, T.; Yan, J.; Liao, Y.; Chen, X.; Tan, J. Small RNA cargo armed extracellular vesicles alleviate periodontitis in OVX mice via M2 macrophage polarization. Chem. Eng. J. 2022, 435, 134870. [Google Scholar] [CrossRef]
- Oh, S.Y.; Kim, D.Y.; Lee, K.Y.; Ha, D.L.; Kim, T.L.; Kwon, T.G.; Kim, J.W.; Lee, H.J.; Choi, S.Y.; Hong, S.H. Streptococcus mutans-derived extracellular vesicles promote skin wound healing via tRNA cargo. J. Nanobiotechnol. 2025, 23, 322. [Google Scholar] [CrossRef]
- Antich-Rosselló, M.; Munar-Bestard, M.; Forteza-Genestra, M.A.; Calvo, J.; Gayà, A.; Monjo, M.; Ramis, J.M. Evaluation of Platelet-Derived Extracellular Vesicles in Gingival Fibroblasts and Keratinocytes for Periodontal Applications. Int. J. Mol. Sci. 2022, 23, 7668. [Google Scholar] [CrossRef] [PubMed]
- Qiao, X.; Tang, J.; Dou, L.; Yang, S.; Sun, Y.; Mao, H.; Yang, D. Dental Pulp Stem Cell-Derived Exosomes Regulate Anti-Inflammatory and Osteogenesis in Periodontal Ligament Stem Cells and Promote the Repair of Experimental Periodontitis in Rats. Int. J. Nanomed. 2023, 18, 4683–4703. [Google Scholar] [CrossRef]
- Jo, H.; Brito, S.; Kwak, B.M.; Park, S.; Lee, M.G.; Bin, B.H. Applications of Mesenchymal Stem Cells in Skin Regeneration and Rejuvenation. Int. J. Mol. Sci. 2021, 22, 2410. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zou, Y.; Sun, H.; He, Y.; Ye, K.; Li, Y.; Qiu, L.; Mai, Y.; Chen, X.; Mao, Z.; et al. Engineered Enucleated Mesenchymal Stem Cells Regulating Immune Microenvironment and Promoting Wound Healing. Adv. Mater. 2024, 36, e2412253. [Google Scholar] [CrossRef] [PubMed]
- Mazini, L.; Rochette, L.; Admou, B.; Amal, S.; Malka, G. Hopes and Limits of Adipose-Derived Stem Cells (ADSCs) and Mesenchymal Stem Cells (MSCs) in Wound Healing. Int. J. Mol. Sci. 2020, 21, 1306. [Google Scholar] [CrossRef]
- Jankovic, M.G.; Stojkovic, M.; Bojic, S.; Jovicic, N.; Kovacevic, M.M.; Ivosevic, Z.; Juskovic, A.; Kovacevic, V.; Ljujic, B. Scaling up human mesenchymal stem cell manufacturing using bioreactors for clinical uses. Curr. Res. Transl. Med. 2023, 71, 103393. [Google Scholar] [CrossRef]
- Giebel, B. A milestone for the therapeutic EV field: FDA approves Ryoncil, an allogeneic bone marrow-derived mesenchymal stromal cell therapy. Extracell. Vesicles Circ. Nucl. Acids 2025, 6, 183–190. [Google Scholar] [CrossRef]
- Kou, M.; Huang, L.; Yang, J.; Chiang, Z.; Chen, S.; Liu, J.; Guo, L.; Zhang, X.; Zhou, X.; Xu, X.; et al. Mesenchymal stem cell-derived extracellular vesicles for immunomodulation and regeneration: A next generation therapeutic tool? Cell Death Dis. 2022, 13, 580. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.; Jung, Y.; Seo, J.; Bae, Y.; Kim, H.S.; Jeong, W. Roles of extracellular vesicles from mesenchymal stem cells in regeneration. Mol. Cells 2024, 47, 100151. [Google Scholar] [CrossRef]
- Merino-González, C.; Zuñiga, F.A.; Escudero, C.; Ormazabal, V.; Reyes, C.; Nova-Lamperti, E.; Salomón, C.; Aguayo, C. Mesenchymal Stem Cell-Derived Extracellular Vesicles Promote Angiogenesis: Potencial Clinical Application. Front. Physiol. 2016, 7, 24. [Google Scholar] [CrossRef]
- Łabędź-Masłowska, A.; Vergori, L.; Kędracka-Krok, S.; Karnas, E.; Bobis-Wozowicz, S.; Sekuła-Stryjewska, M.; Sarna, M.; Andriantsitohaina, R.; Zuba-Surma, E.K. Mesenchymal stem cell-derived extracellular vesicles exert pro-angiogenic and pro-lymphangiogenic effects in ischemic tissues by transferring various microRNAs and proteins including ITGa5 and NRP1. J. Nanobiotechnol. 2024, 22, 60. [Google Scholar] [CrossRef]
- Kim, J.; Kim, E.H.; Lee, H.; Sung, J.H.; Bang, O.Y. Clinical-Scale Mesenchymal Stem Cell-Derived Extracellular Vesicle Therapy for Wound Healing. Int. J. Mol. Sci. 2023, 24, 4273. [Google Scholar] [CrossRef]
- Kanayama, A.; Seth, R.B.; Sun, L.; Ea, C.K.; Hong, M.; Shaito, A.; Chiu, Y.H.; Deng, L.; Chen, Z.J. TAB2 and TAB3 activate the NF-kappaB pathway through binding to polyubiquitin chains. Mol. Cell 2004, 15, 535–548. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.Z.; Su, W.R.; Shi, S.H.; Wilder-Smith, P.; Xiang, A.P.; Wong, A.; Nguyen, A.L.; Kwon, C.W.; Le, A.D. Human gingiva-derived mesenchymal stem cells elicit polarization of m2 macrophages and enhance cutaneous wound healing. Stem Cells 2010, 28, 1856–1868. [Google Scholar] [CrossRef]
- Yunna, C.; Mengru, H.; Lei, W.; Weidong, C. Macrophage M1/M2 polarization. Eur. J. Pharmacol. 2020, 877, 173090. [Google Scholar] [CrossRef] [PubMed]
- Bashir, S.; Sharma, Y.; Elahi, A.; Khan, F. Macrophage polarization: The link between inflammation and related diseases. Inflamm. Res. 2016, 65, 1–11. [Google Scholar] [CrossRef]
- Kuninaka, Y.; Ishida, Y.; Ishigami, A.; Nosaka, M.; Matsuki, J.; Yasuda, H.; Kofuna, A.; Kimura, A.; Furukawa, F.; Kondo, T. Macrophage polarity and wound age determination. Sci. Rep. 2022, 12, 20327. [Google Scholar] [CrossRef]
- Lyu, L.; Cai, Y.; Zhang, G.; Jing, Z.; Liang, J.; Zhang, R.; Dang, X.; Zhang, C. Exosomes derived from M2 macrophages induce angiogenesis to promote wound healing. Front. Mol. Biosci. 2022, 9, 1008802. [Google Scholar] [CrossRef]
- Luo, G.; Zhou, Z.; Cao, Z.; Huang, C.; Li, C.; Li, X.; Deng, C.; Wu, P.; Yang, Z.; Tang, J.; et al. M2 macrophage-derived exosomes induce angiogenesis and increase skin flap survival through HIF1AN/HIF-1α/VEGFA control. Arch. Biochem. Biophys. 2024, 751, 109822. [Google Scholar] [CrossRef]
- Zeng, J.; Sun, Z.; Zeng, F.; Gu, C.; Chen, X. M2 macrophage-derived exosome-encapsulated microneedles with mild photothermal therapy for accelerated diabetic wound healing. Mater. Today Bio 2023, 20, 100649. [Google Scholar] [CrossRef] [PubMed]
- Deng, F.; Yan, J.; Lu, J.; Luo, M.; Xia, P.; Liu, S.; Wang, X.; Zhi, F.; Liu, D. M2 Macrophage-Derived Exosomal miR-590-3p Attenuates DSS-Induced Mucosal Damage and Promotes Epithelial Repair via the LATS1/YAP/ β-Catenin Signalling Axis. J. Crohns Colitis 2021, 15, 665–677. [Google Scholar] [CrossRef] [PubMed]
- Tracy, L.E.; Minasian, R.A.; Caterson, E.J. Extracellular Matrix and Dermal Fibroblast Function in the Healing Wound. Adv. Wound Care 2016, 5, 119–136. [Google Scholar] [CrossRef] [PubMed]
- Galbiati, M.; Maiullari, F.; Ceraolo, M.G.; Bousselmi, S.; Fratini, N.; Gega, K.; Recchia, S.; Ferretti, A.M.; Scala, G.; Costantini, M.; et al. Bioactive Hydrogel Supplemented with Stromal Cell-Derived Extracellular Vesicles Enhance Wound Healing. Pharmaceutics 2025, 17, 162. [Google Scholar] [CrossRef]
- Liu, M.; Ye, G.; Li, R.; Gao, T.; Zhuang, C.; Huang, S.; Wang, S.; Hu, J.; Xiang, A.P.; Jiang, M.H. Fibroblast-Derived Extracellular Vesicles Ameliorate the Skin Injury Microenvironment to Promote Wound Healing. Cell Biol. Int. 2025, 49, 1425–1440. [Google Scholar] [CrossRef]
- Ahangar, P.; Mills, S.J.; Smith, L.E.; Gronthos, S.; Cowin, A.J. Human gingival fibroblast secretome accelerates wound healing through anti-inflammatory and pro-angiogenic mechanisms. npj Regen. Med. 2020, 5, 24. [Google Scholar] [CrossRef]
- Sadeghi Moghaddam Bijari, A.; Alijanianzadeh, M.; Keshmiri Neghab, H.; Soheilifar, M.H. A Mini-Review on Fibroblast-Derived Exosomes as Wound Healing Stimulators. Int. J. Hematol. Oncol. Stem Cell Res. 2025, 19, 237–247. [Google Scholar] [CrossRef]
- Fadl, A.; Leask, A. Hiding in Plain Sight: Human Gingival Fibroblasts as an Essential, Yet Overlooked, Tool in Regenerative Medicine. Cells 2023, 12, 2021. [Google Scholar] [CrossRef]
- Han, P.; Li, X.; Wei, W.; Ivanovski, S. Saliva Diagnosis Using Small Extracellular Vesicles and Salivaomics. Methods Mol. Biol. 2023, 2588, 25–39. [Google Scholar] [CrossRef]
- Wu, J.; Liu, G.; Jia, R.; Guo, J. Salivary Extracellular Vesicles: Biomarkers and Beyond in Human Diseases. Int. J. Mol. Sci. 2023, 24, 17328. [Google Scholar] [CrossRef]
- Cui, L.; Zheng, J.; Lu, Y.; Lin, P.; Lin, Y.; Zheng, Y.; Xu, R.; Mai, Z.; Guo, B.; Zhao, X. New frontiers in salivary extracellular vesicles: Transforming diagnostics, monitoring, and therapeutics in oral and systemic diseases. J. Nanobiotechnol. 2024, 22, 171. [Google Scholar] [CrossRef]
- Waasdorp, M. How does saliva contribute to wound healing? Ned. Tijdschr. Tandheelkd. 2022, 129, 275–278. [Google Scholar] [CrossRef]
- Qian, J.; Lu, E.; Xiang, H.; Ding, P.; Wang, Z.; Lin, Z.; Pan, B.; Zhang, C.; Zhao, Z. GelMA loaded with exosomes from human minor salivary gland organoids enhances wound healing by inducing macrophage polarization. J. Nanobiotechnol. 2024, 22, 550. [Google Scholar] [CrossRef]
- Mi, B.; Chen, L.; Xiong, Y.; Yan, C.; Xue, H.; Panayi, A.C.; Liu, J.; Hu, L.; Hu, Y.; Cao, F.; et al. Saliva exosomes-derived UBE2O mRNA promotes angiogenesis in cutaneous wounds by targeting SMAD6. J. Nanobiotechnol. 2020, 18, 68. [Google Scholar] [CrossRef]
- Song, S.; Xiang, R.; Chen, S.; Wu, J.; Chen, W.; Li, X. Saliva-derived exosomes regulate fibroblast metabolic reprogramming in skin wound healing. Front. Cell Dev. Biol. 2025, 13, 1606716. [Google Scholar] [CrossRef]
- George, C.T.; Kurien, B.T.; Scofield, R.H. The Potential Utility of Salivary and Tear Proteomics to Discriminate Sjögren’s Disease from Non-Sjögren’s Sicca. Int. J. Mol. Sci. 2023, 24, 17497. [Google Scholar] [CrossRef] [PubMed]
- Chopra, P.; Fatima, A.; Mohapatra, S.; Murugaiyan, K.; Vemuganti, G.K.; Rengan, A.K.; Watson, S.L.; Singh, V.; Basu, S.; Singh, S. Extracellular vesicles in dry eye disease and Sjögren’s syndrome: A systematic review on their diagnostic and therapeutic role. Surv. Ophthalmol. 2025, 70, 499–515. [Google Scholar] [CrossRef] [PubMed]
- Marsh, S.R.; Amin, M.R.; Toldo, S.; Beard, C.; Dogan, A.B.; Mezzaroma, E.; Andres, E.; Stout, R.F.; Bannon, M.S.; Payne, L.B.; et al. Orally Delivered Milk-Derived Nanovesicles Loaded with Connexin 43 Peptides for Targeted Cardiac Ischemia-Reperfusion Therapy. bioRxiv 2025. [Google Scholar] [CrossRef]
- Prasadani, M.; Kodithuwakku, S.; Pennarossa, G.; Fazeli, A.; Brevini, T.A.L. Therapeutic Potential of Bovine Milk-Derived Extracellular Vesicles. Int. J. Mol. Sci. 2024, 25, 5543. [Google Scholar] [CrossRef]
- Ruan, J.; Xia, Y.; Ma, Y.; Xu, X.; Luo, S.; Yi, J.; Wu, B.; Chen, R.; Wang, H.; Yu, H.; et al. Milk-derived exosomes as functional nanocarriers in wound healing: Mechanisms, applications, and future directions. Mater. Today Bio 2025, 32, 101715. [Google Scholar] [CrossRef]
- Li, X.; Le, Y.; Zhang, Z.; Nian, X.; Liu, B.; Yang, X. Viral Vector-Based Gene Therapy. Int. J. Mol. Sci. 2023, 24, 7736. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.; Yan, R.; Li, A.; Zhang, Y.; Li, J.; Du, H.; Chen, B.; Wei, W.; Zhang, Y.; Sumners, C.; et al. Lentiviral Vectors Mediate Long-Term and High Efficiency Transgene Expression in HEK 293T cells. Int. J. Med. Sci. 2015, 12, 407–415. [Google Scholar] [CrossRef]
- Johnson, K.E.; Wilgus, T.A. Vascular Endothelial Growth Factor and Angiogenesis in the Regulation of Cutaneous Wound Repair. Adv. Wound Care 2014, 3, 647–661. [Google Scholar] [CrossRef]
- Hosgood, G. Wound healing. The role of platelet-derived growth factor and transforming growth factor beta. Vet. Surg. 1993, 22, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Nugent, M.A.; Iozzo, R.V. Fibroblast growth factor-2. Int. J. Biochem. Cell Biol. 2000, 32, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-H.; Gessler, D.J.; Zhan, W.; Gallagher, T.L.; Gao, G. Adeno-associated virus as a delivery vector for gene therapy of human diseases. Signal Transduct. Target. Ther. 2024, 9, 78. [Google Scholar] [CrossRef]
- Xiu, C.; Zheng, H.; Jiang, M.; Li, J.; Zhou, Y.; Mu, L.; Liu, W. MSCs-Derived miR-150-5p-Expressing Exosomes Promote Skin Wound Healing by Activating PI3K/AKT Pathway through PTEN. Int. J. Stem Cells 2022, 15, 359–371. [Google Scholar] [CrossRef]
- Garcia, J.P.; Avila, F.R.; Torres, R.A.; Maita, K.C.; Eldaly, A.S.; Rinker, B.D.; Zubair, A.C.; Forte, A.J.; Sarabia-Estrada, R. Hypoxia-preconditioning of human adipose-derived stem cells enhances cellular proliferation and angiogenesis: A systematic review. J. Clin. Transl. Res. 2022, 8, 61–70. [Google Scholar] [PubMed]
- Gunton, J.E. Hypoxia-inducible factors and diabetes. J. Clin. Investig. 2020, 130, 5063–5073. [Google Scholar] [CrossRef]
- Sung, D.K.; Sung, S.I.; Ahn, S.Y.; Chang, Y.S.; Park, W.S. Thrombin Preconditioning Boosts Biogenesis of Extracellular Vesicles from Mesenchymal Stem Cells and Enriches Their Cargo Contents via Protease-Activated Receptor-Mediated Signaling Pathways. Int. J. Mol. Sci. 2019, 20, 2899. [Google Scholar] [CrossRef]
- Sung, D.K.; Chang, Y.S.; Sung, S.I.; Ahn, S.Y.; Park, W.S. Thrombin Preconditioning of Extracellular Vesicles Derived from Mesenchymal Stem Cells Accelerates Cutaneous Wound Healing by Boosting Their Biogenesis and Enriching Cargo Content. J. Clin. Med. 2019, 8, 533. [Google Scholar] [CrossRef]
- Shah, D.C.; DeVeaux, S.D.A.; Zhang, H.; Liu, A.Y.; Adedipe, T.A.; Chiappa, N.F.; Patel, K.A.; Jang, Y.C.; Goudy, S.L.; Sulchek, T.; et al. Exogenous Delivery of Sphingomyelinase Mediates Mesenchymal Stromal Cell-Extracellular Vesicle Biogenesis, Alters Cargo Sorting, and Therapeutic Potency in vitro. bioRxiv 2025. [Google Scholar] [CrossRef]
- Mukherjee, D.; Paul, D.; Sarker, S.; Hasan, M.N.; Ghosh, R.; Prasad, S.E.; Vemula, P.K.; Das, R.; Adhikary, A.; Pal, S.K.; et al. Polyethylene Glycol-Mediated Fusion of Extracellular Vesicles with Cationic Liposomes for the Design of Hybrid Delivery Systems. ACS Appl. Bio Mater. 2021, 4, 8259–8266. [Google Scholar] [CrossRef]
- Rakshit, T.; Pal, S. Extracellular Vesicles for Drug Delivery and Theranostics In Vivo. JACS Au 2024, 4, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Sutaria, D.S.; Badawi, M.; Phelps, M.A.; Schmittgen, T.D. Achieving the Promise of Therapeutic Extracellular Vesicles: The Devil is in Details of Therapeutic Loading. Pharm. Res. 2017, 34, 1053–1066. [Google Scholar] [CrossRef]
- Liao, Y.; Li, Z.; Zhou, Q.; Sheng, M.; Qu, Q.; Shi, Y.; Yang, J.; Lv, L.; Dai, X.; Shi, X. Saponin surfactants used in drug delivery systems: A new application for natural medicine components. Int. J. Pharm. 2021, 603, 120709. [Google Scholar] [CrossRef]
- Wang, Y.; Cao, Z.; Wei, Q.; Ma, K.; Hu, W.; Huang, Q.; Su, J.; Li, H.; Zhang, C.; Fu, X. VH298-loaded extracellular vesicles released from gelatin methacryloyl hydrogel facilitate diabetic wound healing by HIF-1α-mediated enhancement of angiogenesis. Acta Biomater. 2022, 147, 342–355. [Google Scholar] [CrossRef] [PubMed]
- Lennaárd, A.J.; Mamand, D.R.; Wiklander, R.J.; El Andaloussi, S.; Wiklander, O.P.B. Optimised Electroporation for Loading of Extracellular Vesicles with Doxorubicin. Pharmaceutics 2021, 14, 38. [Google Scholar] [CrossRef] [PubMed]
- Lowe, N.M.; Mizenko, R.R.; Nguyen, B.B.; Chiu, K.L.; Arun, V.; Panitch, A.; Carney, R.P. Orthogonal analysis reveals inconsistencies in cargo loading of extracellular vesicles. J. Extracell. Biol. 2024, 3, e70003. [Google Scholar] [CrossRef]
- Wang, X.; Hu, S.; Zhu, D.; Li, J.; Cheng, K.; Liu, G. Comparison of extruded cell nanovesicles and exosomes in their molecular cargos and regenerative potentials. Nano Res. 2023, 16, 7248–7259. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Huang, Q.; Jiang, Y.; Tang, H.; Zhang, L.; Li, D.; Xu, Y. Emerging technologies for engineering of extracellular vesicles. Front. Bioeng. Biotechnol. 2023, 11, 1298746. [Google Scholar] [CrossRef]
- Cipolla, D.; Wu, H.; Eastman, S.; Redelmeier, T.; Gonda, I.; Chan, H.K. Tuning Ciprofloxacin Release Profiles from Liposomally Encapsulated Nanocrystalline Drug. Pharm. Res. 2016, 33, 2748–2762. [Google Scholar] [CrossRef]
- Liu, Q.; Li, D.; Pan, X.; Liang, Y. Targeted therapy using engineered extracellular vesicles: Principles and strategies for membrane modification. J. Nanobiotechnol. 2023, 21, 334. [Google Scholar] [CrossRef]
- Kooijmans, S.A.A.; Fliervoet, L.A.L.; van der Meel, R.; Fens, M.H.A.M.; Heijnen, H.F.G.; van Bergen en Henegouwen, P.M.P.; Vader, P.; Schiffelers, R.M. PEGylated and targeted extracellular vesicles display enhanced cell specificity and circulation time. J. Control. Release 2016, 224, 77–85. [Google Scholar] [CrossRef]
- Hao, D.; Lu, L.; Song, H.; Duan, Y.; Chen, J.; Carney, R.; Li, J.J.; Zhou, P.; Nolta, J.; Lam, K.S.; et al. Engineered extracellular vesicles with high collagen-binding affinity present superior in situ retention and therapeutic efficacy in tissue repair. Theranostics 2022, 12, 6021–6037. [Google Scholar] [CrossRef]
- Xu, G.; Jin, J.; Fu, Z.; Wang, G.; Lei, X.; Xu, J.; Wang, J. Extracellular vesicle-based drug overview: Research landscape, quality control and nonclinical evaluation strategies. Signal Transduct. Target. Ther. 2025, 10, 255. [Google Scholar] [CrossRef]
- Rai, A.; Claridge, B.; Lozano, J.; Greening, D.W. The Discovery of Extracellular Vesicles and Their Emergence as a Next-Generation Therapy. Circ. Res. 2024, 135, 198–221. [Google Scholar] [CrossRef] [PubMed]
- Eftekhari, Z.; Zohrabi, H.; Oghalaie, A.; Ebrahimi, T.; Shariati, F.S.; Behdani, M.; Kazemi-Lomedasht, F. Advancements and challenges in mRNA and ribonucleoprotein-based therapies: From delivery systems to clinical applications. Mol. Ther. Nucleic Acids 2024, 35, 102313. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Zhang, Q.; Lin, S.; Li, J. Water: The soul of hydrogels. Prog. Mater. Sci. 2025, 148, 101378. [Google Scholar] [CrossRef]
- Carton, F.; Rizzi, M.; Canciani, E.; Sieve, G.; Di Francesco, D.; Casarella, S.; Di Nunno, L.; Boccafoschi, F. Use of Hydrogels in Regenerative Medicine: Focus on Mechanical Properties. Int. J. Mol. Sci. 2024, 25, 11426. [Google Scholar] [CrossRef] [PubMed]
- Chewchuk, S.; Soucy, N.; Wan, F.; Harden, J.; Godin, M. pH controlled release of extracellular vesicles from a hydrogel scaffold for therapeutic applications. Biomed. Mater. 2025, 20, 065006. [Google Scholar] [CrossRef]
- Bashir, S.; Hina, M.; Iqbal, J.; Rajpar, A.H.; Mujtaba, M.A.; Alghamdi, N.A.; Wageh, S.; Ramesh, K.; Ramesh, S. Fundamental Concepts of Hydrogels: Synthesis, Properties, and Their Applications. Polymers 2020, 12, 2702. [Google Scholar] [CrossRef]
- Zhao, L.; Zhou, Y.; Zhang, J.; Liang, H.; Chen, X.; Tan, H. Natural Polymer-Based Hydrogels: From Polymer to Biomedical Applications. Pharmaceutics 2023, 15, 2514. [Google Scholar] [CrossRef]
- Wu, D.T.; Munguia-Lopez, J.G.; Cho, Y.W.; Ma, X.; Song, V.; Zhu, Z.; Tran, S.D. Polymeric Scaffolds for Dental, Oral, and Craniofacial Regenerative Medicine. Molecules 2021, 26, 7043. [Google Scholar] [CrossRef]
- Amengual-Tugores, A.M.; Ráez-Meseguer, C.; Forteza-Genestra, M.A.; Monjo, M.; Ramis, J.M. Extracellular Vesicle-Based Hydrogels for Wound Healing Applications. Int. J. Mol. Sci. 2023, 24, 4104. [Google Scholar] [CrossRef]
- Shi, Q.; Qian, Z.; Liu, D.; Sun, J.; Wang, X.; Liu, H.; Xu, J.; Guo, X. GMSC-Derived Exosomes Combined with a Chitosan/Silk Hydrogel Sponge Accelerates Wound Healing in a Diabetic Rat Skin Defect Model. Front. Physiol. 2017, 8, 904. [Google Scholar] [CrossRef]
- Yao, W.D.; Zhou, J.N.; Tang, C.; Zhang, J.L.; Chen, Z.Y.; Li, Y.; Gong, X.J.; Qu, M.Y.; Zeng, Q.; Jia, Y.L.; et al. Hydrogel Microneedle Patches Loaded with Stem Cell Mitochondria-Enriched Microvesicles Boost the Chronic Wound Healing. ACS Nano 2024, 18, 26733–26750. [Google Scholar] [CrossRef]
- Ma, Y.; Dong, J.; Li, M.; Du, X.; Yan, Z.; Tian, W. An antimicrobial microneedle patch promotes functional healing of infected wounds through controlled release of adipose tissue-derived apoptotic vesicles. J. Nanobiotechnol. 2024, 22, 579. [Google Scholar] [CrossRef]
- He, M.; Wei, Y.; Chen, X.; Li, J.; Kang, J.; Li, B.; Liu, D.; Cai, H. Incorporating Stem Cell-Derived Small Extracellular Vesicles into a Multifunctional OHAMA-EPL Hydrogel Spray for Rapid Healing of Infected Skin Wounds. Adv. Healthc. Mater. 2025, 14, e04147. [Google Scholar] [CrossRef]
- Ma, J.; Sun, X.; Wang, X.; Liu, B.; Shi, K. Factors Affecting Patient Adherence to Inhalation Therapy: An Application of SEIPS Model 2.0. Patient Prefer. Adherence 2023, 17, 531–545. [Google Scholar] [CrossRef]
- Paganini, C.; Capasso Palmiero, U.; Pocsfalvi, G.; Touzet, N.; Bongiovanni, A.; Arosio, P. Scalable Production and Isolation of Extracellular Vesicles: Available Sources and Lessons from Current Industrial Bioprocesses. Biotechnol. J. 2019, 14, e1800528. [Google Scholar] [CrossRef]
- Burbidge, K.; Zwikelmaier, V.; Cook, B.; Long, M.M.; Balva, B.; Lonigro, M.; Ispas, G.; Rademacher, D.J.; Campbell, E.M. Cargo and cell-specific differences in extracellular vesicle populations identified by multiplexed immunofluorescent analysis. J. Extracell. Vesicles 2020, 9, 1789326. [Google Scholar] [CrossRef]
- Silva, T.F.; Hutchins, E.; Zhao, W.; Ciani, Y.; Kim, M.; Mariscal, J.; Qiu, Z.; Kittel, A.; Zhou, B.; Wang, Y.; et al. Extracellular Vesicles heterogeneity through the lens of multiomics. bioRxiv 2024. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.P.; Mardini, O.; Ericsson, M.; Prabhakar, S.; Maguire, C.; Chen, J.W.; Tannous, B.A.; Breakefield, X.O. Dynamic biodistribution of extracellular vesicles in vivo using a multimodal imaging reporter. ACS Nano 2014, 8, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Ahmadian, S.; Jafari, N.; Tamadon, A.; Ghaffarzadeh, A.; Rahbarghazi, R.; Mahdipour, M. Different storage and freezing protocols for extracellular vesicles: A systematic review. Stem Cell Res. Ther. 2024, 15, 453. [Google Scholar] [CrossRef] [PubMed]
- Colceriu-Șimon, I.M.; Feștilă, D.; Emoke, H.; Pancsur, A.; Șimon, M.; Olteanu, C.D.; Păstrav, M.; Bunta, O.; Ghergie, M. The Effects of Non-Steroidal Anti-Inflammatory Drugs Used for Orthodontic Pain Management on Tooth Movement: A Comprehensive Review of the Literature. J. Clin. Med. 2025, 14, 2920. [Google Scholar] [CrossRef]
- Domaszewska-Szostek, A.P.; Krzyżanowska, M.O.; Czarnecka, A.M.; Siemionow, M. Local Treatment of Burns with Cell-Based Therapies Tested in Clinical Studies. J. Clin. Med. 2021, 10, 396. [Google Scholar] [CrossRef] [PubMed]
- Schurr, M.J.; Foster, K.N.; Centanni, J.M.; Comer, A.R.; Wicks, A.; Gibson, A.L.; Thomas-Virnig, C.L.; Schlosser, S.J.; Faucher, L.D.; Lokuta, M.A.; et al. Phase I/II clinical evaluation of StrataGraft: A consistent, pathogen-free human skin substitute. J. Trauma Acute Care Surg. 2009, 66, 866–874. [Google Scholar] [CrossRef]
- Wang, C.K.; Tsai, T.H.; Lee, C.H. Regulation of exosomes as biologic medicines: Regulatory challenges faced in exosome development and manufacturing processes. Clin. Transl. Sci. 2024, 17, e13904. [Google Scholar] [CrossRef] [PubMed]
- Hahm, J.; Kim, J.; Park, J. Strategies to Enhance Extracellular Vesicle Production. Tissue Eng. Regen. Med. 2021, 18, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Trenkenschuh, E.; Richter, M.; Heinrich, E.; Koch, M.; Fuhrmann, G.; Friess, W. Enhancing the Stabilization Potential of Lyophilization for Extracellular Vesicles. Adv. Healthc. Mater. 2022, 11, e2100538. [Google Scholar] [CrossRef] [PubMed]
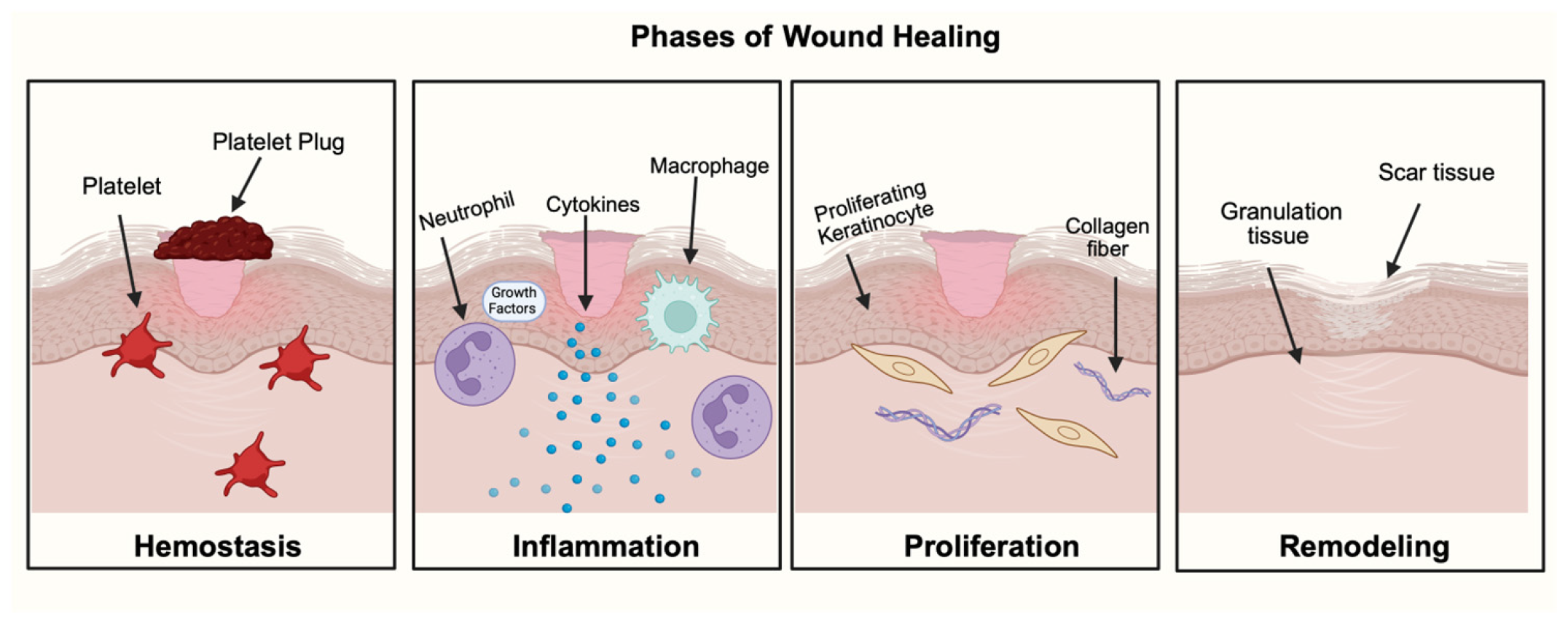
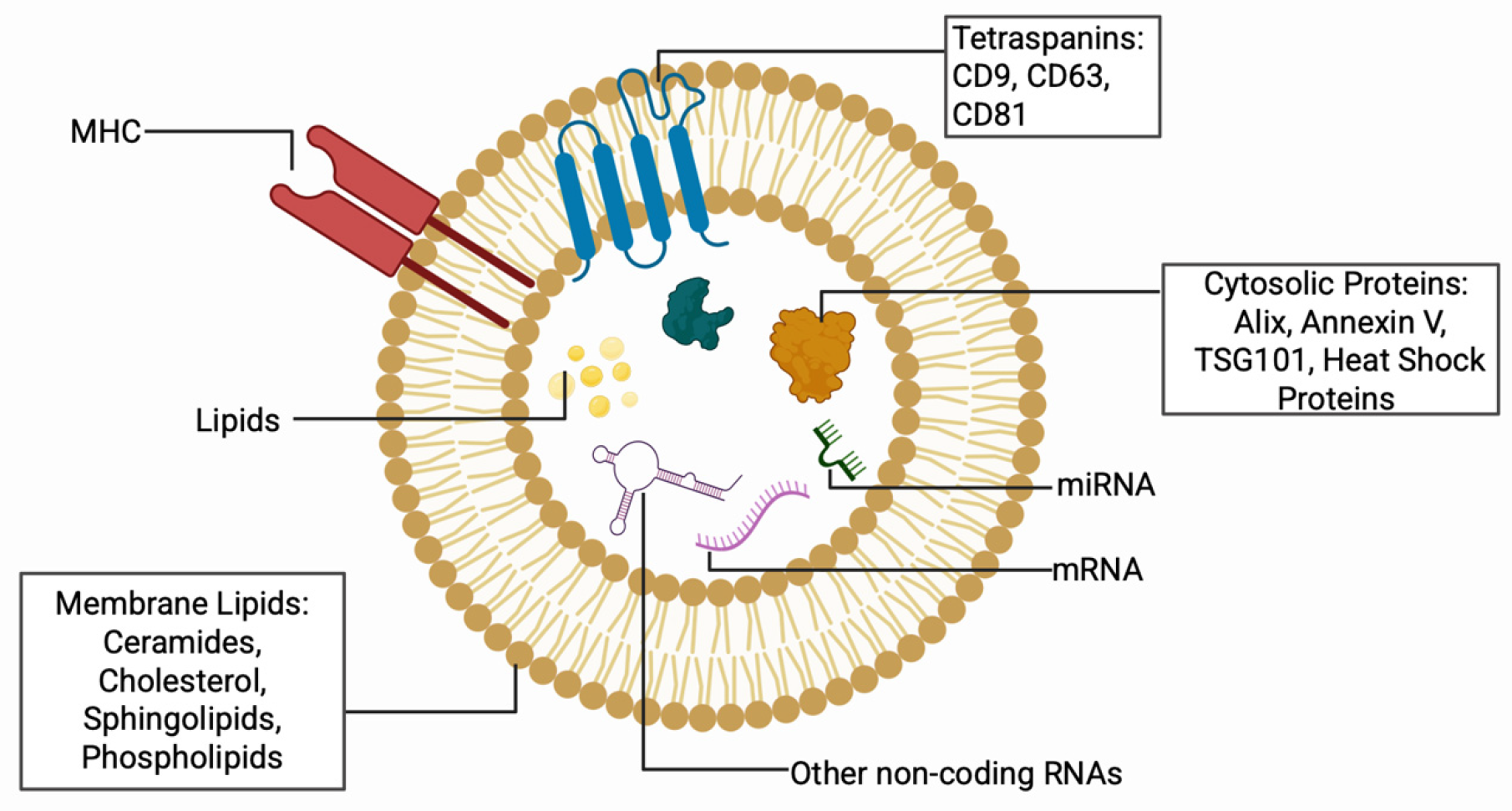
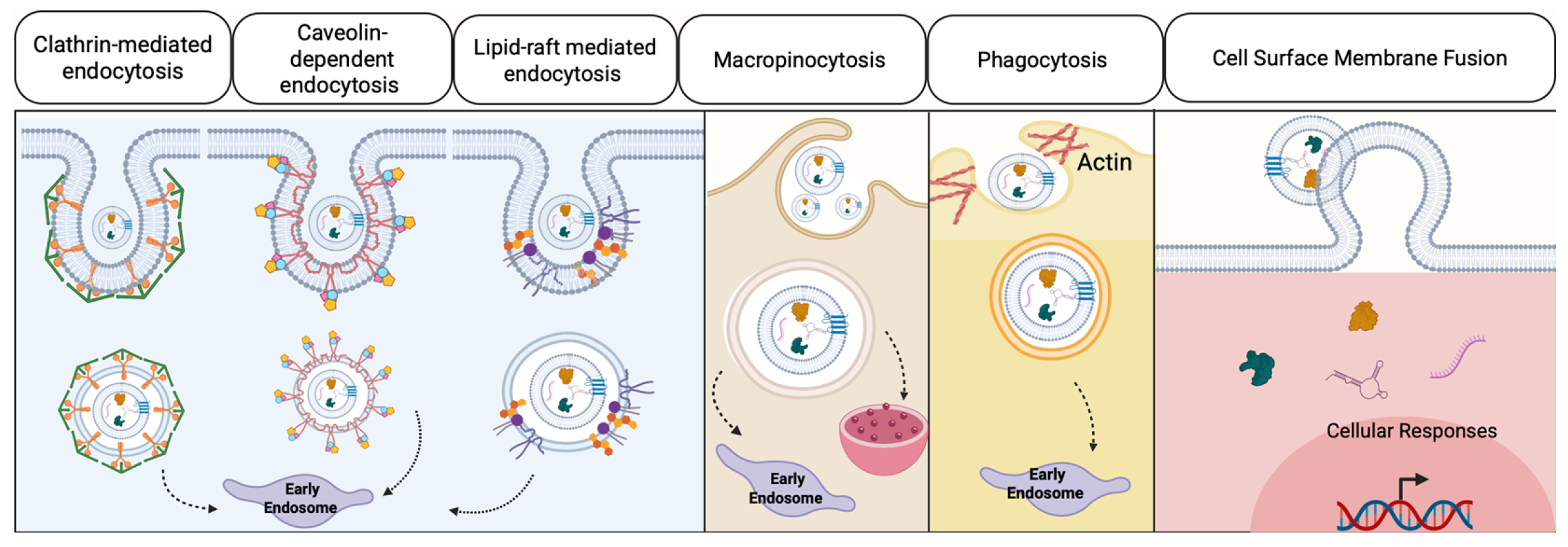

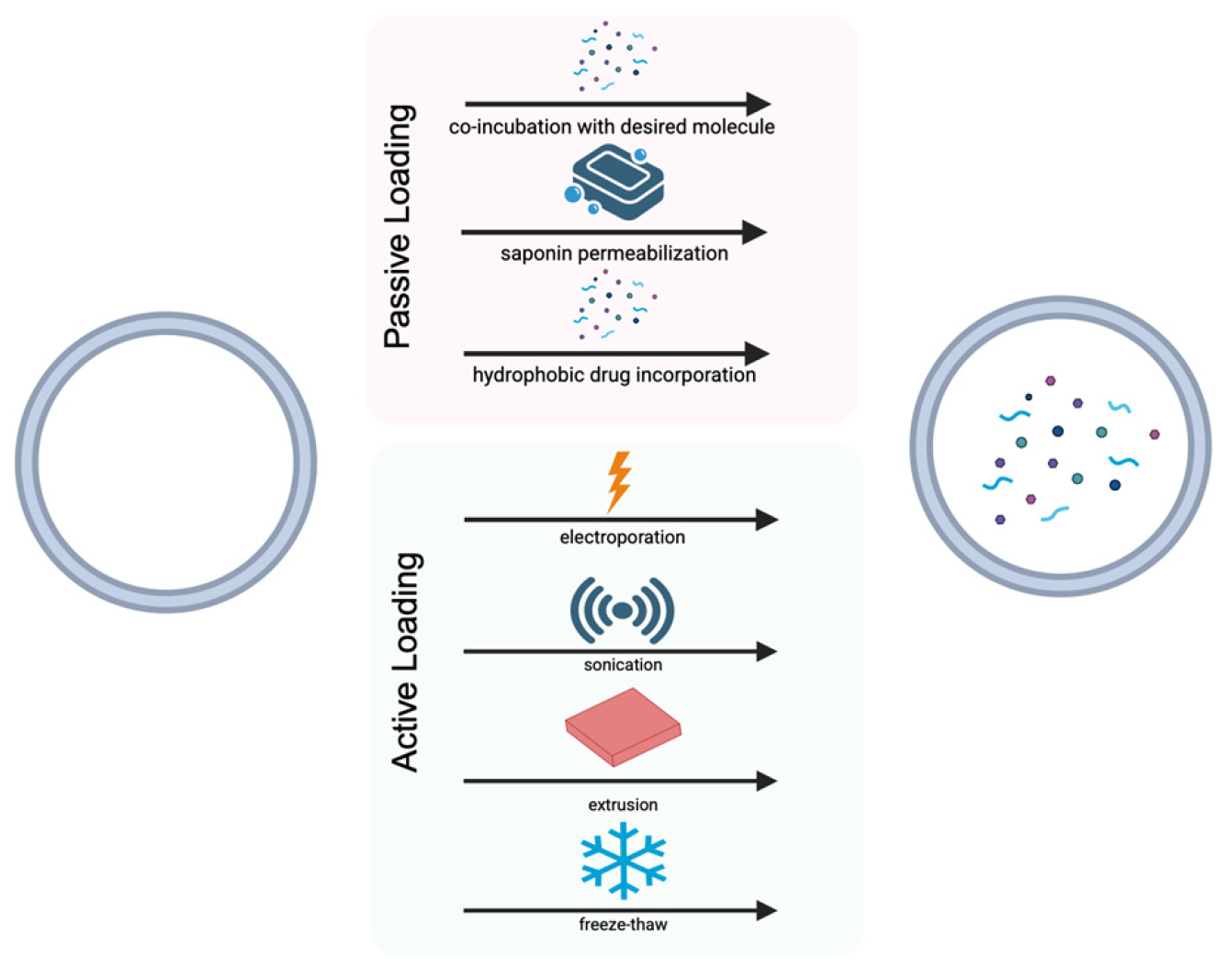
| First Author & Year | Phases | Key Cellular Events | Role of EVs | Molecular Pathway |
|---|---|---|---|---|
| Setua 2022 [28] Owens 2011 [29] Berckmans 2001 [30] Tao 2017 [31] Aatonen 2012 [32] | Hemostasis | Coagulation and initiation of repair | EVs derived from platelet and endothelial cells promote coagulation by providing procoagulant phospholipid surfaces and tissue factor activity. | Enhance thrombin generation and fibrin formation via tissue factor and phosphatidylserine exposure. Promote procoagulant activities and thrombin generation time by facilitating assembly of tenase and prothrombinase complexes. |
| Ding 2023 [33] Sun 2022 [34] Li 2016 [35] Peng 2024 [36] Shen 2021 [37] | Inflammation | Immune cell recruitment, cytokine modulation, and inflammation resolution | EVs derived from immune and stromal modulate cytokine release and promote macrophage M1 to M2 transition. | MSC and oral tissue-derived EVs regulate inflammatory cytokines and the NF-κB activity. Downregulate TLR4 pathway, TNF-α, IL-1β and elevate IL-10 and TGF-β to resolve inflammation. |
| Gao 2022 [38] Shahsavandi 2025 [39] Li 2025 [40] Ju 2023 [41] Zeng 2021 [42] Li 2025 [43] | Proliferation | Epithelial migration, fibroblast activation, angiogenesis/vasculogenesis | EVs transfer miRNAs and growth factors that enhance keratinocyte migration, angiogenesis, and fibroblast proliferation. | MSC-, epithelial, and fibroblast-derived EVs promote angiogenesis via VEGF pathway, PTEN/PI3K/AKT signaling, and NF-kB/MAPK pathway inhibition. Promote fibroblast migration through miR-21, PTEN/AKT pathway. |
| Yuan 2021 [44] Nawaz 2018 [45] Wang 2017 [46] Ahmadpour 2023 [47] Oh 2021 [48] | Remodeling | Extracellular matrix reorganization, scar reduction, tissue regeneration | EVs coordinate fibroblast to myofibroblast differentiation and accelerate wound closure via extracellular matrix and collagen remodeling. | EV miRNAs regulate TGF-β and Smad signaling leading to reduced fibrosis and scarring. EVs are active carriers of matrix-degrading enzymes (MMPS, ADAMs, cathepsins) and regulate extracellular matrix remodeling. |
| Factors | Normal Function | Pathology | Mechanism of Impaired Healing | Examples |
|---|---|---|---|---|
| Oral microbiome | Maintains epithelial turnover and primes immune responses for rapid repair [84,85] | Dysbiosis—imbalance favoring pathogenic species [80]. | Pathogen release virulence factors (e.g., LPS) that activate inflammatory pathways such as NLRP3 inflammasome [81], caspase-1 activation, and proinflammatory cytokine secretion [83]. Aberrant NLRP3 activation disrupts stromal cell function, delays epithelial closure, prolongs inflammation, and increases infection susceptibility. | — |
| Pathogenic bacteria | Balanced microbial community supports healing. | Overgrowth of pathogens causes chronic inflammation and epithelial stress. | Chronic inflammation damages epithelial barrier and delays healing. | P. gingivalis induces EVs carrying TNF-α, IL-1β, and promotes inflammation [86]; E. faecalis EVs trigger M1 macrophage polarization via NOD2/RIPK2 pathway, sustaining inflammation [87]; S. mutans EVs contain virulence proteins that promote biofilm formation and disease progression [88]. |
| Probiotic bacteria | Supports microbial balance and anti-inflammatory effects. | Loss or absence reduces regenerative capacity. | L. reuteri EVs decrease TNF-α, IL-1β, IL-6; increase CD206+ and M2 macrophages, promoting anti-inflammatory healing [79,89]. Restores microbial balance, enhances stem cell activity, and accelerates tissue repair. | — |
| Saliva | Contains growth factors, histatins, and antimicrobial peptides that stimulate keratinocyte migration and protect from infection [90]. | Reduced salivary flow (e.g., radiotherapy, autoimmune disease, medication use). | Xerostomia leads to increased microbial colonization, slower re-epithelialization, and higher infection risk [91]. | In diabetes, salivary dysfunction delays wound closure [92]. |
| First Author & Year | EV Source | Oral Injury | In Vitro Oral Wound Model | In Vivo Oral Wound Model | Main Findings |
|---|---|---|---|---|---|
| Sun 2022 [34] | Human Gingival Mesenchymal Stem Cells | Periodontitis | LPS was used to exacerbate inflammation in periodontal ligament stem cells | — | The human gingival mesenchymal stem cell exosomes dampened the inflammatory response by decreasing the expression of NF-kB and Wnt5a. |
| Gao 2022 [38] | Human Umbilical Mesenchymal Stem Cells | Oral Mucositis | — | Wistar rats (male 6–8 weeks) were treated with glacial acetic acid in the inner mucosa of the lower lipEV | EV treatment group resulted in a reduced immunostaining intensity of NF-kB, IL-6, and TNF-a. |
| Lin 2024 [171] | Human Umbilical Mesenchymal Stem Cells | Oral Mucositis | LPS-induced human oral keratinocytes | Hamster model for Oral mucositis | Human umbilical cord mesenchymal stem cell extracellular vesicles reduced inflammation after human oral keratinocyte exposure to LPS. EV treatment reduced OM phenotype in vivo. Bioinformatic analysis showed the overexpression of has-let-7e-5p in EV. |
| Zhang 2025 [172] | Human Gingiva-Derived Mesenchymal Stem Cell | Tongue Defect | — | Rat Tongue Defect Model | iGMSC-derived secretome applied to the rat tongue defect wound promoted regeneration without fibrosis and shape deformity. Secretome had increased IL-10 and suppressed TNF-a expression following LPS stimulation in macrophages. |
| Guo 2025 [173] | Human Adipose-Derived Stem Cell | Salivary glands injured with 14 Gy | Irradiated submandibular gland epithelial C6 (SMG-CG) cells | Irradiated injured salivary glands C57BL/6 mice | Mice with exosome treatment increased saliva secretion, cell proliferation, and tissue repair genes. miR-199a-3p was increased in mice with exosome treatment and reduced epithelial to mesenchymal transition and inactivated the TGFB1-Smad3p. |
| Li 2022 [174] | Macrophage | Periodontitis | — | Mice that have undergone an ovariectomy surgery with an induction of periodontitis | Therapeutic delivery of miR-30e-5p inhibitor in macrophage extracellular vesicles can treat periodontitis in an estrogen deficiency model. |
| Chen 2024 [89] | Lactobacillus reuteri | Oral Mucosa Wound Healing | — | C57BL/6 mice received a circular ulcer on the tongue | Lactobacillus reuteri extracellular vesicles were uptaken by macrophages shifted their phenotype to M2 by regulating mitochondrial permeability and decreasing oxidative stress. |
| Oh 2025 [175] | Streptococcus mutans | Oral Mucosal Organoids | Human oral tissue including buccal mucosa, tongue, and mandible mucosa were used to create an organoid | — | Streptococcus mutans-derived extracellular vesicles promoted epithelial proliferation and wound healing through the TLR3 mechanism facilitated by tRNA-Met variant. |
| Antich-Rossello 2022 [176] | Platelet | — | Human gingival keratinocyte and fibroblast scratch assay | — | Platelet-derived extracellular vesicles increased wound closure in both gingival keratinocyte and fibroblasts. |
| Qiao 2023 [177] | Dental Pulp Stem Cell | Periodontitis | — | Rat Periodontitis Model | Dental Pulp Stem Cell exosomes prompted healing of the periodontal epithelium by potentially inhibiting the IL-6/JAK2/STAT3 pathway. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Patel, H.A.; Schmiliver, B.; Sekar, K.P.C.; Dogini, M.; Onyeagoro, C.; Shah, D.C.; Robinson, M.H.; Giwa-Otusajo, B.; Wu, D.T.; Goudy, S.L. Harnessing the Therapeutic Potential of Extracellular Vesicles for Oral Wound Healing. Bioengineering 2026, 13, 148. https://doi.org/10.3390/bioengineering13020148
Patel HA, Schmiliver B, Sekar KPC, Dogini M, Onyeagoro C, Shah DC, Robinson MH, Giwa-Otusajo B, Wu DT, Goudy SL. Harnessing the Therapeutic Potential of Extracellular Vesicles for Oral Wound Healing. Bioengineering. 2026; 13(2):148. https://doi.org/10.3390/bioengineering13020148
Chicago/Turabian StylePatel, Helly A., Bianca Schmiliver, Keerthi Priya Chinniyampalayam Sekar, Mirelle Dogini, Chidubem Onyeagoro, Daniel C. Shah, M. Hope Robinson, Babatunde Giwa-Otusajo, David T. Wu, and Steven L. Goudy. 2026. "Harnessing the Therapeutic Potential of Extracellular Vesicles for Oral Wound Healing" Bioengineering 13, no. 2: 148. https://doi.org/10.3390/bioengineering13020148
APA StylePatel, H. A., Schmiliver, B., Sekar, K. P. C., Dogini, M., Onyeagoro, C., Shah, D. C., Robinson, M. H., Giwa-Otusajo, B., Wu, D. T., & Goudy, S. L. (2026). Harnessing the Therapeutic Potential of Extracellular Vesicles for Oral Wound Healing. Bioengineering, 13(2), 148. https://doi.org/10.3390/bioengineering13020148









