The Effects of Ankle Versus Plantar Vibrotactile Orthoses on Joint Position Sense and Postural Control in Individuals with Functional Ankle Instability: A Pilot Randomized Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Trial Design, Setting, and Registration
2.2. Eligibility Criteria
2.3. Interventions
Fabrication of Vibrotactile Orthoses
2.4. Randomization and Blinding
2.5. Outcomes
2.5.1. Joint Position Sense
2.5.2. Static Postural Control
2.5.3. Dynamic Postural Control
2.6. Harms
2.7. Procedure
2.8. Statistical Analysis
3. Results
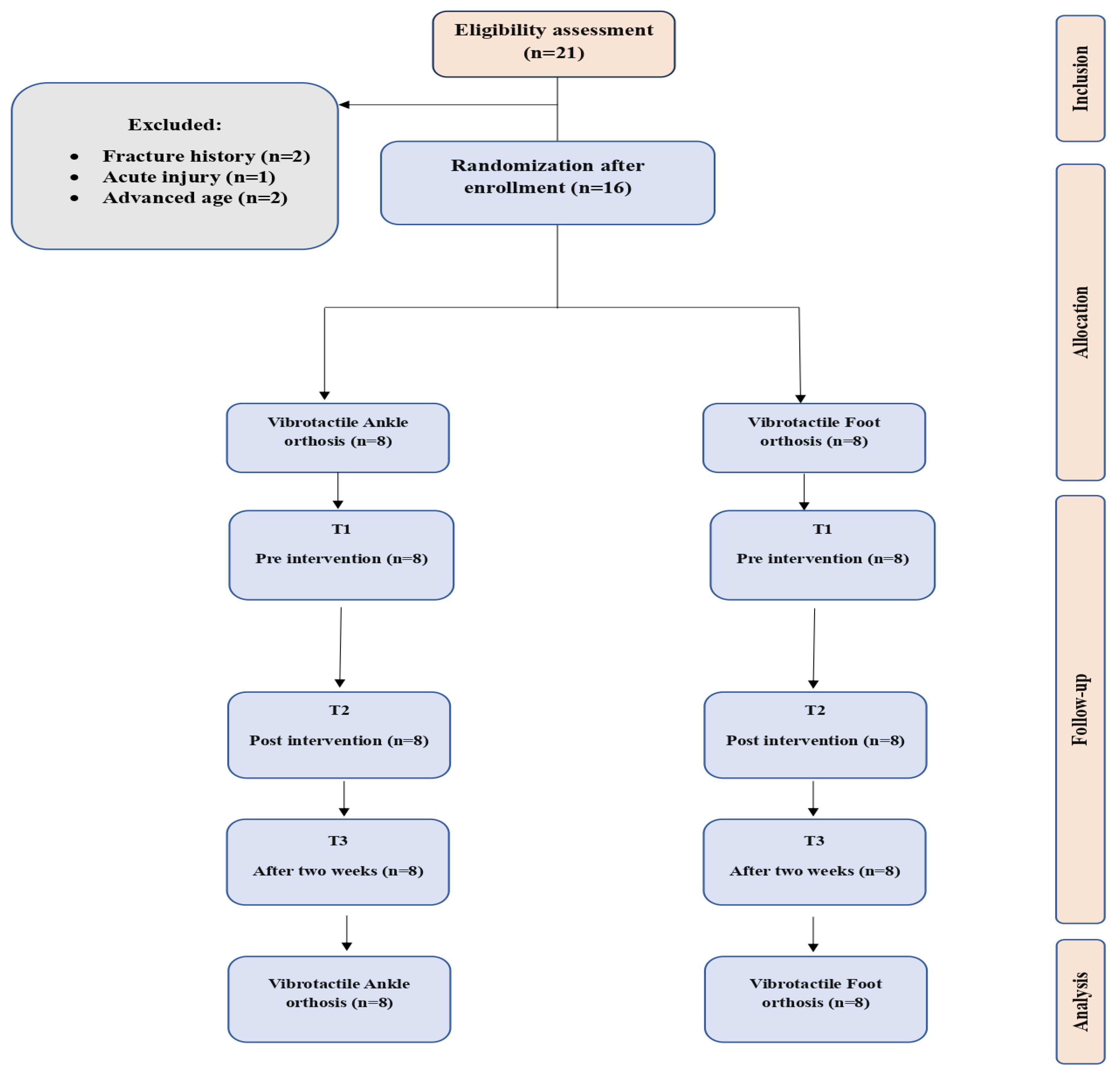
3.1. Participants Flow
3.2. Baseline Data
3.3. Effects of Interventions
3.4. Change in Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| LAS | Lateral ankle sprain |
| FAI | Functional ankle instability |
| JPS | Joint position sense |
| GRF | Ground reaction force |
| CNS | Central nervous system |
| FOs | Foot orthoses |
| AOs | Ankle orthoses |
| VAO | vibrotactile ankle orthoses |
| VFO | vibrotactile foot orthoses |
| PWM | Pulse width modulation |
| EVA | Ethylene-vinyl acetate |
| SEBT | Star Excursion Balance Test |
| SMHT | Six-Meter Hop Test |
| A | Anterior |
| PM | Posteromedial |
| PL | Posterolateral |
| AP | Anteroposterior |
| ML | Mediolateral |
| η2 | partial eta squared |
| CAIT | Cumberland ankle instability tool |
| COP-R | Center of pressure resultant |
| MCID | minimal clinically important difference |
References
- Wang, S.; Qian, Z.; Liu, X.; Song, G.; Jiang, Z.; Wang, K.; Wu, J.; Liu, J.; Ren, L.; Ren, L. Effects of additional weight-bearing on the in vivo kinematics of the human ankle joint complex during walking. Sci. Rep. 2024, 14, 29049. [Google Scholar] [CrossRef]
- Van Royen, A.; Shahabpour, M.; Al Jahed, D.; Abid, W.; Vanhoenacker, F.; De Maeseneer, M. Injuries of the Ligaments and Tendons in Ankle and Foot. In Imaging of Orthopedic Sports Injuries; Springer International Publishing: Cham, Switzerland, 2021; pp. 511–555. [Google Scholar] [CrossRef]
- Fenn, B.P.; Song, J.; Casey, J.; Waryasz, G.R.; DiGiovanni, C.W.; Lubberts, B.; Guss, D. Worldwide epidemiology of foot and ankle injuries during military training: A systematic review. BMJ Mil. Health 2021, 167, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Galasso, A.; Caughman, A.M.; Griffith, A.; Hoch, C.; Rex, J.; Scott, D.J.; Gross, C.E. A detailed analysis of workplace foot and ankle injuries. Foot Ankle Spec. 2024, 19, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Herzog, M.M.; Kerr, Z.Y.; Marshall, S.W.; Wikstrom, E.A. Epidemiology of ankle sprains and chronic ankle instability. J. Athl. Train. 2019, 54, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Munn, J.; Sullivan, S.J.; Schneiders, A.G. Evidence of sensorimotor deficits in functional ankle instability: A systematic review with meta-analysis. J. Sci. Med. Sport. 2010, 13, 2–12. [Google Scholar] [CrossRef]
- Liu, Y.; Dong, S.; Wang, Q.; Liu, Z.; Song, Q.; Shen, P. Deficits in proprioception and strength may contribute to the impaired postural stability among individuals with functional ankle instability. Front. Physiol. 2024, 15, 1342636. [Google Scholar] [CrossRef]
- Hertel, J.; Corbett, R.O. An updated model of chronic ankle instability. J. Athl. Train. 2019, 54, 572–588. [Google Scholar] [CrossRef]
- Liu, N.; Yang, C.; Song, Q.; Yang, F.; Chen, Y. Patients with chronic ankle instability exhibit increased sensorimotor cortex activation and correlation with poorer lateral balance control ability during single-leg stance: A FNIRS study. Front. Hum. Neurosci. 2024, 18, 1366443. [Google Scholar] [CrossRef]
- Hu, X.; Feng, T.; Li, P.; Liao, J.; Wang, L. Bilateral sensorimotor impairments in individuals with unilateral chronic ankle instability: A systematic review and meta-analysis. Sports Med. Open 2024, 10, 33. [Google Scholar] [CrossRef]
- Hoch, M.C.; McKeon, P.O. Peroneal reaction time after ankle sprain: A systematic review and meta-analysis. Med. Sci. Sports Exerc. 2014, 46, 546–556. [Google Scholar] [CrossRef]
- Kim, K.M.; Kim, J.S.; Needle, A.R. Soleus arthrogenic muscle inhibition following acute lateral ankle sprain correlates with symptoms and ankle disability but not with postural control. J. Sport Health Sci. 2024, 13, 559–568. [Google Scholar] [CrossRef]
- Lin, J.Z.; Hung, M.H.; Ko, B.J.; Lee, H.J. Analysing lower limb motion and muscle activation in athletes with ankle instability during dual-task drop-jump. Sports Biomech. 2025, 24, 1899–1913. [Google Scholar] [CrossRef]
- Rittweger, J. (Ed.) Manual of Vibration Exercise and Vibration Therapy; Springer Nature: Cham, Switzerland, 2020; pp. 87–99. [Google Scholar] [CrossRef]
- Proske, U.; Gandevia, S.C. The proprioceptive senses: Their roles in signaling body shape, body position and movement, and muscle force. Physiol. Rev. 2012, 92, 1651–1697. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Gong, Y.; Xu, L.; Wang, L. Influence of short-term plantar–sensory stimulation with different body loadings on postural control and sensory strategies in individuals with chronic ankle instability. Sport Sci. Health 2025, 21, 245–256. [Google Scholar] [CrossRef]
- Grillner, S. The motor infrastructure: From ion channels to neuronal networks. Nat. Rev. Neurosci. 2003, 4, 573–586. [Google Scholar] [CrossRef] [PubMed]
- Garza-Lombó, C.; Pappa, A.; Panayiotidis, M.I.; Franco, R. Redox homeostasis, oxidative stress and mitophagy. Mitochondrion 2020, 51, 105–117. [Google Scholar] [CrossRef]
- Lo, W.T.; Wong, D.P.; Yick, K.L.; Ng, S.P.; Yip, J. Effects of custom-made textile insoles on plantar pressure distribution and lower limb EMG activity during turning. J. Foot Ankle Res. 2016, 9, 22. [Google Scholar] [CrossRef]
- Chen, H.; Sun, D.; Fang, Y.; Gao, S.; Zhang, Q.; Bíró, I.; Tafferner-Gulyás, V.; Gu, Y. Effect of orthopedic insoles on lower limb motion kinematics and kinetics in adults with flat foot: A systematic review. Front. Bioeng. Biotechnol. 2024, 12, 1435554. [Google Scholar] [CrossRef]
- Haris, F.; Liau, B.Y.; Jan, Y.K.; Akbari, V.B.; Primanda, Y.; Lin, K.H.; Lung, C.W. A review of the plantar pressure distribution effects from insole materials and at different walking speeds. Appl. Sci. 2021, 11, 11851. [Google Scholar] [CrossRef]
- Cordova, M.L.; Scott, B.D.; Ingersoll, C.D.; LeBlanc, M.J. Effects of ankle support on lower-extremity functional performance: A meta-analysis. Med. Sci. Sports Exerc. 2005, 37, 635–641. [Google Scholar] [CrossRef]
- Tsikopoulos, K.; Sidiropoulos, K.; Kitridis, D.; Metaxiotis, D.; Ali, A. Do external supports improve dynamic balance in patients with chronic ankle instability? A network meta-analysis. Clin. Orthop. Relat. Res. 2020, 478, 359–377. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Li, J.; Wang, Q.; Liu, D.; Guo, L. Comparative analysis of sensory-motor function and its correlation with gait biomechanics in patients with unilateral chronic ankle instability. J. Orthop. Surg. Res. 2025, 20, 396. [Google Scholar] [CrossRef] [PubMed]
- Rowe, P.L.; Bryant, A.L.; Egerton, T.; Paterson, K.L. External ankle support and ankle biomechanics in chronic ankle instability: Systematic review and meta-analysis. J. Athl. Train. 2023, 58, 635–647. [Google Scholar] [CrossRef] [PubMed]
- Hopewell, S.; Chan, A.W.; Collins, G.S.; Hróbjartsson, A.; Moher, D.; Schulz, K.F.; Tunn, R.; Aggarwal, R.; Berkwits, M.; Berlin, J.A.; et al. CONSORT 2025 statement: Updated guideline for reporting randomised trials. Lancet 2025, 405, 1633–1640. [Google Scholar] [CrossRef]
- Gribble, P.A.; Delahunt, E.; Bleakley, C.; Caulfield, B.; Docherty, C.; Fourchet, F.; Fong, D.; Hertel, J.; Hiller, C.; Kaminski, T.; et al. Selection criteria for patients with chronic ankle instability in controlled research: A position statement of the International Ankle Consortium. J. Orthop. Sports Phys. Ther. 2013, 43, 585–591. Available online: https://www.jospt.org/doi/10.2519/jospt.2013.0303 (accessed on 20 January 2026). [CrossRef]
- Attenborough, A.S.; Hiller, C.E.; Smith, R.M.; Stuelcken, M.; Greene, A.; Sinclair, P.J. Chronic ankle instability in sporting populations. Sports Med. 2014, 44, 1545–1556. [Google Scholar] [CrossRef]
- Ghazi, M.; Rippetoe, J.; Chandrashekhar, R.; Wang, H. Focal vibration therapy: Vibration parameters of effective wearable devices. Appl. Sci. 2021, 11, 2969. [Google Scholar] [CrossRef]
- Kynsburg, A.; Halasi, T.; Tallay, A.; Berkes, I. Changes in joint position sense after conservatively treated chronic lateral ankle instability. Knee Surg. Sports Traumatol. Arthrosc. 2006, 14, 1299–1306. [Google Scholar] [CrossRef]
- Asgari, N.; Yeowell, G.; Sadeghi-Demneh, E. A comparison of the efficacy of textured insoles on balance performance in older people with versus without plantar callosities. Gait Posture 2022, 94, 217–221. [Google Scholar] [CrossRef]
- Kawaguchi, K.; Taketomi, S.; Mizutani, Y.; Inui, H.; Yamagami, R.; Kono, K.; Kage, T.; Takei, S.; Fujiwara, S.; Ogata, T.; et al. Dynamic postural stability is decreased during the single-leg drop landing task in male collegiate soccer players with chronic ankle instability. Orthop. J. Sports Med. 2022, 10, 23259671221107343. [Google Scholar] [CrossRef]
- Lin, J.Z.; Lin, Y.A.; Tai, W.H.; Chen, C.Y. Influence of Landing in Neuromuscular Control and Ground Reaction Force with Ankle Instability: A Narrative Review. Bioengineering 2022, 9, 68. [Google Scholar] [CrossRef] [PubMed]
- Xue, X.A.; Wang, Y.; Xu, X.; Li, H.; Li, Q.; Na, Y.; Tao, W.; Yu, L.; Jin, Z.; Li, H.; et al. Postural control deficits during static single-leg stance in chronic ankle instability: A systematic review and meta-analysis. Sports Health 2024, 16, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Fereshtenejad, N.; Saberi, S.; Pol, F.; Yeowell, G.; Sadeghi-Demneh, E. Intersession reliability of center of pressure measurement during bipedal standing with different foot placement angles. J. Bodyw. Mov. Ther. 2024, 39, 410–414. [Google Scholar] [CrossRef] [PubMed]
- Powden, C.J.; Dodds, T.K.; Gabriel, E.H. The reliability of the star excursion balance test and lower quarter y-balance test in healthy adults: A systematic review. Int. J. Sports Phys. Ther. 2019, 14, 683. Available online: https://pmc.ncbi.nlm.nih.gov/articles/PMC6769278/ (accessed on 20 January 2026).
- Ko, J.; Wikstrom, E.; Li, Y.; Weber, M.; Brown, C.N. Performance differences between the modified star excursion balance test and the Y-balance test in individuals with chronic ankle instability. J. Sport Rehabil. 2019, 29, 748–753. [Google Scholar] [CrossRef]
- Eechaute, C.; Vaes, P.; Duquet, W. The dynamic postural control is impaired in patients with chronic ankle instability: Reliability and validity of the multiple hop test. Clin. J. Sport Med. 2009, 19, 107–114. [Google Scholar] [CrossRef]
- Hadadi, M.; Ebrahimi, I.; Mousavi, M.E.; Aminian, G.; Esteki, A.; Rahgozar, M. The effect of combined mechanism ankle support on postural control of patients with chronic ankle instability. Prosthet. Orthot. Int. 2017, 41, 58–64. [Google Scholar] [CrossRef]
- O’Neill, B. Sample size determination with a pilot study. PLoS ONE 2022, 17, e0262804. [Google Scholar] [CrossRef]
- Orlando, G.; Brown, S.; Jude, E.; Bowling, F.L.; Boulton, A.J.; Reeves, N.D. Acute effects of vibrating insoles on dynamic balance and gait quality in individuals with diabetic peripheral neuropathy: A randomized crossover study. Diabetes Care 2024, 47, 1004–1011. [Google Scholar] [CrossRef]
- Hatton, A.L.; Chatfield, M.D.; Cattagni, T.; Vicenzino, B. The effects of vibrating shoe insoles on standing balance, walking, and ankle-foot muscle activity in adults with diabetic peripheral neuropathy. Gait Posture 2024, 111, 8–13. [Google Scholar] [CrossRef]
- Novak, P.; Novak, V. Effect of step-synchronized vibration stimulation of soles on gait in Parkinson’s disease: A pilot study. J. Neuroeng. Rehabil. 2006, 3, 9. [Google Scholar] [CrossRef]
- Barati, K.; Kamyab, M.; Takamjani, I.E.; Bidari, S.; Parnianpour, M. Effect of equipping an unloader knee orthosis with vibrators on pain, function, stiffness, and knee adduction moment in people with knee osteoarthritis: A pilot randomized trial. Gait Posture 2023, 99, 83–89. [Google Scholar] [CrossRef]
- Liang, J.N.; Ho, K.Y.; Hung, V.; Reilly, A.; Wood, R.; Yuskov, N.; Lee, Y.J. Effects of augmented somatosensory input using vibratory insoles to improve walking in individuals with chronic post-stroke hemiparesis. Gait Posture 2021, 86, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Jiryaei, Z.; Amini, M.; Sanjari, M.A.; Hajiaghaei, B.; Babazadeh, R.; Jalali, M. The effect of vibration-hinged ankle foot orthoses on gait and spasticity in children with cerebral palsy: A randomized clinical trial design. Prosthet. Orthot. Int. 2024, 48, 387–399. [Google Scholar] [CrossRef] [PubMed]
- Lipsitz, L.A.; Lough, M.; Niemi, J.; Travison, T.; Howlett, H.; Manor, B. A shoe insole delivering subsensory vibratory noise improves balance and gait in healthy elderly people. Arch. Phys. Med. Rehabil. 2015, 96, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Miranda, D.L.; Hsu, W.H.; Gravelle, D.C.; Petersen, K.; Ryzman, R.; Niemi, J.; Lesniewski-Laas, N. Sensory enhancing insoles improve athletic performance during a hexagonal agility task. J. Biomech. 2016, 49, 1058–1063. [Google Scholar] [CrossRef]
- Khaliliyan, H.; Sharafatvaziri, A.; Safaeepour, Z.; Bahramizadeh, M. Gait and muscle activity measures after biomechanical device therapy in subjects with ankle instability: A systematic review. Foot 2024, 59, 102083. [Google Scholar] [CrossRef]
- Rasman, B.G.; Forbes, P.A.; Tisserand, R.; Blouin, J.S. Sensorimotor manipulations of the balance control loop–beyond imposed external perturbations. Front. Neurol. 2018, 9, 899. [Google Scholar] [CrossRef]
- Hertel, J.; Braham, R.A.; Hale, S.A.; Olmsted, L.C. Effect of lateral ankle joint anesthesia on center of balance, postural sway, and joint position sense. J. Sport Rehabil. 2000, 9, 111–125. [Google Scholar] [CrossRef]
- McKeon, P.O.; Booi, M.J.; Branam, B.; Johnson, D.L.; Mattacola, C.G. Lateral ankle ligament anesthesia significantly alters single limb postural control. Gait Posture 2010, 32, 374–377. [Google Scholar] [CrossRef]
- Stehle, S.A.; Aubonnet, R.; Hassan, M.; Recenti, M.; Jacob, D.; Petersen, H.; Gargiulo, P. Predicting postural control adaptation measuring EEG, EMG, and center of pressure changes: BioVRSea paradigm. Front. Hum. Neurosci. 2022, 16, 1038976. [Google Scholar] [CrossRef]
- Picot, B.; Terrier, R.; Forestier, N.; Fourchet, F.; McKeon, P.O. The star excursion balance test: An update review and practical guidelines. Int. J. Athl. Ther. Train. 2021, 26, 285–293. [Google Scholar] [CrossRef]
- Picot, B.; Hardy, A.; Terrier, R.; Tassignon, B.; Lopes, R.; Fourchet, F. Which functional tests and self-reported questionnaires can help clinicians make valid return to sport decisions in patients with chronic ankle instability? A narrative review and expert opinion. Front. Sports Act. Living 2022, 4, 902886. [Google Scholar] [CrossRef]
- Mollà-Casanova, S.; Inglés, M.; Serra-Añó, P. Effects of balance training on functionality, ankle instability, and dynamic balance outcomes in people with chronic ankle instability: Systematic review and meta-analysis. Clin. Rehabil. 2021, 35, 1694–1709. [Google Scholar] [CrossRef]


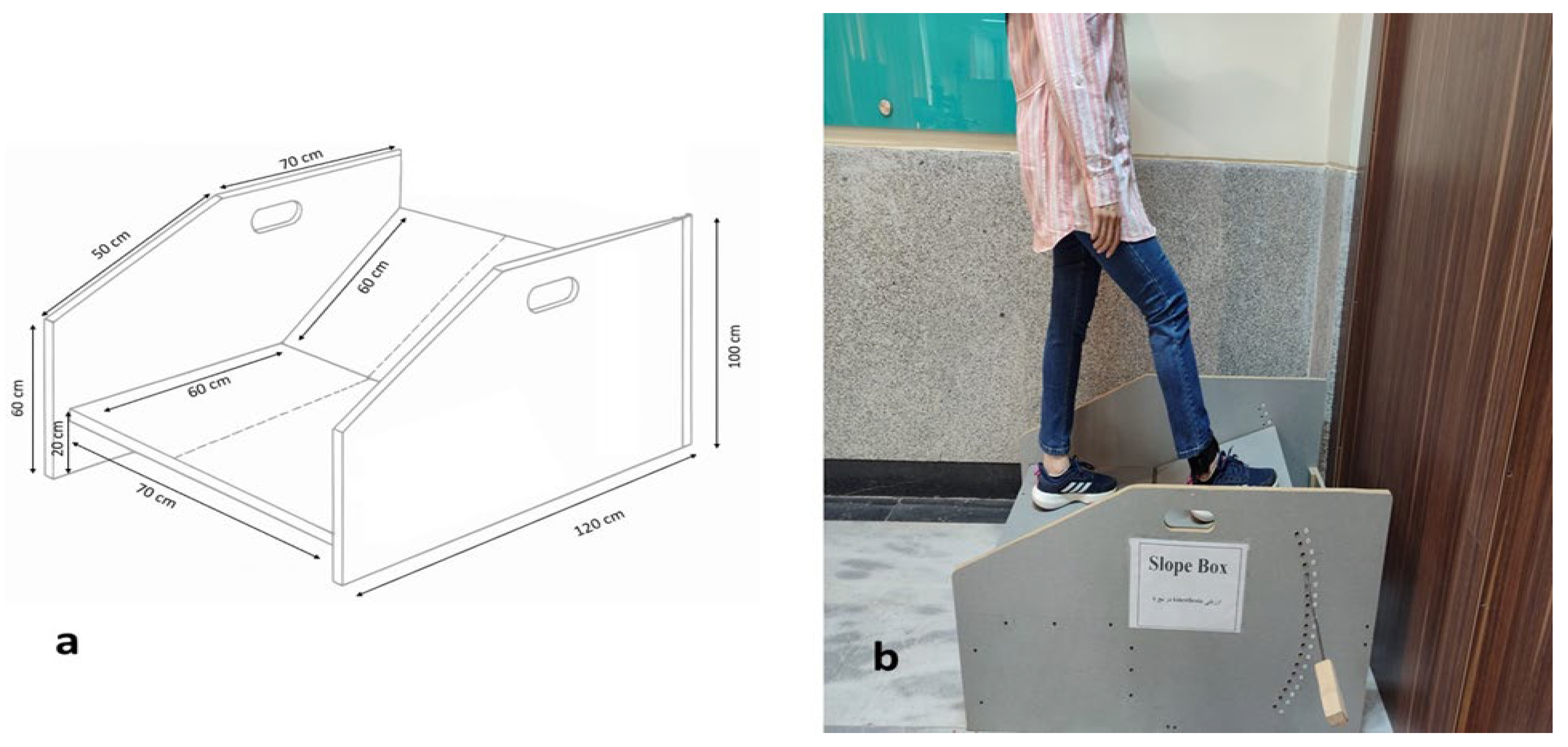


| Characteristics | VAO M ± SD or No. | VFO M ± SD or No. |
|---|---|---|
| Age (years) | 22.6 ± 6.3 | 23.8 ± 5.8 |
| Sex (male/female) | 3/5 | 4/4 |
| Body mass index (kg/m2) | 24.6 ± 4.2 | 23.1 ± 5.8 |
| CAIT (0–30) | 19.3 ± 3.1 | 20.3 ± 1.9 |
| Time since last ankle sprain (months) | 4.3 ± 7.9 | 5.8 ± 6.32 |
| Number of previous ankle sprains | 4.09 ± 5.02 | 5.52 ± 3.26 |
| Dominant side (right/left) | 7/1 | 5/3 |
| Affected side (right/left) | 6/2 | 6/2 |
| Outcome | Groups | Time | Group × Time | Time | Group | ||
|---|---|---|---|---|---|---|---|
| T1 (M ± SD) | T2 (M ± SD) | T3 (M ± SD) | |||||
| Error of ankle JPS (Degrees) | VFO | 2.84 ± 1.04 | 2.5 ± 0.96 | 1.12 ± 1.03 | Wilk’s Lambda = 0.57 F (2,13) = 4.81 p = 0.02 * η2 = 0.42 | Wilk’s Lambda = 0.54 F (2,13) = 5.53 p = 0.01 * η2 = 0.46 | F (2,13) = 2.74 p = 0.12 η2 = 0.16 |
| VAO | 2.96 ± 1.08 | 2.84 ± 1.07 | 2.87 ± 1.06 | ||||
| Mean velocity of COP-AP (mm/s) | VFO | 5.49 ± 1.28 | 5.31 ± 1 | 4.48 ± 1.23 | Wilk’s Lambda = 0.84 F (2,13) = 1.24 p = 0.32 η2 = 0.16 | Wilk’s Lambda = 0.21 F (2,13) = 23.66 p ˂ 0.001 * η2 = 0.78 | F (2,13) = 1.68 p = 0.21 η2 = 0.10 |
| VAO | 6.65 ± 1.19 | 5.83 ± 1.39 | 5.26 ± 1.73 | ||||
| Mean velocity of COP-ML (mm/s) | VFO | 10.61 ± 2.86 | 10.46 ± 2.91 | 10.53 ± 2.74 | Wilk’s Lambda = 0.93 F (2,13) = 0.42 p = 0.66 η2 = 0.06 | Wilk’s Lambda = 0.76 F (2,13) = 2.05 p = 0.16 η2 = 0.24 | F (2,13) = 0.2 p = 0.65 η2 = 0.01 |
| VAO | 11.38 ± 2.21 | 10.97 ± 2.59 | 11.06 ± 2.61 | ||||
| Mean velocity of COP-Resultant (mm/s) | VFO | 11.99 ± 2.94 | 11.76 ±2.93 | 11.42 ± 2.92 | Wilk’s Lambda = 0.78 F (2,13) = 1.73 p = 0.21 η2 = 0.21 | Wilk’s Lambda = 0.48 F (2,13) = 6.93 p = 0.009 * η2 = 0.51 | F (2,13) = 0.64 p = 0.43 η2 = 0.04 |
| VAO | 13.29 ± 1.71 | 12.55 ± 2.16 | 12.38 ± 2.47 | ||||
| SEBT-A (normalized to leg length) | VFO | 0.9 ± 0.13 | 0.9 ± 0.41 | 0.91 ± 0.03 | Wilk’s Lambda= 0.9 F (2,13) = 0.06 p= 0.93 η2 = 0.01 | Wilk’s Lambda = 0.9 F (2,13) = 0.69 p = 0.51 η2 = 0.09 | F (2,13) = 0.31 p = 0.58 η2 = 0.02 |
| VAO | 0.88 ± 0.12 | 0.9 ± 0.05 | 0.91 ± 0.04 | ||||
| SEBT-PM (normalized to leg length) | VFO | 0.7 ± 0.04 | 0.7 ± 0.05 | 0.71 ± 0.05 | Wilk’s Lambda = 0.95 F (2,13) = 0.29 p = 0.74 η2 = 0.04 | Wilk’s Lambda= 0.97 F (2,13) = 0.18 p = 0.83 η2 = 0.02 | F (2,13) = 3.84 p = 0.07 η2 = 0.21 |
| VAO | 0.73 ± 0.02 | 0.74 ±0.01 | 0.74 ± 0.06 | ||||
| SEBT-PL (normalized to leg length) | VFO | 0.75 ± 0.03 | 0.75 ± 0.04 | 0.76 ± 0.04 | Wilk’s Lambda= 0.85 F (2,13) = 1.06 p = 0.37 η2 = 0.14 | Wilk’s Lambda= 0.75 F (2,13) = 2.1 p = 0.16 η2 = 0.14 | F (2,13) = 0.01 p = 0.92 η2 = 0.001 |
| VAO | 0.75 ± 0.04 | 0.76 ± 0.04 | 0.76 ± 0.04 | ||||
| SMHT (s) | VFO | 5.35 ± 1.14 | 4.88 ± 1 | 4.95 ± 0.97 | Wilk’s Lambda= 0.95 F (2,13) = 0.3 p = 0.75 η2 = 0.04 | Wilk’s Lambda= 0.72 F (2,13) = 2.50 p = 0.12 η2 = 0.27 | F (2,13) = 1.69 p = 0.21 η2 = 0.1 |
| VAO | 4.59 ± 1.12 | 4.30 ± 1.15 | 4.27 ± 1.15 | ||||
| Pairwise Between-Group Comparisons at Each Time Point | ||||
|---|---|---|---|---|
| Outcome | Time | Conditions | MD ± SE (95%CI) | p Value (Cohen’s d) |
| Error of ankle JPS (Degrees) | T1 | VFO vs. VAO | −0.12 ± 0.53 | 0.81 (−0.11) |
| (−1.26 to 1.01) | ||||
| T2 | VFO vs. VAO | −0.34 ± 0.51 | 0.51 (−0.33) | |
| (−1.44 to 0.75) | ||||
| T3 | VFO vs. VAO | −1.75 ± 0.52 | 0.005 * (−1.68) | |
| (−2.87 to −0.62) | ||||
| Pairwise comparisons of time points within each group | ||||
| Outcome | Group | Conditions | MD ± SE (95%CI) | p value (Cohen’s d) |
| Error of ankle JPS (Degrees) | VFO | T1 vs. T2 | 0.34 ± 0.27 | 0.68 (0.45) |
| (−0.39 to 1.08) | ||||
| T1 vs. T3 | 1.71 ± 0.37 | 0.001 * (1.64) | ||
| (0.7 to 2.73) | ||||
| T2 vs. T3 | 1.37 ± 0.34 | 0.004 * (1.42) | ||
| (0.43 to 2.31) | ||||
| VAO | T1 vs. T2 | 0.12 ± 0.27 | 1 (0.16) | |
| (−0.61 to 0.86) | ||||
| T1 vs. T3 | 0.09 ± 0.37 | 1 (0.09) | ||
| (−0.61 to 0.86) | ||||
| T2 vs. T3 | −0.31 ± 0.34 | 1 (−0.32) | ||
| (−0.97 to 0.91) | ||||
| Mean velocity of COP-AP (mm/s) | VFO | T1 vs. T2 | 0.18 ± 0.28 | 1 (0.23) |
| (−0.58 to 0.94) | ||||
| T1 vs. T3 | 1 ± 0.24 | 0.003 * (1.47) | ||
| (0.33 to 1.67) | ||||
| T2 vs. T3 | 0.82 ± 0.23 | 0.01 (1.26) | ||
| (0.17 to 1.47) | ||||
| VAO | T1 vs. T2 | 0.82 ± 0.28 | 0.03 (1.03) | |
| (0.06 to 1.58) | ||||
| T1 vs. T3 | 1.39 ± 0.24 | ˂0.001 * (2.05) | ||
| (0.72 to 2.06) | ||||
| T2 vs. T3 | 0.56 ± 0.23 | 0.09 (0.86) | ||
| (−0.08 to 1.21) | ||||
| Mean velocity of COP-R (mm/s) | VFO | T1 vs. T2 | 0.23 ± 0.19 | 0.72 (0.43) |
| (−0.28 to 0.75) | ||||
| T1 vs. T3 | 0.57 ± 0.32 | 0.3 (0.63) | ||
| (−0.31 to 1.46) | ||||
| T2 vs. T3 | 0.34 ± 0.26 | 0.65 (0.46) | ||
| (−1.06 to 0.37) | ||||
| VAO | T1 vs. T2 | 0.74 ± 0.19 | 0.005 * (1.38) | |
| (0.22 to 1.25) | ||||
| T1 vs. T3 | 0.91 ± 0.32 | 0.04 (1.01) | ||
| (0.03 to 1.08) | ||||
| T2 vs. T3 | 0.17 ± 0.26 | 1 (0.23) | ||
| (−0.54 to 0.89) | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Khaliliyan, H.; Bahramizadeh, M.; Sadeghi-Demneh, E. The Effects of Ankle Versus Plantar Vibrotactile Orthoses on Joint Position Sense and Postural Control in Individuals with Functional Ankle Instability: A Pilot Randomized Trial. Bioengineering 2026, 13, 138. https://doi.org/10.3390/bioengineering13020138
Khaliliyan H, Bahramizadeh M, Sadeghi-Demneh E. The Effects of Ankle Versus Plantar Vibrotactile Orthoses on Joint Position Sense and Postural Control in Individuals with Functional Ankle Instability: A Pilot Randomized Trial. Bioengineering. 2026; 13(2):138. https://doi.org/10.3390/bioengineering13020138
Chicago/Turabian StyleKhaliliyan, Hanieh, Mahmood Bahramizadeh, and Ebrahim Sadeghi-Demneh. 2026. "The Effects of Ankle Versus Plantar Vibrotactile Orthoses on Joint Position Sense and Postural Control in Individuals with Functional Ankle Instability: A Pilot Randomized Trial" Bioengineering 13, no. 2: 138. https://doi.org/10.3390/bioengineering13020138
APA StyleKhaliliyan, H., Bahramizadeh, M., & Sadeghi-Demneh, E. (2026). The Effects of Ankle Versus Plantar Vibrotactile Orthoses on Joint Position Sense and Postural Control in Individuals with Functional Ankle Instability: A Pilot Randomized Trial. Bioengineering, 13(2), 138. https://doi.org/10.3390/bioengineering13020138





