A Novel Mixed Stimulation Pattern for Balanced Pulmonary EIT Imaging Performance
Abstract
1. Introduction
- Proposing a weight-adjustable mixed stimulation pattern to resolve the long-standing trade-off dilemma between anti-noise performance and image interpretability in traditional pulmonary EIT stimulation patterns.
- Achieving a balanced optimization of real-time capability, signal anti-noise performance, image interpretability, and artifact reduction.
- Providing a flexible trade-off parameter to improve EIT’s adaptability to diverse acquisition environments.
- Validating stable imaging in low-current scenarios, expanding the practical applicability of pulmonary EIT imaging.
2. Materials and Methods
2.1. Mixed Stimulation Pattern
2.1.1. Measurement Channels of the Mixed Stimulation Pattern
2.1.2. EIT Image Reconstruction Algorithm for the Mixed Stimulation Pattern
2.2. Performance Evaluation of the Mixed Stimulation Pattern via Simulation and Human Experiments
2.2.1. Performance Parameters
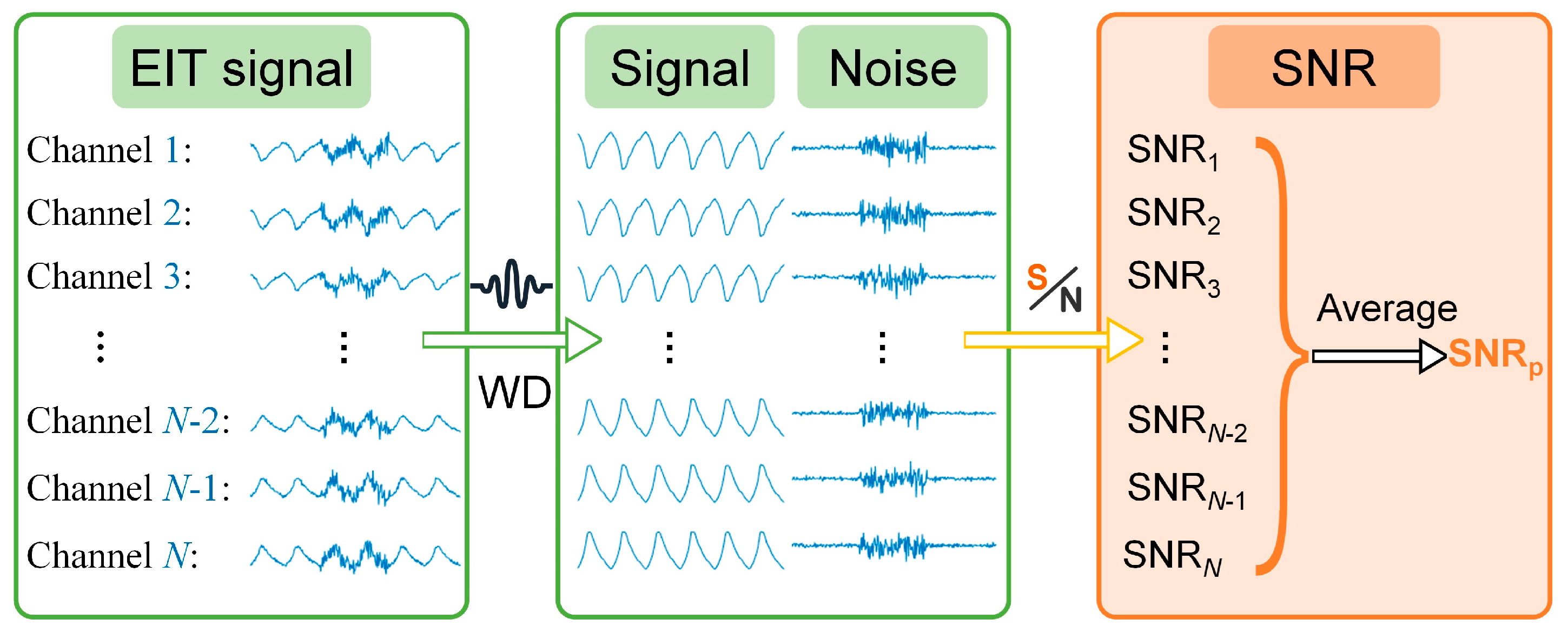
- Measurement Sensitivity and EIT Signal SNR
- 2.
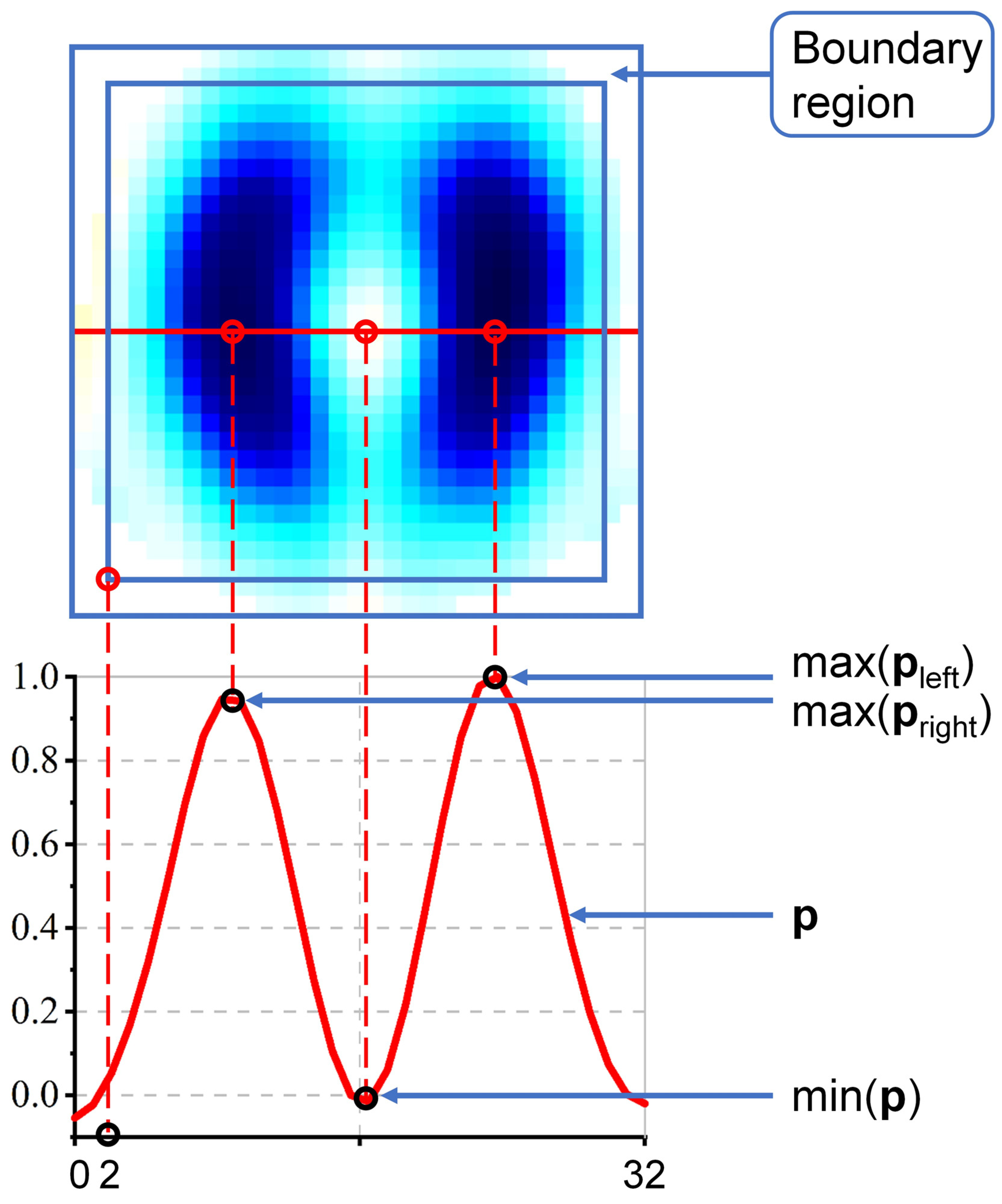
- Lung Ventilation EIT Image Indicators
- 3.
- Lung Ventilation EIT Clinical Indicators
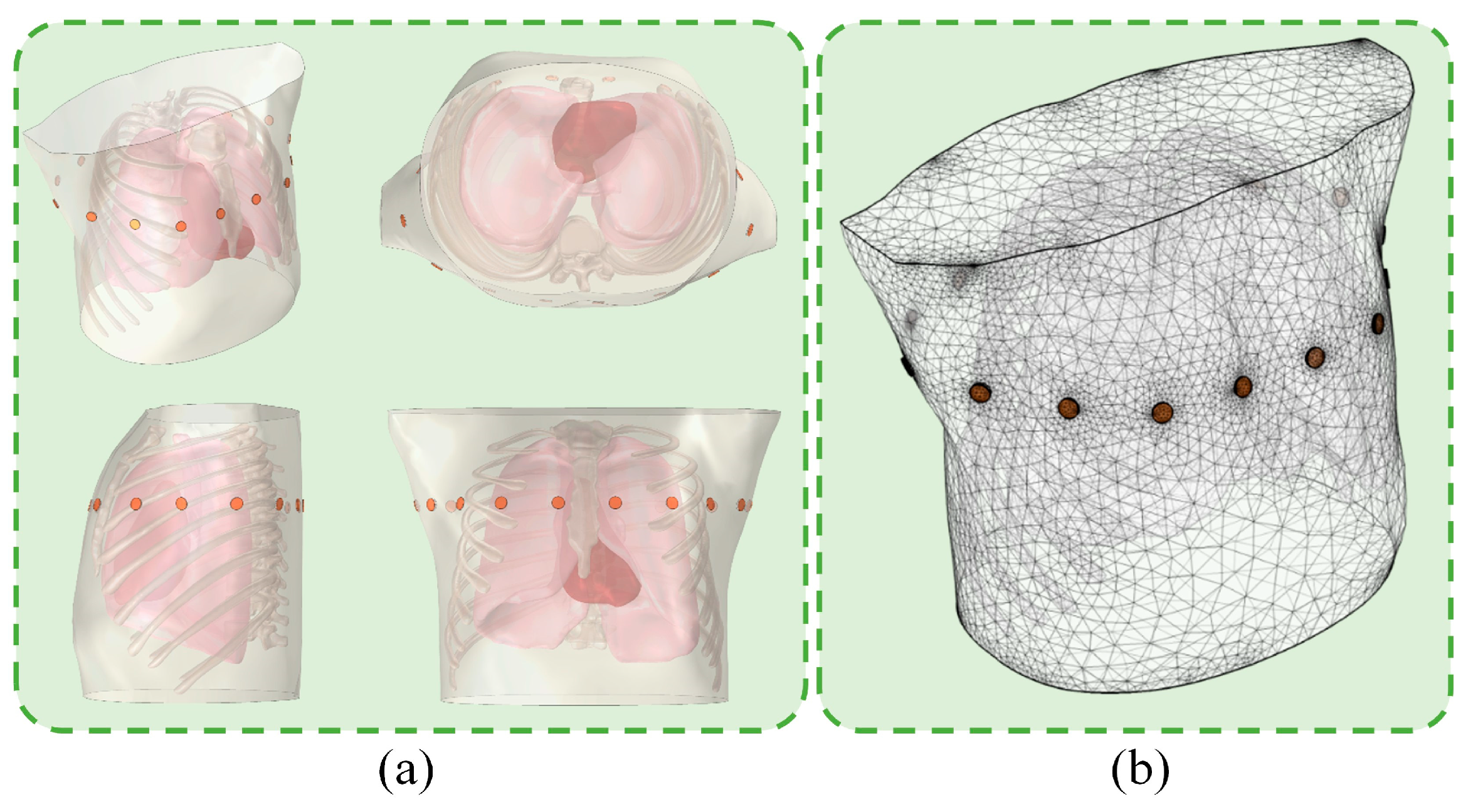
2.2.2. Details of the Simulation Model
2.2.3. Human Experiment Protocol
- EIT Electrode Setup: In accordance with expert consensus, the 16-electrode belt was placed on the horizontal plane of the 4th–5th intercostal space of the subjects’ chest. The 16 electrodes were distributed equidistantly in a clockwise direction (from the foot-to-head view), with electrode 1 and electrode 16 located on the left and right sides of the median sagittal plane of the human body, respectively.
- EIT Data Acquisition Equipment: We used a wireless wearable electrical impedance tomography system to collect EIT data from 30 subjects. The current stimulation frequency of this system is 50 kHz, the sampling rate is 20 frames/s, and the current range is 100 μA to 1 mA. The results of resistance phantom tests showed that the measurement SNR of the device is greater than 70 dB, and the relative change in measurement within 3 h is less than 0.1% [36].
- EIT Data Acquisition Process: During the entire data acquisition period, subjects were required to maintain a sitting posture and breathe calmly. Opposite stimulation and adjacent stimulation were selected, respectively, with a stimulation current amplitude of 1 mA set. In addition, the mixed stimulation pattern was selected, and three stimulation current amplitudes (1 mA, 600 μA, and 200 μA) were configured in sequence. There were a total of 5 acquisition configurations above, and for each configuration, EIT data were collected for more than 2 min.
- Statistical Analysis: Data from male and female subjects were analyzed independently, with 15 sets of data for each gender. For the pulmonary EIT signals collected for more than 2 min under the three stimulation patterns with a stimulation current amplitude of 1 mA, their signal SNR and image indicators were calculated. In addition, for the pulmonary EIT signals collected for more than 2 min under the mixed stimulation pattern with three stimulation current amplitudes (1 mA, 600 μA, and 200 μA), their image indicators and clinical indicators were calculated. First, normality test and homogeneity of variance test were performed. On this basis, traditional ANOVA was applied to compare the differences in signal SNR and image indicators among different stimulation patterns (aiming to evaluate the anti-noise performance and imaging capability of the mixed stimulation pattern), and to compare the differences in image indicators and clinical indicators among different current amplitudes (aiming to evaluate the stability of the mixed stimulation pattern under low-current conditions), with Tukey’s HSD test used for multiple comparisons. In addition, for data that failed the homogeneity of variance test, Welch’s ANOVA and Games–Howell test were used for multiple comparisons. For data that failed the normality test, the Friedman test was used for analysis, and pairwise comparisons were conducted via the corrected paired Wilcoxon signed-rank test.
3. Results
3.1. Simulation Experiments
3.2. Human Experiments
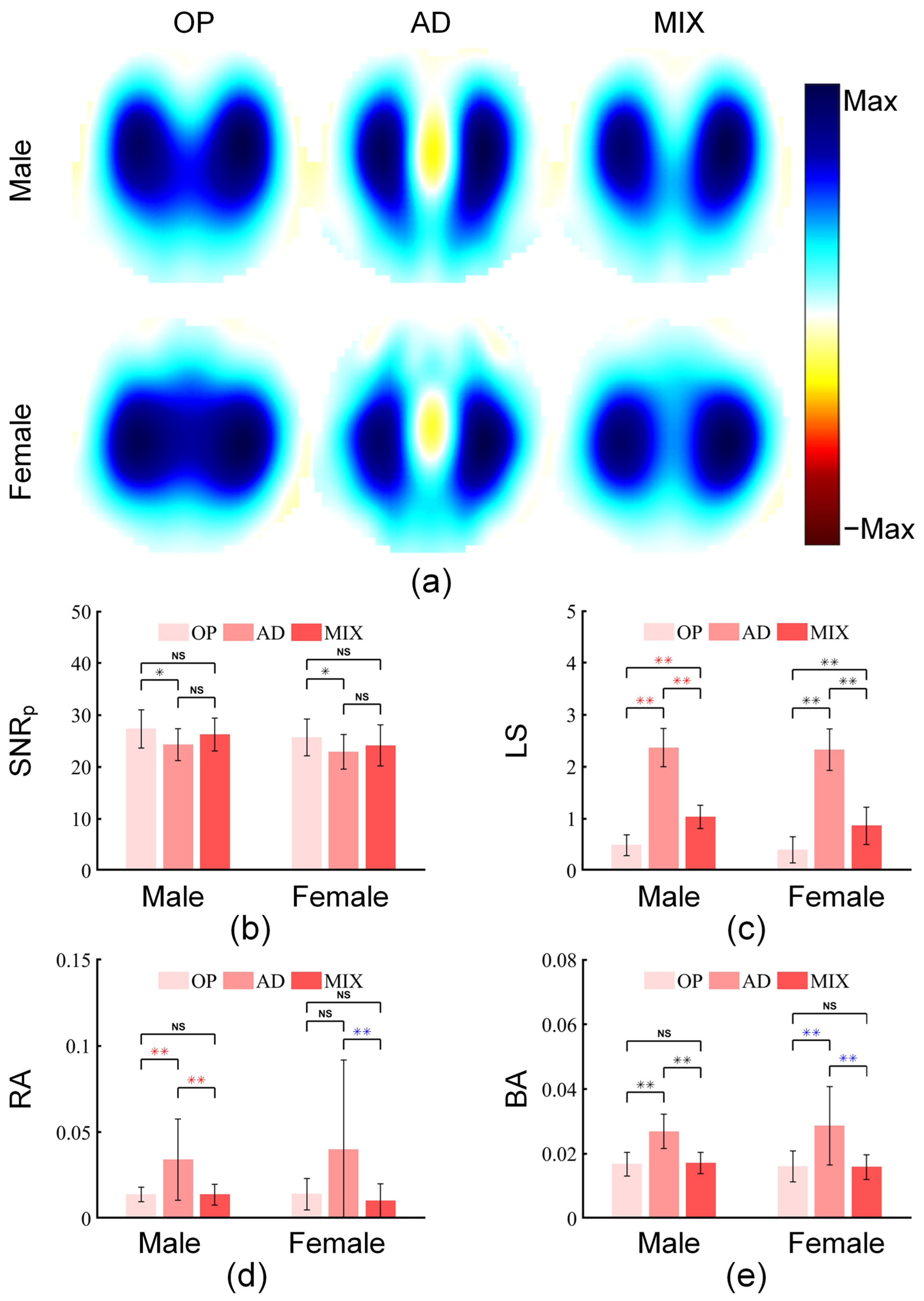
3.2.1. Comparison of Three Stimulation Patterns
3.2.2. Comparison of the Mixed Stimulation Pattern Under Different Stimulation Current Amplitudes
4. Discussion
4.1. Analysis of Simulation and Human Experiment Results
4.2. Weighted Mixed Stimulation Pattern
4.3. Advantages and Limitations of the Mixed Stimulation Pattern
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A

References
- Henderson, R.P.; Webster, J.G. An impedance camera for spatially specific measurements of the thorax. IEEE Trans. Biomed. Eng. 2007, 25, 250–254. [Google Scholar] [CrossRef] [PubMed]
- Adler, A.; Boyle, A. Electrical impedance tomography: Tissue properties to image measures. IEEE Trans. Biomed. Eng. 2017, 64, 2494–2504. [Google Scholar] [CrossRef]
- Dimas, C.; Alimisis, V.; Uzunoglu, N.; Sotiriadis, P.P. Advances in electrical impedance tomography inverse problem solution methods: From traditional regularization to deep learning. IEEE Access 2024, 12, 47797–47829. [Google Scholar] [CrossRef]
- Lan, L.; Ni, Y.; Zhou, Y.; Fu, L.; Wu, W.; Li, P.; Yu, H.; Liang, G.; Luo, F. PEEP-Induced Lung Recruitment Maneuver Combined with Prone Position for ARDS: A Single-Center, Prospective, Randomized Clinical Trial. J. Clin. Med. 2024, 13, 853. [Google Scholar] [CrossRef]
- Jonkman, A.H.; Alcala, G.C.; Pavlovsky, B.; Roca, O.; Spadaro, S.; Scaramuzzo, G.; Chen, L.; Dianti, J.; Sousa, M.L.; Sklar, M.C.; et al. Lung recruitment assessed by electrical impedance tomography (RECRUIT): A multicenter study of COVID-19 acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 2023, 208, 25–38. [Google Scholar] [CrossRef]
- Pequignot, B.; Combes, A.; Lescroart, M.; Levy, B.; Koszutski, M. Contribution of electrical impedance tomography to personalize positive end-expiratory pressure under ECCO2R. Crit. Care 2024, 28, 124. [Google Scholar] [CrossRef]
- Zhang, H.; Wu, Y.; Gao, X.; Peng, C.; Li, R.; Wang, A.; Zhang, J.; Yuan, S.; Yang, L.; Zou, X.; et al. Effects of different VV ECMO blood flow rates on lung perfusion assessment by hypertonic saline bolus-based electrical impedance tomography. Crit. Care 2024, 28, 274. [Google Scholar] [CrossRef]
- Hou, H.; Ning, F.; Li, W.; Zhai, Q.; Wei, J.; Wang, C. Coherent Noise Denoising in Beamforming Based on Non-Convex Robust Principal Component Analysis. J. Theor. Comput. Acoust. 2024, 32, 2340004. [Google Scholar] [CrossRef]
- Liu, Y.; Du, B.; Chen, Y.; Zhang, L.; Gong, M.; Tao, D. Convex–concave tensor robust principal component analysis. Int. J. Comput. Vis. 2024, 132, 1721–1747. [Google Scholar] [CrossRef]
- Kimura, S.; Sato, M.; Sano, K.; Shimoyama, K.; Honda, T.; Yasukawa, K.; Takanashi, J.I. Electrical impedance tomography for evaluating child respiratory rehabilitation. Pediatr. Int. 2023, 65, e15572. [Google Scholar] [CrossRef]
- Aydeniz, E.; van Bussel, B.C.; de Jongh, S.; Schellens, J.; Heines, S.J.; van Kuijk, S.M.; Tas, J.; van Rosmalen, F.; van der Horst, I.C.; Bergmans, D.C.; et al. Serial electrical impedance tomography course in different treatment groups; The MaastrICCht cohort. J. Crit. Care 2024, 80, 154506. [Google Scholar] [CrossRef]
- Florio, G.; Zanella, A.; Slobod, D.; Guzzardella, A.; Protti, I.; Carlesso, E.; Canakoglu, A.; Fumagalli, J.; Scaravilli, V.; Colombo, S.M.; et al. Impact of Positive End-Expiratory Pressure and FiO2 on Lung Mechanics and Intrapulmonary Shunt in Mechanically Ventilated Patients with ARDS Due to COVID-19 Pneumonia. J. Intensive Care Med. 2024, 39, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Öner, Ö.; Ergan, B.; Kizil, A.S.; Gurkok, M.C.; Dugral, E.; Gökmen, N. Investigation of high flow nasal cannule efficiency with electric impedance tomography based parameters in COVID-19 adults patients: A retrospective study. PeerJ 2023, 11, e15555. [Google Scholar] [CrossRef] [PubMed]
- Gremme, Y.; Derlien, S.; Katzer, K.; Reuken, P.A.; Stallmach, A.; Lewejohann, J.C.; Lemhöfer, C. Visualizing exertional dyspnea in a post-COVID patient using electrical impedance tomography. Infection 2023, 51, 1841–1845. [Google Scholar] [CrossRef]
- He, H.; Zhao, Z.; Becher, T.; Bellani, G.; Yoshida, T.; Amato, M.B.; Long, Y.; Frerichs, I.; Bayford, R.; Berra, L.; et al. Recommendations for lung ventilation and perfusion assessment with chest electrical impedance tomography in critically ill adult patients: An international evidence-based and expert Delphi consensus study. EClinicalMedicine 2025, 89, 103575. [Google Scholar] [CrossRef]
- Adler, A.; Gaggero, P.O.; Maimaitijiang, Y. Adjacent stimulation and measurement patterns considered harmful. Physiol. Meas. 2011, 32, 731. [Google Scholar] [CrossRef]
- Barber, C.C.; Brown, B.H.; Freeston, I.L. Imaging spatial distributions of resistivity using applied potential tomography. Electron. Lett. 1983, 19, 933–935. [Google Scholar] [CrossRef]
- Cheney, M.; Isaacson, D. Distinguishability in impedance imaging. IEEE Trans. Biomed. Eng. 2002, 39, 852–860. [Google Scholar] [CrossRef]
- Bera, T.K.; Nagaraju, J. Studying the resistivity imaging of chicken tissue phantoms with different current patterns in Electrical Impedance Tomography (EIT). Measurement 2012, 45, 663–682. [Google Scholar] [CrossRef]
- Tarabi, N.; Mousazadeh, H.; Jafari, A.; Taghizadeh-Tameh, J.; Kiapey, A. Experimental evaluation of some current injection-voltage reading patterns in electrical impedance tomography (EIT) and comparison to simulation results-case study: Large scales. Flow Meas. Instrum. 2022, 83, 102087. [Google Scholar] [CrossRef]
- Ren, S.; Sun, K.; Liu, D.; Dong, F. A statistical shape-constrained reconstruction framework for electrical impedance tomography. IEEE Trans. Med. Imaging 2019, 38, 2400–2410. [Google Scholar] [CrossRef]
- Page, M.I.; Nicholson, R.; Tawhai, M.H.; Clark, A.R.; Kumar, H. Improved electrical impedance tomography reconstruction via a Bayesian approach with an anatomical statistical shape model. IEEE Trans. Biomed. Eng. 2023, 70, 2486–2495. [Google Scholar] [CrossRef]
- Chen, R.; Krueger-Ziolek, S.; Lovas, A.; Benyó, B.; Rupitsch, S.J.; Moeller, K. Structural priors represented by discrete cosine transform improve EIT functional imaging. PLoS ONE 2023, 18, e0285619. [Google Scholar] [CrossRef]
- Russo, S.; Nefti-Meziani, S.; Carbonaro, N.; Tognetti, A. A quantitative evaluation of drive pattern selection for optimizing EIT-based stretchable sensors. Sensors 2017, 17, 1999. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, H. Single source current drive patterns for electrical impedance tomography. In Proceedings of the 2010 IEEE Instrumentation & Measurement Technology Conference, Austin, TX, USA, 3–6 May 2010; IEEE: New York City, NY, USA; pp. 1477–1480. [Google Scholar]
- Silva, O.L.; Lima, R.G.; Martins, T.C.; de Moura, F.S.; Tavares, R.S.; Tsuzuki, M.S. Influence of current injection pattern and electric potential measurement strategies in electrical impedance tomography. Control. Eng. Pract. 2017, 58, 276–286. [Google Scholar] [CrossRef]
- Geselowitz, D.B. An application of electrocardiographic lead theory to impedance plethysmography. IEEE Trans. Biomed. Eng. 2008, 18, 38–41. [Google Scholar] [CrossRef] [PubMed]
- Aebli, J.; Bohnenblust, V.; Koepp-Medina, G.; Ahsani-Nasab, S.; Huber, M.; Greif, R.; Disma, N.; Riva, T.; Riedel, T.; Fuchs, A. Lung Volume Change Under Apnoeic Oxygenation With Different Flow Rates in Children: A Single-Centre Prospective Randomized Controlled Non-Inferiority Trial. Pediatr. Anesth. 2026, 36, 36–42. [Google Scholar] [CrossRef]
- Adler, A.; Arnold, J.H.; Bayford, R.; Borsic, A.; Brown, B.; Dixon, P.; Faes, T.J.; Frerichs, I.; Gagnon, H.; Gärber, Y.; et al. GREIT: A unified approach to 2D linear EIT reconstruction of lung images. Physiol. Meas. 2009, 30, S35. [Google Scholar] [CrossRef] [PubMed]
- Grychtol, B.; Müller, B.; Adler, A. 3D EIT image reconstruction with GREIT. Physiol. Meas. 2016, 37, 785. [Google Scholar] [CrossRef]
- Zhao, Z.; Dai, M.; Zhao, Z.; Wang, H.; Cao, X.; Dai, J.; Wang, C.; Fu, F.; Yang, L. Real-time quality assessment of clinical pulmonary EIT signals during ventilation monitoring. Measurement 2025, 257, 118920. [Google Scholar] [CrossRef]
- Yang, L.; Gao, Z.; Wang, C.; Wang, H.; Dai, J.; Liu, Y.; Qin, Y.; Dai, M.; Cao, X.; Zhao, Z. Evaluation of adjacent and opposite current injection patterns for a wearable chest electrical impedance tomography system. Physiol. Meas. 2024, 45, 025004. [Google Scholar] [CrossRef]
- Borsic, A.; Adler, A. A primal–dual interior-point framework for using the L1 or L2 norm on the data and regularization terms of inverse problems. Inverse Probl. 2012, 28, 095011. [Google Scholar] [CrossRef]
- Blankman, P.; Hasan, D.; Erik, G.J.; Gommers, D. Detection of ‘best’positive end-expiratory pressure derived from electrical impedance tomography parameters during a decremental positive end-expiratory pressure trial. Crit. Care 2014, 18, R95. [Google Scholar] [CrossRef] [PubMed]
- Darbas, M.; Heleine, J.; Mendoza, R.; Velasco, A.C. Sensitivity analysis of the complete electrode model for electrical impedance tomography. Aims Math. 2021, 6, 7333–7366. [Google Scholar] [CrossRef]
- Yang, L.; Dai, M.; Wang, H.; Cao, X.; Li, S.; Fu, F.; Xia, J.; Zhao, Z. A wireless, low-power, and miniaturized EIT system for remote and long-term monitoring of lung ventilation in the isolation ward of ICU. IEEE Trans. Instrum. Meas. 2021, 70, 4005311. [Google Scholar] [CrossRef]
- Adler, A.; Guardo, R. Electrical impedance tomography: Regularized imaging and contrast detection. IEEE Trans. Med. Imaging 1996, 15, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wu, H.; Liu, K.; Chen, B.; Han, W.; Yao, J. Image reconstruction improvement with optimal driven-measurement pattern selection for electrical impedance tomography. IEEE Sens. J. 2021, 21, 13530–13539. [Google Scholar] [CrossRef]
- Bader, O.; Amara, N.E.; Ernst, O.G.; Kanoun, O. Rotating radial injection pattern for highly sensitive electrical impedance tomography of human lung anomalies. Physiol. Meas. 2025, 13, 025001. [Google Scholar] [CrossRef]
- Borsic, A.; Halter, R.; Wan, Y.; Hartov, A.; Paulsen, K.D. Sensitivity study and optimization of a 3D electric impedance tomography prostate probe. Physiol. Meas. 2009, 30, S1. [Google Scholar] [CrossRef]
- Malone, E.; Jehl, M.; Arridge, S.; Betcke, T.; Holder, D. Stroke type differentiation using spectrally constrained multifrequency EIT: Evaluation of feasibility in a realistic head model. Physiol. Meas. 2014, 35, 1051–1066. [Google Scholar] [CrossRef]
- Faulkner, M.; Jehl, M.; Aristovich, K.; Avery, J.; Witkowska-Wrobel, A.; Holder, D. Optimisation of current injection protocol based on a region of interest. Physiol. Meas. 2017, 38, 1158–1175. [Google Scholar] [CrossRef] [PubMed]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhao, Z.; Gao, Z.; Zhu, H.; Zhao, Z.; Dai, M.; Liu, Z.; Fu, F.; Yang, L. A Novel Mixed Stimulation Pattern for Balanced Pulmonary EIT Imaging Performance. Bioengineering 2026, 13, 72. https://doi.org/10.3390/bioengineering13010072
Zhao Z, Gao Z, Zhu H, Zhao Z, Dai M, Liu Z, Fu F, Yang L. A Novel Mixed Stimulation Pattern for Balanced Pulmonary EIT Imaging Performance. Bioengineering. 2026; 13(1):72. https://doi.org/10.3390/bioengineering13010072
Chicago/Turabian StyleZhao, Zhibo, Zhijun Gao, Heyao Zhu, Zhanqi Zhao, Meng Dai, Zilong Liu, Feng Fu, and Lin Yang. 2026. "A Novel Mixed Stimulation Pattern for Balanced Pulmonary EIT Imaging Performance" Bioengineering 13, no. 1: 72. https://doi.org/10.3390/bioengineering13010072
APA StyleZhao, Z., Gao, Z., Zhu, H., Zhao, Z., Dai, M., Liu, Z., Fu, F., & Yang, L. (2026). A Novel Mixed Stimulation Pattern for Balanced Pulmonary EIT Imaging Performance. Bioengineering, 13(1), 72. https://doi.org/10.3390/bioengineering13010072








