Production of Carbon Sources Through Anaerobic Fermentation Using the Liquid Phase of Food Waste Three-Phase Separation: Influencing Factors and Microbial Community Structure
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents Used
2.2. Anaerobic Fermentation Experiments
2.3. Denitrification Experiments
2.4. Physicochemical Analysis
2.5. Microbial Community Analysis
3. Results and Discussion
3.1. Substrate and Inoculum Properties
3.2. Effect of Temperature on Carbon Source Production
3.2.1. VFAs’ Production and Composition Under Different Temperatures
3.2.2. Microbial Community Structure Under Different Temperatures
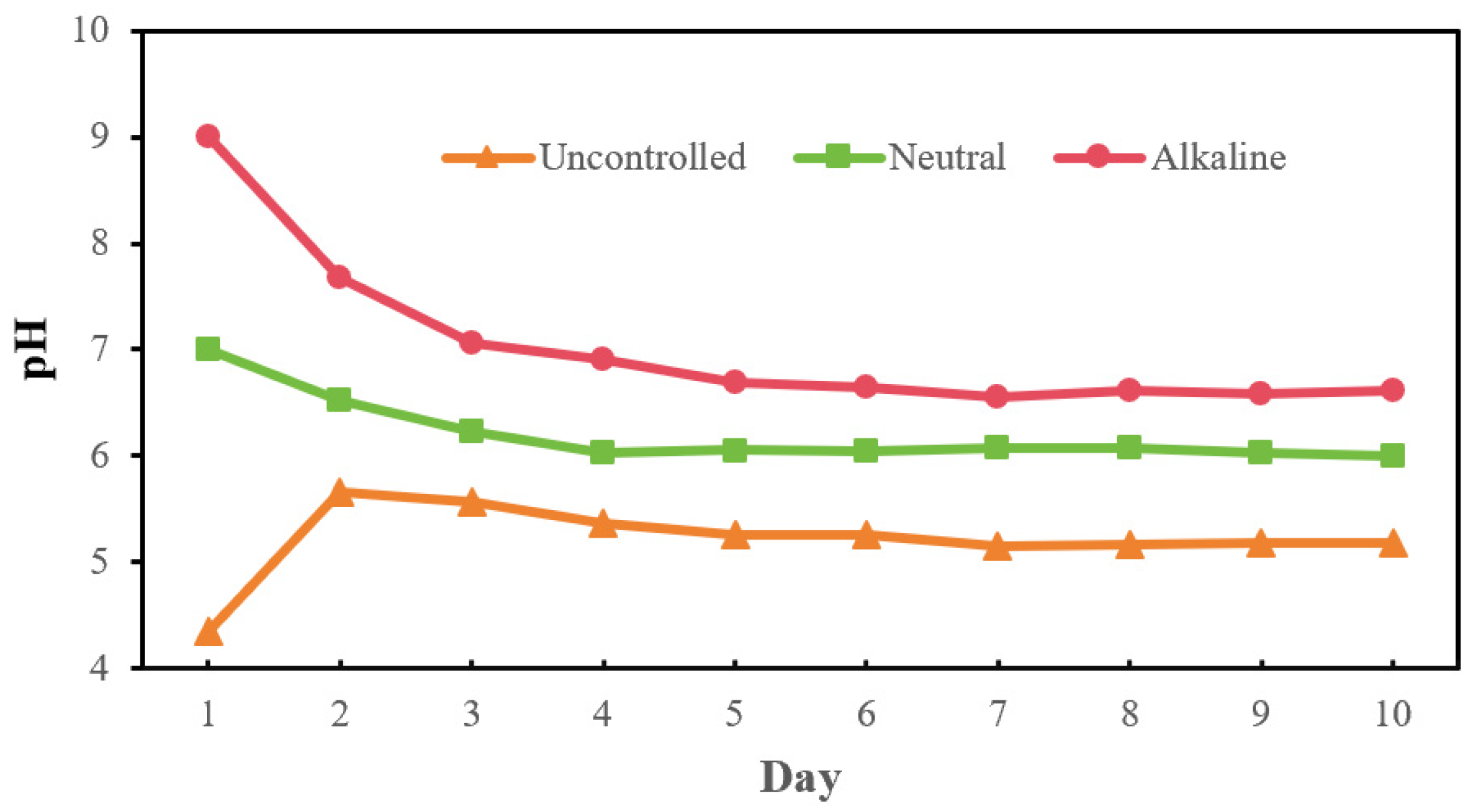
3.3. Effect of Initial pH in Carbon Source Production
3.3.1. VFAs’ Production and Composition Under Different Initial pH Conditions
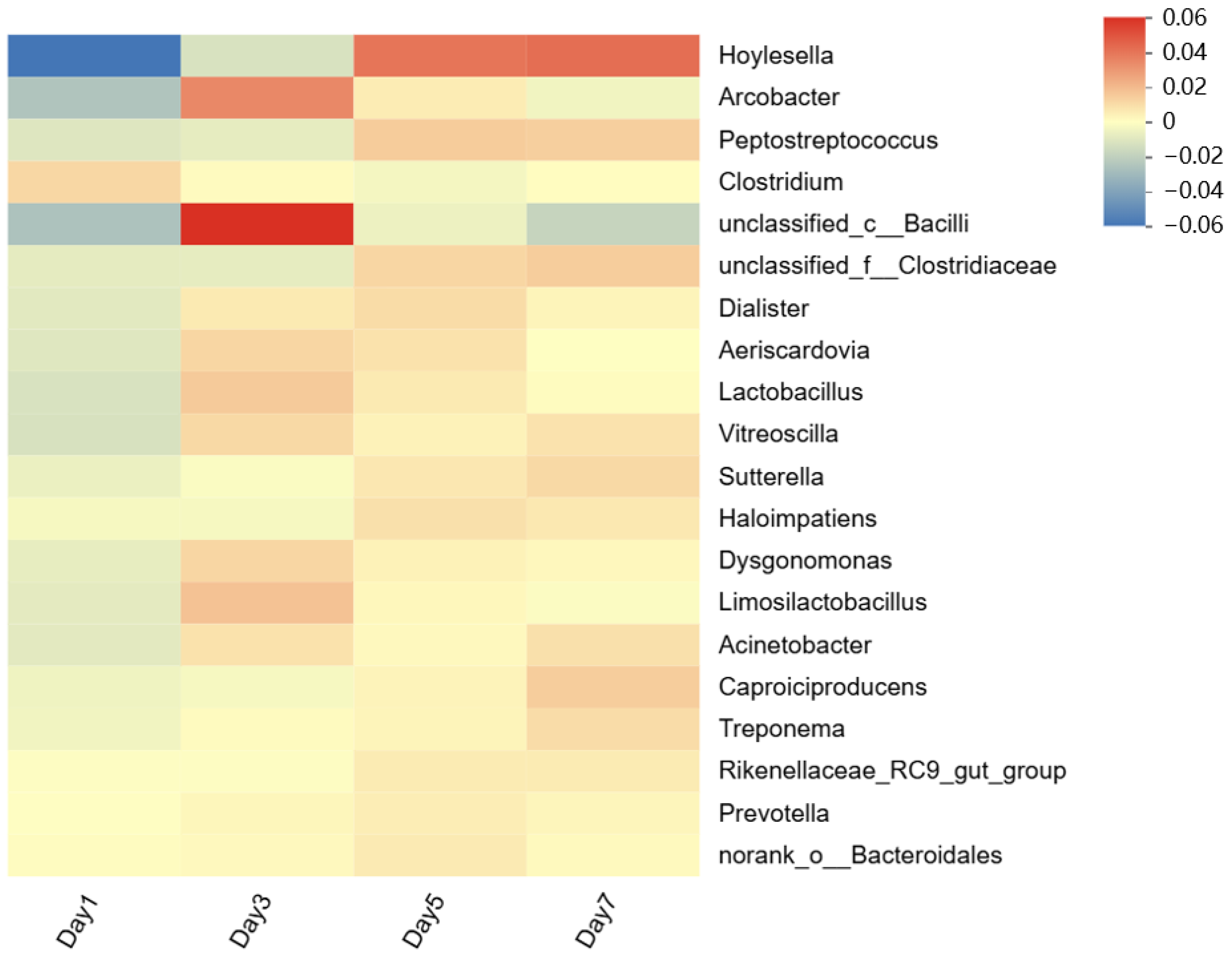
3.3.2. Microbial Community Structure Under Different Initial pH Conditions
3.4. Denitrification Performance
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| VFAs | Volatile Fatty Acids. |
References
- Akhter, S.; Rather, M.I.; Zargar, U.R. Understanding the Food Waste Behaviour in University Students: An Application of the Theory of Planned Behaviour. J. Clean. Prod. 2024, 437, 140632. [Google Scholar] [CrossRef]
- Xiao, Y.; Yang, L.; Sun, C.; Li, H. Efficient Conversion from Food Waste to Composite Carbon Source through Rapid Fermentation and Ceramic Membrane Filtration. Chemosphere 2024, 367, 143601. [Google Scholar] [CrossRef]
- Yu, Q.; Li, H. Life Cycle Environmental Performance of Two Restaurant Food Waste Management Strategies at Shenzhen, China. J. Mater. Cycles Waste Manag. 2021, 23, 826–839. [Google Scholar] [CrossRef]
- Hu, Y.; Zhou, Z.; Shen, C. Microbial Community Acclimation during Anaerobic Digestion of High-Oil Food Waste. Sci. Rep. 2024, 14, 25364. [Google Scholar] [CrossRef]
- Tang, J.; Wang, X.C.; Hu, Y.; Pu, Y.; Huang, J.; Hao Ngo, H.; Zeng, Y.; Li, Y. Nitrogen Removal Enhancement Using Lactic Acid Fermentation Products from Food Waste as External Carbon Sources: Performance and Microbial Communities. Bioresour. Technol. 2018, 256, 259–268. [Google Scholar] [CrossRef]
- Frison, N.; Di Fabio, S.; Cavinato, C.; Pavan, P.; Fatone, F. Best Available Carbon Sources to Enhance the Via-Nitrite Biological Nutrients Removal from Supernatants of Anaerobic Co-Digestion. Chem. Eng. J. 2013, 215–216, 15–22. [Google Scholar] [CrossRef]
- Zhang, H.; Jiang, J.; Li, M.; Yan, F.; Gong, C.; Wang, Q. Biological Nitrate Removal Using a Food Waste-Derived Carbon Source in Synthetic Wastewater and Real Sewage. J. Environ. Manag. 2016, 166, 407–413. [Google Scholar] [CrossRef]
- Qi, S.; Yuan, S.; Wang, W.; Xiao, L.; Zhan, X.; Hu, Z. Effect of Solid-Liquid Separation on Food Waste Fermentation Products as External Carbon Source for Denitrification. J. Clean. Prod. 2021, 284, 124687. [Google Scholar] [CrossRef]
- Elefsiniotis, P.; Wareham, D.G. Utilization Patterns of Volatile Fatty Acids in the Denitrification Reaction. Enzym. Microb. Technol. 2007, 41, 92–97. [Google Scholar] [CrossRef]
- Zhou, M.; Yan, B.; Wong, J.W.C.; Zhang, Y. Enhanced Volatile Fatty Acids Production from Anaerobic Fermentation of Food Waste: A Mini-Review Focusing on Acidogenic Metabolic Pathways. Bioresour. Technol. 2018, 248, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Zhang, Y.; Li, K.; Wang, Q.; Gong, C.; Li, M. Volatile Fatty Acids Production from Food Waste: Effects of pH, Temperature, and Organic Loading Rate. Bioresour. Technol. 2013, 143, 525–530. [Google Scholar] [CrossRef]
- Feng, L.; Yan, Y.; Chen, Y. Co-Fermentation of Waste Activated Sludge with Food Waste for Short-Chain Fatty Acids Production: Effect of pH at Ambient Temperature. Front. Environ. Sci. Eng. China 2011, 5, 623–632. [Google Scholar] [CrossRef]
- Wu, Q.-L.; Guo, W.-Q.; Zheng, H.-S.; Luo, H.-C.; Feng, X.-C.; Yin, R.-L.; Ren, N.-Q. Enhancement of Volatile Fatty Acid Production by Co-Fermentation of Food Waste and Excess Sludge without pH Control: The Mechanism and Microbial Community Analyses. Bioresour. Technol. 2016, 216, 653–660. [Google Scholar] [CrossRef]
- Cheah, Y.-K.; Vidal-Antich, C.; Dosta, J.; Mata-Álvarez, J. Volatile Fatty Acid Production from Mesophilic Acidogenic Fermentation of Organic Fraction of Municipal Solid Waste and Food Waste under Acidic and Alkaline pH. Environ. Sci. Pollut. Res. 2019, 26, 35509–35522. [Google Scholar] [CrossRef]
- Chen, L.; Yang, L.; Peng, C.; Chen, Z.; Li, H.; Deng, Z. Feeding Black Fly Soldier Larva Using Fermented Solid Residue Generated from Food Waste Three-Phase Separation. J. Mater. Cycles Waste Manag. 2025, 27, 2431–2443. [Google Scholar] [CrossRef]
- Chen, L.; He, Z.; Yang, L.; Wang, L.; Li, Y.; Chen, T.; Li, H. Optimal Utilization of Solid Residue from Phase-Separation Pretreatment before Food Waste Anaerobic Digestion. J. Clean. Prod. 2022, 372, 133795. [Google Scholar] [CrossRef]
- Chaitanya, N.; Sivaramakrishna, D.; Kumar, B.S.; Himabindu, V.; Lakshminarasu, M.; Vishwanadham, M. Selection of Pretreatment Method for Enriching Hydrogen-Producing Bacteria Using Anaerobic Sewage Sludge with Three Different Substrates. Biofuels 2016, 7, 163–171. [Google Scholar] [CrossRef]
- Gandhi, B.P.; Otite, S.V.; Fofie, E.A.; Lag-Brotons, A.J.; Ezemonye, L.I.; Semple, K.T.; Martin, A.D. Kinetic Investigations into the Effect of Inoculum to Substrate Ratio on Batch Anaerobic Digestion of Simulated Food Waste. Renew. Energy 2022, 195, 311–321. [Google Scholar] [CrossRef]
- Zhong, M. Preparation of Carbon Source from Food Waste Hydrolysis-Acidification Liquid and Investigation of Its Denitrification Performance. Master’s Thesis, Harbin Institute of Technology, Harbin, China, 2024. [Google Scholar]
- Pawlowski, L. Standard Methods for the Examination of Water and Wastewater, 18th Edition. Sci. Total Environ. 1994, 142, 227–228. [Google Scholar] [CrossRef]
- Hu, Y.; Wang, F.; Chi, Y. The Evolution of Microbial Community during Acclimation for High Sodium Food Waste Anaerobic Digestion. Waste Biomass Valorization 2019, 11, 6057–6063. [Google Scholar] [CrossRef]
- Hu, Y.; Shen, C. Thermophilic-Mesophilic Temperature Phase Anaerobic Co-Digestion Compared with Single Phase Co-Digestion of Sewage Sludge and Food Waste. Sci. Rep. 2024, 14, 11967. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.R.; Li, Y.Z.; Ding, Y.M.; Zhao, B.; Liu, Z.C.; Pang, J.H.; Wang, T.H.; Shang, C. Effects of Mesophilic and Thermophilic Temperature on Hydrolysis and Acidification of Organic Wastes Two-Phase Anaerobic Digestion. Appl. Mech. Mater. 2013, 448–453, 1599–1604. [Google Scholar] [CrossRef]
- Oren, A.; Garrity, G.M. Valid Publication of the Names of Forty-Two Phyla of Prokaryotes. Int. J. Syst. Evol. Microbiol. 2021, 71, 5056. [Google Scholar] [CrossRef] [PubMed]
- Ariesyady, H.; Ito, T.; Okabe, S. Functional Bacterial and Archaeal Community Structures of Major Trophic Groups in a Full-Scale Anaerobic Sludge Digester. Water Res. 2007, 41, 1554–1568. [Google Scholar] [CrossRef]
- Zhao, Z.; Wang, J.; Li, Y.; Zhu, T.; Yu, Q.; Wang, T.; Liang, S.; Zhang, Y. Why Do DIETers like Drinking: Metagenomic Analysis for Methane and Energy Metabolism during Anaerobic Digestion with Ethanol. Water Res. 2020, 171, 115425. [Google Scholar] [CrossRef]
- Li, Q.; Yuwen, C.; Cheng, X.; Yang, X.; Chen, R.; Wang, X.C. Responses of Microbial Capacity and Community on the Performance of Mesophilic Co-Digestion of Food Waste and Waste Activated Sludge in a High-Frequency Feeding CSTR. Bioresour. Technol. 2018, 260, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Feng, L.; Chen, Y.; Sun, H.; Shen, Q.; Li, X.; Chen, H. Alkyl Polyglucose Enhancing Propionic Acid Enriched Short-Chain Fatty Acids Production during Anaerobic Treatment of Waste Activated Sludge and Mechanisms. Water Res. 2015, 73, 332–341. [Google Scholar] [CrossRef]
- Campanaro, S.; Treu, L.; Kougias, P.G.; Luo, G.; Angelidaki, I. Metagenomic Binning Reveals the Functional Roles of Core Abundant Microorganisms in Twelve Full-Scale Biogas Plants. Water Res. 2018, 140, 123–134. [Google Scholar] [CrossRef]
- Waite, D.W.; Vanwonterghem, I.; Rinke, C.; Parks, D.H.; Zhang, Y.; Takai, K.; Sievert, S.M.; Simon, J.; Campbell, B.J.; Hanson, T.E.; et al. Comparative Genomic Analysis of the Class Epsilonproteobacteria and Proposed Reclassification to Epsilonbacteraeota (Phyl. Nov.). Front. Microbiol. 2017, 8, 682, Correction in Front. Microbiol. 2018, 9, 772. https://doi.org/10.3389/fmicb.2018.00772. [Google Scholar] [CrossRef]
- Lee, J.; Han, G.; Shin, S.G.; Koo, T.; Cho, K.; Kim, W.; Hwang, S. Seasonal Monitoring of Bacteria and Archaea in a Full-Scale Thermophilic Anaerobic Digester Treating Food Waste-Recycling Wastewater: Correlations between Microbial Community Characteristics and Process Variables. Chem. Eng. J. 2016, 300, 291–299. [Google Scholar] [CrossRef]
- Hitch, T.C.A.; Bisdorf, K.; Afrizal, A.; Riedel, T.; Overmann, J.; Strowig, T.; Clavel, T. A Taxonomic Note on the Genus Prevotella: Description of Four Novel Genera and Emended Description of the Genera Hallella and Xylanibacter. Syst. Appl. Microbiol. 2022, 45, 126354. [Google Scholar] [CrossRef]
- Ezaki, T.; Yamamoto, N.; Ninomiya, K.; Suzuki, S.; Yabuuchi, E. Transfer of Peptococcus Indolicus, Peptococcus Asaccharolyticus, Peptococcus Prevotii, and Peptococcus Magnus to the Genus Peptostreptococcus and Proposal of Peptostreptococcus Tetradius Sp. Nov. Int. J. Syst. Bacteriol. 1983, 33, 683–698. [Google Scholar] [CrossRef]
- Jiang, W.; Jiang, Y.; Tao, J.; Luo, J.; Xie, W.; Zhou, X.; Yang, L.; Kang, J.; Ye, Y. Enhanced Volatile Fatty Acids Production through Anaerobic Co-Fermentation of Dewatered Sludge and Food Waste: Insights into Performance, Microbial Dynamics and Metabolic Pathways. J. Environ. Chem. Eng. 2025, 13, 119189. [Google Scholar] [CrossRef]
- Fu, Z.; Zhao, J.; Zhong, Z.; Li, B.; Sun, Y.; Fan, H.; Wang, H.; Xie, J.; Hu, Y.; Su, Y.; et al. Novel Insights into Liquid Digestate-Derived Hydrochar Enhances Volatile Fatty Acids Production from Anaerobic Co-Digestion of Sludge and Food Waste. Chem. Eng. J. 2025, 507, 160635. [Google Scholar] [CrossRef]
- Kim, M.; Gomec, C.Y.; Ahn, Y.; Speece, R.E. Hydrolysis and Acidogenesis of Particulate Organic Material in Mesophilic and Thermophilic Anaerobic Digestion. Environ. Technol. 2003, 24, 1183–1190. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, D.; Liu, Y.; Ngo, H.H.; Guo, W.; Yang, Q.; Li, X. Novel Stepwise pH Control Strategy to Improve Short Chain Fatty Acid Production from Sludge Anaerobic Fermentation. Bioresour. Technol. 2018, 249, 431–438. [Google Scholar] [CrossRef]
- Wu, Q.; Guo, W.; Bao, X.; Meng, X.; Yin, R.; Du, J.; Zheng, H.; Feng, X.; Luo, H.; Ren, N. Upgrading Liquor-Making Wastewater into Medium Chain Fatty Acid: Insights into Co-Electron Donors, Key Microflora, and Energy Harvest. Water Res. 2018, 145, 650–659. [Google Scholar] [CrossRef]
- Chuvochina, M.; Mussig, A.J.; Chaumeil, P.-A.; Skarshewski, A.; Rinke, C.; Parks, D.H.; Hugenholtz, P. Proposal of Names for 329 Higher Rank Taxa Defined in the Genome Taxonomy Database under Two Prokaryotic Codes. FEMS Microbiol. Lett. 2023, 370, fnad071. [Google Scholar] [CrossRef] [PubMed]
- Oren, A.; Arahal, D.R.; Ventosa, A. Emended Descriptions of Genera of the Family Halobacteriaceae. Int. J. Syst. Evol. Microbiol. 2009, 59, 637–642. [Google Scholar] [CrossRef]
- Mayumi, D.; Mochimaru, H.; Tamaki, H.; Yamamoto, K.; Yoshioka, H.; Suzuki, Y.; Kamagata, Y.; Sakata, S. Methane Production from Coal by a Single Methanogen. Science 2016, 354, 222–225. [Google Scholar] [CrossRef] [PubMed]
- Conrad, R. Importance of Hydrogenotrophic, Aceticlastic and Methylotrophic Methanogenesis for Methane Production in Terrestrial, Aquatic and Other Anoxic Environments: A Mini Review. Pedosphere 2020, 30, 25–39. [Google Scholar] [CrossRef]
- Sage, M.; Daufin, G.; Gésan-Guiziou, G. Denitrification Potential and Rates of Complex Carbon Source from Dairy Effluents in Activated Sludge System. Water Res. 2006, 40, 2747–2755. [Google Scholar] [CrossRef] [PubMed]
- Tian, Z.; Li, G.; Xiong, Y.; Cao, X.; Pang, H.; Tang, W.; Liu, Y.; Bai, M.; Zhu, Q.; Du, C.; et al. Step-Feeding Food Waste Fermentation Liquid as Supplementary Carbon Source for Low C/N Municipal Wastewater Treatment: Bench Scale Performance and Response of Microbial Community. J. Environ. Manag. 2023, 345, 118434. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, E.; Ren, Y.; Chen, P.; Gaballah, M.S.; Wei, Q.; Dong, R.; Guo, J. Volatile Fatty Acids from Food Waste as External Carbon Sources: Denitrification Efficiency, Microbial Pathways, and Techno-Economic Assessment. Bioresour. Technol. 2025, 438, 133257. [Google Scholar] [CrossRef]
- Li, S.; Yang, H. Optimizing Denitrification with Volatile Fatty Acids from Hydrolysis Acidification-Treated Domestic Wastewater: Comparative Effects of Nitrate and Nitrite Using Immobilized Biofiller. J. Environ. Manag. 2025, 378, 124812. [Google Scholar] [CrossRef] [PubMed]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Hu, Y.; Lin, E.; Weng, X.; Wang, F.; Chen, Z.; Lv, G. Production of Carbon Sources Through Anaerobic Fermentation Using the Liquid Phase of Food Waste Three-Phase Separation: Influencing Factors and Microbial Community Structure. Bioengineering 2026, 13, 60. https://doi.org/10.3390/bioengineering13010060
Hu Y, Lin E, Weng X, Wang F, Chen Z, Lv G. Production of Carbon Sources Through Anaerobic Fermentation Using the Liquid Phase of Food Waste Three-Phase Separation: Influencing Factors and Microbial Community Structure. Bioengineering. 2026; 13(1):60. https://doi.org/10.3390/bioengineering13010060
Chicago/Turabian StyleHu, Yangqing, Enwei Lin, Xianming Weng, Fei Wang, Zhenghui Chen, and Guojun Lv. 2026. "Production of Carbon Sources Through Anaerobic Fermentation Using the Liquid Phase of Food Waste Three-Phase Separation: Influencing Factors and Microbial Community Structure" Bioengineering 13, no. 1: 60. https://doi.org/10.3390/bioengineering13010060
APA StyleHu, Y., Lin, E., Weng, X., Wang, F., Chen, Z., & Lv, G. (2026). Production of Carbon Sources Through Anaerobic Fermentation Using the Liquid Phase of Food Waste Three-Phase Separation: Influencing Factors and Microbial Community Structure. Bioengineering, 13(1), 60. https://doi.org/10.3390/bioengineering13010060




