High-Rate Bioelectrochemical Anaerobic Digester for Biomethane Production from Food Waste
Abstract
1. Introduction
2. Materials and Methods
2.1. Inoculum and Feedstock Characteristics
2.2. Reactor Design and Location
2.3. Reactor Startup and Operating Conditions
2.4. Analytical Methods and Calculations
3. Results and Discussion
3.1. Operation of BEAD and Control Reactors on Food Waste Leachate
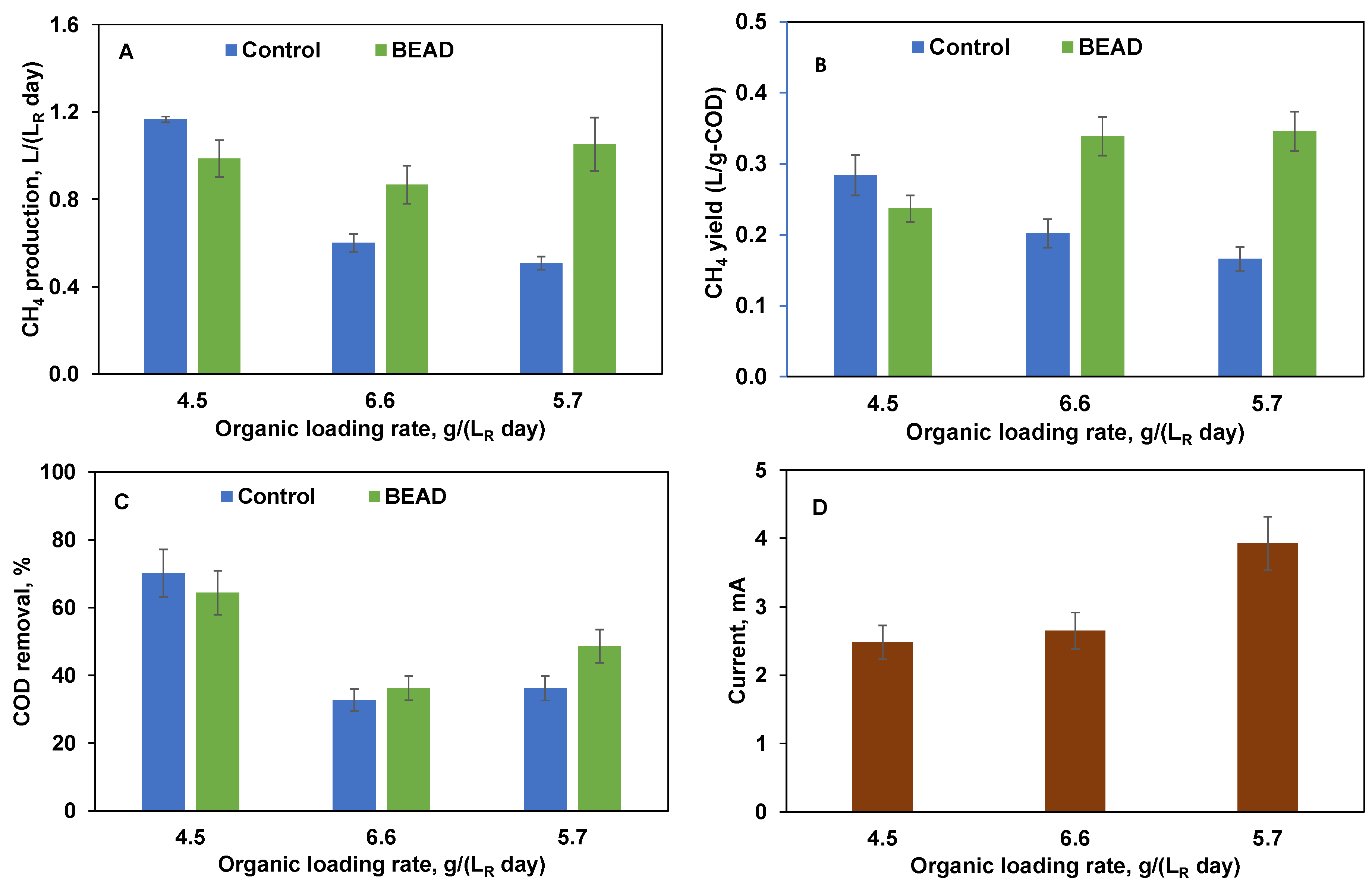
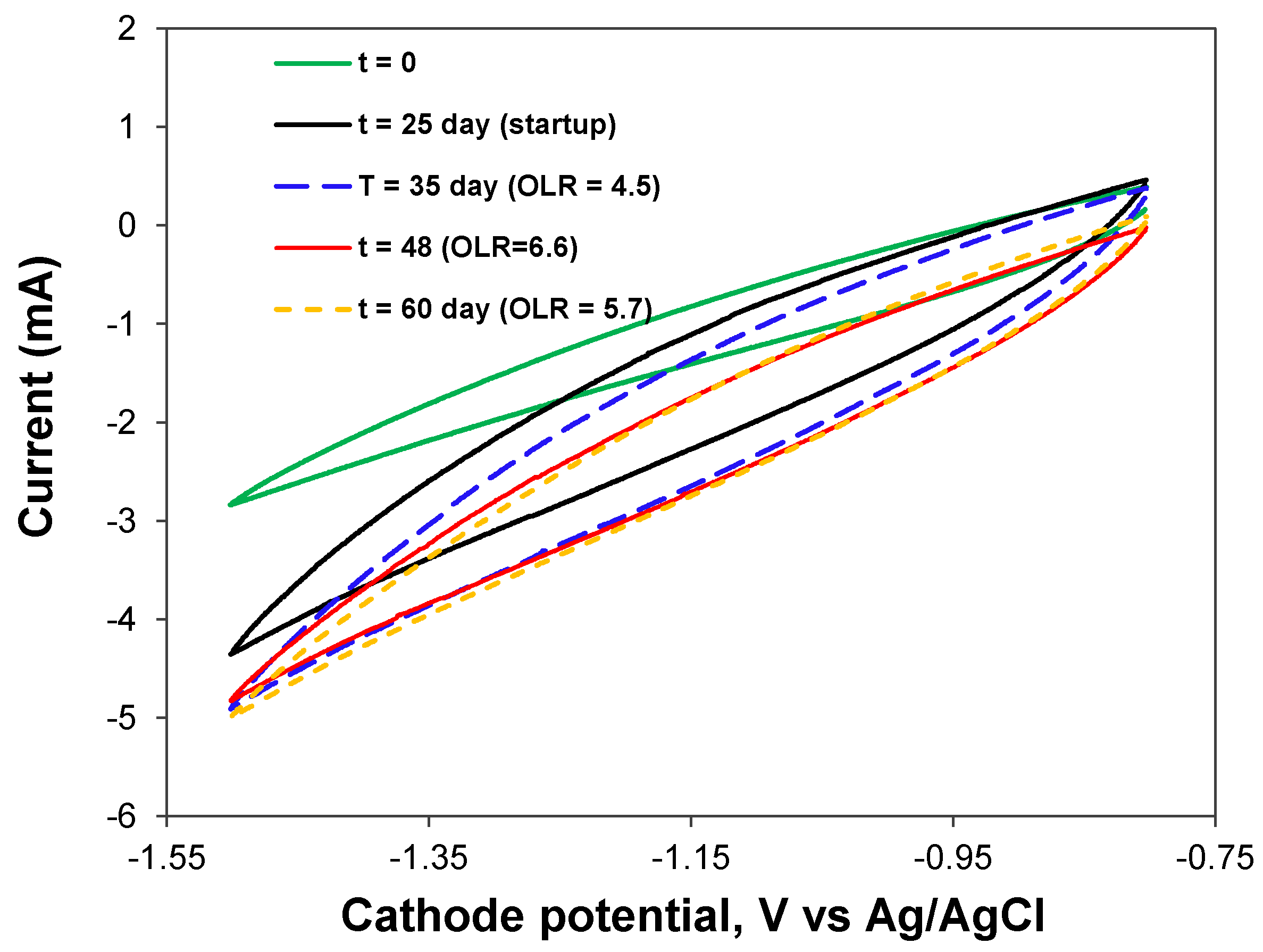
3.2. Biogas Production from SPS Liquid
3.3. Implications of Using BEAD for Food Waste Conversion to Biogas
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De Gioannis, G.; Muntoni, A.; Polettini, A.; Pomi, R.; Spiga, D. Energy recovery from one- and two-stage anaerobic digestion of food waste. Waste Manag. 2017, 68, 595–602. [Google Scholar] [CrossRef]
- International Energy Agency. Outlook for Biogas and Biomethane; International Energy Agency: Paris, France, 2020. [Google Scholar]
- Liu, C.; Wang, W.; Anwar, N.; Ma, Z.; Liu, G.; Zhang, R. Effect of Organic Loading Rate on Anaerobic Digestion of Food Waste under Mesophilic and Thermophilic Conditions. Energy Fuels 2017, 31, 2976–2984. [Google Scholar] [CrossRef]
- Phillip, A.; Bhatt, S.M.; Sharma, N.; Poudel, B. Enhanced Biogas Production Using Anaerobic Co-digestion of Animal Waste and Food Waste: A Review. J. Sci. Res. Rep. 2024, 30, 761–781. [Google Scholar] [CrossRef]
- Lim, B.S.; Kim, B.; Chung, I. Anaerobic Treatment of Food Waste Leachate for Biogas Production Using a Novel Digestion System. Environ. Eng. Res. 2012, 17, 41–46. [Google Scholar] [CrossRef]
- Kim, M.-S.; Kim, D.-H.; Yun, Y.-M. Effect of operation temperature on anaerobic digestion of food waste: Performance and microbial analysis. Fuel 2017, 209, 598–605. [Google Scholar] [CrossRef]
- Rusín, J.; Chamrádová, K.; Basinas, P. Two-stage psychrophilic anaerobic digestion of food waste: Comparison to conventional single-stage mesophilic process. Waste Manag. 2021, 119, 172–182. [Google Scholar] [CrossRef]
- Bi, S.; Hong, X.; Yang, H.; Yu, X.; Fang, S.; Bai, Y.; Liu, J.; Gao, Y.; Yan, L.; Wang, W.; et al. Effect of hydraulic retention time on anaerobic co-digestion of cattle manure and food waste. Renew. Energy 2020, 150, 213–220. [Google Scholar] [CrossRef]
- Ma, J.; Frear, C.; Wang, Z.-W.; Yu, L.; Zhao, Q.; Li, X.; Chen, S. A simple methodology for rate-limiting step determination for anaerobic digestion of complex substrates and effect of microbial community ratio. Bioresour. Technol. 2013, 134, 391–395. [Google Scholar] [CrossRef]
- Yuan, H.; Zhu, N. Progress in inhibition mechanisms and process control of intermediates and by-products in sewage sludge anaerobic digestion. Renew. Sustain. Energy Rev. 2016, 58, 429–438. [Google Scholar] [CrossRef]
- Atelge, M.R.; Atabani, A.E.; Banu, J.R.; Krisa, D.; Kaya, M.; Eskicioglu, C.; Kumar, G.; Lee, C.; Yildiz, Y.Ş.; Unalan, S.; et al. A critical review of pretreatment technologies to enhance anaerobic digestion and energy recovery. Fuel 2020, 270, 117494. [Google Scholar] [CrossRef]
- Chang Bejarano, A.; Champagne, P. Optimization of biogas production during start-up with electrode-assisted anaerobic digestion. Chemosphere 2022, 3012, 134739. [Google Scholar] [CrossRef] [PubMed]
- Bo, T.; Zhu, X.; Zhang, L.; Tao, Y.; He, X.; Li, D.; Yan, Z. A new upgraded biogas production process: Coupling microbial electrolysis cell and anaerobic digestion in single-chamber, barrel-shape stainless steel reactor. Electrochem. Commun. 2014, 45, 67–70. [Google Scholar] [CrossRef]
- De Vrieze, J.; Arends, J.B.A.; Verbeeck, K.; Gildemyn, S.; Rabaey, K. Interfacing anaerobic digestion with (bio)electrochemical systems: Potentials and challenges. Water Res. 2018, 146, 244–255. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, L.; Chen, S.; Buisman, C.J.N.; ter Heijne, A. Bioelectrochemical enhancement of methane production in low temperature anaerobic digestion at 10C. Water Res. 2016, 99, 281–287. [Google Scholar] [CrossRef]
- Ning, X.; Lin, R.; Mao, J.; Deng, C.; Ding, L.; O’Shea, R.; Wall, D.M.; Murphy, J.D. Improving the efficiency of anaerobic digestion and optimising in-situ CO2 bioconversion through the enhanced local electric field at the microbe-electrode interface. Energy Convers. Manag. 2024, 304, 118245. [Google Scholar] [CrossRef]
- Park, J.; Lee, B.; Tian, D.; Jun, H. Bioelectrochemical enhancement of methane production from highly concentrated food waste in a combined anaerobic digester and microbial electrolysis cell. Biores. Technol. 2018, 247, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Villano, M.; Monaco, G.; Aulenta, F.; Majone, M. Electrochemically assisted methane production in a biofilm reactor. J. Power Sources 2011, 196, 9467–9472. [Google Scholar] [CrossRef]
- Wang, W.; Chang, J.-S.; Lee, D.-J. Integrating anaerobic digestion with bioelectrochemical system for performance enhancement: A mini review. Bioresour. Technol. 2022, 345, 126519. [Google Scholar] [CrossRef]
- Huang, Q.; Liu, Y.; Dhar, B.R. A critical review of microbial electrolysis cells coupled with anaerobic digester for enhanced biomethane recovery from high-strength feedstocks. Crit. Rev. Environ. Sci. Technol. 2022, 52, 50–89. [Google Scholar] [CrossRef]
- Clauwaert, P.; Verstraete, W. Methanogenesis in membraneless microbial electrolysis cells. Appl. Microbiol. Biotechnol. 2008, 82, 829–836. [Google Scholar] [CrossRef]
- Liu, W.; Cai, W.; Guo, Z.; Wang, L.; Yang, C.; Varrone, C.; Wang, A. Microbial electrolysis contribution to anaerobic digestion of waste activated sludge, leading to accelerated methane production. Renew. Energ. 2016, 91, 334–339. [Google Scholar] [CrossRef]
- Rader, G.K.; Logan, B.E. Multi-electrode continuous flow microbial electrolysis cell for biogas production from acetate. Int. J. Hydrogen Energy 2010, 35, 8848–8854. [Google Scholar] [CrossRef]
- van Eerten-Jansen, M.; Jansen, N.; Plugge, C.M.; de Wilde, V.; Buisman, C.; ter Heijne, A. Analysis of the mechanisms of bioelectrochemical methane production by mixed cultures. J. Chem. Technol. Biotechnol. 2014, 90, 963–970. [Google Scholar] [CrossRef]
- Rozendal, R.A.; Hamelers, H.V.M.; Rabaey, K.; Keller, J.; Buisman, C.J.N. Towards practical implementation of bioelectrochemical wastewater treatment. Trends Biotechnol. 2008, 26, 450–459. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Xing, D.; Logan, B. Direct biological conversion of electrical current into methane by electromethanogenesis. Environ. Sci. Technol. 2009, 43, 3953–3958. [Google Scholar] [CrossRef] [PubMed]
- Villano, M.; Aulenta, F.; Ciucci, C.; Ferri, T.; Giuliano, A.; Majone, M. Bioelectrochemical reduction of CO2 to CH4 via direct and indirect extracellular electron transfer by a hydrogenophilic methanogenic culture. Bioresour. Technol. 2010, 101, 3085–3090. [Google Scholar] [CrossRef]
- Colantoni, S.; Molognoni, D.; Sánchez-Cueto, P.; De Soto, C.; Bosch-Jimenez, P.; Ghemis, R.; Borràs, E. Bioelectrochemically-improved anaerobic digestion of fishery processing industrial wastewater. J. Water Process Eng. 2024, 65, 105848. [Google Scholar] [CrossRef]
- Flores-Rodriguez, C.; Nagendranatha Reddy, C.; Min, B. Enhanced methane production from acetate intermediate by bioelectrochemical anaerobic digestion at optimal applied voltages. Biomass Bioenergy 2019, 127, 105261. [Google Scholar] [CrossRef]
- Singh, V.; Tartakovsky, B.; Örmeci, B.; Li, H.; Hussain, A. Combining a solid-state submerged fermenter with bioelectrochemically enhanced anaerobic digestion (BEAD) process for enhanced methane (CH4) production from food waste: Effects of the organic loading rates and applied voltages. J. Environ. Chem. Eng. 2025, 13, 116801. [Google Scholar] [CrossRef]
- Liu, Q.; Ren, Z.J.; Huang, C.; Liu, B.; Ren, N.; Xing, D. Multiple syntrophic interactions drive biohythane production from waste sludge in microbial electrolysis cells. Biotechnol. Biofuels 2016, 9, 162. [Google Scholar] [CrossRef] [PubMed]
- Johansen, J.E.; Bakke, R. Enhancing hydrolysis with microaeration. Water Sci. Technol. 2006, 53, 43–50. [Google Scholar] [CrossRef]
- Kim, H.J.; Choi, G.; Lee, C. Enhancing anaerobic digestion of dewatered sewage sludge through thermal hydrolysis pretreatment: Performance evaluation and microbial community analysis. J. Water Process Eng. 2024, 57, 104617. [Google Scholar] [CrossRef]
- Yenigün, O.; Demirel, B. Ammonia inhibition in anaerobic digestion: A review. Process Biochem. 2013, 48, 901–911. [Google Scholar] [CrossRef]
- Zhang, W.; Xing, W.; Li, R. Real-time recovery strategies for volatile fatty acid-inhibited anaerobic digestion of food waste for methane production. Bioresour. Technol. 2018, 265, 82–92. [Google Scholar] [CrossRef]
- Srisowmeya, G.; Chakravarthy, M.; Nandhini Devi, G. Critical considerations in two-stage anaerobic digestion of food waste—A review. Renew. Sustain. Energy Rev. 2020, 119, 109587. [Google Scholar] [CrossRef]
- Radadiya, P.; Lee, J.; Venkateshwaran, K.; Benn, N.; Lee, H.-S.; Hussain, A. Acidogenic fermentation of food waste in a leachate bed reactor (LBR) at high volumetric organic Loading: Effect of granular activated carbon (GAC) and sequential enrichment of inoculum. Bioresour. Technol. 2022, 361, 127705. [Google Scholar] [CrossRef]
- Singh, V.; Örmeci, B.; Singh, A.; Saha, S.; Hussain, A. A novel solid-state submerged fermenter (3SF) for acidogenic fermentation of food waste at high volumetric loading: Effect of inoculum to substrate ratio, design optimization, and inoculum enrichment. Chem. Eng. 2023, 475, 146173. [Google Scholar] [CrossRef]
- Tanguay-Rioux, F.; Spreutels, L.; Roy, C.; Frigon, J.-C. Assessment of the Feasibility of Converting the Liquid Fraction Separated from Fruit and Vegetable Waste in a UASB Digester. Bioengineering 2024, 11, 6. [Google Scholar] [CrossRef]
- González, R.; Ellacuriaga, M.; Aguilar-Pesantes, A.; Carrillo-Peña, D.; García-Cascallana, J.; Smith, R.; Gómez, X. Feasibility of Coupling Anaerobic Digestion and Hydrothermal Carbonization: Analyzing Thermal Demand. Appl. Sci. 2021, 11, 11660. [Google Scholar] [CrossRef]
- Mehari, B.B.; Chang, S.; Hong, Y.; Chen, H. Temperature-Phased Biological Hydrolysis and Thermal Hydrolysis Pretreatment for Anaerobic Digestion Performance Enhancement. Water 2018, 10, 1812. [Google Scholar] [CrossRef]
- Sharma, I.; Rackemann, D.; Ramirez, J.; Cronin, D.J.; Moghaddam, L.; Beltramini, J.N.; Te’O, J.; Li, K.; Shi, C.; Doherty, W.O.S. Exploring the potential for biomethane production by the hybrid anaerobic digestion and hydrothermal gasification process: A review. J. Clean. Prod. 2022, 362, 132507. [Google Scholar] [CrossRef]
- Wang, S.; Han, Y.; Lu, X.; Zhi, Z.; Zhang, R.; Cai, T.; Zhang, Z.; Qin, X.; Song, Y.; Zhen, G. Microbial mechanism underlying high methane production of coupled alkali-microwave-H2O2-oxidation pretreated sewage sludge by in-situ bioelectrochemical regulation. J. Clean. Prod. 2021, 305, 127195. [Google Scholar] [CrossRef]
- Wu, B.; Wang, Y.; He, L.; Liu, M.; Xiang, J.; Chen, Y.; Gu, L.; Li, J.; Li, L.; Pan, W.; et al. Enhancing Anaerobic Digestion of Food Waste by Combining Carriers and Microaeration: Performance and Potential Mechanisms. ACS ES&T Eng. 2024, 4, 2506–2519. [Google Scholar] [CrossRef]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 19th ed.; American Public Health Association: Washington, DC, USA, 1995. [Google Scholar]
- Gil-Carrera, L.; Escapa, A.; Mehta, P.; Santoyo, G.; Guiot, S.R.; Morán, A.; Tartakovsky, B. Microbial electrolysis cell scale-up for combined wastewater treatment and hydrogen production. Biores. Technol. 2012, 130, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Blasco-Gomez, R.; Batlle-Vilanova, P.; Villano, M.; Balaguer, M.D.; Colprim, J.; Puig, S. On the Edge of Research and Technological Application: A Critical Review of Electromethanogenesis. Int. J. Mol. Sci. 2017, 18, 874. [Google Scholar] [CrossRef]
- Kracke, F.; Deutzmann, J.S.; Gu, W.; Spormann, A.M. In situ electrochemical H2 production for efficient and stable power-to-gas electromethanogenesis. Green Chem. 2020, 22, 6194–6203. [Google Scholar] [CrossRef]
- Van Eerten-Jansen, M.; Ter Heijne, A.; Buisman, C.J.N.; Hamelers, H.V.M. Microbial electrolysis cells for production of methane from CO2: Long-term performance and perspectives. Int. J. Energy Res. 2012, 36, 809–819. [Google Scholar] [CrossRef]
- Franco, A.; Giovannini, C. Recent and Future Advances in Water Electrolysis for Green Hydrogen Generation: Critical Analysis and Perspectives. Sustainability 2023, 15, 16917. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, Y.; Jia, H.; Yong, X.; Zhang, L.; Zhou, J.; Cao, Z.; Kruse, A.; Wei, P. Effects of different biofilm carriers on biogas production during anaerobic digestion of corn straw. Bioresour. Technol. 2017, 244, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Habouzit, F.; Hamelin, J.; Santa-Catalina, G.; Steyer, J.P.; Bernet, N. Biofilm development during the start-up period of anaerobic biofilm reactors: The biofilm Archaea community is highly dependent on the support material. Microb. Biotechnol. 2014, 7, 257–264. [Google Scholar] [CrossRef]
- Nevin, K.P.; Woodard, T.L.; Franks, A.E.; Summers, Z.M.; Lovley, D.R. Microbial Electrosynthesis: Feeding Microbes Electricity To Convert Carbon Dioxide and Water to Multicarbon Extracellular Organic Compounds. mBio 2010, 1, e00103-10. [Google Scholar] [CrossRef] [PubMed]
- Xafenias, N.; Mapelli, V. Performance and bacterial enrichment of bioelectrochemical systems during methane and acetate production. Int. J. Hydrogen Energy 2014, 39, 21864–21875. [Google Scholar] [CrossRef]
- Zabranska, J.; Pokorna, D. Bioconversion of carbon dioxide to methane using hydrogen and hydrogenotrophic methanogens. Biotech. Adv. 2018, 36, 707–720. [Google Scholar] [CrossRef]
- Colantoni, S.; Santiago, Ó.; Weiler, J.R.; Knoll, M.T.; Lapp, C.J.; Gescher, J.; Kerzenmacher, S. Comparative study of bioanodes for microbial electrolysis cells operation in anaerobic digester conditions. J. Environ. Chem. Eng. 2024, 12, 113071. [Google Scholar] [CrossRef]
- Liu, C.; Xiao, J.; Li, H.; Chen, Q.; Sun, D.; Cheng, X.; Li, P.; Dang, Y.; Smith, J.A.; Holmes, D.E. High efficiency in-situ biogas upgrading in a bioelectrochemical system with low energy input. Water Res. 2021, 197, 117055. [Google Scholar] [CrossRef]
- Chan, S.; Nishi, K.; Koyama, M.; Toda, T.; Matsuyama, T. Combined effects of various conductive materials and substrates on enhancing methane production performance. Biomass Bioenergy 2023, 178, 106977. [Google Scholar] [CrossRef]
- Li, J.; Xu, X.; Chen, C.; Xu, L.; Du, Z.; Gu, L.; Xiang, P.; Shi, D.; Huangfu, X.; Liu, F. Conductive materials enhance microbial salt-tolerance in anaerobic digestion of food waste: Microbial response and metagenomics analysis. Environ. Res. 2023, 227, 115779. [Google Scholar] [CrossRef]
- De Vrieze, J.; Gildemyn, S.; Arends, J.B.A.; Vanwonterghem, I.; Verbeken, K.; Boon, N.; Verstraete, W.; Tyson, G.W.; Hennebel, T.; Rabaey, K. Biomass retention on electrodes rather than electrical current enhances stability in anaerobic digestion. Water Res. 2014, 54, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Yin, Q.; Zhu, X.; Zhan, G.; Bo, T.; Yang, Y.; Tao, Y.; He, X.; Li, D.; Yan, Z. Enhanced methane production in an anaerobic digestion and microbial electrolysis cell coupled system with co-cultivation of Geobacter and Methanosarcina. J. Environ. Sci. 2016, 42, 210–214. [Google Scholar] [CrossRef]
- Feng, Y.; Zhang, Y.; Chen, S.; Quan, X. Enhanced production of methane from waste activated sludge by the combination of high-solid anaerobic digestion and microbial electrolysis cell with iron–graphite electrode. Chem. Eng. J. 2015, 259, 787–794. [Google Scholar] [CrossRef]
- Zhang, C.; Xu, T.; Feng, H.; Chen, S. Greenhouse Gas Emissions from Landfills: A Review and Bibliometric Analysis. Sustainability 2019, 11, 2282. [Google Scholar] [CrossRef]




| Test # | Location | Feedstock | OLR g (LR d)−1 | HRT Day | Temperature, °C | BEAD Voltage (V) | Duration Day |
|---|---|---|---|---|---|---|---|
| 1-1 | Montreal | FW leachate, centrifuged | 4.3 | 4 | 22 | 1.4 | 16 |
| 1-2 | Montreal | FW leachate, non-centrifuged | 8.3 | 4 | 22 | 1.4 | 18 |
| 1-3 | Montreal | FW leachate, non-centrifuged | 7.7 | 4 | 22 | 1.4 | 13 |
| 2-1 | Ottawa | FW leachate, centrifuged | 3 | 4 | 22 | 1.4 | 12 |
| 2-2 | Ottawa | FW leachate, non-centrifuged | 3 | 4 | 22 | 1.4 | 15 |
| 2-3 | Ottawa | FW leachate, non-centrifuged | 5 | 4 | 22 | 1.4 | 18 |
| 2-4 | Ottawa | FW leachate, non-centrifuged | 7 | 4 | 22 | 1.4 | 23 |
| 3-1 | Montreal | FW SPS liquid | 4.5 | 8.8 | 35 | 1.2 | 8 |
| 3-2 | Montreal | FW SPS liquid | 6.6 | 6.4 | 35 | 1.2 | 12 |
| 3-3 | Montreal | FW SPS liquid | 5.7 | 6.5 | 35 | 1.2 | 6 |
| Test | Substrate | tCOD g L−1 | sCOD g L−1 | OLR (g (LR d)−1 | Acetate mg L−1 | Propionate mg L−1 | Butyrate mg L−1 |
|---|---|---|---|---|---|---|---|
| 1-1 | Centrifuged leachate (Montreal) | n/a | 18.4 | 4.3 | 6510 | 1150 | 3100 |
| 1-2 | Non-centrifuged leachate (Montreal) | 22.8 | 10.6 | 7.7; 8.3 | 3615 | 570 | 1780 |
| 1-3 | |||||||
| 2-1 | Centrifuged leachate (Ottawa) | n/a | 15 | 3 | 4788 | 2268 | 5166 |
| 2-2 | Non-centrifuged leachate (Ottawa) | 15.1 | 10.3 | 3 | 3990 | 1890 | 4305 |
| 2-3 | Non-centrifuged leachate (Ottawa) | 24.8 | 17.4 | 5 | 6597 | 2951 | 7991 |
| 2-4 | Non-centrifuged leachate (Ottawa) | 35.2 | 24.5 | 7 | 9310 | 4165 | 10,291 |
| 3-1, 3-2, 3-3 | SPS liquid (Montreal) | n/a | 16.3 | 4.5–6.6 | 1490 | 648 | 1530 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Singh, V.; Hussain, A.; Örmeci, B.; Pauzé-Foixet, J.; Nwanebu, E.; Li, H.; Tartakovsky, B. High-Rate Bioelectrochemical Anaerobic Digester for Biomethane Production from Food Waste. Bioengineering 2026, 13, 31. https://doi.org/10.3390/bioengineering13010031
Singh V, Hussain A, Örmeci B, Pauzé-Foixet J, Nwanebu E, Li H, Tartakovsky B. High-Rate Bioelectrochemical Anaerobic Digester for Biomethane Production from Food Waste. Bioengineering. 2026; 13(1):31. https://doi.org/10.3390/bioengineering13010031
Chicago/Turabian StyleSingh, Virender, Abid Hussain, Banu Örmeci, Julien Pauzé-Foixet, Emmanuel Nwanebu, Hongbo Li, and Boris Tartakovsky. 2026. "High-Rate Bioelectrochemical Anaerobic Digester for Biomethane Production from Food Waste" Bioengineering 13, no. 1: 31. https://doi.org/10.3390/bioengineering13010031
APA StyleSingh, V., Hussain, A., Örmeci, B., Pauzé-Foixet, J., Nwanebu, E., Li, H., & Tartakovsky, B. (2026). High-Rate Bioelectrochemical Anaerobic Digester for Biomethane Production from Food Waste. Bioengineering, 13(1), 31. https://doi.org/10.3390/bioengineering13010031









