3D Printing for Tissue Engineering: Printing Techniques, Biomaterials, Challenges, and the Emerging Role of 4D Bioprinting
Abstract
1. Introduction
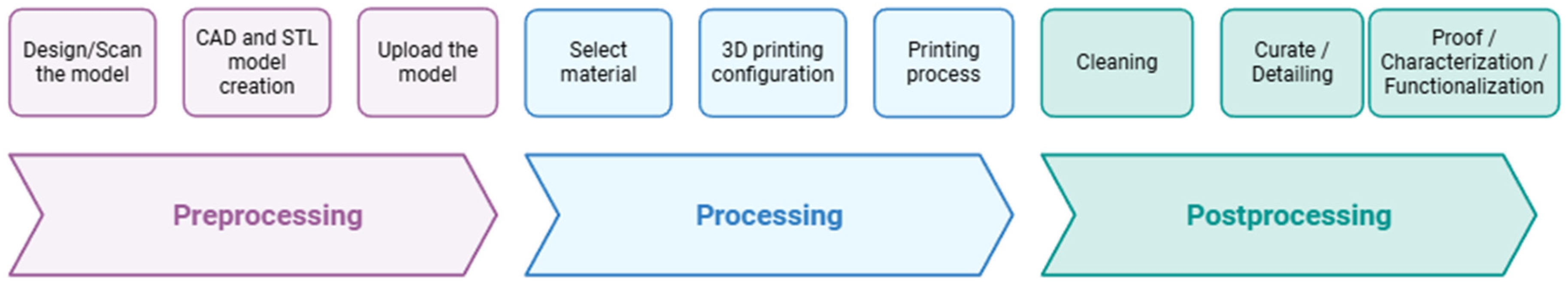
2. Fundamentals of 3D Printing in Tissue Engineering
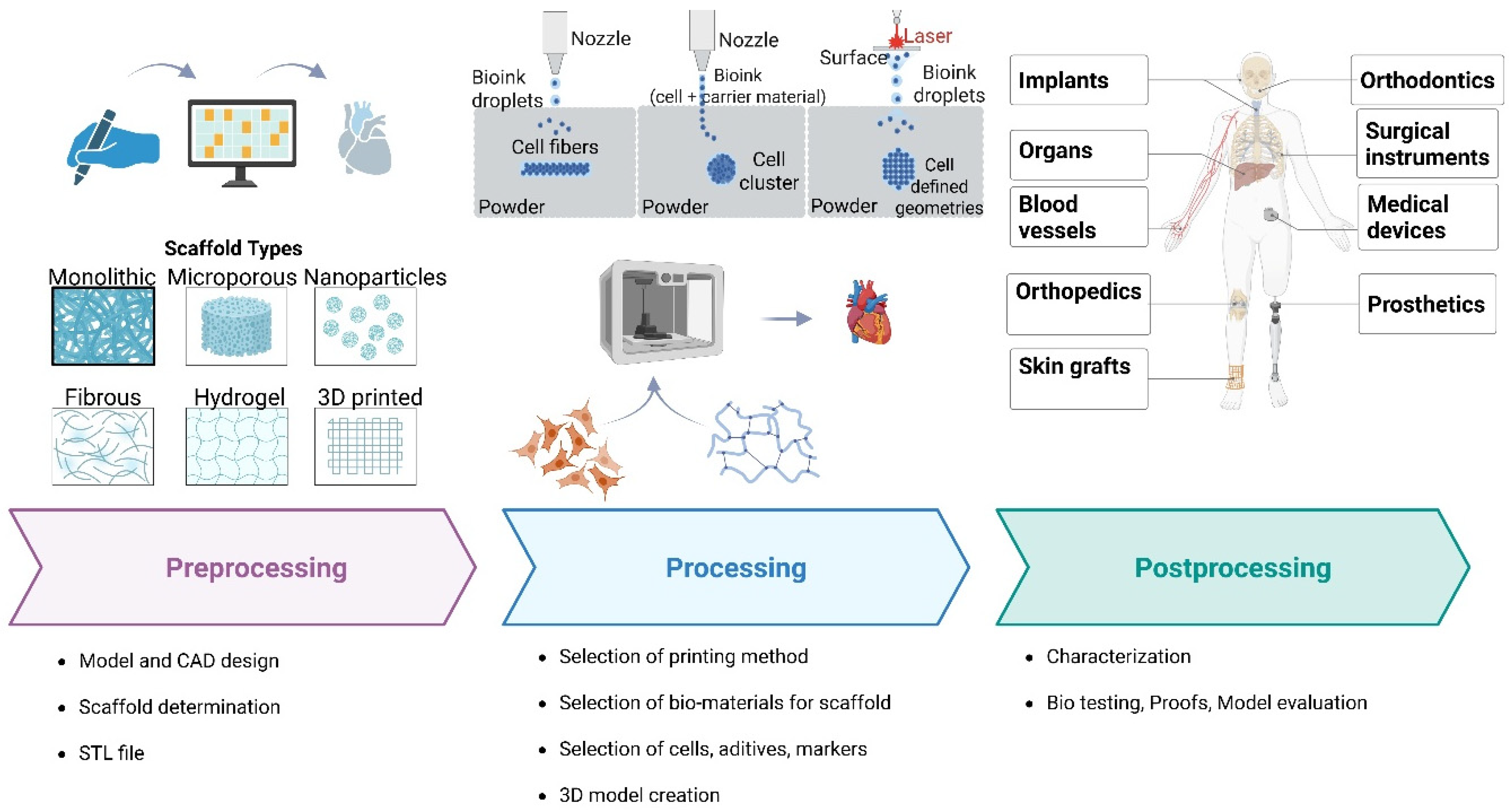
2.1. Three-Dimensional Printing Principle
2.2. Techniques Behind 3D Printing
2.3. Materials for 3D Printing in Tissue Engineering
2.3.1. Natural Polymers
| Polymer | 3DP Techniques | Cell Viability | Properties | Limitations | Reference |
|---|---|---|---|---|---|
| Natural | |||||
| Chitosan | Extrusion | ~75% | Highly biocompatible. | Low mechanical strength | [6] |
| Collagen/gelatin | Extrusion Laser additive bioprinting | 70–99% | Supports cell–matrix interactions and dissolves in water at body temperature. | - | [59,60,61] |
| Agar/agarose | Extrusion | 60–90% | Facilitates printing due to its low viscosity and wide gelling temperature range. | - | [59,62] |
| Alginate | Extrusion | 77–100% | Exhibits biocompatibility and enables rapid ionic cross-linking. | - | [59,63] |
| Fibrin | Extrusion | 74–100% | Offers blood-clotting properties and strong tissue adhesion. | Limited long-term stability | [59,60] |
| Hyaluronic acid | Extrusion | 64.4% | High viscosity and hydrated environment. | Requires modification to enable gelation | [30,64,65] |
| Starch | Inkjet Printing | 70–90% | Forms viscous paste when heated. | - | [66,67] |
| Synthetic | |||||
| PGA, PLA, and PLGA | Stereolithography Extrusion | 70–90% | The degradation rate can be adjusted based on the ratio of comonomers and molecular weight, which contains hydrolysable ester bonds. | - | [59,68,69] |
| PCL | Stereolithography Laser Additive Bioprinting Extrusion | 70–95% | Well-established biocompatibility and safety within the body at a rate that aligns with the formation of new bone tissue. | Slow degradation rate (1 to 3 years in vivo conditions), limited bioactivity. | [59,68,70] |
| PVA | Laser additive bioprinting | 60–85% | Offers high mechanical strength and minimal thermal degradation. | Limited cell adhesion (60–100%) | [27,59] |
| PEG derivatives | Stereolithography Extrusion | 80–95% | Well-established biocompatibility offers a hydrophilic and highly bioinert surface and can also be covalently cross-linked. | Limited bioactivity | [59,63,68,69] |
2.3.2. Hydrogels
2.3.3. Synthetic Polymers
2.3.4. Bio-Inks
2.4. Parameters for 3D Printing in Tissue Engineering
2.4.1. Design Inputs: General Considerations, Materials, and Cell Types
2.4.2. Scaffold Properties to Consider
2.4.3. Selection of Biomaterials
2.4.4. Biological Interactions and Cell Types
2.4.5. Print Outputs: Infill, Resolution, Porous Architecture
2.4.6. Print Resolution
2.4.7. Porous Architecture
2.4.8. Assessment of Scaffold Fidelity
3. Three-Dimensional Printing Applications in Tissue Engineering
3.1. Dermal Regeneration
3.2. Bone Tissues
3.3. Cardiovascular Tissues
3.4. Neural Regeneration
4. Advantages of 3D Printing in Tissue Engineering
4.1. Quality and Reproducibility in Complicated Fabrication Systems
4.2. Development Prototypes with Features That Can Be Rapidly Modified
4.3. Providing Cells with Excellent Precision
4.4. Engineering in Highly Controllable Microenvironments
4.5. Patterning of Multiple Inks
5. Present Technical Challenges and Deficiencies in 3D Printing
5.1. Biocompatibility and Quality of Materials
5.2. Selection of the Materials
5.3. Enhancements in Cell Behavior of Bioprinted Structures
5.4. Regulation and Ethical Issues
6. Four-Dimensional Bioprinting for Biomedical Applications: Engineering Responsive and Dynamic Living Systems
7. Comparative Analysis of 3D and 4D Printing Technologies for Biomedical Applications and Tissue Engineering
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Deng, C.; Chang, J.; Wu, C. Bioactive scaffolds for osteochondral regeneration. J. Orthop. Transl. 2019, 17, 15–25. [Google Scholar] [CrossRef]
- Jammalamadaka, U.; Tappa, K. Recent Advances in Biomaterials for 3D Printing and Tissue Engineering. J. Funct. Biomater. 2018, 9, 22. [Google Scholar] [CrossRef] [PubMed]
- Mabrouk, M.; Beherei, H.H.; Das, D.B. Recent progress in the fabrication techniques of 3D scaffolds for tissue engineering. Mater. Sci. Eng. C 2020, 110, 110716. [Google Scholar] [CrossRef]
- Han, F.; Wang, J.; Ding, L.; Hu, Y.; Li, W.; Yuan, Z.; Guo, Q.; Zhu, C.; Yu, L.; Wang, H.; et al. Tissue Engineering and Regenerative Medicine: Achievements, Future, and Sustainability in Asia. Front. Bioeng. Biotechnol. 2020, 8, 83. [Google Scholar] [CrossRef]
- Do, A.; Khorsand, B.; Geary, S.M.; Salem, A.K. 3D Printing of Scaffolds for Tissue Regeneration Applications. Adv. Healthc. Mater. 2015, 4, 1742–1762. [Google Scholar] [CrossRef]
- Lai, J.; Wang, C.; Wang, M. 3D printing in biomedical engineering: Processes, materials, and applications. Appl. Phys. Rev. 2021, 8, 021322. [Google Scholar] [CrossRef]
- Zaszczyńska, A.; Moczulska-Heljak, M.; Gradys, A.; Sajkiewicz, P. Advances in 3D Printing for Tissue Engineering. Materials 2021, 14, 3149. [Google Scholar] [CrossRef]
- Seoane-Viaño, I.; Januskaite, P.; Alvarez-Lorenzo, C.; Basit, A.W.; Goyanes, A. Semi-solid extrusion 3D printing in drug delivery and biomedicine: Personalised solutions for healthcare challenges. J. Controlled Release 2021, 332, 367–389. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Forgham, H.; Shen, A.; Wang, J.; Zhu, J.; Huang, X.; Tang, S.-Y.; Xu, C.; Davis, T.P.; Qiao, R. Nanomaterial integrated 3D printing for biomedical applications. J. Mater. Chem. B 2022, 10, 7473–7490. [Google Scholar] [CrossRef]
- Lee, V.; Singh, G.; Trasatti, J.P.; Bjornsson, C.; Xu, X.; Tran, T.N.; Yoo, S.-S.; Dai, G.; Karande, P. Design and Fabrication of Human Skin by Three-Dimensional Bioprinting. Tissue Eng. Part C Methods 2014, 20, 473–484. [Google Scholar] [CrossRef]
- Markstedt, K.; Mantas, A.; Tournier, I.; Martínez Ávila, H.; Hägg, D.; Gatenholm, P. 3D Bioprinting Human Chondrocytes with Nanocellulose–Alginate Bioink for Cartilage Tissue Engineering Applications. Biomacromolecules 2015, 16, 1489–1496. [Google Scholar] [CrossRef]
- Cui, H.; Zhu, W.; Nowicki, M.; Zhou, X.; Khademhosseini, A.; Zhang, L.G. Hierarchical Fabrication of Engineered Vascularized Bone Biphasic Constructs via Dual 3D Bioprinting: Integrating Regional Bioactive Factors into Architectural Design. Adv. Healthc. Mater. 2016, 5, 2174–2181. [Google Scholar] [CrossRef]
- Laronda, M.M.; Rutz, A.L.; Xiao, S.; Whelan, K.A.; Duncan, F.E.; Roth, E.W.; Woodruff, T.K.; Shah, R.N. A bioprosthetic ovary created using 3D printed microporous scaffolds restores ovarian function in sterilized mice. Nat. Commun. 2017, 8, 15261. [Google Scholar] [CrossRef]
- Pavan Kalyan, B.; Kumar, L. 3D Printing: Applications in Tissue Engineering, Medical Devices, and Drug Delivery. AAPS PharmSciTech 2022, 23, 92. [Google Scholar] [CrossRef]
- Thompson, M.K.; Moroni, G.; Vaneker, T.; Fadel, G.; Campbell, R.I.; Gibson, I.; Bernard, A.; Schulz, J.; Graf, P.; Ahuja, B.; et al. Design for Additive Manufacturing: Trends, opportunities, considerations, and constraints. CIRP Ann. 2016, 65, 737–760. [Google Scholar] [CrossRef]
- Tetsuka, H.; Shin, S.R. Materials and technical innovations in 3D printing in biomedical applications. J. Mater. Chem. B 2020, 8, 2930–2950. [Google Scholar] [CrossRef]
- Halper, J. Narrative Review and Guide: State of the Art and Emerging Opportunities of Bioprinting in Tissue Regeneration and Medical Instrumentation. Bioengineering 2025, 12, 71. [Google Scholar] [CrossRef] [PubMed]
- Mirsky, N.A.; Ehlen, Q.T.; Greenfield, J.A.; Antonietti, M.; Slavin, B.V.; Nayak, V.V.; Pelaez, D.; Tse, D.T.; Witek, L.; Daunert, S.; et al. Three-Dimensional Bioprinting: A Comprehensive Review for Applications in Tissue Engineering and Regenerative Medicine. Bioengineering 2024, 11, 777. [Google Scholar] [CrossRef] [PubMed]
- Ji, S.; Guvendiren, M. Recent Advances in Bioink Design for 3D Bioprinting of Tissues and Organs. Front. Bioeng. Biotechnol. 2017, 5, 23. [Google Scholar] [CrossRef] [PubMed]
- Surmen, H.K.; Ortes, F.; Arslan, Y.Z. Fundamentals of 3D Printing and Its Applications in Biomedical Engineering. In 3D Printing in Biomedical Engineering; Singh, S., Prakash, C., Singh, R., Eds.; Materials Horizons: From Nature to Nanomaterials; Springer: Singapore, 2020; pp. 23–41. [Google Scholar] [CrossRef]
- Xing, J.-F.; Zheng, M.-L.; Duan, X.-M. Two-photon polymerization microfabrication of hydrogels: An advanced 3D printing technology for tissue engineering and drug delivery. Chem. Soc. Rev. 2015, 44, 5031–5039. [Google Scholar] [CrossRef]
- Mott, E.J.; Busso, M.; Luo, X.; Dolder, C.; Wang, M.O.; Fisher, J.P.; Dean, D. Digital micromirror device (DMD)-based 3D printing of poly(propylene fumarate) scaffolds. Mater. Sci. Eng. C 2016, 61, 301–311. [Google Scholar] [CrossRef]
- Hong, H.; Seo, Y.B.; Kim, D.Y.; Lee, J.S.; Lee, Y.J.; Lee, H.; Ajiteru, O.; Sultan, M.T.; Lee, O.J.; Kim, S.H.; et al. Digital light processing 3D printed silk fibroin hydrogel for cartilage tissue engineering. Biomaterials 2020, 232, 119679. [Google Scholar] [CrossRef]
- Adamidis, O.; Alber, S.; Anastasopoulos, I. Investigation into 3D printing of granular media. In Physical Modelling in Geotechnics; McNamara, A., Divall, S., Goodey, R., Taylor, N., Stallebrass, S., Panchal, J., Eds.; CRC Press: Boca Raton, FL, USA, 2018; pp. 113–118. [Google Scholar] [CrossRef]
- Tan, J.Y.; Chua, C.K.; Leong, K.F. Indirect fabrication of gelatin scaffolds using rapid prototyping technology. Virtual Phys. Prototyp. 2010, 5, 45–53. [Google Scholar] [CrossRef]
- Singh, R. Process capability study of polyjet printing for plastic components. J. Mech. Sci. Technol. 2011, 25, 1011–1015. [Google Scholar] [CrossRef]
- Goyanes, A.; Wang, J.; Buanz, A.; Martínez-Pacheco, R.; Telford, R.; Gaisford, S.; Basit, A.W. 3D Printing of Medicines: Engineering Novel Oral Devices with Unique Design and Drug Release Characteristics. Mol. Pharm. 2015, 12, 4077–4084. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.; Radacsi, N.; Robert, C.; McCarthy, E.D.; Callanan, A.; Conlisk, N.; Hoskins, P.R.; Koutsos, V. On the optimization of low-cost FDM 3D printers for accurate replication of patient-specific abdominal aortic aneurysm geometry. 3D Print. Med. 2018, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.H.Y.; Naficy, S.; Yue, Z.; Kapsa, R.; Quigley, A.; Moulton, S.E.; Wallace, G.G. Bio-ink properties and printability for extrusion printing living cells. Biomater. Sci. 2013, 1, 763. [Google Scholar] [CrossRef] [PubMed]
- Panwar, A.; Tan, L. Current Status of Bioinks for Micro-Extrusion-Based 3D Bioprinting. Molecules 2016, 21, 685. [Google Scholar] [CrossRef]
- Russias, J.; Saiz, E.; Deville, S.; Gryn, K.; Liu, G.; Nalla, R.K.; Tomsia, A.P. Fabrication and in vitro characterization of three-dimensional organic/inorganic scaffolds by robocasting. J. Biomed. Mater. Res. A 2007, 83A, 434–445. [Google Scholar] [CrossRef] [PubMed]
- Dorj, B.; Park, J.-H.; Kim, H.-W. Robocasting chitosan/nanobioactive glass dual-pore structured scaffolds for bone engineering. Mater. Lett. 2012, 73, 119–122. [Google Scholar] [CrossRef]
- Eqtesadi, S.; Motealleh, A.; Pajares, A.; Miranda, P. Effect of milling media on processing and performance of 13-93 bioactive glass scaffolds fabricated by robocasting. Ceram. Int. 2015, 41, 1379–1389. [Google Scholar] [CrossRef]
- Shao, H.; Liu, A.; Ke, X.; Sun, M.; He, Y.; Yang, X.; Fu, J.; Zhang, L.; Yang, G.; Liu, Y.; et al. 3D robocasting magnesium-doped wollastonite/TCP bioceramic scaffolds with improved bone regeneration capacity in critical sized calvarial defects. J. Mater. Chem. B 2017, 5, 2941–2951. [Google Scholar] [CrossRef]
- Lei, L.; Wei, Y.; Wang, Z.; Han, J.; Sun, J.; Chen, Y.; Yang, X.; Wu, Y.; Chen, L.; Gou, Z. Core–Shell Bioactive Ceramic Robocasting: Tuning Component Distribution Beneficial for Highly Efficient Alveolar Bone Regeneration and Repair. ACS Biomater. Sci. Eng. 2020, 6, 2376–2387. [Google Scholar] [CrossRef]
- Cesarano, J. A Review of Robocasting Technology. MRS Proc. 1998, 542, 133. [Google Scholar] [CrossRef]
- Duan, B.; Wang, M. Selective laser sintering and its application in biomedical engineering. MRS Bull. 2011, 36, 998–1005. [Google Scholar] [CrossRef]
- Gayer, C.; Ritter, J.; Bullemer, M.; Grom, S.; Jauer, L.; Meiners, W.; Pfister, A.; Reinauer, F.; Vučak, M.; Wissenbach, K.; et al. Development of a solvent-free polylactide/calcium carbonate composite for selective laser sintering of bone tissue engineering scaffolds. Mater. Sci. Eng. C 2019, 101, 660–673. [Google Scholar] [CrossRef]
- Chua, C.K.; Yeong, W.Y. Bioprinting: Principles and Applications; World Scientific: Singapore, 2015. [Google Scholar] [CrossRef]
- Zhu, W.; Qu, X.; Zhu, J.; Ma, X.; Patel, S.; Liu, J.; Wang, P.; Lai, C.S.E.; Gou, M.; Xu, Y.; et al. Direct 3D bioprinting of prevascularized tissue constructs with complex microarchitecture. Biomaterials 2017, 124, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Bedell, M.L.; Navara, A.M.; Du, Y.; Zhang, S.; Mikos, A.G. Polymeric Systems for Bioprinting. Chem. Rev. 2020, 120, 10744–10792. [Google Scholar] [CrossRef] [PubMed]
- Gautam, S.; Chou, C.-F.; Dinda, A.K.; Potdar, P.D.; Mishra, N.C. Surface modification of nanofibrous polycaprolactone/gelatin composite scaffold by collagen type I grafting for skin tissue engineering. Mater. Sci. Eng. C 2014, 34, 402–409. [Google Scholar] [CrossRef]
- Li, W.-J.; Tuli, R.; Okafor, C.; Derfoul, A.; Danielson, K.G.; Hall, D.J.; Tuan, R.S. A three-dimensional nanofibrous scaffold for cartilage tissue engineering using human mesenchymal stem cells. Biomaterials 2005, 26, 599–609. [Google Scholar] [CrossRef]
- Li, X.; Feng, Q.; Wang, W.; Cui, F. Chemical characteristics and cytocompatibility of collagen-based scaffold reinforced by chitin fibers for bone tissue engineering. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 77B, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, S.; Ang, L.-T.; Cho-Hong Goh, J.; Toh, S.-L. Bioactive nanofibers for fibroblastic differentiation of mesenchymal precursor cells for ligament/tendon tissue engineering applications. Differentiation 2010, 79, 102–110. [Google Scholar] [CrossRef]
- Hu, J.; Sun, X.; Ma, H.; Xie, C.; Chen, Y.E.; Ma, P.X. Porous nanofibrous PLLA scaffolds for vascular tissue engineering. Biomaterials 2010, 31, 7971–7977. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Morshed, M.; Nasr-Esfahani, M.-H.; Ramakrishna, S. Electrospun poly(ɛ-caprolactone)/gelatin nanofibrous scaffolds for nerve tissue engineering. Biomaterials 2008, 29, 4532–4539. [Google Scholar] [CrossRef]
- Kim, B.-S.; Park, I.-K.; Hoshiba, T.; Jiang, H.-L.; Choi, Y.-J.; Akaike, T.; Cho, C.-S. Design of artificial extracellular matrices for tissue engineering. Prog. Polym. Sci. 2011, 36, 238–268. [Google Scholar] [CrossRef]
- Khan, M.U.A.; Aslam, M.A.; Bin Abdullah, M.F.; Hasan, A.; Shah, S.A.; Stojanović, G.M. Recent perspective of polymeric biomaterial in tissue engineering—A review. Mater. Today Chem. 2023, 34, 101818. [Google Scholar] [CrossRef]
- Khan, F.; Ahmad, S.R. Polysaccharides and Their Derivatives for Versatile Tissue Engineering Application. Macromol. Biosci. 2013, 13, 395–421. [Google Scholar] [CrossRef]
- Lee, H.M.; Fischer, K.M. Cost-effective synthesis of polyethylene glycol diacrylate-acrylic acid (PEGDA-AA), a tissue engineering hydrogel ideal for C2C12 mouse muscle cells. FASEB J. 2019, 33, 493.6. [Google Scholar] [CrossRef]
- Gayathri, V.; Khan, T.; Gowtham, M.; Balan, R.; Sebaey, T.A. Functionalized conductive polymer composites for tissue engineering and biomedical applications—A mini review. Front. Bioeng. Biotechnol. 2025, 13, 1533944. [Google Scholar] [CrossRef]
- Li, X.; Feng, Q.; Liu, X.; Dong, W.; Cui, F. Collagen-based implants reinforced by chitin fibres in a goat shank bone defect model. Biomaterials 2006, 27, 1917–1923. [Google Scholar] [CrossRef]
- Serrano, M.C.; Nardecchia, S.; García-Rama, C.; Ferrer, M.L.; Collazos-Castro, J.E.; Del Monte, F.; Gutiérrez, M.C. Chondroitin sulphate-based 3D scaffolds containing MWCNTs for nervous tissue repair. Biomaterials 2014, 35, 1543–1551. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, X.; Dong, W.; Feng, Q.; Cui, F.; Uo, M.; Akasaka, T.; Watari, F. In vitro evaluation of porous poly(L-lactic acid) scaffold reinforced by chitin fibers. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 90B, 503–509. [Google Scholar] [CrossRef]
- Tamura, H.; Furuike, T.; Nair, S.V.; Jayakumar, R. Biomedical applications of chitin hydrogel membranes and scaffolds. Carbohydr. Polym. 2011, 84, 820–824. [Google Scholar] [CrossRef]
- Islam, A.; Yasin, T.; Rehman, I.U. Synthesis of hybrid polymer networks of irradiated chitosan/poly(vinyl alcohol) for biomedical applications. Radiat. Phys. Chem. 2014, 96, 115–119. [Google Scholar] [CrossRef]
- Li, X.; Feng, Q.; Jiao, Y.; Cui, F. Collagen-based scaffolds reinforced by chitosan fibres for bone tissue engineering. Polym. Int. 2005, 54, 1034–1040. [Google Scholar] [CrossRef]
- Chia, H.N.; Wu, B.M. Recent advances in 3D printing of biomaterials. J. Biol. Eng. 2015, 9, 4. [Google Scholar] [CrossRef]
- Wan, J. Microfluidic-Based Synthesis of Hydrogel Particles for Cell Microencapsulation and Cell-Based Drug Delivery. Polymers 2012, 4, 1084–1108. [Google Scholar] [CrossRef]
- Savsani, K.; Aitchison, A.H.; Allen, N.B.; Adams, E.A.; Adams, S.B. The Use of Gelatin Methacrylate (GelMA) in Cartilage Tissue Engineering: A Comprehensive Review. Bioengineering 2025, 12, 700. [Google Scholar] [CrossRef]
- Wei, J.; Wang, J.; Su, S.; Wang, S.; Qiu, J.; Zhang, Z.; Christopher, G.; Ning, F.; Cong, W. 3D printing of an extremely tough hydrogel. RSC Adv. 2015, 5, 81324–81329. [Google Scholar] [CrossRef]
- Hong, S.; Sycks, D.; Chan, H.F.; Lin, S.; Lopez, G.P.; Guilak, F.; Leong, K.W.; Zhao, X. 3D Printing: 3D Printing of Highly Stretchable and Tough Hydrogels into Complex, Cellularized Structures. Adv. Mater. 2015, 27, 4034. [Google Scholar] [CrossRef]
- Ouyang, L.; Highley, C.B.; Rodell, C.B.; Sun, W.; Burdick, J.A. 3D Printing of Shear-Thinning Hyaluronic Acid Hydrogels with Secondary Cross-Linking. ACS Biomater. Sci. Eng. 2016, 2, 1743–1751. [Google Scholar] [CrossRef] [PubMed]
- Highley, C.B.; Song, K.H.; Daly, A.C.; Burdick, J.A. Jammed Microgel Inks for 3D Printing Applications. Adv. Sci. 2019, 6, 1801076. [Google Scholar] [CrossRef] [PubMed]
- Lam, C.X.F.; Mo, X.M.; Teoh, S.H.; Hutmacher, D.W. Scaffold development using 3D printing with a starch-based polymer. Mater. Sci. Eng. C 2002, 20, 49–56. [Google Scholar] [CrossRef]
- Ventola, C.L. Medical Applications for 3D Printing: Current and Projected Uses. Pharm. Ther. 2014, 39, 704–711. [Google Scholar]
- Bose, S.; Vahabzadeh, S.; Bandyopadhyay, A. Bone tissue engineering using 3D printing. Mater. Today 2013, 16, 496–504. [Google Scholar] [CrossRef]
- Liu, X.; Ma, P.X. Polymeric Scaffolds for Bone Tissue Engineering. Ann. Biomed. Eng. 2004, 32, 477–486. [Google Scholar] [CrossRef]
- Temple, J.P.; Hutton, D.L.; Hung, B.P.; Huri, P.Y.; Cook, C.A.; Kondragunta, R.; Jia, X.; Grayson, W.L. Engineering anatomically shaped vascularized bone grafts with hASCs and 3D-printed PCL scaffolds: Engineering Anatomically Shaped Vascularized Bone Grafts. J. Biomed. Mater. Res. A 2014, 102, 4317–4325. [Google Scholar] [CrossRef]
- Singh, S.R. Principles of Regenerative Medicine; Atala, A., Lanza, R., Thompson, J., Nerem, R., Eds.; Elsevier Inc.: New York, NY, USA, 2008; p. 1448. ISBN 978-0-12-369410-2. [Google Scholar]
- Slaughter, B.V.; Khurshid, S.S.; Fisher, O.Z.; Khademhosseini, A.; Peppas, N.A. Hydrogels in Regenerative Medicine. Adv. Mater. 2009, 21, 3307–3329. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.P.; Leong, K.W. Scaffolding in tissue engineering: General approaches and tissue-specific considerations. Eur. Spine J. 2008, 17, 467–479. [Google Scholar] [CrossRef]
- Anderson, J.M. Biological Responses to Materials. Annu. Rev. Mater. Res. 2001, 31, 81–110. [Google Scholar] [CrossRef]
- Saroia, J.; Yanen, W.; Wei, Q.; Zhang, K.; Lu, T.; Zhang, B. A review on biocompatibility nature of hydrogels with 3D printing techniques, tissue engineering application and its future prospective. Bio-Des. Manuf. 2018, 1, 265–279. [Google Scholar] [CrossRef]
- Zein, I.; Hutmacher, D.W.; Tan, K.C.; Teoh, S.H. Fused deposition modeling of novel scaffold architectures for tissue engineering applications. Biomaterials 2002, 23, 1169–1185. [Google Scholar] [CrossRef]
- Gregor, A.; Filová, E.; Novák, M.; Kronek, J.; Chlup, H.; Buzgo, M.; Blahnová, V.; Lukášová, V.; Bartoš, M.; Nečas, A.; et al. Designing of PLA scaffolds for bone tissue replacement fabricated by ordinary commercial 3D printer. J. Biol. Eng. 2017, 11, 31. [Google Scholar] [CrossRef]
- Jain, R.A. The manufacturing techniques of various drug loaded biodegradable poly(lactide-co-glycolide) (PLGA) devices. Biomaterials 2000, 21, 2475–2490. [Google Scholar] [CrossRef]
- Dong, L.; Wang, S.-J.; Zhao, X.-R.; Zhu, Y.-F.; Yu, J.-K. 3D-Printed Poly(ε-caprolactone) Scaffold Integrated with Cell-laden Chitosan Hydrogels for Bone Tissue Engineering. Sci. Rep. 2017, 7, 13412. [Google Scholar] [CrossRef] [PubMed]
- Luis, E.; Pan, H.M.; Bastola, A.K.; Bajpai, R.; Sing, S.L.; Song, J.; Yeong, W.Y. 3D Printed Silicone Meniscus Implants: Influence of the 3D Printing Process on Properties of Silicone Implants. Polymers 2020, 12, 2136. [Google Scholar] [CrossRef]
- Tan, Y.; Tan, X.; Yeong, W.; Tor, S. Additive Manufacturing of Patient-Customizable Scaffolds for Tubular Tissues Using the Melt-Drawing Method. Materials 2016, 9, 893. [Google Scholar] [CrossRef]
- Haleem, A.; Javaid, M. Polyether ether ketone (PEEK) and its 3D printed implants applications in medical field: An overview. Clin. Epidemiol. Glob. Health 2019, 7, 571–577. [Google Scholar] [CrossRef]
- Groll, J.; Burdick, J.A.; Cho, D.-W.; Derby, B.; Gelinsky, M.; Heilshorn, S.C.; Jüngst, T.; Malda, J.; Mironov, V.A.; Nakayama, K.; et al. A definition of bioinks and their distinction from biomaterial inks. Biofabrication 2018, 11, 013001. [Google Scholar] [CrossRef]
- Levato, R.; Webb, W.R.; Otto, I.A.; Mensinga, A.; Zhang, Y.; Van Rijen, M.; Van Weeren, R.; Khan, I.M.; Malda, J. The bio in the ink: Cartilage regeneration with bioprintable hydrogels and articular cartilage-derived progenitor cells. Acta Biomater. 2017, 61, 41–53. [Google Scholar] [CrossRef]
- Fatimi, A.; Okoro, O.V.; Podstawczyk, D.; Siminska-Stanny, J.; Shavandi, A. Natural Hydrogel-Based Bio-Inks for 3D Bioprinting in Tissue Engineering: A Review. Gels 2022, 8, 179. [Google Scholar] [CrossRef]
- Hospodiuk, M.; Dey, M.; Sosnoski, D.; Ozbolat, I.T. The bioink: A comprehensive review on bioprintable materials. Biotechnol. Adv. 2017, 35, 217–239. [Google Scholar] [CrossRef]
- Donderwinkel, I.; Van Hest, J.C.M.; Cameron, N.R. Bio-inks for 3D bioprinting: Recent advances and future prospects. Polym. Chem. 2017, 8, 4451–4471. [Google Scholar] [CrossRef]
- Bedell, M.L.; Guo, J.L.; Xie, V.Y.; Navara, A.M.; Mikos, A.G. Polymer scaffold fabrication. In Principles of Tissue Engineering; Elsevier: New York, NY, USA, 2020; pp. 295–315. [Google Scholar] [CrossRef]
- Muschler, G.F.; Nakamoto, C.; Griffith, L.G. Engineering Principles of clinical cell-based tissue engineering. J. Bone Jt. Surg.-Am. Vol. 2004, 86, 1541–1558. [Google Scholar] [CrossRef] [PubMed]
- Velema, J.; Kaplan, D. Biopolymer-Based Biomaterials as Scaffolds for Tissue Engineering. In Tissue Engineering I; Lee, K., Kaplan, D., Eds.; Advances in Biochemical Engineering/Biotechnology; Springer: Berlin/Heidelberg, Germany, 2006; Volume 102, pp. 187–238. [Google Scholar] [CrossRef]
- Rickert, D.; Lendlein, A.; Peters, I.; Moses, M.A.; Franke, R.-P. Biocompatibility testing of novel multifunctional polymeric biomaterials for tissue engineering applications in head and neck surgery: An overview. Eur. Arch. Otorhinolaryngol. 2006, 263, 215–222. [Google Scholar] [CrossRef]
- Bittner, S.M.; Guo, J.L.; Melchiorri, A.; Mikos, A.G. Three-dimensional printing of multilayered tissue engineering scaffolds. Mater. Today 2018, 21, 861–874. [Google Scholar] [CrossRef] [PubMed]
- Rana, D.; Arulkumar, S.; Vishwakarma, A.; Ramalingam, M. Considerations on Designing Scaffold for Tissue Engineering. In Stem Cell Biology and Tissue Engineering in Dental Sciences; Elsevier: New York, NY, USA, 2015; pp. 133–148. [Google Scholar] [CrossRef]
- Mikos, A.G.; Herring, S.W.; Ochareon, P.; Elisseeff, J.; Lu, H.H.; Kandel, R.; Schoen, F.J.; Toner, M.; Mooney, D.; Atala, A.; et al. Engineering Complex Tissues. Tissue Eng. 2006, 12, 3307–3339. [Google Scholar] [CrossRef] [PubMed]
- Potter, C.M.F.; Lao, K.H.; Zeng, L.; Xu, Q. Role of Biomechanical Forces in Stem Cell Vascular Lineage Differentiation. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 2184–2190. [Google Scholar] [CrossRef] [PubMed]
- Tibbitt, M.W.; Anseth, K.S. Hydrogels as extracellular matrix mimics for 3D cell culture. Biotechnol. Bioeng. 2009, 103, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Lutolf, M.P.; Hubbell, J.A. Synthetic biomaterials as instructive extracellular microenvironments for morphogenesis in tissue engineering. Nat. Biotechnol. 2005, 23, 47–55. [Google Scholar] [CrossRef]
- Bini, F.; D’Alessandro, S.; Pica, A.; Marinozzi, F.; Cidonio, G. Harnessing Biofabrication Strategies to Re-Surface Osteochondral Defects: Repair, Enhance, and Regenerate. Biomimetics 2023, 8, 260. [Google Scholar] [CrossRef] [PubMed]
- Bittner, S.M.; Guo, J.L.; Mikos, A.G. Spatiotemporal control of growth factors in three-dimensional printed scaffolds. Bioprinting 2018, 12, e00032. [Google Scholar] [CrossRef] [PubMed]
- Jose, R.R.; Rodriguez, M.J.; Dixon, T.A.; Omenetto, F.; Kaplan, D.L. Evolution of Bioinks and Additive Manufacturing Technologies for 3D Bioprinting. ACS Biomater. Sci. Eng. 2016, 2, 1662–1678. [Google Scholar] [CrossRef] [PubMed]
- Koch, L.; Kuhn, S.; Sorg, H.; Gruene, M.; Schlie, S.; Gaebel, R.; Polchow, B.; Reimers, K.; Stoelting, S.; Ma, N.; et al. Laser Printing of Skin Cells and Human Stem Cells. Tissue Eng. Part C Methods 2010, 16, 847–854. [Google Scholar] [CrossRef]
- Koons, G.L.; Mikos, A.G. Progress in three-dimensional printing with growth factors. J. Controlled Release 2019, 295, 50–59. [Google Scholar] [CrossRef]
- Pataky, K.; Braschler, T.; Negro, A.; Renaud, P.; Lutolf, M.P.; Brugger, J. Microdrop Printing of Hydrogel Bioinks into 3D Tissue-Like Geometries. Adv. Mater. 2012, 24, 391–396. [Google Scholar] [CrossRef]
- Schuurman, W.; Khristov, V.; Pot, M.W.; Van Weeren, P.R.; Dhert, W.J.A.; Malda, J. Bioprinting of hybrid tissue constructs with tailorable mechanical properties. Biofabrication 2011, 3, 021001. [Google Scholar] [CrossRef]
- Cohen, D.L.; Malone, E.; Lipson, H.; Bonassar, L.J. Direct Freeform Fabrication of Seeded Hydrogels in Arbitrary Geometries. Tissue Eng. 2006, 12, 1325–1335. [Google Scholar] [CrossRef]
- Wu, C.; Luo, Y.; Cuniberti, G.; Xiao, Y.; Gelinsky, M. Three-dimensional printing of hierarchical and tough mesoporous bioactive glass scaffolds with a controllable pore architecture, excellent mechanical strength and mineralization ability. Acta Biomater. 2011, 7, 2644–2650. [Google Scholar] [CrossRef]
- Zhang, J.; Zhao, S.; Zhu, Y.; Huang, Y.; Zhu, M.; Tao, C.; Zhang, C. Three-dimensional printing of strontium-containing mesoporous bioactive glass scaffolds for bone regeneration. Acta Biomater. 2014, 10, 2269–2281. [Google Scholar] [CrossRef]
- Choudhury, D.; Anand, S.; Naing, M.W. The arrival of commercial bioprinters—Towards 3D bioprinting revolution! Int. J. Bioprinting 2024, 4, 139. [Google Scholar] [CrossRef]
- Derakhshanfar, S.; Mbeleck, R.; Xu, K.; Zhang, X.; Zhong, W.; Xing, M. 3D bioprinting for biomedical devices and tissue engineering: A review of recent trends and advances. Bioact. Mater. 2018, 3, 144–156. [Google Scholar] [CrossRef]
- Zafaryab, M.; Vig, K. Three-Dimensional Printing of Hydrogel as Skin Substitute and Comparative Evaluation of Melanin Production. Bioengineering 2025, 12, 270. [Google Scholar] [CrossRef]
- Daikuara, L.Y.; Chen, X.; Yue, Z.; Skropeta, D.; Wood, F.M.; Fear, M.W.; Wallace, G.G. 3D Bioprinting Constructs to Facilitate Skin Regeneration. Adv. Funct. Mater. 2022, 32, 2105080. [Google Scholar] [CrossRef]
- Ma, J.; Qin, C.; Wu, J.; Zhang, H.; Zhuang, H.; Zhang, M.; Zhang, Z.; Ma, L.; Wang, X.; Ma, B.; et al. 3D Printing of Strontium Silicate Microcylinder-Containing Multicellular Biomaterial Inks for Vascularized Skin Regeneration. Adv. Healthc. Mater. 2021, 10, 2100523. [Google Scholar] [CrossRef] [PubMed]
- Afghah, F.; Ullah, M.; Seyyed Monfared Zanjani, J.; Akkuş Süt, P.; Sen, O.; Emanet, M.; Saner Okan, B.; Culha, M.; Menceloglu, Y.; Yildiz, M.; et al. 3D printing of silver-doped polycaprolactone-poly propylene succinate composite scaffolds for skin tissue engineering. Biomed. Mater. 2020, 15, 035015. [Google Scholar] [CrossRef]
- Singh, M.; Jonnalagadda, S. Design and characterization of 3D printed, neomycin-eluting poly-L-lactide mats for wound-healing applications. J. Mater. Sci. Mater. Med. 2021, 32, 44. [Google Scholar] [CrossRef] [PubMed]
- Pourchet, L.J.; Thepot, A.; Albouy, M.; Courtial, E.J.; Boher, A.; Blum, L.J.; Marquette, C.A. Human Skin 3D Bioprinting Using Scaffold-Free Approach. Adv. Healthc. Mater. 2017, 6, 1601101. [Google Scholar] [CrossRef]
- Cubo, N.; Garcia, M.; Del Cañizo, J.F.; Velasco, D.; Jorcano, J.L. 3D bioprinting of functional human skin: Production and in vivo analysis. Biofabrication 2016, 9, 015006. [Google Scholar] [CrossRef]
- Zhao, X.; Lang, Q.; Yildirimer, L.; Lin, Z.Y.; Cui, W.; Annabi, N.; Ng, K.W.; Dokmeci, M.R.; Ghaemmaghami, A.M.; Khademhosseini, A. Photocrosslinkable Gelatin Hydrogel for Epidermal Tissue Engineering. Adv. Healthc. Mater. 2016, 5, 108–118. [Google Scholar] [CrossRef]
- Ng, W.L.; Yeong, W.Y.; Naing, M.W. Polyelectrolyte gelatin-chitosan hydrogel optimized for 3D bioprinting in skin tissue engineering. Int. J. Bioprinting 2024, 2, 53. [Google Scholar] [CrossRef]
- Ashammakhi, N.; Hasan, A.; Kaarela, O.; Byambaa, B.; Sheikhi, A.; Gaharwar, A.K.; Khademhosseini, A. Advancing Frontiers in Bone Bioprinting. Adv. Healthc. Mater. 2019, 8, 1801048. [Google Scholar] [CrossRef]
- Leukers, B.; Gülkan, H.; Irsen, S.H.; Milz, S.; Tille, C.; Schieker, M.; Seitz, H. Hydroxyapatite scaffolds for bone tissue engineering made by 3D printing. J. Mater. Sci. Mater. Med. 2005, 16, 1121–1124. [Google Scholar] [CrossRef]
- Zhang, J.; Zhao, S.; Zhu, M.; Zhu, Y.; Zhang, Y.; Liu, Z.; Zhang, C. 3D-printed magnetic Fe3O4/MBG/PCL composite scaffolds with multifunctionality of bone regeneration, local anticancer drug delivery and hyperthermia. J. Mater. Chem. B 2014, 2, 7583–7595. [Google Scholar] [CrossRef] [PubMed]
- Lan, W.; Huang, X.; Huang, D.; Wei, X.; Chen, W. Progress in 3D printing for bone tissue engineering: A review. J. Mater. Sci. 2022, 57, 12685–12709. [Google Scholar] [CrossRef]
- Pahlevanzadeh, F.; Emadi, R.; Valiani, A.; Kharaziha, M.; Poursamar, S.A.; Bakhsheshi-Rad, H.R.; Ismail, A.F.; RamaKrishna, S.; Berto, F. Three-Dimensional Printing Constructs Based on the Chitosan for Tissue Regeneration: State of the Art, Developing Directions and Prospect Trends. Materials 2020, 13, 2663. [Google Scholar] [CrossRef] [PubMed]
- Fradique, R.; Correia, T.R.; Miguel, S.P.; De Sá, K.D.; Figueira, D.R.; Mendonça, A.G.; Correia, I.J. Production of new 3D scaffolds for bone tissue regeneration by rapid prototyping. J. Mater. Sci. Mater. Med. 2016, 27, 69. [Google Scholar] [CrossRef]
- Kim, H.; Yang, G.H.; Choi, C.H.; Cho, Y.S.; Kim, G. Gelatin/PVA scaffolds fabricated using a 3D-printing process employed with a low-temperature plate for hard tissue regeneration: Fabrication and characterizations. Int. J. Biol. Macromol. 2018, 120, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Heo, D.N.; Castro, N.J.; Lee, S.-J.; Noh, H.; Zhu, W.; Zhang, L.G. Enhanced bone tissue regeneration using a 3D printed microstructure incorporated with a hybrid nano hydrogel. Nanoscale 2017, 9, 5055–5062. [Google Scholar] [CrossRef] [PubMed]
- Sudarmadji, N.; Tan, J.Y.; Leong, K.F.; Chua, C.K.; Loh, Y.T. Investigation of the mechanical properties and porosity relationships in selective laser-sintered polyhedral for functionally graded scaffolds. Acta Biomater. 2011, 7, 530–537. [Google Scholar] [CrossRef]
- Seyednejad, H.; Vermonden, T.; Fedorovich, N.E.; Van Eijk, R.; Van Steenbergen, M.J.; Dhert, W.J.A.; Van Nostrum, C.F.; Hennink, W.E. Synthesis and Characterization of Hydroxyl-Functionalized Caprolactone Copolymers and Their Effect on Adhesion, Proliferation, and Differentiation of Human Mesenchymal Stem Cells. Biomacromolecules 2009, 10, 3048–3054. [Google Scholar] [CrossRef]
- Seok, J.M.; Jeong, J.E.; Lee, S.J.; Im, S.H.; Lee, J.H.; Kim, W.D.; Lee, K.; Park, S.A. Bio-plotted hydrogel scaffold with core and sheath strand-enhancing mechanical and biological properties for tissue regeneration. Colloids Surf. B Biointerfaces 2021, 205, 111919. [Google Scholar] [CrossRef]
- Sithole, M.N.; Kumar, P.; Du Toit, L.C.; Marimuthu, T.; Choonara, Y.E.; Pillay, V. A 3D bioprinted in situ conjugated- co -fabricated scaffold for potential bone tissue engineering applications. J. Biomed. Mater. Res. A 2018, 106, 1311–1321. [Google Scholar] [CrossRef]
- Heo, E.Y.; Ko, N.R.; Bae, M.S.; Lee, S.J.; Choi, B.-J.; Kim, J.H.; Kim, H.K.; Park, S.A.; Kwon, I.K. Novel 3D printed alginate–BFP1 hybrid scaffolds for enhanced bone regeneration. J. Ind. Eng. Chem. 2017, 45, 61–67. [Google Scholar] [CrossRef]
- Choudhury, S.; Joshi, A.; Agrawal, A.; Nain, A.; Bagde, A.; Patel, A.; Syed, Z.Q.; Asthana, S.; Chatterjee, K. NIR-Responsive Deployable and Self-Fitting 4D-Printed Bone Tissue Scaffold. ACS Appl. Mater. Interfaces 2024, 16, 49135–49147. [Google Scholar] [CrossRef]
- Ni, X.; Cui, Y.; Salehi, M.; Nai, M.L.S.; Zhou, K.; Vyas, C.; Huang, B.; Bartolo, P. Piezoelectric Biomaterials for Bone Regeneration: Roadmap from Dipole to Osteogenesis. Adv. Sci. 2025, e14969. [Google Scholar] [CrossRef]
- Zheng, T.; Huang, Y.; Zhang, X.; Cai, Q.; Deng, X.; Yang, X. Mimicking the electrophysiological microenvironment of bone tissue using electroactive materials to promote its regeneration. J. Mater. Chem. B 2020, 8, 10221–10256. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Ye, X.; Yao, B.; Zhao, M.; Wu, P.; Liu, G.; Zhuang, D.; Jiang, H.; Chen, X.; He, Y.; et al. Advances in 3D bioprinting technology for cardiac tissue engineering and regeneration. Bioact. Mater. 2021, 6, 1388–1401. [Google Scholar] [CrossRef] [PubMed]
- Jabri, A.; Taftafa, B.; Mhannayeh, A.; Alsharif, M.; Abbad, T.; Ahmed, S.; Alshehri, E.A.; Elsalti, A.; Khan, J.; Mir, T.A.; et al. Cardiac Tissue Engineering for Translational Cardiology: From In Vitro Models to Regenerative Therapies. Bioengineering 2025, 12, 518. [Google Scholar] [CrossRef]
- Goo, H.W.; Park, S.J.; Yoo, S.-J. Advanced Medical Use of Three-Dimensional Imaging in Congenital Heart Disease: Augmented Reality, Mixed Reality, Virtual Reality, and Three-Dimensional Printing. Korean J. Radiol. 2020, 21, 133. [Google Scholar] [CrossRef] [PubMed]
- Al Jabbari, O.; Abu Saleh, W.K.; Patel, A.P.; Igo, S.R.; Reardon, M.J. Use of three-dimensional models to assist in the resection of malignant cardiac tumors. J. Card. Surg. 2016, 31, 581–583. [Google Scholar] [CrossRef]
- Gardin, C.; Ferroni, L.; Latremouille, C.; Chachques, J.C.; Mitrečić, D.; Zavan, B. Recent Applications of Three Dimensional Printing in Cardiovascular Medicine. Cells 2020, 9, 742. [Google Scholar] [CrossRef]
- Lee, A.; Hudson, A.R.; Shiwarski, D.J.; Tashman, J.W.; Hinton, T.J.; Yerneni, S.; Bliley, J.M.; Campbell, P.G.; Feinberg, A.W. 3D bioprinting of collagen to rebuild components of the human heart. Science 2019, 365, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.S.; Arneri, A.; Bersini, S.; Shin, S.-R.; Zhu, K.; Goli-Malekabadi, Z.; Aleman, J.; Colosi, C.; Busignani, F.; Dell’Erba, V.; et al. Bioprinting 3D microfibrous scaffolds for engineering endothelialized myocardium and heart-on-a-chip. Biomaterials 2016, 110, 45–59. [Google Scholar] [CrossRef]
- Yeong, W.Y.; Sudarmadji, N.; Yu, H.Y.; Chua, C.K.; Leong, K.F.; Venkatraman, S.S.; Boey, Y.C.F.; Tan, L.P. Porous polycaprolactone scaffold for cardiac tissue engineering fabricated by selective laser sintering. Acta Biomater. 2010, 6, 2028–2034. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.; Gungor-Ozkerim, P.S.; Zhang, Y.S.; Yue, K.; Zhu, K.; Liu, W.; Pi, Q.; Byambaa, B.; Dokmeci, M.R.; Shin, S.R.; et al. Direct 3D bioprinting of perfusable vascular constructs using a blend bioink. Biomaterials 2016, 106, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Jin, Q.; Fu, Y.; Zhang, G.; Xu, L.; Jin, G.; Tang, L.; Ju, J.; Zhao, W.; Hou, R. Nanofiber Electrospinning Combined with Rotary Bioprinting for Fabricating Small-Diameter Vessels with Endothelium and Smooth Muscle. Compos. Part B Eng. 2022, 234, 109691. [Google Scholar] [CrossRef]
- Hann, S.Y.; Cui, H.; Esworthy, T.; Zhou, X.; Lee, S.; Plesniak, M.W.; Zhang, L.G. Dual 3D printing for vascularized bone tissue regeneration. Acta Biomater. 2021, 123, 263–274. [Google Scholar] [CrossRef]
- Liu, S.; Sun, L.; Zhang, H.; Hu, Q.; Wang, Y.; Ramalingam, M. High-resolution combinatorial 3D printing of gelatin-based biomimetic triple-layered conduits for nerve tissue engineering. Int. J. Biol. Macromol. 2021, 166, 1280–1291. [Google Scholar] [CrossRef]
- Ye, W.; Li, H.; Yu, K.; Xie, C.; Wang, P.; Zheng, Y.; Zhang, P.; Xiu, J.; Yang, Y.; Zhang, F.; et al. 3D printing of gelatin methacrylate-based nerve guidance conduits with multiple channels. Mater. Des. 2020, 192, 108757. [Google Scholar] [CrossRef]
- Tao, J.; Hu, Y.; Wang, S.; Zhang, J.; Liu, X.; Gou, Z.; Cheng, H.; Liu, Q.; Zhang, Q.; You, S.; et al. A 3D-engineered porous conduit for peripheral nerve repair. Sci. Rep. 2017, 7, 46038. [Google Scholar] [CrossRef] [PubMed]
- Dilla, R.A.; Motta, C.M.M.; Snyder, S.R.; Wilson, J.A.; Wesdemiotis, C.; Becker, M.L. Synthesis and 3D Printing of PEG–Poly(propylene fumarate) Diblock and Triblock Copolymer Hydrogels. ACS Macro Lett. 2018, 7, 1254–1260. [Google Scholar] [CrossRef]
- Chen, J.; Huang, D.; Wang, L.; Hou, J.; Zhang, H.; Li, Y.; Zhong, S.; Wang, Y.; Wu, Y.; Huang, W. 3D bioprinted multiscale composite scaffolds based on gelatin methacryloyl (GelMA)/chitosan microspheres as a modular bioink for enhancing 3D neurite outgrowth and elongation. J. Colloid Interface Sci. 2020, 574, 162–173. [Google Scholar] [CrossRef]
- Zhu, W.; Harris, B.T.; Zhang, L.G. Gelatin methacrylamide hydrogel with graphene nanoplatelets for neural cell-laden 3D bioprinting. In Proceedings of the 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 16–20 August 2016; IEEE: Orlando, FL, USA, 2016; pp. 4185–4188. [Google Scholar] [CrossRef]
- Lee, Y.-B.; Polio, S.; Lee, W.; Dai, G.; Menon, L.; Carroll, R.S.; Yoo, S.-S. Bio-printing of collagen and VEGF-releasing fibrin gel scaffolds for neural stem cell culture. Exp. Neurol. 2010, 223, 645–652. [Google Scholar] [CrossRef]
- Tan, B.; Gan, S.; Wang, X.; Liu, W.; Li, X. Applications of 3D bioprinting in tissue engineering: Advantages, deficiencies, improvements, and future perspectives. J. Mater. Chem. B 2021, 9, 5385–5413. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, V.; Gonçalves, I.; Lage, T.; Rodrigues, R.O.; Minas, G.; Teixeira, S.F.C.F.; Moita, A.S.; Hori, T.; Kaji, H.; Lima, R.A. 3D Printing Techniques and Their Applications to Organ-on-a-Chip Platforms: A Systematic Review. Sensors 2021, 21, 3304. [Google Scholar] [CrossRef]
- Vijayavenkataraman, S.; Yan, W.-C.; Lu, W.F.; Wang, C.-H.; Fuh, J.Y.H. 3D bioprinting of tissues and organs for regenerative medicine. Adv. Drug Deliv. Rev. 2018, 132, 296–332. [Google Scholar] [CrossRef]
- Rider, P.; Kačarević, Ž.P.; Alkildani, S.; Retnasingh, S.; Barbeck, M. Bioprinting of tissue engineering scaffolds. J. Tissue Eng. 2018, 9, 2041731418802090. [Google Scholar] [CrossRef]
- Howard, D.; Buttery, L.D.; Shakesheff, K.M.; Roberts, S.J. Tissue engineering: Strategies, stem cells and scaffolds. J. Anat. 2008, 213, 66–72. [Google Scholar] [CrossRef]
- Hockaday, L.A.; Kang, K.H.; Colangelo, N.W.; Cheung, P.Y.C.; Duan, B.; Malone, E.; Wu, J.; Girardi, L.N.; Bonassar, L.J.; Lipson, H.; et al. Rapid 3D printing of anatomically accurate and mechanically heterogeneous aortic valve hydrogel scaffolds. Biofabrication 2012, 4, 035005. [Google Scholar] [CrossRef] [PubMed]
- Seol, Y.-J.; Kang, H.-W.; Lee, S.J.; Atala, A.; Yoo, J.J. Bioprinting technology and its applications. Eur. J. Cardiothorac. Surg. 2014, 46, 342–348. [Google Scholar] [CrossRef]
- Griffith, L.G.; Naughton, G. Tissue Engineering--Current Challenges and Expanding Opportunities. Science 2002, 295, 1009–1014. [Google Scholar] [CrossRef]
- Agarwal, S.; Saha, S.; Balla, V.K.; Pal, A.; Barui, A.; Bodhak, S. Current Developments in 3D Bioprinting for Tissue and Organ Regeneration–A Review. Front. Mech. Eng. 2020, 6, 589171. [Google Scholar] [CrossRef]
- Kim, J.E.; Kim, S.H.; Jung, Y. Current status of three-dimensional printing inks for soft tissue regeneration. Tissue Eng. Regen. Med. 2016, 13, 636–646. [Google Scholar] [CrossRef]
- Mandrycky, C.; Wang, Z.; Kim, K.; Kim, D.-H. 3D bioprinting for engineering complex tissues. Biotechnol. Adv. 2016, 34, 422–434. [Google Scholar] [CrossRef]
- Wang, Z.; Abdulla, R.; Parker, B.; Samanipour, R.; Ghosh, S.; Kim, K. A simple and high-resolution stereolithography-based 3D bioprinting system using visible light crosslinkable bioinks. Biofabrication 2015, 7, 045009. [Google Scholar] [CrossRef]
- Koivisto, J.T.; Gering, C.; Karvinen, J.; Maria Cherian, R.; Belay, B.; Hyttinen, J.; Aalto-Setälä, K.; Kellomäki, M.; Parraga, J. Mechanically Biomimetic Gelatin–Gellan Gum Hydrogels for 3D Culture of Beating Human Cardiomyocytes. ACS Appl. Mater. Interfaces 2019, 11, 20589–20602. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Leong, K.-F.; Du, Z.; Chua, C.-K. The Design of Scaffolds for Use in Tissue Engineering. Part I. Traditional Factors. Tissue Eng. 2001, 7, 679–689. [Google Scholar] [CrossRef]
- Jones, N. Science in three dimensions: The print revolution. Nature 2012, 487, 22–23. [Google Scholar] [CrossRef] [PubMed]
- Fedorovich, N.E.; De Wijn, J.R.; Verbout, A.J.; Alblas, J.; Dhert, W.J.A. Three-Dimensional Fiber Deposition of Cell-Laden, Viable, Patterned Constructs for Bone Tissue Printing. Tissue Eng. Part A 2008, 14, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Peltola, S.M.; Melchels, F.P.W.; Grijpma, D.W.; Kellomäki, M. A review of rapid prototyping techniques for tissue engineering purposes. Ann. Med. 2008, 40, 268–280. [Google Scholar] [CrossRef]
- Marga, F.; Jakab, K.; Khatiwala, C.; Shephard, B.; Dorfman, S.; Forgacs, G. Organ Printing: A Novel Tissue Engineering Paradigm. In Proceedings of the 5th European Conference of the International Federation for Medical and Biological Engineering, Budapest, Hungary, 14–18 September 2011; Jobbágy, Á., Ed.; IFMBE Proceedings; Springer: Berlin/Heidelberg, Germany, 2011; Volume 37, pp. 27–30. [Google Scholar] [CrossRef]
- Luu, Y.K.; Kim, K.; Hsiao, B.S.; Chu, B.; Hadjiargyrou, M. Development of a nanostructured DNA delivery scaffold via electrospinning of PLGA and PLA–PEG block copolymers. J. Controlled Release 2003, 89, 341–353. [Google Scholar] [CrossRef] [PubMed]
- Sodhi, H.; Panitch, A. Glycosaminoglycans in Tissue Engineering: A Review. Biomolecules 2020, 11, 29. [Google Scholar] [CrossRef] [PubMed]
- Loh, Q.L.; Choong, C. Three-Dimensional Scaffolds for Tissue Engineering Applications: Role of Porosity and Pore Size. Tissue Eng. Part B Rev. 2013, 19, 485–502. [Google Scholar] [CrossRef]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar] [CrossRef] [PubMed]
- Chevalier, E.; Chulia, D.; Pouget, C.; Viana, M. Fabrication of porous substrates: A review of processes using pore forming agents in the biomaterial field. J. Pharm. Sci. 2008, 97, 1135–1154. [Google Scholar] [CrossRef]
- Yeong, W.-Y.; Chua, C.-K.; Leong, K.-F.; Chandrasekaran, M. Rapid prototyping in tissue engineering: Challenges and potential. Trends Biotechnol. 2004, 22, 643–652. [Google Scholar] [CrossRef]
- Koch, L.; Deiwick, A.; Schlie, S.; Michael, S.; Gruene, M.; Coger, V.; Zychlinski, D.; Schambach, A.; Reimers, K.; Vogt, P.M.; et al. Skin tissue generation by laser cell printing. Biotechnol. Bioeng. 2012, 109, 1855–1863. [Google Scholar] [CrossRef]
- Guillotin, B.; Souquet, A.; Catros, S.; Duocastella, M.; Pippenger, B.; Bellance, S.; Bareille, R.; Rémy, M.; Bordenave, L.; Amédée, J.; et al. Laser assisted bioprinting of engineered tissue with high cell density and microscale organization. Biomaterials 2010, 31, 7250–7256. [Google Scholar] [CrossRef]
- Kirillova, A.; Maxson, R.; Stoychev, G.; Gomillion, C.T.; Ionov, L. 4D Biofabrication Using Shape-Morphing Hydrogels. Adv. Mater. 2017, 29, 1703443. [Google Scholar] [CrossRef]
- Hollister, S.J. Porous scaffold design for tissue engineering. Nat. Mater. 2005, 4, 518–524. [Google Scholar] [CrossRef]
- Hardin, J.O.; Ober, T.J.; Valentine, A.D.; Lewis, J.A. Microfluidic Printheads for Multimaterial 3D Printing of Viscoelastic Inks. Adv. Mater. 2015, 27, 3279–3284. [Google Scholar] [CrossRef]
- Atala, A.; Kasper, F.K.; Mikos, A.G. Engineering Complex Tissues. Sci. Transl. Med. 2012, 4, 160rv12. [Google Scholar] [CrossRef]
- Murphy, S.V.; Atala, A. 3D bioprinting of tissues and organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.N.; Lancaster, K.Z.; Zhen, G.; He, J.; Gupta, M.K.; Kong, Y.L.; Engel, E.A.; Krick, K.D.; Ju, A.; Meng, F.; et al. 3D Printed Anatomical Nerve Regeneration Pathways. Adv. Funct. Mater. 2015, 25, 6205–6217. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.K.; Meng, F.; Johnson, B.N.; Kong, Y.L.; Tian, L.; Yeh, Y.-W.; Masters, N.; Singamaneni, S.; McAlpine, M.C. 3D Printed Programmable Release Capsules. Nano Lett. 2015, 15, 5321–5329. [Google Scholar] [CrossRef]
- Xie, Z.; Gao, M.; Lobo, A.O.; Webster, T.J. 3D Bioprinting in Tissue Engineering for Medical Applications: The Classic and the Hybrid. Polymers 2020, 12, 1717. [Google Scholar] [CrossRef]
- Zhu, W.; Ma, X.; Gou, M.; Mei, D.; Zhang, K.; Chen, S. 3D printing of functional biomaterials for tissue engineering. Curr. Opin. Biotechnol. 2016, 40, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Connell, J.L.; Ritschdorff, E.T.; Whiteley, M.; Shear, J.B. 3D printing of microscopic bacterial communities. Proc. Natl. Acad. Sci. USA 2013, 110, 18380–18385. [Google Scholar] [CrossRef]
- Soman, P.; Chung, P.H.; Zhang, A.P.; Chen, S. Digital microfabrication of user-defined 3D microstructures in cell-laden hydrogels. Biotechnol. Bioeng. 2013, 110, 3038–3047. [Google Scholar] [CrossRef]
- Tumbleston, J.R.; Shirvanyants, D.; Ermoshkin, N.; Janusziewicz, R.; Johnson, A.R.; Kelly, D.; Chen, K.; Pinschmidt, R.; Rolland, J.P.; Ermoshkin, A.; et al. Continuous liquid interface production of 3D objects. Science 2015, 347, 1349–1352. [Google Scholar] [CrossRef]
- Ousterout, D.G.; Perez-Pinera, P.; Thakore, P.I.; Kabadi, A.M.; Brown, M.T.; Qin, X.; Fedrigo, O.; Mouly, V.; Tremblay, J.P.; Gersbach, C.A. Reading Frame Correction by Targeted Genome Editing Restores Dystrophin Expression in Cells from Duchenne Muscular Dystrophy Patients. Mol. Ther. 2013, 21, 1718–1726. [Google Scholar] [CrossRef]
- Zhang, X.; Xu, B.; Puperi, D.S.; Yonezawa, A.L.; Wu, Y.; Tseng, H.; Cuchiara, M.L.; West, J.L.; Grande-Allen, K.J. Integrating valve-inspired design features into poly(ethylene glycol) hydrogel scaffolds for heart valve tissue engineering. Acta Biomater. 2015, 14, 11–21. [Google Scholar] [CrossRef]
- Shin, S.R.; Jung, S.M.; Zalabany, M.; Kim, K.; Zorlutuna, P.; Kim, S.B.; Nikkhah, M.; Khabiry, M.; Azize, M.; Kong, J.; et al. Carbon-Nanotube-Embedded Hydrogel Sheets for Engineering Cardiac Constructs and Bioactuators. ACS Nano 2013, 7, 2369–2380. [Google Scholar] [CrossRef] [PubMed]
- Kim, P.; Yuan, A.; Nam, K.-H.; Jiao, A.; Kim, D.-H. Fabrication of poly(ethylene glycol): Gelatin methacrylate composite nanostructures with tunable stiffness and degradation for vascular tissue engineering. Biofabrication 2014, 6, 024112. [Google Scholar] [CrossRef]
- Cui, X.; Boland, T.; D’Lima, D.D.; Lotz, M.K. Thermal Inkjet Printing in Tissue Engineering and Regenerative Medicine. Recent Pat. Drug Deliv. Formul. 2012, 6, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Demy, T.J.; Lucas, G.R.; Strawser, B.J. Military Ethics and Emerging Technologies; Routledge: London, UK, 2014. [Google Scholar]
- Gilbert, F.; O’Connell, C.D.; Mladenovska, T.; Dodds, S. Print Me an Organ? Ethical and Regulatory Issues Emerging from 3D Bioprinting in Medicine. Sci. Eng. Ethics 2018, 24, 73–91. [Google Scholar] [CrossRef]
- Ecorys Nederland BV; University Utrecht; Consumers, Health, Agriculture and Food Executive Agency. Study on the Regulation of Advanced Therapies in Selected Jurisdictions: Final Report; Publications Office: Luxemburg, 2016; Available online: https://op.europa.eu/en/publication-detail/-/publication/78af6082-bc4a-11e6-a237-01aa75ed71a1/language-en (accessed on 4 June 2025).
- Center for Biologics Evaluation and Research. Technical Considerations for Additive Manufactured Medical Devices—Guidance for Industry and Food and Drug Administration Staff; Center for Biologics Evaluation and Research: Silver Spring, MD, USA, 2017. [Google Scholar]
- Li, J.; Chen, M.; Fan, X.; Zhou, H. Recent advances in bioprinting techniques: Approaches, applications and future prospects. J. Transl. Med. 2016, 14, 271. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-C.; Zhang, Y.S.; Akpek, A.; Shin, S.R.; Khademhosseini, A. 4D bioprinting: The next-generation technology for biofabrication enabled by stimuli-responsive materials. Biofabrication 2016, 9, 012001. [Google Scholar] [CrossRef] [PubMed]
- Faber, L.; Yau, A.; Chen, Y. Translational biomaterials of four-dimensional bioprinting for tissue regeneration. Biofabrication 2024, 16, 012001. [Google Scholar] [CrossRef]
- Douillet, C.; Nicodeme, M.; Hermant, L.; Bergeron, V.; Guillemot, F.; Fricain, J.-C.; Oliveira, H.; Garcia, M. From local to global matrix organization by fibroblasts: A 4D laser-assisted bioprinting approach. Biofabrication 2022, 14, 025006. [Google Scholar] [CrossRef]
- Kumar, M.S.; Varma, P.; Kandasubramanian, B. From lab to life: Advances in in-situ bioprinting and bioink technology. Biomed. Mater. 2025, 20, 012004. [Google Scholar] [CrossRef]
- Afzali Naniz, M.; Askari, M.; Zolfagharian, A.; Afzali Naniz, M.; Bodaghi, M. 4D printing: A cutting-edge platform for biomedical applications. Biomed. Mater. 2022, 17, 062001. [Google Scholar] [CrossRef] [PubMed]
- Pazhamannil, R.V.; Alkhedher, M. Advances in additive manufacturing for bone tissue engineering: Materials, design strategies, and applications. Biomed. Mater. 2025, 20, 012002. [Google Scholar] [CrossRef] [PubMed]
- Gharehdaghi, N.; Nokhbatolfoghahaei, H.; Khojasteh, A. 4D printing of smart scaffolds for bone regeneration: A systematic review. Biomed. Mater. 2025, 20, 012003. [Google Scholar] [CrossRef]
- Mehta, P.P. Advances in Drug Delivery Systems for Healthcare: From Concept to Clinic, 1st ed.; IPEM-IOP Series in Physics and Engineering in Medicine and Biology Series; Institute of Physics Publishing: Bristol, UK, 2024. [Google Scholar]
- Amiri, E.; Sanjarnia, P.; Sadri, B.; Jafarkhani, S.; Khakbiz, M. Recent advances and future directions of 3D to 6D printing in brain cancer treatment and neural tissue engineering. Biomed. Mater. 2023, 18, 052005. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Zhang, X.; Guo, Q.; Wu, T.; Wang, Y. 3D Bioprinted Scaffolds for Tissue Repair and Regeneration. Front. Mater. 2022, 9, 925321. [Google Scholar] [CrossRef]
- Ramezani, M.; Mohd Ripin, Z. 4D Printing in Biomedical Engineering: Advancements, Challenges, and Future Directions. J. Funct. Biomater. 2023, 14, 347. [Google Scholar] [CrossRef]

| 3D-Printing Method | Materials | Resolution | Advantages | Limitations | Ref. |
|---|---|---|---|---|---|
| SLA | Photo-curable polymer resins | 50–100 µm | High precision and smooth surfaces. | Vulnerability to brittleness, reduced impact strength, and a limited lifespan due to the gradual degradation of physical properties over time. | [20,21] |
| DLP | Photo-curable polymer resins | 10–50 µm | It offers higher printing speeds and is less influenced by oxygen inhibition than SLA. | It requires the use of low-viscosity resins, which may limit the mechanical properties of the printed object. | [22,23] |
| Inkjet printing | Polymers and hydrogels | 50–300 µm | Cost-effective, offers high resolution, enables rapid printing, and is compatible with a diverse range of materials. | The materials used must be in liquid form, which leads to lower printing density and limits the ability to produce large or highly complex structures. | [6,20] |
| Poly-jet | Photo-curable polymer resins | 20 µm | Smooth surfaces and intricate geometries, with support materials that can be easily removed. | The range of materials is highly restricted, leading to higher costs. | [24,25,26] |
| FDM | Polymers and their composites in filament form | 100–150 µm | User-friendly, environmentally sustainable, and relatively inexpensive, with high mechanical, thermal, and chemical properties. | It has a slower printing speed than SLA, provides lower dimensional accuracy, and requires high temperatures to function effectively. | [27,28] |
| 3D Dispensing | Polymers, hydrogels, ceramics, and their composites | 100 µm | Ability to process a broad spectrum of materials with varying viscosities, capable of printing bio-inks that contain living cells. | It produces rough surface finishes and generally offers lower printing resolution. | [29,30] |
| Robocasting | Dense ceramics and their composites | 200 µm | Allow the processing of highly dense ceramic pastes. | It creates relatively rudimentary objects and faces difficulties in producing complex structures. | [31,32,33,34,35,36] |
| SLS | Polymer powders, ceramic powders, and composite powders | 50–100 µm | Compatible with a wide range of materials and does not require the design of support structures. | It requires high temperatures, rough surface finishes, and necessitates post-processing. | [37,38] |
| 3D Bioprinting | Hydrogels, biomolecules, and living cells | 50–300 µm | Allows the creation of 3D structures incorporating living substances. | Expensive, involves complex operations, and requires a sterile environment for printing. | [39,40,41] |
| Material | 3DP Method | Cells | Layer Thickness | Tensile Modulus | Elongation at Break | Cell Viability | Degradation Time | Ref. |
|---|---|---|---|---|---|---|---|---|
| Gellam gum, sodium alginate, and methyl cellulose | 3D Bioprinting | HDFs, HUVECs, and MUVECs | 1.4 mm (in vitro) and 1 mm (in vivo) | - | High | - | [112] | |
| PCL and PPS doped with silver nitrate | Extrusion | HDFs | 330 µm | - | - | Reduced viability at high concentrations of AgNO3 (5% wt/wt), but acceptable at lower concentrations (1% and 2.5% wt/wt) | Entirely degraded in 11 days | [113] |
| PLLA and PEGs | Hot melt-extrusion | - | 0.4 mm | 50 ± 20 MPa | 10 ± 5% | - | - | [114] |
| Gelatin, alginate, and fibrinogen | Extrusion | HDFs and HEK, | 5 mm | - | - | High | - | [115] |
| Human plasma/fibrin | Extrusion | HDFs and HKCs | - | - | - | Viable | - | [116] |
| GelMA hydrogels | - | HaCaT | 20 µm (2 weeks), 50 µm (4 weeks), and 100 µm (6 weeks) | 9 kPa (5% GelMA) to 194 kPa (20% GelMA) | 40% (5% GelMA) to 22% (20% GelMA). | >90% for all GelMA concentrations (5% to 20%) over 1, 4, and 7 days | Ranged from a few days (5% GelMA) to over 8 weeks (20% GelMA) | [117] |
| Collagen | Solid freeform fabrication | Fibroblasts and HaCaT | ~140 µm | - | - | ~98% | - | [10] |
| Polyelectrolyte gelating-chitosan | Extrusion | Neonatal human foreskin fibroblasts | 160 µm | - | - | High (at 5% PGC hydrogel concentration) | - | [118] |
| Material | Degradation Time | 3DP Method | Cells | Layer Thickness | Tensile Modulus | Elongation at Break | Cell Viability | Reference |
|---|---|---|---|---|---|---|---|---|
| Natural | ||||||||
| Sodium alginate and hyaluronic acid | - | - | MC-3T3 osteoblast-like cells | - | - | - | High with live/dead assay showing mostly live cells after 7 days | [129] |
| Alginate and peptide | - | Extrusion | NIH3T3 HADSCs | - | - | - | >95% at 7 days for hADSCs cells >85% at 1–5% CaCl2 for NIH3T3 cells | [131] |
| Synthetic | ||||||||
| PVA | No degradation rate after 3 days | Low temperature 3DP | MG63 | - | - | - | 129% after 7 days | [125] |
| PCL | - | Selective Laser Sintering | Osteoblasts | - | - | - | High cell viability, scaffolds were nontoxic and biocompatible | [127] |
| PCL | - | FDM | - | ~0.254 mm | ~400 MPa | 80% | - | [76] |
| PHMGCL | - | - | HMSCs | - | - | - | 97 ± 1% at day 1, 99 ± 1% at day 7 | [128] |
| PLMC + PDA | 240–280 °C | 3D printing | In vivo tests | 3–4 mm | - | - | 6–12 w upon bone formation | [132] |
| Synthetic + Natural | ||||||||
| Gelatin and PVA | No degradation rate after 3 days | Low temperature 3DP | MG63 | - | - | - | 129% after 7 days | [125] |
| PLA and Gelatin Methacryloyl (GelMA) | - | Casting + FDM | ADSCs | 250 μm | - | - | No significant cytotoxic effects | [126] |
| Sodium alginate and poly(ethyleneimine) | Scaffolds maintained structure for 28 days with controlled degradation | - | - | 0.4 mm | 18.37 MPa | - | - | [130] |
| Material | 3DP Method | Cells | Layer Thickness | Tensile Modulus | Elongation at Break | Cell Viability | Degradation Time | Reference |
|---|---|---|---|---|---|---|---|---|
| Collagen | 3D Bioprinting | Human cardiomyocytes, C2C12, HUVECs, cardiac fibroblasts | 20 to 200 µm | - | - | ~96% post-printing viability | No degradation rate after 3 days | [140] |
| Alginate and GelMA | 3D Bioprinting | HUVECs, neonatal rat cardiomyocytes | 0.75 mm | 5.2 ± 0.9 kPa | - | High | GelMA degradation observed after 33 days | [141] |
| PCL | SLS | C2C12 myoblast cells | - | 0.43 ± 0.15 MPa | ~89% | High cell density of 1.2 × 106 cells/mL after 4 days | - | [142] |
| GelMA, sodium alginate and PEGTA | 3D Bioprinting | HUVECs, MSCs | - | - | - | >80% cell viability after 1, 3, and 7 days of culture for UV crosslinking times of 20 s and 30 s | >60% mass remaining after 14 days for 40 s UV exposure, <40% mass remaining for 20 s and 30 s UV exposure | [143] |
| PCL and GelMA | 3D Bioprinting | HUVECs, SMCs | 0.78 mm GelMA outer layer | - | 1384 ± 76.22% | 90% viability | - | [144] |
| PVA | SLA + FDM | HMSCs | 250 μm | - | - | High | In vitro biodegradation showed no significant change up to 20 days | [145] |
| Material | 3DP Method | Cells | Layer Thickness | Tensile Modulus | Elongation at Break | Cell Viability | Degradation Time | Reference |
|---|---|---|---|---|---|---|---|---|
| Collagen | Jet Printing | PC-12 HUVECs | Triple layered with a wall thickness of 1.2 mm | 13.365 ± 0.086 MPa | 623.511 ± 23.035% | High | - | [146] |
| Alginate and GelMA | Digital Light Processing | PC-12 NCSCs | - | - | - | >95% after 1 day | - | [147] |
| PCL | - | NIH3T3 | - | - | - | Good cell viability on the surface | Completely degraded after 5 weeks | [148] |
| GelMA, sodium alginate and PEGTA | Digital Light Processing | MC3T3-E1 NIH3T3 | - | 8.9 ± 0.1 kPa | - | High | - | [149] |
| PCL and GelMA | Extrusion | PC-12 RSC96 | - | - | - | 96.9 ± 1.52% (3 days) | - | [150] |
| PVA | Inkjet | Murine neural stem cells | ~50 µm | - | - | 98.05 ± 0.37% | - | [152] |
| GelMA with graphene nanoplatelets | Stereolitography | PC-12 | - | - | - | High cell viability observed in 10% GelMA hydrogels | - | [151] |
| Aspect | 3D Printing | 4D Printing |
|---|---|---|
| Definition | Fabrication of fixed structures through additive layer-by-layer deposition. | 3D-printed structures that change shape or function over time. |
| Material Type | Biocompatible polymers, ceramics, or hydrogels. | Stimuli-responsive smart materials (e.g., shape-memory polymers, hydrogels). |
| Shape Dynamics | Static does not change after printing. | Dynamic, responds to stimuli such as heat, moisture, or pH. |
| Stimuli Required | None. | External stimuli needed (e.g., temperature, humidity, magnetic field). |
| Applications | Prosthetics, anatomical models, static tissue scaffolds. | Smart implants, adaptive scaffolds, soft robotics, controlled drug delivery. |
| Advantages | High precision, reproducibility, clinically accessible. | Adaptive behavior, enhanced functionality, potential for real-time response. |
| Limitations | Inflexibility, limited to static functions. | Complex design, material constraints, hight. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arias-Peregrino, V.M.; Tenorio-Barajas, A.Y.; Mendoza-Barrera, C.O.; Román-Doval, J.; Lavariega-Sumano, E.F.; Torres-Arellanes, S.P.; Román-Doval, R. 3D Printing for Tissue Engineering: Printing Techniques, Biomaterials, Challenges, and the Emerging Role of 4D Bioprinting. Bioengineering 2025, 12, 936. https://doi.org/10.3390/bioengineering12090936
Arias-Peregrino VM, Tenorio-Barajas AY, Mendoza-Barrera CO, Román-Doval J, Lavariega-Sumano EF, Torres-Arellanes SP, Román-Doval R. 3D Printing for Tissue Engineering: Printing Techniques, Biomaterials, Challenges, and the Emerging Role of 4D Bioprinting. Bioengineering. 2025; 12(9):936. https://doi.org/10.3390/bioengineering12090936
Chicago/Turabian StyleArias-Peregrino, Victor M., Aldo Y. Tenorio-Barajas, Claudia O. Mendoza-Barrera, Jesús Román-Doval, Esteban F. Lavariega-Sumano, Sandra P. Torres-Arellanes, and Ramón Román-Doval. 2025. "3D Printing for Tissue Engineering: Printing Techniques, Biomaterials, Challenges, and the Emerging Role of 4D Bioprinting" Bioengineering 12, no. 9: 936. https://doi.org/10.3390/bioengineering12090936
APA StyleArias-Peregrino, V. M., Tenorio-Barajas, A. Y., Mendoza-Barrera, C. O., Román-Doval, J., Lavariega-Sumano, E. F., Torres-Arellanes, S. P., & Román-Doval, R. (2025). 3D Printing for Tissue Engineering: Printing Techniques, Biomaterials, Challenges, and the Emerging Role of 4D Bioprinting. Bioengineering, 12(9), 936. https://doi.org/10.3390/bioengineering12090936







