Feasibility and Preliminary Efficacy of Wearable Focal Vibration Therapy on Gait and Mobility in People with Multiple Sclerosis: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
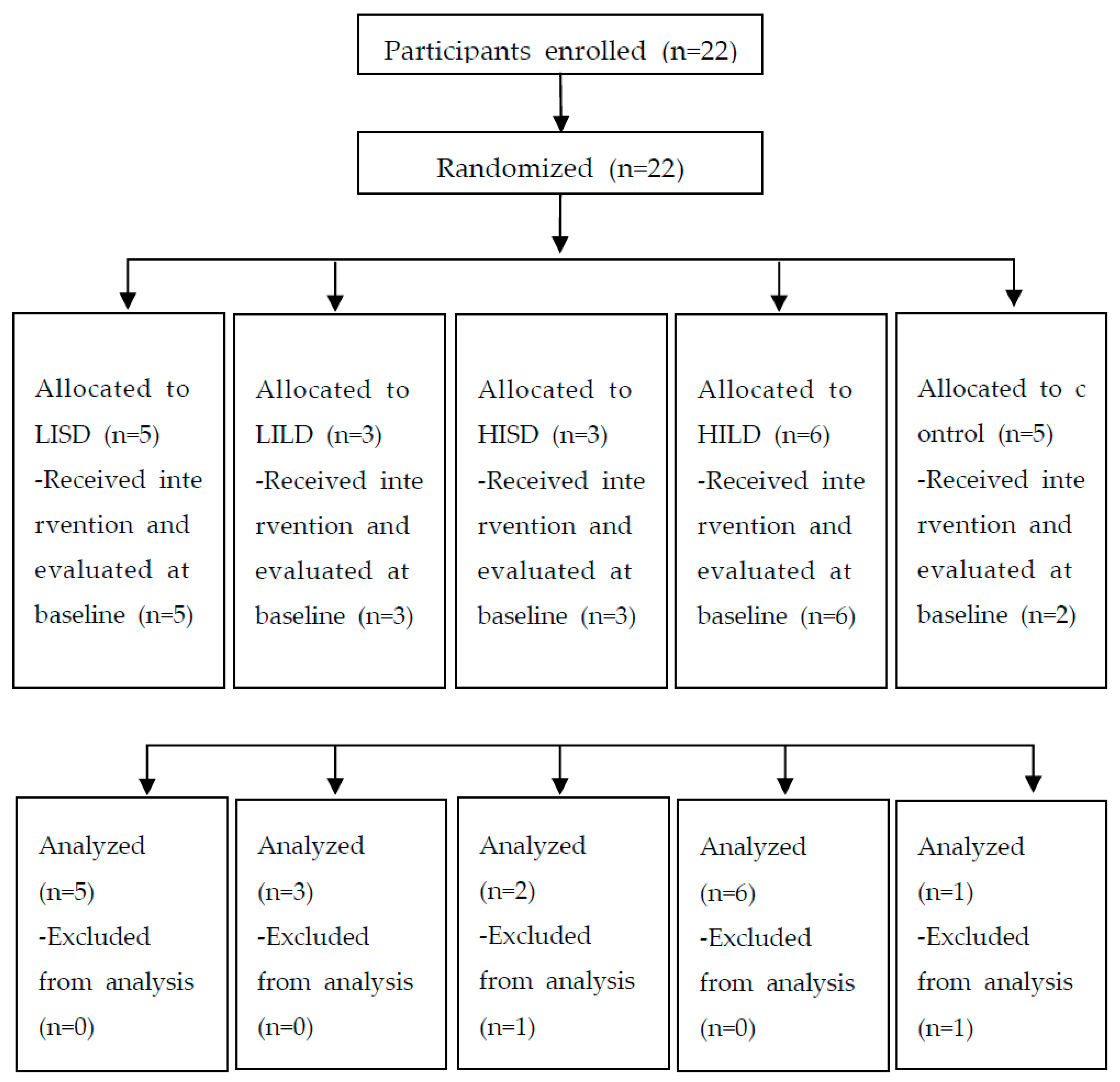
2.2. Participants
2.3. Intervention
2.4. Outcome Measurements
2.5. Statistical Analysis
3. Results
3.1. Feasibility of the Wearable FVT Intervention
3.2. Gait Parameters
3.3. Mobility
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Filippi, M.; Bar-Or, A.; Piehl, F.; Preziosa, P.; Solari, A.; Vukusic, S.; Rocca, M.A. Multiple sclerosis. Nat. Rev. Dis. Primers 2018, 4, 43. [Google Scholar] [CrossRef]
- Lassmann, H.; Brück, W.; Lucchinetti, C.F. The immunopathology of multiple sclerosis: An overview. Brain Pathol. 2007, 17, 210–218. [Google Scholar] [CrossRef]
- Lubetzki, C.; Stankoff, B. Demyelination in multiple sclerosis. Handb. Clin. Neurol. 2014, 122, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Chee, J.N.; Sosnoff, J.J.; Sandroff, B.M.; Motl, R.W. Influence of multiple sclerosis on spatiotemporal gait parameters: A systematic review and meta-regression. Arch. Phys. Med. Rehabil. 2021, 102, 1801–1815. [Google Scholar] [CrossRef] [PubMed]
- Braley, T.J.; Chervin, R.D. Fatigue in multiple sclerosis: Mechanisms, evaluation, and treatment. Sleep 2010, 33, 1061–1067. [Google Scholar] [CrossRef]
- Comber, L.; Galvin, R.; Coote, S. Gait deficits in people with multiple sclerosis: A systematic review and meta-analysis. Gait Posture 2017, 51, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Cameron, M.H.; Nilsagard, Y. Balance, gait, and falls in multiple sclerosis. Handb. Clin. Neurol. 2018, 159, 237–250. [Google Scholar]
- Matsuda, P.N.; Shumway-Cook, A.; Bamer, A.M.; Johnson, S.L.; Amtmann, D.; Kraft, G.H. Falls in multiple sclerosis. PM R Phys. Med. Rehabil. 2011, 3, 624–632. [Google Scholar] [CrossRef]
- Finlayson, M.; Peterson, E.; Matsuda, P.N. Participation as an outcome in multiple sclerosis falls-prevention research: Consensus recommendation from the International MS Falls Prevention Research Network. Int. J. MS Care 2014, 16, 171–177. [Google Scholar] [CrossRef]
- Van der Feen, F.E.; De Haan, G.A.; van der Lijn, I.; Heersema, D.J.; Meilof, J.F.; Heutink, J. Independent outdoor mobility of persons with multiple sclerosis—A systematic review. Mult. Scler. Relat. Disord. 2020, 37, 101463. [Google Scholar] [CrossRef]
- Zhang, E.; Tian, X.; Li, R.; Chen, C.; Li, M.; Ma, L.; Wei, R.; Zhou, Y.; Cui, Y. Dalfampridine in the treatment of multiple sclerosis: A meta-analysis of randomised controlled trials. Orphanet J. Rare Dis. 2021, 16, 87. [Google Scholar] [CrossRef] [PubMed]
- Dunn, J.; Blight, A. Dalfampridine: A brief review of its mechanism and efficacy in multiple sclerosis. Drugs 2011, 71, 471–480. [Google Scholar]
- Du, L.; Xi, H.; Zhang, S.; Zhou, Y.; Tao, X.; Lv, Y.; Hou, X.; Yu, L. Effects of exercise in people with multiple sclerosis: A systematic review and meta-analysis. Front. Public Health 2024, 12, 1387658. [Google Scholar] [CrossRef]
- Zou, L.; Wang, H.; Xiao, Z.; Fang, Q.; Zhang, M.; Li, T.; Du, G.; Liu, Y. Tai chi for health benefits in patients with multiple sclerosis: A systematic review. PLoS ONE 2017, 12, e0170212. [Google Scholar] [CrossRef]
- Diaz-Quiroz, M.; Chicue-Cuervo, P.C.; Garcia-Moreno, L.; Gaviria-Carrillo, M.; Talero-Gutierrez, C.; Palacios-Espinosa, X. Fatigue in multiple sclerosis: A scoping review of pharmacological and nonpharmacological interventions. Mult. Scler. J. Exp. Transl. Clin. 2025, 11, 20552173241312527. [Google Scholar] [CrossRef]
- Fattorini, L.; Pettorossi, V.E.; Marchetti, E.; Rodio, A.; Filippi, G.M. A review about muscle focal vibration contribution on spasticity recovery. Front. Neurol. 2025, 16, 1579118. [Google Scholar] [CrossRef]
- Moggio, L.; de Sire, A.; Marotta, N.; Demeco, A.; Ammendolia, A. Vibration therapy role in neurological diseases rehabilitation: An umbrella review of systematic reviews. Disabil. Rehabil. 2022, 44, 5741–5749. [Google Scholar] [CrossRef] [PubMed]
- Bessaguet, H.; Fernandez, B.; Malejac, V.; Nerriere, E.; Velarde, M.; Coiffet, A.; Rimaud, D.; Lapole, T. Effect of tibialis anterior focal muscle vibration for gait rehabilitation in hemiplegic individuals during the subacute phase after stroke: The NEUROVIB-AVC study protocol—A multicentric randomised controlled trial. BMJ Open 2025, 15, e102838. [Google Scholar] [CrossRef] [PubMed]
- Alashram, A.R.; Padua, E.; Romagnoli, C.; Raju, M.; Annino, G. Clinical effectiveness of focal muscle vibration on gait and postural stability in individuals with neurological disorders: A systematic review. Physiother. Res. Int. 2022, 27, e1945. [Google Scholar] [CrossRef]
- Ferrara, P.E.; Gatto, D.M.; Codazza, S.; Zordan, P.; Stefinlongo, G.; Coraci, D.; Lo Monaco, M.R.; Ricciardi, D.; Ronconi, G. Effects of focal muscle vibration on gait and balance in Parkinson patients: Preliminary results. Appl. Sci. 2022, 12, 10486. [Google Scholar] [CrossRef]
- Camerota, F.; Celletti, C.; Di Sipio, E.; De Fino, C.; Simbolotti, C.; Germanotta, M.; Mirabella, M.; Padua, L.; Nociti, V. Focal muscle vibration, an effective rehabilitative approach in severe gait impairment due to multiple sclerosis. J. Neurol. Sci. 2017, 372, 33–39. [Google Scholar] [CrossRef]
- Spina, E.; Carotenuto, A.; Aceto, M.G.; Cerillo, I.; Silvestre, F.; Arace, F.; Paone, P.; Orefice, G.; Iodice, R. The effects of mechanical focal vibration on walking impairment in multiple sclerosis patients: A randomized, double-blinded vs placebo study. Restor. Neurol. Neurosci. 2016, 34, 869–876. [Google Scholar] [CrossRef]
- Paoloni, M.; Mangone, M.; Scettri, P.; Procaccianti, R.; Cometa, A.; Santilli, V. Segmental muscle vibration improves walking in chronic stroke patients with foot drop: A randomized controlled trial. Neurorehabil. Neural Repair 2010, 24, 254–262. [Google Scholar] [CrossRef]
- Ayvat, F.; Özçakar, L.; Ayvat, E.; Aksu Yıldırım, S.; Kılınç, M. Effects of low vs. high frequency local vibration on mild-moderate muscle spasticity: Ultrasonographical and functional evaluation in patients with multiple sclerosis. Mult. Scler. Relat. Disord. 2021, 51, 102930. [Google Scholar] [CrossRef]
- Thompson, A.J.; Banwell, B.L.; Barkhof, F.; Carroll, W.M.; Coetzee, T.; Comi, G.; Correale, J.; Fazekas, F.; Filippi, M.; Freedman, M.S.; et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018, 17, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Li, N.; Gao, J.; Wang, L.N.; Zhao, Y.M.; Yu, B.C.; Du, W.; Zhang, W.J.; Cui, L.Q.; Wang, Q.S.; et al. Optimal cutoff scores for dementia and mild cognitive impairment of the Montreal Cognitive Assessment among elderly and oldest-old Chinese population. J. Alzheimers Dis. 2015, 43, 1403–1412. [Google Scholar] [CrossRef] [PubMed]
- Kurtzke, J.F. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 1983, 33, 1444–1452. [Google Scholar] [CrossRef]
- Kuspinar, A.; Mayo, N.E. A review of the psychometric properties of generic utility measures in multiple sclerosis. Pharmacoeconomics 2014, 32, 759–773. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Moock, S.; Feng, Y.S.; Maeurer, M.; Dippel, F.W.; Kohlmann, T. Systematic literature review and validity evaluation of the Expanded Disability Status Scale (EDSS) and the Multiple Sclerosis Functional Composite (MSFC) in patients with multiple sclerosis. BMC Neurol. 2014, 14, 58. [Google Scholar] [CrossRef]
- Demers, L.; Weiss-Lambrou, R.; Ska, B. Development of the Quebec User Evaluation of Satisfaction with assistive Technology (QUEST). Assist. Technol. 1996, 8, 3–13. [Google Scholar] [CrossRef]
- Demers, L.; Weiss-Lambrou, R.; Ska, B. Item analysis of the Quebec User Evaluation of Satisfaction with Assistive Technology (QUEST). Assist. Technol. 2000, 12, 96–105. [Google Scholar] [CrossRef]
- Demers, L.; Monette, M.; Lapierre, Y.; Arnold, D.L.; Wolfson, C. Reliability, validity, and applicability of the Quebec User Evaluation of Satisfaction with assistive Technology (QUEST 2.0) for adults with multiple sclerosis. Disabil. Rehabil. 2002, 24, 21–30. [Google Scholar] [CrossRef]
- Goldman, M.D.; Motl, R.W.; Scagnelli, J.; Pula, J.H.; Sosnoff, J.J.; Cadavid, D. Clinically meaningful performance benchmarks in MS: Timed 25-foot walk and the real world. Neurology 2013, 81, 1856–1863. [Google Scholar] [CrossRef] [PubMed]
- Oosterveer, D.M.; Wouda, N.C.; Volker, G.; van den Berg, C.; Terluin, B.; Hoitsma, E. Reliability parameters of the Timed 25-Foot-Walk (T25FW) in patients with Multiple Sclerosis: Lower walking speed is associated with greater Smallest Detectable Change. Mult. Scler. Relat. Disord. 2024, 88, 105731. [Google Scholar] [CrossRef] [PubMed]
- C-Motion. Temporal Distance Calculations for Gait. Visual3D Wiki Documentation. Available online: https://c-motion.com/v3dwiki/index.php?title=Temporal_Distance_Calculations_for_Gait#Cadence (accessed on 6 October 2022).
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988; pp. 19–27. [Google Scholar]
- Coleman, C.I.; Sobieraj, D.M.; Marinucci, L.N. Minimally important clinical difference of the Timed 25-Foot Walk Test: Results from a randomized controlled trial in patients with multiple sclerosis. Curr. Med. Res. Opin. 2012, 28, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Horder, J.; Mrotek, L.A.; Casadio, M.; Bassindale, K.D.; McGuire, J.; Scheidt, R.A. Utility and usability of a wearable system and progressive-challenge cued exercise program for encouraging use of the more involved arm at-home after stroke—A feasibility study with case reports. J. Neuroeng. Rehabil. 2024, 21, 66. [Google Scholar] [CrossRef]
- Martinez-Hernandez, U.; Metcalfe, B.; Assaf, T.; Jabban, L.; Male, J.; Zhang, D. Wearable assistive robotics: A perspective on current challenges and future trends. Sensors 2021, 21, 6751. [Google Scholar] [CrossRef]
- Rodgers, M.M.; Alon, G.; Pai, V.M.; Conroy, R.S. Wearable technologies for active living and rehabilitation: Current research challenges and future opportunities. J. Rehabil. Assist. Technol. Eng. 2019, 6, 2055668319839607. [Google Scholar] [CrossRef]
- Hebert, J.R.; Corboy, J.R. The association between multiple sclerosis-related fatigue and balance as a function of central sensory integration. Gait Posture 2013, 38, 37–42. [Google Scholar] [CrossRef]
- Layne, C.S.; Malaya, C.A.; Levine, J.T. The effects of muscle vibration on gait control: A review. Somatosens. Mot. Res. 2019, 36, 212–222. [Google Scholar] [CrossRef]
- Kerschan-Schindl, K.; Grampp, S.; Henk, C.; Resch, H.; Preisinger, E.; Fialka-Moser, V.; Imhof, H. Whole-body vibration exercise leads to alterations in muscle blood volume. Clin. Physiol. 2001, 21, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Lizrova Preiningerova, J.; Novotna, K.; Rusz, J.; Sucha, L.; Ruzicka, E.; Havrdova, E. Spatial and temporal characteristics of gait as outcome measures in multiple sclerosis. J. Neuroeng. Rehabil. 2015, 12, 14. [Google Scholar] [CrossRef]
- Wonneberger, M.; Schmidt, H. Gait analysis in neurological diseases. J. Neurol. 2015, 262, 869–876. [Google Scholar]
- Sokhangu, M.K.; Rahnama, N.; Etemadifar, M.; Rafeii, M.; Saberi, A. Effect of Neuromuscular Exercises on Strength, Proprioceptive Receptors, and Balance in Females with Multiple Sclerosis. Int. J. Prev. Med. 2021, 12, 5. [Google Scholar] [CrossRef] [PubMed]
- Giorgi, F.; Donati, D.; Platano, D.; Tedeschi, R. Focal Vibration Therapy for Motor Deficits and Spasticity Management in Post-Stroke Rehabilitation. Brain Sci. 2024, 14, 1060. [Google Scholar] [CrossRef]
- Lienhard, K.; Cabasson, A.; Meste, O.; Colson, S.S. Determination of the optimal parameters maximizing muscle activity of the lower limbs during vertical synchronous whole-body vibration. Eur. J. Appl. Physiol. 2014, 114, 1493–1501. [Google Scholar] [CrossRef]
- Goyal, D.; Hunt, X.; Kuper, H.; Shakespeare, T.; Banks, L.M. Impact of the COVID-19 pandemic on people with disabilities and implications for health services research. J. Health Serv. Res. Policy 2023, 28, 77–79. [Google Scholar] [CrossRef] [PubMed]


| Experimental Group (n = 17) | Control Group (n = 2) | |
|---|---|---|
| Age (mean, (SD)) | 49.65 (9.59) | 49.50 (9.19) |
| Gender (n, (%)) Male Female | 3 (17.65%) 14 (82.35%) | 1 (50.00%) 1 (50.00%) |
| BMI (mean, (SD)) | 33.31 (7.86) | 22.27 (0.97) |
| Ethnicity (n, (%)) African American Caucasian | 6 (35.29%) 11 (64.71%) | 1 (50.00%) 1 (50.00%) |
| Education (n, (%)) GED or High School Diploma Some college Bachelor’s Degree Master’s Degree Doctorate Associate degree Technical Certification | 4 (23.53%) 2 (11.76%) 5 (29.41%) 3 (17.65%) 1 (5.88%) 1 (5.88%) 1 (5.88%) | 1 (50.00%) 0 (0.00%) 0 (0.00%) 0 (0.00%) 0 (0.00%) 1 (50.00%) 0 (0.00%) |
| Years of MS (mean, (SD)) | 9.76 (7.53) | 8 (9.90) |
| Baseline EDSS score (mean, (SD)) | 3.22 (0.93) ++ | 4.00 (0.00) |
| Ankle-foot orthoses usage (n, (%)) Yes No | 1 (5.88%) 16 (94.12%) | 0 (0.00%) 2 (100.00%) |
| Walking aids usage (n, (%)) Yes No | 6 (35.29%) 11 (64.71%) | 1 (50.00%) 1 (50.00%) |
| Using MS medication Yes No | 16 (94.12%) 1 (5.88%) | 2 (100.00%) 0 (0.00%) |
| Perceived importance of medication use (mean, (SD)) | 9.82 (0.53) | 9.00 (1.41) |
| Physical limitations affecting self-care (n, (%)) Problems with hand Problems with feet Vision loss Hearing loss | 3 (17.65%) 8 (47.06%) 1 (5.88%) 1 (5.88%) | 0 (0.00%) 1 (50.00%) 1 (50.00%) 0 (0.00%) |
| Low-Intensity Vibration Group (n = 8) | High-Intensity Vibration Group (n = 9) | Short-Duration Vibration Group (n = 8) | Long-Duration Vibration Group (n = 9) | |
|---|---|---|---|---|
| Age (mean, (SD)) | 50.00 (9.74) | 49.33 (10.04) | 50.88 (9.63) | 48.56 (10.00) |
| Gender (n, (%)) Male Female | 2 (25.00%) 6 (75.00%) | 1 (11.11%) 8 (88.89%) | 2 (25.00%) 6 (75.00%) | 1 (11.11%) 8 (88.89%) |
| BMI (mean, (SD)) | 36.24 (7.28) | 30.71 (7.82) | 34.75 (7.94) | 32.03 (8.03) |
| Ethnicity (n, (%)) African American Caucasian | 2 (25.00%) 6 (75.00%) | 4 (44.44%) 5 (55.56%) | 2 (25.00%) 6 (75.00%) | 4 (44.44%) 5 (55.56%) |
| Education (n, (%)) GED or High School Diploma Some college Bachelor’s Degree Master’s Degree Doctorate Associate degree Technical Certification | 1 (12.50%) 2 (25.00%) 3 (37.50%) 1 (12.50%) 1 (12.50%) 0 (0.00%) 0 (0.00%) | 3 (33.33%) 0 (0.00%) 2 (22.22%) 2 (22.22%) 0 (0.00%) 1 (11.11%) 1 (11.11%) | 1 (12.50%) 1 (12.50%) 3 (32.50%) 2 (25.00%) 1 (12.50%) 0 (0.00%) 0 (0.00%) | 3 (33.33%) 1 (11.11%) 2 (22.22%) 1 (11.11%) 0 (0.00%) 1 (11.11%) 1 (11.11%) |
| Years of MS (mean, (SD)) | 7.75 (8.48) | 11.56 (6.54) | 8.13 (7.94) | 11.22 (7.29) |
| Baseline EDSS score (mean, (SD)) | 2.79 (0.57) + | 3.56 (1.04) | 3.36 (0.80) + | 3.11 (1.05) |
| Ankle-foot orthoses usage (n, (%)) Yes No | 0 (0.00%) 8 (100.00%) | 1 (11.11%) 8 (88.89%) | 0 (0.00%) 8 (100.00%) | 1 (11.11%) 8 (88.89%) |
| Walking aids usage (n, (%)) Yes No | 1 (12.50%) 7 (0.00%) | 5 (55.56%) 4 (44.44%) | 3 (32.50%) 5 (67.50%) | 3 (33.33%) 6 (66.67%) |
| Using MS medication Yes No | 8 (100.00%) 0 (0.00%) | 8 (88.89%) 1 (11.11%) | 8 (100.00%) 0 (0.00%) | 8 (88.89%) 1 (11.11%) |
| Perceived importance of medication use (mean, (SD)) | 9.75 (0.71) | 9.89 (0.33) | 9.63 (0.74) | 10.00 (0.00) |
| Physical limitations affecting the ability to perform self-care (n, (%)) Problems with hand Problems with feet Vision loss Hearing loss | 2 (25.00%) 5 (62.50%) 1 (12.50%) 1 (12.50%) | 1 (11.11%) 3 (33.33%) 0 (0.00%) 0 (0.00%) | 1 (12.50%) 4 (50.00%) 1 (12.50%) 0 (0.00%) | 2 (22.22%) 4 (44.44%) 0 (0.00%) 1 (11.11%) |
| Questions | Mean (SD) | Min Score |
|---|---|---|
| Satisfaction with the dimensions of the assistive device | 4.76 (0.44) | 4 |
| Satisfaction with the weight | 4.88 (0.33) | 4 |
| Satisfaction with the ease of adjusting the device | 4.29 (1.05) | 2 |
| Satisfaction with how safe and secure of device is | 4.71 (0.59) | 3 |
| Satisfaction with the durability | 4.47 (1.07) | 2 |
| Satisfaction with ease of use | 4.82 (0.39) | 4 |
| Satisfaction with the comfort of the device | 4.71 (0.59) | 3 |
| Satisfaction with how effective the device is | 4.53 (0.80) | 3 |
| Total satisfaction | 37.18 (3.89) | 27 |
| n | |
|---|---|
| Willing to use FVT | 15 |
| Willing to purchase the device | 9 |
| Will not purchase the device | 2 |
| Willing to purchase the device after improvements (stronger intensity, clear evidence, appropriate cost) | 3 |
| Missed using the device | 3 |
| No pain during device use | 13 |
| Potential adverse events during device use (spasm or pain) | 2 |
| Device preference All the aspects Ease of use Flexibility Easy to use Simplicity Fun to use Lightweight | 6 5 1 2 1 1 1 |
| Parameter: m = Meters; s = Second | All Participants Mean (Standard Deviation) | p Values | Estimated Effect Size | |
|---|---|---|---|---|
| Gait speed (m/s) | pre | 0.92 (0.30) | 0.014 | 0.34 |
| post | 0.98 (0.27) | |||
| Stride length (m) | pre | 1.05 (0.23) | 0.004 | 0.44 |
| post | 1.11 (0.19) | |||
| Stride width (m) | pre | 0.19 ± 0.04 | 0.613 | 0.02 |
| post | 0.19 ± 0.03 | |||
| Stride time (s) | pre | 1.20 (0.19) | 0.76 | 0.01 |
| post | 1.19 (0.23) | |||
| More involved cadence (steps/min) | pre | 101.01 (16.18) | 0.53 | 0.03 |
| post | 102.36 (18.26) | |||
| Less involved cadence (steps/min) | pre | 105.34 (15.53) | 0.74 | 0.01 |
| post | 105.83 (13.97) | |||
| More involved stance (s) | pre | 0.79 (0.15) | 0.62 | 0.02 |
| post | 0.78 (0.16) | |||
| Less involved stance (s) | pre | 0.81 (0.15) | 0.63 | 0.02 |
| post | 0.79 (0.18) | |||
| More involved swing time (s) a | pre | 0.40 (0.05) | 0.57 | 0.02 |
| post | 0.41 (0.07) | |||
| Less involved swing time (s) a | pre | 0.40 (0.06) | 0.97 | <0.01 |
| post | 0.40 (0.05) | |||
| Double limb support (s) a | pre | 0.39 (0.11) | 0.77 | 0.01 |
| post | 0.39 (0.12) | |||
| Parameter: LI-Less Involved; MI-More Involved | All Participants Mean (Standard Deviation) | p Values | Estimated Effect Size | |
|---|---|---|---|---|
| LI peak dorsiflexion at heel-off (°) a | pre | 21.09 (2.41) | 0.89 | 0.001 |
| post | 21.24 (4.33) | |||
| LI peak plantarflexion before heel strike (°) a | pre | −5.02 (6.26) | 0.18 | 0.13 |
| post | −7.03 (6.36) | |||
| LI peak dorsiflexion before heel-off (°) a | pre | 11.55 (3.51) | 0.97 | <0.01 |
| post | 11.52 (2.88) | |||
| LI peak plantarflexion during pre-swing (°) a | pre | −6.48 (7.30) | 0.28 | 0.08 |
| post | −8.42 (7.38) | |||
| MI peak dorsiflexion at heel-off (°) a | pre | 19.41 (2.96) | 0.59 | 0.02 |
| post | 19.79 (2.94) | |||
| MI peak plantarflexion before heel strike (°) a | pre | −7.02 (8.38) | 0.056 | 0.24 |
| post | −8.99 (8.07) | |||
| MI peak dorsiflexion before heel-off (°) a | pre | 10.60 (4.19) | 0.62 | 0.02 |
| post | 10.24 (4.97) | |||
| MI peak plantarflexion during pre-swing (°) a | pre | −9.61 (9.87) | 0.043 | 0.26 |
| post | −11.88 (9.69) | |||
| LI knee flexion at heel stike (°) | pre | 8.75 (9.93) | 0.41 | 0.05 |
| post | 10.10 (8.65) | |||
| LI maximum knee flexion at stance (°) | pre | 43.68 (8.91) | 0.86 | 0.002 |
| post | 43.26 (5.53) | |||
| LI minum knee flexion at stance (°) | pre | 4.58 (7.82) | 0.51 | 0.03 |
| post | 5.61 (7.23) | |||
| LI maximum knee flexion at swing (°) | pre | 54.36 (11.26) | 0.53 | 0.03 |
| post | 55.80 (6.44) | |||
| LI minimum knee flexion at swing (°) | pre | 6.44 (10.75) | 0.50 | 0.03 |
| post | 7.59 (9.83) | |||
| MI knee flexion at heel stike (°) | pre | 8.12 (9.59) | 0.44 | 0.04 |
| post | 9.52 (8.23) | |||
| MI maximum knee flexion at stance (°) | pre | 40.62 (7.73) | 0.31 | 0.07 |
| post | 42.82 (4.68) | |||
| MI minimum knee flexion at stance (°) | pre | 2.39 (7.02) | 0.41 | 0.05 |
| post | 3.67 (4.83) | |||
| MI maximum knee flexion at swing (°) | pre | 52.46 (10.78) | 0.31 | 0.07 |
| post | 54.70 (8.03) | |||
| MI minimum knee flexion at swing (°) | pre | 5.89 (10.53) | 0.80 | 0.004 |
| post | 6.32 (8.87) | |||
| LI peak hip flexion at stance (°) | pre | 23.63 (10.55) | 0.18 | 0.12 |
| post | 28.12 (8.51) | |||
| LI peak hip extension at stance (°) | pre | −13.73 (11.94) | 0.36 | 0.06 |
| post | −10.90 (10.11) | |||
| LI peak hip flexion at swing (°) | pre | 24.75 (11.03) | 0.16 | 0.13 |
| post | 29.64 (8.46) | |||
| LI peak hip extension at swing (°) | pre | −2.44 (12.86) | 0.49 | 0.05 |
| post | −0.08 (10.02) | |||
| MI peak hip flexion at stance (°) | pre | 21.69 (12.09) | 0.08 | 0.19 |
| post | 27.41 (9.18) | |||
| MI peak hip extension at stance (°) | pre | −14.86 (12.78) | 0.29 | 0.07 |
| post | −11.66 (9.84) | |||
| MI peak hip flexion at swing (°) | pre | 23.20 (12.99) | 0.11 | 0.16 |
| post | 28.60 (8.91) | |||
| MI peak hip extension at swing (°) | pre | −4.03 (13.97) | 0.32 | 0.07 |
| post | −0.34 (10.64) | |||
| LI minimum knee flexion at swing (°) | pre | 6.44 (10.75) | 0.50 | 0.03 |
| post | 7.59 (9.83) | |||
| Parameter: LI-Less Involved; MI-More Involved | All Participants Mean (Standard Deviation) | p Values | Estimated Effect Size | |
|---|---|---|---|---|
| LI peak plantar flexor moment (N · m/kg) | pre | 1.10 (0.20) | 0.15 | 0.16 |
| post | 1.15 (0.16) | |||
| LI peak ankle power in late stance (W/kg) | pre | 1.89 (0.77) | 0.058 | 0.27 |
| post | 2.13 (0.82) | |||
| LI peak ankle power in early stance (W/kg) | pre | −0.84 (0.41) | 0.74 | 0.01 |
| post | −0.86 (0.31) | |||
| MI peak plantar flexor moment (N · m/kg) | pre | 1.10 (0.26) | 0.37 | 0.07 |
| post | 1.14 (0.22) | |||
| MI peak ankle power in late stance (W/kg) | pre | 1.81 (0.95) | 0.086 | 0.23 |
| post | 2.03 (1.11) | |||
| MI peak ankle power in early stance (W/kg) | pre | −0.71 (0.27) | 0.077 | 0.24 |
| post | −0.79 (0.32) | |||
| LI peak knee extensor moment in early stance (N · m/kg) | pre | 0.85 (0.29) | 0.88 | 0.002 |
| post | 0.85 (0.21) | |||
| LI peak knee flexor moment in mid-stance (N · m/kg) | pre | −0.17 (0.08) | 0.57 | 0.03 |
| post | −0.18 (0.06) | |||
| LI peak knee flexor moment in early swing (N · m/kg) | pre | 0.11 (0.06) | 0.86 | 0.003 |
| post | 0.11 (0.04) | |||
| LI peak knee extensor moment in late swing (N · m/kg) | pre | −0.28 (0.09) | 0.19 | 0.14 |
| post | −0.29 (0.08) | |||
| LI peak knee power generation in stance (W/kg) | pre | 0.55 (0.40) | 0.13 | 0.18 |
| post | 0.71 (0.47) | |||
| LI peak knee power absorption in stance (W/kg) | pre | −1.38 (0.65) | 0.69 | 0.01 |
| post | −1.43 (0.49) | |||
| LI peak knee power absorption in swing (W/kg) | pre | −0.97 (0.55) | 0.55 | 0.03 |
| post | −1.01 (0.52) | |||
| MI peak knee extensor moment in early stance (N · m/kg) | pre | 0.74 (0.28) | 0.19 | 0.14 |
| post | 0.81 (0.27) | |||
| MI peak knee flexor moment in mid-stance (N · m/kg) | pre | −0.18 (0.07) | 0.67 | 0.02 |
| post | −0.19 (0.08) | |||
| MI peak knee flexor moment in early swing (N · m/kg) | pre | 0.08 (0.05) | 0.076 | 0.24 |
| post | 0.09 (0.06) | |||
| MI peak knee extensor moment in late swing (N · m/kg) | pre | −0.26 (0.09) | 0.27 | 0.10 |
| post | −0.28 (0.10) | |||
| MI peak knee power generation in stance (W/kg) | pre | 0.50 (0.29) | 0.28 | 0.10 |
| post | 0.57 (0.37) | |||
| MI peak knee power absorption in stance (W/kg) | pre | −1.17 (0.58) | 0.18 | 0.15 |
| post | −1.39 (0.61) | |||
| MI peak knee power absorption in swing (W/kg) | pre | −0.87 (0.45) | 0.065 | 0.26 |
| post | −1.03 (0.48) | |||
| LI peak hip flexion moment in late stance (N · m/kg) | pre | 1.19 (0.31) | 0.31 | 0.09 |
| post | 1.10 (0.33) | |||
| LI peak hip extension moment in stance (N · m/kg) | pre | −0.33 (0.16) | 0.26 | 0.10 |
| post | −0.37 (0.11) | |||
| LI peak hip flexor moment in early swing (N · m/kg) | pre | 0.21 (0.08) | 0.25 | 0.11 |
| post | 0.30 (0.25) | |||
| LI peak hip power generation in terminal stance (W/kg) | pre | 1.05 (0.36) | 0.57 | 0.03 |
| post | 1.09 (0.46) | |||
| LI peak hip power absorption in stance (W/kg) | pre | −1.68 (0.78) | 0.21 | 0.13 |
| post | −1.42 (0.56) | |||
| LI peak hip power generation in swing (W/kg) | pre | 0.61 (0.32) | 0.61 | 0.02 |
| post | 0.66 (0.30) | |||
| MI peak hip flexion moment in late stance (N · m/kg) | pre | 1.13 (0.38) | 0.52 | 0.04 |
| post | 1.07 (0.38) | |||
| MI peak hip extension moment in stance (N · m/kg) | pre | −0.40 (0.17) | 0.27 | 0.10 |
| post | −0.42 (0.16) | |||
| MI peak hip flexor moment in early swing (N · m/kg) | pre | 0.20 (0.09) | 0.86 | 0.003 |
| post | 0.20 (0.09) | |||
| MI peak hip power generation in terminal stance (W/kg) | pre | 1.00 (0.48) | 0.67 | 0.02 |
| post | 1.03 (0.38) | |||
| MI peak hip power absorption in stance (W/kg) | pre | −1.54 (0.76) | 0.33 | 0.08 |
| post | −1.36 (0.56) | |||
| MI peak hip power generation in swing (W/kg) | pre | 0.53 (0.30) | 0.91 | 0.001 |
| post | 0.54 (0.24) | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Shin, Y.C.; Tester, N.J.; Rempe, T. Feasibility and Preliminary Efficacy of Wearable Focal Vibration Therapy on Gait and Mobility in People with Multiple Sclerosis: A Pilot Study. Bioengineering 2025, 12, 932. https://doi.org/10.3390/bioengineering12090932
Wang H, Shin YC, Tester NJ, Rempe T. Feasibility and Preliminary Efficacy of Wearable Focal Vibration Therapy on Gait and Mobility in People with Multiple Sclerosis: A Pilot Study. Bioengineering. 2025; 12(9):932. https://doi.org/10.3390/bioengineering12090932
Chicago/Turabian StyleWang, Hongwu, Yun Chan Shin, Nicole J. Tester, and Torge Rempe. 2025. "Feasibility and Preliminary Efficacy of Wearable Focal Vibration Therapy on Gait and Mobility in People with Multiple Sclerosis: A Pilot Study" Bioengineering 12, no. 9: 932. https://doi.org/10.3390/bioengineering12090932
APA StyleWang, H., Shin, Y. C., Tester, N. J., & Rempe, T. (2025). Feasibility and Preliminary Efficacy of Wearable Focal Vibration Therapy on Gait and Mobility in People with Multiple Sclerosis: A Pilot Study. Bioengineering, 12(9), 932. https://doi.org/10.3390/bioengineering12090932









