Integration of Artificial Intelligence and Wearable Devices in Pediatric Clinical Care: A Review
Abstract
1. Introduction
2. Methods
2.1. Study Design
- What types of wearable biosensors are used in pediatric hospital and outpatient settings?
- Which clinical domains have seen the use of wearable devices?
- How and to what extent have pediatric wearable devices incorporated AI beyond basic physiological metric estimation (e.g., HR, HRV, RR) to support outcome prediction, event detection, or clinical decision support?
- What barriers remain to their broader clinical adoption?
2.2. Search Strategy
2.3. Eligibility Criteria
2.4. Data Extraction and Analysis
3. Results
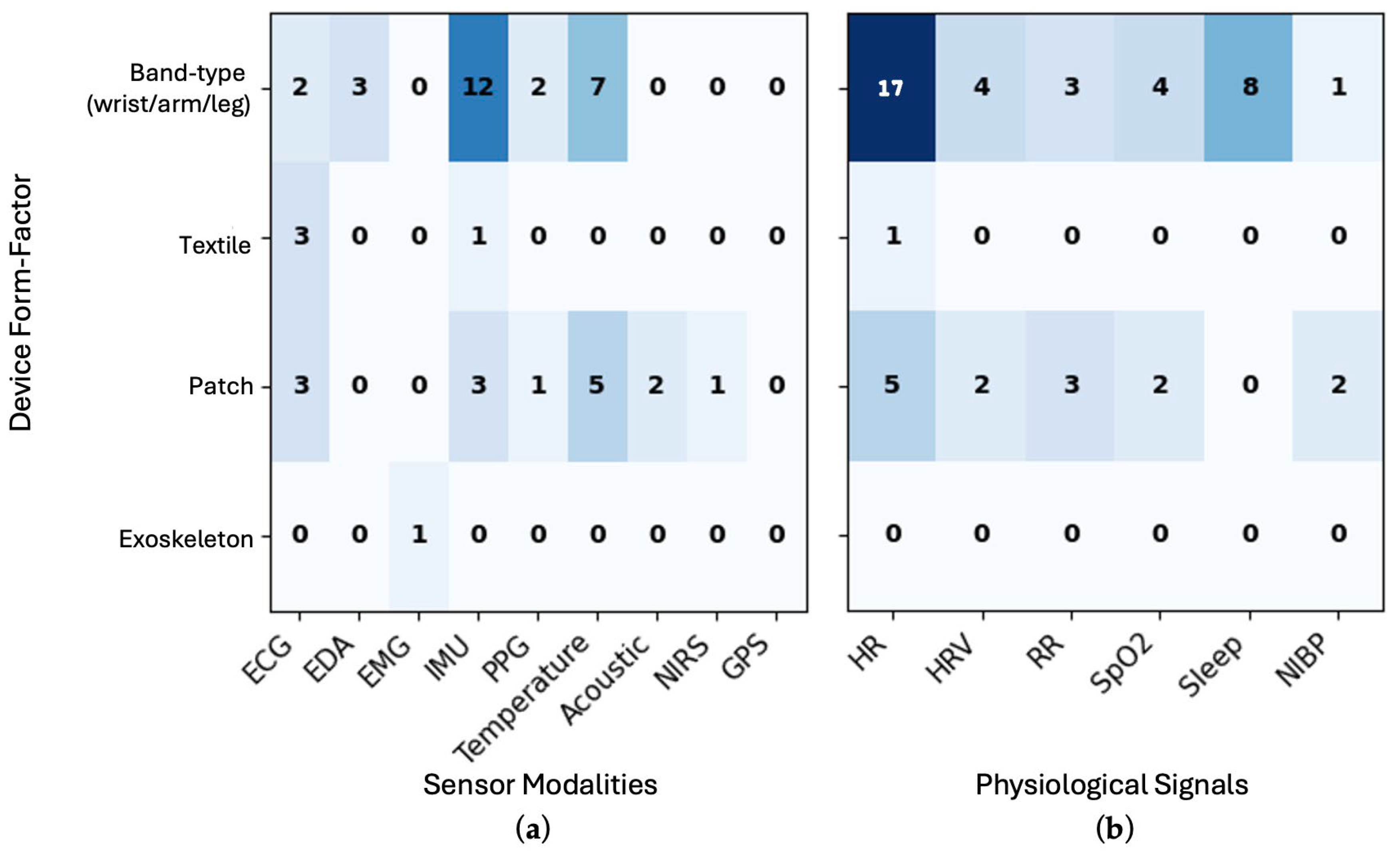
3.1. Device Types and Measures
3.1.1. Band-Type WDs
3.1.2. Patch WDs
3.1.3. Textile-Based WDs
3.1.4. Special-Purpose WDs
3.2. Clinical Domains and AI Applications
3.2.1. Infection and Sepsis
3.2.2. Surgery and Recovery
3.2.3. Seizure and Neuromonitoring
3.2.4. Respiratory Illness
3.2.5. Cardiology
3.2.6. Rehabilitation
3.2.7. Intensive Care
3.2.8. Enuresis
3.2.9. Psychiatric Dysregulation
3.3. Regulatory Landscape of Pediatric Wearables
4. Practical Challenges
4.1. Comfort, Fit, and Adherence
4.2. Signal Quality and Reliability
4.3. Regulatory and Labeling Constraints
4.4. Workflow Integration and Alert Governance
4.5. Ethics and Equity Related Challenges
4.6. AI Resource and Performance Trade-Offs
5. Future Directions and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Spatz, E.S.; Ginsburg, G.S.; Rumsfeld, J.S.; Turakhia, M.P. Wearable Digital Health Technologies for Monitoring in Cardiovascular Medicine. N. Engl. J. Med. 2024, 390, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Abdelrazik, A.; Eldesouky, M.; Antoun, I.; Lau, E.Y.M.; Koya, A.; Vali, Z.; Suleman, S.A.; Donaldson, J.; Ng, G.A. Wearable Devices for Arrhythmia Detection: Advancements and Clinical Implications. Sensors 2025, 25, 2848. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, A.; Changolkar, S.; Patel, M.S. Wearable Devices to Monitor and Reduce the Risk of Cardiovascular Disease: Evidence and Opportunities. Annu. Rev. Med. 2021, 72, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Tandon, A.; Avari Silva, J.N.; Bhatt, A.B.; Drummond, C.K.; Hill, A.C.; Paluch, A.E.; Waits, S.; Zablah, J.E.; Harris, K.C.; on behalf of the Council on Lifelong Congenital Heart Disease and Heart Health in the Young (Young Hearts); et al. Advancing Wearable Biosensors for Congenital Heart Disease: Patient and Clinician Perspectives: A Science Advisory From the American Heart Association. Circulation 2024, 149, e1134–e1142. [Google Scholar] [CrossRef]
- Amin, T.; Mobbs, R.J.; Mostafa, N.; Sy, L.W.; Choy, W.J. Wearable Devices for Patient Monitoring in the Early Postoperative Period: A Literature Review. Mhealth 2021, 7, 50. [Google Scholar] [CrossRef]
- Nino, V.; Claudio, D.; Schiel, C.; Bellows, B. Coupling Wearable Devices and Decision Theory in the United States Emergency Department Triage Process: A Narrative Review. Int. J. Environ. Res. Public Health 2020, 17, 9561. [Google Scholar] [CrossRef]
- Mattison, G.; Canfell, O.; Forrester, D.; Dobbins, C.; Smith, D.; Töyräs, J.; Sullivan, C. The Influence of Wearables on Health Care Outcomes in Chronic Disease: Systematic Review. J. Med. Internet Res. 2022, 24, e36690. [Google Scholar] [CrossRef]
- McErlane, F.; Davies, E.H.; Ollivier, C.; Mayhew, A.; Anyanwu, O.; Harbottle, V.; Donald, A. Wearable Technologies for Children with Chronic Illnesses: An Exploratory Approach. Ther. Innov. Regul. Sci. 2021, 55, 799–806. [Google Scholar] [CrossRef]
- Menassa, M.; Wilmont, I.; Beigrezaei, S.; Knobbe, A.; Arita, V.A.; Valderrama, J.F.; Bridge, L.; Verschuren, W.M.M.; Rennie, K.L.; Franco, O.H.; et al. The Future of Healthy Ageing: Wearables in Public Health, Disease Prevention and Healthcare. Maturitas 2025, 196, 108254. [Google Scholar] [CrossRef]
- Lee, Y.J.; Park, C.; Kim, H.; Cho, S.J.; Yeo, W.-H. Artificial Intelligence on Biomedical Signals: Technologies, Applications, and Future Directions. Med-X 2024, 2, 25. [Google Scholar] [CrossRef]
- Hyun, A.; Takashima, M.; Hall, S.; Lee, L.; Dufficy, M.; Ruppel, H.; Ullman, A. Wearable Biosensors for Pediatric Hospitals: A Scoping Review. Pediatr. Res. 2025, 98, 90–99. [Google Scholar] [CrossRef]
- Grooby, E.; Sitaula, C.; Chang Kwok, T.; Sharkey, D.; Marzbanrad, F.; Malhotra, A. Artificial Intelligence-Driven Wearable Technologies for Neonatal Cardiorespiratory Monitoring: Part 1 Wearable Technology. Pediatr. Res. 2023, 93, 413–425. [Google Scholar] [CrossRef]
- Sitaula, C.; Grooby, E.; Kwok, T.C.; Sharkey, D.; Marzbanrad, F.; Malhotra, A. Artificial Intelligence-Driven Wearable Technologies for Neonatal Cardiorespiratory Monitoring. Part 2: Artificial Intelligence. Pediatr. Res. 2023, 93, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Magsayo, K.-A.; Khatami Firoozabadi, S.F. Non-Invasive Wearables in Pediatric Healthcare: A Comprehensive Review of Uses and Implications. Children 2025, 12, 1233. [Google Scholar] [CrossRef] [PubMed]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Sharma, P.; Johnson, M.; Danieletto, M.; Alleva, E.; Charney, A.; Glicksberg, B. Advancements in Artificial Intelligence and Wearable Devices in Pediatric Clinical Care: A Scoping Review Protocol. Available online: https://inplasy.com/inplasy-2025-10-0096/ (accessed on 3 November 2025).
- Jeong, J.; Yeo, W.; Akhtar, A.; Norton, J.J.S.; Kwack, Y.; Li, S.; Jung, S.; Su, Y.; Lee, W.; Xia, J.; et al. Materials and Optimized Designs for Human-Machine Interfaces Via Epidermal Electronics. Adv. Mater. 2013, 25, 6839–6846. [Google Scholar] [CrossRef]
- Xu, C.; Yang, Y.; Gao, W. Skin-Interfaced Sensors in Digital Medicine: From Materials to Applications. Matter 2020, 2, 1414–1445. [Google Scholar] [CrossRef]
- Charlton, P.H.; Allen, J.; Bailón, R.; Baker, S.; Behar, J.A.; Chen, F.; Clifford, G.D.; Clifton, D.A.; Davies, H.J.; Ding, C.; et al. The 2023 Wearable Photoplethysmography Roadmap. Physiol. Meas. 2023, 44, 111001. [Google Scholar] [CrossRef]
- Cesari, P.; Cristani, M.; Demrozi, F.; Pascucci, F.; Picotti, P.M.; Pravadelli, G.; Tomazzoli, C.; Turetta, C.; Workneh, T.C.; Zenti, L. Towards Posture and Gait Evaluation through Wearable-Based Biofeedback Technologies. Electronics 2023, 12, 644. [Google Scholar] [CrossRef]
- Zheng, Q.; Lee, J.; Shen, X.; Chen, X.; Kim, J.-K. Graphene-Based Wearable Piezoresistive Physical Sensors. Mater. Today 2020, 36, 158–179. [Google Scholar] [CrossRef]
- Jin, L.; Liu, Z.; Altintas, M.; Zheng, Y.; Liu, Z.; Yao, S.; Fan, Y.; Li, Y. Wearable Piezoelectric Airflow Transducers for Human Respiratory and Metabolic Monitoring. ACS Sens. 2022, 7, 2281–2292. [Google Scholar] [CrossRef]
- Arman Kuzubasoglu, B.; Kursun Bahadir, S. Flexible Temperature Sensors: A Review. Sens. Actuators A Phys. 2020, 315, 112282. [Google Scholar] [CrossRef]
- Shinde, S.; Kim, K.H.; Park, S.Y.; Kim, J.H.; Kim, J.; Joe, D.J.; Lee, H.E. Wearable Sweat-Sensing Patches for Non-Invasive and Continuous Health Tracking. Sens. Actuators Rep. 2025, 9, 100265. [Google Scholar] [CrossRef]
- Zahedivash, A.; Chubb, H.; Giacone, H.; Boramanand, N.K.; Dubin, A.M.; Trela, A.; Lencioni, E.; Motonaga, K.S.; Goodyer, W.; Navarre, B.; et al. Utility of Smart Watches for Identifying Arrhythmias in Children. Commun. Med. 2023, 3, 167. [Google Scholar] [CrossRef] [PubMed]
- Robles-Rubio, C.A.; Brown, K.A.; Kearney, R.E. A New Movement Artifact Detector for Photoplethysmographic Signals. In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; IEEE: Piscataway, NJ, USA, 2013; pp. 2295–2299. [Google Scholar]
- Carter, M.; Linton, S.C.; Zeineddin, S.; Pitt, J.B.; De Boer, C.; Figueroa, A.; Gosain, A.; Lanning, D.; Lesher, A.; Islam, S.; et al. Impact of Consumer Wearables Data on Pediatric Surgery Clinicians’ Management: Multi-Institutional Scenario-Based Usability Study. JMIR Perioper. Med. 2024, 7, e58663. [Google Scholar] [CrossRef]
- Onorati, F.; Regalia, G.; Caborni, C.; LaFrance, W.C.; Blum, A.S.; Bidwell, J.; De Liso, P.; El Atrache, R.; Loddenkemper, T.; Mohammadpour-Touserkani, F.; et al. Prospective Study of a Multimodal Convulsive Seizure Detection Wearable System on Pediatric and Adult Patients in the Epilepsy Monitoring Unit. Front. Neurol. 2021, 12, 724904. [Google Scholar] [CrossRef]
- Littell, L.; Roelle, L.; Dalal, A.; Van Hare, G.F.; Orr, W.B.; Miller, N.; Avari Silva, J.N. Assessment of Apple Watch Series 6 Pulse Oximetry and Electrocardiograms in a Pediatric Population. PLoS Digit. Health 2022, 1, e0000051. [Google Scholar] [CrossRef]
- Garbern, S.C.; Mamun, G.M.S.; Shaima, S.N.; Hakim, N.; Wegerich, S.; Alla, S.; Sarmin, M.; Afroze, F.; Sekaric, J.; Genisca, A.; et al. A Novel Digital Health Approach to Improving Global Pediatric Sepsis Care in Bangladesh Using Wearable Technology and Machine Learning. PLoS Digit. Health 2024, 3, e0000634. [Google Scholar] [CrossRef]
- Nessle, C.N.; Flora, C.; Sandford, E.; Choi, S.W.; Tewari, M. High-frequency Temperature Monitoring at Home Using a Wearable Device: A Case Series of Early Fever Detection and Antibiotic Administration for Febrile Neutropenia with Bacteremia. Pediatr. Blood Cancer 2022, 69, e29835. [Google Scholar] [CrossRef]
- Tan, R.; Ang, Y.Y.; Ang, A.Q.X.; Wai, K.; Ramamurthy, M.B.; Goh, D.Y.T.; Lim, M.T.C. Evaluating a Wearable Device for Wheeze Detection in the Pediatric Emergency Department: A Feasibility Study. Am. J. Respir. Crit. Care Med. 2025, 211, A2296. [Google Scholar] [CrossRef]
- Chung, H.U.; Rwei, A.Y.; Hourlier-Fargette, A.; Xu, S.; Lee, K.; Dunne, E.C.; Xie, Z.; Liu, C.; Carlini, A.; Kim, D.H.; et al. Skin-Interfaced Biosensors for Advanced Wireless Physiological Monitoring in Neonatal and Pediatric Intensive-Care Units. Nat. Med. 2020, 26, 418–429. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Kim, J.; Kwak, S.S.; Hourlier-Fargette, A.; Avila, R.; Vogl, J.; Tzavelis, A.; Chung, H.U.; Lee, J.Y.; Kim, D.H.; et al. Wireless, Skin-Interfaced Devices for Pediatric Critical Care: Application to Continuous, Noninvasive Blood Pressure Monitoring. Adv. Healthc. Mater. 2021, 10, 2100383. [Google Scholar] [CrossRef] [PubMed]
- Rwei, A.Y.; Lu, W.; Wu, C.; Human, K.; Suen, E.; Franklin, D.; Fabiani, M.; Gratton, G.; Xie, Z.; Deng, Y.; et al. A Wireless, Skin-Interfaced Biosensor for Cerebral Hemodynamic Monitoring in Pediatric Care. Proc. Natl. Acad. Sci. USA 2020, 117, 31674–31684. [Google Scholar] [CrossRef]
- Tzavelis, A.; Palla, J.; Mathur, R.; Bedford, B.; Wu, Y.-H.; Trueb, J.; Shin, H.S.; Arafa, H.; Jeong, H.; Ouyang, W.; et al. Development of a Miniaturized Mechanoacoustic Sensor for Continuous, Objective Cough Detection, Characterization and Physiologic Monitoring in Children with Cystic Fibrosis. IEEE J. Biomed. Health Inform. 2024, 28, 5941–5952. [Google Scholar] [CrossRef]
- Breteler, M.J.M.; KleinJan, E.J.; Dohmen, D.A.J.; Leenen, L.P.H.; Van Hillegersberg, R.; Ruurda, J.P.; Van Loon, K.; Blokhuis, T.J.; Kalkman, C.J. Vital Signs Monitoring with Wearable Sensors in High-Risk Surgical Patients: A Clinical Validation Study. Anesthesiology 2020, 132, 424–439. [Google Scholar] [CrossRef]
- Morgado Areia, C.; Santos, M.; Vollam, S.; Pimentel, M.; Young, L.; Roman, C.; Ede, J.; Piper, P.; King, E.; Gustafson, O.; et al. A Chest Patch for Continuous Vital Sign Monitoring: Clinical Validation Study During Movement and Controlled Hypoxia. J. Med. Internet Res. 2021, 23, e27547. [Google Scholar] [CrossRef]
- Lund, C. Medical Adhesives in the NICU. Newborn Infant. Nurs. Rev. 2014, 14, 160–165. [Google Scholar] [CrossRef]
- Nikolova, E.; Ganev, B.; Gieva, E. Wearable Intelligent Textile Suits for Telemetry Monitoring in Pediatrics. In Proceedings of the 2021 XXX International Scientific Conference Electronics (ET), Sozopol, Bulgaria, 15–17 September 2021; IEEE: Piscataway, NJ, USA, 2021; pp. 1–6. [Google Scholar]
- Montazeri Ghahjaverstan, N.; Balmer-Minnes, D.; Taghibeyglou, B.; Moineau, B.; Chaves, G.; Alizadeh-Meghrazi, M.; Cifra, B.; Jeewa, A.; Yadollahi, A. Textile-Based Wearable to Monitor Heart Activity in Paediatric Population: A Pilot Study. CJC Pediatr. Congenit. Heart Dis. 2023, 2, 187–195. [Google Scholar] [CrossRef]
- Dangerfield, M.I.; Ward, K.; Davidson, L.; Adamian, M. Initial Experience and Usage Patterns with the Owlet Smart Sock Monitor in 47,495 Newborns. Glob. Pediatr. Health 2017, 4, 2333794X17742751. [Google Scholar] [CrossRef]
- Ritchie, H.; Kang, M.; Feng, J.; Morshedzadeh, E.; Luis Contreras-vidal, J.; Sarkar, S.; Judith Aguilar Herrera, A.; Willson, P. Designing Adaptive Wearables for Children with Cerebral Palsy: A Holistic Approach to Lifestyle-Centric Garment Design. In Proceedings of the 16th International Conference on Applied Human Factors and Ergonomics (AHFE 2025), Orlando, FL, USA, 26–30 July 2025. [Google Scholar]
- Butzer, T.; Dittli, J.; Lieber, J.; Van Hedel, H.J.A.; Meyer-Heim, A.; Lambercy, O.; Gassert, R. PEXO—A Pediatric Whole Hand Exoskeleton for Grasping Assistance in Task-Oriented Training. In Proceedings of the 2019 IEEE 16th International Conference on Rehabilitation Robotics (ICORR), Toronto, ON, Canada, 24–28 June 2019; IEEE: Piscataway, NJ, USA, 2019; pp. 108–114. [Google Scholar]
- Spar, D.S.; Bianco, N.R.; Knilans, T.K.; Czosek, R.J.; Anderson, J.B. The US Experience of the Wearable Cardioverter-Defibrillator in Pediatric Patients. Circ. Arrhythmia Electrophysiol. 2018, 11, e006163. [Google Scholar] [CrossRef]
- Hua, R.; O’Brien, M.K.; Carter, M.; Pitt, J.B.; Kwon, S.; Ghomrawi, H.M.K.; Jayaraman, A.; Abdullah, F. Improving Early Prediction of Abnormal Recovery after Appendectomy in Children Using Real-World Data from Wearables. In Proceedings of the 2024 46th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 15–19 July 2024; IEEE: Piscataway, NJ, USA, 2024; pp. 1–4. [Google Scholar]
- Ghomrawi, H.M.K.; O’Brien, M.K.; Carter, M.; Macaluso, R.; Khazanchi, R.; Fanton, M.; DeBoer, C.; Linton, S.C.; Zeineddin, S.; Pitt, J.B.; et al. Applying Machine Learning to Consumer Wearable Data for the Early Detection of Complications after Pediatric Appendectomy. npj Digit. Med. 2023, 6, 148. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; El Atrache, R.; Tang, J.; Jackson, M.; Makarucha, A.; Cantley, S.; Sheehan, T.; Vieluf, S.; Zhang, B.; Rogers, J.L.; et al. Artificial Intelligence-enhanced Epileptic Seizure Detection by Wearables. Epilepsia 2023, 64, 3213–3226. [Google Scholar] [CrossRef] [PubMed]
- Teich, L.; Franke, D.; Michaelis, A.; Dähnert, I.; Gebauer, R.A.; Markel, F.; Paech, C. Development of an AI Based Automated Analysis of Pediatric Apple Watch iECGs. Front. Pediatr. 2023, 11, 1185629. [Google Scholar] [CrossRef] [PubMed]
- Koenig, C.; Ammann, R.A.; Kuehni, C.E.; Roessler, J.; Brack, E. Continuous Recording of Vital Signs with a Wearable Device in Pediatric Patients Undergoing Chemotherapy for Cancer—An Operational Feasibility Study. Support. Care Cancer 2021, 29, 5283–5292. [Google Scholar] [CrossRef]
- Haemmerli, M.; Ammann, R.A.; Roessler, J.; Koenig, C.; Brack, E. Vital Signs in Pediatric Oncology Patients Assessed by Continuous Recording with a Wearable Device, NCT04134429. Sci. Data 2022, 9, 89. [Google Scholar] [CrossRef]
- Mack, I.; Juchler, N.; Rey, S.; Hirsch, S.; Hoelz, B.; Eckstein, J.; Bielicki, J. Wearable Technologies for Pediatric Patients with Surgical Infections-More than Counting Steps? Biosensors 2022, 12, 634. [Google Scholar] [CrossRef]
- Koenig, C.; Ammann, R.A.; Schneider, C.; Wyss, J.; Roessler, J.; Brack, E. Continuous Timely Monitoring of Core Temperature with Two Wearable Devices in Pediatric Patients Undergoing Chemotherapy for Cancer—A Comparison Study. Support. Care Cancer 2024, 32, 188. [Google Scholar] [CrossRef]
- Farner, L.; König, C.; Rössler, J.; Schneider, C.; Wyss, J.; Ammann, R.A.; Brack, E. Vital Signs Continuously Monitored by Two Wearable Devices in Pediatric Oncology Patients, NCT04914702. Sci. Data 2025, 12, 807. [Google Scholar] [CrossRef]
- De Boer, C.; Ghomrawi, H.; Many, B.; Bouchard, M.E.; Linton, S.; Figueroa, A.; Kwon, S.; Abdullah, F. Utility of Wearable Sensors to Assess Postoperative Recovery in Pediatric Patients After Appendectomy. J. Surg. Res. 2021, 263, 160–166. [Google Scholar] [CrossRef]
- Hua, R.; Carter, M.; O’Brien, M.K.; Pitt, J.B.; Kwon, S.; Manworren, R.C.B.; Oscherwitz, G.; Edobor, A.; Chen, A.; Ghomrawi, H.M.K.; et al. Biorhythms Derived from Consumer Wearables Predict Postoperative Complications in Children. Sci. Adv. 2025, 11, eadv2643. [Google Scholar] [CrossRef]
- Pitt, J.B.; Carter, M.; Zeineddin, S.; Bai, I.; Hua, R.; Kwon, S.; Ghomrawi, H.; Abdullah, F. Association of Wearable Derived Pulse Rate Variability with Pediatric Appendectomy Complications. J. Pediatr. Surg. 2025, 60, 162016. [Google Scholar] [CrossRef] [PubMed]
- Zeineddin, S.; Figueroa, A.; Pitt, J.B.; Carter, M.; DeTella, M.; Perez, A.; Park, E.; Kwon, S.; Ghomrawi, H.; Abdullah, F. Using Wearable Devices to Profile Demographic-Specific Recovery After Pediatric Appendectomy. J. Surg. Res. 2024, 295, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Pelizzo, G.; Guddo, A.; Puglisi, A.; De Silvestri, A.; Comparato, C.; Valenza, M.; Bordonaro, E.; Calcaterra, V. Accuracy of a Wrist-Worn Heart Rate Sensing Device during Elective Pediatric Surgical Procedures. Children 2018, 5, 38. [Google Scholar] [CrossRef] [PubMed]
- Davies, E.H.; Fieggen, K.; Wilmshurst, J.; Anyanwu, O.; Burman, R.J.; Komarzynski, S. Demonstrating the Feasibility of Digital Health to Support Pediatric Patients in South Africa. Epilepsia Open 2021, 6, 653–662. [Google Scholar] [CrossRef]
- Kruizinga, M.D.; Moll, A.; Zhuparris, A.; Ziagkos, D.; Stuurman, F.E.; Nuijsink, M.; Cohen, A.F.; Driessen, G.J.A. PEXO—A Pediatric Whole Hand Exoskeleton forCan Be Quantified with Digital Biomarkers. Respiration 2021, 100, 979–988. [Google Scholar] [CrossRef]
- Avitabile, C.M.; Mota, J.P.; Yeaman, K.M.; Andrieux, S.J.; Lechtenberg, L.; Escobar, E.; Chuo, J.; Xanthopoulos, M.S.; Faig, W.; O’Malley, S.M.; et al. Creating a Digital Approach for Promoting Physical Activity in Pediatric Pulmonary Hypertension: A Framework for Future Interventions. Pulm. Circ. 2024, 14, e12402. [Google Scholar] [CrossRef]
- Yan, K.; Tracie, B.; Marie-Ève, M.; Mélanie, H.; Jean-Luc, B.; Benoit, T.; St-Onge, M.; Marie, L. Innovation through Wearable Sensors to Collect Real-Life Data among Pediatric Patients with Cardiometabolic Risk Factors. Int. J. Pediatr. 2014, 2014, 328076. [Google Scholar] [CrossRef]
- Lee, S.; Moon, J.; Lee, Y.S.; Shin, S.; Lee, K. Wearable-Based Integrated System for In-Home Monitoring and Analysis of Nocturnal Enuresis. Sensors 2024, 24, 3330. [Google Scholar] [CrossRef]
- Romanowicz, M.; Croarkin, K.S.; Elmaghraby, R.; Skime, M.; Mayo Clinic Inpatient Child and Adolescent Psychiatry Registered Nurse Staff; Croarkin, P.E.; Vande Voort, J.L.; Shekunov, J.; Athreya, A.P. Machine Learning Identifies Smartwatch-Based Physiological Biomarker for Predicting Disruptive Behavior in Children: A Feasibility Study. J. Child. Adolesc. Psychopharmacol. 2023, 33, 387–392. [Google Scholar] [CrossRef]
- Empatic Embrace 510(k) Premarket Notification. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K181861 (accessed on 8 October 2025).
- TempTraq TT100 510(k) Premarket Notification. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K143267 (accessed on 8 October 2025).
- ANNE Pediatric 510(k) Premarket Notification. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K221530 (accessed on 8 October 2025).
- ActiGraph LEAP 510(k) Premarket Notification. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=k231532 (accessed on 8 October 2025).
- Owlet Dream Sock Device Classification Under Section 513(f)(2)(De Novo). Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/denovo.cfm?id=DEN220091 (accessed on 8 October 2025).
- Lifevest Wearable Defibrillator Premarket Approval (PMA). Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpma/pma.cfm?id=P010030S056 (accessed on 8 October 2025).
- AeviceMD 510(k) Premarket Notification. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K223382 (accessed on 8 October 2025).
- Tonino, R.P.B.; Larimer, K.; Eissen, O.; Schipperus, M.R. Remote Patient Monitoring in Adults Receiving Transfusion or Infusion for Hematological Disorders Using the VitalPatch and accelerateIQ Monitoring System: Quantitative Feasibility Study. JMIR Hum. Factors 2019, 6, e15103. [Google Scholar] [CrossRef]
- Silverstein, L.; Dreger, N.; Anza, O.A.; Behere, S. Resource Use and Clinical Outcomes in Infants with Supraventricular Tachycardia Monitored with the Owlet Smart Sock. J. Pediatr. 2024, 268, 113946. [Google Scholar] [CrossRef]
- Alzghaibi, H. Nurses’ Perspectives on AI-Enabled Wearable Health Technologies: Opportunities and Challenges in Clinical Practice. BMC Nurs. 2025, 24, 799. [Google Scholar] [CrossRef]
- Swaroop, S.; Buçinca, Z.; Gajos, K.Z.; Doshi-Velez, F. Accuracy-Time Tradeoffs in AI-Assisted Decision Making under Time Pressure. In Proceedings of the 29th International Conference on Intelligent User Interfaces, Greenville, SC, USA, 18 March 2024; ACM: New York, NY, USA, 2024; pp. 138–154. [Google Scholar]
- Wang, X.; Tang, Z.; Guo, J.; Meng, T.; Wang, C.; Wang, T.; Jia, W. Empowering Edge Intelligence: A Comprehensive Survey on On-Device AI Models. ACM Comput. Surv. 2025, 57, 1–39. [Google Scholar] [CrossRef]
- Erion, G.; Janizek, J.D.; Hudelson, C.; Utarnachitt, R.B.; McCoy, A.M.; Sayre, M.R.; White, N.J.; Lee, S.-I. A Cost-Aware Framework for the Development of AI Models for Healthcare Applications. Nat. Biomed. Eng. 2022, 6, 1384–1398. [Google Scholar] [CrossRef]
- Alsahfi, T.; Badshah, A.; Aboulola, O.I.; Daud, A. Optimizing Healthcare Big Data Performance through Regional Computing. Sci. Rep. 2025, 15, 3129. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, H.; Sharma, P.; Johnson, M.; Danieletto, M.; Alleva, E.; Charney, A.W.; Nadkarni, G.N.; Sarabu, C.; Eskofier, B.M.; Ahuja, Y.; et al. Integration of Artificial Intelligence and Wearable Devices in Pediatric Clinical Care: A Review. Bioengineering 2025, 12, 1320. https://doi.org/10.3390/bioengineering12121320
Zheng H, Sharma P, Johnson M, Danieletto M, Alleva E, Charney AW, Nadkarni GN, Sarabu C, Eskofier BM, Ahuja Y, et al. Integration of Artificial Intelligence and Wearable Devices in Pediatric Clinical Care: A Review. Bioengineering. 2025; 12(12):1320. https://doi.org/10.3390/bioengineering12121320
Chicago/Turabian StyleZheng, Huili, Pragya Sharma, Matthew Johnson, Matteo Danieletto, Eugenia Alleva, Alexander W. Charney, Girish N. Nadkarni, Chethan Sarabu, Bjoern M. Eskofier, Yuri Ahuja, and et al. 2025. "Integration of Artificial Intelligence and Wearable Devices in Pediatric Clinical Care: A Review" Bioengineering 12, no. 12: 1320. https://doi.org/10.3390/bioengineering12121320
APA StyleZheng, H., Sharma, P., Johnson, M., Danieletto, M., Alleva, E., Charney, A. W., Nadkarni, G. N., Sarabu, C., Eskofier, B. M., Ahuja, Y., Richter, F., Klang, E., Gangadharan, S., Richter, F., Holmes, E., & Glicksberg, B. S. (2025). Integration of Artificial Intelligence and Wearable Devices in Pediatric Clinical Care: A Review. Bioengineering, 12(12), 1320. https://doi.org/10.3390/bioengineering12121320







