Extreme Serum Titanium Concentration Induced by Acetabular Cup Failure: Unveiling a Unique Scenario of Titanium Alloy Debris Accumulation
Abstract
1. Introduction
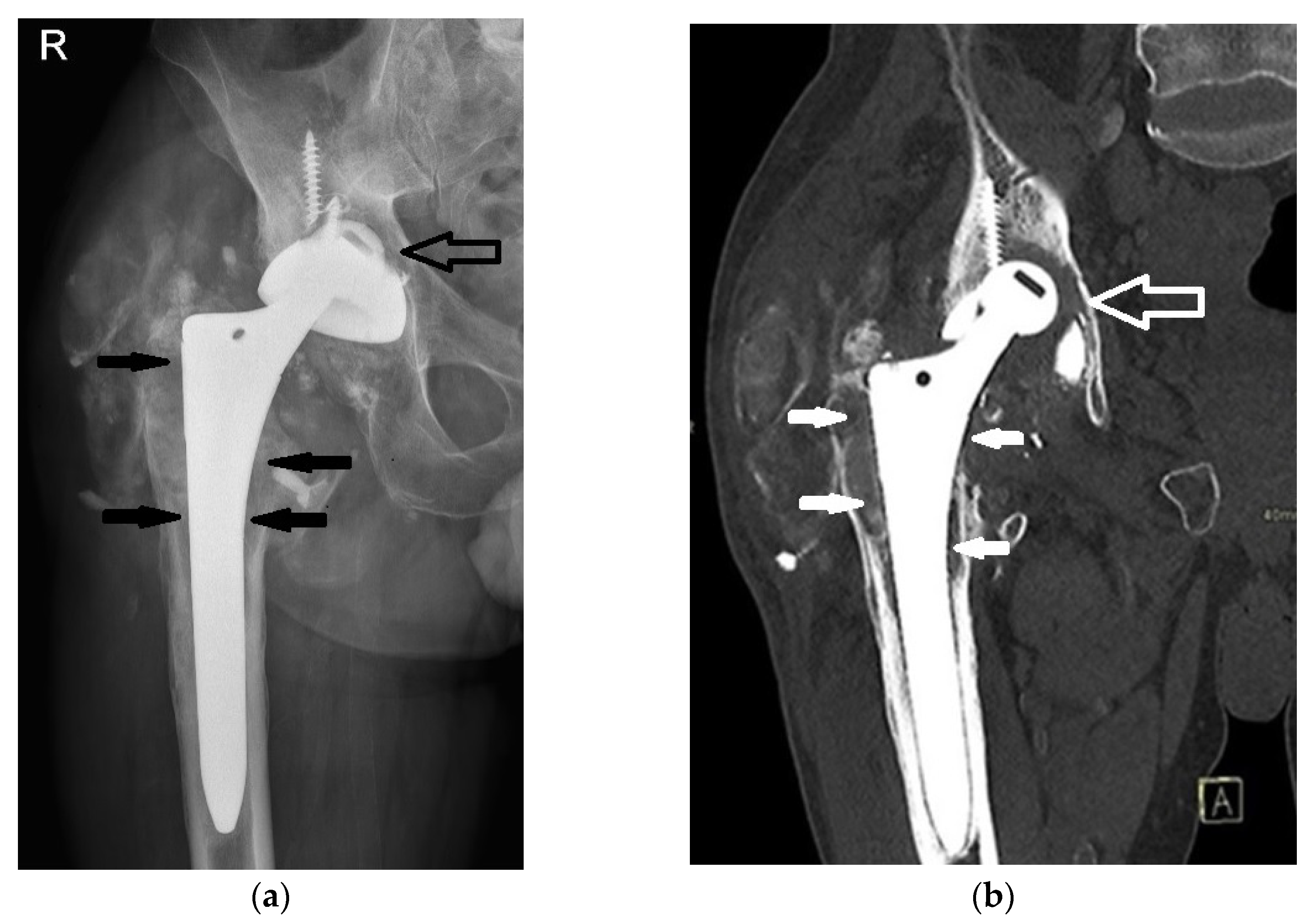
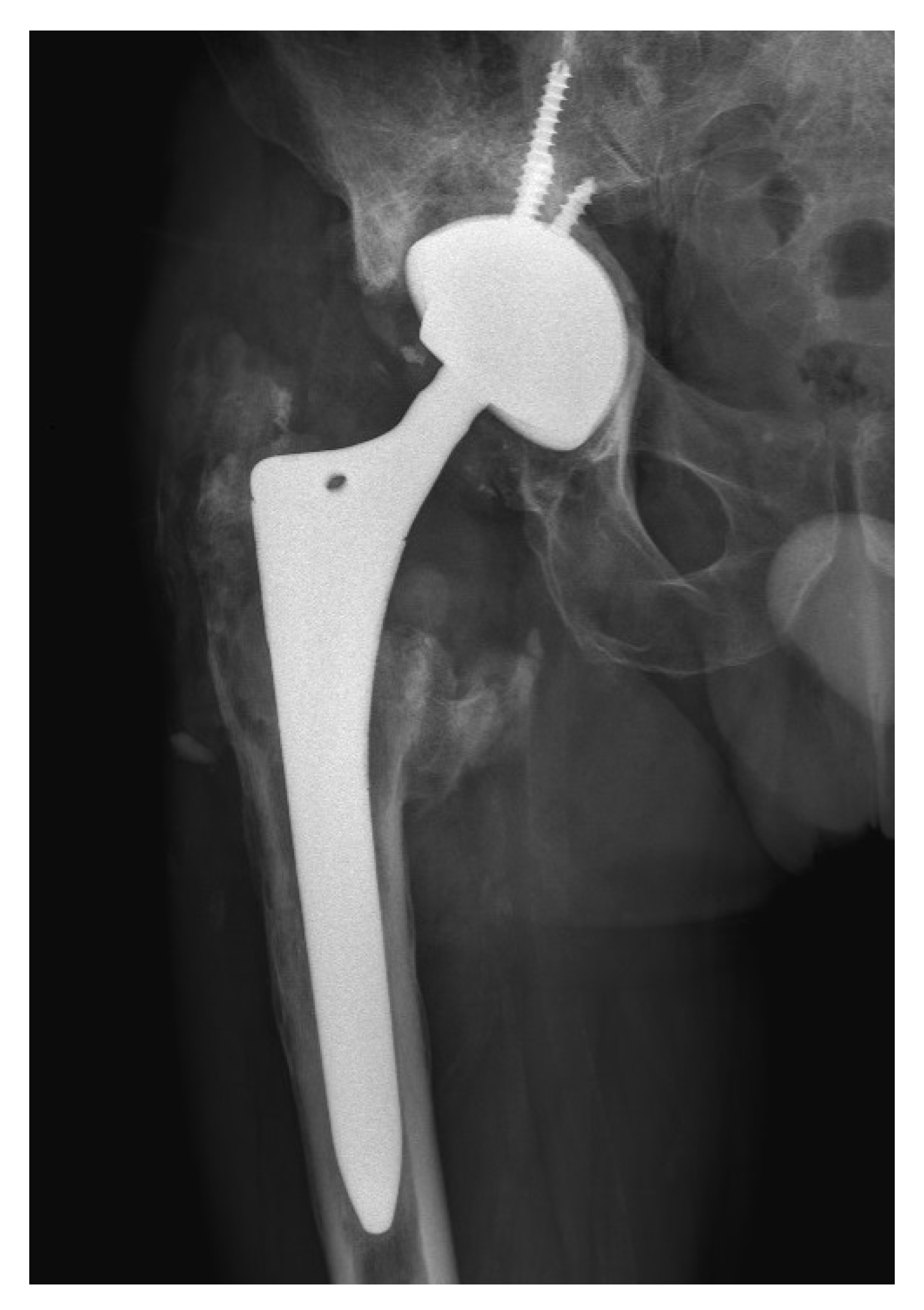
2. Case Presentation
2.1. Serum Trace Element Analysis
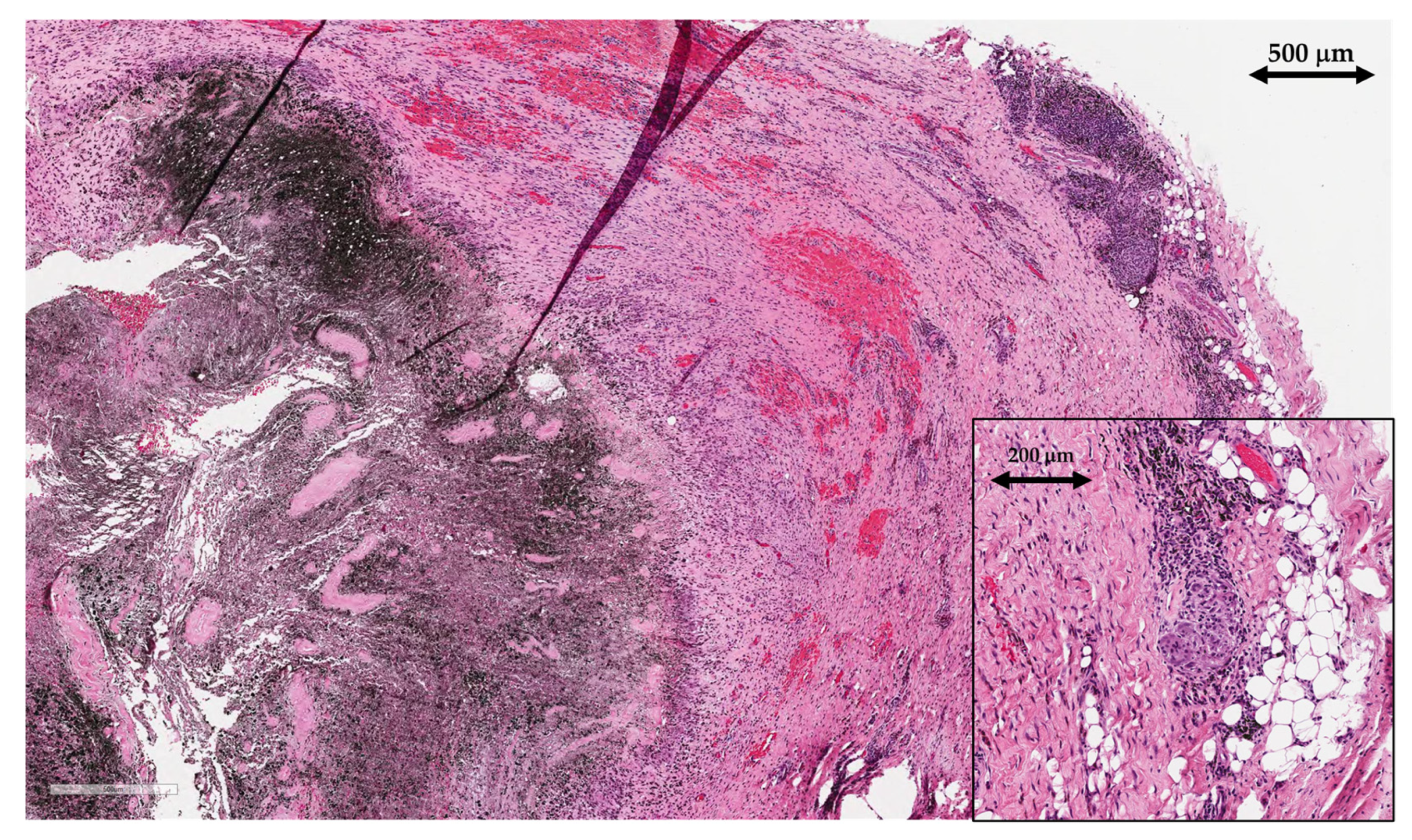
2.2. Histological Analysis
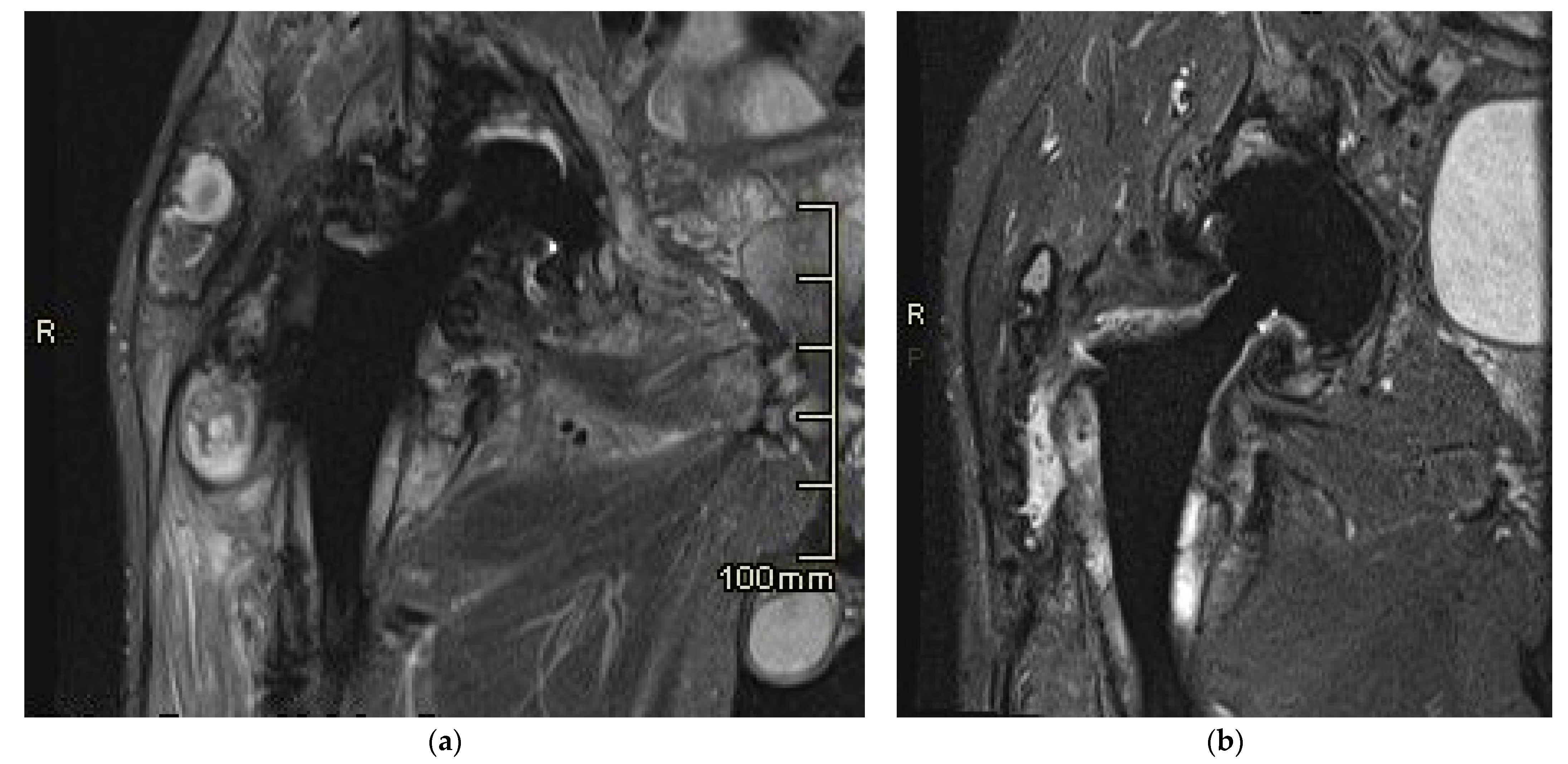
2.3. MR Imaging
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Katz, J.N.; Arant, K.R.; Loeser, R.F. Diagnosis and Treatment of Hip and Knee Osteoarthritis: A Review. JAMA 2021, 325, 568–578. [Google Scholar] [CrossRef] [PubMed]
- Long, M.; Rack, H.J. Titanium alloys in total joint replacement—A materials science perspective. Biomaterials 1998, 19, 1621–1639. [Google Scholar] [CrossRef] [PubMed]
- Mistry, J.B.; Chughtai, M.; Elmallah, R.K.; Diedrich, A.; Le, S.; Thomas, M.; Mont, M.A. Trunnionosis in total hip arthroplasty: A review. J. Orthop. Traumatol. Off. J. Ital. Soc. Orthop. Traumatol. 2016, 17, 1–6. [Google Scholar] [CrossRef]
- Van Doesburg, P.G.; van Langelaan, E.J.; Apachitei, I.; Bénard, M.R.; Verdegaal, S.H.M. Femoral prosthesis neck fracture following total hip arthroplasty—A systematic review. Arthroplasty 2020, 2, 28. [Google Scholar] [CrossRef]
- Berstock, J.R.; Whitehouse, M.R.; Duncan, C.P. Trunnion corrosion: What surgeons need to know in 2018. Bone Jt. J. 2018, 100 (1 Suppl. A), 44–49. [Google Scholar] [CrossRef]
- Nugent, M.; Young, S.W.; Frampton, C.M.; Hooper, G.J. The lifetime risk of revision following total hip arthroplasty. Bone Jt. J. 2021, 103, 479–485. [Google Scholar] [CrossRef]
- Prokopetz, J.J.; Losina, E.; Bliss, R.L.; Wright, J.; Baron, J.A.; Katz, J.N. Risk factors for revision of primary total hip arthroplasty: A systematic review. BMC Musculoskel. Disord. 2012, 13, 251. [Google Scholar] [CrossRef]
- Rowan, F.E.; Wach, A.; Wright, T.M.; Padgett, D.E. The onset of fretting at the head-stem connection in hip arthroplasty is affected by head material and trunnion design under simulated corrosion conditions. J. Orthop. Traumatol. Off. J. Ital. Soc. Orthop. Traumatol. 2018, 36, 1630–1636. [Google Scholar] [CrossRef]
- Podlipec, R.; Punzón-Quijorna, E.; Pirker, L.; Kelemen, M.; Vavpetič, P.; Kavalar, R.; Hlawacek, G.; Štrancar, J.; Pelicon, P.; Fokter, S.K. Revealing Inflammatory Indications Induced by Titanium Alloy Wear Debris in Periprosthetic Tissue by Label-Free Correlative High-Resolution Ion, Electron and Optical Microspectroscopy. Materials 2021, 14, 3048. [Google Scholar] [CrossRef]
- Solou, K.; Panagopoulos, A.; Tatani, I.; Megas, P. Fracture of femoral neck in modular total hip arthroplasty: A systematic review of the literature. Hip Int. J. Clin. Exp. Res. Hip Pathol. Ther. 2023, 11207000231211253, Advance online publication. [Google Scholar] [CrossRef] [PubMed]
- Kavalar, R.; Fokter, S.K.; Lamovec, J. Total hip arthroplasty-related osteogenic osteosarcoma: Case report and review of the literature. Eur. J. Med. Res. 2016, 21, 8. [Google Scholar] [CrossRef]
- Yao, J.J.; Lewallen, E.A.; Thaler, R.; Dudakovic, A.; Wermers, M.; Day, P.; Eckdahl, S.; Jannetto, P.; Bornhorst, J.A.; Larson, A.N.; et al. Challenges in the Measurement and Interpretation of Serum Titanium Concentrations. Biol. Trace Elem. Res. 2020, 196, 20–26. [Google Scholar] [CrossRef]
- Fokter, S.K.; Zajc, J.; Merc, M. Interchangeable neck failures of bi-modular femoral stems in primary total hip arthroplasty cannot be predicted from serum trace element analysis. Int. Orthop. 2021, 45, 877–881. [Google Scholar] [CrossRef]
- Peterson, S.M.; O’Byrne, T.J.; Brennan, P.C.; Jannetto, P.J.; Pavelko, K.D.; Lewallen, D.G.; Vassilaki, M.; Maradit Kremers, H. Cross-sectional association between systemic metal concentrations and immune markers in patients with total joint arthroplasty. Front. Immunol. 2023, 14, 1130209. [Google Scholar] [CrossRef]
- Krenn, V.; Perino, G. Histological Diagnosis of Implant-Associated Pathologies; Springer: Berlin/Heidelberg, Germany, 2017; pp. 1–44. [Google Scholar] [CrossRef]
- Jacobs, J.J.; Skipor, A.K.; Black, J.; Urban, R.M.; Galante, J.O. Release and excretion of metal in patients who have a total hip-replacement component made of titanium-base alloy. J. Bone Jt. Surg. Am. Vol. 1991, 73, 1475–1486. [Google Scholar] [CrossRef]
- Omlor, G.W.; Kretzer, J.P.; Reinders, J.; Streit, M.R.; Bruckner, T.; Gotterbarm, T.; Aldinger, P.R.; Merle, C. In vivo serum titanium ion levels following modular neck total hip arthroplasty—10 year results in 67 patients. Acta Biomater. 2013, 9, 6278–6282. [Google Scholar] [CrossRef]
- McAlister, I.P.; Abdel, M.P. Elevated Serum Titanium Level as a Marker for Failure in a Titanium Modular Fluted Tapered Stem. Orthopedics 2016, 39, e768–e770. [Google Scholar] [CrossRef] [PubMed]
- Swiatkowska, I.; Martin, N.; Hart, A.J. Blood titanium level as a biomarker of orthopaedic implant wear. J. Trace Elem. Med. Biol. Organ Soc. Miner. Trace Elem. 2019, 53, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, M.; Watanabe, H.; Higashi, H.; Kubosawa, H. Pseudotumor Caused by Titanium Particles from a Total Hip Prosthesis. Orthopedics 2016, 39, e162–e165. [Google Scholar] [CrossRef] [PubMed]
- Agins, H.J.; Alcock, N.W.; Bansal, M.; Salvati, E.A.; Wilson, P.D., Jr.; Pellicci, P.M.; Bullough, P.G. Metallic wear in failed titanium-alloy total hip replacements. A histological and quantitative analysis. J. Bone Jt. Surg. 1988, 70, 347–356. [Google Scholar] [CrossRef]
- Moran, C.A.; Mullick, F.G.; Ishak, K.G.; Johnson, F.B.; Hummer, W.B. Identification of titanium in human tissues: Probable role in pathologic processes. Hum. Pathol. 1991, 22, 450–454. [Google Scholar] [CrossRef]
- Koller, D.; Bramhall, P.; Devoy, J.; Goenaga-Infante, H.; Harrington, C.F.; Leese, E.; Morton, J.; Nuñez, S.; Rogers, J.; Sampson, B.; et al. Analysis of soluble or titanium dioxide derived titanium levels in human whole blood: Consensus from an inter-laboratory comparison. Analyst 2018, 143, 5520–5529. [Google Scholar] [CrossRef]
- Perino, G.; Sunitsch, S.; Huber, M.; Ramirez, D.; Gallo, J.; Vaculova, J.; Natu, S.; Kretzer, J.P.; Müller, S.; Thomas, P.; et al. Diagnostic guidelines for the histological particle algorithm in the periprosthetic neo-synovial tissue. BMC Clin. Pathol. 2018, 18, 7. [Google Scholar] [CrossRef]
- Fagotti, L.; Vicente, J.R.; Miyahara, H.S.; de Oliveira, P.V.; Bernabé, A.C.; Croci, A.T. Formation of a pseudotumor in total hip arthroplasty using a tribological metal-polyethylene pair. Rev. Bras. Ortop. 2015, 50, 747–751. [Google Scholar] [CrossRef][Green Version]
- Mao, X.; Tay, G.H.; Godbolt, D.B.; Crawford, R.W. Pseudotumor in a well-fixed metal-on-polyethylene uncemented hip arthroplasty. J. Arthroplast. 2012, 27, 493.e13–493.e4.93E17. [Google Scholar] [CrossRef]
- Snyder, M.J.; Weber, M.A.; Kromka, J.J.; Sims, M.M.; Smith, C.N.; Daji, A.V.; Kumar, D.; Borrero, C.G.; Cordle, A.C.; DiGioia, A.M.; et al. Predictors of Adverse Local Tissue Reaction in a High-Risk Population. Arthroplast. Today 2022, 13, 125–129. [Google Scholar] [CrossRef]
- Taunton, M.J. How to Interpret Metal Ions in THA. J. Arthroplast. 2020, 35 (Suppl. S6), S60–S62. [Google Scholar] [CrossRef]
- Bisseling, P.; Tan, T.; Lu, Z.; Campbell, P.A.; Susante, J.L. The absence of a metal-on-metal bearing does not preclude the formation of a destructive pseudotumor in the hip—A case report. Acta Orthop. 2013, 84, 437–441. [Google Scholar] [CrossRef]
- von Schroeder, H.P.; Smith, D.C.; Gross, A.E.; Pilliar, R.M.; Kandel, R.A.; Chernecky, R.; Lugowski, S.J. Titanemia from total knee arthroplasty. A case resulting from a failed patellar component. J. Arthroplast. 1966, 11, 620–625. [Google Scholar] [CrossRef]
- Nam, D.; Keeney, J.A.; Nunley, R.M.; Johnson, S.R.; Clohisy, J.C.; Barrack, R.L. Metal Ion Concentrations in Young, Active Patients Following Total Hip Arthroplasty with the Use of Modern Bearing Couples. J. Arthroplast. 2015, 30, 2227–2232. [Google Scholar] [CrossRef]
- Jacobs, J.J.; Silverton, C.; Hallab, N.J.; Skipor, A.K.; Patterson, L.; Black, J.; Galante, J.O. Metal release and excretion from cementless titanium alloy total knee replacements. Clin. Orthop. Relat. Res. 1999, 358, 173–180. [Google Scholar] [CrossRef]
- Ude, C.C.; Schmidt, S.J.; Laurencin, S.; Shah, S.; Esdaille, J.; Kan, H.M.; Holt, B.D.; Arnold, A.M.; Wolf, M.E.; Nair, L.S.; et al. Hyaluronic acid-British anti-Lewisite as a safer chelation therapy for the treatment of arthroplasty-related metallosis. Proc. Natl. Acad. Sci. USA 2023, 120, e2309156120. [Google Scholar] [CrossRef]
- Konopitski, A.; Okafor, C.; Smith, B.; Baldwin, K.; Sheth, N.P. Evolution of total hip arthroplasty in patients younger than 30 years of age: A systematic review and meta-analysis. Arch. Orthop. Trauma Surg. 2023, 143, 1081–1094. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, A.V., Jr.; Byrd, Z.O.; Berend, K.R.; Morris, M.J.; Adams, J.B.; Crawford, D.A. Mid-Term Survival of Total Hip Arthroplasty in Patients Younger Than 55-year-old. J. Arthroplast. 2022, 37, S517–S523. [Google Scholar] [CrossRef] [PubMed]
- Vendittoli, P.A.; Shahin, M.; Rivière, C.; Barry, J.; Lavoie, P.; Duval, N. Ceramic-on-ceramic total hip arthroplasty is superior to metal-on-conventional polyethylene at 20-year follow-up: A randomised clinical trial. Orthop. Traumatol. Surg. Res. OTSR 2021, 107, 102744. [Google Scholar] [CrossRef]
- Murali, R.; Bonar, S.F.; Kirsh, G.; Walter, W.K.; Walter, W.L. Osteolysis in third-generation alumina ceramic-on-ceramic hip bearings with severe impingement and titanium metallosis. J. Arthroplast. 2008, 23, 1240.e13–1240.e1.24E19. [Google Scholar] [CrossRef] [PubMed]
- Chappard, D.; Rony, L.; Ducellier, F.; Steiger, V.; Hubert, L. Wear debris released by hip prosthesis analysed by microcomputed tomography. J. Microsc. 2021, 282, 13–20. [Google Scholar] [CrossRef]
- Ude, C.C.; Esdaille, C.J.; Ogueri, K.S.; Ho-Man, K.; Laurencin, S.J.; Nair, L.S.; Laurencin, C.T. The Mechanism of Metallosis After Total Hip Arthroplasty. Regen. Eng. Transl. Med. 2021, 7, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Paiement, G.D.; Penenberg, B.L.; Rajaee, S.S. Metallosis in Total Hip Arthroplasty. JBJS Rev. 2023, 11, e23.00105. [Google Scholar] [CrossRef]



| Investigation | Norm | Unit | At Revision | +6 Months |
|---|---|---|---|---|
| ESR | <15 | mm/h | 5 | 2 |
| CRP | <5 | mg/L | 4 | 6 |
| Leukocytes, total | 4.00–10.00 | 109/L | 6.93 | 6.92 |
| Neutrophils, total | 1.50–7.40 | 109/L | NA | 4.57 |
| Neutrophils, segmented | <0.03 | 109/L | NA | 0.03 |
| Lymphocytes | 1.10–3.50 | 109/L | NA | 1.25 |
| Monocytes | 0.21–0.92 | 109/L | NA | 0.75 |
| Eosinophils | 0.02–0.67 | 109/L | NA | 0.31 |
| Basophils | 0.00–0.13 | 109/L | NA | 0.04 |
| Erythrocytes | 4.50–5.50 | 1012/L | 5.57 | 6.21 |
| Hemoglobin | 130–170 | g/L | 154 | 167 |
| Hematocrit | 0.40–0.50 | - | 0.47 | 0.51 |
| MCV | 83.0–101.0 | fL | 83.8 | 82.4 |
| MCH | 27.0–32.0 | pg | 27.6 | 26.9 |
| MCHC | 315–345 | g/L | 330 | 326 |
| Platelets | 150–410 | 109/L | 545 | 355 |
| Glucose | 3.6–6.1 | mmol/L | 6.0 | 5.9 |
| Urea nitrogen | 2.8–7.5 | mmol/L | 2.6 | 2.1 |
| Creatinine | 64–104 | μmol/L | 46 | 56 |
| GFR | 80–120 | mL/min/1.73m2 | >90 | >90 |
| Bilirubin—total | <17 | μmol/L | 5 | 12 |
| Bilirubin—direct | <5 | μmol/L | 2 | 3 |
| AST | <0.58 | μkat/L | 0.77 | 0.54 |
| ALT | <0.74 | μkat/L | 0.49 | 0.47 |
| γ-GT | <0.92 | μkat/L | 0.85 | 1.05 |
| Sodium | 135–145 | μmol/L | 136 | 136 |
| Potassium | 3.5–5.3 | μmol/L | 4.91 | 4.24 |
| Chlorides | 97–110 | μmol/L | 103 | 106 |
| Serum Trace Element | At Revision | +4 Months | +6 Months |
|---|---|---|---|
| Titanium * | 891.4 | 1237.6 | 879.1 |
| Aluminum * | 11 | NA | 15 |
| Vanadium * | 6.33 | NA | 5.38 |
| Cobalt * | <0.07 | NA | 0.1 |
| Chromium * | <0.257 | NA | <0.257 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fokter, S.K.; Ledinek, Ž.; Kljaić Dujić, M.; Novak, I. Extreme Serum Titanium Concentration Induced by Acetabular Cup Failure: Unveiling a Unique Scenario of Titanium Alloy Debris Accumulation. Bioengineering 2024, 11, 235. https://doi.org/10.3390/bioengineering11030235
Fokter SK, Ledinek Ž, Kljaić Dujić M, Novak I. Extreme Serum Titanium Concentration Induced by Acetabular Cup Failure: Unveiling a Unique Scenario of Titanium Alloy Debris Accumulation. Bioengineering. 2024; 11(3):235. https://doi.org/10.3390/bioengineering11030235
Chicago/Turabian StyleFokter, Samo K., Živa Ledinek, Milka Kljaić Dujić, and Igor Novak. 2024. "Extreme Serum Titanium Concentration Induced by Acetabular Cup Failure: Unveiling a Unique Scenario of Titanium Alloy Debris Accumulation" Bioengineering 11, no. 3: 235. https://doi.org/10.3390/bioengineering11030235
APA StyleFokter, S. K., Ledinek, Ž., Kljaić Dujić, M., & Novak, I. (2024). Extreme Serum Titanium Concentration Induced by Acetabular Cup Failure: Unveiling a Unique Scenario of Titanium Alloy Debris Accumulation. Bioengineering, 11(3), 235. https://doi.org/10.3390/bioengineering11030235








