Gaining New Insights into Fundamental Biological Pathways by Bacterial Toxin-Based Genetic Screens
Abstract
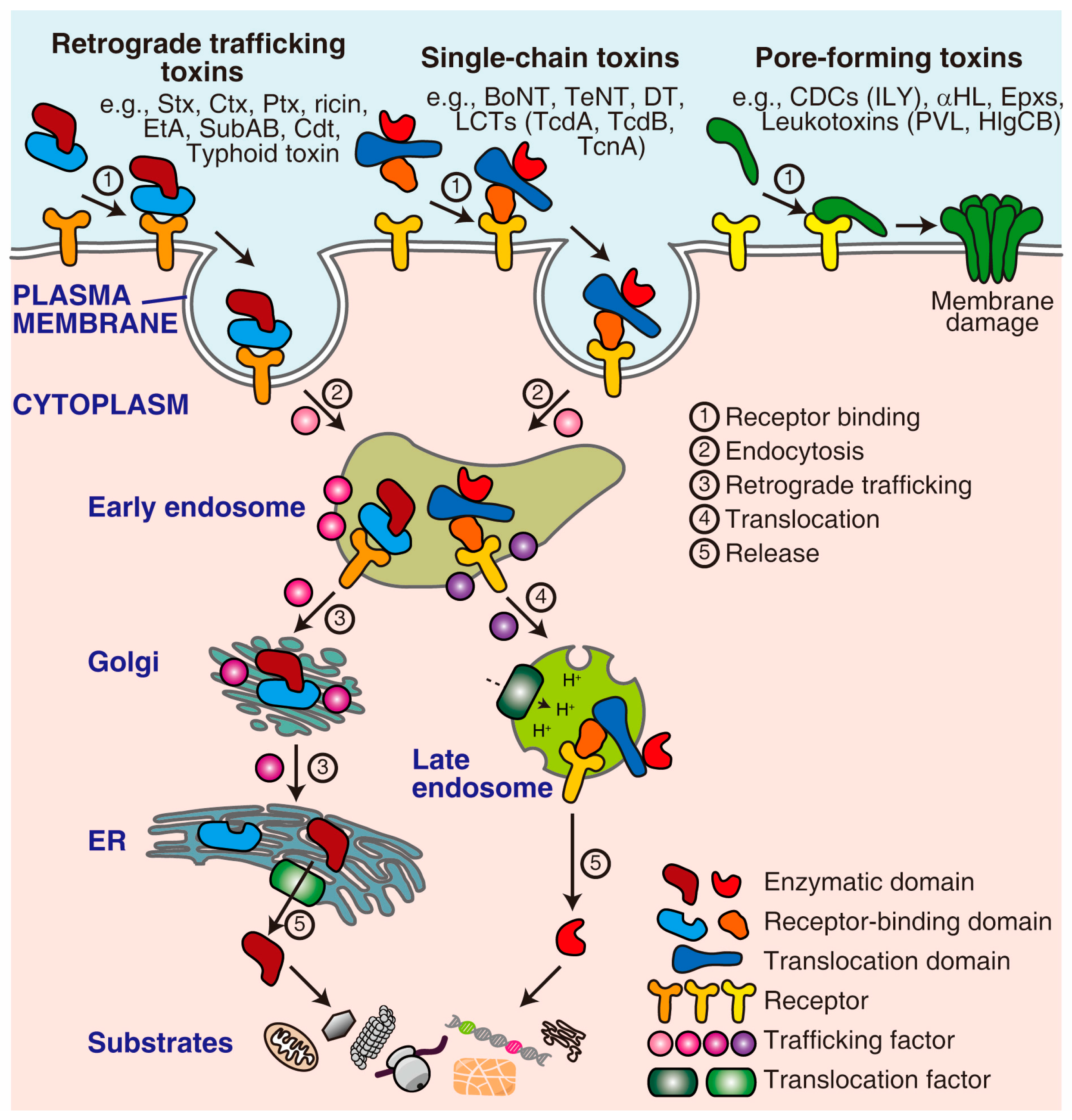
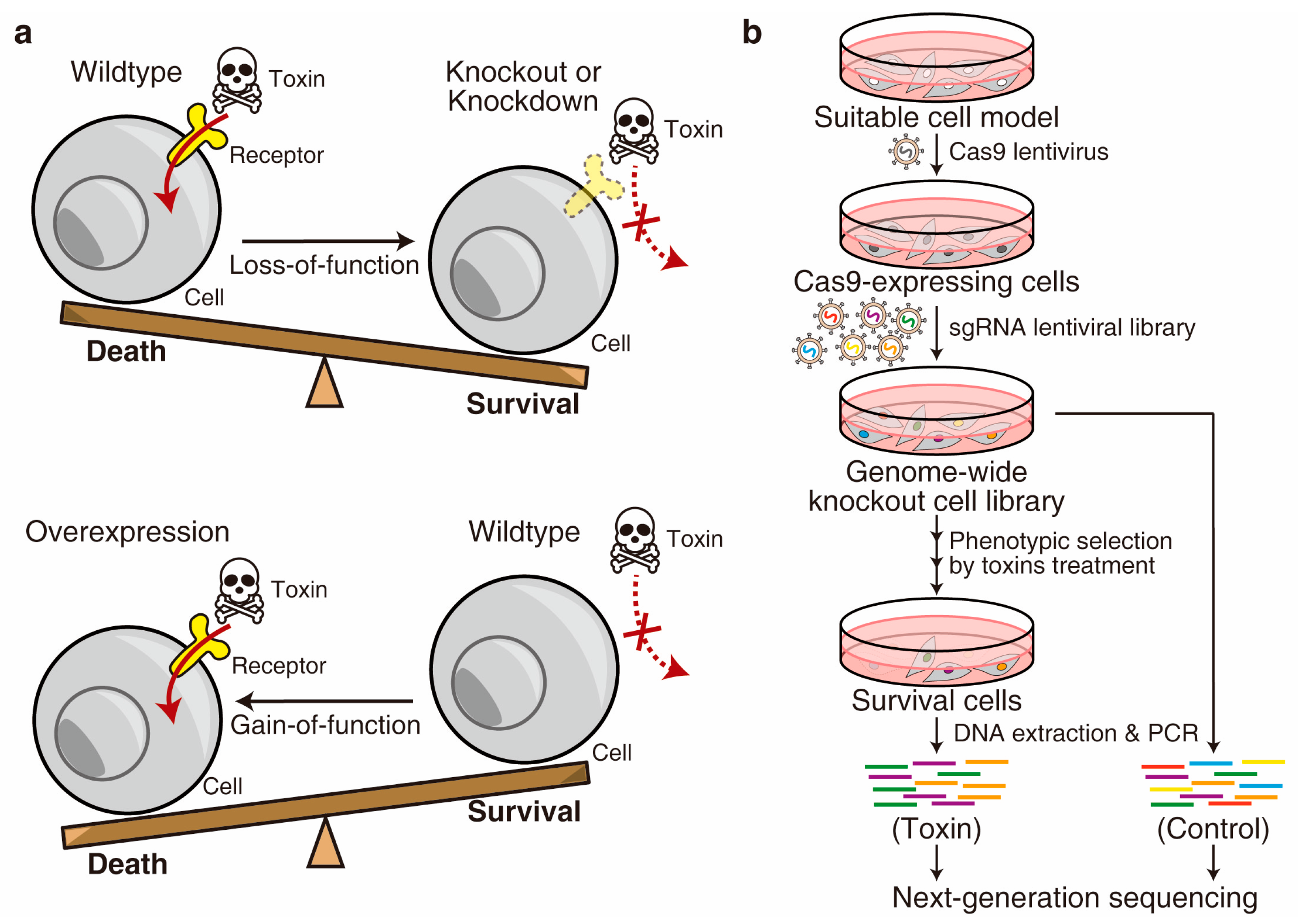
1. Introduction
2. Factors Required for the Biosynthesis of Glycosphingolipids
2.1. The Biology of Stxs
2.2. Genetic Screens for Stxs
2.3. Ctx and the Related Screens
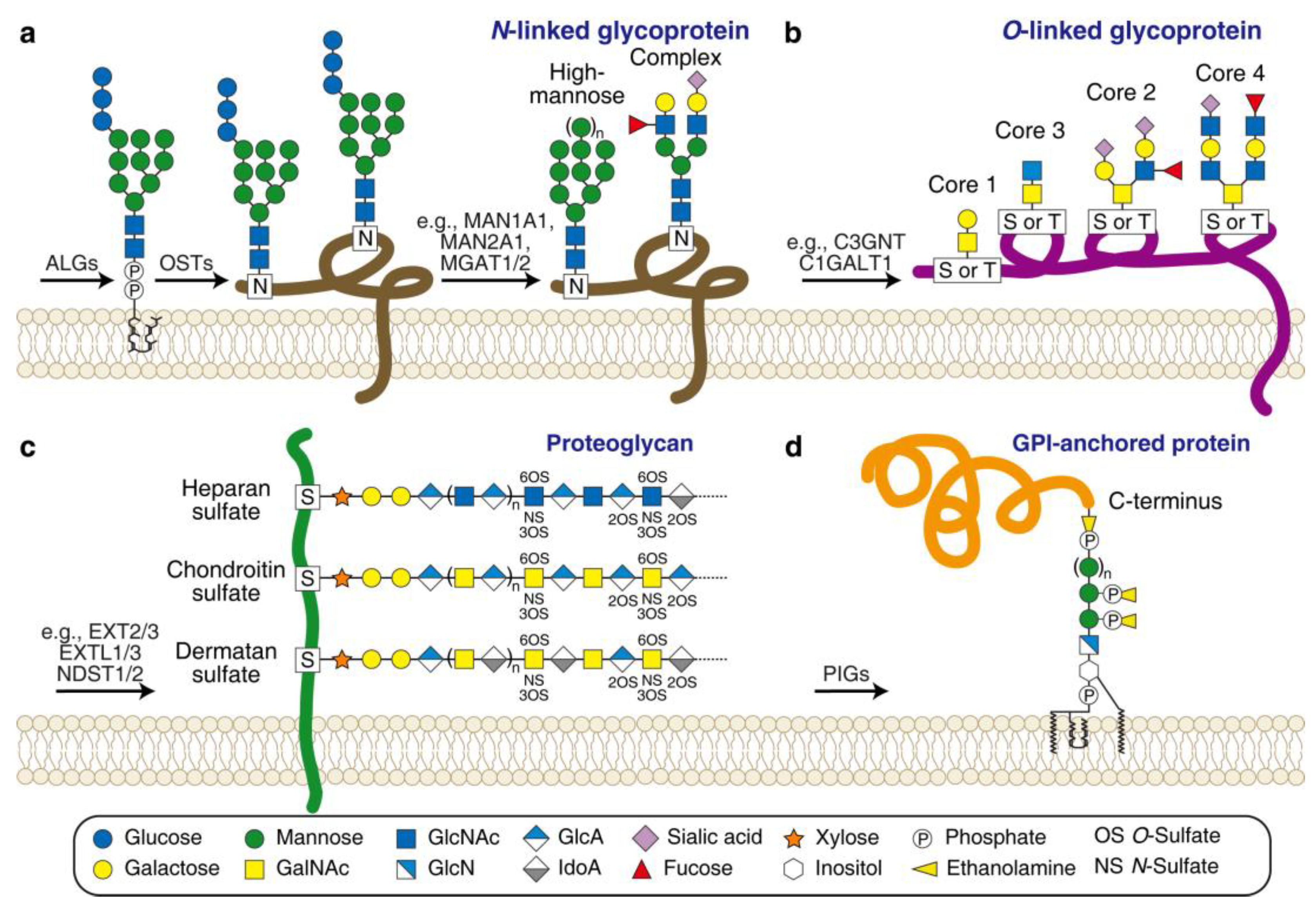
3. Factors Involved in Protein Glycosylation
3.1. Ricin and Related Screens for N-Linked Glycosylation
3.2. TcdA and Related Screens for Biosynthesis of sGAGs
3.3. TcdB and Related Screens for Biosynthesis of GPI Anchor
3.4. Other Toxin-Based Screens for Glycosylation Pathways
4. Factors Involved in Membrane Vesicle Trafficking
4.1. Screens Using Ricin as a Probe
4.2. Screens Using Stx as a Probe
4.3. Screens Using EtA as a Probe
4.4. Screens Using Other Toxins as Probes
5. Factors Involved in Unique Pathways
6. Summary
| Screens | Toxins | Screen Strategy * | Identified Host Factors and Functions |
|---|---|---|---|
| Reported by Tian et al. [31] | Stx1 Stx2 ricin |
|
|
| Reported by Yamaji et al. [68] | Stx1 |
|
|
| Reported by Sakuma et al. [69] | Stx1 |
|
|
| Reported by Majumder et al. [70] | Stx |
|
|
| Reported by Pacheco et al. [71] | EHEC *** |
|
|
| Reported by Kono et al. [72] | Stx |
|
|
| Reported by Selyunin et al. [148] | Stx1 Stx2 |
|
|
| Reported by Guimaraes et al. [83] | Ctx-DTA |
|
|
| Reported by Gilbert et al. [26] | Ctx-DTA |
|
|
| Reported by Morgens et al. [97] | ricin |
|
|
| Reported by Bassik et al. [98] | ricin |
|
|
| Reported by Tafesse et al. [152] | EtA |
|
|
| Reported by Moreau et al. [99] | Ricin EtA |
|
|
| Reported by Yamaji et al. [132] | SubAB |
|
|
| Reported by Chang et al. [153] | typhoid toxin |
|
|
| Reported by Anwar et al. [155] | Anthrax toxin |
|
|
| Reported by Carette et al. [18] | DT LFN-DTA |
|
|
| Reported by Tao et al. [113,154] | TcdB |
|
|
| Reported by Tao et al. [33] | TcdA |
|
|
| Reported by Zhou et al. [117] | TcnA |
|
|
| Reported by Tian et al. [35] | TcdB4 |
|
|
| Reported by Luo et al. [114] | TcdB4 |
|
|
| Reported by Tromp et al. [134] | PVL HlgCB |
|
|
| Reported by Jeon et al. [156] | PVL |
|
|
| Reported by Drabavicius et al. [138] | ILY |
|
|
| Reported by Shahi et al. [141] | ILY |
|
|
| Reported by Virreira Winter et al. [149] | αHL |
|
|
| Reported by Song et al. [144] | Tc toxins |
|
|
7. Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Stx | Shiga toxin |
| Ctx | cholera toxin |
| DT | diphtheria toxin |
| DTA | enzymatic domain of diphtheria toxin |
| PA | anthrax protective antigen |
| LFN | anthrax lethal factor N-terminal |
| EtA | Pseudomonas aeruginosa exotoxin A |
| LCT | large clostridial toxin |
| TcdA | Clostridioides difficile toxin A |
| TcdB | Clostridioides difficile toxin B |
| TcnA | Clostridium novyi alpha-toxin |
| CROPs | combined repetitive oligopeptides |
| SubAB | Escherichia coli subtilase cytotoxin |
| CDC | cholesterol-dependent cytolysin |
| Tc toxin | insecticidal toxin complex |
| αHL | Staphylococcus aureus α-hemolysin |
| PVL | Staphylococcus aureus Panton-Valentine leucocidin |
| HlgCB | Staphylococcus aureus γ-haemolysin CB |
| ILY | Streptococcus intermedius intermedilysin |
| Cdt | cytolethal distending toxin |
| T3SS | type III secretion system |
| LPS | lipopolysaccharides |
| EHEC | enterohemorrhagic Escherichia coli |
| RNAi | RNA interference |
| CRISPR | clustered regularly interspaced short palindromic repeat |
| CRISPRi | CRISPR repression |
| CRISPRa | CRISPR activation |
| sgRNA | single guide RNA |
| shRNA | short hairpin RNA |
| siRNA | small interfering RNA |
| cDNA | complementary DNA |
| cAMP | 3′,5′-cyclic AMP |
| GalNAc | N-acetylgalactosamine |
| GlcNAc | N-acetylglucosamine |
| GSL | glycosphingolipid |
| GlcCer | glucosylceramide |
| LacCer | lactosylceramide |
| Gb3 | globotriaosylceramide (also known as CD77) |
| GM1 | monosialotetrahexosylganglioside |
| S1P | sphingosine-1-phosphate |
| GPI | glycosylphosphatidylinositol |
| GAG | glycosaminoglycan |
| sGAG | sulfated glycosaminoglycan |
| ER | endoplasmic reticulum |
| TGN | trans-Golgi network |
| ERAD | ER-associated protein degradation |
| GPCR | G-protein coupled receptor |
| COPI | coat protein complex I |
| COPII | coat protein complex II |
| TRAPP | transport protein particle |
| GARP | Golgi-associated retrograde protein |
| COG | conserved oligomeric Golgi complex |
| EMC | ER membrane protein complex |
| UGCG | UDP-glucose ceramide glucosyltransferase |
| B4GALT5 | β-1,4-galactosyltransferase 5 |
| SLC35A2 | solute carrier family 35 member A2 |
| A4GALT | α-1,4-galactosyltransferase (also known as Gb3 synthetase) |
| SPTSSA | serine palmitoyltransferase small subunit A |
| SPTLC1 | serine palmitoyltransferase long chain base subunit 1 |
| UGP2 | UDP-glucose pyrophosphorylase 2 |
| SPPL3 | Signal peptide peptidase-like 3 |
| LAPTM4A | lysosomal-associated protein transmembrane 4 A |
| LAPTM4B | lysosomal-associated protein transmembrane 4 B |
| TMEM165 | transmembrane protein 165 |
| TM9SF2 | transmembrane 9 superfamily member 2 |
| AHR | aryl hydrocarbon receptor |
| PLPP3 | phospholipid phosphatase 3 (also known as PPAP2B) |
| SGPP1 | S1P phosphatase 1 |
| CFTR | cystic fibrosis transmembrane conductance regulator |
| ST3GAL5 | ST3 β-galactoside α-2,3-sialyltransferase 5 |
| B4GALNT1 | β-1,4-N-acetylgalactosaminyltransferase 1 |
| B3GALT4 | β-1,3-galactosyltransferase 4 |
| eEF-2 | eukaryotic elongation factor 2 |
| CSPG4 | chondroitin sulfate proteoglycan 4 |
| FZDs | frizzled receptors |
| SLC39A9 | solute carrier family 39 member A9 |
| GBF1 | Golgi Brefeldin A resistant guanine nucleotide exchange factor 1 |
| ADAM10 | ADAM metalloproteinase domain 10 |
| KDELR1 | KDEL receptors 1 |
| KDELR2 | KDEL receptors 2 |
| JTB | jumping translocation breakpoint |
| TMED2 | transmembrane p24 trafficking protein 2 |
| FBXO11 | F-box protein 11 |
| D+HUS | post-diarrheal hemolytic uremic syndrome |
| CDG | congenital disorders of glycosylation |
| HPMRS | hyperphosphatasia with mental retardation syndrome |
| PNH | paroxysmal nocturnal hemoglobinuria |
References
- Ala’Aldeen, D.A.A.; Wooldridge, K.G. Bacterial pathogenicity. In Medical Microbiology; Elsevier: Amsterdam, The Netherlands, 2012; pp. 156–167. [Google Scholar] [CrossRef]
- Barbieri, J.T. Exotoxins. In Encyclopedia of Microbiology; Elsevier: Amsterdam, The Netherlands, 2009; pp. 355–364. [Google Scholar] [CrossRef]
- Orrell, K.E.; Melnyk, R.A. Large Clostridial Toxins: Mechanisms and Roles in Disease. Microbiol. Mol. Biol. Rev. 2021, 85, e0006421. [Google Scholar] [CrossRef]
- Dal Peraro, M.; van der Goot, F.G. Pore-forming toxins: Ancient, but never really out of fashion. Nat. Rev. Microbiol. 2016, 14, 77–92. [Google Scholar] [CrossRef]
- Odumosu, O.; Nicholas, D.; Yano, H.; Langridge, W. AB toxins: A paradigm switch from deadly to desirable. Toxins 2010, 2, 1612–1645. [Google Scholar] [CrossRef] [PubMed]
- Beddoe, T.; Paton, A.W.; Le Nours, J.; Rossjohn, J.; Paton, J.C. Structure, biological functions and applications of the AB5 toxins. Trends Biochem. Sci. 2010, 35, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Pons, B.J.; Vignard, J.; Mirey, G. Cytolethal Distending Toxin Subunit B: A Review of Structure-Function Relationship. Toxins 2019, 11, 595. [Google Scholar] [CrossRef] [PubMed]
- Fowler, C.C.; Chang, S.J.; Gao, X.; Geiger, T.; Stack, G.; Galan, J.E. Emerging insights into the biology of typhoid toxin. Curr. Opin. Microbiol. 2017, 35, 70–77. [Google Scholar] [CrossRef]
- Dong, M.; Masuyer, G.; Stenmark, P. Botulinum and Tetanus Neurotoxins. Annu. Rev. Biochem. 2019, 88, 811–837. [Google Scholar] [CrossRef]
- Moresco, E.M.; Li, X.; Beutler, B. Going forward with genetics: Recent technological advances and forward genetics in mice. Am. J. Pathol. 2013, 182, 1462–1473. [Google Scholar] [CrossRef]
- Grimm, S. The art and design of genetic screens: Mammalian culture cells. Nat. Rev. Genet. 2004, 5, 179–189. [Google Scholar] [CrossRef]
- Strachan, T.; Read, A.P.; Strachan, T. Human Molecular Genetics, 2nd ed.; BIOS Scientific Publishers Ltd.: Oxford, UK, 1999; p. xxiii. 576p. [Google Scholar]
- Collaboration, O.R. The ORFeome Collaboration: A genome-scale human ORF-clone resource. Nat. Methods 2016, 13, 191–192. [Google Scholar] [CrossRef]
- Yang, X.; Boehm, J.S.; Yang, X.; Salehi-Ashtiani, K.; Hao, T.; Shen, Y.; Lubonja, R.; Thomas, S.R.; Alkan, O.; Bhimdi, T.; et al. A public genome-scale lentiviral expression library of human ORFs. Nat. Methods 2011, 8, 659–661. [Google Scholar] [CrossRef] [PubMed]
- Naglich, J.G.; Metherall, J.E.; Russell, D.W.; Eidels, L. Expression cloning of a diphtheria toxin receptor: Identity with a heparin-binding EGF-like growth factor precursor. Cell 1992, 69, 1051–1061. [Google Scholar] [CrossRef] [PubMed]
- Berns, K.; Hijmans, E.M.; Mullenders, J.; Brummelkamp, T.R.; Velds, A.; Heimerikx, M.; Kerkhoven, R.M.; Madiredjo, M.; Nijkamp, W.; Weigelt, B.; et al. A large-scale RNAi screen in human cells identifies new components of the p53 pathway. Nature 2004, 428, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Paddison, P.J.; Silva, J.M.; Conklin, D.S.; Schlabach, M.; Li, M.; Aruleba, S.; Balija, V.; O’Shaughnessy, A.; Gnoj, L.; Scobie, K.; et al. A resource for large-scale RNA-interference-based screens in mammals. Nature 2004, 428, 427–431. [Google Scholar] [CrossRef]
- Carette, J.E.; Guimaraes, C.P.; Varadarajan, M.; Park, A.S.; Wuethrich, I.; Godarova, A.; Kotecki, M.; Cochran, B.H.; Spooner, E.; Ploegh, H.L.; et al. Haploid genetic screens in human cells identify host factors used by pathogens. Science 2009, 326, 1231–1235. [Google Scholar] [CrossRef]
- Carette, J.E.; Guimaraes, C.P.; Wuethrich, I.; Blomen, V.A.; Varadarajan, M.; Sun, C.; Bell, G.; Yuan, B.; Muellner, M.K.; Nijman, S.M.; et al. Global gene disruption in human cells to assign genes to phenotypes by deep sequencing. Nat. Biotechnol. 2011, 29, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Shalem, O.; Sanjana, N.E.; Hartenian, E.; Shi, X.; Scott, D.A.; Mikkelson, T.; Heckl, D.; Ebert, B.L.; Root, D.E.; Doench, J.G.; et al. Genome-scale CRISPR-Cas9 knockout screening in human cells. Science 2014, 343, 84–87. [Google Scholar] [CrossRef]
- Wang, T.; Wei, J.J.; Sabatini, D.M.; Lander, E.S. Genetic screens in human cells using the CRISPR-Cas9 system. Science 2014, 343, 80–84. [Google Scholar] [CrossRef]
- Shalem, O.; Sanjana, N.E.; Zhang, F. High-throughput functional genomics using CRISPR-Cas9. Nat. Rev. Genet. 2015, 16, 299–311. [Google Scholar] [CrossRef]
- Puschnik, A.S.; Majzoub, K.; Ooi, Y.S.; Carette, J.E. A CRISPR toolbox to study virus-host interactions. Nat. Rev. Microbiol. 2017, 15, 351–364. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, L.A.; Larson, M.H.; Morsut, L.; Liu, Z.; Brar, G.A.; Torres, S.E.; Stern-Ginossar, N.; Brandman, O.; Whitehead, E.H.; Doudna, J.A.; et al. CRISPR-mediated modular RNA-guided regulation of transcription in eukaryotes. Cell 2013, 154, 442–451. [Google Scholar] [CrossRef]
- Qi, L.S.; Larson, M.H.; Gilbert, L.A.; Doudna, J.A.; Weissman, J.S.; Arkin, A.P.; Lim, W.A. Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression. Cell 2013, 152, 1173–1183. [Google Scholar] [CrossRef]
- Gilbert, L.A.; Horlbeck, M.A.; Adamson, B.; Villalta, J.E.; Chen, Y.; Whitehead, E.H.; Guimaraes, C.; Panning, B.; Ploegh, H.L.; Bassik, M.C.; et al. Genome-Scale CRISPR-Mediated Control of Gene Repression and Activation. Cell 2014, 159, 647–661. [Google Scholar] [CrossRef]
- Boutros, M.; Ahringer, J. The art and design of genetic screens: RNA interference. Nat. Rev. Genet. 2008, 9, 554–566. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, J.; Moreira, E.; Sequeira, I.J.; Rodrigues, A.S.; Rueff, J.; Bras, A. Integration of HIV in the Human Genome: Which Sites Are Preferential? A Genetic and Statistical Assessment. Int. J. Genom. 2016, 2016, 2168590. [Google Scholar] [CrossRef] [PubMed]
- Konermann, S.; Brigham, M.D.; Trevino, A.E.; Joung, J.; Abudayyeh, O.O.; Barcena, C.; Hsu, P.D.; Habib, N.; Gootenberg, J.S.; Nishimasu, H.; et al. Genome-scale transcriptional activation by an engineered CRISPR-Cas9 complex. Nature 2015, 517, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Foden, J.A.; Khayter, C.; Maeder, M.L.; Reyon, D.; Joung, J.K.; Sander, J.D. High-frequency off-target mutagenesis induced by CRISPR-Cas nucleases in human cells. Nat. Biotechnol. 2013, 31, 822–826. [Google Scholar] [CrossRef]
- Tian, S.; Muneeruddin, K.; Choi, M.Y.; Tao, L.; Bhuiyan, R.H.; Ohmi, Y.; Furukawa, K.; Furukawa, K.; Boland, S.; Shaffer, S.A.; et al. Genome-wide CRISPR screens for Shiga toxins and ricin reveal Golgi proteins critical for glycosylation. PLoS Biol. 2018, 16, e2006951. [Google Scholar] [CrossRef]
- Tian, S.; Liu, Y.; Wu, H.; Liu, H.; Zeng, J.; Choi, M.Y.; Chen, H.; Gerhard, R.; Dong, M. Genome-Wide CRISPR Screen Identifies Semaphorin 6A and 6B as Receptors for Paeniclostridium sordellii Toxin TcsL. Cell Host Microbe 2020, 27, 782–792.e7. [Google Scholar] [CrossRef]
- Tao, L.; Tian, S.; Zhang, J.; Liu, Z.; Robinson-McCarthy, L.; Miyashita, S.I.; Breault, D.T.; Gerhard, R.; Oottamasathien, S.; Whelan, S.P.J.; et al. Sulfated glycosaminoglycans and low-density lipoprotein receptor contribute to Clostridium difficile toxin A entry into cells. Nat. Microbiol. 2019, 4, 1760–1769. [Google Scholar] [CrossRef]
- Xiong, X.; Tian, S.; Yang, P.; Lebreton, F.; Bao, H.; Sheng, K.; Yin, L.; Chen, P.; Zhang, J.; Qi, W.; et al. Emerging enterococcus pore-forming toxins with MHC/HLA-I as receptors. Cell 2022, 185, 1157–1171.e22. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Xiong, X.; Zeng, J.; Wang, S.; Tremblay, B.J.; Chen, P.; Chen, B.; Liu, M.; Chen, P.; Sheng, K.; et al. Identification of TFPI as a receptor reveals recombination-driven receptor switching in Clostridioides difficile toxin B variants. Nat. Commun. 2022, 13, 6786. [Google Scholar] [CrossRef]
- Holthuis, J.C.; Menon, A.K. Lipid landscapes and pipelines in membrane homeostasis. Nature 2014, 510, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Yamaji, T.; Hanada, K. Sphingolipid metabolism and interorganellar transport: Localization of sphingolipid enzymes and lipid transfer proteins. Traffic 2015, 16, 101–122. [Google Scholar] [CrossRef] [PubMed]
- Kolter, T.; Sandhoff, K. Sphingolipid metabolism diseases. Biochim. Biophys. Acta 2006, 1758, 2057–2079. [Google Scholar] [CrossRef] [PubMed]
- Green, C.D.; Maceyka, M.; Cowart, L.A.; Spiegel, S. Sphingolipids in metabolic disease: The good, the bad, and the unknown. Cell Metab. 2021, 33, 1293–1306. [Google Scholar] [CrossRef]
- Chester, M.A. IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN). Nomenclature of glycolipids—Recommendations 1997. Eur. J. Biochem. 1998, 257, 293–298. [Google Scholar] [CrossRef]
- Qasba, P.K.; Ramakrishnan, B. Letter to the Glyco-Forum: Catalytic domains of glycosyltransferases with ‘add-on’ domains. Glycobiology 2007, 17, 7G–9G. [Google Scholar] [CrossRef]
- Kitatani, K.; Idkowiak-Baldys, J.; Hannun, Y.A. The sphingolipid salvage pathway in ceramide metabolism and signaling. Cell. Signal. 2008, 20, 1010–1018. [Google Scholar] [CrossRef]
- Merrill, A.H., Jr. Sphingolipid and glycosphingolipid metabolic pathways in the era of sphingolipidomics. Chem. Rev. 2011, 111, 6387–6422. [Google Scholar] [CrossRef]
- Geny, B.; Popoff, M.R. Bacterial protein toxins and lipids: Role in toxin targeting and activity. Biol. Cell 2006, 98, 633–651. [Google Scholar] [CrossRef] [PubMed]
- Johannes, L.; Romer, W. Shiga toxins—From cell biology to biomedical applications. Nat. Rev. Microbiol. 2010, 8, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Bergan, J.; Dyve Lingelem, A.B.; Simm, R.; Skotland, T.; Sandvig, K. Shiga toxins. Toxicon 2012, 60, 1085–1107. [Google Scholar] [CrossRef]
- Lingwood, C.A. Verotoxins and their glycolipid receptors. Adv. Lipid Res. 1993, 25, 189–211. [Google Scholar] [PubMed]
- Ling, H.; Boodhoo, A.; Hazes, B.; Cummings, M.D.; Armstrong, G.D.; Brunton, J.L.; Read, R.J. Structure of the shiga-like toxin I B-pentamer complexed with an analogue of its receptor Gb3. Biochemistry 1998, 37, 1777–1788. [Google Scholar] [CrossRef] [PubMed]
- Kojima, Y.; Fukumoto, S.; Furukawa, K.; Okajima, T.; Wiels, J.; Yokoyama, K.; Suzuki, Y.; Urano, T.; Ohta, M.; Furukawa, K. Molecular cloning of globotriaosylceramide/CD77 synthase, a glycosyltransferase that initiates the synthesis of globo series glycosphingolipids. J. Biol. Chem. 2000, 275, 15152–15156. [Google Scholar] [CrossRef]
- Keusch, J.J.; Manzella, S.M.; Nyame, K.A.; Cummings, R.D.; Baenziger, J.U. Cloning of Gb3 synthase, the key enzyme in globo-series glycosphingolipid synthesis, predicts a family of alpha 1, 4-glycosyltransferases conserved in plants, insects, and mammals. J. Biol. Chem. 2000, 275, 25315–25321. [Google Scholar] [CrossRef]
- Okuda, T.; Tokuda, N.; Numata, S.; Ito, M.; Ohta, M.; Kawamura, K.; Wiels, J.; Urano, T.; Tajima, O.; Furukawa, K.; et al. Targeted disruption of Gb3/CD77 synthase gene resulted in the complete deletion of globo-series glycosphingolipids and loss of sensitivity to verotoxins. J. Biol. Chem. 2006, 281, 10230–10235. [Google Scholar] [CrossRef]
- Kannagi, R.; Fukuda, M.N.; Hakomori, S. A new glycolipid antigen isolated from human erythrocyte membranes reacting with antibodies directed to globo-N-tetraosylceramide (globoside). J. Biol. Chem. 1982, 257, 4438–4442. [Google Scholar] [CrossRef]
- Gregory, C.D.; Dive, C.; Henderson, S.; Smith, C.A.; Williams, G.T.; Gordon, J.; Rickinson, A.B. Activation of Epstein-Barr virus latent genes protects human B cells from death by apoptosis. Nature 1991, 349, 612–614. [Google Scholar] [CrossRef]
- Liu, Y.; Tian, S.; Thaker, H.; Dong, M. Shiga Toxins: An Update on Host Factors and Biomedical Applications. Toxins 2021, 13, 222. [Google Scholar] [CrossRef] [PubMed]
- Obrig, T.G.; Karpman, D. Shiga toxin pathogenesis: Kidney complications and renal failure. Curr. Top. Microbiol. Immunol. 2012, 357, 105–136. [Google Scholar] [CrossRef]
- Masson, C.; Cisse, I.; Simon, V.; Insalaco, P.; Audran, M. Fabry disease: A review. Joint Bone Spine 2004, 71, 381–383. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, H.M. Fabry’s disease—A comprehensive review on pathogenesis, diagnosis and treatment. J. Pak. Med. Assoc. 2014, 64, 189–194. [Google Scholar] [PubMed]
- Zarate, Y.A.; Hopkin, R.J. Fabry’s disease. Lancet 2008, 372, 1427–1435. [Google Scholar] [CrossRef] [PubMed]
- Voss, M.; Kunzel, U.; Higel, F.; Kuhn, P.H.; Colombo, A.; Fukumori, A.; Haug-Kroper, M.; Klier, B.; Grammer, G.; Seidl, A.; et al. Shedding of glycan-modifying enzymes by signal peptide peptidase-like 3 (SPPL3) regulates cellular N-glycosylation. EMBO J. 2014, 33, 2890–2905. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, P.H.; Voss, M.; Haug-Kroper, M.; Schroder, B.; Schepers, U.; Brase, S.; Haass, C.; Lichtenthaler, S.F.; Fluhrer, R. Secretome analysis identifies novel signal Peptide peptidase-like 3 (Sppl3) substrates and reveals a role of Sppl3 in multiple Golgi glycosylation pathways. Mol. Cell. Proteom. 2015, 14, 1584–1598. [Google Scholar] [CrossRef]
- Hogue, D.L.; Ellison, M.J.; Young, J.D.; Cass, C.E. Identification of a novel membrane transporter associated with intracellular membranes by phenotypic complementation in the yeast Saccharomyces cerevisiae. J. Biol. Chem. 1996, 271, 9801–9808. [Google Scholar] [CrossRef]
- Cabrita, M.A.; Hobman, T.C.; Hogue, D.L.; King, K.M.; Cass, C.E. Mouse transporter protein, a membrane protein that regulates cellular multidrug resistance, is localized to lysosomes. Cancer Res. 1999, 59, 4890–4897. [Google Scholar]
- Potelle, S.; Morelle, W.; Dulary, E.; Duvet, S.; Vicogne, D.; Spriet, C.; Krzewinski-Recchi, M.A.; Morsomme, P.; Jaeken, J.; Matthijs, G.; et al. Glycosylation abnormalities in Gdt1p/TMEM165 deficient cells result from a defect in Golgi manganese homeostasis. Hum. Mol. Genet. 2016, 25, 1489–1500. [Google Scholar] [CrossRef]
- Foulquier, F.; Amyere, M.; Jaeken, J.; Zeevaert, R.; Schollen, E.; Race, V.; Bammens, R.; Morelle, W.; Rosnoblet, C.; Legrand, D.; et al. TMEM165 deficiency causes a congenital disorder of glycosylation. Am. J. Hum. Genet. 2012, 91, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Dulary, E.; Potelle, S.; Legrand, D.; Foulquier, F. TMEM165 deficiencies in Congenital Disorders of Glycosylation type II (CDG-II): Clues and evidences for roles of the protein in Golgi functions and ion homeostasis. Tissue Cell 2017, 49, 150–156. [Google Scholar] [CrossRef]
- Schimmoller, F.; Diaz, E.; Muhlbauer, B.; Pfeffer, S.R. Characterization of a 76 kDa endosomal, multispanning membrane protein that is highly conserved throughout evolution. Gene 1998, 216, 311–318. [Google Scholar] [CrossRef]
- Tanaka, A.; Tumkosit, U.; Nakamura, S.; Motooka, D.; Kishishita, N.; Priengprom, T.; Sa-Ngasang, A.; Kinoshita, T.; Takeda, N.; Maeda, Y. Genome-Wide Screening Uncovers the Significance of N-Sulfation of Heparan Sulfate as a Host Cell Factor for Chikungunya Virus Infection. J. Virol. 2017, 91, e00432-17. [Google Scholar] [CrossRef] [PubMed]
- Yamaji, T.; Sekizuka, T.; Tachida, Y.; Sakuma, C.; Morimoto, K.; Kuroda, M.; Hanada, K. A CRISPR Screen Identifies LAPTM4A and TM9SF Proteins as Glycolipid-Regulating Factors. iScience 2019, 11, 409–424. [Google Scholar] [CrossRef]
- Sakuma, C.; Sekizuka, T.; Kuroda, M.; Hanada, K.; Yamaji, T. Identification of SYS1 as a Host Factor Required for Shiga Toxin-Mediated Cytotoxicity in Vero Cells. Int. J. Mol. Sci. 2021, 22, 4936. [Google Scholar] [CrossRef]
- Majumder, S.; Kono, M.; Lee, Y.T.; Byrnes, C.; Li, C.; Tuymetova, G.; Proia, R.L. A genome-wide CRISPR/Cas9 screen reveals that the aryl hydrocarbon receptor stimulates sphingolipid levels. J. Biol. Chem. 2020, 295, 4341–4349. [Google Scholar] [CrossRef]
- Pacheco, A.R.; Lazarus, J.E.; Sit, B.; Schmieder, S.; Lencer, W.I.; Blondel, C.J.; Doench, J.G.; Davis, B.M.; Waldor, M.K. CRISPR Screen Reveals that EHEC’s T3SS and Shiga Toxin Rely on Shared Host Factors for Infection. mBio 2018, 9, e01003-18. [Google Scholar] [CrossRef]
- Kono, M.; Hoachlander-Hobby, L.E.; Majumder, S.; Schwartz, R.; Byrnes, C.; Zhu, H.; Proia, R.L. Identification of two lipid phosphatases that regulate sphingosine-1-phosphate cellular uptake and recycling. J. Lipid Res. 2022, 63, 100225. [Google Scholar] [CrossRef] [PubMed]
- De, S.N.; Sarkar, J.K.; Tribedi, B.P. An experimental study of the action of cholera toxin. J. Pathol. Bacteriol. 1951, 63, 707–717. [Google Scholar] [CrossRef]
- Lencer, W.I.; Tsai, B. The intracellular voyage of cholera toxin: Going retro. Trends Biochem. Sci. 2003, 28, 639–645. [Google Scholar] [CrossRef]
- Schafer, D.E.; Lust, W.D.; Sircar, B.; Goldberg, N.D. Elevated concentration of adenosine 3′:5′-cyclic monophosphate in intestinal mucosa after treatment with cholera toxin. Proc. Natl. Acad. Sci. USA 1970, 67, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Kassis, S.; Hagmann, J.; Fishman, P.H.; Chang, P.P.; Moss, J. Mechanism of action of cholera toxin on intact cells. Generation of A1 peptide and activation of adenylate cyclase. J. Biol. Chem. 1982, 257, 12148–12152. [Google Scholar] [CrossRef] [PubMed]
- Kahn, R.A.; Gilman, A.G. ADP-ribosylation of Gs promotes the dissociation of its alpha and beta subunits. J. Biol. Chem. 1984, 259, 6235–6240. [Google Scholar] [CrossRef]
- Bharati, K.; Ganguly, N.K. Cholera toxin: A paradigm of a multifunctional protein. Indian J. Med. Res. 2011, 133, 179–187. [Google Scholar]
- Heyningen, S.V. Cholera toxin: Interaction of subunits with ganglioside GM1. Science 1974, 183, 656–657. [Google Scholar] [CrossRef] [PubMed]
- Merritt, E.A.; Sarfaty, S.; van den Akker, F.; L’Hoir, C.; Martial, J.A.; Hol, W.G. Crystal structure of cholera toxin B-pentamer bound to receptor GM1 pentasaccharide. Protein Sci. 1994, 3, 166–175. [Google Scholar] [CrossRef]
- Kenworthy, A.K.; Schmieder, S.S.; Raghunathan, K.; Tiwari, A.; Wang, T.; Kelly, C.V.; Lencer, W.I. Cholera Toxin as a Probe for Membrane Biology. Toxins 2021, 13, 543. [Google Scholar] [CrossRef]
- Ledeen, R.W.; Wu, G. The multi-tasked life of GM1 ganglioside, a true factotum of nature. Trends Biochem. Sci. 2015, 40, 407–418. [Google Scholar] [CrossRef]
- Guimaraes, C.P.; Carette, J.E.; Varadarajan, M.; Antos, J.; Popp, M.W.; Spooner, E.; Brummelkamp, T.R.; Ploegh, H.L. Identification of host cell factors required for intoxication through use of modified cholera toxin. J. Cell Biol. 2011, 195, 751–764. [Google Scholar] [CrossRef]
- Collier, R.J. Understanding the mode of action of diphtheria toxin: A perspective on progress during the 20th century. Toxicon 2001, 39, 1793–1803. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Milne, G.T.; Kuremsky, J.G.; Fink, G.R.; Leppla, S.H. Identification of the proteins required for biosynthesis of diphthamide, the target of bacterial ADP-ribosylating toxins on translation elongation factor 2. Mol. Cell. Biol. 2004, 24, 9487–9497. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Hashiramoto, A.; Haluzik, M.; Mizukami, H.; Beck, S.; Norton, A.; Kono, M.; Tsuji, S.; Daniotti, J.L.; Werth, N.; et al. Enhanced insulin sensitivity in mice lacking ganglioside GM3. Proc. Natl. Acad. Sci. USA 2003, 100, 3445–3449. [Google Scholar] [CrossRef]
- Stanley, P. Golgi glycosylation. Cold Spring Harb. Perspect. Biol. 2011, 3, a005199. [Google Scholar] [CrossRef] [PubMed]
- Varki, A.; Cummings, R.D.; Esko, J.D.; Freeze, H.H.; Stanley, P.; Bertozzi, C.R.; Hart, G.W.; Etzler, M.E. (Eds.) Essentials of Glycobiology, 2nd ed.; Cold Spring Harbor (NY): New York, NY, USA, 2009. [Google Scholar]
- Freeze, H.H.; Chong, J.X.; Bamshad, M.J.; Ng, B.G. Solving glycosylation disorders: Fundamental approaches reveal complicated pathways. Am. J. Hum. Genet. 2014, 94, 161–175. [Google Scholar] [CrossRef]
- Cantarel, B.L.; Coutinho, P.M.; Rancurel, C.; Bernard, T.; Lombard, V.; Henrissat, B. The Carbohydrate-Active EnZymes database (CAZy): An expert resource for Glycogenomics. Nucleic Acids Res. 2009, 37, D233–D238. [Google Scholar] [CrossRef]
- Jaeken, J.; Carchon, H. The carbohydrate-deficient glycoprotein syndromes: An overview. J. Inherit. Metab. Dis. 1993, 16, 813–820. [Google Scholar] [CrossRef]
- Rutishauser, U.; Sachs, L. Cell-to-cell binding induced by different lectins. J. Cell Biol. 1975, 65, 247–257. [Google Scholar] [CrossRef]
- Audi, J.; Belson, M.; Patel, M.; Schier, J.; Osterloh, J. Ricin poisoning: A comprehensive review. JAMA 2005, 294, 2342–2351. [Google Scholar] [CrossRef]
- Schaffrath, R.; Schmitt, M.J.; SpringerLink (Online service). Microbial Protein Toxins. In Topics in Current Genetics; Springer: Berlin/Heidelberg, Germany, 2005; Volume xiii, 237p. [Google Scholar]
- Baenziger, J.U.; Fiete, D. Structural determinants of Ricinus communis agglutinin and toxin specificity for oligosaccharides. J. Biol. Chem. 1979, 254, 9795–9799. [Google Scholar] [CrossRef]
- Taubenschmid, J.; Stadlmann, J.; Jost, M.; Klokk, T.I.; Rillahan, C.D.; Leibbrandt, A.; Mechtler, K.; Paulson, J.C.; Jude, J.; Zuber, J.; et al. A vital sugar code for ricin toxicity. Cell Res. 2017, 27, 1351–1364. [Google Scholar] [CrossRef] [PubMed]
- Morgens, D.W.; Wainberg, M.; Boyle, E.A.; Ursu, O.; Araya, C.L.; Tsui, C.K.; Haney, M.S.; Hess, G.T.; Han, K.; Jeng, E.E.; et al. Genome-scale measurement of off-target activity using Cas9 toxicity in high-throughput screens. Nat. Commun. 2017, 8, 15178. [Google Scholar] [CrossRef] [PubMed]
- Bassik, M.C.; Kampmann, M.; Lebbink, R.J.; Wang, S.; Hein, M.Y.; Poser, I.; Weibezahn, J.; Horlbeck, M.A.; Chen, S.; Mann, M.; et al. A systematic mammalian genetic interaction map reveals pathways underlying ricin susceptibility. Cell 2013, 152, 909–922. [Google Scholar] [CrossRef]
- Moreau, D.; Kumar, P.; Wang, S.C.; Chaumet, A.; Chew, S.Y.; Chevalley, H.; Bard, F. Genome-wide RNAi screens identify genes required for Ricin and PE intoxications. Dev. Cell 2011, 21, 231–244. [Google Scholar] [CrossRef]
- Chen, W.; Tang, J.; Stanley, P. Suppressors of alpha(1,3)fucosylation identified by expression cloning in the LEC11B gain-of-function CHO mutant. Glycobiology 2005, 15, 259–269. [Google Scholar] [CrossRef]
- Elling, U.; Taubenschmid, J.; Wirnsberger, G.; O’Malley, R.; Demers, S.P.; Vanhaelen, Q.; Shukalyuk, A.I.; Schmauss, G.; Schramek, D.; Schnuetgen, F.; et al. Forward and reverse genetics through derivation of haploid mouse embryonic stem cells. Cell Stem Cell 2011, 9, 563–574. [Google Scholar] [CrossRef] [PubMed]
- Potvin, B.; Stanley, P. Activation of two new alpha(1,3)fucosyltransferase activities in Chinese hamster ovary cells by 5-azacytidine. Cell Regul. 1991, 2, 989–1000. [Google Scholar] [CrossRef]
- Voth, D.E.; Ballard, J.D. Clostridium difficile toxins: Mechanism of action and role in disease. Clin. Microbiol. Rev. 2005, 18, 247–263. [Google Scholar] [CrossRef]
- Aktories, K.; Schwan, C.; Jank, T. Clostridium difficile Toxin Biology. Annu. Rev. Microbiol. 2017, 71, 281–307. [Google Scholar] [CrossRef]
- Kordus, S.L.; Thomas, A.K.; Lacy, D.B. Clostridioides difficile toxins: Mechanisms of action and antitoxin therapeutics. Nat. Rev. Microbiol. 2022, 20, 285–298. [Google Scholar] [CrossRef]
- Jank, T.; Aktories, K. Structure and mode of action of clostridial glucosylating toxins: The ABCD model. Trends Microbiol. 2008, 16, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Lam, K.H.; Liu, Z.; Mindlin, F.A.; Chen, B.; Gutierrez, C.B.; Huang, L.; Zhang, Y.; Hamza, T.; Feng, H.; et al. Structure of the full-length Clostridium difficile toxin B. Nat. Struct. Mol. Biol. 2019, 26, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Basak, S.; Chen, P.; Zhang, C.; Perry, K.; Tian, S.; Yu, C.; Dong, M.; Huang, L.; Bowen, M.E.; et al. Structure and conformational dynamics of Clostridioides difficile toxin A. Life Sci. Alliance 2022, 5, e202201383. [Google Scholar] [CrossRef]
- Aminzadeh, A.; Larsen, C.E.; Boesen, T.; Jorgensen, R. High-resolution structure of native toxin A from Clostridioides difficile. EMBO Rep. 2022, 23, e53597. [Google Scholar] [CrossRef] [PubMed]
- Krivan, H.C.; Clark, G.F.; Smith, D.F.; Wilkins, T.D. Cell surface binding site for Clostridium difficile enterotoxin: Evidence for a glycoconjugate containing the sequence Gal alpha 1-3Gal beta 1-4GlcNAc. Infect. Immun. 1986, 53, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Tucker, K.D.; Wilkins, T.D. Toxin A of Clostridium difficile binds to the human carbohydrate antigens I, X, and Y. Infect. Immun. 1991, 59, 73–78. [Google Scholar] [CrossRef]
- Teneberg, S.; Lonnroth, I.; Torres Lopez, J.F.; Galili, U.; Halvarsson, M.O.; Angstrom, J.; Karlsson, K.A. Molecular mimicry in the recognition of glycosphingolipids by Gal alpha 3 Gal beta 4 GlcNAc beta-binding Clostridium difficile toxin A, human natural anti alpha-galactosyl IgG and the monoclonal antibody Gal-13: Characterization of a binding-active human glycosphingolipid, non-identical with the animal receptor. Glycobiology 1996, 6, 599–609. [Google Scholar] [CrossRef]
- Tao, L.; Zhang, J.; Meraner, P.; Tovaglieri, A.; Wu, X.; Gerhard, R.; Zhang, X.; Stallcup, W.B.; Miao, J.; He, X.; et al. Frizzled proteins are colonic epithelial receptors for C. difficile toxin B. Nature 2016, 538, 350–355. [Google Scholar] [CrossRef]
- Luo, J.; Yang, Q.; Zhang, X.; Zhang, Y.; Wan, L.; Zhan, X.; Zhou, Y.; He, L.; Li, D.; Jin, D.; et al. TFPI is a colonic crypt receptor for TcdB from hypervirulent clade 2 C. difficile. Cell 2022, 185, 980–994.e15. [Google Scholar] [CrossRef]
- Lee, H.; Beilhartz, G.L.; Kucharska, I.; Raman, S.; Cui, H.; Lam, M.H.Y.; Liang, H.; Rubinstein, J.L.; Schramek, D.; Julien, J.P.; et al. Recognition of Semaphorin Proteins by P. sordellii Lethal Toxin Reveals Principles of Receptor Specificity in Clostridial Toxins. Cell 2020, 182, 345–356.e16. [Google Scholar] [CrossRef]
- Kreuger, J.; Kjellen, L. Heparan sulfate biosynthesis: Regulation and variability. J. Histochem. Cytochem. 2012, 60, 898–907. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Li, D.; Luo, J.; Chen, A.; Li, X.; Pan, Z.; Wan, L.; He, L.; Li, D.; Li, Y.; et al. Sulfated glycosaminoglycans and low-density lipoprotein receptor mediate the cellular entry of Clostridium novyi alpha-toxin. Cell Res. 2021, 31, 935–938. [Google Scholar] [CrossRef]
- Iozzo, R.V. Matrix proteoglycans: From molecular design to cellular function. Annu. Rev. Biochem. 1998, 67, 609–652. [Google Scholar] [CrossRef] [PubMed]
- Mizumoto, S.; Yamada, S. Congenital Disorders of Deficiency in Glycosaminoglycan Biosynthesis. Front. Genet. 2021, 12, 717535. [Google Scholar] [CrossRef] [PubMed]
- Yuan, P.; Zhang, H.; Cai, C.; Zhu, S.; Zhou, Y.; Yang, X.; He, R.; Li, C.; Guo, S.; Li, S.; et al. Chondroitin sulfate proteoglycan 4 functions as the cellular receptor for Clostridium difficile toxin B. Cell Res. 2015, 25, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Tao, L.; Wang, T.; Zhang, J.; He, A.; Lam, K.H.; Liu, Z.; He, X.; Perry, K.; Dong, M.; et al. Structural basis for recognition of frizzled proteins by Clostridium difficile toxin B. Science 2018, 360, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Zeng, J.; Liu, Z.; Thaker, H.; Wang, S.; Tian, S.; Zhang, J.; Tao, L.; Gutierrez, C.B.; Xing, L.; et al. Structural basis for CSPG4 as a receptor for TcdB and a therapeutic target in Clostridioides difficile infection. Nat. Commun. 2021, 12, 3748. [Google Scholar] [CrossRef]
- Chen, P.; Jin, R. Receptor binding mechanisms of Clostridioides difficile toxin B and implications for therapeutics development. FEBS J. 2023, 290, 962–969. [Google Scholar] [CrossRef]
- Jiang, M.; Shin, J.; Simeon, R.; Chang, J.Y.; Meng, R.; Wang, Y.; Shinde, O.; Li, P.; Chen, Z.; Zhang, J. Structural dynamics of receptor recognition and pH-induced dissociation of full-length Clostridioides difficile Toxin B. PLoS Biol. 2022, 20, e3001589. [Google Scholar] [CrossRef]
- Henkel, D.; Tatge, H.; Schottelndreier, D.; Tao, L.; Dong, M.; Gerhard, R. Receptor Binding Domains of TcdB from Clostridioides difficile for Chondroitin Sulfate Proteoglycan-4 and Frizzled Proteins Are Functionally Independent and Additive. Toxins 2020, 12, 736. [Google Scholar] [CrossRef]
- Shen, E.; Zhu, K.; Li, D.; Pan, Z.; Luo, Y.; Bian, Q.; He, L.; Song, X.; Zhen, Y.; Jin, D.; et al. Subtyping analysis reveals new variants and accelerated evolution of Clostridioides difficile toxin B. Commun. Biol. 2020, 3, 347. [Google Scholar] [CrossRef]
- Mansfield, M.J.; Tremblay, B.J.; Zeng, J.; Wei, X.; Hodgins, H.; Worley, J.; Bry, L.; Dong, M.; Doxey, A.C. Phylogenomics of 8,839 Clostridioides difficile genomes reveals recombination-driven evolution and diversification of toxin A and B. PLoS Pathog. 2020, 16, e1009181. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.P.; Ellery, P.E.; Maroney, S.A.; Mast, A.E. Biology of tissue factor pathway inhibitor. Blood 2014, 123, 2934–2943. [Google Scholar] [CrossRef] [PubMed]
- Mast, A.E. Tissue Factor Pathway Inhibitor: Multiple Anticoagulant Activities for a Single Protein. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 9–14. [Google Scholar] [CrossRef]
- Kinoshita, T.; Fujita, M. Biosynthesis of GPI-anchored proteins: Special emphasis on GPI lipid remodeling. J. Lipid Res. 2016, 57, 6–24. [Google Scholar] [CrossRef]
- Ferguson, M.A.J.; Hart, G.W.; Kinoshita, T. Glycosylphosphatidylinositol Anchors. In Essentials of Glycobiology, 3rd ed.; Varki, A., Cummings, R.D., Esko, J.D., Stanley, P., Hart, G.W., Aebi, M., Darvill, A.G., Kinoshita, T., Packer, N.H., Prestegard, J.H., et al., Eds.; Cold Spring Harbor (NY): New York, NY, USA, 2015; pp. 137–150. [Google Scholar]
- Yamaji, T.; Hanamatsu, H.; Sekizuka, T.; Kuroda, M.; Iwasaki, N.; Ohnishi, M.; Furukawa, J.I.; Yahiro, K.; Hanada, K. A CRISPR Screen Using Subtilase Cytotoxin Identifies SLC39A9 as a Glycan-Regulating Factor. iScience 2019, 15, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Byres, E.; Paton, A.W.; Paton, J.C.; Lofling, J.C.; Smith, D.F.; Wilce, M.C.; Talbot, U.M.; Chong, D.C.; Yu, H.; Huang, S.; et al. Incorporation of a non-human glycan mediates human susceptibility to a bacterial toxin. Nature 2008, 456, 648–652. [Google Scholar] [CrossRef]
- Tromp, A.T.; Van Gent, M.; Jansen, J.P.; Scheepmaker, L.M.; Velthuizen, A.; De Haas, C.J.C.; Van Kessel, K.P.M.; Bardoel, B.W.; Boettcher, M.; McManus, M.T.; et al. Host-Receptor Post-Translational Modifications Refine Staphylococcal Leukocidin Cytotoxicity. Toxins 2020, 12, 106. [Google Scholar] [CrossRef]
- Spaan, A.N.; Henry, T.; van Rooijen, W.J.M.; Perret, M.; Badiou, C.; Aerts, P.C.; Kemmink, J.; de Haas, C.J.C.; van Kessel, K.P.M.; Vandenesch, F.; et al. The staphylococcal toxin Panton-Valentine Leukocidin targets human C5a receptors. Cell Host Microbe 2013, 13, 584–594. [Google Scholar] [CrossRef]
- Spaan, A.N.; Vrieling, M.; Wallet, P.; Badiou, C.; Reyes-Robles, T.; Ohneck, E.A.; Benito, Y.; de Haas, C.J.; Day, C.J.; Jennings, M.P.; et al. The staphylococcal toxins gamma-haemolysin AB and CB differentially target phagocytes by employing specific chemokine receptors. Nat. Commun. 2014, 5, 5438. [Google Scholar] [CrossRef]
- Reyes-Robles, T.; Alonzo, F., 3rd; Kozhaya, L.; Lacy, D.B.; Unutmaz, D.; Torres, V.J. Staphylococcus aureus leukotoxin ED targets the chemokine receptors CXCR1 and CXCR2 to kill leukocytes and promote infection. Cell Host Microbe 2013, 14, 453–459. [Google Scholar] [CrossRef]
- Drabavicius, G.; Daelemans, D. Intermedilysin cytolytic activity depends on heparan sulfates and membrane composition. PLoS Genet. 2021, 17, e1009387. [Google Scholar] [CrossRef]
- Wade, K.R.; Hotze, E.M.; Kuiper, M.J.; Morton, C.J.; Parker, M.W.; Tweten, R.K. An intermolecular electrostatic interaction controls the prepore-to-pore transition in a cholesterol-dependent cytolysin. Proc. Natl. Acad. Sci. USA 2015, 112, 2204–2209. [Google Scholar] [CrossRef]
- Lawrence, S.L.; Gorman, M.A.; Feil, S.C.; Mulhern, T.D.; Kuiper, M.J.; Ratner, A.J.; Tweten, R.K.; Morton, C.J.; Parker, M.W. Structural Basis for Receptor Recognition by the Human CD59-Responsive Cholesterol-Dependent Cytolysins. Structure 2016, 24, 1488–1498. [Google Scholar] [CrossRef] [PubMed]
- Shahi, I.; Llaneras, C.N.; Perelman, S.S.; Torres, V.J.; Ratner, A.J. Genome-Wide CRISPR-Cas9 Screen Does Not Identify Host Factors Modulating Streptococcus agalactiae beta-Hemolysin/Cytolysin-Induced Cell Death. Microbiol. Spectr. 2022, 10, e0218621. [Google Scholar] [CrossRef] [PubMed]
- Piper, S.J.; Brillault, L.; Rothnagel, R.; Croll, T.I.; Box, J.K.; Chassagnon, I.; Scherer, S.; Goldie, K.N.; Jones, S.A.; Schepers, F.; et al. Cryo-EM structures of the pore-forming A subunit from the Yersinia entomophaga ABC toxin. Nat. Commun. 2019, 10, 1952. [Google Scholar] [CrossRef]
- Roderer, D.; Brocker, F.; Sitsel, O.; Kaplonek, P.; Leidreiter, F.; Seeberger, P.H.; Raunser, S. Glycan-dependent cell adhesion mechanism of Tc toxins. Nat. Commun. 2020, 11, 2694. [Google Scholar] [CrossRef]
- Song, N.; Chen, L.; Ren, X.; Waterfield, N.R.; Yang, J.; Yang, G. N-Glycans and sulfated glycosaminoglycans contribute to the action of diverse Tc toxins on mammalian cells. PLoS Pathog. 2021, 17, e1009244. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Viswanatha, R.; Sitsel, O.; Roderer, D.; Zhao, H.; Ashwood, C.; Voelcker, C.; Tian, S.; Raunser, S.; Perrimon, N.; et al. CRISPR screens in Drosophila cells identify Vsg as a Tc toxin receptor. Nature 2022, 610, 349–355. [Google Scholar] [CrossRef]
- Stechmann, B.; Bai, S.K.; Gobbo, E.; Lopez, R.; Merer, G.; Pinchard, S.; Panigai, L.; Tenza, D.; Raposo, G.; Beaumelle, B.; et al. Inhibition of retrograde transport protects mice from lethal ricin challenge. Cell 2010, 141, 231–242. [Google Scholar] [CrossRef]
- Yarwood, R.; Hellicar, J.; Woodman, P.G.; Lowe, M. Membrane trafficking in health and disease. Dis. Model. Mech. 2020, 13, dmm043448. [Google Scholar] [CrossRef] [PubMed]
- Selyunin, A.S.; Iles, L.R.; Bartholomeusz, G.; Mukhopadhyay, S. Genome-wide siRNA screen identifies UNC50 as a regulator of Shiga toxin 2 trafficking. J. Cell Biol. 2017, 216, 3249–3262. [Google Scholar] [CrossRef] [PubMed]
- Virreira Winter, S.; Zychlinsky, A.; Bardoel, B.W. Genome-wide CRISPR screen reveals novel host factors required for Staphylococcus aureus alpha-hemolysin-mediated toxicity. Sci. Rep. 2016, 6, 24242. [Google Scholar] [CrossRef]
- Kouzel, I.U.; Kehl, A.; Berger, P.; Liashkovich, I.; Steil, D.; Makalowski, W.; Suzuki, Y.; Pohlentz, G.; Karch, H.; Mellmann, A.; et al. RAB5A and TRAPPC6B are novel targets for Shiga toxin 2a inactivation in kidney epithelial cells. Sci. Rep. 2020, 10, 4945. [Google Scholar] [CrossRef]
- Michalska, M.; Wolf, P. Pseudomonas Exotoxin A: Optimized by evolution for effective killing. Front. Microbiol. 2015, 6, 963. [Google Scholar] [CrossRef] [PubMed]
- Tafesse, F.G.; Guimaraes, C.P.; Maruyama, T.; Carette, J.E.; Lory, S.; Brummelkamp, T.R.; Ploegh, H.L. GPR107, a G-protein-coupled receptor essential for intoxication by Pseudomonas aeruginosa exotoxin A, localizes to the Golgi and is cleaved by furin. J. Biol. Chem. 2014, 289, 24005–24018. [Google Scholar] [CrossRef]
- Chang, S.J.; Jin, S.C.; Jiao, X.; Galan, J.E. Unique features in the intracellular transport of typhoid toxin revealed by a genome-wide screen. PLoS Pathog. 2019, 15, e1007704. [Google Scholar] [CrossRef]
- Tian, S.; Wu, Q.; Zhou, B.; Choi, M.Y.; Ding, B.; Yang, W.; Dong, M. Proteomic Analysis Identifies Membrane Proteins Dependent on the ER Membrane Protein Complex. Cell Rep. 2019, 28, 2517–2526.e2515. [Google Scholar] [CrossRef] [PubMed]
- Anwar, M.U.; Sergeeva, O.A.; Abrami, L.; Mesquita, F.S.; Lukonin, I.; Amen, T.; Chuat, A.; Capolupo, L.; Liberali, P.; D’Angelo, G.; et al. ER-Golgi-localized proteins TMED2 and TMED10 control the formation of plasma membrane lipid nanodomains. Dev. Cell 2022, 57, 2334–2346.e2338. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.; Chow, S.H.; Stuart, I.; Weir, A.; Yeung, A.T.; Hale, C.; Sridhar, S.; Dougan, G.; Vince, J.E.; Naderer, T. FBXO11 governs macrophage cell death and inflammation in response to bacterial toxins. Life Sci. Alliance 2023, 6, e202201735. [Google Scholar] [CrossRef]
- Alouf, J.E. Bacterial protein toxins. An overview. Methods Mol. Biol. 2000, 145, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Gill, D.M. Bacterial toxins: A table of lethal amounts. Microbiol. Rev. 1982, 46, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Liu, Y.; Appleton, E.; Wang, H.; Church, G.M.; Dong, M. Targeted intracellular delivery of Cas13 and Cas9 nucleases using bacterial toxin-based platforms. Cell Rep. 2022, 38, 110476. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, S.; Zhou, N. Gaining New Insights into Fundamental Biological Pathways by Bacterial Toxin-Based Genetic Screens. Bioengineering 2023, 10, 884. https://doi.org/10.3390/bioengineering10080884
Tian S, Zhou N. Gaining New Insights into Fundamental Biological Pathways by Bacterial Toxin-Based Genetic Screens. Bioengineering. 2023; 10(8):884. https://doi.org/10.3390/bioengineering10080884
Chicago/Turabian StyleTian, Songhai, and Nini Zhou. 2023. "Gaining New Insights into Fundamental Biological Pathways by Bacterial Toxin-Based Genetic Screens" Bioengineering 10, no. 8: 884. https://doi.org/10.3390/bioengineering10080884
APA StyleTian, S., & Zhou, N. (2023). Gaining New Insights into Fundamental Biological Pathways by Bacterial Toxin-Based Genetic Screens. Bioengineering, 10(8), 884. https://doi.org/10.3390/bioengineering10080884







