Revisiting the Role of NAG across the Continuum of Kidney Disease
Abstract
1. Introduction
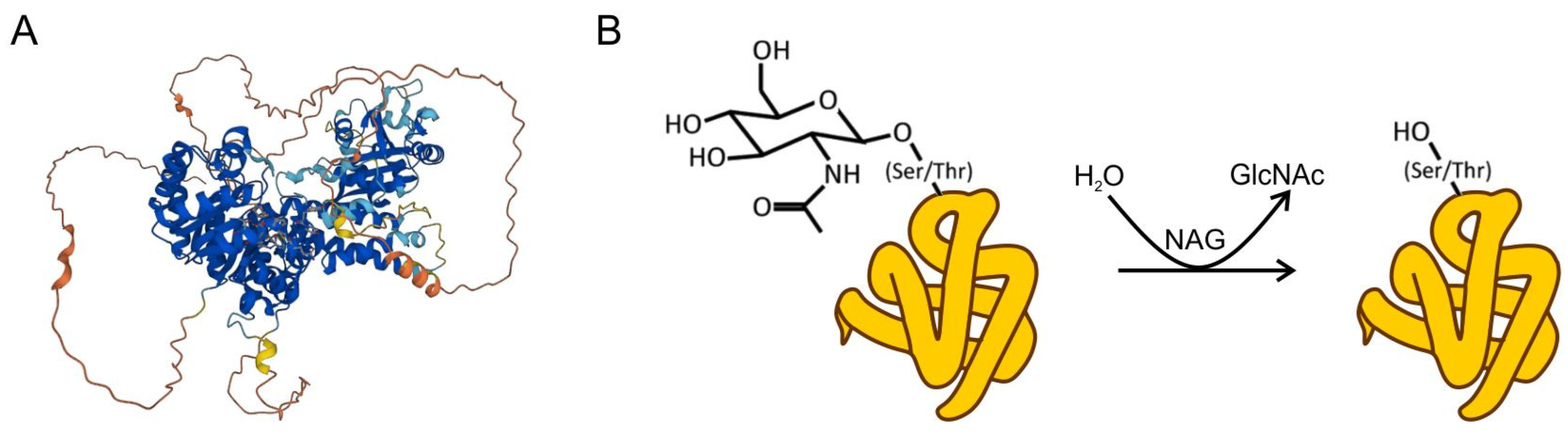
2. Molecular Biology of NAG
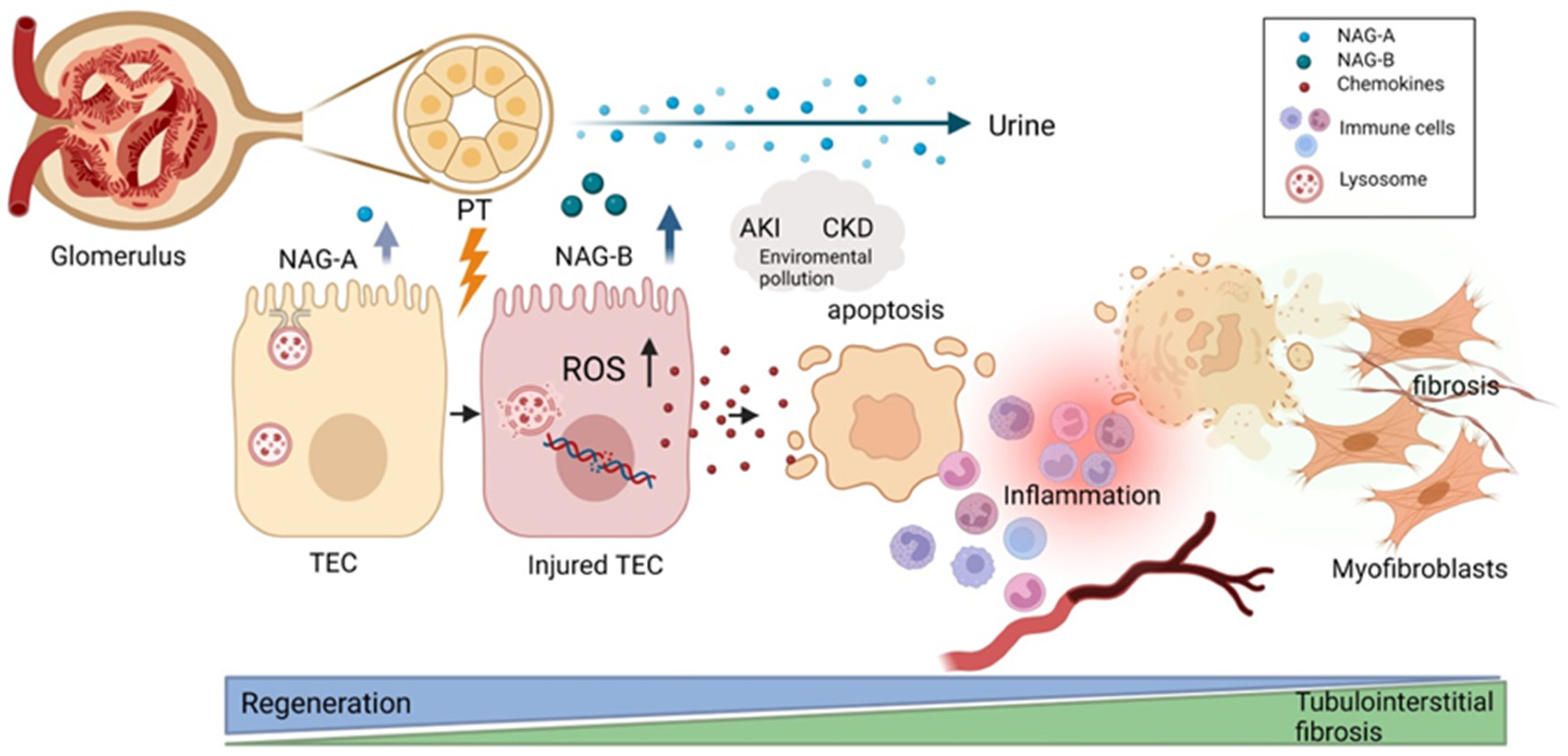
3. NAG in Kidney Injury
3.1. NAG in the Setting of AKI
3.2. NAG in the Setting of CKD
3.3. NAG in Kidney Injury Related to Environmental Nephrotoxins
4. Identifying Potential Drug Targets in Kidney-Disease-Associated Signaling Pathways
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Koeppen, B.M.; Stanton, B. Renal Physiology, 5th ed.; Elsevier: Philadelphia, PA, USA, 2013. [Google Scholar]
- Gheshlaghi, F. Toxic renal injury at a glance. J. Ren. Inj. Prev. 2012, 1, 15–16. [Google Scholar]
- Grgic, I.; Campanholle, G.; Bijol, V.; Wang, C.; Sabbisetti, V.S.; Ichimura, T.; Humphreys, B.D.; Bonventre, J.V. Targeted proximal tubule injury triggers interstitial fibrosis and glomerulosclerosis. Kidney Int. 2012, 82, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Menez, S.; Moledina, D.G.; Garg, A.X.; Thiessen-Philbrook, H.; McArthur, E.; Jia, Y.; Liu, C.; Obeid, W.; Mansour, S.G.; Koyner, J.L.; et al. Results from the TRIBE-AKI Study found associations between post-operative blood biomarkers and risk of chronic kidney disease after cardiac surgery. Kidney Int. 2021, 99, 716–724. [Google Scholar] [CrossRef] [PubMed]
- Chawla, L.S. Acute kidney injury leading to chronic kidney disease and long-term outcomes of acute kidney injury: The best opportunity to mitigate acute kidney injury? Contrib. Nephrol. 2011, 174, 182–190. [Google Scholar]
- Liu, B.-C.; Tang, T.-T.; Lv, L.-L.; Lan, H.-Y. Renal tubule injury: A driving force toward chronic kidney disease. Kidney Int. 2018, 93, 568–579. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.N.; Kalia, K. Efficacy of urinary N-acetyl-β-D-glucosaminidase to evaluate early renal tubular damage as a consequence of type 2 diabetes mellitus: A cross-sectional study. Int. J. Diabetes Dev. Ctries. 2015, 35, 449–457. [Google Scholar] [CrossRef]
- Schlondorff, D.O. Overview of factors contributing to the pathophysiology of progressive renal disease. Kidney Int. 2008, 74, 860–866. [Google Scholar] [CrossRef] [PubMed]
- Schnaper, H.W. The Tubulointerstitial Pathophysiology of Progressive Kidney Disease. Adv. Chronic Kidney Dis. 2017, 24, 107–116. [Google Scholar] [CrossRef]
- Hewitson, T.D. Fibrosis in the kidney: Is a problem shared a problem halved? Fibrogenesis Tissue Repair. 2012, 5, S14. [Google Scholar] [CrossRef]
- Eddy, S.; Mariani, L.H.; Kretzler, M. Integrated multi-omics approaches to improve classification of chronic kidney disease. Nat. Rev. Nephrol. 2020, 16, 657–668. [Google Scholar] [CrossRef]
- Bhatraju, P.K.; Zelnick, L.R.; Katz, R.; Mikacenic, C.; Kosamo, S.; Hahn, W.O.; Dmyterko, V.; Kestenbaum, B.; Christiani, D.C.; Liles, W.C.; et al. A Prediction Model for Severe AKI in Critically Ill Adults That Incorporates Clinical and Biomarker Data. Clin. J. Am. Soc. Nephrol. 2019, 14, 506–514. [Google Scholar] [CrossRef]
- Katz, N.; Ronco, C. Acute kidney stress—A useful term based on evolution in the understanding of acute kidney injury. Crit. Care. 2016, 20, 23. [Google Scholar] [CrossRef] [PubMed]
- Albert, C.; Albert, A.; Kube, J.; Bellomo, R.; Wettersten, N.; Kuppe, H.; Westphal, S.; Haase, M.; Haase-Fielitz, A. Urinary biomarkers may provide prognostic information for subclinical acute kidney injury after cardiac surgery. J. Thorac. Cardiovasc. Surg. 2018, 155, 2441–2452.e13. [Google Scholar] [CrossRef] [PubMed]
- Kashani, K.; Al-Khafaji, A.; Ardiles, T.; Artigas, A.; Bagshaw, S.M.; Bell, M.; Bihorac, A.; Birkhahn, R.; Cely, C.M.; Chawla, L.S.; et al. Discovery and validation of cell cycle arrest biomarkers in human acute kidney injury. Crit. Care. 2013, 17, R25. [Google Scholar] [CrossRef] [PubMed]
- Esmeijer, K.; Schoe, A.; Ruhaak, L.R.; Hoogeveen, E.K.; Soonawala, D.; Romijn, F.P.H.T.M.; Shirzada, M.R.; van Dissel, J.T.; Cobbaert, C.M.; de Fijter, J.W. The predictive value of TIMP-2 and IGFBP7 for kidney failure and 30-day mortality after elective cardiac surgery. Sci. Rep. 2021, 11, 1071. [Google Scholar] [CrossRef] [PubMed]
- Herget-Rosenthal, S.; Poppen, D.; Hüsing, J.; Marggraf, G.; Pietruck, F.; Jakob, H.-G.; Philipp, T.; Kribben, A. Prognostic value of tubular proteinuria and enzymuria in nonoliguric acute tubular necrosis. Clin. Chem. 2004, 50, 552–558. [Google Scholar] [CrossRef]
- Argyropoulos, C.P.; Chen, S.S.; Ng, Y.-H.; Roumelioti, M.-E.; Shaffi, K.; Singh, P.P.; Tzamaloukas, A.H. Rediscovering Beta-2 Microglobulin As a Biomarker across the Spectrum of Kidney Diseases. Front. Med. 2017, 4, 73. [Google Scholar] [CrossRef]
- Paragas, N.; Qiu, A.; Zhang, Q.; Samstein, B.; Deng, S.-X.; Schmidt-Ott, K.M.; Viltard, M.; Yu, W.; Forster, C.S.; Gong, G.; et al. The Ngal reporter mouse detects the response of the kidney to injury in real time. Nat. Med. 2011, 17, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Parikh, C.R.; Coca, S.G.; Thiessen-Philbrook, H.; Shlipak, M.G.; Koyner, J.L.; Wang, Z.; Edelstein, C.L.; Devarajan, P.; Patel, U.D.; Zappitelli, M.; et al. Postoperative biomarkers predict acute kidney injury and poor outcomes after adult cardiac surgery. J. Am. Soc. Nephrol. 2011, 22, 1748–1757. [Google Scholar] [CrossRef]
- Schrezenmeier, E.V.; Barasch, J.; Budde, K.; Westhoff, T.; Schmidt-Ott, K.M. Biomarkers in acute kidney injury—Pathophysiological basis and clinical performance. Acta Physiol. 2017, 219, 554–572. [Google Scholar] [CrossRef]
- Vaidya, V.S.; Ferguson, M.A.; Bonventre, J.V. Biomarkers of acute kidney injury. Annu. Rev. Pharmacol. Toxicol. 2008, 48, 463–493. [Google Scholar] [CrossRef]
- Dance, N.; Price, R.G.; Robinson, D.; Stirling, J.L. β-Galactosidase, β-glucosidase and N-acetyl-β-glucosaminidase in human kidney. Clin. Chim. Acta 1969, 24, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Lockwood, T.D.; Bosmann, H.B. The use of urinary N-acetyl-beta-glucosaminidase in human renal toxicology. II. Elevation in human excretion after aspirin and sodium salicylate. Toxicol. Appl. Pharmacol. 1979, 49, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Ellis, E.N.; Brouhard, B.H.; LaGrone, L. Urinary N-acetyl-beta-D-glucosaminidase in streptozotocin-induced diabetic rats. Biochem. Med. 1984, 31, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Lockwood, T.D.; Bosmann, H.B. The use of urinary N-acetyl-beta-glucosaminidase in human renal toxicology. I. Partial biochemical characterization and excretion in humans and release from the isolated perfused rat kidney. Toxicol. Appl. Pharmacol. 1979, 49, 323–336. [Google Scholar] [CrossRef]
- Sabath, E. Arsenic, Kidney, and Urinary Bladder Disorders. In Handbook of Arsenic Toxicology; Academic Press: Cambridge, MA, USA, 2015; pp. 429–442. [Google Scholar]
- Gao, Y.; Wells, L.; Comer, F.I.; Parker, G.J.; Hart, G.W. Dynamic O-Glycosylation of Nuclear and Cytosolic Proteins: Cloning and characterization of a neutral, cytosolic β-n-acetylglucosaminidase from human brain. J. Biol. Chem. 2001, 276, 9838–9845. [Google Scholar] [CrossRef] [PubMed]
- Fehl, C.; Hanover, J.A. Tools, tactics and objectives to interrogate cellular roles of O-GlcNAc in disease. Nat. Chem. Biol. 2022, 18, 8–17. [Google Scholar] [CrossRef]
- Ma, J.; Li, Y.; Hou, C.; Wu, C. O-GlcNAcAtlas: A database of experimentally identified O-GlcNAc sites and proteins. Glycobiology. 2021, 31, 719–723. [Google Scholar] [CrossRef] [PubMed]
- Hart, G.W. Nutrient regulation of signaling and transcription. J. Biol. Chem. 2019, 294, 2211–2231. [Google Scholar] [CrossRef]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- Li, J.; Huang, C.; Zhang, L.; Lin, L.; Li, Z.; Zhang, F.; Wang, P. Isoforms of human O-GlcNAcase show distinct catalytic efficiencies. Biochemistry 2010, 75, 938–943. [Google Scholar] [CrossRef]
- Gibey, R.; Dupond, J.L.; Henry, J.C. Urinary N-acetyl-beta-D-glucosaminidase (NAG) isoenzyme profiles: A tool for evaluating nephrotoxicity of aminoglycosides and cephalosporins. Clin. Chim. Acta. 1984, 137, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Çuhadar, S.; Semerci, T. Renal Biomarkers N-Acetyl-Beta-D- Glucosaminidase (NAG), Endothelin, and Their Application. Biomark. Kidney Dis. 2016, 369–396. [Google Scholar]
- Mandić, L.M.; Aćimović, J.M.; Jovanović, V.B. The possibility of determining N-acetyl-beta-D-glucosaminidase isoenzymes under alkaline conditions. Clin. Biochem. 2005, 38, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Lousa, I.; Reis, F.; Beirão, I.; Alves, R.; Belo, L.; Santos-Silva, A. New potential biomarkers for chronic kidney disease management—A review of the literature. Int. J. Mol. Sci. 2021, 22, 43. [Google Scholar] [CrossRef]
- Yan, F.; Tian, X.; Luan, Z.; Feng, L.; Ma, X.; James, T.D. NAG-targeting fluorescence based probe for precision diagnosis of kidney injury. Chem. Commun. 2019, 55, 1955–1958. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.N.; Alborn, W.E.; Knierman, M.D.; Konrad, R.J. Alloxan is an inhibitor of O-GlcNAc-selective N-acetyl-beta-D-glucosaminidase. Biochem. Biophys. Res. Commun. 2006, 350, 1038–1043. [Google Scholar] [CrossRef]
- Lee, T.N.; Alborn, W.E.; Knierman, M.D.; Konrad, R.J. The diabetogenic antibiotic streptozotocin modifies the tryptic digest pattern for peptides of the enzyme O-GlcNAc-selective N-acetyl-beta-d-glucosaminidase that contain amino acid residues essential for enzymatic activity. Biochem. Pharmacol. 2006, 72, 710–718. [Google Scholar] [CrossRef]
- Uhlén, M.; Fagerberg, L.; Hallström, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, Å.; Kampf, C.; Sjöstedt, E.; Asplund, A.; et al. Tissue-based map of the human proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef]
- Bosomworth, M.P.; Aparicio, S.R.; Hay, A.W. Urine N-acetyl-beta-D-glucosaminidase--a marker of tubular damage? Nephrol. Dial. Transplant. 1999, 14, 620–626. [Google Scholar] [CrossRef]
- Kim, S.R.; Lee, Y.; Lee, S.-G.; Kang, E.S.; Cha, B.-S.; Kim, J.-H.; Lee, B.W. Urinary N-acetyl-β-D-glucosaminidase, an early marker of diabetic kidney disease, might reflect glucose excursion in patients with type 2 diabetes. Medicine 2016, 95, e4114. [Google Scholar] [CrossRef]
- Kadokura, T.; Saito, M.; Utsuno, A.; Kazuta, K.; Yoshida, S.; Kawasaki, S.; Nagase, I.; Kageyama, S. Ipragliflozin (ASP1941), a selective sodium-dependent glucose cotransporter 2 inhibitor, safely stimulates urinary glucose excretion without inducing hypoglycemia in healthy Japanese subjects. Diabetol. Int. 2011, 2, 172–182. [Google Scholar] [CrossRef]
- Beker, B.M.; Corleto, M.G.; Fieiras, C.; Musso, C.G. Novel acute kidney injury biomarkers: Their characteristics, utility and concerns. Int. Urol. Nephrol. 2018, 50, 705–713. [Google Scholar] [CrossRef]
- Khwaja, A. KDIGO Clinical Practice Guidelines for Acute Kidney Injury. Nephron Clin. Pract. 2012, 120, c179–c184. [Google Scholar] [CrossRef]
- Ostermann, M.; Bellomo, R.; Burdmann, E.A.; Doi, K.; Endre, Z.H.; Goldstein, S.L.; Kane-Gill, S.L.; Liu, K.D.; Prowle, J.R.; Shaw, A.D.; et al. Controversies in acute kidney injury: Conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Conference. Kidney Int. 2020, 98, 294–309. [Google Scholar] [CrossRef]
- Ma, J.; Deng, Y.; Lao, H.; Ouyang, X.; Liang, S.; Wang, Y.; Yao, F.; Deng, Y.; Chen, C. A nomogram incorporating functional and tubular damage biomarkers to predict the risk of acute kidney injury for septic patients. BMC Nephrol. 2021, 22, 176. [Google Scholar] [CrossRef]
- Deng, Y.; Ma, J.; Hou, Y.; Zhou, D.; Hou, T.; Li, J.; Liang, S.; Tan, N.; Chen, C. Combining Serum Cystatin C and Urinary N-Acetyl-Beta-D-Glucosaminidase Improves the Precision for Acute Kidney Injury Diagnosis after Resection of Intracranial Space-Occupying Lesions. Kidney Blood Press. Res. 2020, 45, 142–156. [Google Scholar] [CrossRef]
- Gobe, G.C.; Coombes, J.S.; Fassett, R.G.; Endre, Z.H. Biomarkers of drug-induced acute kidney injury in the adult. Expert. Opin. Drug. Metab. Toxicol. 2015, 11, 1683–1694. [Google Scholar] [CrossRef]
- Skálová, S. The diagnostic role of urinary N-acetyl-beta-D-glucosaminidase (NAG) activity in the detection of renal tubular impairment. Acta Med. Hradec Kral. 2005, 48, 75–80. [Google Scholar]
- Kaufmann, M.; Schlossbauer, M.; Hubauer, U.; Stadler, S.; Fischer, M.; Wallner, S.; Hupf, J.; Zimmermann, M.; Orso, E.; Zeman, F.; et al. N-acety-b-D-glucosaminidase: A potential biomarker for early detection of acute kidney injury in acute chest pain. Nephrology 2020, 25, 135–143. [Google Scholar] [CrossRef]
- Jungbauer, C.G.; Birner, C.; Jung, B.; Buchner, S.; Lubnow, M.; von Bary, C.; Endemann, D.; Banas, B.; Mack, M.; Böger, C.A.; et al. Kidney injury molecule-1 and N-acetyl-β-D-glucosaminidase in chronic heart failure: Possible biomarkers of cardiorenal syndrome. Eur. J. Heart Fail. 2011, 13, 1104–1110. [Google Scholar] [CrossRef]
- Fujigaki, Y.; Tamura, Y.; Nagura, M.; Arai, S.; Ota, T.; Shibata, S.; Kondo, F.; Yamaguchi, Y.; Uchida, S. Unique proximal tubular cell injury and the development of acute kidney injury in adult patients with minimal change nephrotic syndrome. BMC Nephrol. 2017, 18, 339. [Google Scholar] [CrossRef]
- Kim, T.H.; Lee, H.A.; Seo, Y.S.; Lee, Y.R.; Yim, S.Y.; Lee, Y.S.; Suh, S.J.; Jung, Y.K.; Kim, J.H.; An, H.; et al. Assessment and prediction of acute kidney injury in patients with decompensated cirrhosis with serum cystatin C and urine N-acetyl-β-D-glucosaminidase. J. Gastroenterol. Hepatol. 2019, 34, 234–240. [Google Scholar] [CrossRef]
- Deng, Y.; Chi, R.; Chen, S.; Ye, H.; Yuan, J.; Wang, L.; Zhai, Y.; Gao, L.; Zhang, D.; Hu, L.; et al. Evaluation of clinically available renal biomarkers in critically ill adults: A prospective multicenter observational study. Crit. Care. 2017, 21, 46. [Google Scholar] [CrossRef]
- Bíró, E.; Szegedi, I.; Kiss, C.; Oláh, A.V.; Dockrell, M.; Price, R.G.; Szabó, T. The role of urinary N-acetyl-β-D-glucosaminidase in early detection of acute kidney injury among pediatric patients with neoplastic disorders in a retrospective study. BMC Pediatr. 2022, 22, 429. [Google Scholar] [CrossRef]
- Mishra, O.P.; Rai, A.K.; Srivastava, P.; Pandey, K.; Abhinay, A.; Prasad, R.; Mishra, R.N.; Schaefer, F. Predictive ability of urinary biomarkers for outcome in children with acute kidney injury. Pediatr. Nephrol. 2017, 32, 521–527. [Google Scholar] [CrossRef]
- Brouhard, B.H.; LaGrone, L.; Travis, L.B.; Pollard, T.G. Response of urinary N-acetyl-beta-D-glucosaminidase to rapid decreases in blood glucose. Clin. Chim. Acta. 1984, 140, 197–202. [Google Scholar] [CrossRef]
- Tominaga, M.; Fujiyama, K.; Hoshino, T.; Tanaka, Y.; Takeuchi, T.; Honda, M.; Mokuda, O.; Ikeda, T.; Mashiba, H. Urinary N-acetyl-beta-D-glucosaminidase in the patients with hyperthyroidism. Horm. Metab. Res. Horm. Stoffwechselforsch Horm. Metab. 1989, 21, 438–440. [Google Scholar] [CrossRef]
- Wang, L.; Deng, Y.; Zhai, Y.; Xu, F.; Li, J.; Zhang, D.; Gao, L.; Hou, Y.; OuYang, X.; Hu, L.; et al. Impact of blood glucose levels on the accuracy of urinary N-acety-β-D-glucosaminidase for acute kidney injury detection in critically ill adults: A multicenter, prospective, observational study. BMC Nephrol. 2019, 20, 186. [Google Scholar] [CrossRef]
- Liang, S.; Luo, D.; Hu, L.; Fang, M.; Li, J.; Deng, J.; Fang, H.; Zhang, H.; He, L.; Xu, J.; et al. Variations of urinary N-acetyl-β-D-glucosaminidase levels and its performance in detecting acute kidney injury under different thyroid hormones levels: A prospectively recruited, observational study. BMJ Open 2022, 12, e055787. [Google Scholar] [CrossRef]
- KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Off. J. Int. Soc. Nephrol. 2013, 3, 81–90.
- Bikbov, B.; Purcell, C.A.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M.; et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef]
- Vojtusek, I.K.; Laganovic, M.; Burek Kamenaric, M.; Bulimbasic, S.; Hrkac, S.; Salai, G.; Ivkovic, V.; Coric, M.; Novak, R.; Grgurevic, L. First Characterization of ADAMTS-4 in Kidney Tissue and Plasma of Patients with Chronic Kidney Disease—A Potential Novel Diagnostic Indicator. Diagnostics 2022, 12, 648. [Google Scholar] [CrossRef]
- Cañadas-Garre, M.; Anderson, K.; Cappa, R.; Skelly, R.; Smyth, L.J.; McKnight, A.J.; Maxwell, A.P. Genetic Susceptibility to Chronic Kidney Disease—Some More Pieces for the Heritability Puzzle. Front. Genet. 2019, 10, 453. [Google Scholar] [CrossRef]
- Atkins, R.C. The epidemiology of chronic kidney disease. Kidney Int. 2005, 67, S14–S18. [Google Scholar] [CrossRef]
- Kovesdy, C.P. Epidemiology of chronic kidney disease: An update 2022. Kidney Int. Suppl. 2022, 12, 7–11. [Google Scholar] [CrossRef]
- Manns, B.; Hemmelgarn, B.; Tonelli, M.; Au, F.; So, H.; Weaver, R.; Quinn, A.E.; Klarenbach, S. The Cost of Care for People with Chronic Kidney Disease. Can. J. Kidney Health Dis. 2019, 6, 1–11. [Google Scholar] [CrossRef]
- Lopez-Giacoman, S.; Madero, M. Biomarkers in chronic kidney disease, from kidney function to kidney damage. World J. Nephrol. 2015, 4, 57–73. [Google Scholar] [CrossRef]
- Fassett, R.G.; Venuthurupalli, S.K.; Gobe, G.C.; Coombes, J.S.; Cooper, M.A.; Hoy, W.E. Biomarkers in chronic kidney disease: A review. Kidney Int. 2011, 80, 806–821. [Google Scholar] [CrossRef]
- Hallan, S.I.; Ritz, E.; Lydersen, S.; Romundstad, S.; Kvenild, K.; Orth, S.R. Combining GFR and albuminuria to classify CKD improves prediction of ESRD. J. Am. Soc. Nephrol. 2009, 20, 1069–1077. [Google Scholar] [CrossRef]
- Caplin, B.; Nitsch, D. Urinary biomarkers of tubular injury in chronic kidney disease. Kidney Int. 2017, 91, 21–23. [Google Scholar] [CrossRef]
- Hsu, C.-Y.; Xie, D.; Waikar, S.S.; Bonventre, J.V.; Zhang, X.; Sabbisetti, V.; Mifflin, T.E.; Coresh, J.; Diamantidis, C.J.; He, J.; et al. Urine biomarkers of tubular injury do not improve on the clinical model predicting chronic kidney disease progression. Kidney Int. 2017, 91, 196–203. [Google Scholar] [CrossRef]
- Grgurevic, L.; Novak, R.; Salai, G.; Hrkac, S.; Mocibob, M.; Vojtusek, I.K.; Laganovic, M. Stage II of Chronic Kidney Diseas—A Tipping Point in Disease Progression? Biomedicines 2022, 10, 1522. [Google Scholar] [CrossRef]
- Govender, M.A.; Brandenburg, J.-T.; Fabian, J.; Ramsay, M. The Use of ’Omics for Diagnosing and Predicting Progression of Chronic Kidney Disease: A Scoping Review. Front. Genet. 2021, 12, 682929. [Google Scholar] [CrossRef]
- Provenzano, M.; Serra, R.; Garofalo, C.; Michael, A.; Crugliano, G.; Battaglia, Y.; Ielapi, N.; Bracale, U.M.; Faga, T.; Capitoli, G.; et al. OMICS in Chronic Kidney Disease: Focus on Prognosis and Prediction. Int. J. Mol. Sci. 2021, 23, 336. [Google Scholar] [CrossRef]
- Kern, E.F.O.; Erhard, P.; Sun, W.; Genuth, S.; Weiss, M.F. Early urinary markers of diabetic kidney disease: A nested case-control study from the Diabetes Control and Complications Trial (DCCT). Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2010, 55, 824–834. [Google Scholar] [CrossRef]
- Vaidya, V.S.; Niewczas, M.A.; Ficociello, L.H.; Johnson, A.C.; Collings, F.B.; Warram, J.H.; Krolewski, A.S.; Bonventre, J.V. Regression of microalbuminuria in type 1 diabetes is associated with lower levels of urinary tubular injury biomarkers, kidney injury molecule-1, and N-acetyl-β-D-glucosaminidase. Kidney Int. 2011, 79, 464–470. [Google Scholar] [CrossRef]
- Lobato, G.R.; Lobato, M.R.; Thomé, F.S.; Veronese, F.V. Performance of urinary kidney injury molecule-1, neutrophil gelatinase-associated lipocalin, and N-acetyl-β-D-glucosaminidase to predict chronic kidney disease progression and adverse outcomes. Brazilian J. Med. Biol. Res. Rev. Bras. Pesqui. Med. Biol. 2017, 50, e6106. [Google Scholar] [CrossRef]
- Fufaa, G.D.; Weil, E.J.; Nelson, R.G.; Hanson, R.L.; Bonventre, J.V.; Sabbisetti, V.; Waikar, S.S.; Mifflin, T.E.; Zhang, X.; Xie, D.; et al. Association of urinary KIM-1, L-FABP, NAG and NGAL with incident end-stage renal disease and mortality in American Indians with type 2 diabetes mellitus. Diabetologia 2015, 58, 188–198. [Google Scholar] [CrossRef]
- Holdt-Lehmann, B.; Lehmann, A.; Korten, G.; Nagel, H.; Nizze, H.; Schuff-Werner, P. Diagnostic value of urinary alanine aminopeptidase and N-acetyl-beta-D-glucosaminidase in comparison to alpha 1-microglobulin as a marker in evaluating tubular dysfunction in glomerulonephritis patients. Clin. Chim. Acta. 2000, 297, 93–102. [Google Scholar] [CrossRef]
- Bazzi, C.; Petrini, C.; Rizza, V.; Arrigo, G.; Napodano, P.; Paparella, M.; D’Amico, G. Urinary N-acetyl-beta-glucosaminidase excretion is a marker of tubular cell dysfunction and a predictor of outcome in primary glomerulonephritis. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2002, 17, 1890–1896. [Google Scholar] [CrossRef]
- Jungbauer, C.G.; Uecer, E.; Stadler, S.; Birner, C.; Buchner, S.; Maier, L.S.; Luchner, A. N-acteyl-ß-D-glucosaminidase and kidney injury molecule-1: New predictors for long-term progression of chronic kidney disease in patients with heart failure. Nephrology 2016, 21, 490–498. [Google Scholar] [CrossRef]
- An, C.; Akankwasa, G.; Liu, J.; Wang, D.; Cheng, G.; Zhang, J.; Qin, X. Urine markers of renal tubular injury in idiopathic membranous nephropathy: A cross sectional study. Clin. Chim. Acta. 2019, 492, 7–11. [Google Scholar] [CrossRef]
- Ráduly, Z.; Price, R.G.; Dockrell, M.E.C.; Csernoch, L.; Pócsi, I. Urinary Biomarkers of Mycotoxin Induced Nephrotoxicity—Current Status and Expected Future Trends. Toxins 2021, 13, 848. [Google Scholar]
- Yan, L.-J.; Allen, D.C. Cadmium-Induced Kidney Injury: Oxidative Damage as a Unifying Mechanism. Biomolecules 2021, 11, 1575. [Google Scholar] [CrossRef]
- Tripathi, S.; Srivastav, A.K. Cytoarchitectural alterations in kidney of Wistar rat after oral exposure to cadmium chloride. Tissue Cell 2011, 43, 131–136. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, P.; Liang, X.; Tan, C.S.; Tan, J.; Wang, J.; Huang, Q.; Huang, R.; Li, Z.X.; Chen, W.C.; et al. Associations between Urinary Excretion of Cadmium and Renal Biomarkers in Nonsmoking Females: A Cross-Sectional Study in Rural Areas of South China. Int. J. Environ. Res. Public Health 2015, 12, 11988–12001. [Google Scholar] [CrossRef]
- Akerstrom, M.; Sallsten, G.; Lundh, T.; Barregard, L. Associations between urinary excretion of cadmium and proteins in a nonsmoking population: Renal toxicity or normal physiology? Environ. Health Perspect. 2013, 121, 187–191. [Google Scholar] [CrossRef]
- Liu, C.; Li, Y.; Zhu, C.; Dong, Z.; Zhang, K.; Zhao, Y.; Xu, Y. Benchmark dose for cadmium exposure and elevated N-acetyl-β-D-glucosaminidase: A meta-analysis. Environ. Sci. Pollut. Res. Int. 2016, 23, 20528–20538. [Google Scholar] [CrossRef]
- Genchi, G.; Sinicropi, M.S.; Lauria, G.; Carocci, A.; Catalano, A. The Effects of Cadmium Toxicity. Int. J. Environ. Res. Public Health 2020, 17, 3782. [Google Scholar] [CrossRef]
- Eom, S.-Y.; Yim, D.-H.; Huang, M.; Park, C.-H.; Kim, G.-B.; Yu, S.-D.; Choi, B.S.; Park, J.D.; Kim, Y.D.; Kim, H. Copper–zinc imbalance induces kidney tubule damage and oxidative stress in a population exposed to chronic environmental cadmium. Int. Arch. Occup. Environ. Health 2020, 93, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Parvez, F.; Liu, M.; Pesola, G.R.; Gamble, M.V.; Slavkovich, V.; Islam, T.; Ahmed, A.; Hasan, R.; Graziano, J.H.; et al. Association between arsenic exposure from drinking water and proteinuria: Results from the Health Effects of Arsenic Longitudinal Study. Int. J. Epidemiol. 2011, 40, 828–835. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Kuo, C.-C.; Fadrowski, J.; Agnew, J.; Weaver, V.M.; Navas-Acien, A. Arsenic and Chronic Kidney Disease: A Systematic Review. Curr. Environ. Health Rep. 2014, 1, 192–207. [Google Scholar] [CrossRef]
- Farkhondeh, T.; Naseri, K.; Esform, A.; Aramjoo, H.; Naghizadeh, A. Drinking water heavy metal toxicity and chronic kidney diseases: A systematic review. Rev. Environ. Health 2021, 36, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Pfohl-Leszkowicz, A.; Manderville, R.A. Ochratoxin A: An overview on toxicity and carcinogenicity in animals and humans. Mol. Nutr. Food Res. 2007, 51, 61–99. [Google Scholar] [CrossRef]
- Grosman, M.E.; Elías, M.M.; Comin, E.J.; Rodriguez Garay, E.A. Alterations in renal function induced by aflatoxin B1 in the rat. Toxicol. Appl. Pharmacol. 1983, 69, 319–325. [Google Scholar] [CrossRef]
- Rached, E.; Hoffmann, D.; Blumbach, K.; Weber, K.; Dekant, W.; Mally, A. Evaluation of putative biomarkers of nephrotoxicity after exposure to ochratoxin a in vivo and in vitro. Toxicol. Sci. 2008, 103, 371–381. [Google Scholar] [CrossRef]
- Perazella, M.A.; Rosner, M.H. Drug-Induced Acute Kidney Injury. Clin. J. Am. Soc. Nephrol. 2022, 17, 1220–1233. [Google Scholar] [CrossRef]
- Wilkinson, R.; Wang, X.; Roper, K.E.; Healy, H. Activated human renal tubular cells inhibit autologous immune responses. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2011, 26, 1483–1492. [Google Scholar] [CrossRef]
- Vormann, M.K.; Vriend, J.; Lanz, H.L.; Gijzen, L.; van den Heuvel, A.; Hutter, S.; Joore, J.; Trietsch, S.J.; Stuut, C.; Nieskens, T.T.G.; et al. Implementation of a Human Renal Proximal Tubule on a Chip for Nephrotoxicity and Drug Interaction Studies. J. Pharm. Sci. 2021, 110, 1601–1614. [Google Scholar] [CrossRef]
- Basnakian, A.G.; Kaushal, G.P.; Shah, S.V. Apoptotic pathways of oxidative damage to renal tubular epithelial cells. Antioxid. Redox Signal. 2002, 4, 915–924. [Google Scholar] [CrossRef]
- White, S.; Lin, L.; Hu, K. NF-κB and tPA Signaling in Kidney and Other Diseases. Cells 2020, 9, 1348. [Google Scholar] [CrossRef]
- Pașatu-Cornea, A.-M.; Ciciu, E.; Tuță, L.-A. Perforin: An intriguing protein in allograft rejection immunology (Review). Exp. Ther. Med. 2022, 24, 519. [Google Scholar] [CrossRef]
- Li, S.-S.; Sun, Q.; Hua, M.-R.; Suo, P.; Chen, J.-R.; Yu, X.-Y.; Zhao, Y.Y. Targeting the Wnt/β-Catenin Signaling Pathway as a Potential Therapeutic Strategy in Renal Tubulointerstitial Fibrosis. Front. Pharmacol. 2021, 12, 719880. [Google Scholar] [CrossRef]
- Yuan, Q.; Tang, B.; Zhang, C. Signaling pathways of chronic kidney diseases, implications for therapeutics. Signal. Transduct. Target. Ther. 2022, 7, 182. [Google Scholar] [CrossRef] [PubMed]
- Guzzi, F.; Cirillo, L.; Roperto, R.M.; Romagnani, P.; Lazzeri, E. Molecular Mechanisms of the Acute Kidney Injury to Chronic Kidney Disease Transition: An Updated View. Int. J. Mol. Sci. 2019, 20, 4941. [Google Scholar] [CrossRef]
- Cisek, K.; Krochmal, M.; Klein, J.; Mischak, H. The application of multi-omics and systems biology to identify therapeutic targets in chronic kidney disease. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2016, 31, 2003–2011. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, S.M.H.; Chen, W.; Liu, Y.; Awal, M.A.; Ahmed, K.; Rahman, M.H.; Moni, M.A. PreDTIs: Prediction of drug–target interactions based on multiple feature information using gradient boosting framework with data balancing and feature selection techniques. Brief. Bioinform. 2021, 22, bbab046. [Google Scholar] [CrossRef]
- Hasan, M.T.; Hassan, M.; Ahmed, K.; Islam, M.R.; Islam, K.; Bhuyian, T.; Uddin, M.S.; Paul, B.K. Network based study to explore genetic linkage between diabetes mellitus and myocardial ischemia: Bioinformatics approach. Gene Rep. 2020, 21, 100809. [Google Scholar] [CrossRef]
- Kawsar, M.; Taz, T.A.; Paul, B.K.; Ahmed, K.; Habib, M.A.; Bhuyian, T. Identification of vital regulatory genes with network pathways among Huntington’s, Parkinson’s, and Alzheimer’s diseases. Netw. Model. Anal. Health. Inform. Bioinform. 2020, 9, 50. [Google Scholar] [CrossRef]
| Authors, Year | Type of Study | Conclusions |
|---|---|---|
| Holdt-Lehmann et al., 2000 [82] | Cross-sectional study | uNAG was significantly elevated in patients with glomerulonephritis compared with healthy controls. |
| Bazzi et al., 2002 [83] | Cross-sectional study | uNAG excretion can be considered as a reliable marker of the tubulo-toxicity of proteinuria in the early stage of IMN, FSGS, and MCD. |
| Kern et al., 2010 [78] | Nested case-control during 1-9 years of follow-up | Elevated uNAG at baseline independently predicted micro- and macroalbuminuria in patients with type I diabetes metllitus. |
| Vaidya et al., 2011 [79] | Prospective study during 2 years of follow-up | Lower uNAG levels were associated with regression of microalbuminuria in patients with type I diabetes mellitus. |
| Kim et al., 2016 [43] | Cross-sectional study | uNAG may be related to glycemic parameters reflecting glucose fluctuation and decreased insulin secretory capacity in patients with type II diabetes mellitus. |
| Jungbauer et al., 2016 [84] | Prospective study during 5-year follow-up | Elevated uNAG was an independent predictor of ESRD and all-cause mortality in HF patients. |
| Lobato et al., 2017 [80] | Prospective study during median 15-months follow-up | uNAG levels were weakly correlated with CKD stages I-V defined by KDIGO; uNAG was NOT associated with CKD progression or renal adverse outcomes. |
| Hsu et al., 2017 [74] | Prospective study (9433 person-years) | uNAG was associated with CKD progression before adjustment. After adjustment for established risk factors of CKD, there was no independent association. |
| An et al., 2019 [85] | Cross-sectional study | uNAG was increased in patients with IMN, compared with healthy controls. No correlation between histological grade and uNAG was observed. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Novak, R.; Salai, G.; Hrkac, S.; Vojtusek, I.K.; Grgurevic, L. Revisiting the Role of NAG across the Continuum of Kidney Disease. Bioengineering 2023, 10, 444. https://doi.org/10.3390/bioengineering10040444
Novak R, Salai G, Hrkac S, Vojtusek IK, Grgurevic L. Revisiting the Role of NAG across the Continuum of Kidney Disease. Bioengineering. 2023; 10(4):444. https://doi.org/10.3390/bioengineering10040444
Chicago/Turabian StyleNovak, Ruder, Grgur Salai, Stela Hrkac, Ivana Kovacevic Vojtusek, and Lovorka Grgurevic. 2023. "Revisiting the Role of NAG across the Continuum of Kidney Disease" Bioengineering 10, no. 4: 444. https://doi.org/10.3390/bioengineering10040444
APA StyleNovak, R., Salai, G., Hrkac, S., Vojtusek, I. K., & Grgurevic, L. (2023). Revisiting the Role of NAG across the Continuum of Kidney Disease. Bioengineering, 10(4), 444. https://doi.org/10.3390/bioengineering10040444








