Adipose Tissue-Derived Components: From Cells to Tissue Glue to Treat Dermal Damage
Abstract
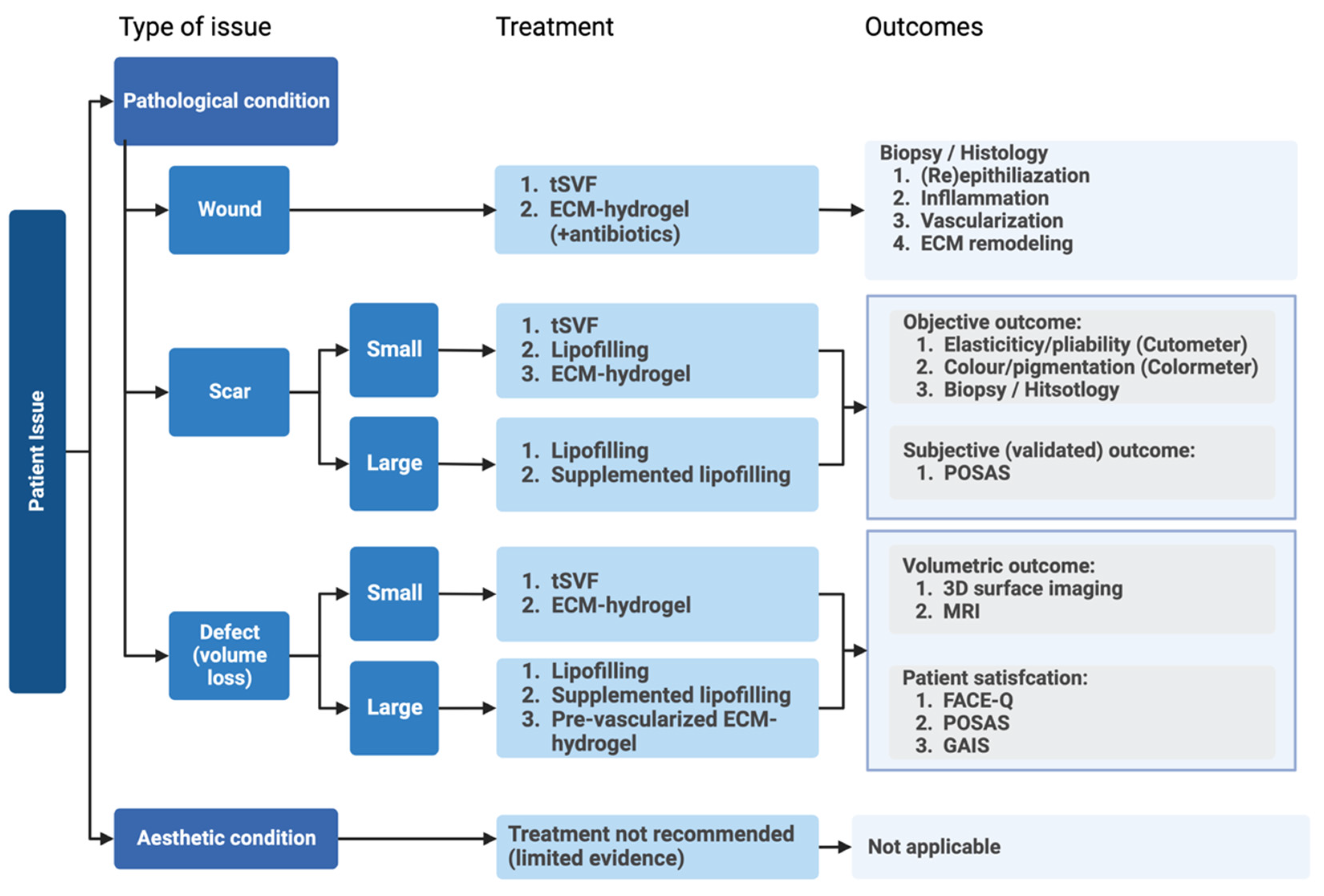
1. Introduction
2. Regenerative Components of Adipose Tissue
3. The Extracellular Matrix as an Instructive Scaffold and Delivery Vehicle for Trophic Factors
4. Conclusions and Future Perspective
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Singer, A.J.; Clark, R.A. Cutaneous wound healing. N. Engl. J. Med. 1999, 341, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Furie, B.; Furie, B.C. Mechanisms of thrombus formation. N. Engl. J. Med. 2008, 359, 938–949. [Google Scholar] [CrossRef] [PubMed]
- Eming, S.A.; Krieg, T.; Davidson, J.M. Inflammation in wound repair: Molecular and cellular mechanisms. J. Investig. Dermatol. 2007, 127, 514–525. [Google Scholar] [CrossRef]
- Hosseini, M.; Brown, J.; Khosrotehrani, K.; Bayat, A.; Shafiee, A. Skin biomechanics: A potential therapeutic intervention target to reduce scarring. Burns Trauma 2022, 10, tkac036. [Google Scholar] [CrossRef]
- Condé-Green, A.; Marano, A.A.; Lee, E.S.; Reisler, T.; Price, L.A.; Milner, S.M.; Granick, M.S. Fat Grafting and Adipose-Derived Regenerative Cells in Burn Wound Healing and Scarring: A Systematic Review of the Literature. Plast. Reconstr. Surg. 2016, 137, 302–312. [Google Scholar] [CrossRef]
- Tedesco, M.; Bellei, B.; Garelli, V.; Caputo, S.; Latini, A.; Giuliani, M.; Cota, C.; Chichierchia, G.; Romani, C.; Foddai, M.L.; et al. Adipose tissue stromal vascular fraction and adipose tissue stromal vascular fraction plus platelet-rich plasma grafting: New regenerative perspectives in genital lichen sclerosus. Dermatol. Ther. 2020, 33, e14277. [Google Scholar] [CrossRef]
- Bellei, B.; Migliano, E.; Picardo, M. Research update of adipose tissue-based therapies in regenerative dermatology. Stem Cell Rev. Rep. 2022, 18, 1956–1973. [Google Scholar] [CrossRef]
- Bellei, B.; Migliano, E.; Picardo, M. Therapeutic potential of adipose tissue-derivatives in modern dermatology. Exp. Dermatol. 2022, 31, 1837–1852. [Google Scholar] [CrossRef]
- Walocko, F.M.; Eber, A.E.; Kirsner, R.S.; Badiavas, E.; Nouri, K. Systematic review of the therapeutic roles of adipose tissue in dermatology. J. Am. Acad. Dermatol. 2018, 79, 935–944. [Google Scholar] [CrossRef]
- Negenborn, V.L.; Groen, J.W.; Smit, J.M.; Niessen, F.B.; Mullender, M.G. The Use of Autologous Fat Grafting for Treatment of Scar Tissue and Scar-Related Conditions: A Systematic Review. Plast. Reconstr. Surg. 2016, 137, 31e–43e. [Google Scholar] [CrossRef]
- Krastev, T.K.; Schop, S.J.; Hommes, J.; Piatkowski, A.; van der Hulst, R.R.W.J. Autologous fat transfer to treat fibrosis and scar-related conditions: A systematic review and meta-analysis. J. Plast. Reconstr. Aesthet. Surg. 2020, 73, 2033–2048. [Google Scholar] [CrossRef]
- Stachura, A.; Paskal, W.; Pawlik, W.; Mazurek, M.J.; Jaworowski, J. The Use of Adipose-Derived Stem Cells (ADSCs) and Stromal Vascular Fraction (SVF) in Skin Scar Treatment-A Systematic Review of Clinical Studies. J. Clin. Med. 2021, 10, 3637. [Google Scholar] [CrossRef]
- Yoshimura, K.; Shigeura, T.; Matsumoto, D.; Sato, T.; Takaki, Y.; Aiba-Kojima, E.; Sato, K.; Inoue, K.; Nagase, T.; Koshima, I.; et al. Characterization of freshly isolated and cultured cells derived from the fatty and fluid portions of liposuction aspirates. J. Cell Physiol. 2006, 208, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Corselli, M.; Chen, C.W.; Sun, B.; Yap, S.; Rubin, J.P.; Péault, B. The tunica adventitia of human arteries and veins as a source of mesenchymal stem cells. Stem Cells Dev. 2012, 21, 1299–1308. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.; Garcia, M.; Ning, H.; Banie, L.; Guo, Y.L.; Lue, T.F.; Lin, C.S. Defining stem and progenitor cells within adipose tissue. Stem Cells Dev. 2008, 17, 1053–1063. [Google Scholar] [CrossRef]
- Ferroni, L.; Gardin, C.; Tocco, I.; Epis, R.; Casadei, A.; Vindigni, V.; Mucci, G.; Zavan, B. Potential for neural differentiation of mesenchymal stem cells. Adv. Biochem. Eng. Biotechnol. 2013, 129, 89–115. [Google Scholar] [PubMed]
- Baer, P.C. Adipose-derived stem cells and their potential to differentiate into the epithelial lineage. Stem Cells Dev. 2011, 20, 1805–1816. [Google Scholar] [CrossRef] [PubMed]
- Zuk, P.A.; Zhu, M.; Ashjian, P.; De Ugarte, D.A.; Huang, J.I.; Mizuno, H.; Alfonso, Z.C.; Fraser, J.K.; Benhaim, P.; Hedrick, M.H. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell. 2002, 13, 4279–4295. [Google Scholar] [CrossRef] [PubMed]
- Spiekman, M.; van Dongen, J.A.; Willemsen, J.C.; Hoppe, D.L.; van der Lei, B.; Harmsen, M.C. The power of fat and its adipose-derived stromal cells: Emerging concepts for fibrotic scar treatment. J. Tissue Eng. Regen. Med. 2017, 11, 3220–3235. [Google Scholar] [CrossRef]
- Weidenbusch, M.; Anders, H.J. Tissue microenvironments define and get reinforced by macrophage phenotypes in homeostasis or during inflammation, repair and fibrosis. J. Innate Immun. 2012, 4, 463–477. [Google Scholar] [CrossRef]
- Le Blanc, K.; Mougiakakos, D. Multipotent mesenchymal stromal cells and the innate immune system. Nat. Rev. Immunol. 2012, 12, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Jariwala, N.; Ozols, M.; Bell, M.; Bradley, E.; Gilmore, A.; Debelle, L.; Sherratt, M.J. Matrikines as mediators of tissue remodelling. Adv. Drug Deliv. Rev. 2022, 185, 114240. [Google Scholar] [CrossRef]
- Wynn, T.A.; Vannella, K.M. Macrophages in Tissue Repair, Regeneration, and Fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Badylak, S.F.; Gilbert, T.W. Immune response to biologic scaffold materials. Semin. Immunol. 2008, 20, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Badylak, S.F.; Valentin, J.E.; Ravindra, A.K.; McCabe, G.P.; Stewart-Akers, A.M. Macrophage phenotype as a determinant of biologic scaffold remodeling. Tissue Eng. Part A 2008, 14, 1835–1842. [Google Scholar] [CrossRef]
- van Dongen, J.A.; Boxtel, J.V.; Uguten, M.; Brouwer, L.A.; Vermeulen, K.M.; Melenhorst, W.B.; Niessen, F.B.; Harmsen, M.C.; Stevens, H.P.; van der Lei, B. Tissue Stromal Vascular Fraction Improves Early Scar Healing: A Prospective Randomized Multicenter Clinical Trial. Aesthet. Surg. J. 2021, 42, NP477–NP488. [Google Scholar] [CrossRef]
- Xue, M.; Jackson, C.J. Extracellular Matrix Reorganization during Wound Healing and Its Impact on Abnormal Scarring. Adv. Wound Care (New Rochelle) 2015, 4, 119–136. [Google Scholar] [CrossRef]
- Hayakawa, T.; Hashimoto, Y.; Myokei, Y.; Aoyama, H.; Izawa, Y. Changes in type of collagen during the development of human post-burn hypertrophic scars. Clin. Chim. Acta 1979, 93, 119–125. [Google Scholar] [CrossRef]
- Rehman, J.; Traktuev, D.; Li, J.; Merfeld-Clauss, S.; Temm-Grove, C.J.; Bovenkerk, J.E.; Pell, C.L.; Johnstone, B.H.; Considine, R.V.; March, K.L. Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation 2004, 109, 1292–1298. [Google Scholar] [CrossRef]
- Amos, P.J.; Kapur, S.K.; Stapor, P.C.; Shang, H.; Bekiranov, S.; Khurgel, M.; Rodeheaver, G.T.; Peirce, S.M.; Katz, A.J. Human adipose-derived stromal cells accelerate diabetic wound healing: Impact of cell formulation and delivery. Tissue Eng. Part A 2010, 16, 1595–1606. [Google Scholar] [CrossRef]
- Vanderstichele, S.; Vranckx, J.J. Anti-fibrotic effect of adipose-derived stem cells on fibrotic scars. World J. Stem Cells 2022, 14, 200–213. [Google Scholar] [CrossRef] [PubMed]
- Spiekman, M.; Przybyt, E.; Plantinga, J.A.; Gibbs, S.; van der Lei, B.; Harmsen, M.C. Adipose tissue-derived stromal cells inhibit TGF-β1-induced differentiation of human dermal fibroblasts and keloid scar-derived fibroblasts in a paracrine fashion. Plast. Reconstr. Surg. 2014, 134, 699–712. [Google Scholar] [CrossRef]
- Desmoulière, A.; Chaponnier, C.; Gabbiani, G. Tissue repair, contraction, and the myofibroblast. Wound Repair. Regen. 2005, 13, 7–12. [Google Scholar] [CrossRef]
- Rong, Y.H.; Zhang, G.A.; Wang, C.; Ning, F.G. Quantification of type I and III collagen content in normal human skin in different age groups. Zhonghua Shao Shang Za Zhi 2008, 24, 51–53. (In Chinese) [Google Scholar] [PubMed]
- Skorochod, R.; Fteiha, B.; Gronovich, Y. Perforation of Abdominal Viscera Following Liposuction: A Systemic Literature Review. Aesthetic Plast. Surg. 2022, 46, 774–785. [Google Scholar] [CrossRef]
- Alicka, M.; Major, P.; Wysocki, M.; Marycz, K. Adipose-Derived Mesenchymal Stem Cells Isolated from Patients with Type 2 Diabetes Show Reduced “Stemness” through an Altered Secretome Profile, Impaired Anti-Oxidative Protection, and Mitochondrial Dynamics Deterioration. J. Clin. Med. 2019, 8, 765. [Google Scholar] [CrossRef]
- Abu-Shahba, N.; Mahmoud, M.; El-Erian, A.M.; Husseiny, M.I.; Nour-Eldeen, G.; Helwa, I.; Amr, K.; ElHefnawi, M.; Othman, A.I.; Ibrahim, S.A.; et al. Impact of type 2 diabetes mellitus on the immunoregulatory characteristics of adipose tissue-derived mesenchymal stem cells. Int. J. Biochem. Cell Biol. 2021, 140, 106072. [Google Scholar] [CrossRef]
- Andjelkov, K.; Conde-Green, A.; Mosahebi, A. Smoking and Physical Activity Significantly Influence Stromal Vascular Fraction Cell Yield and Viability. Aesthetic Plast. Surg. 2021, 45, 315–321. [Google Scholar] [CrossRef]
- Dos-Anjos Vilaboa, S.; Navarro-Palou, M.; Llull, R. Age influence on stromal vascular fraction cell yield obtained from human lipoaspirates. Cytotherapy 2014, 16, 1092–1097. [Google Scholar] [CrossRef]
- Alaaeddine, N.; El Atat, O.; Saliba, N.; Feghali, Z.; Nasr, M.; Tarabey, B.; Hilal, G.; Hashim, H. Effect of age and body mass index on the yield of stromal vascular fraction. J. Cosmet. Dermatol. 2018, 17, 1233–1239. [Google Scholar] [CrossRef]
- van Dongen, J.A.; Stevens, H.P.; Parvizi, M.; van der Lei, B.; Harmsen, M.C. The fractionation of adipose tissue procedure to obtain stromal vascular fractions for regenerative purposes. Wound Repair. Regen. 2016, 24, 994–1003. [Google Scholar] [CrossRef]
- van Dongen, J.A.; Tuin, A.J.; Spiekman, M.; Jansma, J.; van der Lei, B.; Harmsen, M.C. Comparison of intraoperative procedures for isolation of clinical grade stromal vascular fraction for regenerative purposes: A systematic review. J. Tissue Eng. Regen. Med. 2018, 12, e261–e274. [Google Scholar] [CrossRef] [PubMed]
- van Dongen, J.A.; Getova, V.; Brouwer, L.A.; Liguori, G.R.; Sharma, P.K.; Stevens, H.P.; van der Lei, B.; Harmsen, M.C. Adipose tissue-derived extracellular matrix hydrogels as a release platform for secreted paracrine factors. J. Tissue Eng. Regen. Med. 2019, 13, 973–985. [Google Scholar] [CrossRef]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef]
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. Molecular Biology of the Cell, 4th ed.; Chapter ECM; Garland Science: New York, NY, USA, 2002. [Google Scholar]
- Martinez-Garcia, F.D.; de Hilster, R.H.J.; Sharma, P.K.; Borghuis, T.; Hylkema, M.N.; Burgess, J.K.; Harmsen, M.C. Architecture and Composition Dictate Viscoelastic Properties of Organ-Derived Extracellular Matrix Hydrogels. Polymers 2021, 13, 3113. [Google Scholar] [CrossRef]
- Frantz, C.; Stewart, K.M.; Weaver, V.M. The extracellular matrix at a glance. J. Cell Sci. 2010, 123 Pt 24, 4195–4200. [Google Scholar] [CrossRef]
- Clause, K.C.; Barker, T.H. Extracellular matrix signaling in morphogenesis and repair. Curr. Opin. Biotechnol. 2013, 24, 830–833. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Garcia, F.D.; Valk, M.M.; Sharma, P.K.; Burgess, J.K.; Harmsen, M.C. Adipose Tissue-Derived Stromal Cells Alter the Mechanical Stability and Viscoelastic Properties of Gelatine Methacryloyl Hydrogels. Int. J. Mol. Sci 2021, 22, 10153. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Garcia, F.D.; van Dongen, J.A.; Burgess, J.K.; Harmsen, M.C. Matrix Metalloproteases from Adipose Tissue-Derived Stromal Cells Are Spatiotemporally Regulated by Hydrogel Mechanics in a 3D Microenvironment. Bioengineering 2022, 9, 340. [Google Scholar] [CrossRef]
- Zhang, M.; Getova, V.E.; Martinez-Garcia, F.D.; Borghuis, T.; Burgess, J.K.; Harmsen, M.C. From Macro to Micro: Comparison of Imaging Techniques to Detect Vascular Network Formation in Left Ventricle Decellularized Extracellular Matrix Hydrogels. Gels 2022, 8, 729. [Google Scholar] [CrossRef]
- Liguori, T.T.A.; Liguori, G.R.; van Dongen, J.A.; Moreira, L.F.P.; Harmsen, M.C. Bioactive decellularized cardiac extracellular matrix-based hydrogel as a sustained-release platform for human adipose tissue-derived stromal cell-secreted factors. Biomed. Mater. 2021, 16, 025022. [Google Scholar] [CrossRef] [PubMed]
- Rozario, T.; DeSimone, D.W. The extracellular matrix in development and morphogenesis: A dynamic view. Dev. Biol. 2010, 341, 126–140. [Google Scholar] [CrossRef]
- Vriend, L.; van Dongen, J.A.; Sinkunas, V.; Brouwer, L.A.; Buikema, H.J.; Moreira, L.F.; Gemperli, R.; Bongiovanni, L.; de Bruin, A.; van der Lei, B.; et al. Limited Efficacy of Adipose Stromal Cell Secretome-Loaded Skin-Derived Hydrogels to Augment Skin Flap Regeneration in Rats. Stem Cells Dev. 2022, 31, 630–640. [Google Scholar] [CrossRef] [PubMed]
- Vriend, L.; Sinkunas, V.; Camargo, C.P.; van der Lei, B.; Harmsen, M.C.; van Dongen, J.A. Extracellular Matrix-Derived Hydrogels to Augment Dermal Wound Healing: A Systematic Review. Tissue Eng. Part B Rev. 2022, 28, 1093–1108. [Google Scholar] [CrossRef]
- Chan, B.P.; Leong, K.W. Scaffolding in tissue engineering: General approaches and tissue-specific considerations. Eur. Spine J. 2008, 17 (Suppl. 4), 467–479. [Google Scholar] [CrossRef] [PubMed]
- Gu, X. Progress and perspectives of neural tissue engineering. Front. Med. 2015, 9, 401–411. [Google Scholar] [CrossRef]
- Yi, S.; Ding, F.; Gong, L.; Gu, X. Extracellular Matrix Scaffolds for Tissue Engineering and Regenerative Medicine. Curr. Stem Cell Res. Ther. 2017, 12, 233–246. [Google Scholar] [CrossRef]
- Getova, V.E.; van Dongen, J.A.; Brouwer, L.A.; Harmsen, M.C. Adipose tissue-derived ECM hydrogels and their use as 3D culture scaffold. Artif. Cells Nanomed. Biotechnol. 2019, 47, 1693–1701. [Google Scholar] [CrossRef]
- Seif-Naraghi, S.B.; Singelyn, J.M.; Salvatore, M.A.; Osborn, K.G.; Wang, J.J.; Sampat, U.; Kwan, O.L.; Strachan, G.M.; Wong, J.; Schup-Magoffin, P.J.; et al. Safety and efficacy of an injectable extracellular matrix hydrogel for treating myocardial infarction. Sci. Transl. Med. 2013, 5, 173ra25. [Google Scholar] [CrossRef]
- Singelyn, J.M.; DeQuach, J.A.; Seif-Naraghi, S.B.; Littlefield, R.B.; Schup-Magoffin, P.J.; Christman, K.L. Naturally derived myocardial matrix as an injectable scaffold for cardiac tissue engineering. Biomaterials 2009, 30, 5409–5416. [Google Scholar] [CrossRef]
- Pouliot, R.A.; Link, P.A.; Mikhaiel, N.S.; Schneck, M.B.; Valentine, M.S.; Kamga Gninzeko, F.J.; Herbert, J.A.; Sakagami, M.; Heise, R.L. Development and characterization of a naturally derived lung extracellular matrix hydrogel. J. Biomed. Mater. Res. A 2016, 104, 1922–1935. [Google Scholar] [CrossRef]
- Nizamoglu, M.; de Hilster, R.H.J.; Zhao, F.; Sharma, P.K.; Borghuis, T.; Harmsen, M.C.; Burgess, J.K. An in vitro model of fibrosis using crosslinked native extracellular matrix-derived hydrogels to modulate biomechanics without changing composition. Acta Biomater. 2022, 147, 50–62. [Google Scholar] [CrossRef]
- de Hilster, R.H.J.; Sharma, P.K.; Jonker, M.R.; White, E.S.; Gercama, E.A.; Roobeek, M.; Timens, W.; Harmsen, M.C.; Hylkema, M.N.; Burgess, J.K. Human lung extracellular matrix hydrogels resemble the stiffness and viscoelasticity of native lung tissue. Am. J. Physiol. Lung Cell Mol. Physiol. 2020, 318, L698–L704. [Google Scholar] [CrossRef] [PubMed]
- Evangelista-Leite, D.; Carreira, A.C.O.; Gilpin, S.E.; Miglino, M.A. Protective Effects of Extracellular Matrix-Derived Hydrogels in Idiopathic Pulmonary Fibrosis. Tissue Eng. Part B Rev. 2022, 28, 517–530. [Google Scholar] [CrossRef]
- Wang, D.; Zhu, Y.; Huang, Y.; Zhu, J.; Zhu, B.; Zhao, Y.; Lu, Y.; Wang, Z.; Guo, Y. Pancreatic Extracellular Matrix/Alginate Hydrogels Provide a Supportive Microenvironment for Insulin-Producing Cells. ACS Biomater. Sci. Eng. 2021, 7, 3793–3805. [Google Scholar] [CrossRef]
- Zhu, S.; Chen, P.; Chen, Y.; Li, M.; Chen, C.; Lu, H. 3D-Printed Extracellular Matrix/Polyethylene Glycol Diacrylate Hydrogel Incorporating the Anti-inflammatory Phytomolecule Honokiol for Regeneration of Osteochondral Defects. Am. J. Sports Med. 2020, 48, 2808–2818. [Google Scholar] [CrossRef]
- Crapo, P.M.; Gilbert, T.W.; Badylak, S.F. An overview of tissue and whole organ decellularization processes. Biomaterials 2011, 32, 3233–3243. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, Y.; Cui, K.; Guo, Y.; Zhang, X.; Qin, J. Advances in Hydrogels in Organoids and Organs-on-a-Chip. Adv. Mater. 2019, 31, e1902042. [Google Scholar] [CrossRef] [PubMed]
| Product | Benefits | Disadvantages/Challenges |
|---|---|---|
| Lipofilling | Large quantities; easy procedure; limited patient morbidity | Autologous; intra-operative procedure warranted; limited availability slim persons; fat graft retention unclear; influential intra-personal factors; specialized medicine |
| Supplemented Lipofilling | Large quantities; easy procedure; limited patient morbidity; possibly better fat graft retention | Autologous; intra-operative procedure warranted; limited availability slim persons; fat graft retention unclear; influential intra-personal factors; specialized medicine |
| Tissue-SVF | Fast, easy, cheap procedure; obtained 1. intra-operatively or 2. from donor tissue; application possibilities for small or medium sizes with high regenerative potential; dose-changeable | Autologous; quantity limited to available adipose tissue; mass cell death after defrosting; specialized medicine |
| Cellular-SVF | Application possibilities for small or medium sizes with high regenerative potential; dose-changeable | Autologous; expensive, long procedure; chemical use; the need for lab facility, equipment; mass cell death after defrosting; storage; specialized medicine |
| ASC | Multipotent cell differentiation; high regenerative potential; dose-changeable | Autologous; governmental approval; obtained 1. intra-operatively or 2. from donor tissue or lab culturing; the need for lab facility and equipment; mass cell death after defrosting; storage; specialized medicine |
| ASC Secretome | Allogenic; dose-changeable; prolonged preservation; various medical indications; no patient morbidity; application possibilities of small, medium, and large sizes | Governmental approval; obtained 1. intra-operatively or 2. from donor tissue or lab culturing; the need for lab facility and equipment; experimental setting |
| ECM hydrogels | Allogenic; dose-changeable; unlimited preservation; various medical indications; no patient morbidity; application possibilities of small, medium, and large sizes; availability in a large group of patients; specialized medicine and GP | Scalability; governmental approval; indications and therapeutic application not yet determined; experimental setting |
| ECM hydrogels + ASC secretome | Allogenic; dose-changeable; unlimited preservation; various medical indications; no patient morbidity; application possibilities of small, medium, and large sizes; availability in a large group of patients; specialized medicine and GP | Scalability; governmental approval; Indications and therapeutic application not yet determined; experimental setting |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vriend, L.; van der Lei, B.; Harmsen, M.C.; van Dongen, J.A. Adipose Tissue-Derived Components: From Cells to Tissue Glue to Treat Dermal Damage. Bioengineering 2023, 10, 328. https://doi.org/10.3390/bioengineering10030328
Vriend L, van der Lei B, Harmsen MC, van Dongen JA. Adipose Tissue-Derived Components: From Cells to Tissue Glue to Treat Dermal Damage. Bioengineering. 2023; 10(3):328. https://doi.org/10.3390/bioengineering10030328
Chicago/Turabian StyleVriend, Linda, Berend van der Lei, Martin C. Harmsen, and Joris A. van Dongen. 2023. "Adipose Tissue-Derived Components: From Cells to Tissue Glue to Treat Dermal Damage" Bioengineering 10, no. 3: 328. https://doi.org/10.3390/bioengineering10030328
APA StyleVriend, L., van der Lei, B., Harmsen, M. C., & van Dongen, J. A. (2023). Adipose Tissue-Derived Components: From Cells to Tissue Glue to Treat Dermal Damage. Bioengineering, 10(3), 328. https://doi.org/10.3390/bioengineering10030328








