Utilization of Tryptophan-like Fluorescence as a Proxy for E. coli Contamination in a Mixed-Land-Use Karst Basin
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
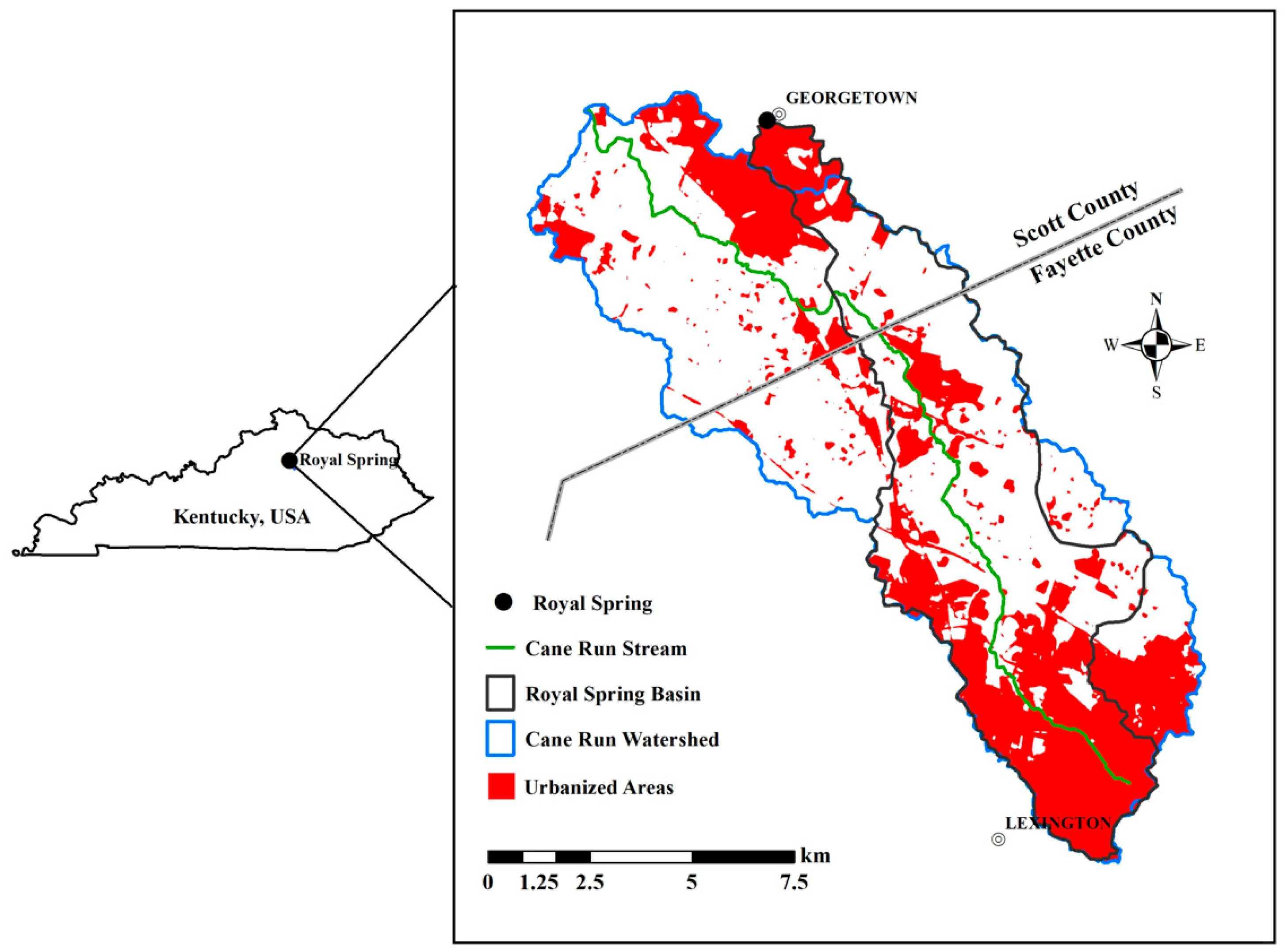
2.2. Data Collection and Processing
2.3. Manual Monitoring and Laboratory Analyses
2.4. Data Sources and Treatment
2.5. Statistical Analysis
3. Results
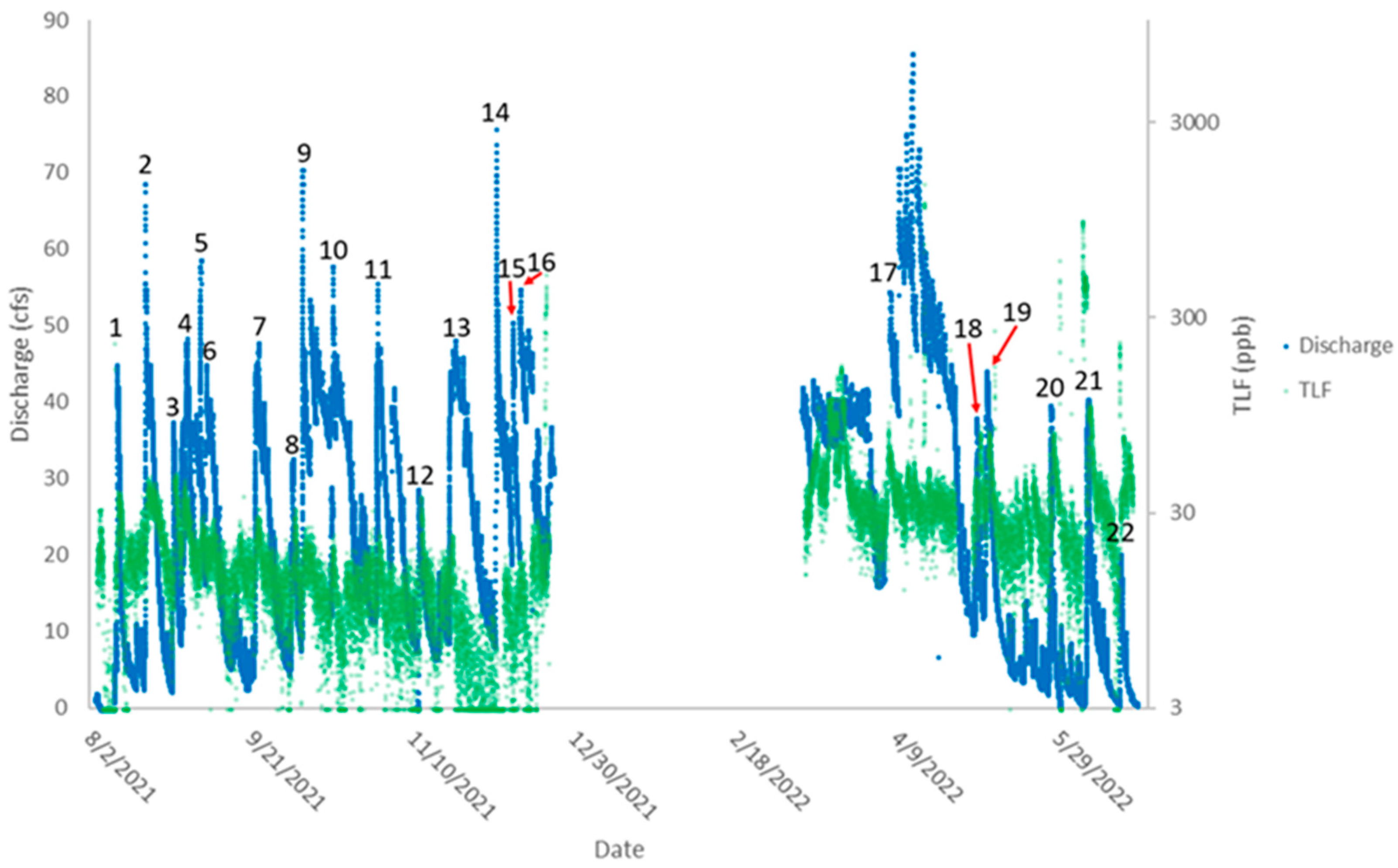
3.1. Precipitation and Discharge
3.2. Relationships of TLF and Turbidity to E. coli
3.3. Linear Regression Modeling and AIC
3.4. TLF and Discharge Peak Timing
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Macler, B.A.; Merkle, J.C. Current knowledge on groundwater microbial pathogens and their control. Hydrogeol. J. 2000, 8, 29–40. [Google Scholar] [CrossRef]
- Ford, D.; Williams, P. Karst Hydrogeology and Geomorphology; John Wiley & Sons: New York, NY, USA, 2007. [Google Scholar]
- Green, R.T.; Painter, S.L.; Sun, A.; Worthington, S.R. Groundwater contamination in karst terranes. Water Air Soil Poll. Focus 2006, 6, 157–170. [Google Scholar] [CrossRef]
- Gunn, J.; Tranter, J.; Perkins, J.; Hunter, C. Sanitary bacterial dynamics in a mixed karst aquifer. In Karst Hydrology; Leibundgut, C., Gunn, J., Dassargues, A., Eds.; International Association of Hydrological Sciences Publication 247, IAHS Press: Wallingford, UK, 1997; pp. 61–70. [Google Scholar]
- Marshall, D.; Brahana, J.V.; Davis, R.K. Resuspension of viable sediment-bound enteric pathogens in shallow karst aquifers. In Gambling with Groundwater—Physical, Chemical and Biological Aspects of Aquifer-Stream Relations; Brahana, J.V., Eckstein, Y., Ongley, L.K., Schneider, R., Moore, J.E., Eds.; American Institute of Hydrology: St. Paul, MN, USA, 1998; pp. 179–186. [Google Scholar]
- Sherer, B.M.; Miner, J.R.; Moore, J.A.; Buckhouse, J.C. Resuspending organisms from a rangeland stream bottom. T. ASAE 1988, 31, 1217–1222. [Google Scholar] [CrossRef]
- Sherer, B.M.; Miner, J.R.; Moore, J.A.; Buckhouse, J.C. Indicator bacterial survival in stream sediments. J. Environ. Qual. 1992, 21, 591–595. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Drinking-Water Quality: Fourth Edition Incorporating the First and Second Addenda; World Health Organization: Geneva, Switzerland, 2022. [Google Scholar]
- Feng, P.; Weagant, S.D.; Grant, M.A.; Burkhardt, W. BAM Chapter 4: Enumeration of Escherichia coli and the coliform bacteria. In Bacteriological Analytical Manual; U.S. Food and Drug Administration: Washington, DC, USA, 2020. [Google Scholar]
- Colilert 18—IDEXX US. Available online: https://www.idexx.com/en/water/water-products-services/colilert-18/ (accessed on 22 February 2023).
- Dorner, S.M.; Anderson, W.B.; Gaulin, T.; Candon, H.L.; Slawson, R.M.; Payment, P.; Huck, P.M. Pathogen and indicator variability in a heavily impacted watershed. J. Water Health 2007, 5, 241–257. [Google Scholar] [CrossRef]
- Vidon, P.; Tedesco, L.P.; Wilson, J.; Campbell, M.A.; Casey, L.R.; Gray, M. Direct and indirect hydrological controls on E. coli concentration and loading in midwestern streams. J. Environ. Qual. 2008, 37, 1761–1768. [Google Scholar] [CrossRef]
- Sorensen, J.P.; Carr, A.F.; Nayebare, J.; Diongue, D.M.; Pouye, A.; Roffo, R.; Gwengweya, G.; Ward, J.S.; Kanoti, J.; Okotto-Okotto, J.; et al. Tryptophan-like and humic-like fluorophores are extracellular in groundwater: Implications as real-time faecal indicators. Sci. Rep. 2020, 10, 15379. [Google Scholar] [CrossRef]
- Sorensen, J.P.; Nayebare, J.; Carr, A.F.; Lyness, R.; Campos, L.C.; Ciric, L.; Goodall, T.; Kulabako, R.; Curran, C.M.; MacDonald, A.M.; et al. In-situ fluorescence spectroscopy is a more rapid and resilient indicator of faecal contamination risk in drinking water than faecal indicator organisms. Water Res. 2021, 206, 117734. [Google Scholar] [CrossRef]
- Cammack, W.K.; Kalff, J.; Prairie, Y.T.; Smith, E.M. Fluorescent dissolved organic matter in lakes: Relationships with heterotrophic metabolism. Limnol. Oceanogr. 2004, 49, 2034–2045. [Google Scholar] [CrossRef]
- Lapworth, D.J.; Gooddy, D.C.; Morris, B.L.; Butcher, A. Investigating groundwater movement and pathogen transport in sandstone aquifers using intrinsic fluorescence spectroscopy. In Proceedings of the Poster Session, Geological Society of London Bicentennial Conference, London, UK, 10–12 September 2007; Available online: https://nora.nerc.ac.uk/id/eprint/13995/1/GS2007.pdf (accessed on 22 February 2023).
- Hudson, N.; Baker, A.; Reynolds, D. Fluorescence analysis of dissolved organic matter in natural, waste and polluted waters—A review. River Res. Appl. 2007, 23, 631–649. [Google Scholar] [CrossRef]
- Hudson, N.; Baker, A.; Ward, D.; Reynolds, D.M.; Brunsdon, C.; Carliell-Marquet, C.; Browning, S. Can fluorescence spectrometry be used as a surrogate for the biochemical oxygen demand (BOD) test in water quality assessment? An example from southwest England. Sci. Total Environ. 2008, 391, 149–158. [Google Scholar] [CrossRef]
- Vucinic, L.; O’Connell, D.; Dubber, D.; Coxon, C.; Gill, L. Multiple fluorescence approaches to identify rapid changes in microbial indicators at karst springs. J. Contam. Hydrol. 2023, 254, 104129. [Google Scholar] [CrossRef]
- Sorensen, J.P.; Baker, A.; Cumberland, S.A.; Lapworth, D.J.; MacDonald, A.M.; Pedley, S.; Taylor, R.G.; Ward, J.S. Real-time detection of faecally contaminated drinking water with tryptophan-like fluorescence: Defining threshold values. Sci. Total Environ. 2018, 622–623, 1250–1257. [Google Scholar] [CrossRef]
- Sorensen, J.; Lapworth, D.; Marchant, B.; Nkhuwa, D.; Pedley, S.; Stuart, M.; Bell, R.; Chirwa, M.; Kabika, J.; Liemisa, M.; et al. In-situ tryptophan-like fluorescence: A real-time indicator of faecal contamination in drinking water supplies. Water Res. 2015, 81, 38–46. [Google Scholar] [CrossRef]
- Sorensen, J.; Vivanco, A.; Ascott, M.; Gooddy, D.; Lapworth, D.; Read, D.; Rushworth, C.; Bucknall, J.; Herbert, K.; Karapanos, I.; et al. Online fluorescence spectroscopy for the real-time evaluation of the microbial quality of drinking water. Water Res. 2018, 137, 301–309. [Google Scholar] [CrossRef]
- Frank, S.; Goeppert, N.; Goldscheider, N. Fluorescence-based multi-parameter approach to characterize dynamics of organic carbon, faecal bacteria and particles at alpine karst springs. Sci. Total Environ. 2018, 615, 1446–1459. [Google Scholar] [CrossRef]
- Ward, J.S.; Lapworth, D.J.; Read, D.S.; Pedley, S.; Banda, S.T.; Monjerezi, M.; Gwengweya, G.; MacDonald, A.M. Tryptophan-like fluorescence as a high-level screening tool for detecting microbial contamination in drinking water. Sci. Total Environ. 2021, 750, 141284. [Google Scholar] [CrossRef]
- Baker, A.; Cumberland, S.A.; Bradley, C.; Buckley, C.; Bridgeman, J. To what extent can portable fluorescence spectroscopy be used in the real-time assessment of microbial water quality? Sci. Total Environ. 2015, 532, 14–19. [Google Scholar] [CrossRef]
- Mendoza, L.M.; Mladenov, N.; Kinoshita, A.M.; Pinongcos, F.; Verbyla, M.E.; Gersberg, R. Fluorescence-based monitoring of anthropogenic pollutant inputs to an urban stream in southern California, USA. Sci. Total Environ. 2020, 718, 137206. [Google Scholar] [CrossRef]
- Nowicki, S.; Lapworth, D.J.; Ward, J.S.; Thomson, P.; Charles, K. Tryptophan-like fluorescence as a measure of microbial contamination risk in groundwater. Sci. Total Environ. 2019, 646, 782–791. [Google Scholar] [CrossRef]
- Ward, J.S.; Lapworth, D.J.; Read, D.S.; Pedley, S.; Banda, S.T.; Monjerezi, M.; Gwengweya, G.; MacDonald, A.M. Large-scale survey of seasonal drinking water quality in Malawi using in situ tryptophan-like fluorescence and conventional water quality indicators. Sci. Total Environ. 2020, 744, 140674. [Google Scholar] [CrossRef] [PubMed]
- Frank, S.; Fahrmeier, N.; Goeppert, N.; Goldscheider, N. High-resolution multi-parameter monitoring of microbial water quality and particles at two alpine karst springs as a basis for an early-warning system. Hydrogeol. J. 2022, 30, 2285–2298. [Google Scholar] [CrossRef]
- Khamis, K.; Sorensen, J.; Bradley, C.; Hannah, D.; Lapworth, D.J.; Stevens, R. In situ tryptophan-like fluorometers: Assessing turbidity and temperature effects for freshwater applications. Environ. Sci. Process. Impacts 2015, 17, 740–752. [Google Scholar] [CrossRef] [PubMed]
- Husic, A.; Fox, J.; Adams, E.; Backus, J.; Pollock, E.; Ford, W.; Agouridis, C. Inland impacts of atmospheric river and tropical cyclone extremes on nitrate transport and stable isotope measurements. Environ. Earth Sci. 2019, 78, 36. [Google Scholar] [CrossRef]
- McFarlan, A.C. Geology of Kentucky; University of Kentucky: Lexington, KY, USA, 1943. [Google Scholar]
- Clepper, M.L. Lithostratigraphic and Paleoenvironmental Framework of the Upper Ordovician Lexington Limestone, Bluegrass Region, Central Kentucky. Doctoral Dissertation, University of Kentucky, Lexington, KY, USA, 2011. [Google Scholar]
- Barton, A.M.; Black Eagle, C.W.; Fryar, A.E. Bourbon and springs in the Inner Bluegrass region of Kentucky. In On and around the Cincinnati Arch and Niagara Escarpment: Geological Field Trips in Ohio and Kentucky for the GSA North-Central Section Meeting, Dayton, Ohio, 2012; Sandy, M.R., Goldman, D., Eds.; Field Guide 27; Geological Society of America: Boulder, CO, USA, 2012; pp. 19–31. [Google Scholar]
- cli-MATE: MRCC Application Tools Environment. Available online: https://mrcc.purdue.edu/CLIMATE/ (accessed on 22 February 2023).
- Source Water Protection. Available online: https://eec.ky.gov/Environmental-Protection/Water/Protection/Pages/SWP.aspx (accessed on 22 February 2023).
- Currens, B.J.; Hall, A.; Brion, G.M.; Fryar, A.E. Use of acetaminophen and sucralose as co-analytes to differentiate sources of human excreta in surface waters. Water Res. 2019, 157, 1–7. [Google Scholar] [CrossRef]
- Bandy, A.M.; Cook, K.; Fryar, A.E.; Zhu, J. Differential transport of Escherichia coli isolates compared to abiotic tracers in a karst aquifer. Groundwater 2020, 58, 70–78. [Google Scholar] [CrossRef]
- Class V Wells for Injection of Non-Hazardous Fluids into or Above Underground Sources of Drinking Water. Available online: https://www.epa.gov/uic/class-v-wells-injection-non-hazardous-fluids-or-above-underground-sources-drinking-water (accessed on 22 February 2023).
- University of Kentucky Department of Biosystems and Agricultural Engineering. Cane Run and Royal Spring Watershed-Based Plan. In Report Prepared for U.S. Environmental Protection Agency under Project Number C9994861-06; University of Kentucky: Lexington, KY, USA, 2016. [Google Scholar]
- Paylor, R.L.; Currens, J.C. Final report: Royal Spring Karst Groundwater Travel Time Investigation. In Report submitted to Georgetown Municipal Water and Sewer Service; Kentucky Geological Survey; University of Kentucky: Lexington, KY, USA, 2004. [Google Scholar]
- Zhu, J.; Currens, J.C.; Dinger, J.S. Challenges of using electrical resistivity method to locate karst conduits—A field case in the Inner Bluegrass region, Kentucky. J. Appl. Geophys. 2011, 75, 523–530. [Google Scholar] [CrossRef]
- Sawyer, A.H.; Zhu, J.; Currens, J.C.; Atcher, C.; Binley, A. Time-lapse electrical resistivity imaging of solute transport in a karst conduit. Hydrol. Process. 2015, 29, 4968–4976. [Google Scholar] [CrossRef]
- Tripathi, G.N.; Fryar, A.E. Integrated surface geophysical approach to locate a karst conduit: A case study from Royal Spring basin, Kentucky, USA. J. Nepal Geol. Soc. 2016, 51, 27–37. [Google Scholar] [CrossRef]
- Husic, A.; Fox, J.; Agouridis, C.; Currens, J.; Ford, W.; Taylor, C. Sediment carbon fate in phreatic karst (Part 1): Conceptual model development. J. Hydrol. 2017, 549, 179–193. [Google Scholar] [CrossRef]
- Younan, L. (Turner Designs, Sunnyvale, CA, USA). Personal communication, 2021.
- Younan, L. (Turner Designs, Sunnyvale, CA, USA). Personal communication, 2022.
- Gentry, R.W.; McCarthy, J.; Layton, A.; McKay, L.D.; Williams, D.; Koirala, S.R.; Sayler, G.S. Escherichia coli loading at or near base flow in a mixed-use watershed. J. Environ. Qual. 2006, 35, 2244–2249. [Google Scholar] [CrossRef]
- Buckerfield, S.J.; Quilliam, R.S.; Waldron, S.; Naylor, L.A.; Li, S.; Oliver, D.M. Rainfall-driven E. coli transfer to the stream-conduit network observed through increasing spatial scales in mixed land-use paddy farming karst terrain. Water Res. X 2019, 5, 100038. [Google Scholar] [CrossRef]
- Royal Springs at Georgetown, KY. Available online: https://waterdata.usgs.gov/monitoring-location/03288110/ (accessed on 22 February 2023).
- Behrouj-Peely, A.; Mohammadi, Z.; Scheiber, L.; Vázquez-Suñé, E. An integrated approach to estimate the mixing ratios in a karst system under different hydrogeological conditions. J. Hydrol. Reg. Stud. 2020, 30, 100693. [Google Scholar] [CrossRef]
- Tobin, B.W.; Polk, J.S.; Arpin, S.M.; Shelley, A.; Taylor, C. A conceptual model of epikarst processes across sites, seasons, and storm events. J. Hydrol. 2021, 596, 125692. [Google Scholar] [CrossRef]
- Anderson, D.R.; Burnham, K.P. Avoiding pitfalls when using information-theoretic methods. J. Wildlife Manage. 2002, 66, 912–918. [Google Scholar] [CrossRef]
- Brion, G.M. (Department of Civil Engineering, University of Kentucky, Lexington, KY, USA). Personal communication, 2022.
- R Core Team. R: A Language and Environment for Statistical Computing, Version 4.1.2; R Foundation for Statistical Computing: Vienna, Austria, 2021.
- Dapkus, R. Tryptophan-Like Fluorescence and Non-Point Source Pollution in Karst Basins, Inner Bluegrass Region, Kentucky. Master’s Thesis, University of Kentucky, Lexington, KY, USA, 2022. [Google Scholar]
- Bettel, L.; Fox, J.; Husic, A.; Zhu, J.; Al Aamery, N.; Mahoney, T.; Gold-McCoy, A. Sediment transport investigation in a karst aquifer hypothesizes controls on internal versus external sediment origin and saturation impact on hysteresis. J. Hydrol. 2022, 613, 128391. [Google Scholar] [CrossRef]

| Variable | Data Source |
|---|---|
| E. coli | IDEXX analyses |
| TLF | 7F and C3 fluorometers |
| Turbidity | YSI multiparameter sonde and smart sensor |
| Instrument effect | 7F or C3 fluorometer |
| Air temperature | Blue Grass Airport [35] |
| 48 h antecedent precipitation | Blue Grass Airport [35] |
| Model | Model Parameters | AICc | Delta AIC | Weight |
|---|---|---|---|---|
| 1 | Ecoli = f(TLF * Instrument) | 991.8 | 11.54 | 0.002 |
| 2 | Ecoli = f(TLF * Inst + rain_48 h) | 980.3 | 0 | 0.481 |
| 3 | Ecoli = f(turbidity) | 1011.8 | 31.45 | 0 |
| 4 | Ecoli = f(rain_48 h) | 983.4 | 3.13 | 0.101 |
| 5 | Ecoli = f(turb + rain_48 h) | 985.6 | 5.28 | 0.034 |
| 6 | Ecoli = f(TLF) | 1013.6 | 33.26 | 0 |
| 7 | Ecoli = f(airT + rain_48 h) | 982.2 | 1.86 | 0.190 |
| 8 | Ecoli = f(TLF * Inst + rain_48 h + turb + airT) | 982.1 | 1.84 | 0.192 |
| Model | Intercept | TLF | Instrument | rain_48 h | turb | airT | TLF * Inst | F-Statistic | r2 |
|---|---|---|---|---|---|---|---|---|---|
| 2 | −194.76 | 14.44 | −350.44 | 888.09 | N/A | N/A | 47.62 | 17.61 | 0.52 |
| 8 | −971.38 | 17.49 | −120.3 | 936.78 | −28.21 | 11.36 | 41.39 | 12.46 | 0.53 |
| 7 | −713.19 | N/A | N/A | 1258.92 | N/A | 14.95 | N/A | 29.71 | 0.48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dapkus, R.T.; Fryar, A.E.; Tobin, B.W.; Byrne, D.M.; Sarker, S.K.; Bettel, L.; Fox, J.F. Utilization of Tryptophan-like Fluorescence as a Proxy for E. coli Contamination in a Mixed-Land-Use Karst Basin. Hydrology 2023, 10, 74. https://doi.org/10.3390/hydrology10040074
Dapkus RT, Fryar AE, Tobin BW, Byrne DM, Sarker SK, Bettel L, Fox JF. Utilization of Tryptophan-like Fluorescence as a Proxy for E. coli Contamination in a Mixed-Land-Use Karst Basin. Hydrology. 2023; 10(4):74. https://doi.org/10.3390/hydrology10040074
Chicago/Turabian StyleDapkus, Ryan T., Alan E. Fryar, Benjamin W. Tobin, Diana M. Byrne, Shishir K. Sarker, Leonie Bettel, and James F. Fox. 2023. "Utilization of Tryptophan-like Fluorescence as a Proxy for E. coli Contamination in a Mixed-Land-Use Karst Basin" Hydrology 10, no. 4: 74. https://doi.org/10.3390/hydrology10040074
APA StyleDapkus, R. T., Fryar, A. E., Tobin, B. W., Byrne, D. M., Sarker, S. K., Bettel, L., & Fox, J. F. (2023). Utilization of Tryptophan-like Fluorescence as a Proxy for E. coli Contamination in a Mixed-Land-Use Karst Basin. Hydrology, 10(4), 74. https://doi.org/10.3390/hydrology10040074








