Facile Lignin Extraction and Application as Natural UV Blockers in Cosmetic Formulations
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
2.2. Lignin Preparation
2.3. Characterization of Lignin
2.4. Preparation and UV Absorption Measurement of Creams with Added Lignin
2.5. UV Irradiation Effects
2.6. Solar Radiation Effects
3. Results and Discussion
3.1. Characteristics of the Obtained Lignin Samples
3.2. Ultraviolet Absorption Potential of Lignin
3.3. Assessing the UV Protection of Lignin in Combination with a Commercial Moisturizing Cream
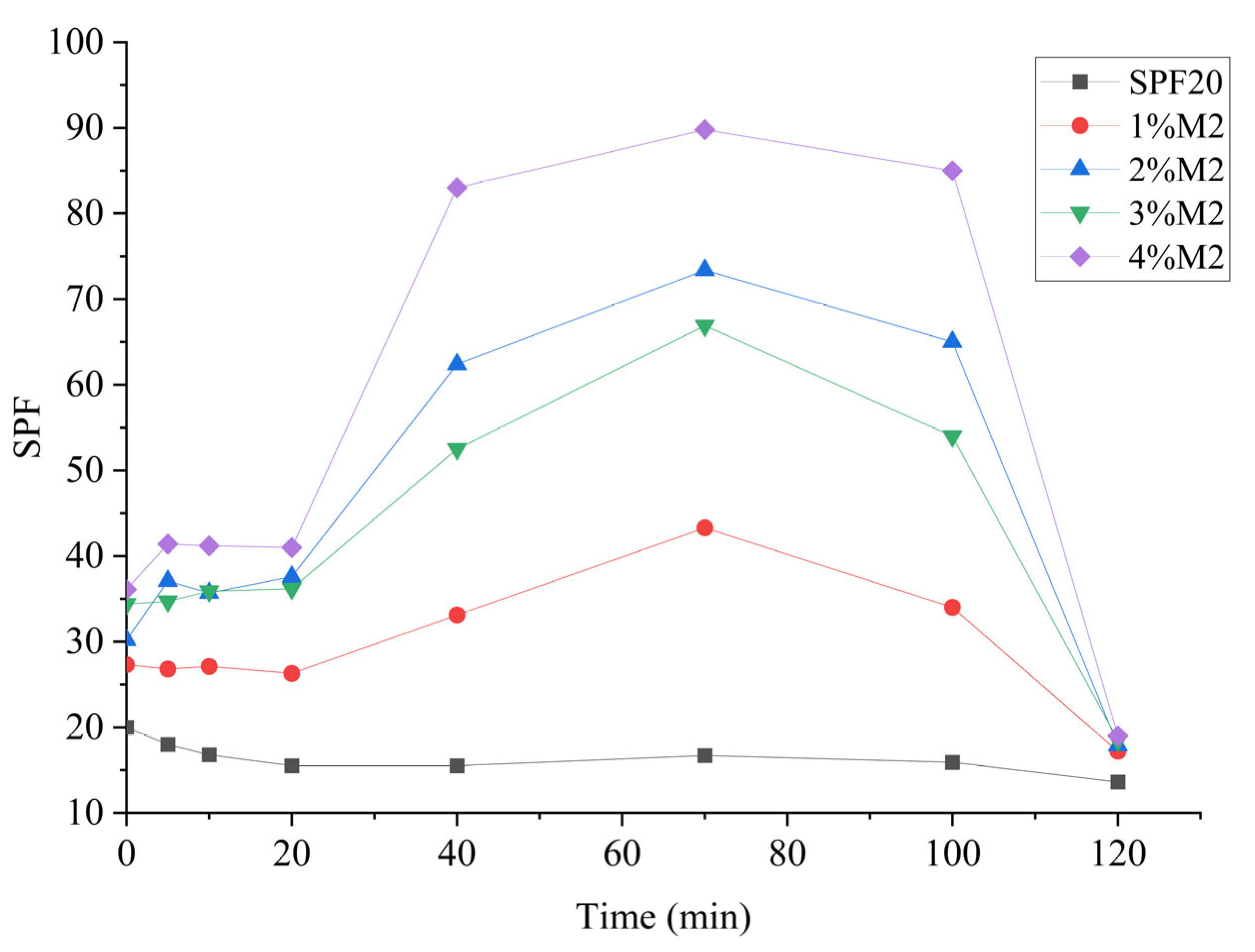
3.4. Mixture Effects of Lignin with Commercial Sunscreen
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hudson, L.; Rashdan, E.; Bonn, C.A.; Chavan, B.; Rawlings, D.; Birch-Machin, M.A. Individual and combined effects of the infrared, visible, and ultraviolet light components of solar radiation on damage biomarkers in human skin cells. FASEB J. 2020, 34, 3874–3883. [Google Scholar] [CrossRef]
- Hirst, N.G.; Gordon, L.G.; Scuffham, P.A.; Green, A.C. Lifetime cost-effectiveness of skin cancer prevention through promotion of daily sunscreen use. Value Health 2012, 15, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Lyons, A.B.; Trullas, C.; Kohli, I.; Hamzavi, I.H.; Lim, H.W. Photoprotection beyond ultraviolet radiation: A review of tinted sunscreens. J. Am. Acad. Dermatol. 2021, 84, 1393–1397. [Google Scholar] [CrossRef] [PubMed]
- Diaz, J.H.; Nesbitt, L.T., Jr. Sun exposure behavior and protection: Recommendations for travelers. J. Travel Med. 2013, 20, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Beisl, S.; Friedl, A.; Miltner, A. Lignin from Micro- to Nanosize: Applications. Int. J. Mol. Sci. 2017, 18, 2367. [Google Scholar] [CrossRef] [PubMed]
- Gause, S.; Chauhan, A. UV-blocking potential of oils and juices. Int. J. Cosmet. Sci. 2016, 38, 354–363. [Google Scholar] [CrossRef]
- Rabinovich, L.; Kazlouskaya, V. Herbal sun protection agents: Human studies. Clin. Dermatol. 2018, 36, 369–375. [Google Scholar] [CrossRef]
- Butt, S.T.; Christensen, T. Toxicity and Phototoxicity of Chemical Sun Filters. Radiat. Prot. Dosim. 2000, 91, 283–286. [Google Scholar] [CrossRef]
- Corinaldesi, C.; Marcellini, F.; Nepote, E.; Damiani, E.; Danovaro, R. Impact of inorganic UV filters contained in sunscreen products on tropical stony corals (Acropora spp.). Sci. Total Environ. 2018, 637–638, 1279–1285. [Google Scholar] [CrossRef]
- Norgren, M.; Edlund, H. Lignin: Recent advances and emerging applications. Curr. Opin. Colloid Interface Sci. 2014, 19, 409–416. [Google Scholar] [CrossRef]
- Shankar, S.; Rhim, J.W.; Won, K. Preparation of poly(lactide)/lignin/silver nanoparticles composite films with UV light barrier and antibacterial properties. Int. J. Biol. Macromol. 2018, 107, 1724–1731. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Tian, D.; Shen, F.; Yang, G.; Long, L.; He, J.; Song, C.; Zhang, J.; Zhu, Y.; Huang, C.; et al. Transparent Cellulose/Technical Lignin Composite Films for Advanced Packaging. Polymers 2019, 11, 1455. [Google Scholar] [CrossRef] [PubMed]
- Tortora, M.; Cavalieri, F.; Mosesso, P.; Ciaffardini, F.; Melone, F.; Crestini, C. Ultrasound driven assembly of lignin into microcapsules for storage and delivery of hydrophobic molecules. Biomacromolecules 2014, 15, 1634–1643. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Qiu, X.; Zhu, S. Lignin: A nature-inspired sun blocker for broad-spectrum sunscreens. Green Chem. 2015, 17, 320–324. [Google Scholar] [CrossRef]
- Qian, Y.; Qiu, X.; Zhu, S. Sunscreen Performance of Lignin from Different Technical Resources and Their General Synergistic Effect with Synthetic Sunscreens. ACS Sustain. Chem. Eng. 2016, 4, 4029–4035. [Google Scholar] [CrossRef]
- Lee, S.C.; Tran, T.M.T.; Choi, J.W.; Won, K. Lignin for white natural sunscreens. Int. J. Biol. Macromol. 2019, 122, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Yoo, E.; Lee, S.H.; Won, K. Preparation and Application of Light-Colored Lignin Nanoparticles for Broad-Spectrum Sunscreens. Polymers 2020, 12, 699. [Google Scholar] [CrossRef] [PubMed]
- Kai, D.; Tan, M.J.; Chee, P.L.; Chua, Y.K.; Yap, Y.L.; Loh, X.J. Towards lignin-based functional materials in a sustainable world. Green Chem. 2016, 18, 1175–1200. [Google Scholar] [CrossRef]
- Wang, J.; Deng, Y.; Qian, Y.; Qiu, X.; Ren, Y.; Yang, D. Reduction of lignin color via one-step UV irradiation. Green Chem. 2016, 18, 695–699. [Google Scholar] [CrossRef]
- Tran, M.H.; Phan, D.-P.; Lee, E.Y. Review on lignin modifications toward natural UV protection ingredient for lignin-based sunscreens. Green Chem. 2021, 23, 4633–4646. [Google Scholar] [CrossRef]
- Yao, C.; Yongming, F.; Jianmin, G.; Houkun, L. Coloring characteristics of in situ lignin during heat treatment. Wood Sci. Technol. 2010, 46, 33–40. [Google Scholar] [CrossRef]
- Qiu, X.; Yu, J.; Yang, D.; Wang, J.; Mo, W.; Qian, Y. Whitening Sulfonated Alkali Lignin via H2O2/UV Radiation and Its Application As Dye Dispersant. ACS Sustain. Chem. Eng. 2017, 6, 1055–1060. [Google Scholar] [CrossRef]
- Zhang, H.; Bai, Y.; Yu, B.; Liu, X.; Chen, F. A practicable process for lignin color reduction: Fractionation of lignin using methanol/water as a solvent. Green Chem. 2017, 19, 5152–5162. [Google Scholar] [CrossRef]
- Zhang, H.; Bai, Y.; Zhou, W.; Chen, F. Color reduction of sulfonated eucalyptus kraft lignin. Int. J. Biol. Macromol. 2017, 97, 201–208. [Google Scholar] [CrossRef]
- Duy, N.V.; Tsygankov, P.Y.; Menshutina, N.V. Hybrid Aerogels Based on Lignin, Derived from Vegetable Raw Materials. ChemChemTech 2023, 66, 75–83. [Google Scholar] [CrossRef]
- Reddy, N. Sustainable Applications of Coir and Other Coconut By-Products; Springer International Publishing: Cham, Switzerland, 2019; p. 63. [Google Scholar] [CrossRef]
- Serrano, L.; Esakkimuthu, E.S.; Marlin, N.; Brochier-Salon, M.-C.; Mortha, G.; Bertaud, F. Fast, Easy, and Economical Quantification of Lignin Phenolic Hydroxyl Groups: Comparison with Classical Techniques. Energy Fuels 2018, 32, 5969–5977. [Google Scholar] [CrossRef]
- Solihat, N.N.; Santoso, E.B.; Karimah, A.; Madyaratri, E.W.; Sari, F.P.; Falah, F.; Iswanto, A.H.; Ismayati, M.; Lubis, M.A.R.; Fatriasari, W.; et al. Physical and Chemical Properties of Acacia mangium Lignin Isolated from Pulp Mill Byproduct for Potential Application in Wood Composites. Polymers 2022, 14, 491. [Google Scholar] [CrossRef] [PubMed]
- Wendel, V.; Uhlmann, B.; Mann, T.; Klette, E. In Vitro Test Method to Assess the UVA Protection Performance of Sun Care Products. European Patent EP1291640A1, 12 March 2003. [Google Scholar]
- ISO 24444:2019; Cosmetics Sun Protection Test Methods In Vivo Determination of the Sun Protection Factor (SPF). International Organization for Standardization: Geneva, Switzerland, 2019.
- Gers-Barlag, H.; Klette, E.; Bimczok, R.; Springob, C.; Finkel, P.; Rudolph, T.; Gonzenbach, H.U.; Schneider, P.H.; Kockott, D.; Heinrich, U.; et al. In vitro testing to assess the UVA protection performance of sun care products. Int. J. Cosmet. Sci. 2001, 23, 3–14. [Google Scholar] [CrossRef]
- Ferrero, L.; Pissavini, M.; Marguerie, S.; Zastrow, L. Sunscreen in vitro spectroscopy: Application to UVA protection assessment and correlation with in vivo persistent pigment darkening. Int. J. Cosmet. Sci. 2002, 24, 63–70. [Google Scholar] [CrossRef]
- Constant, S.; Wienk, H.L.J.; Frissen, A.E.; Peinder, P.d.; Boelens, R.; van Es, D.S.; Grisel, R.J.H.; Weckhuysen, B.M.; Huijgen, W.J.J.; Gosselink, R.J.A.; et al. New insights into the structure and composition of technical lignins: A comparative characterisation study. Green Chem. 2016, 18, 2651–2665. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, L.; Wang, D.; Li, D. Structure elucidation and properties of different lignins isolated from acorn shell of Quercus variabilis Bl. Int. J. Biol. Macromol. 2018, 107, 1193–1202. [Google Scholar] [CrossRef] [PubMed]
- Farid, T.; Wang, Y.; Rafiq, M.I.; Ali, A.; Tang, W. Porous flexible wood scaffolds designed for high-performance electrochemical energy storage. ACS Sustain. Chem. Eng. 2022, 10, 7078–7090. [Google Scholar] [CrossRef]
- Song, Y.; Ji, H.; Shi, X.; Yang, X.; Zhang, X. Successive organic solvent fractionation and structural characterization of lignin extracted from hybrid poplar by deep eutectic solvent for improving the homogeneity and isolating narrow fractions. Renew. Energy 2020, 157, 1025–1034. [Google Scholar] [CrossRef]
- Su, X.; Fu, Y.; Shao, Z.; Qin, M.; Li, X.; Zhang, F. Light-colored lignin isolated from poplar by ultrasound-assisted ethanol extraction: Structural features and anti-ultraviolet and anti-oxidation activities. Ind. Crops Prod. 2022, 176, 114359. [Google Scholar] [CrossRef]
- Singh, S.K.; Dhepe, P.L. Isolation of lignin by organosolv process from different varieties of rice husk: Understanding their physical and chemical properties. Bioresour. Technol. 2016, 221, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Bock, P.; Nousiainen, P.; Elder, T.; Blaukopf, M.; Amer, H.; Zirbs, R.; Potthast, A.; Gierlinger, N. Infrared and Raman spectra of lignin substructures: Dibenzodioxocin. J. Raman Spectrosc. 2020, 51, 422–431. [Google Scholar] [CrossRef] [PubMed]
- Melro, E.; Filipe, A.; Sousa, D.; Medronho, B.; Romano, A. Revisiting lignin: A tour through its structural features, characterization methods and applications. New J. Chem. 2021, 45, 6986–7013. [Google Scholar] [CrossRef]
- Deepa, A.K.; Dhepe, P.L. Lignin depolymerization into aromatic monomers over solid acid catalysts. ACS Catal. 2015, 5, 365–379. [Google Scholar] [CrossRef]
- Li, L.; Wu, Z.; Xi, X.; Liu, B.; Cao, Y.; Xu, H.; Hu, Y. A bifunctional brønsted acidic deep eutectic solvent to dissolve and catalyze the depolymerization of alkali lignin. J. Renew. Mater. 2021, 9, 219–235. [Google Scholar] [CrossRef]
- Rashid, T.; Kait, C.F.; Regupathi, I.; Murugesan, T. Dissolution of kraft lignin using Protic Ionic Liquids and characterization. Ind. Crops Prod. 2016, 84, 284–293. [Google Scholar] [CrossRef]
- Abd Latif, N.H.; Brosse, N.; Ziegler-Devin, I.; Chrusiel, L.; Hashim, R.; Hussin, M.H. Structural characterization of modified coconut husk lignin via steam explosion pretreatment as a renewable phenol substitutes. Int. J. Biol. Macromol. 2023, 253, 127210. [Google Scholar] [CrossRef]
- Cassales, A.; Ramos, L.A.; Frollini, E. Synthesis of bio-based polyurethanes from Kraft lignin and castor oil with simultaneous film formation. Int. J. Biol. Macromol. 2020, 145, 28–41. [Google Scholar] [CrossRef]
- Kim, D.; Cheon, J.; Kim, J.; Hwang, D.; Hong, I.; Kwon, O.H.; Park, W.H.; Cho, D. Extraction and characterization of lignin from black liquor and preparation of biomass-based activated carbon there-from. Carbon Lett. 2017, 22, 81–88. [Google Scholar] [CrossRef]
- Mankar, A.R.; Pandey, A.; Pant, K. Microwave-assisted extraction of lignin from coconut coir using deep eutectic solvents and its valorization to aromatics. Bioresour. Technol. 2022, 345, 126528. [Google Scholar] [CrossRef] [PubMed]
- Oruganti, R.K.; Sunar, S.L.; Panda, T.K.; Shee, D.; Bhattacharyya, D. Kraft lignin recovery from de-oiled Jatropha curcas seed by potassium hydroxide pretreatment and optimization using response surface methodology. Bioresour. Technol. Rep. 2023, 23, 101572. [Google Scholar] [CrossRef]
- Nivedha, M.; Manisha, M.; Gopinath, M.; Baskar, G.; Tamilarasan, K. Fractionation, characterization, and economic evaluation of alkali lignin from saw industry waste. Bioresour. Technol. 2021, 335, 125260. [Google Scholar] [CrossRef]
- Dhara, S.; Samanta, N.S.; Uppaluri, R.; Purkait, M. High-purity alkaline lignin extraction from Saccharum ravannae and optimization of lignin recovery through response surface methodology. Int. J. Biol. Macromol. 2023, 234, 123594. [Google Scholar] [CrossRef] [PubMed]
- Alzagameem, A.; Khaldi-Hansen, B.E.; Buchner, D.; Larkins, M.; Kamm, B.; Witzleben, S.; Schulze, M. Lignocellulosic Biomass as Source for Lignin-Based Environmentally Benign Antioxidants. Molecules 2018, 23, 2664. [Google Scholar] [CrossRef]
- Rumpf, J.; Do, X.T.; Burger, R.; Monakhova, Y.B.; Schulze, M. Extraction of High-Purity Lignins via Catalyst-free Organosolv Pulping from Low-Input Crops. Biomacromolecules 2020, 21, 1929–1942. [Google Scholar] [CrossRef]
- Zhang, Y.; Naebe, M. Lignin: A Review on Structure, Properties, and Applications as a Light-Colored UV Absorber. ACS Sustain. Chem. Eng. 2021, 9, 1427–1442. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, X.; Fu, S.; Chen, Y. Fabrication of Light-Colored Lignin Microspheres for Developing Natural Sunscreens with Favorable UV Absorbability and Staining Resistance. Ind. Eng. Chem. Res. 2019, 58, 13858–13867. [Google Scholar] [CrossRef]
- Egambaram, O.P.; Kesavan Pillai, S.; Ray, S.S. Materials Science Challenges in Skin UV Protection: A Review. Photochem. Photobiol. 2020, 96, 779–797. [Google Scholar] [CrossRef] [PubMed]
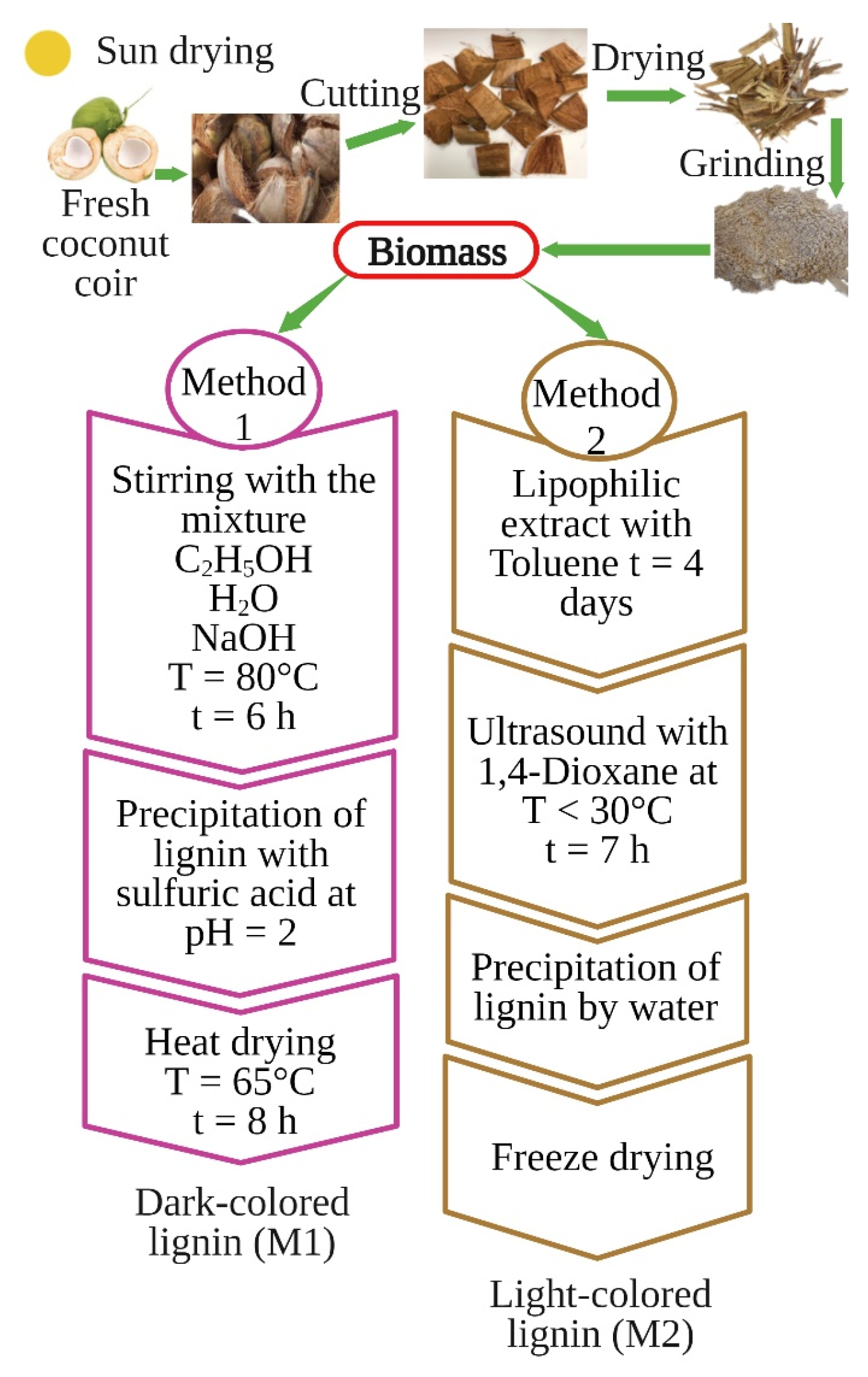



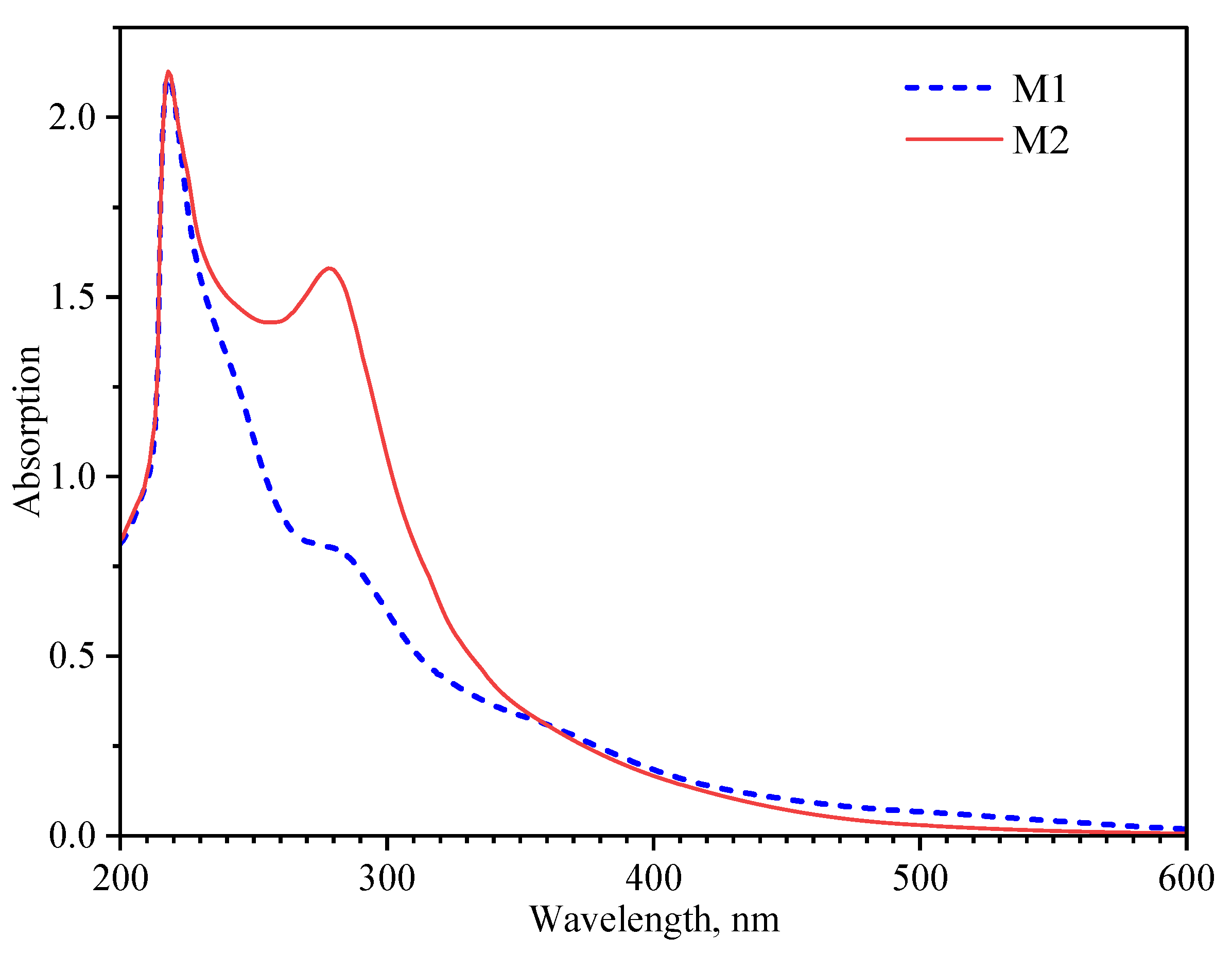
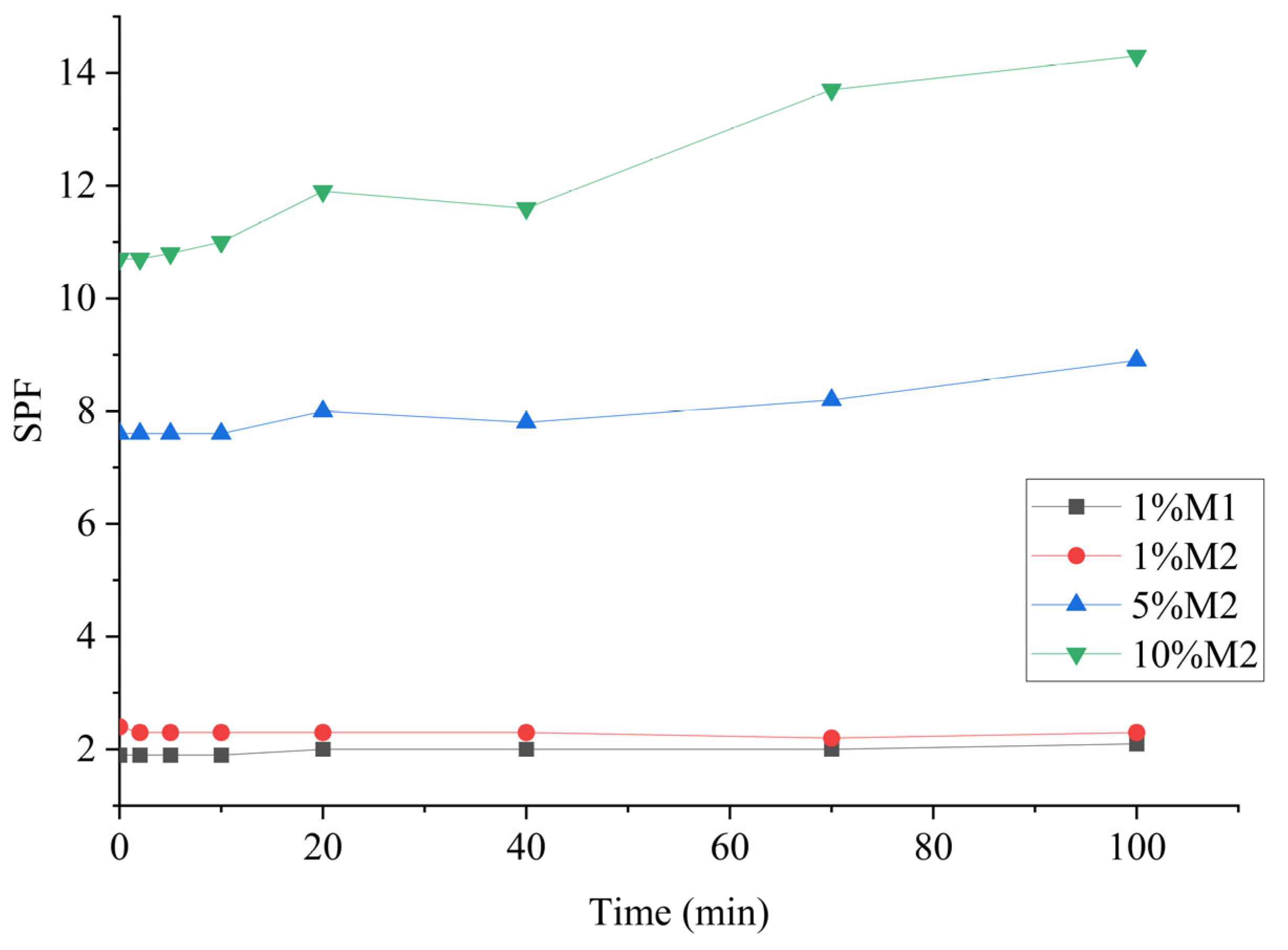
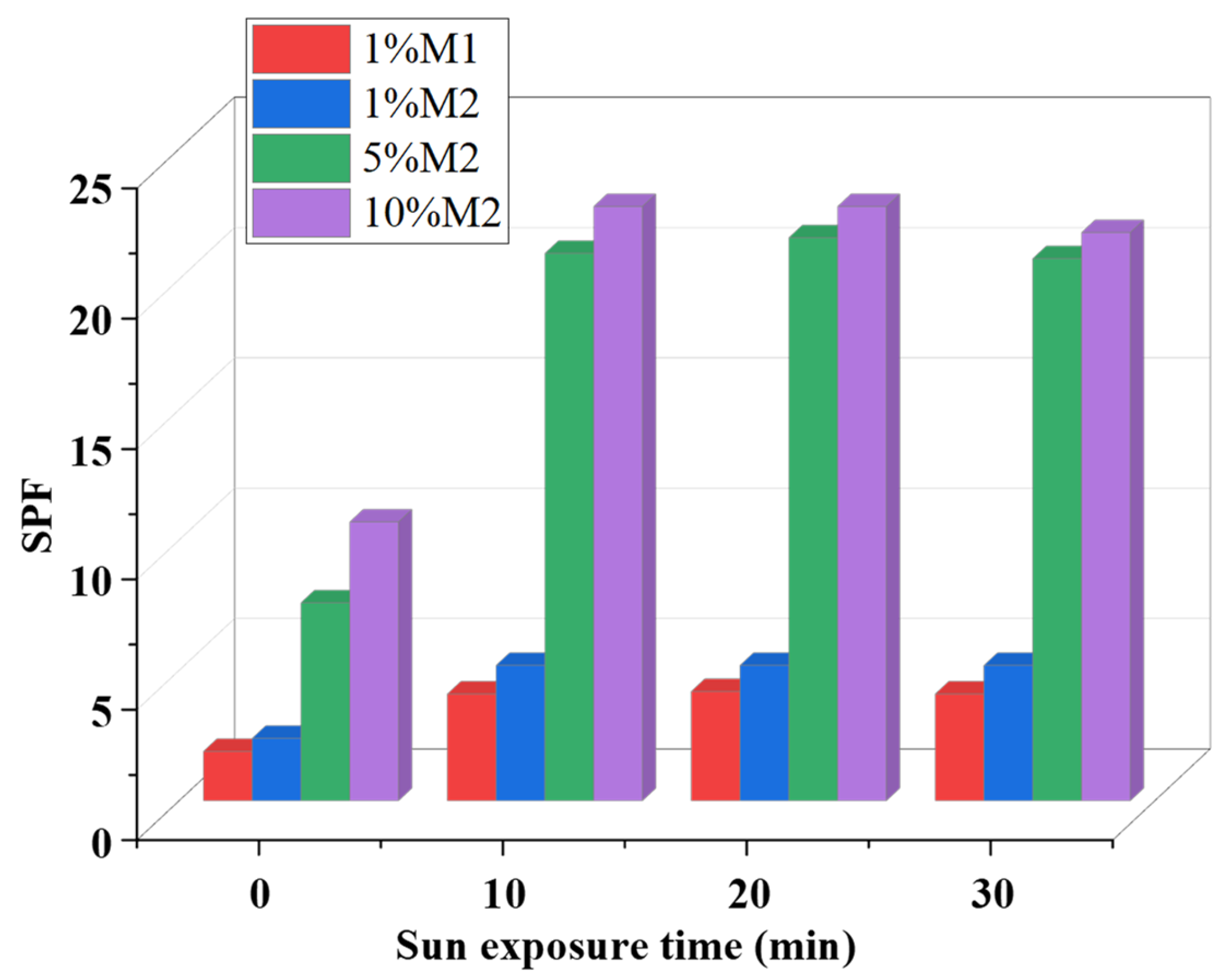

| Sample | Total phOH (mmol/g) |
|---|---|
| M1 | 2.82 ± 0.22 |
| M2 | 3.15 ± 0.10 |
| Lignin | 0% Lignin (wt.%) | 1% Lignin (wt.%) | 5% Lignin (wt.%) | 10% Lignin (wt.%) | |
|---|---|---|---|---|---|
| SPF Value | |||||
| SPF of cream with lignin M1 | 1.1 ± 0.1 1 | 1.9 ± 0.1 | 3.7 ± 0.1 | 8.7 ± 0.3 | |
| SPF of cream with lignin M2 | 2.4 ± 0.1 | 7.6 ± 0.2 | 10.7 ± 0.4 | ||
| Lignin (wt.%) | M1 | M2 | ||
|---|---|---|---|---|
| SPF (290–400 nm) | UVA PF (320–400 nm) | SPF (290–400 nm) | UVA PF (320–400 nm) | |
| 0% | 20.0 ± 0.3 2 | 3.2 ± 0.1 3 | ||
| 1% | 24.6 ± 0.4 | 3.8 ± 0.1 | 27.3 ± 0.5 | 4.0 ± 0.2 |
| 2% | 26.7 ± 0.3 | 4.9 ± 0.2 | 30.2 ± 0.2 | 5.4 ± 0.2 |
| 3% | 29.8 ± 0.6 | 5.6 ± 0.1 | 34.4 ± 0.5 | 6.2 ± 0.1 |
| 4% | 32.7 ± 0.7 | 7.8 ± 0.2 | 36.1 ± 0.8 | 8.9 ± 0.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duy, N.V.; Tsygankov, P.Y.; Menshutina, N.V. Facile Lignin Extraction and Application as Natural UV Blockers in Cosmetic Formulations. ChemEngineering 2024, 8, 69. https://doi.org/10.3390/chemengineering8040069
Duy NV, Tsygankov PY, Menshutina NV. Facile Lignin Extraction and Application as Natural UV Blockers in Cosmetic Formulations. ChemEngineering. 2024; 8(4):69. https://doi.org/10.3390/chemengineering8040069
Chicago/Turabian StyleDuy, Nguyen Van, Pavel Y. Tsygankov, and Natalia V. Menshutina. 2024. "Facile Lignin Extraction and Application as Natural UV Blockers in Cosmetic Formulations" ChemEngineering 8, no. 4: 69. https://doi.org/10.3390/chemengineering8040069
APA StyleDuy, N. V., Tsygankov, P. Y., & Menshutina, N. V. (2024). Facile Lignin Extraction and Application as Natural UV Blockers in Cosmetic Formulations. ChemEngineering, 8(4), 69. https://doi.org/10.3390/chemengineering8040069








