PharmActa: Empowering Patients to Avoid Clinical Significant Drug–Herb Interactions
Abstract
1. Introduction
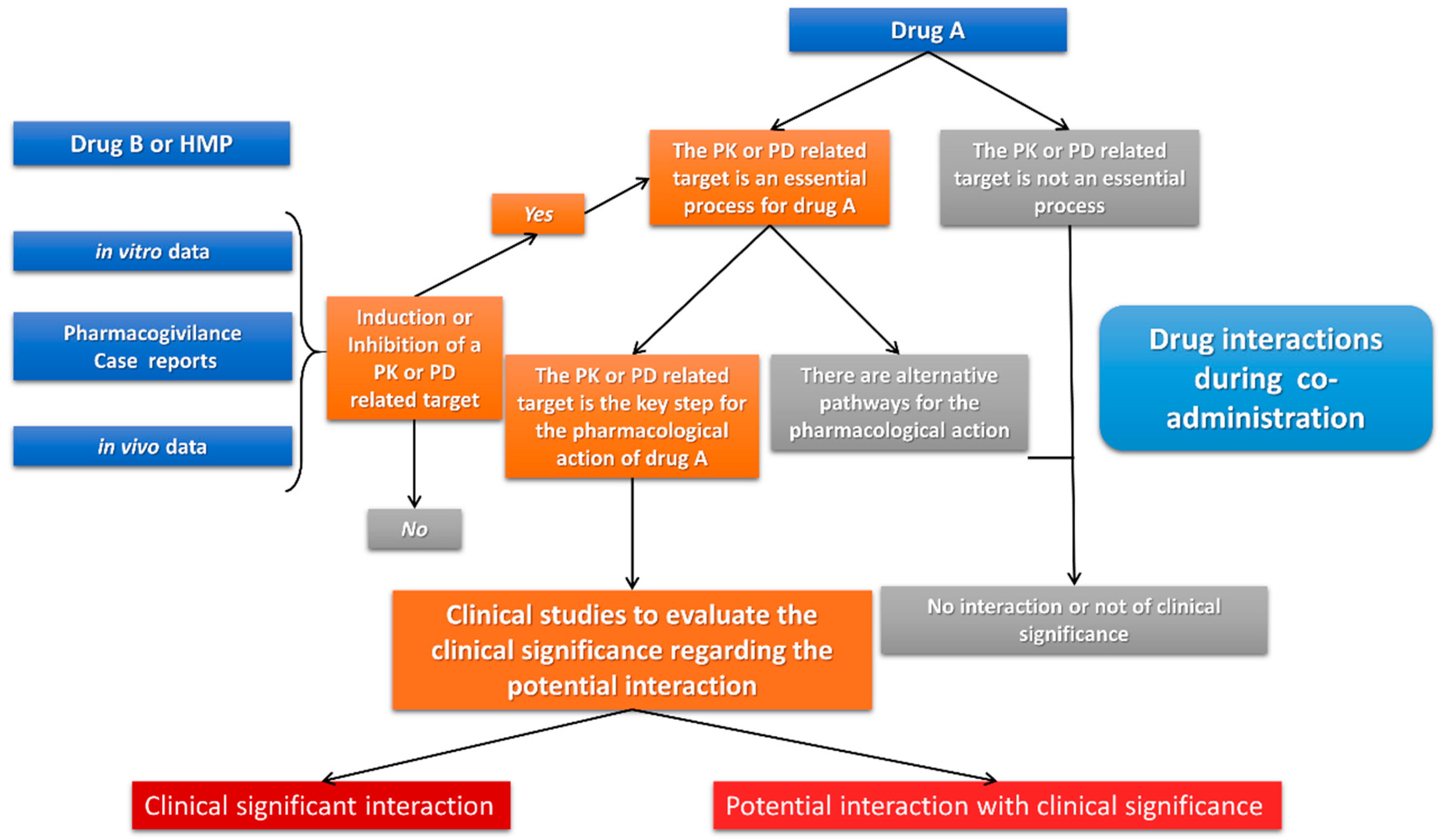
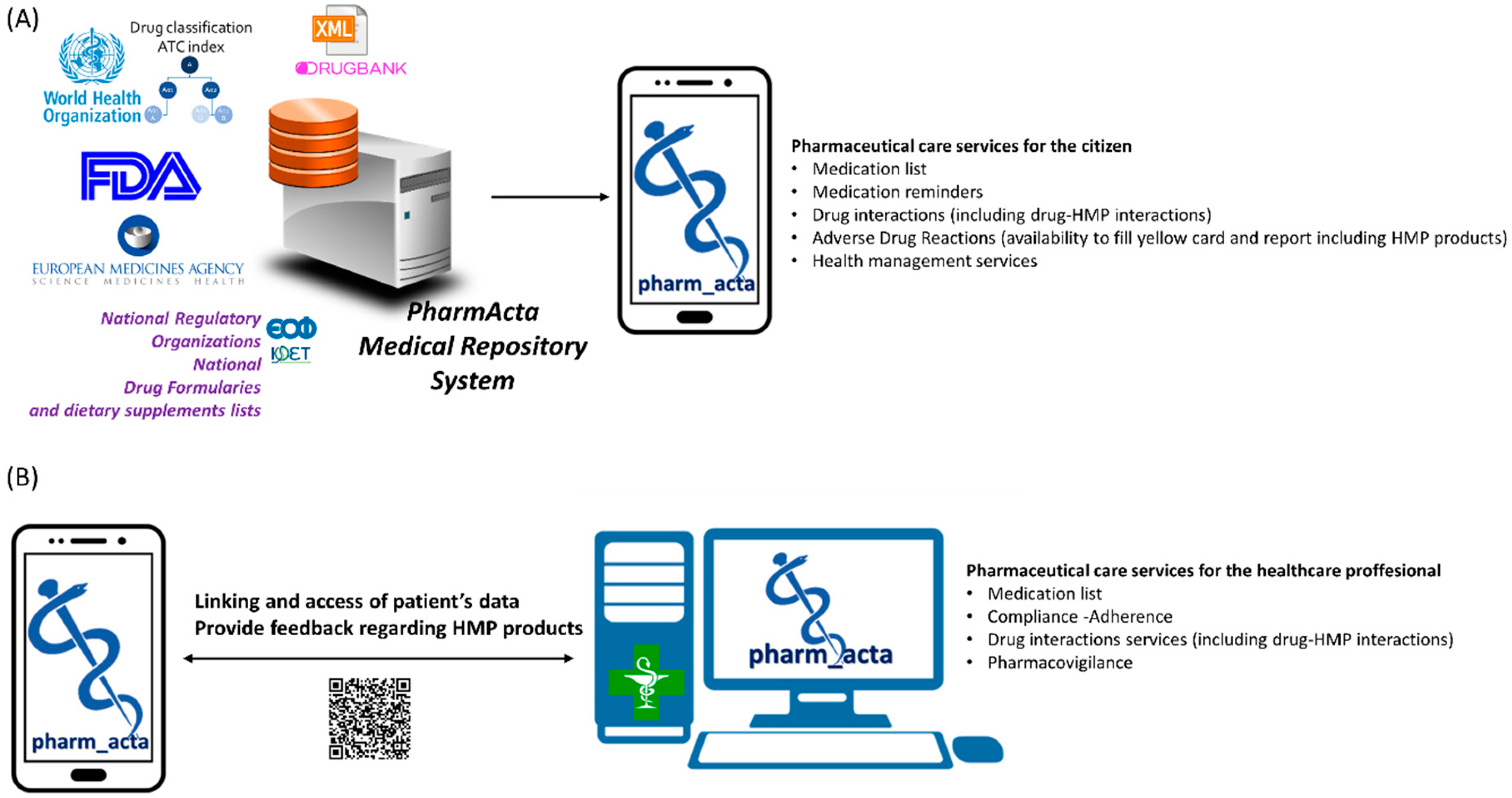
2. Materials and Methods
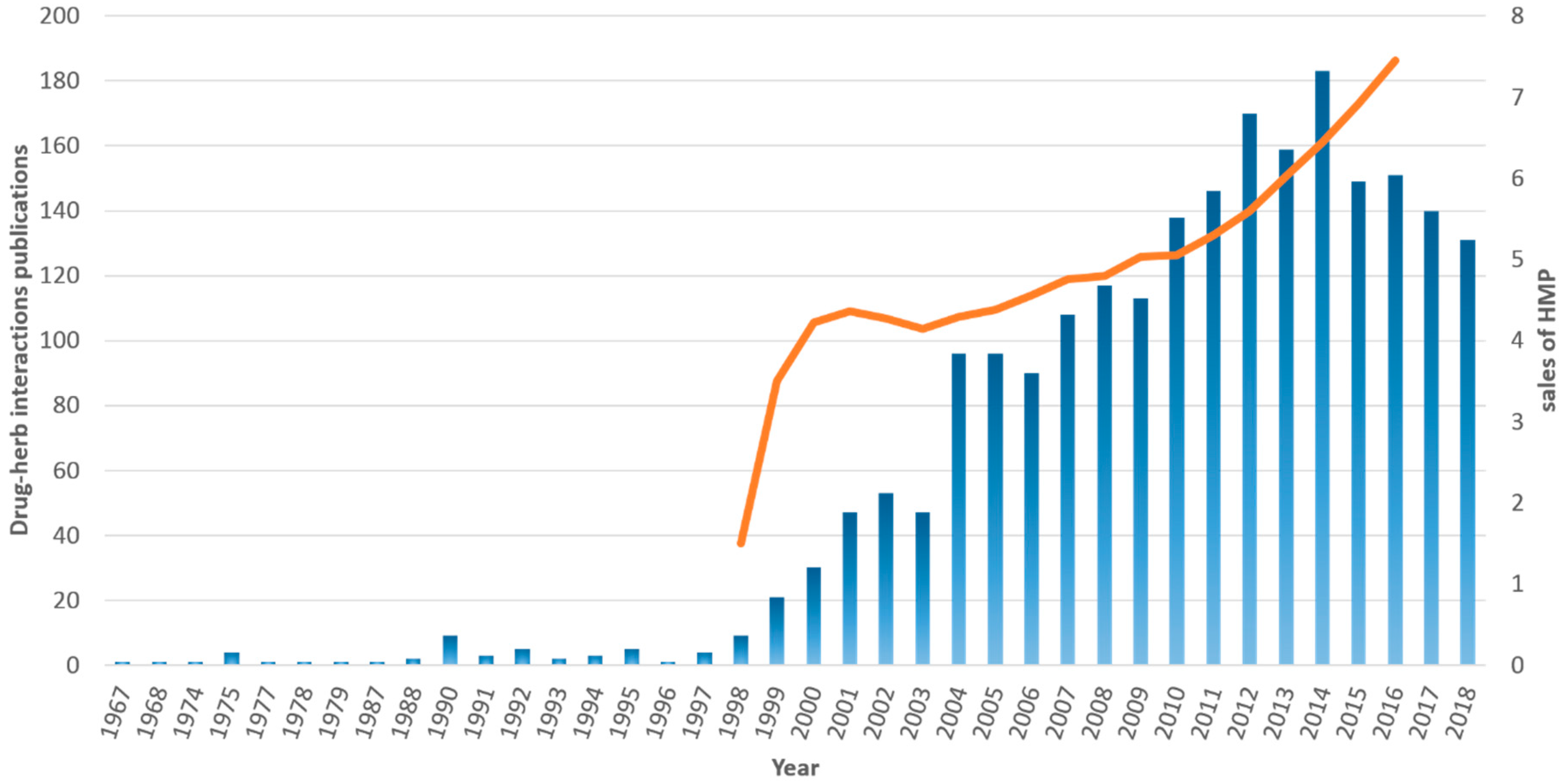
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Chugh, N.A.; Bali, S.; Koul, A. Integration of botanicals in contemporary medicine: Road blocks, checkpoints and go-ahead signals. Integr. Med. Res. 2018, 7, 109–125. [Google Scholar] [CrossRef] [PubMed]
- Jones, W.P.; Chin, Y.W.; Kinghorn, A.D. The role of pharmacognosy in modern medicine and pharmacy. Curr. Drug Targets 2006, 7, 247–264. [Google Scholar] [CrossRef] [PubMed]
- Orhan, I.E. Pharmacognosy: Science of natural products in drug discovery. BioImpacts 2014, 4, 109–110. [Google Scholar] [CrossRef] [PubMed]
- Clarke, T.C.; Black, L.I.; Stussman, B.J.; Barnes, P.M.; Nahin, R.L. Trends in the Use of Complementary Health Approaches among Adults: United States, 2002–2012. 2015. Available online: http://klinikfarmakoloji.com/files/ABD%20complementary%20medicine%20statistic.pdf (accessed on 15 February 2019).
- Harris, P.E.; Cooper, K.L.; Relton, C.; Thomas, K.J. Prevalence of complementary and alternative medicine (CAM) use by the general population: A systematic review and update. Int. J. Clin. Pract. 2012, 66, 924–939. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.; Kawa, K.; Eckl, V.; Morton, C.; Stredney, R. Herbal supplement sales in US increase 7.7% in 2016: Consumer preferences shifting toward ingredients with general wellness benefits, driving growth of adaptogens and digestive health products. HerbalGram J. Am. Bot. Counc. 2017, 115, 56–65. [Google Scholar]
- Bauer, B.A. Chinese medicine and integrative medicine in the United States. Chin. J. Integr. Med. 2015, 21, 569–570. [Google Scholar] [CrossRef] [PubMed]
- Chon, T.Y.; Lee, M.C. Acupuncture. Mayo Clin. Proc. 2013, 88, 1141–1146. [Google Scholar] [CrossRef]
- Cui, J.; Wang, S.; Ren, J.; Zhang, J.; Jing, J. Use of acupuncture in the USA: Changes over a decade (2002–2012). Acupunct. Med. 2017, 35, 200–207. [Google Scholar] [CrossRef]
- Fleischer, T.; Su, Y.C.; Lin, S.J. How do government regulations influence the ability to practice Chinese herbal medicine in western countries. J. Ethnopharmacol. 2017, 196, 104–109. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, Z.; Cheng, J.; Fan, D. Barriers and countermeasures in developing traditional Chinese medicine in Europe. Front. Med. 2016, 10, 360–376. [Google Scholar] [CrossRef]
- Williamson, E.M.; Lorenc, A.; Booker, A.; Robinson, N. The rise of traditional Chinese medicine and its materia medica: A comparison of the frequency and safety of materials and species used in Europe and China. J. Ethnopharmacol. 2013, 149, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Adams, J.; Sibbritt, D. Prevalence and factors associated with the use of acupuncture and Chinese medicine: Results of a nationally representative survey of 17161 Australian women. Acupunct. Med. 2017, 35, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Furst, R.; Zundorf, I. Evidence-Based Phytotherapy in Europe: Where Do We Stand? Planta Med. 2015, 81, 962–967. [Google Scholar] [CrossRef] [PubMed]
- Vizirianakis, I.S.; Spanakis, M.; Termentzi, A.; Niopas, I.; Kokkalou, E. Clinical and pharmacogenomic assessment of herb-drug interactions to improve drug delivery and pharmacovigilance. In Plants in Traditional and Modern Medicine: Chemistry and Activity; Kokkalou, E., Ed.; Transworld Research Network: Kerala, India, 2010. [Google Scholar]
- Kwan, D.; Boon, H.S.; Hirschkorn, K.; Welsh, S.; Jurgens, T.; Eccott, L.; Heschuk, S.; Griener, G.G.; Cohen-Kohler, J.C. Exploring consumer and pharmacist views on the professional role of the pharmacist with respect to natural health products: A study of focus groups. BMC Complementary Altern. Med. 2008, 8, 40. [Google Scholar] [CrossRef] [PubMed]
- Tran, S.; Calabretto, J.P.; Sorich, M. Consumer-pharmacist interactions around complementary medicines: Agreement between pharmacist and consumer expectations, satisfaction and pharmacist influence. Int. J. Pharm. Pract. 2013, 21, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Braun, L.A.; Tiralongo, E.; Wilkinson, J.M.; Spitzer, O.; Bailey, M.; Poole, S.; Dooley, M. Perceptions, use and attitudes of pharmacy customers on complementary medicines and pharmacy practice. BMC Complementary Altern. Med. 2010, 10, 38. [Google Scholar] [CrossRef] [PubMed]
- Radimer, K.; Bindewald, B.; Hughes, J.; Ervin, B.; Swanson, C.; Picciano, M.F. Dietary supplement use by US adults: Data from the National Health and Nutrition Examination Survey, 1999–2000. Am. J. Epidemiol. 2004, 160, 339–349. [Google Scholar] [CrossRef]
- Tsai, H.H.; Lin, H.W.; Simon Pickard, A.; Tsai, H.Y.; Mahady, G.B. Evaluation of documented drug interactions and contraindications associated with herbs and dietary supplements: A systematic literature review. Int. J. Clin. Pract. 2012, 66, 1056–1078. [Google Scholar] [CrossRef]
- Bandaranayake, W.M. Quality Control, Screening, Toxicity, and Regulation of Herbal Drugs. In Modern Phytomedicine; Ahmad, I., Aqil, F., Owais, M., Eds.; WILEY-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006. [Google Scholar] [CrossRef]
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 177. [Google Scholar] [CrossRef]
- Dwyer, J.T.; Coates, P.M.; Smith, M.J. Dietary Supplements: Regulatory Challenges and Research Resources. Nutrients 2018, 10, 41. [Google Scholar] [CrossRef]
- Lam, Y.W.F.; Huang, S.M.; Hall, S.D. Herbal Supplements-Drug Interactions: Scientific and Regulatory Perspectives; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Amadi, C.N.; Mgbahurike, A.A. Selected Food/Herb-Drug Interactions: Mechanisms and Clinical Relevance. Am. J. Ther. 2018, 25, e423–e433. [Google Scholar] [CrossRef] [PubMed]
- Asher, G.N.; Corbett, A.H.; Hawke, R.L. Common Herbal Dietary Supplement-Drug Interactions. Am. Fam. Physician 2017, 96, 101–107. [Google Scholar] [PubMed]
- Izzo, A.A. Interactions between herbs and conventional drugs: Overview of the clinical data. Med. Princ. Pract. 2012, 21, 404–428. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A.; Ernst, E. Interactions between herbal medicines and prescribed drugs: A systematic review. Drugs 2001, 61, 2163–2175. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.A.; Seely, D. Clinically based evidence of drug–herb interactions: A systematic review. Expert Opin. Drug Saf. 2010, 9, 79–124. [Google Scholar] [CrossRef]
- Posadzki, P.; Watson, L.; Ernst, E. Herb-drug interactions: An overview of systematic reviews. Br. J. Clin. Pharmacol. 2013, 75, 603–618. [Google Scholar] [CrossRef]
- Singh, A.; Zhao, K. Herb-Drug Interactions of Commonly Used Chinese Medicinal Herbs. Int. Rev. Neurobiol. 2017, 135, 197–232. [Google Scholar] [CrossRef]
- Singh, D.; Gupta, R.; Saraf, S.A. Herbs-are they safe enough? An overview. Crit. Rev. Food Sci. Nutr. 2012, 52, 876–898. [Google Scholar] [CrossRef]
- EMA. DIRECTIVE 2004/24/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL Amending, as Regards Traditional Herbal Medicinal Products, Directive 2001/83/EC on the Community Code Relating to Medicinal Products for Human Use. 2004. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2004:136:0085:0090:en:PDF (accessed on 11 October 2017).
- FDA. Botanical Drug Development Guidance for Industry; FDA: Silver Spring, MD, USA, 2016.
- World Health Organization. Research Guidelines for Evaluating the Safety and Efficacy of Herbal Medicines; World Health Organization: Manila, Philippines, 1993; p. 94. Available online: http://apps.who.int/medicinedocs/en/d/Jh2946e/ (accessed on 11 October 2017).
- World Health Organization. WHO Guidelines for Assessing Quality of Herbal Medicines with Reference to Contaminants and Residues; World Health Organization: Geneva, Switzerland, 2007; p. 118. [Google Scholar]
- Chiarugi, F.; Trypakis, D.; Spanakis, E.G. Problems and solutions for storing and sharing data from medical devices in eHealth applications. In Proceedings of the 2nd OpenECG Workshop 2004, Berlin, Germany, 1–3 April 2004. [Google Scholar]
- Kondylakis, H.; Spanakis, E.G.; Sfakianakis, S.; Sakkalis, V.; Tsiknakis, M.; Marias, K.; Xia, Z.; Hong Qing, Y.; Feng, D. Digital patient: Personalized and translational data management through the MyHealthAvatar EU project. In Proceedings of the 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015. [Google Scholar] [CrossRef]
- Prashad, R. The Role of Personal Health Record Systems in Chronic Disease Management. Stud. Health Technol. Inform. 2017, 234, 275–279. [Google Scholar]
- Spanakis, E.G.; Chiarugi, F.; Kouroubali, A.; Spat, S.; Beck, P.; Asanin, S.; Rosengren, P.; Gergely, T.; Thestrup, J. Diabetes management using modern information and communication technologies and new care models. Interact. J. Med Res. 2012, 1, e8. [Google Scholar] [CrossRef]
- Spanakis, E.G.; Psaraki, M.; Sakkalis, V. Congestive Heart Failure Risk Assessment Monitoring through Internet of things and mobile Personal Health Systems. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 18–21 July 2018; pp. 2925–2928. [Google Scholar] [CrossRef]
- Spanakis, E.G.; Santana, S.; Tsiknakis, M.; Marias, K.; Sakkalis, V.; Teixeira, A.; Janssen, J.H.; de Jong, H.; Tziraki, C. Technology-Based Innovations to Foster Personalized Healthy Lifestyles and Well-Being: A Targeted Review. J. Med Internet Res. 2016, 18, e128. [Google Scholar] [CrossRef]
- Traganitis, A.; Trypakis, D.; Spanakis, E.G.; Condos, S.; Stamkopoulos, T.G.; Tsiknakis, M.; Orphanoudakis, S.C. Home monitoring and personal health management services in a regional health telematics network. In Proceedings of the 23rd Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Istanbul, Turkey, 25–28 October 2001. [Google Scholar] [CrossRef]
- Chatsisvili, A.; Sapounidis, I.; Pavlidou, G.; Zoumpouridou, E.; Karakousis, V.A.; Spanakis, M.; Teperikidis, L.; Niopas, I. Potential drug–drug interactions in prescriptions dispensed in community pharmacies in Greece. Pharm. World Sci. 2010, 32, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Muhic, N.; Mrhar, A.; Brvar, M. Comparative analysis of three drug–drug interaction screening systems against probable clinically relevant drug–drug interactions: A prospective cohort study. Eur. J. Clin. Pharmacol. 2017, 73, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Spanakis, M.; Spanakis, E.G.; Kondylakis, H.; Sfakianakis, S.; Genitsaridi, I.; Sakkalis, V.; Tsiknakis, M.; Marias, K. Addressing drug–drug and drug-food interactions through personalized empowerment services for healthcare. In Proceedings of the 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 16–20 August 2016. [Google Scholar] [CrossRef]
- FDA. Clinical Drug Interaction Studies—Study Design, Data Analysis, and Clinical Implications Guidance for Industry; FDA: Silver Spring, MD, USA, 2017; p. 32.
- Hansten, P.D.; Horn, J.R. Drug Interactions: Analysis and Management; Wolters Kluwer Health: St. Louis, MO, USA, 2006. [Google Scholar]
- Awortwe, C.; Makiwane, M.; Reuter, H.; Muller, C.; Louw, J.; Rosenkranz, B. Critical evaluation of causality assessment of herb-drug interactions in patients. Br. J. Clin. Pharmacol. 2018, 84, 679–693. [Google Scholar] [CrossRef] [PubMed]
- Fasinu, P.S.; Bouic, P.J.; Rosenkranz, B. An overview of the evidence and mechanisms of herb-drug interactions. Front. Pharmacol. 2012, 3, 69. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Liu, K. Pharmacokinetic interactions between herbal medicines and prescribed drugs: Focus on drug metabolic enzymes and transporters. Curr. Drug Metab. 2014, 15, 791–807. [Google Scholar] [CrossRef] [PubMed]
- Greenblatt, D.J. Mechanisms and Consequences of Drug–drug Interactions. Clin. Pharmacol. Drug Dev. 2017, 6, 118–124. [Google Scholar] [CrossRef]
- Zanger, U.M.; Schwab, M. Cytochrome P450 enzymes in drug metabolism: Regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol. Ther. 2013, 138, 103–141. [Google Scholar] [CrossRef]
- Di, Y.M.; Li, C.G.; Xue, C.C.; Zhou, S.F. Clinical drugs that interact with St. John’s wort and implication in drug development. Curr. Pharm. Des. 2008, 14, 1723–1742. [Google Scholar] [CrossRef]
- Rahimi, R.; Abdollahi, M. An update on the ability of St. John’s wort to affect the metabolism of other drugs. Expert Opin. Drug Metab. Toxicol. 2012, 8, 691–708. [Google Scholar] [CrossRef]
- Soleymani, S.; Bahramsoltani, R.; Rahimi, R.; Abdollahi, M. Clinical risks of St John’s Wort (Hypericum perforatum) co-administration. Expert Opin. Drug Metab. Toxicol. 2017, 13, 1047–1062. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.; Scicchitano, F.; Whalley, B.J.; Mazzitello, C.; Ciriaco, M.; Esposito, S.; Patane, M.; Upton, R.; Pugliese, M.; Chimirri, S.; et al. Hypericum perforatum: Pharmacokinetic, mechanism of action, tolerability, and clinical drug–drug interactions. Phytother. Res. 2014, 28, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Gorman, G.S.; Coward, L.; Darby, A.; Rasberry, B. Effects of herbal supplements on the bioactivation of chemotherapeutic agents. J. Pharm. Pharmacol. 2013, 65, 1014–1025. [Google Scholar] [CrossRef] [PubMed]
- Rengelshausen, J.; Banfield, M.; Riedel, K.D.; Burhenne, J.; Weiss, J.; Thomsen, T.; Walter-Sack, I.; Haefeli, W.E.; Mikus, G. Opposite effects of short-term and long-term St John’s wort intake on voriconazole pharmacokinetics. Clin. Pharmacol. Ther. 2005, 78, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Schellander, R.; Donnerer, J. Antidepressants: Clinically relevant drug interactions to be considered. Pharmacology 2010, 86, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A.; Hoon-Kim, S.; Radhakrishnan, R.; Williamson, E.M. A Critical Approach to Evaluating Clinical Efficacy, Adverse Events and Drug Interactions of Herbal Remedies. Phytother. Res. 2016, 30, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Chrubasik-Hausmann, S.; Vlachojannis, J.; McLachlan, A.J. Understanding drug interactions with St John’s wort (Hypericum perforatum L.): Impact of hyperforin content. J. Pharm. Pharmacol. 2019, 71, 129–138. [Google Scholar] [CrossRef]
- Chen, L.; Lv, D.; Wang, D.; Chen, X.; Zhu, Z.; Cao, Y.; Chai, Y. A novel strategy of profiling the mechanism of herbal medicines by combining network pharmacology with plasma concentration determination and affinity constant measurement. Mol. Biosyst. 2016, 12, 3347–3356. [Google Scholar] [CrossRef]
- Scheife, R.T.; Hines, L.E.; Boyce, R.D.; Chung, S.P.; Momper, J.D.; Sommer, C.D.; Abernethy, D.R.; Horn, J.R.; Sklar, S.J.; Wong, S.K.; et al. Consensus recommendations for systematic evaluation of drug–drug interaction evidence for clinical decision support. Drug Saf. 2015, 38, 197–206. [Google Scholar] [CrossRef]
- Chronaki, C.E.; Chiarugi, F.; Mavrogiannaki, E.; Demou, C.; Lelis, P.; Trypakis, D.; Spanakis, M.; Tsiknakis, M.; Orphanoudakis, S.C. An eHealth platform for instant interaction among health professionals. In Proceedings of the Computers in Cardiology, 2003, Thessaloniki Chalkidiki, Greece, 21–24 September 2003; pp. 101–104. [Google Scholar]
- Maniadi, E.; Kondylakis, H.; Spanakis, E.G.; Spanakis, M.; Tsiknakis, M.; Marias, K.; Dong, F. Designing a digital patient avatar in the context of the MyHealthAvatar project initiative. In Proceedings of the 13th IEEE International Conference on BioInformatics and BioEngineering, Chania, Greece, 10–13 November 2013. [Google Scholar] [CrossRef]
- Ogultarhan, V.; Shoshi, A.; Magnucki, R.; Kormeier, B.; Hofestadt, R. KATIS: An eHealth System for Complementary Medicine. Stud. Health Technol. Inform. 2016, 223, 167–173. [Google Scholar]
- Lin, S.S.; Tsai, C.L.; Tu, C.Y.; Hsieh, C.L. Reducing drug–herb interaction risk with a computerized reminder system. Ther. Clin. Risk Manag. 2015, 11, 247–253. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pourroy, B.; Letellier, C.; Helvig, A.; Chanet, B.; De Crozals, F.; Alessandra, C. Development of a rapid risk evaluation tool for herbs/drugs interactions in cancer patients: A multicentric experience in south of France. Eur. J. Cancer Care 2017, 26. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.; Finlay, I.G.; Cook, A.; Thomas, K.J.; Hood, K. Complementary and alternative medicine use by patients with cancer in Wales: A cross sectional survey. Complementary Ther. Med. 2003, 11, 249–253. [Google Scholar] [CrossRef]
- Spanakis, M.; Sfakianakis, S.; Spanakis, E.G.; Kallergis, G.; Sakkalis, V. PDCA: An eHealth service for the management of drug interactions with complementary and alternative medicines. In Proceedings of the 2018 IEEE EMBS International Conference on Biomedical & Health Informatics (BHI), Las Vegas, NV, USA, 4–7 March 2018; pp. 373–376. [Google Scholar]
- Wishart, D.S.; Knox, C.; Guo, A.C.; Shrivastava, S.; Hassanali, M.; Stothard, P.; Chang, Z.; Woolsey, J. DrugBank: A comprehensive resource for in silico drug discovery and exploration. Nucleic Acids Res. 2006, 34, D668–D672. [Google Scholar] [CrossRef] [PubMed]
- International Standards Organization (ISO). Health Informatics—Identification of medicinal products—Data elements and structures for the unique identification and exchange of regulated information on substances. ISO/TC 215. In Health informatics; ISO Press BV: Amsterdam, The Netherlands, 2018; Volume 2, p. 61. [Google Scholar]
- Heinrich, M.; Modarai, M.; Kortenkamp, A. Herbal extracts used for upper respiratory tract infections: Are there clinically relevant interactions with the cytochrome P450 enzyme system? Planta Med. 2008, 74, 657–660. [Google Scholar] [CrossRef] [PubMed]
- Srinivas, N.R. Cranberry juice ingestion and clinical drug–drug interaction potentials; review of case studies and perspectives. J. Pharm. Pharm. Sci. 2013, 16, 289–303. [Google Scholar] [CrossRef]
- Albassam, A.A.; Markowitz, J.S. An Appraisal of Drug–drug Interactions with Green Tea (Camellia sinensis). Planta Med. 2017, 83, 496–508. [Google Scholar] [CrossRef]
- Basch, E.; Bent, S.; Collins, J.; Dacey, C.; Hammerness, P.; Harrison, M.; Smith, M.; Szapary, P.; Ulbricht, C.; Vora, M.; et al. Flax and flaxseed oil (Linum usitatissimum): A review by the Natural Standard Research Collaboration. J. Soc. Integr. Oncol. 2007, 5, 92–105. [Google Scholar] [CrossRef]
- Langhammer, A.J.; Nilsen, O.G. In vitro inhibition of human CYP1A2, CYP2D6, and CYP3A4 by six herbs commonly used in pregnancy. Phytother. Res. 2014, 28, 603–610. [Google Scholar] [CrossRef]
- Rehman, S.U.; Kim, I.S.; Choi, M.S.; Kim, S.H.; Zhang, Y.; Yoo, H.H. Time-dependent Inhibition of CYP2C8 and CYP2C19 by Hedera helix Extracts, A Traditional Respiratory Herbal Medicine. Molecules 2017, 22, 1241. [Google Scholar] [CrossRef]
- Bahramsoltani, R.; Rahimi, R.; Farzaei, M.H. Pharmacokinetic interactions of curcuminoids with conventional drugs: A review. J. Ethnopharmacol. 2017, 209, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Agbabiaka, T.B.; Spencer, N.H.; Khanom, S.; Goodman, C. Prevalence of drug–herb and drug-supplement interactions in older adults: A cross-sectional survey. Br. J. Gen. Pract. 2018, 68, e711–e717. [Google Scholar] [CrossRef] [PubMed]
- Al-Jenoobi, F.I.; Ahad, A.; Mahrous, G.M.; Al-Mohizea, A.M.; AlKharfy, K.M.; Al-Suwayeh, S.A. Effects of fenugreek, garden cress, and black seed on theophylline pharmacokinetics in beagle dogs. Pharm. Biol. 2015, 53, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Djuv, A.; Nilsen, O.G.; Steinsbekk, A. The co-use of conventional drugs and herbs among patients in Norwegian general practice: A cross-sectional study. BMC Complementary Altern. Med. 2013, 13, 295. [Google Scholar] [CrossRef] [PubMed]
- Ulbricht, C.; Basch, E.; Bent, S.; Boon, H.; Corrado, M.; Foppa, I.; Hashmi, S.; Hammerness, P.; Kingsbury, E.; Smith, M.; et al. Evidence-based systematic review of saw palmetto by the Natural Standard Research Collaboration. J. Soc. Integr. Oncol. 2006, 4, 170–186. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.E.; Zhang, S. Flavonoid-drug interactions: Effects of flavonoids on ABC transporters. Life Sci. 2006, 78, 2116–2130. [Google Scholar] [CrossRef] [PubMed]
- Hajda, J.; Rentsch, K.M.; Gubler, C.; Steinert, H.; Stieger, B.; Fattinger, K. Garlic extract induces intestinal P-glycoprotein, but exhibits no effect on intestinal and hepatic CYP3A4 in humans. Eur. J. Pharm. Sci. 2010, 41, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Taheri, A.; Lavasani, H.; Kasirzadeh, S.; Sheikholeslami, B.; Ardakani, Y.H.; Rouini, M.R. Changes in CYP2D enzyme activity following induction of type 2 diabetes, and administration of cinnamon and metformin: An experimental animal study. Xenobiotica 2018, 48, 984–989. [Google Scholar] [CrossRef] [PubMed]
- Rahman, H.; Kim, M.; Leung, G.; Green, J.A.; Katz, S. Drug–herb Interactions in the Elderly Patient with IBD: A Growing Concern. Curr. Treat. Options Gastroenterol. 2017, 15, 618–636. [Google Scholar] [CrossRef]
- Malina, D.M.; Fonseca, F.A.; Barbosa, S.A.; Kasmas, S.H.; Machado, V.A.; Franca, C.N.; Borges, N.C.; Moreno, R.A.; Izar, M.C. Additive effects of plant sterols supplementation in addition to different lipid-lowering regimens. J. Clin. Lipidol. 2015, 9, 542–552. [Google Scholar] [CrossRef]
- Wang, M.T.; Li, I.H.; Lee, W.J.; Huang, T.Y.; Leu, H.B.; Chan, A.L. Exposure to sennoside-digoxin interaction and risk of digoxin toxicity: A population-based nested case-control study. Eur. J. Heart Fail. 2011, 13, 1238–1243. [Google Scholar] [CrossRef] [PubMed]
- Ventura, S.; Rodrigues, M.; Falcao, A.; Alves, G. Effects of Paullinia cupana extract on lamotrigine pharmacokinetics in rats: A herb-drug interaction on the gastrointestinal tract with potential clinical impact. Food Chem. Toxicol. 2018, 115, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Fung, W.T.; Subramaniam, G.; Lee, J.; Loh, H.M.; Leung, P.H. Assessment of extracts from red yeast rice for herb-drug interaction by in-vitro and in-vivo assays. Sci. Rep. 2012, 2, 298. [Google Scholar] [CrossRef] [PubMed]
- Hellum, B.H.; Tosse, A.; Hoybakk, K.; Thomsen, M.; Rohloff, J.; Georg Nilsen, O. Potent in vitro inhibition of CYP3A4 and P-glycoprotein by Rhodiola rosea. Planta Med. 2010, 76, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Maniscalco, I.; Toffol, E.; Giupponi, G.; Conca, A. The interaction of Rhodiola rosea and antidepressants. A case report. Neuropsychiatrie 2015, 29, 36–38. [Google Scholar] [CrossRef] [PubMed]
- Spanakis, M.; Vizirianakis, I.S.; Batzias, G.; Niopas, I. Pharmacokinetic interaction between losartan and Rhodiola rosea in rabbits. Pharmacology 2013, 91, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Thu, O.K.; Spigset, O.; Nilsen, O.G.; Hellum, B. Effect of commercial Rhodiola rosea on CYP enzyme activity in humans. Eur. J. Clin. Pharmacol. 2016, 72, 295–300. [Google Scholar] [CrossRef]
- Xu, W.; Zhang, T.; Wang, Z.; Liu, T.; Liu, Y.; Cao, Z.; Sui, Z. Two potent cytochrome P450 2D6 inhibitors found in Rhodiola rosea. Die Pharm. 2013, 68, 974–976. [Google Scholar]
- Dietz, B.M.; Hajirahimkhan, A.; Dunlap, T.L.; Bolton, J.L. Botanicals and Their Bioactive Phytochemicals for Women’s Health. Pharmacol. Rev. 2016, 68, 1026–1073. [Google Scholar] [CrossRef]
- Barton, C.; Vigor, K.; Scott, R.; Jones, P.; Lentfer, H.; Bax, H.J.; Josephs, D.H.; Karagiannis, S.N.; Spicer, J.F. Beta-glucan contamination of pharmaceutical products: How much should we accept? Cancer Immunol. Immunother. 2016, 65, 1289–1301. [Google Scholar] [CrossRef]
- Potterat, O. Goji (Lycium barbarum and L. chinense): Phytochemistry, pharmacology and safety in the perspective of traditional uses and recent popularity. Planta Med. 2010, 76, 7–19. [Google Scholar] [CrossRef] [PubMed]
- Walji, R.; Boon, H.; Barnes, J.; Welsh, S.; Austin, Z.; Baker, G.R. Reporting natural health product related adverse drug reactions: Is it the pharmacist’s responsibility? Int. J. Pharm. Pract. 2011, 19, 383–391. [Google Scholar] [CrossRef] [PubMed]
- El Morabet, N.; Uitvlugt, E.B.; van den Bemt, B.J.F.; van den Bemt, P.; Janssen, M.J.A.; Karapinar-Carkit, F. Prevalence and Preventability of Drug-Related Hospital Readmissions: A Systematic Review. J. Am. Geriatr. Soc. 2018, 66, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Liobikiene, G.; Bernatoniene, J. The determinants of access to information on the Internet and knowledge of health related topics in European countries. Health Policy 2018, 122, 1348–1355. [Google Scholar] [CrossRef] [PubMed]
- Santana, S.; Lausen, B.; Bujnowska-Fedak, M.; Chronaki, C.E.; Prokosch, H.U.; Wynn, R. Informed citizen and empowered citizen in health: Results from an European survey. BMC Fam. Pract. 2011, 12, 20. [Google Scholar] [CrossRef]
- Ansari, J. Drug interaction and pharmacist. J. Young Pharm. 2010, 2, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Zhou, S.Y.; Fabriaga, E.; Zhang, P.H.; Zhou, Q. Food-drug interactions precipitated by fruit juices other than grapefruit juice: An update review. J. Food Drug Anal. 2018, 26, S61–S71. [Google Scholar] [CrossRef] [PubMed]
| Severity | Clinical Guidance | Presentation in PharmActa |
|---|---|---|
| Prohibited | Use alternative-Major interaction -Avoid co-administration | Avoid it and seek medical advice |
| It should be avoided | Contraindicated-Major interaction-Combine only under specific circumstances | |
| Minimize the risk | Use with caution-Moderate interaction-Precautions to minimize any risk | Use it only after medical advice |
| No clinical impact | Minor interaction-research data suggest no clinical impact | Use it but always inform your treating physician |
| No interactions | Clinical /research results suggest that there is no interaction |
| HMP | Proposed Use | Potential Drug Interactions | Pharmacological Mechanisms | Patients-Diseases | Clinical Significance | PharmActa | Reference |
|---|---|---|---|---|---|---|---|
| Horehound | respiratory ailments | - | - | - | - | monitor for clinical significant DHIs | - |
| Cranberry | bladder and kidney ailments | anticoagulant and antiplatelet therapy | PK and PD | CVD and clot disorders | Research level-clinical data suggest no interaction | use with caution | [75] |
| Echinacea | immune stimulant | antipsychotics, antidepressants, immunosuppressants | PK and PD | CNS disorders | Under specific circumstances-Precautions to minimize any risk | monitor for clinical significant DHIs | [17,26,27,28,61] |
| Green Tea | antioxidant | anticoagulant and antiplatelet therapy | PD | CVD and clot disorders | Research level-clinical data suggest no interaction | use with caution | [76] |
| Black Cohosh | PMS and menopause symptoms | amiodarone, fexofenadine, glyburide, simvastatin, atorvastatin | PK | CVD and clot disorders | Research level-clinical data suggest no interaction | use with caution | [26,27,51,58] |
| Garcinia | weight loss | ciprofloxacin, quinine | PK | bacterial infections and muscular disorders | Research level-clinical data suggest no interaction | use with caution | [25] |
| Flax Seed/Flax oil | CVD problems and diabetes | anticoagulant and antiplatelet therapy | PD | CVD and clot disorders | Research level-clinical data suggest no interaction | use with caution | [77] |
| Ginger | reduce nausea and inflammation | tacrolimus | PK | allogeneic organ transplant | Under specific circumstances-Precautions to minimize any risk | monitor for clinical significant DHIs | [32,58,61,78] |
| Ivy leaf | expectorant | - | - | - | Research level-clinical data suggest no interaction | use with caution | [32,79] |
| Turmeric | antiinflamantory | CVD drugs, antidepressants, anticoagulants, antibiotics, chemotherapeutic agents, and antihistamines | PK | CVD and clot disorders, CNS disorders, Cancer, bacterial infections, allergies | Under specific circumstances-Precautions to minimize any risk | monitor for clinical significant DHIs | [32,80] |
| Valerian | anxiety and insomnia | benzodiazepines and sedatives | PD | Under specific circumstances-Precautions to minimize any risk | monitor for clinical significant DHIs | [26,27,81] | |
| Fenugreek | diabetes and PMS | hypoglycaemic, anticoagulant, and antiplatelet therapy | PD | diabetes, CVD and clot disorders | research level-clinical data suggest no interaction | use with caution | [82] |
| Yohimbe | erectile dysfunction - weight loss | MAOIs, clonidine, | PD | CNS disorders, prostate hypertrophy, kidney disease | avoid co-administration | use alternative due to clinical significant DHIs | [20] |
| Aloe | constipation, diabetes, acne and inflamation | digoxin, antidiabetic drugs | PD | HF, diabetes | under specific circumstances-precautions to minimize any risk | monitor for clinically significant DHIs | [61,83] |
| Saw Palmeto | prostate surgery | antibiotics, anticoagulant, and antiplatelet therapy | PD | CVD and clot disorders, bacterial infections, | research level-clinical data suggest no interaction | use with caution | [26,27,28,84] |
| Milk Thistle | liver problems | losartan, warfarin, phenytoin, diazepam | PK | CVD and preoperative period | research level-clinical data suggest no interaction | use with caution | [26,51,85] |
| Garlic | reducing cholesterol and blood pressure | colchicine, digoxin, doxorubicin, qunidine, rosuvastatin, dexamethasone, nifedipine, verapamil, saquinavir, indinavir, ritonavir, rifampicin, reserpine, carbamazepine | PK | arthritis, CVD, cancer, HIV | under specific circumstances-precautions to minimize any risk | monitor for clinically significant DHIs | [26,28,32,51,83,86] |
| Coconut Oil | eczema and improve HDL cholesterol levels | - | - | - | - | use with caution | - |
| Eldeberry | Constipation and flu symptoms | immunosuprresants, hypoglycemic | PD | transplantation, diabetes | research level-clinical data suggest no interaction | use with caution | [78] |
| Cinnamon | diabetes and lowering cholesterol | anticoagulant and antiplatelet therapy | PD | CVD and clot disorders | research level-clinical data suggest no interaction | use with caution | [87] |
| Green Coffee Extract | antioxidant for diabetes and heart disease | adenosine, anticoagulant and antiplatelet therapy, MAOIs, CVD drugs | PD | CVD and clot disorders, CNS disorders, Inflammatory bowel disease | under specific circumstances-precautions to minimize any risk | monitor for clinical significant DHIs | [88] |
| Boswellia | anti-inflammatory in asthma and arthritis | - | - | - | - | monitor for clinical significant DHIs | - |
| Ginkgo | adaptogen for dementia and fatigue | clopidogrel, aspirin, warfarin | PD | under specific circumstances-precautions to minimize any risk | monitor for clinical significant DHIs | [20,26,28,30,51,61,85] | |
| Plant Sterols | lower cholesterol levels | LLTs | PD | dyslipidemias | research level-clinical data suggest no interaction | use with caution | [89] |
| Senna | constipation | digoxin, warfarin | PD | avoid co-administration | use alternative due to clinical significant DHIs | [90] | |
| Acaí | antioxidant | - | - | - | - | - | |
| Guarana | CNS stimulant | adenosine, anticoagulant and antiplatelet therapy, MAOIs, CNS drugs | PD | CVD and clot disorders, CNS disorders | under specific circumstances-precautions to minimize any risk | monitor for clinically significant DHIs | [91] |
| Rhodiola | adaptogen | loasartan, warfarin, clopidogrel | PK | CVD and clot disorders | under specific circumstances-precautions to minimize any risk | monitor for clinically significant DHIs | [92,93,94,95,96,97] |
| Bioflavonoid Complex | antioxidant | digoxin, anticoagulant and antiplatelet therapy, CVD, cancer medications | PK | CVD and clot disorders, Cancer | moderate | monitor for clinical significant DHIs | [85] |
| Red Yeast rice | lower cholesterol levels | LLTs, CVD drugs | PK and PD | dyslipidemias, CVD | under specific circumstances-precautions to minimize any risk | monitor for clinically significant DHIs | [92,96] |
| Ginseng (siberian) | adaptogen and heart disease | digoxin | PK | heart failure | under specific circumstances-precautions to minimize any risk | monitor for clinically significant DHIs | [26,28,30,51,61,68] |
| Horny Goat Weed | erectile dysfunction, PMS, osteoporosis | CVD, anticoagulant and antiplatelet therapy | PD | CVD and clot disorders | research level-clinical data suggest no interaction | use with caution | [98] |
| Yerba Mate | CNS stimulant | adenosine, anticoagulant and antiplatelet therapy, MAOIs, CNS drugs | PD | CVD and clot disorders, CNS disorders | research level-clinical data suggest no interaction | use with caution | - |
| Fennel | digestive problems, pregnancy | Contraceptives, tamoxifen, ciprofloxacin | PK | birth control, breast cancer, infections | under specific circumstances-precautions to minimize any risk | monitor for clinically significant DHIs | [78] |
| Beta Glucans | lower blood cholesterol | LLTs, CVD drugs | PK and PD | dyslipidemias, CVD | under specific circumstances-precautions to minimize any risk | monitor for clinically significant DHIs | [99] |
| Maca | female hormone imbalance, menstrual problems, chronic fatigue syndrome | - | - | - | - | - | - |
| St. John’s Wort | antidepressant | anticancer, CNS drugs, LLTs, PPIs, antiretroviral, hypoglycemic, antihistamine, CVD drugs, antimicrobials, hormonal agents, immunosuppressants | PK and PD | arthritis, CVD, cancer, CNS disorders, diabetes, HIV, infections, allergies, transplantation | avoid co-administration | use alternative due to clinical significant DHIs | [15,28,56,58,59,61,62] |
| Wheatgrass/Barley | diabetes, lower cholesterol and for weight loss | - | - | Inflammatory bowel disease | under specific circumstances-precautions to minimize any risk | monitor for clinical significant DHIs | [88] |
| Goji Berry | antioxidant, diabetes, blood pressure | warfarin and antiplatelet | PD | CVD and clot disorders | under specific circumstances-precautions to minimize any risk | monitor for clinical significant DHIs | [100] |
| Chia Seed/Chia oil | omega-3 fatty acids and antioxidants | - | - | - | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spanakis, M.; Sfakianakis, S.; Sakkalis, V.; Spanakis, E.G. PharmActa: Empowering Patients to Avoid Clinical Significant Drug–Herb Interactions. Medicines 2019, 6, 26. https://doi.org/10.3390/medicines6010026
Spanakis M, Sfakianakis S, Sakkalis V, Spanakis EG. PharmActa: Empowering Patients to Avoid Clinical Significant Drug–Herb Interactions. Medicines. 2019; 6(1):26. https://doi.org/10.3390/medicines6010026
Chicago/Turabian StyleSpanakis, Marios, Stelios Sfakianakis, Vangelis Sakkalis, and Emmanouil G. Spanakis. 2019. "PharmActa: Empowering Patients to Avoid Clinical Significant Drug–Herb Interactions" Medicines 6, no. 1: 26. https://doi.org/10.3390/medicines6010026
APA StyleSpanakis, M., Sfakianakis, S., Sakkalis, V., & Spanakis, E. G. (2019). PharmActa: Empowering Patients to Avoid Clinical Significant Drug–Herb Interactions. Medicines, 6(1), 26. https://doi.org/10.3390/medicines6010026








